Ciprofloxacin Affects Host Cells by Suppressing Expression of the Endogenous Antimicrobial Peptides Cathelicidins and Beta-Defensin-3 in Colon Epithelia
Abstract
:1. Introduction
2. Results
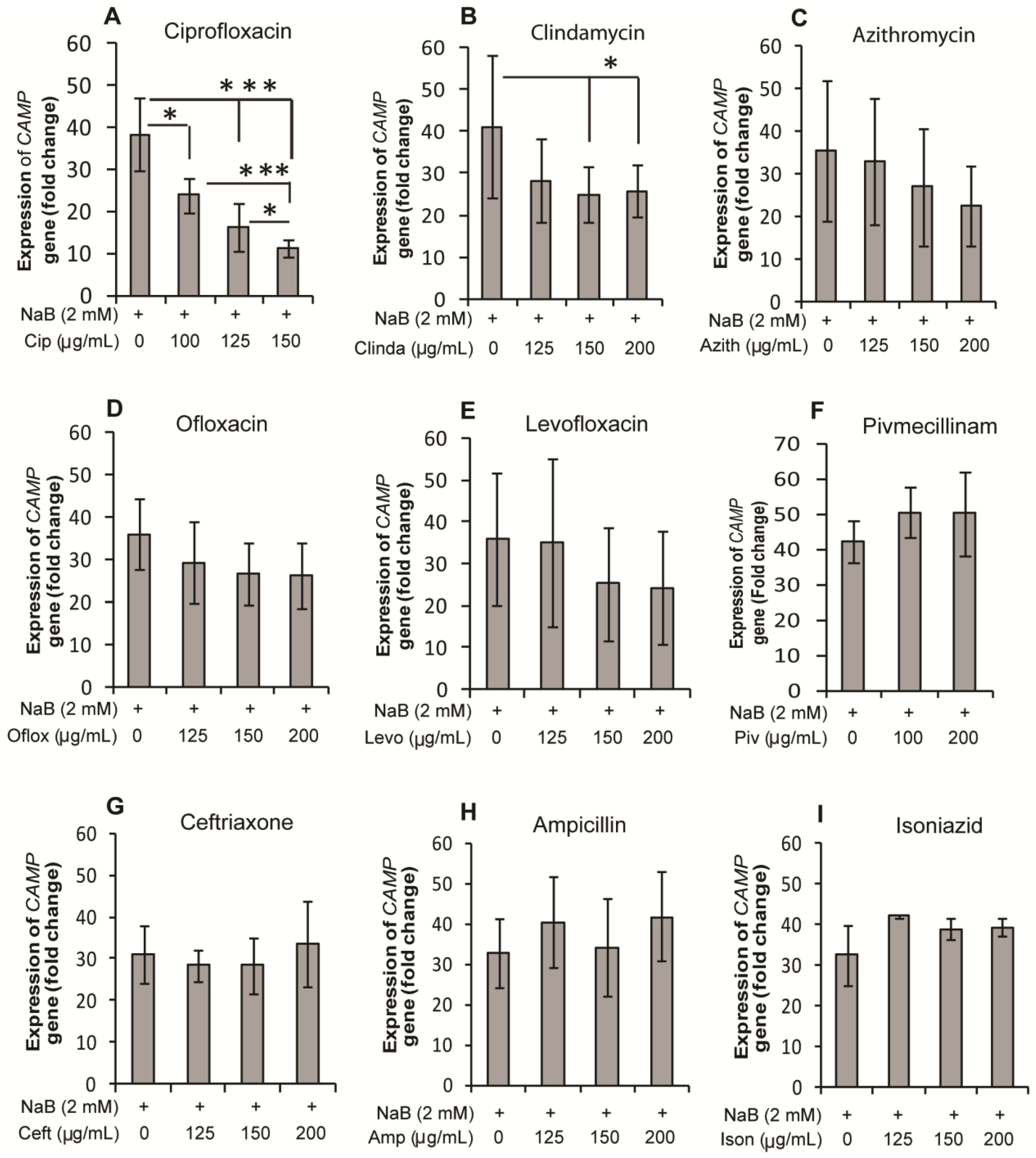
2.1. Effect of Antibiotics on Expression of the CAMP Gene Encoding LL-37 in HT-29, a Colonic Epithelial Cell Line
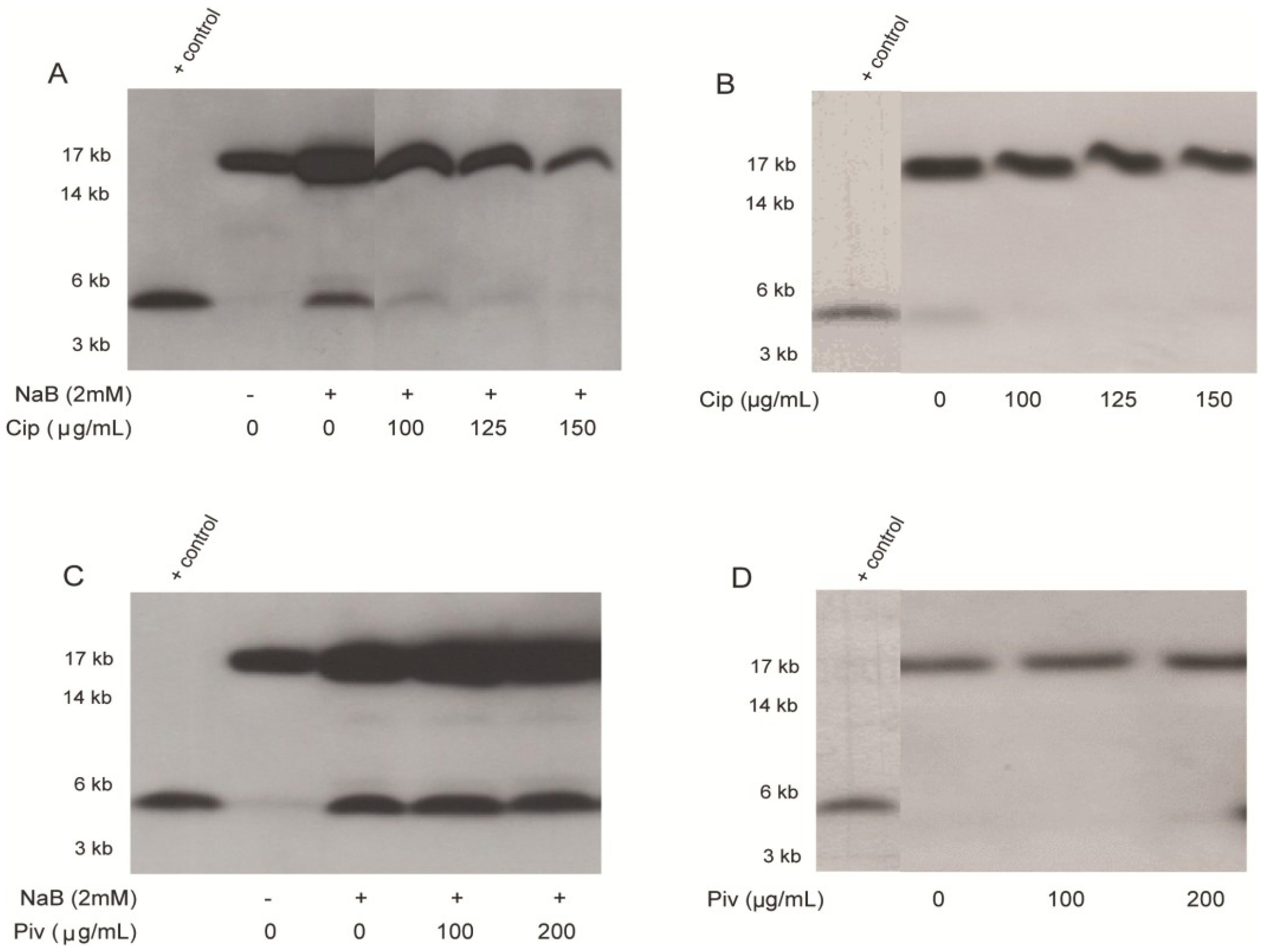
2.2. Effect of Ciprofloxacin and Pivmecillinam on the Expression of LL-37 Peptide and Its Proform hCAP18 in HT-29 Cells
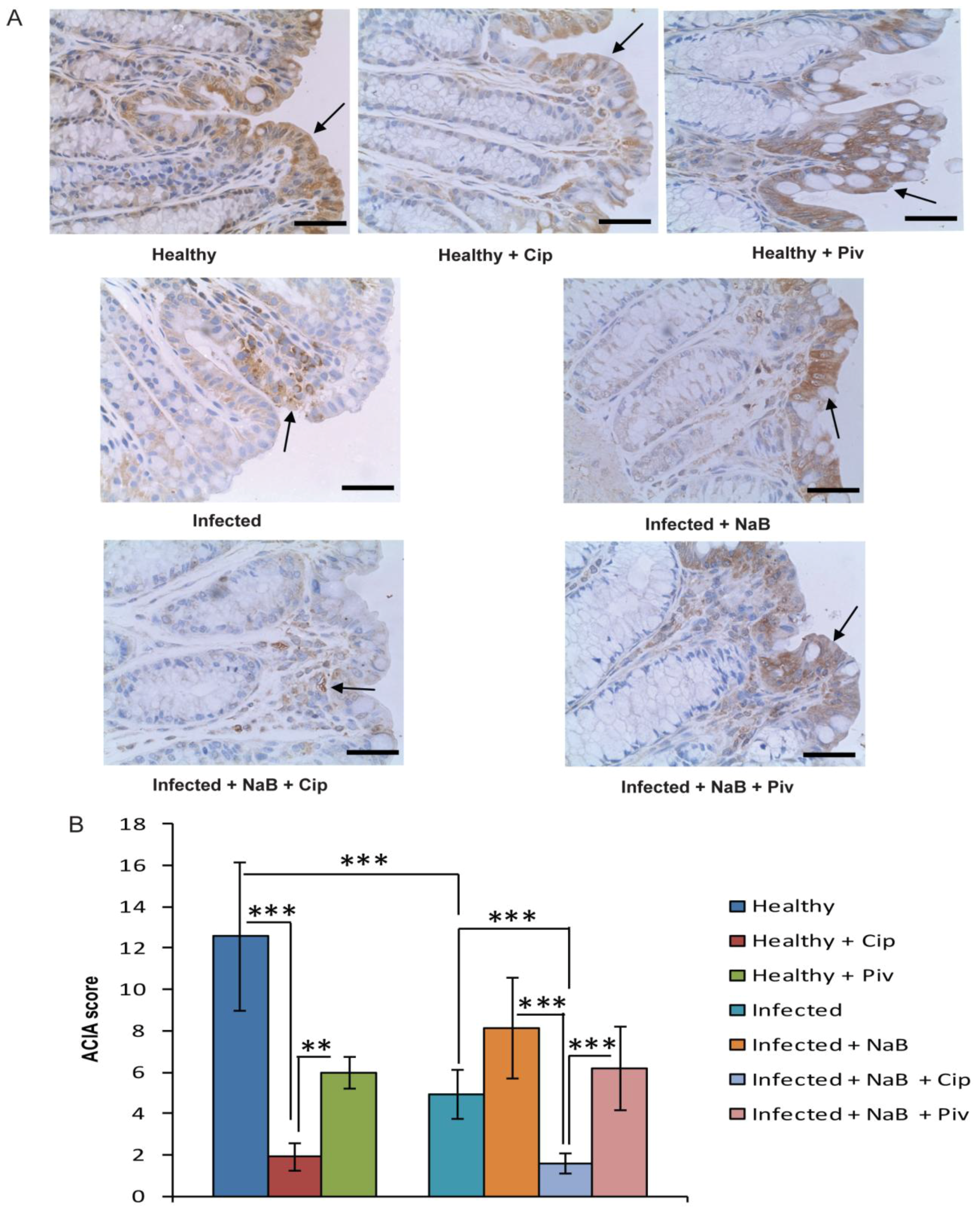
2.3. In Vivo Effect of Ciprofloxacin and Pivmecillinam on Cathelicidin Expression in Colonic Epithelia
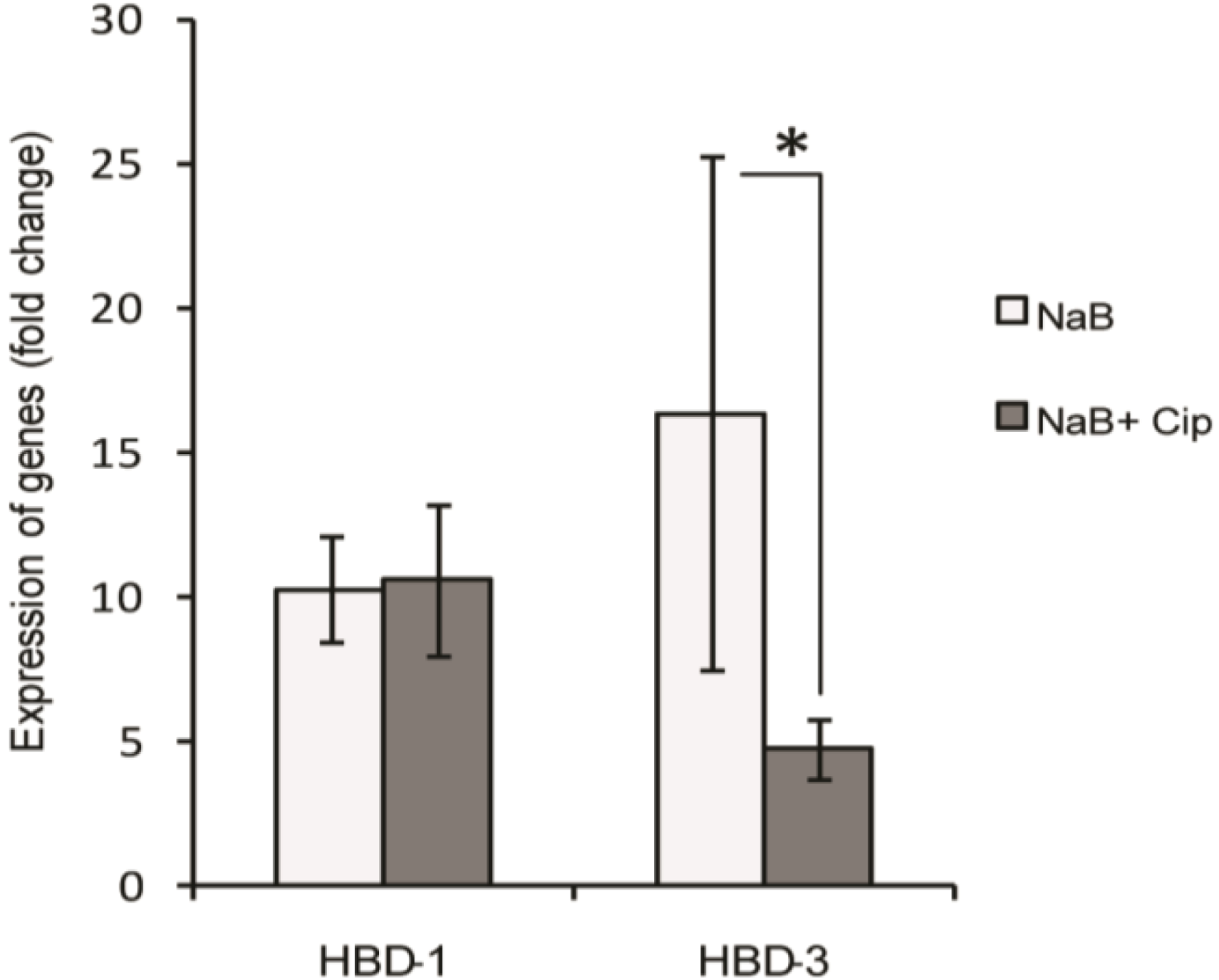
2.4. Effect of Ciprofloxacin on the Butyrate-Induced Expression of Human β-Defensins (HBD) Transcripts in HT-29 Cells
2.5. Suppressive Effect of Ciprofloxacin on Genome-Wide Expression Profile of NaB-Induced Genes in HT-29 Cells
| Entrez gene ID | Gene symbol | Description | Upregulation (NaB vs. unstimulated) | Downregulation (NaB + Cip vs. NaB) | ||
|---|---|---|---|---|---|---|
| Fold change | p-value | Fold change | p-value | |||
| 2840 | GPR17 | G protein-coupled receptor 17 | 4.37 | 0.0002 | −2.93 | 0.0013 |
| 84,873 | GPR128 | G protein-coupled receptor 128 | 2.7 | 0.0098 | −3.4 | 0.006 |
| 3816 | KLK1 | kallikrein 1 | 2.6 | 0.0012 | −1.99 | 0.014 |
| 6035 | RNASE1 | ribonuclease, RNase A family, 1 (pancreatic) | 8.57 | 0.0011 | −1.91 | 0.042 |
| 6274 | S100A3 | S100 calcium binding protein A3 | 10.54 | 0.00005 | −3.02 | 0.016 |
| 6271 | S100A1 | S100 calcium binding protein A1 | 7.72 | 0.0073 | −2.47 | 0.027 |
| 57,402 | S100A14 | S100 calcium binding protein A14 | 2.59 | 0.0056 | −1.95 | 0.015 |
| 4846 | NOS3 | nitric oxide synthase 3 (endothelial cell) | 4.05 | 0.0002 | −3.88 | 0.0008 |
| 143,662 | MUC15 | mucin 15, cell surface associated | 17.2 | 0.03 | −4.7 | 0.05 |
| 394,263 | MUC21 | mucin 21, cell surface associated | 3.27 | 0.0024 | −2.2 | 0.01 |
| 3606 | IL18 | interleukin 18 (interferon-gamma-inducing factor) | 2.93 | 0.0013 | −14.3 | 0.0003 |
| 400,935 | IL17REL | interleukin 17 receptor E-like | 2.57 | 0.0052 | −1.96 | 0.011 |
| 3554 | IL1R1 | interleukin 1 receptor, type I | 6.32 | 0.0071 | −3.26 | 0.013 |
| 3557 | IL1RN | interleukin 1 receptor antagonist | 3.4 | 0.02 | −3.2 | 0.04 |
2.6. Epigenetic Modifications are Involved in the Suppressive Effect of Ciprofloxacin in HT-29 Cells

2.7. In Vitro Effect of Synthetic LL-37 Peptide on C. Difficile

3. Discussion
4. Experimental
4.1. Ethics Statement
4.2. Peptides, Antibodies, Antibiotics and Sodium Butyrate
4.3. Cell Line and Growth Conditions
4.4. Primers
4.5. Bacterial Strains
4.6. Stimulation of Cells
4.7. Real-Time RT-PCR
4.8. Microarray Analysis
4.9. Cell Lysis and Histone Extraction
4.10. Western Blot Analysis
4.11. Immunofluorescence Staining of Phosphorylated Histone H3 at Ser10
4.12. Rabbit Model
4.13. In Situ Immunohistochemical Staining and Quantification of CAP-18 Peptide/Protein Expression in Rectal Mucosa of Rabbits
4.14. In Vitro Bacterial Killing
4.15. Statistical Analyses
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Gottler, L.M.; Ramamoorthy, A. Structure, membrane orientation, mechanism, and function of pexiganan—A highly potent antimicrobial peptide designed from magainin. Biochim. Biophys. Acta 2009, 1788, 1680–1686. [Google Scholar] [CrossRef]
- Hilchie, A.L.; Wuerth, K.; Hancock, R.E. Immune modulation by multifaceted cationic host defense (antimicrobial) peptides. Nat. Chem. Biol. 2013, 9, 761–768. [Google Scholar] [CrossRef]
- Hoskin, D.W.; Ramamoorthy, A. Studies on anticancer activities of antimicrobial peptides. Biochim. Biophys. Acta 2008, 1778, 357–375. [Google Scholar] [CrossRef]
- Zasloff, M. Antimicrobial peptides of multicellular organisms. Nature 2002, 415, 389–395. [Google Scholar] [CrossRef]
- Durr, U.H.; Sudheendra, U.S.; Ramamoorthy, A. LL-37, the only human member of the cathelicidin family of antimicrobial peptides. Biochim. Biophys. Acta 2006, 1758, 1408–1425. [Google Scholar] [CrossRef]
- Dhople, V.; Krukemeyer, A.; Ramamoorthy, A. The human beta-defensin-3, an antibacterial peptide with multiple biological functions. Biochim. Biophys. Acta 2006, 1758, 1499–1512. [Google Scholar] [CrossRef]
- Selsted, M.E.; Ouellette, A.J. Mammalian defensins in the antimicrobial immune response. Nat. Immunol. 2005, 6, 551–557. [Google Scholar] [CrossRef]
- Jarczak, J.; Kosciuczuk, E.M.; Lisowski, P.; Strzalkowska, N.; Jozwik, A.; Horbanczuk, J.; Krzyzewski, J.; Zwierzchowski, L.; Bagnicka, E. Defensins: Natural component of human innate immunity. Human Immunol. 2013, 74, 1069–1079. [Google Scholar] [CrossRef]
- Baroni, A.; Donnarumma, G.; Paoletti, I.; Longanesi-Cattani, I.; Bifulco, K.; Tufano, M.A.; Carriero, M.V. Antimicrobial human beta-defensin-2 stimulates migration, proliferation and tube formation of human umbilical vein endothelial cells. Peptides 2009, 30, 267–272. [Google Scholar] [CrossRef]
- Kai-Larsen, Y.; Agerberth, B. The role of the multifunctional peptide LL-37 in host defense. Front. Biosci. 2008, 13, 3760–3767. [Google Scholar] [CrossRef]
- Koczulla, R.; von Degenfeld, G.; Kupatt, C.; Krotz, F.; Zahler, S.; Gloe, T.; Issbrucker, K.; Unterberger, P.; Zaiou, M.; Lebherz, C.; et al. An angiogenic role for the human peptide antibiotic LL-37/hCAP-18. J. Clin. Invest. 2003, 111, 1665–1672. [Google Scholar] [CrossRef]
- Salzman, N.H.; Hung, K.; Haribhai, D.; Chu, H.; Karlsson-Sjoberg, J.; Amir, E.; Teggatz, P.; Barman, M.; Hayward, M.; Eastwood, D.; et al. Enteric defensins are essential regulators of intestinal microbial ecology. Nat. Immunol. 2010, 11, 76–83. [Google Scholar] [CrossRef]
- Shaykhiev, R.; Beisswenger, C.; Kandler, K.; Senske, J.; Puchner, A.; Damm, T.; Behr, J.; Bals, R. Human endogenous antibiotic LL-37 stimulates airway epithelial cell proliferation and wound closure. Am. J. Physiol. Lung. Cell. Mol. Physiol. 2005, 289, L842–L848. [Google Scholar] [CrossRef]
- Vandamme, D.; Landuyt, B.; Luyten, W.; Schoofs, L. A comprehensive summary of LL-37, the factotum human cathelicidin peptide. Cell. Immunol. 2012, 280, 22–35. [Google Scholar] [CrossRef]
- Schauber, J.; Dorschner, R.A.; Yamasaki, K.; Brouha, B.; Gallo, R.L. Control of the innate epithelial antimicrobial response is cell-type specific and dependent on relevant microenvironmental stimuli. Immunology 2006, 118, 509–519. [Google Scholar]
- Raqib, R.; Sarker, P.; Bergman, P.; Ara, G.; Lindh, M.; Sack, D.A.; Nasirul Islam, K.M.; Gudmundsson, G.H.; Andersson, J.; Agerberth, B. Improved outcome in shigellosis associated with butyrate induction of an endogenous peptide antibiotic. Proc. Natl. Acad. Sci. USA 2006, 103, 9178–9183. [Google Scholar] [CrossRef]
- Schauber, J.; Svanholm, C.; Termen, S.; Iffland, K.; Menzel, T.; Scheppach, W.; Melcher, R.; Agerberth, B.; Luhrs, H.; Gudmundsson, G.H. Expression of the cathelicidin LL-37 is modulated by short chain fatty acids in colonocytes: Relevance of signalling pathways. Gut 2003, 52, 735–741. [Google Scholar] [CrossRef]
- Mandell, L.A.; Afnan, M. Mechanisms of interaction among subinhibitory concentrations of antibiotics, human polymorphonuclear neutrophils, and gram-negative bacilli. Antimicrob. Agents Chemother. 1991, 35, 1291–1297. [Google Scholar] [CrossRef]
- Nau, R.; Eiffert, H. Modulation of release of proinflammatory bacterial compounds by antibacterials: Potential impact on course of inflammation and outcome in sepsis and meningitis. Clin. Microbiol. Rev. 2002, 15, 95–110. [Google Scholar] [CrossRef]
- Tauber, S.C.; Nau, R. Immunomodulatory properties of antibiotics. Curr. Mol. Pharmacol. 2008, 1, 68–79. [Google Scholar] [CrossRef]
- Bartlett, J.G. Clinical practice. Antibiotic-associated diarrhea. N. Engl. J. Med. 2002, 346, 334–339. [Google Scholar] [CrossRef]
- Ubeda, C.; Pamer, E.G. Antibiotics, microbiota, and immune defense. Trends Immunol. 2012, 33, 459–466. [Google Scholar] [CrossRef]
- Blondeau, J.M. What have we learned about antimicrobial use and the risks for Clostridium difficile-associated diarrhoea? J. Antimicrob. Chemother. 2009, 63, 238–242. [Google Scholar] [CrossRef]
- Expression data from HT-29, a human colonic epithelial cell line. Available online: http://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE45220 (accessed on 16 March 2013).
- Kida, Y.; Shimizu, T.; Kuwano, K. Sodium butyrate up-regulates cathelicidin gene expression via activator protein-1 and histone acetylation at the promoter region in a human lung epithelial cell line, EBC-1. Mol. Immunol. 2006, 43, 1972–1981. [Google Scholar] [CrossRef]
- Schauber, J.; Iffland, K.; Frisch, S.; Kudlich, T.; Schmausser, B.; Eck, M.; Menzel, T.; Gostner, A.; Luhrs, H.; Scheppach, W. Histone-deacetylase inhibitors induce the cathelicidin LL-37 in gastrointestinal cells. Mol. Immunol. 2004, 41, 847–854. [Google Scholar] [CrossRef]
- Steinmann, J.; Halldorsson, S.; Agerberth, B.; Gudmundsson, G.H. Phenylbutyrate induces antimicrobial peptide expression. Antimicrob. Agents Chemother. 2009, 53, 5127–5133. [Google Scholar] [CrossRef]
- Bode, A.M.; Dong, Z. Inducible covalent posttranslational modification of histone H3. Sci. STKE 2005, 2005, re4. [Google Scholar]
- Dalhoff, A. Immunomodulatory activities of fluoroquinolones. Infection 2005, 33, 55–70. [Google Scholar] [CrossRef]
- Cacchillo, D.A.; Walters, J.D. Effect of ciprofloxacin on killing of Actinobacillus actinomycetemcomitans by polymorphonuclear leukocytes. Antimicrob. Agents Chemother. 2002, 46, 1980–1984. [Google Scholar] [CrossRef]
- Wong, J.P.; Schnell, G.; Simpson, M.; Saravolac, E. Effects of liposome-encapsulated ciprofloxacin on phagocytosis, nitric oxide and intracellular killing of Staphylcoccus aureus by murine macrophages. Artif. Cells Blood Substit. Immobil. Biotechnol. 2000, 28, 415–428. [Google Scholar] [CrossRef]
- Kaji, M.; Tanaka, J.; Sugita, J.; Kato, N.; Ibata, M.; Shono, Y.; Ohta, S.; Kondo, T.; Asaka, M.; Imamura, M. Ciprofloxacin inhibits lipopolysaccharide-induced toll-like receptor-4 and 8 expression on human monocytes derived from adult and cord blood. Ann. Hematol. 2008, 87, 229–231. [Google Scholar] [CrossRef]
- Katsuno, G.; Takahashi, H.K.; Iwagaki, H.; Sugita, S.; Mori, S.; Saito, S.; Yoshino, T.; Nishibori, M.; Tanaka, N. The effect of ciprofloxacin on CD14 and toll-like receptor-4 expression on human monocytes. Shock 2006, 25, 247–253. [Google Scholar] [CrossRef]
- Kolios, G.; Manousou, P.; Bourikas, L.; Notas, G.; Tsagarakis, N.; Mouzas, I.; Kouroumalis, E. Ciprofloxacin inhibits cytokine-induced nitric oxide production in human colonic epithelium. Eur. J. Clin. Invest. 2006, 36, 720–729. [Google Scholar] [CrossRef]
- Dalhoff, A.; Shalit, I. Immunomodulatory effects of quinolones. Lancet Infect. Dis. 2003, 3, 359–371. [Google Scholar] [CrossRef]
- Islam, D.; Bandholtz, L.; Nilsson, J.; Wigzell, H.; Christensson, B.; Agerberth, B.; Gudmundsson, G. Downregulation of bactericidal peptides in enteric infections: A novel immune escape mechanism with bacterial DNA as a potential regulator. Nat. Med. 2001, 7, 180–185. [Google Scholar] [CrossRef]
- Shalit, I.; Kletter, Y.; Weiss, K.; Gruss, T.; Fabian, I. Enhanced hematopoiesis in sublethally irradiated mice treated with various quinolones. Eur. J. Haematol. 1997, 58, 92–98. [Google Scholar]
- Yamashita, Y.; Ashizawa, T.; Morimoto, M.; Hosomi, J.; Nakano, H. Antitumor quinolones with mammalian topoisomerase II mediated DNA cleavage activity. Cancer Res. 1992, 52, 2818–2822. [Google Scholar]
- Hsu, K.; Champaiboon, C.; Guenther, B.D.; Sorenson, B.S.; Khammanivong, A.; Ross, K.F.; Geczy, C.L.; Herzberg, M.C. Anti-Infective Protective Properties of S100 Calgranulins. Antiinflamm. Antiallergy Agents Med. Chem. 2009, 8, 290–305. [Google Scholar] [CrossRef]
- Dyer, K.D.; Rosenberg, H.F. The RNase a superfamily: Generation of diversity and innate host defense. Mol. Divers. 2006, 10, 585–597. [Google Scholar] [CrossRef]
- Yamasaki, K.; Schauber, J.; Coda, A.; Lin, H.; Dorschner, R.A.; Schechter, N.M.; Bonnart, C.; Descargues, P.; Hovnanian, A.; Gallo, R.L. Kallikrein-mediated proteolysis regulates the antimicrobial effects of cathelicidins in skin. FASEB J. 2006, 20, 2068–2080. [Google Scholar] [CrossRef]
- Kanada, K.N.; Nakatsuji, T.; Gallo, R.L. Doxycycline Indirectly Inhibits Proteolytic Activation of Tryptic Kallikrein-Related Peptidases and Activation of Cathelicidin. J. Invest. Dermatol. 2012, 132, 1435–1442. [Google Scholar] [CrossRef]
- Le Poul, E.; Loison, C.; Struyf, S.; Springael, J.Y.; Lannoy, V.; Decobecq, M.E.; Brezillon, S.; Dupriez, V.; Vassart, G.; van Damme, J.; et al. Functional characterization of human receptors for short chain fatty acids and their role in polymorphonuclear cell activation. J. Biol. Chem. 2003, 278, 25481–25489. [Google Scholar] [CrossRef]
- Maslowski, K.M.; Vieira, A.T.; Ng, A.; Kranich, J.; Sierro, F.; Yu, D.; Schilter, H.C.; Rolph, M.S.; Mackay, F.; Artis, D.; et al. Regulation of inflammatory responses by gut microbiota and chemoattractant receptor GPR43. Nature 2009, 461, 1282–1286. [Google Scholar] [CrossRef]
- Vaishnavi, C. Established and potential risk factors for Clostridum difficile infection. Indian J. Med. Microbiol. 2009, 27, 289–300. [Google Scholar] [CrossRef]
- Hing, T.C.; Ho, S.; Shih, D.Q.; Ichikawa, R.; Cheng, M.; Chen, J.; Chen, X.; Law, I.; Najarian, R.; Kelly, C.P.; et al. The antimicrobial peptide cathelicidin modulates Clostridium difficile-associated colitis and toxin A-mediated enteritis in mice. Gut 2012, 62, 1295–1305. [Google Scholar]
- Giesemann, T.; Guttenberg, G.; Aktories, K. Human alpha-defensins inhibit Clostridium difficile toxin B. Gastroenterology 2008, 134, 2049–2058. [Google Scholar] [CrossRef]
- Brandl, K.; Plitas, G.; Mihu, C.N.; Ubeda, C.; Jia, T.; Fleisher, M.; Schnabl, B.; DeMatteo, R.P.; Pamer, E.G. Vancomycin-resistant enterococci exploit antibiotic-induced innate immune deficits. Nature 2008, 455, 804–807. [Google Scholar] [CrossRef]
- Yoshio, H.; Tollin, M.; Gudmundsson, G.H.; Lagercrantz, H.; Jornvall, H.; Marchini, G.; Agerberth, B. Antimicrobial polypeptides of human vernix caseosa and amniotic fluid: Implications for newborn innate defense. Pediatr. Res. 2003, 53, 211–216. [Google Scholar] [CrossRef]
- Lee, A.Y.; Paweletz, C.P.; Pollock, R.M.; Settlage, R.E.; Cruz, J.C.; Secrist, J.P.; Miller, T.A.; Stanton, M.G.; Kral, A.M.; Ozerova, N.D.; et al. Quantitative analysis of histone deacetylase-1 selective histone modifications by differential mass spectrometry. J. Proteome. Res. 2008, 7, 5177–5186. [Google Scholar] [CrossRef]
- Cunnane, G.; Bjork, L.; Ulfgren, A.K.; Lindblad, S.; FitzGerald, O.; Bresnihan, B.; Andersson, U. Quantitative analysis of synovial membrane inflammation: A comparison between automated and conventional microscopic measurements. Ann. Rheum. Dis. 1999, 58, 493–499. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Sarker, P.; Mily, A.; Mamun, A.A.; Jalal, S.; Bergman, P.; Raqib, R.; Gudmundsson, G.H.; Agerberth, B. Ciprofloxacin Affects Host Cells by Suppressing Expression of the Endogenous Antimicrobial Peptides Cathelicidins and Beta-Defensin-3 in Colon Epithelia. Antibiotics 2014, 3, 353-374. https://doi.org/10.3390/antibiotics3030353
Sarker P, Mily A, Mamun AA, Jalal S, Bergman P, Raqib R, Gudmundsson GH, Agerberth B. Ciprofloxacin Affects Host Cells by Suppressing Expression of the Endogenous Antimicrobial Peptides Cathelicidins and Beta-Defensin-3 in Colon Epithelia. Antibiotics. 2014; 3(3):353-374. https://doi.org/10.3390/antibiotics3030353
Chicago/Turabian StyleSarker, Protim, Akhirunnesa Mily, Abdullah Al Mamun, Shah Jalal, Peter Bergman, Rubhana Raqib, Gudmundur H. Gudmundsson, and Birgitta Agerberth. 2014. "Ciprofloxacin Affects Host Cells by Suppressing Expression of the Endogenous Antimicrobial Peptides Cathelicidins and Beta-Defensin-3 in Colon Epithelia" Antibiotics 3, no. 3: 353-374. https://doi.org/10.3390/antibiotics3030353





