Reevaluation of the Acute Cystitis Symptom Score, a Self-Reporting Questionnaire. Part II. Patient-Reported Outcome Assessment
Abstract
:1. Introduction
2. Patients and Methods
2.1. Clinical Procedures
2.2. Statistical Analysis
3. Results
3.1. Study Population
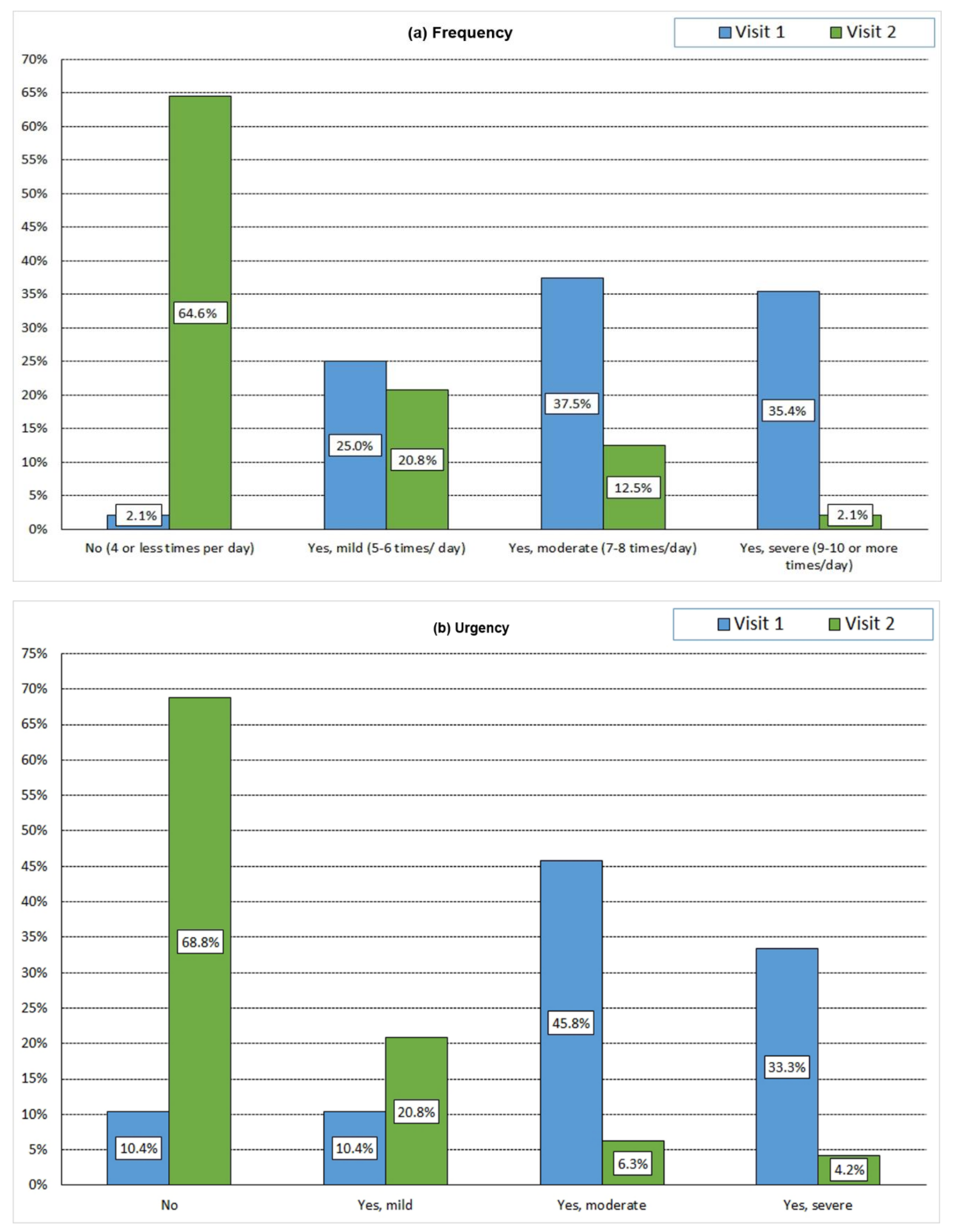
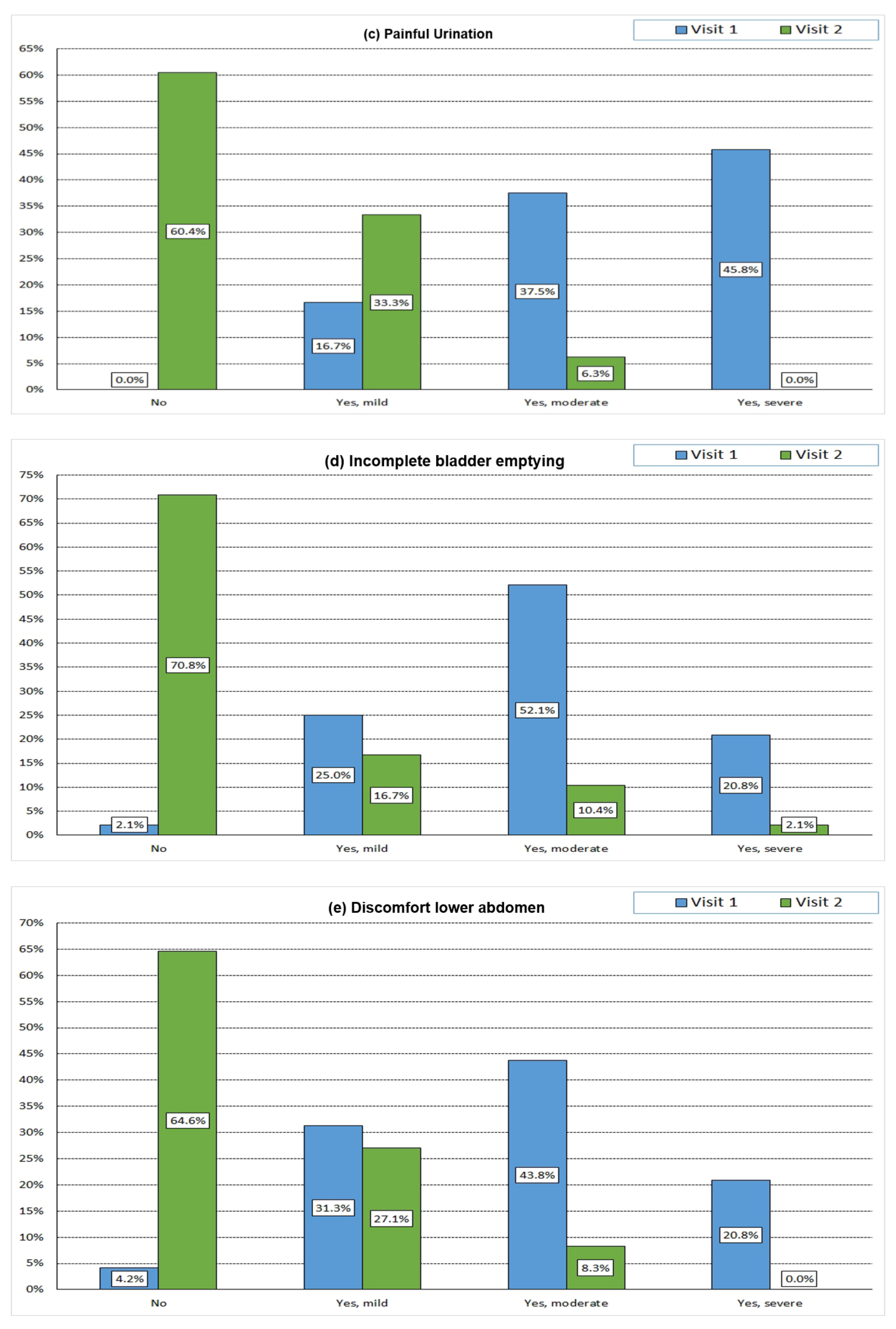
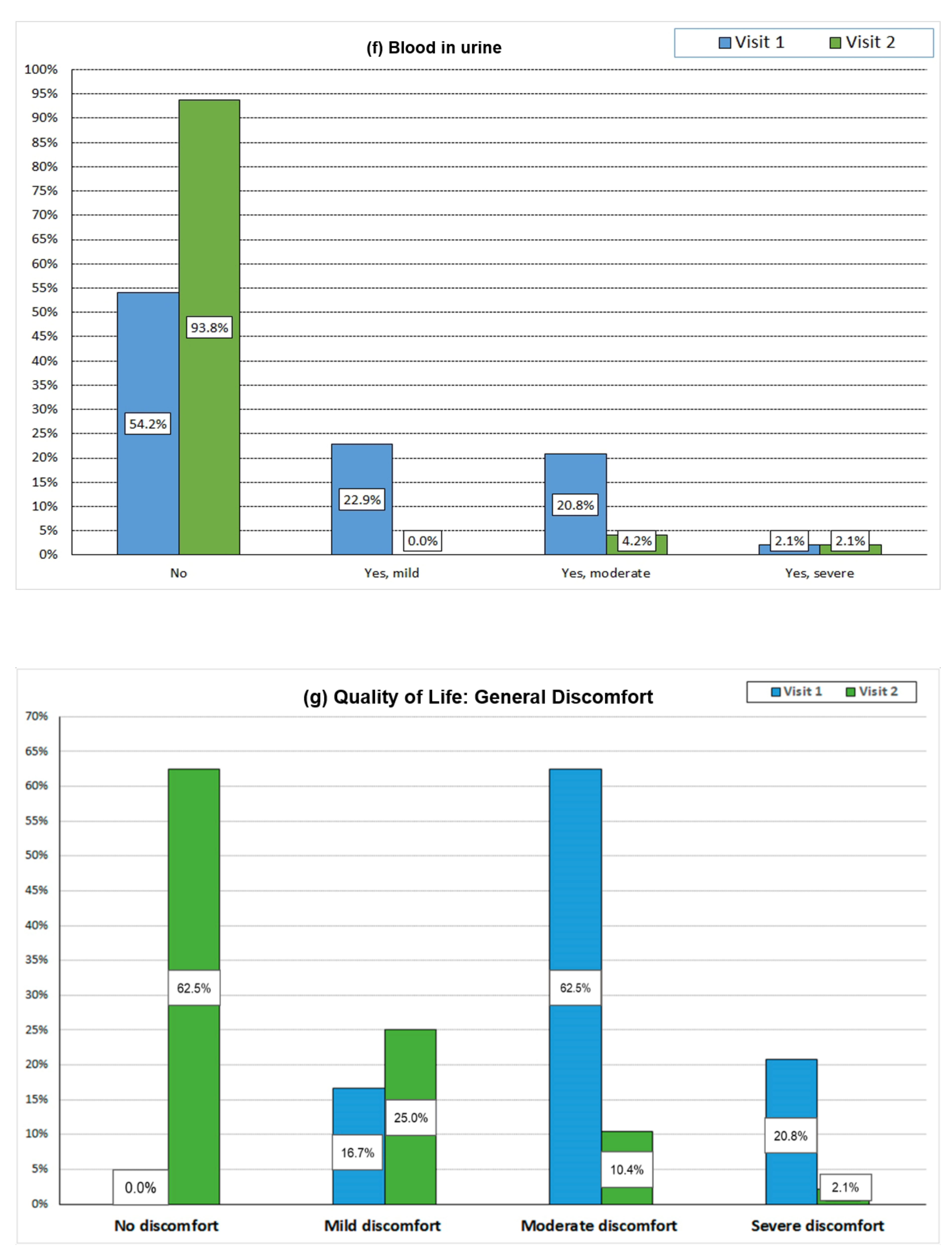
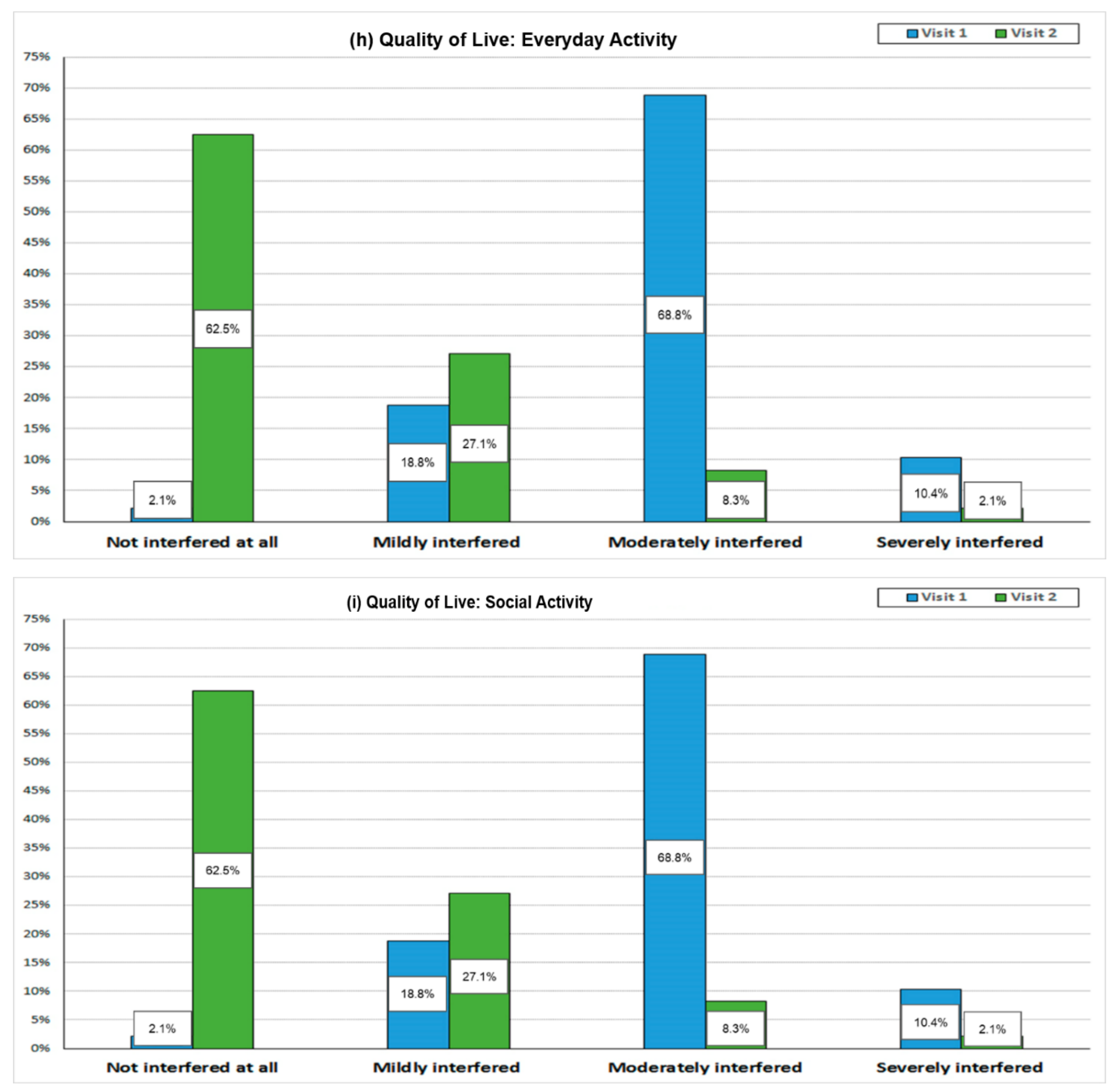
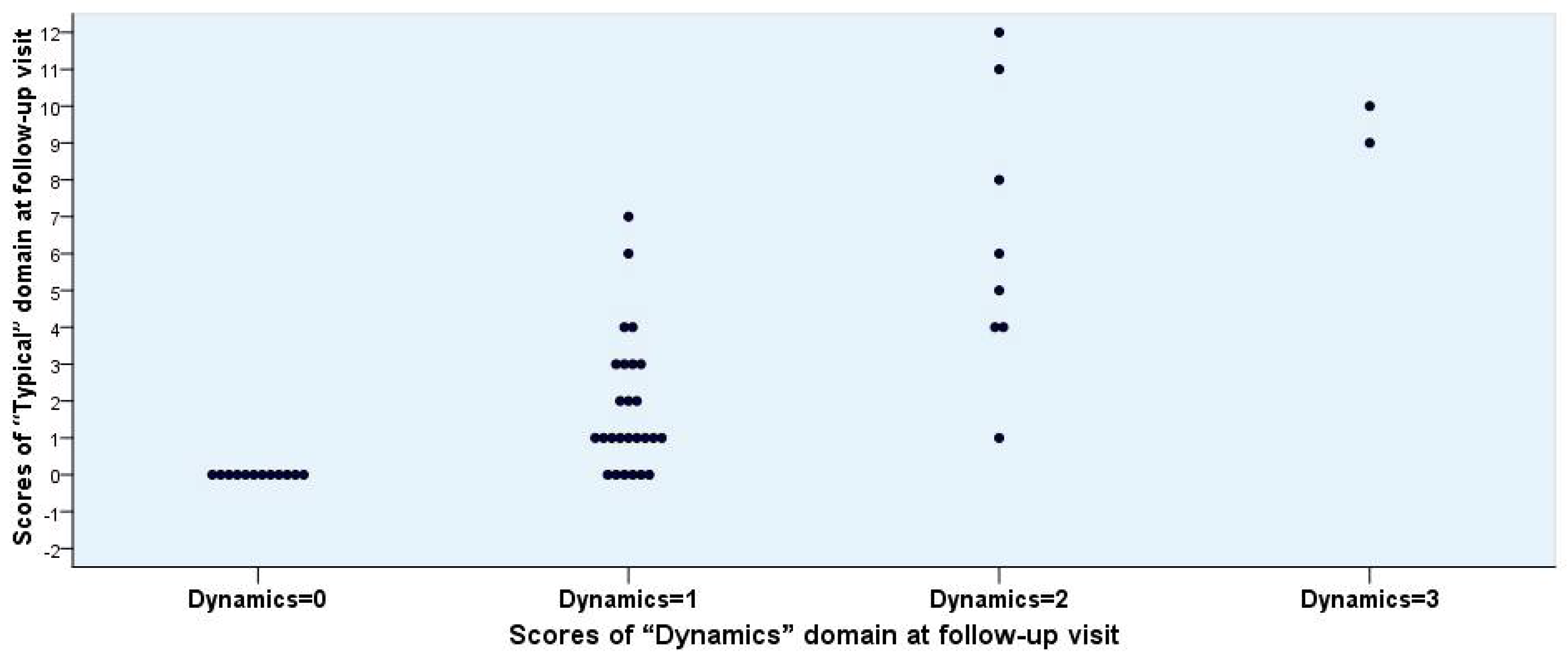
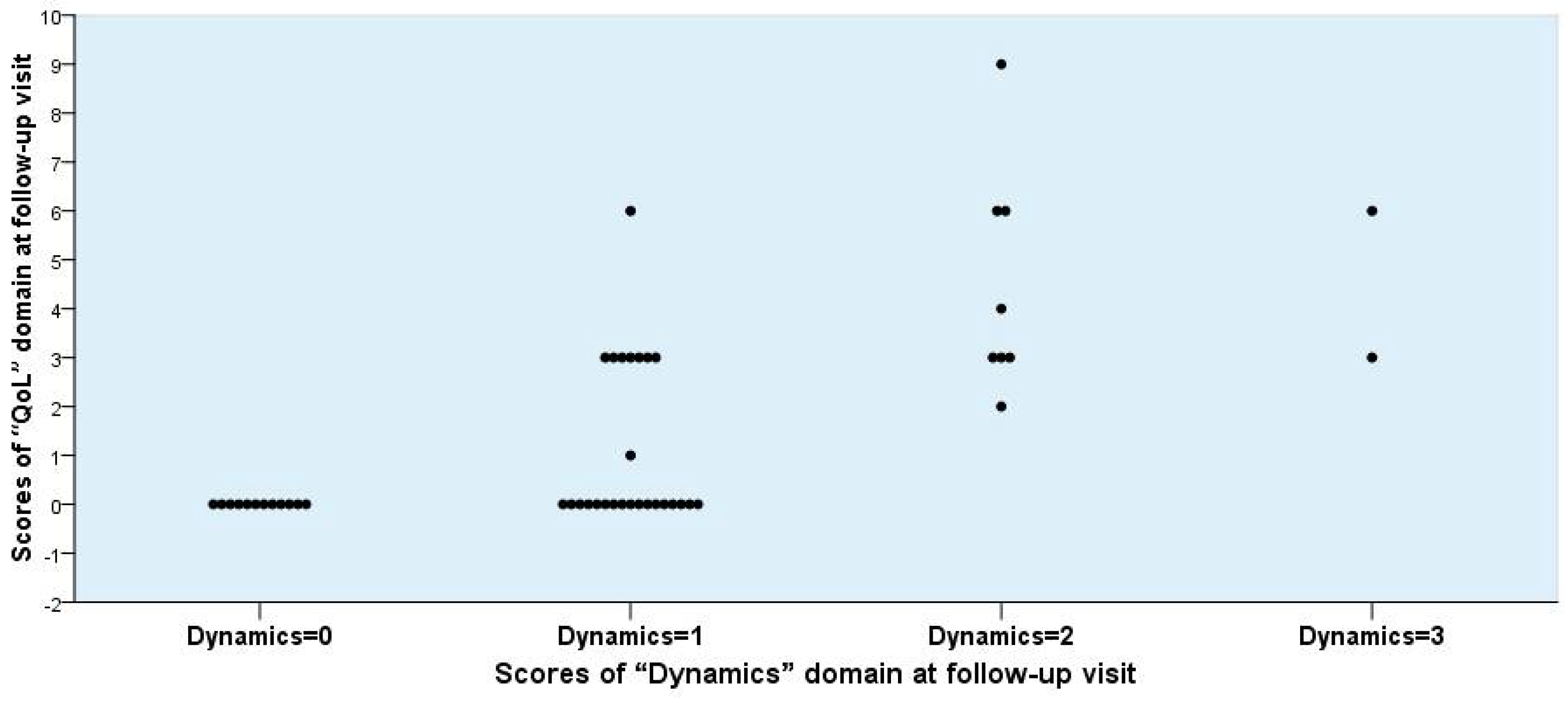
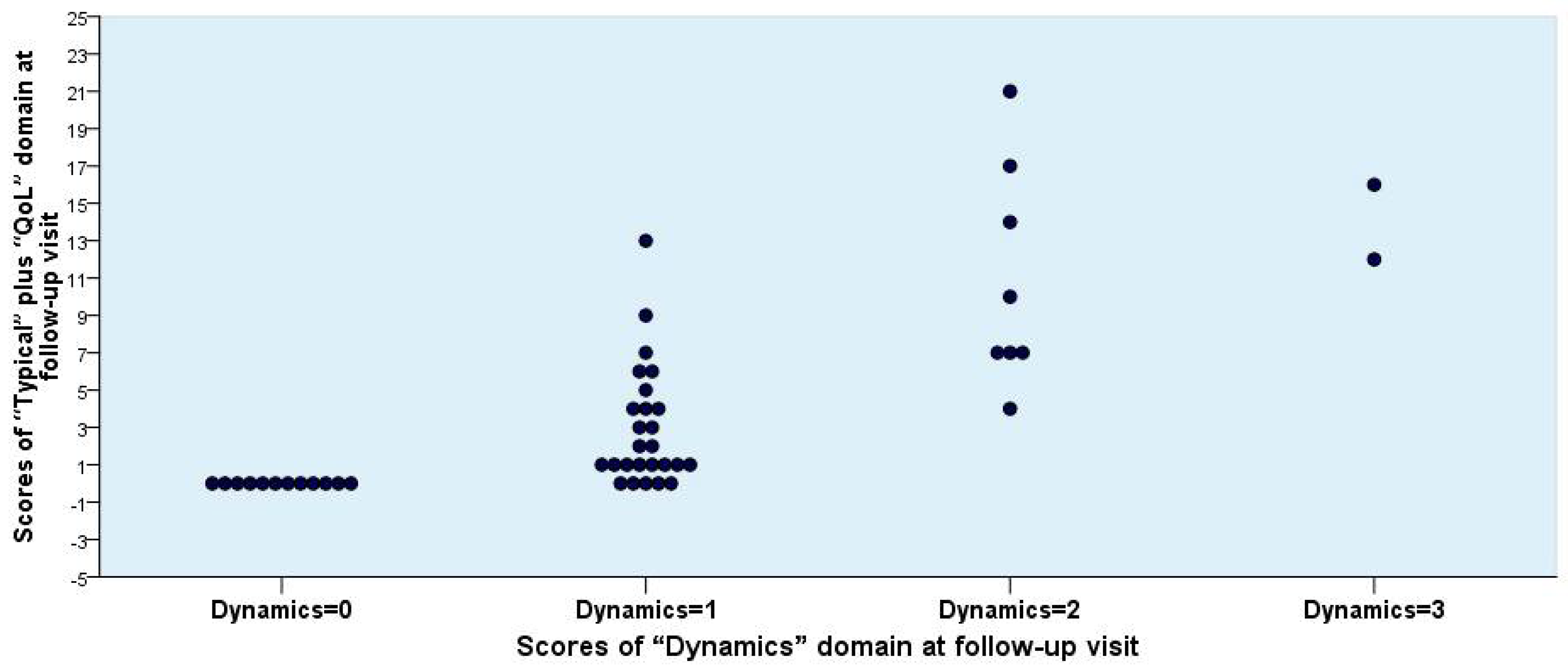
3.2. Dynamics
3.3. Reliability of the Follow-Up form of the ACSS
3.4. ACSS Scores at First and Follow-Up Visits
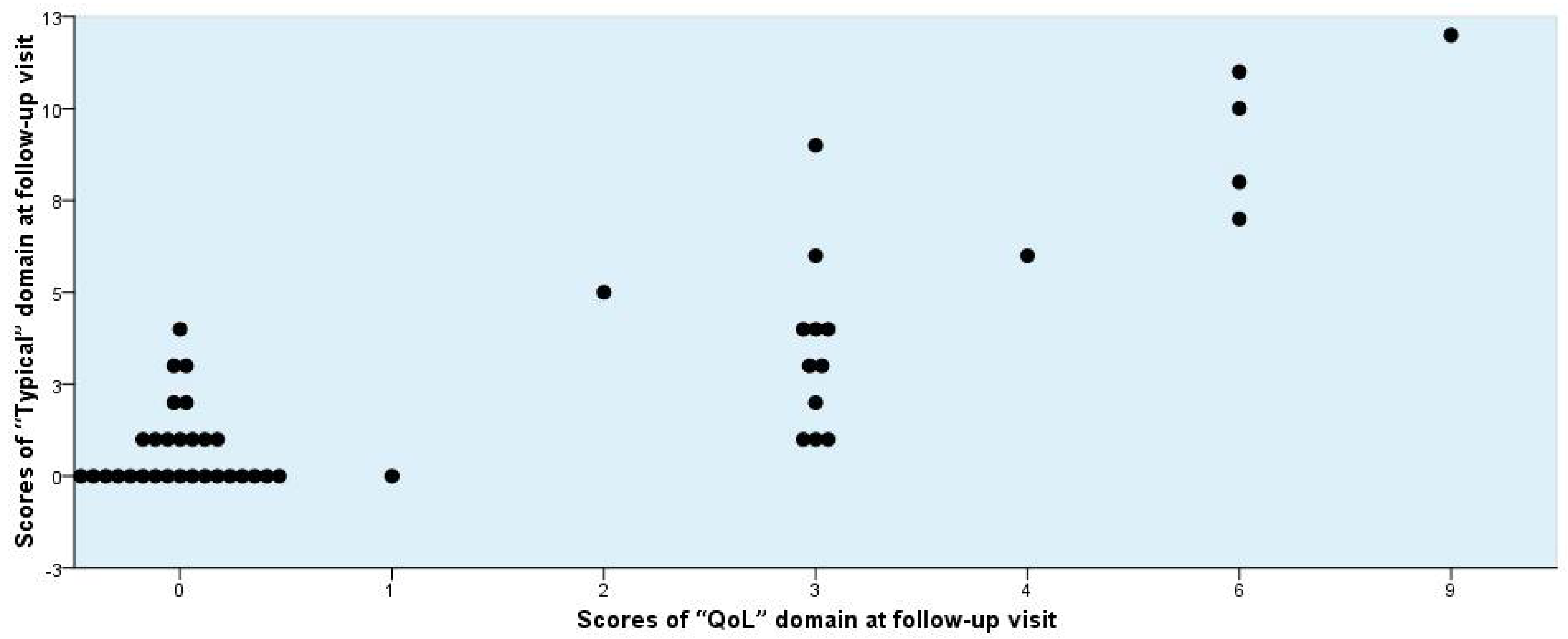
3.5. Correlation between Symptom Scores and Outcome
3.6. Clinical Outcome Categories
3.7. Patient-Reported Outcome Assessment
4. Discussion
- Score 0: Now I feel back to normal (All symptoms are gone);
- Score 1: Now I feel much better (Most of the symptoms are gone);
- Score 2: Now I feel only somewhat better (Only some of the symptoms are gone);
- Score 3: Now, there are barely any changes (I have still about the same symptoms);
- Score 4: Now, I feel worse (My condition is worse)
5. Conclusions
6. Patents: Copyright and Translations of the ACSS in Other Languages
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Alidjanov, J.F.; Abdufattaev, U.A.; Makhsudov, S.A.; Pilatz, A.; Akilov, F.A.; Naber, K.G.; Wagenlehner, F.M. New self-reporting questionnaire to assess urinary tract infections and differential diagnosis: Acute cystitis symptom score. Urol. Int. 2014, 92, 230–236. [Google Scholar] [CrossRef] [PubMed]
- Alidjanov, J.F.; Abdufattaev, U.A.; Makhsudov, S.A.; Pilatz, A.; Akilov, F.A.; Naber, K.G.; Wagenlehner, F.M. The acute cystitis symptom score for patient-reported outcome assessment. Urol. Int. 2016, 97, 402–409. [Google Scholar] [CrossRef] [PubMed]
- Alidjanov, J.F.; Naber, K.G.; Abdufattaev, U.A.; Pilatz, A.; Wagenlehner, F.M. Reevaluation of the acute cystitis symptom score, a self-reporting questionnaire. Part, I. Development, diagnosis and differential diagnosis. Antibiotics 2018, 7, 6. [Google Scholar] [CrossRef]
- Cronbach, L.J. Test reliability; Its meaning and determination. Psychometrika 1947, 12, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Wilcoxon, F. Individual comparisons by ranking methods. Biom. Bull. 1945, 1, 80–83. [Google Scholar] [CrossRef]
- Student. The Probable Error of a Mean. Biometrika 1908, 6, 1–25. Available online: http://www.jstor.org/stable/2331554 (accessed on 18 May 2018).
- Rosenthal, R.; Rosnow, R.L. Essentials of Behavioral Research: Methods and Data Analysis; McGraw-Hill: New York, NY, USA, 1984. [Google Scholar]
- Henry, D.C., Jr.; Bettis, R.B.; Riffer, E.; Haverstock, D.C.; Kowalsky, S.F.; Manning, K.; Hamed, K.A. Church DA: Comparison of once-daily extended-release ciprofloxacin and conventional twice-daily ciprofloxacin for the treatment of uncomplicated urinary tract infection in women. Clin. Ther. 2002, 24, 2088–2104. [Google Scholar] [CrossRef]
- Stamm, W.E.; Counts, G.W.; Running, K.R.; Fihn, S.; Turck, M.; Holmes, K.K. Diagnosis of coliform infection in acutely dysuric women. N. Engl. J. Med. 1982, 307, 463–468. [Google Scholar] [CrossRef] [PubMed]
- Hooton, T.M.; Roberts, P.L.; Cox, M.E.; Stapleton, A.E. Voided midstream urine culture and acute cystitis in premenopausal women. N. Engl. J. Med. 2013, 369, 1883–1891. [Google Scholar] [CrossRef] [PubMed]
- Rubin, R.H.; Shapiro, E.D.; Andriole, V.T.; Davis, R.J.; Stamm, W.E. Evaluation of new anti-infective drugs for the treatment of urinary tract infection. Infectious diseases society of America and the food and drug administration. Clin. Infect. Dis. 1992, 15, S216–S227. [Google Scholar] [CrossRef] [PubMed]
- Gupta, K.; Hooton, T.M.; Naber, K.G.; Wullt, B.; Colgan, R.; Miller, L.G.; Moran, G.J.; Nicolle, L.E.; Raz, R.; Schaeffer, A.J.; et al. Infectious Diseases Society of A, European Society for M, Infectious D: International clinical practice guidelines for the treatment of acute uncomplicated cystitis and pyelonephritis in women: A 2010 update by the infectious diseases society of America and the European society for microbiology and infectious diseases. Clin. Infect. Dis. 2011, 52, e103–e120. [Google Scholar] [PubMed]
- Grabe, M.; Bartoletti, R.; Bjerklund-Johansen, T.E.; Cai, T.; Çek, M.; Köves, B.; Naber, K.G.; Pickard, R.S.; Tenke, P.; Wagenlehner, F.; et al. Guidelines on Urological Infections; Eau Guidelines, Edition Presented at the 30th EAU Annual Congress. Available online: http://uroweb.org/guideline/urological-infections/ (accessed on 19 December 2015).
- Gágyor, I.; Bleidorn, J.; Kochen, M.M.; Schmiemann, G.; Wegscheider, K.; Hummers-Pradier, E. Ibuprofen versus fosfomycin for uncomplicated urinary tract infection in women: Randomised controlled trial. BMJ 2015, 351, h6544. [Google Scholar] [CrossRef] [PubMed]
- Ivanov, D.; Abramov-Sommariva, D.; Moritz, K.; Eskötter, H.; Kostinenko, T.; Martynyuk, L.; Kolesnik, N.; Naber, K.G. An open label, non-controlled, multicentre, interventional trial to investigate the safety and efficacy of canephron® N in the management of uncomplicated urinary tract infections (uIITs). Clin. Phytosci. 2015, 1, 1–11. [Google Scholar] [CrossRef]
- Cai, T.; Mazzoli, S.; Mondaini, N.; Meacci, F.; Nesi, G.; D’Elia, C.; Malossini, G.; Boddi, V.; Bartoletti, R. The role of asymptomatic bacteriuria in young women with recurrent urinary tract infections: To treat or not to treat? Clin. Infect. Dis. 2012, 55, 771–777. [Google Scholar] [CrossRef] [PubMed]
- Wagenlehner, F.M.; Naber, K.G. Editorial commentary: Treatment of asymptomatic bacteriuria might be harmful. Clin. Infect. Dis. 2015, 61, 1662–1663. [Google Scholar] [PubMed]
- Cai, T.; Nesi, G.; Mazzoli, S.; Meacci, F.; Lanzafame, P.; Caciagli, P.; Mereu, L.; Tateo, S.; Malossini, G.; Selli, C.; et al. Asymptomatic bacteriuria treatment is associated with a higher prevalence of antibiotic resistant strains in women with urinary tract infections. Clin. Infect. Dis. 2015, 61, 1655–1661. [Google Scholar] [CrossRef] [PubMed]
- U.S. Department of Health and Human Services; Food and Drug Administration. Guidance for Industry. Patient-Reported Outcome Measures: Use in Medical Product Development to Support Labeling Claims. December 2009. Available online: http://www.fda.gov/downloads/Drugs/.../Guidances/UCM193282.pdf (accessed on 20 December 2015).
| Score | Answer | Follow-Up Visit n = 48 (100%) |
|---|---|---|
| 0 | Yes, I feel myself great better (All symptoms went off) | 12 (25.0%) |
| 1 | Yes, I feel myself much better (Majority of symptoms were solved) | 26 (54.2%) |
| 2 | Yes, I feel myself somewhat better (Some symptoms are remaining) | 8 (16.7%) |
| 3 | No changes, I feel about the same (All symptoms are remaining) | 2 (4.2%) |
| 4 | Yes, I feel worse (My condition is declining) | 0 (0.0%) |
| Items of the ACSS | Correlation Between Item and Entire ACSS | ||
|---|---|---|---|
| Dynamics | Dynamics | Cronbach’s Alpha If Item Deleted | |
| Typical | Frequency | 0.83 | 0.91 |
| Typical Differential | Urgency | 0.62 | 0.92 |
| Painful urination | 0.63 | 0.91 | |
| Incomplete bladder emptying | 0.77 | 0.91 | |
| Suprapubic pain | 0.63 | 0.91 | |
| Visible blood in urine | 0.80 | 0.91 | |
| Flank pain | 0.40 | 0.92 | |
| Differential Quality of Life (QoL) | Vaginal discharge | 0.52 | 0.93 |
| Urethral discharge | 0.19 | 0.93 | |
| Feeling of chill/fever | 0.40 | 0.92 | |
| General discomfort | 0.40 | 0.92 | |
| QoL | Impairment of everyday activity | 0.82 | 0.91 |
| Impairment of social activity | 0.86 | 0.91 | |
| ACSS Items | Mean Scores (95% CI) | p-Value | Effect Size r | |||
| First Visit | Follow-Up Visit | Difference Between Scores | ||||
| Typical | Frequency | 2.06 (1.82 to 2.31) | 0.52 (0.29 to 0.75) | −1.68 (−1.94 to −1.42) | 0.000 | −0.82 |
| Urgency | 2.02 (1.75 to 2.29) | 0.46 (0.23 to 0.69) | −1.56 (−1.87 to −1.26) | 0.000 | −0.79 | |
| Painful urination | 2.29 (2.08 to 2.51) | 0.46 (0.28 to 0.64) | −1.83 (−2.10 to −1.57) | 0.000 | −0.85 | |
| Incomplete bladder emptying | 1.92 (1.70 to 2.13) | 0.44 (0.21 to 0.66) | −1.48 (−1.74 to −1.22) | 0.000 | −0.82 | |
| Suprapubic pain | 1.81 (1.58 to 2.05) | 0.44 (0.25 to 0.63) | −1.38 (−1.63 to −1.12) | 0.000 | −0.82 | |
| Visible blood in urine | 0.71 (0.45 to 0.96) | 0.15 (−0.02 to 0.32) | −0.56 (−0.84 to −0.29) | 0.001 | −0.50 | |
| Differential | Flank pain | 1.27 (0.99 to 1.55) | 0.60 (0.41 to 0.80) | −0.67 (−0.92 to −0.42) | 0.000 | −0.61 |
| Vaginal discharge | 0.38 (0.18 to 0.57) | 0.19 (0.03 to 0.34) | −0.19 (−0.33 to −0.05) | 0.013 | −0.36 | |
| Urethral discharge | 0.25 (0.10 to 0.40) | 0.08 (0.00 to 0.16) | −0.17 (−0.34 to 0.01) | 0.059 | −0.27 | |
| Feeling of chill/fever a | 0.26 (0.09 to 0.43) | 0.07 (−0.01 to 0.14) | −0.20 (−0.34 to −0.05) | 0.014 | −0.36 | |
| QoL | General discomfort | 2.04 (1.86 to 2.22) | 0.52 (0.30 to 0.74) | −1.52 (−1.78 to −1.26) | 0.000 | −0.82 |
| Impairment of everyday activity | 1.88 (1.70 to 2.05) | 0.50 (0.28 to 0.72) | −1.38 (−1.63 to −1.12) | 0.000 | −0.81 | |
| Impairment of social activity | 1.77 (1.57 to 1.97) | 0.50 (0.28 to 0.72) | −1.27 (−1.52 to −1.03) | 0.000 | −0.80 | |
| ACSS Subscales | Mean total Scores and (95% CI) | p-Value | Effect Size r | |||
| First Visit | Follow-Up Visit | Difference Between Scores | ||||
| “Main Symptoms” b | 6.37 (5.85 to 6.90) | 1.44 (0.90 to 1.98) | −4.94 (−5.56 to −4.31) | 0.00 | −0.86 | |
| “Five Typical Symptoms” c | 10.10 (9.27 to 10.93) | 2.31 (1.44 to 3.18) | −8.75 (−8.75 to −6.84) | 0.00 | 0.86 | |
| “Typical” | 10.81 (9.89 to 11.73) | 2.46 (1.52 to 3.39) | −8.35 (−9.42 to 7.29) | 0.00 | −0.86 | |
| “Differential” a | 2.13 (1.66 to 2.61) | 0.85 (0.51 to 1.19) | −1.28 (−1.63 to −0.94) | 0.00 | −0.77 | |
| “Quality of Life (QoL)” | 5.69 (5.21 to 6.17) | 1.52 (0.87 to 2.17) | −4.17 (−4.86 to −3.47) | 0.00 | −0.82 | |
| “Typical” and “QoL” | 16.50 (15.24 to 17.76) | 3.98 (2.45 to 5.51) | −12.52 (−14.15 to −10.89) | 0.00 | −0.86 | |
| Total ACSS a | 18.37 (16.97 to 19.77) | 4.65 (2.93 to 6.37) | −13.72 (−15.46 to −11.98) | 0.00 | −0.86 | |
| ACSS Items | Spearman’s Rho | p-Value a | |
| Typical | Frequency | 0.27 | 0.067 |
| Urgency | 0.34 | 0.017 | |
| Painful urination | 0.4 | 0.005 | |
| Incomplete bladder emptying | 0.375 | 0.009 | |
| Suprapubic pain | 0.409 | 0.004 | |
| Visible blood in urine | 0.301 | 0.037 | |
| Differential b | Flank pain | 0.079 | 0.594 |
| Vaginal discharge | 0.061 | 0.682 | |
| Urethral discharge | −0.059 | 0.689 | |
| Feeling of chill/fever | 0.088 | 0.563 | |
| QoL | General dyscomfort | 0.581 | 0.000 |
| Impairment of everyday activity | 0.613 | 0.000 | |
| Impairment of social activity | 0.499 | 0.000 | |
| Differences in scores for ACSS Subscales | Spearman’s Rho | p | |
| “Main Symptoms” c | 0.435 | 0.002 | |
| “Five Typical Symptoms” d | 0.514 | 0.000 | |
| “Typical” | 0.508 | 0.000 | |
| “Differential” | 0.152 | 0.312 | |
| “QoL” | 0.606 | 0.000 | |
| “Typical” plus “QoL” | 0.598 | 0.000 | |
| Total ACSS score | 0.624 | 0.000 | |
| ACSS Items | Mean Scores (95% CI) | p-Value | |||||
|---|---|---|---|---|---|---|---|
| Total | Dynamics = 0 | Dynamics = 1 | Dynamics = 2 | Dynamics = 3 | Dynamics | ||
| n = 48 | n = 12 | n = 26 | n = 8 | n = 2 | 0 + 1 vs. 2 + 3 | ||
| Typical | Frequency | 0.52 (0.29 to 0.75) | 0.00 (constant) | 0.46 (0.20 to 0.72) | 1.38 (0.49 to 2.26) | 1.00 (−11.71 to 13.71) | 0.008 |
| Urgency | 0.46 (0.23 to 0.69) | 0.00 (constant) | 0.35 (0.15 to 0.54) | 1.13 (−0.01 to 2.26) | 2.00 (constant) | 0.018 | |
| Painful urination | 0.46 (0.28 to 0.64) | 0.00 (constant) | 0.35 (0.15 to 0.54) | 1.13 (0.83 to 1.42) | 2.00 (constant) | 0.000 | |
| Incomplete bladder emptying | 0.44 (0.21 to 0.66) | 0.00 (constant) | 0.35 (0.12 to 0.57) | 1.00 (0.00 to 2.00) | 2.00 (constant) | 0.018 | |
| Suprapubic pain | 0.44 (0.25 to 0.63) | 0.00 (constant) | 0.35 (0.12 to 0.57) | 1.13 (0.59 to 1.66) | 1.50 (−4.85 to 7.85) | 0.000 | |
| Visible blood in urine | 0.15 (−0.02 to 0.32) | 0.00 (constant) | 0.00 (constant) | 0.63 (−0.37 to 1.62) | 1.00 (−11.71 to 13.71) | 0.154 | |
| QoL | General dyscomfort | 0.52 (0.30 to 0.74) | 0.00 (constant) | 0.38 (0.15 to 0.62) | 1.50 (0.73 to 2.27) | 1.50 (−4.85 to 7.85) | 0.000 |
| Impairment of everyday activity | 0.50 (0.28 to 0.72) | 0.00 (constant) | 0.35 (0.12 to 0.57) | 1.50 (0.87 to 2.13) | 1.50 (−4.85 to 7.85) | 0.000 | |
| Impairment of social activity | 0.50 (0.28 to 0.72) | 0.00 (constant) | 0.35 (0.12 to 0.57) | 1.50 (0.87 to 2.13) | 1.50 (−4.85 to 7.85) | 0.000 | |
| ACSS subscales | Mean scores (95% CI) | ||||||
| “Main Symptoms” a | 1.44 (0.90 to 1.98) | 0.00 (constant) | 1.15 (0.72 to 1.59) | 3.63 (1.63 to 5.62) | 5.00 (−7.71 to 17.71) | 0.000 | |
| “Five Typical Symptoms” b | 2.31 (1.44 to 3.18) | 0.00 (constant) | 1.85 (1.10 to 2.59) | 5.75 (2.73 to 8.77) | 8.50 (−10.56 to 27.56) | 0.000 | |
| “Typical” | 2.46 (1.52 to 3.39) | 0.00 (constant) | 1.85 (1.10 to 2.59) | 6.38 (3.25 to 9.50) | 9.50 (3.15 to 15.85) | 0.000 | |
| “Quality of Life (QoL)” | 0.85 (0.51 to 1.19) | 0.00 (constant) | 1.08 (0.40 to 1.75) | 4.50 (2.55 to 6.45) | 4.50 (−14.56 to 23.56) | 0.000 | |
| “Typical” and “QoL” | 1.52 (0.87 to 2.17) | 0.00 (constant) | 2.92 (1.63 to 4.22) | 10.88 (5.95 to 15.80) | 14.00 (−11.41 to 39.41) | 0.000 | |
| Total ACSS | 3.98 (2.45 to 5.51) | 0.42 (−0.01 to 0.84) | 3.08 (1.97 to 4.20) | 12.63 (7.51 to 17.74) | 17 (constant) | 0.000 | |
| Mode | Domain (s) | Definition of Success | Success | Non-Success |
|---|---|---|---|---|
| (Scores) | N (%) | N (%) | ||
| 1 | Dynamics | ≤1 | 38 (79.2%) | 10 (20.8%) |
| 2 | Main Symptoms a | ≤3, but no item >1 (mild) | 39 (81.2%) | 9 (18.8%) |
| 3 | Five Typical Symptoms b | ≤4, but no item >1 (mild) | 37 (77.1%) | 11 (22.9%) |
| 4 | Typical | ≤4, but no item >1 (mild) | 37 (77.1%) | 11 (22.9%) |
| 5 | QoL | ≤3, but no item >1 (mild) | 42 (87.5%) | 6 (12.5%) |
| 6 | Typical plus QoL | ≤7, but no item >1 (mild) | 37 (77.1%) | 11 (22.9%) |
| 7 c | Typical/QoL | ≤4/≤3, but no item >1 (mild) | 37 (77.1%) | 11 (22.9%) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alidjanov, J.F.; Naber, K.G.; Abdufattaev, U.A.; Pilatz, A.; Wagenlehner, F.M. Reevaluation of the Acute Cystitis Symptom Score, a Self-Reporting Questionnaire. Part II. Patient-Reported Outcome Assessment. Antibiotics 2018, 7, 43. https://doi.org/10.3390/antibiotics7020043
Alidjanov JF, Naber KG, Abdufattaev UA, Pilatz A, Wagenlehner FM. Reevaluation of the Acute Cystitis Symptom Score, a Self-Reporting Questionnaire. Part II. Patient-Reported Outcome Assessment. Antibiotics. 2018; 7(2):43. https://doi.org/10.3390/antibiotics7020043
Chicago/Turabian StyleAlidjanov, Jakhongir F., Kurt G. Naber, Ulugbek A. Abdufattaev, Adrian Pilatz, and Florian M. Wagenlehner. 2018. "Reevaluation of the Acute Cystitis Symptom Score, a Self-Reporting Questionnaire. Part II. Patient-Reported Outcome Assessment" Antibiotics 7, no. 2: 43. https://doi.org/10.3390/antibiotics7020043
APA StyleAlidjanov, J. F., Naber, K. G., Abdufattaev, U. A., Pilatz, A., & Wagenlehner, F. M. (2018). Reevaluation of the Acute Cystitis Symptom Score, a Self-Reporting Questionnaire. Part II. Patient-Reported Outcome Assessment. Antibiotics, 7(2), 43. https://doi.org/10.3390/antibiotics7020043





