The Prevalence and Drug Susceptibility of Candida Species and an Analysis of Risk Factors for Oral Candidiasis—A Retrospective Study
Abstract
1. Introduction
2. Results
3. Discussion
4. Materials and Methods
4.1. The Process of Diagnosing Patients
4.2. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Vila, T.; Sultan, A.S.; Montelongo-Jauregui, D.; Jabra-Rizk, M.A. Oral Candidiasis: A Disease of Opportunity. J. Fungi 2020, 6, 15. [Google Scholar] [CrossRef]
- Hertel, M.; Schmidt-Westhausen, A.M.; Strietzel, F.P. Local, systemic, demographic, and health-related factors influencing pathogenic yeast spectrum and antifungal drug administration frequency in oral candidiasis: A retrospective study. Clin. Oral Investig. 2016, 20, 1477–1486. [Google Scholar] [CrossRef]
- Taei, M.; Chadeganipour, M.; Mohammadi, R. An alarming rise of non-albicans Candida species and uncommon yeasts in clinical samples: A combination of various molecular techniques for identification of etiologic agents. BMC Res. Notes 2019, 12, 779. [Google Scholar] [CrossRef]
- Bochniak, M.; Kusiak, A.; Kędzia, A. Analysis of drug resistance in yeast-like fungi isolated from oral mucosa infections. Pol. J. Environ. Stud. 2015, 24, 49–55. [Google Scholar]
- Millsop, J.W.; Fazel, N. Oral candidiasis. Clin. Dermatol. 2016, 34, 487–494. [Google Scholar] [CrossRef] [PubMed]
- Hellstein, J.W.; Marek, C.L. Candidiasis: Red and White Manifestations in the Oral Cavity. Head Neck Pathol. 2019, 13, 25–32. [Google Scholar] [CrossRef]
- Singh, A.; Verma, R.; Murari, A.; Agrawal, A. Oral candidiasis: An overview. J. Oral Maxillofac. Pathol. 2014, 18 (Suppl. S1), 81–85. [Google Scholar] [CrossRef] [PubMed]
- Kozłowski, Z.; Konopka, T.; Karolewska, E.; Mendak, M. Częstość występowania infekcji jamy ustnej grzybami z rodzaju Candida w pacjentów stomatologicznych. Mikol. Lek. 2007, 14, 169–172. [Google Scholar]
- Gómez-Gaviria, M.; Ramírez-Sotelo, U.; Mora-Montes, H.M. Non-albicans Candida Species: Immune Response, Evasion Mechanisms, and New Plant-Derived Alternative Therapies. J. Fungi 2023, 9, 11. [Google Scholar] [CrossRef]
- Shemuel, J.; Bays, D.J.; Thompson, G.R., III; Reef, S.; Snyder, L.; Freifeld, A.J.; Huppert, M.; Salkin, D.; Wilson, M.D.; Galgiani, J.N. Natural history of pulmonary coccidioidomycosis: Further examination of the VA-Armed Forces Database. Med. Mycol. J. 2022, 60, myac054. [Google Scholar] [CrossRef]
- Costa, G.L.d.; Negri, M.; Miranda, R.P.R.d.; Corrêa-Moreira, D.; Pinto, T.C.A.; Ramos, L.d.S.; Ferreira, D.G.; Salomão, B.; Fumian, T.M.; Mannarino, C.F.; et al. Candida palmioleophila: A New Emerging Threat in Brazil? J. Fungi 2023, 9, 770. [Google Scholar] [CrossRef]
- Pfaller, M.A.; Carvalhaes, C.G.; DeVries, S.; Rhomberg, P.R.; Castanheira, M. Impact of COVID-19 on the antifungal susceptibility profiles of isolates collected in a global surveillance program that monitors invasive fungal infections. Med. Mycol. 2022, 60, myac028. [Google Scholar] [CrossRef] [PubMed]
- Prestel, C.; Anderson, E.; Forsberg, K.; Lyman, M.; de Perio, M.A.; Kuhar, D.; Edwards, K.; Rivera, M.; Shugart, A.; Walters, M.; et al. Candida auris Outbreak in a COVID-19 Specialty Care Unit—Florida, July–August 2020. MMWR Morb. Mortal. Wkly. Rep. 2021, 70, 56–57. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, S.; Alfouzan, W. Candida auris: Epidemiology, diagnosis, pathogenesis, antifungal susceptibility, and infection control measures to combat the spread of infections in healthcare facilities. Microorganisms 2021, 9, 807. [Google Scholar] [CrossRef]
- Ayuningtyas, N.F.; Mahdani, F.Y.; Pasaribu, T.A.S.; Chalim, M.; Ayna, V.K.P.; Santosh, A.B.R.; Santacroce, L.; Surboyo, M.D.C. Role of Candida albicans in Oral Carcinogenesis. Pathophysiology 2022, 29, 650–662. [Google Scholar] [CrossRef]
- Ingrosso, G.; Saldi, S.; Marani, S.; Wong, A.Y.W.; Bertelli, M.; Aristei, C.; Zelante, T. Breakdown of Symbiosis in Radiation-Induced Oral Mucositis. J. Fungi 2021, 7, 290. [Google Scholar] [CrossRef]
- Cosio, T.; Pica, F.; Fontana, C.; Pistoia, E.S.; Favaro, M.; Valsecchi, I.; Zarabian, N.; Campione, E.; Botterel, F.; Gaziano, R. Stephanoascus ciferrii Complex: The Current State of Infections and Drug Resistance in Humans. J. Fungi 2024, 10, 294. [Google Scholar] [CrossRef]
- Webb, B.J.; Ferraro, J.P.; Rea, S.; Kaufusi, S.; Goodman, B.E.; Spalding, J. Epidemiology and clinical features of invasive fungal infection in a US health care network. Open Forum Infect. Dis. 2018, 5, ofy187. [Google Scholar] [CrossRef] [PubMed]
- Jebali, A.; Hajjar, F.H.E.; Pourdanesh, F.; Hekmatimoghaddam, S.; Kazemi, B.; Masoudi, A.; Daliri, K.; Sedighi, N. Silver and gold nanostructures: Antifungal property of different shapes of these nanostructures on Candida species. Med. Mycol. 2014, 52, 65–72. [Google Scholar] [CrossRef]
- Kotey, F.C.; Dayie, N.T.; Tetteh-Uarcoo, P.B.; Donkor, E.S. Candida Bloodstream Infections: Changes in Epidemiology and Increase in Drug Resistance. Infect. Dis. Res. Treat. 2021, 14, 11786337211026927. [Google Scholar] [CrossRef]
- Hofs, S.; Mogavero, S.; Hube, B. Interaction of Candida albicans with host cells: Virulence factors, host defense, escape strategies, and the microbiota. J. Microbiol. 2016, 54, 149–169. [Google Scholar] [CrossRef]
- Bamford, C.V.; d’Mello, A.; Nobbs, A.H.; Dutton, L.C.; Vickerman, M.M.; Jenkinson, H.F. Streptococcus gordonii modulates Candida albicans biofilm formation through intergeneric communication. Infect. Immun. 2009, 77, 3696–3704. [Google Scholar] [CrossRef]
- Tsui, C.; Kong, E.F.; Jabra-Rizk, M.A. Pathogenesis of Candida albicans biofilm. Pathog. Dis. 2016, 74, ftw018. [Google Scholar] [CrossRef]
- Neckel, N.; Ohm, C.; Wagendorf, O.; Kielburg, U.; Tröltzsch, D.; Wüster, J.; Preißner, S.; Ronchi, F.; Beck-Broichsitter, B.; Heiland, M.; et al. Swabs versus native specimens in severe head and neck infections: A prospective pilot study and suggestions for clinical management. Oral Maxillofac. Surg. 2025, 29, 81. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Bravo, E.; Arce, M.; Ribeiro-Vidal, H.; Herrera, D.; Sanz, M. The Impact of Candida albicans in the Development, Kinetics, Structure, and Cell Viability of Biofilms on Implant Surfaces—An In Vitro Study with a Validated Multispecies Biofilm Model. Int. J. Mol. Sci. 2024, 25, 3277. [Google Scholar] [CrossRef]
- Montelongo-Jauregui, D.; Srinivasan, A.; Ramasubramanian, A.K.; Lopez-Ribot, J.L. An In Vitro Model for Candida albicans–Streptococcus gordonii Biofilms on Titanium Surfaces. J. Fungi 2018, 4, 66. [Google Scholar] [CrossRef] [PubMed]
- Kubizna, M.; Dawiec, G.; Wiench, R. Efficacy of Curcumin-Mediated Antimicrobial Photodynamic Therapy on Candida spp.—A Systematic Review. Int. J. Mol. Sci. 2024, 25, 8136. [Google Scholar] [CrossRef]
- Wiench, R.; Nowicka, J.; Pajaczkowska, M.; Kuropka, P.; Skaba, D.; Kruczek-Kazibudzka, A.; Kuśka-Kiełbratowska, A.; Grzech-Leśniak, K. Influence of Incubation Time on Ortho-Toluidine Blue Mediated Antimicrobial Photodynamic Therapy Directed against Selected Candida Strains—An In Vitro Study. Int. J. Mol. Sci. 2021, 22, 10971. [Google Scholar] [CrossRef] [PubMed]
- Wiench, R.; Skaba, D.; Matys, J.; Grzech-Leśniak, K. Efficacy of Toluidine Blue—Mediated Antimicrobial Photodynamic Therapy on Candida spp.—A Systematic Review. Antibiotics 2021, 10, 349. [Google Scholar] [CrossRef] [PubMed]
- Kuśka-Kiełbratowska, A.; Wiench, R.; Mertas, A.; Bobela, E.; Kiełbratowski, M.; Łukomska-Szymańska, M.; Tanasiewicz, M.; Skaba, D. Evaluation of the Sensitivity of Selected Candida Strains to Ozonated Water—An In Vitro Study. Medicina 2022, 58, 1731. [Google Scholar] [CrossRef]
- Lu, S.-Y. Oral Candidosis: Pathophysiology and Best Practice for Diagnosis, Classification, and Successful Management. J. Fungi 2021, 7, 555. [Google Scholar] [CrossRef]
- Kubicka-Musiał, M.; Musiał, S.; Wierucka-Młynarczyk, B.; Hüpsch-Marzec, H. Frequency of Occurrence of Fungal Infection and Assessment of Drug Sensitivity of Candida Fungi in Patients with a Burning Sensation in the Oral Mucosa. Dent. Med. Probl. 2011, 48, 364–370. [Google Scholar]
- Bulancea, B.P.; Checherita, L.E.; Foia, G.L.; Stamatin, O.; Teslaru, S.; Lupu, I.C.; Ciobanu, D.G.; Cernei, E.-R.; Carmen, G.; Postolache, M.; et al. The Quantification of Salivary Flow and pH and Stomatognathic System Rehabilitation Interference in Patients with Oral Diseases, Post-Radiotherapy. Appl. Sci. 2022, 12, 3708. [Google Scholar] [CrossRef]
- Bilal, H.; Zhang, D.; Shafiq, M.; Khan, M.N.; Chen, C.; Khan, S.; Wang, Q.; Cai, L.; Islam, R.; Hu, H.; et al. Six-Year Retrospective Analysis of Epidemiology, Risk Factors, and Antifungal Susceptibilities of Candidiasis from a Tertiary Care Hospital in South China. Microbiol. Spectr 2023, 11, e00708–e00723. [Google Scholar] [CrossRef]
- Muzaheed; Alshehri, B.A.; Rabaan, A.A.; El-Masry, O.S.; Acharya, S.; Alzahrani, F.M.; Al Mutair, A.; Alhumaid, S.; Al-Tawfiq, J.A.; Muhammad, J.; et al. A 20-Year Retrospective Clinical Analysis of Candida Infections in a Tertiary Centre: Single-Center Experience. J. Infect. Public Health 2022, 15, 69–74. [Google Scholar] [CrossRef]
- Reinhardt, L.C.; Nascente, P.D.S.; Ribeiro, J.S.; Etges, A.; Lund, R.G. A Single-Center 18-Year Experience with Oral Candidiasis in Brazil: A Retrospective Study of 1,534 Cases. Braz. Oral Res. 2018, 32, e92. [Google Scholar] [CrossRef]
- Hu, L.; He, C.; Zhao, C.; Chen, X.; Hua, H.; Yan, Z. Characterization of Oral Candidiasis and the Candida Species Profile in Patients with Oral Mucosal Diseases. Microb. Pathog. 2019, 134, 103575. [Google Scholar] [CrossRef] [PubMed]
- Sivaramakrishnan, G.; Sridharan, K. Alternatives to Antifungal Therapy for Denture Stomatitis: A Systematic Review and Meta-Analysis. Saudi J. Oral Sci. 2017, 4, 67–71. [Google Scholar] [CrossRef]
- Bochniak, M.; Cydejko, A.; Kusiak, A.; Świetlik, D. Occurrence of Risk Factors for Oral Mucosa Candidiasis and Their Frequency—Retrospective Analysis. Prosthodontics 2020, 70, 289–303. [Google Scholar] [CrossRef]
- Bochniak, M.; Ciesielska, A.; Kusiak, A.; Świetlik, D. Częstość Występowania Czynników Ryzyka Kandydozy Błony Śluzowej Jamy Ustnej: Analiza Retrospektywna. Protet. Stomatol. 2020, 70, 289–303. [Google Scholar] [CrossRef]
- Sardi, J.C.O.; Scorzoni, L.; Bernardi, T.; Fusco-Almeida, A.M.; Mendes Giannini, M.J.S. Candida species: Current epidemiology, pathogenicity, biofilm formation, natural antifungal products and new therapeutic options. J. Med. Microbiol. 2013, 62, 10–24. [Google Scholar] [CrossRef] [PubMed]
- Verma, R.; Pradhan, D.; Hasan, Z.; Singh, H.; Jain, A.K.; Khan, L.A. A systematic review on distribution and antifungal resistance pattern of Candida species in the Indian population. Med. Mycol. 2021, 59, 1145–1165. [Google Scholar] [CrossRef]
- Yang, Z.T.; Wu, L.; Liu, X.Y.; Zhou, M.; Li, J.; Wu, J.Y.; Cai, Y.; Mao, E.Q.; Chen, E.Z.; Lortholary, O. Epidemiology, species distribution and outcome of nosocomial Candida spp. bloodstream infection in Shanghai. BMC Infect. Dis. 2014, 14, 241. [Google Scholar] [CrossRef]
- Mamali, V.; Siopi, M.; Charpantidis, S.; Samonis, G.; Tsakris, A.; Vrioni, G.; on behalf of the Candi-Candi Network. Increasing Incidence and Shifting Epidemiology of Candidemia in Greece: Results from the First Nationwide 10-Year Survey. J. Fungi 2022, 8, 116. [Google Scholar] [CrossRef]
- Clark, T.A.; Hajjeh, R.A. Recent Trends in the Epidemiology of Invasive Mycoses. Curr. Opin. Infect. Dis. 2002, 15, 569–574. [Google Scholar] [CrossRef] [PubMed]
- Dignani, M.C.; Anaissie, E. Human fusariosis. Clin. Microbiol. Infect. 2004, 10 (Suppl. S1), 67–75. [Google Scholar] [CrossRef]
- Maertens, J. Evaluating Prophylaxis of Invasive Fungal Infections in Patients with Haematologic Malignancies. Eur. J. Haematol. 2007, 78, 275–282. [Google Scholar] [CrossRef]
- Ksiezopolska, E.; Gabaldón, T. Evolutionary Emergence of Drug Resistance in Candida Opportunistic Pathogens. Genes 2018, 9, 461. [Google Scholar] [CrossRef]
- Bhattacharya, S.; Sae-Tia, S.; Fries, B.C. Candidiasis and Mechanisms of Antifungal Resistance. Antibiotics 2020, 9, 312. [Google Scholar] [CrossRef] [PubMed]
- Arendrup, M.C. Update on Antifungal Resistance in Aspergillus and Candida. Clin. Microbiol. Infect. 2014, 20, 42–48. [Google Scholar] [CrossRef]
- Calvo, M.; Scalia, G.; Trovato, L. Antifungal Susceptibility Data and Epidemiological Distribution of Candida spp.: An In Vitro Five-Year Evaluation at University Hospital Policlinico of Catania and a Comprehensive Literature Review. Antibiotics 2024, 13, 914. [Google Scholar] [CrossRef]
- Sharma, J.; Rosiana, S.; Razzaq, I.; Shapiro, R.S. Linking Cellular Morphogenesis with Antifungal Treatment and Susceptibility in Candida Pathogens. J. Fungi 2019, 5, 17. [Google Scholar] [CrossRef]
- Campione, E.; Cosio, T.; Lanna, C.; Mazzilli, S.; Ventura, A.; Dika, E.; Gaziano, R.; Dattola, A.; Candi, E.; Bianchi, L. Predictive Role of Vitamin A Serum Concentration in Psoriatic Patients Treated with IL-17 Inhibitors to Prevent Skin and Systemic Fungal Infections. J. Pharmacol. Sci. 2020, 144, 52–56. [Google Scholar] [CrossRef]
- Reich, K.; Warren, R.B.; Lebwohl, M.; Gooderham, M.; Strober, B.; Langley, R.G.; Paul, C.; De Cuyper, D.; Vanvoorden, V.; Madden, C.; et al. Bimekizumab versus Secukinumab in Plaque Psoriasis. N. Engl. J. Med. 2021, 385, 142–152. [Google Scholar] [CrossRef]
- Shulman, J.D.; Rivera-Hidalgo, F.; Beach, M.M. Risk Factors Associated with Denture Stomatitis in the United States. J. Oral Pathol. Med. 2005, 34, 340–346. [Google Scholar] [CrossRef] [PubMed]
- Khan, I.; Ahmad, T.; Manzoor, N.; Rizvi, M.A.; Raza, U.; Premchandani, S. Evaluating the role of local host factors in the candidal colonization of oral cavity: A review update. Natl. J. Maxillofac. Surg. 2020, 11, 169–175. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Abuhajar, E.; Ali, K.; Zulfiqar, G.; Al Ansari, K.; Raja, H.Z.; Bishti, S.; Anweigi, L. Management of Chronic Atrophic Candidiasis (Denture Stomatitis)—A Narrative Review. Int. J. Environ. Res. Public Health 2023, 20, 3029. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ribeiro, A.B.; de Araújo, C.B.; Silva, L.E.V.; Fazan-Junior, R.; Salgado, H.C.; Ribeiro, A.B.; Fortes, C.V.; Bueno, F.L.; de Oliveira, V.C.; de, F.O. Paranhos, H.; et al. Hygiene Protocols for the Treatment of Denture-Related Stomatitis: Local and Systemic Parameters Analysis—A Randomized, Double-Blind Trial Protocol. Trials 2019, 20, 661. [Google Scholar] [CrossRef] [PubMed]
- de Souza, R.; Chaves, C.; Rohani, K.; Bouferguene, S.; Barbeau, J.; Borie, E.; Weber, B.; Fuentes, R.; Crizostomo, L.; Silva-Lovato, C.; et al. Palatal Brushing for the Treatment of Denture Stomatitis: A Multicentre Randomized Controlled Trial. J. Prosthodont. Res. 2022, 67, 93–102. [Google Scholar] [CrossRef]
- Dodds, M.W.; Johnson, D.A.; Yeh, C.K. Health Benefits of Saliva: A Review. J. Dent. 2005, 33, 223–233. [Google Scholar] [CrossRef]
- Perić, M.; Živković, R.; Milić Lemić, A.; Radunović, M.; Miličić, B.; Arsić Arsenijević, V. Severity of Denture-Induced Stomatitis Depending on Risk Factors and Various Candida spp. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2018, 126, 41–47. [Google Scholar] [CrossRef]
- Brantes, M.F.; Azevedo, R.S.; Rozza-de-Menezes, R.E.; Póvoa, H.C.; Tucci, R.; Gouvêa, A.F.; Takahama, A., Jr. Analysis of Risk Factors for Maxillary Denture-Related Oral Mucosal Lesions: A Cross-Sectional Study. Med. Oral Patol. Oral Cir. Bucal 2019, 24, e305–e313. [Google Scholar] [CrossRef]
- Raghavendra Swamy, K.N.; Alla, R.; Mohammed, S.; Konakanchi, A. Role of Antifungal Agents in the Management of Denture Stomatitis. J. Pharm. Technol. 2018, 11, 1365–1369. [Google Scholar]
- De Matteis, V.; Cascione, M.; Toma, C.C.; Albanese, G.; De Giorgi, M.L. Silver Nanoparticles Addition in Poly(Methyl Methacrylate) Dental Matrix: Topographic and Antifungal Studies. Int. J. Mol. Sci. 2019, 20, 4691. [Google Scholar] [CrossRef]
- Yousef, M.; Abdelaziz, A.; Essa, M.; Fahmi, M. Evaluation of Photodynamic Therapy and Miconazole in the Treatment of Denture-Induced Stomatitis. Int. J. Dent. Sci. Res. 2018, 6, 83–87. [Google Scholar] [CrossRef][Green Version]
- Iyer, M.S.; Gujjari, A.K.; Paranthaman, S.; Abu Lila, A.S.; Almansour, K.; Alshammari, F.; Khafagy, E.S.; Arab, H.H.; Gowda, D.V. Development and Evaluation of Supercritical Clove and Cinnamon Extract-Loaded Emulgel Containing Antifungal Agent for Denture-Associated Stomatitis. Gels 2022, 8, 33. [Google Scholar] [CrossRef]
- Feller, L.; Khammissa, R.A.G.; Chandran, R.; Altini, M.; Lemmer, J. Oral Candidosis in Relation to Oral Immunity. J. Oral Pathol. Med. 2014, 43, 563–569. [Google Scholar] [CrossRef] [PubMed]
- Chouhan, S.; Kallianpur, S.; Prabhu, K.T.; Tijare, M.; Kasetty, S.; Gupta, S. Candidal Prevalence in Diabetics and Its Species Identification. Int. J. Appl. Basic Med. Res. 2019, 9, 49–54. [Google Scholar] [CrossRef]
- Jhugroo, C.; Divakar, D.D.; Jhugroo, P.; Al-Amri, S.A.; Alahmari, A.D.; Vijaykumar, S.; Parine, N.R. Characterization of Oral Mucosa Lesions and Prevalence of Yeasts in Diabetic Patients: A Comparative Study. Microb. Pathog. 2019, 126, 363–367. [Google Scholar] [CrossRef] [PubMed]
- Patel, M. Oral Cavity and Candida albicans: Colonisation to the Development of Infection. Pathogens 2022, 11, 335. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, C.F.; Rodrigues, M.E.; Henriques, M. Candida sp. Infections in Patients with Diabetes Mellitus. J. Clin. Med. 2019, 8, 76. [Google Scholar] [CrossRef]
- Ikebe, K.; Morii, K.; Matsuda, K.; Hata, K.; Nokubi, T. Association of Candidal Activity with Denture Use and Salivary Flow in Symptom-Free Adults Over 60 Years. J. Oral Rehabil. 2006, 33, 36–42. [Google Scholar] [CrossRef]
- Drummond, R.A.; Desai, J.V.; Ricotta, E.E.; Swamydas, M.; Deming, C.; Conlan, S.; Quinones, M.; Matei-Rascu, V.; Sherif, L.; Lecky, D.; et al. Long-Term Antibiotic Exposure Promotes Mortality after Systemic Fungal Infection by Driving Lymphocyte Dysfunction and Systemic Escape of Commensal Bacteria. Cell Host Microbe 2022, 30, 1020–1033. [Google Scholar] [CrossRef]
- Pappas, P.G.; Alexander, B.D.; Andes, D.; Hadley, S.; Kauffman, C.A.; Freifeld, A.; Anaissie, E.J.; Brumble, L.M.; Herwaldt, L.; Ito, J.; et al. Invasive Fungal Infections among Organ Transplant Recipients: Results of the Transplant-Associated Infection Surveillance Network (TRANSNET). Clin. Infect. Dis. 2010, 50, 1101–1111. [Google Scholar] [CrossRef]
- Senoner, T.; Breitkopf, R.; Treml, B.; Rajsic, S. Invasive Fungal Infections after Liver Transplantation. J. Clin. Med. 2023, 12, 3238. [Google Scholar] [CrossRef] [PubMed]
- Lalla, R.V.; Latortue, M.C.; Hong, C.H.; Ariyawardana, A.; D’Amato-Palumbo, S.; Fischer, D.J.; Martof, A.; Nicolatou-Galitis, O.; Patton, L.L.; Elting, L.S.; et al. A Systematic Review of Oral Fungal Infections in Patients Receiving Cancer Therapy. Support. Care Cancer 2010, 18, 985–992. [Google Scholar] [CrossRef]
- Pavlova, A.; Sharafutdinov, I. Recognition of Candida albicans and Role of Innate Type 17 Immunity in Oral Candidiasis. Microorganisms 2020, 8, 1340. [Google Scholar] [CrossRef] [PubMed]
- Mühlemann, K.; Wenger, C.; Zenhäusern, R.; Täuber, M.G. Risk Factors for Invasive Aspergillosis in Neutropenic Patients with Hematologic Malignancies. Leukemia 2005, 19, 545–550. [Google Scholar] [CrossRef] [PubMed]
- Mun, M.; Yap, T.; Alnuaimi, A.D.; Adams, G.G.; McCullough, M.J. Oral Candidal Carriage in Asymptomatic Patients. Aust. Dent. J. 2016, 61, 190–195. [Google Scholar] [CrossRef]
- Loster, J.E.; Wieczorek, A.; Loster, B.W. Correlation between Age and Gender in Candida Species Infections of Complete Denture Wearers: A Retrospective Analysis. Clin. Interv. Aging 2016, 11, 1707–1714. [Google Scholar] [CrossRef]
- Mareković, I.; Pleško, S.; Rezo Vranješ, V.; Herljević, Z.; Kuliš, T.; Jandrlić, M. Epidemiology of Candidemia: Three-Year Results from a Croatian Tertiary Care Hospital. J. Fungi 2021, 7, 267. [Google Scholar] [CrossRef] [PubMed]
- Lagunes, L.; Rello, J. Invasive Candidiasis: From Mycobiome to Infection, Therapy, and Prevention. Eur. J. Clin. Microbiol. Infect. Dis. 2016, 35, 1221–1226. [Google Scholar] [CrossRef] [PubMed]
- Ryniewicz, J.; Orczykowska, M.; Gronkiewicz, K.; Pihut, M. Assessment of Oral Hygiene in Patients Using Fixed and Removable Dentures Treated at the University Dental Clinic in Krakow. Int. J. Environ. Res. Public Health 2021, 18, 11986. [Google Scholar] [CrossRef] [PubMed]
- Holmstrup, P.; Axéll, T. Classification and Clinical Manifestations of Oral Yeast Infections. Acta Odontol. Scand. 1990, 48, 57–59. [Google Scholar] [CrossRef]

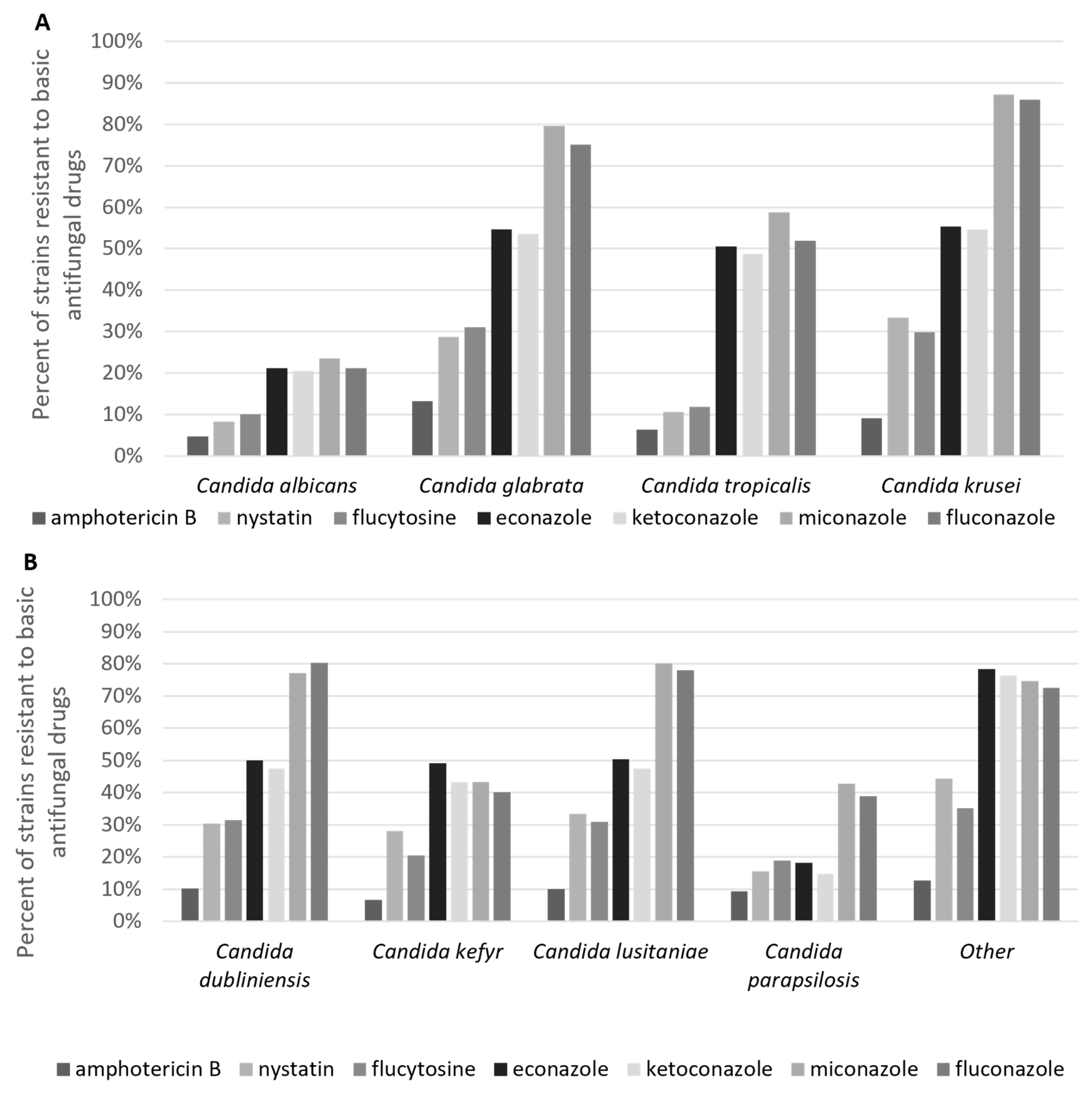
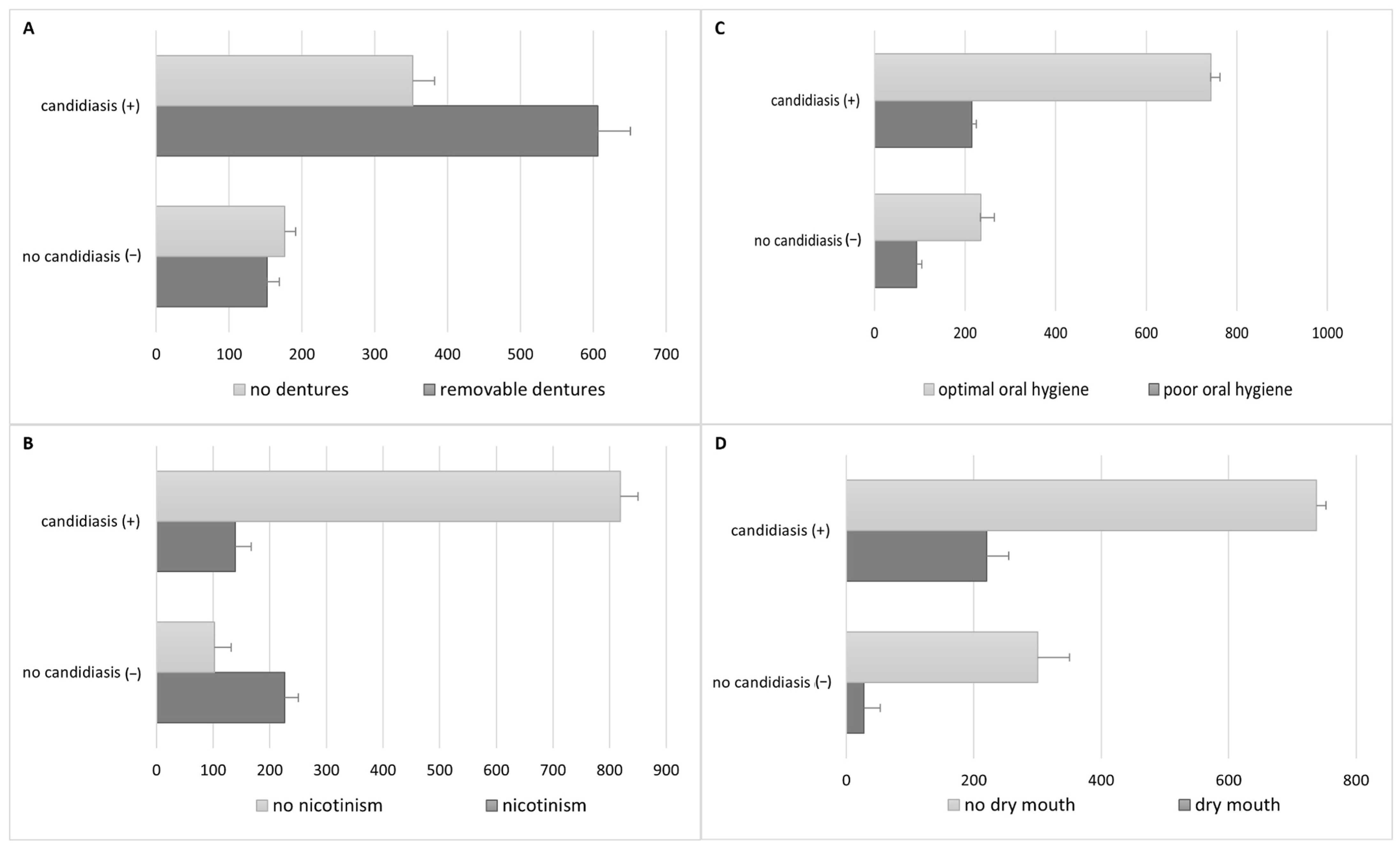
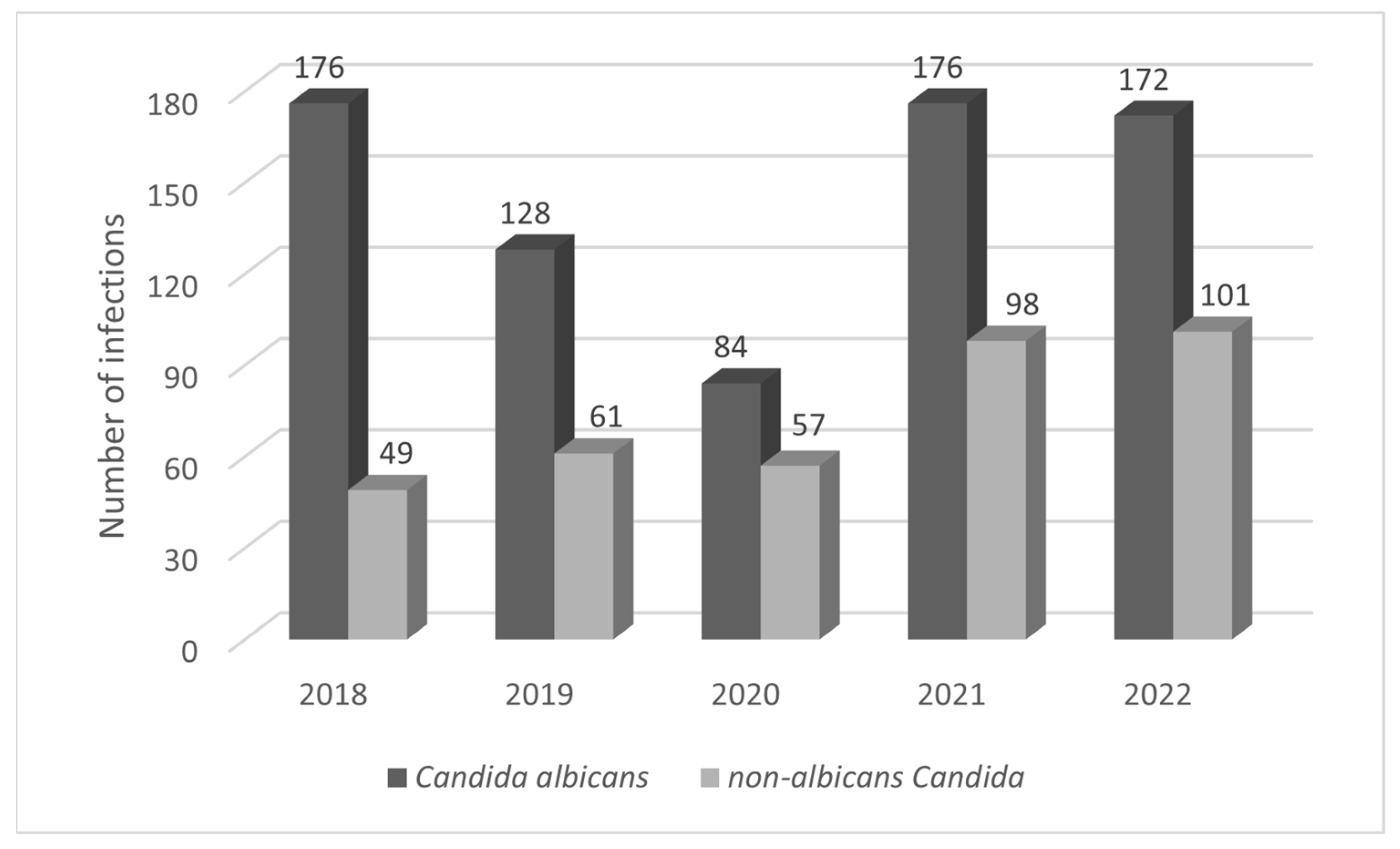
| Strain | Amount | % |
|---|---|---|
| Candida albicans | 736 | 66.79 |
| Non-albicans Candida | 366 | 33.21 |
| Candida glabrata | 108 | 9.80 |
| Candida tropicalis | 80 | 7.26 |
| Candida krusei | 49 | 4.45 |
| Candida dubliniensis | 44 | 3.99 |
| Candida kefyr | 41 | 3.72 |
| Candida lusitaniae | 19 | 1.72 |
| Candida parapsilosis | 16 | 1.45 |
| Candida spp. (other rare strains) | 9 | 0.82 |
| Total | 1102 | 100.00 |
| Local Predisposing Factor | Total | Female | Male |
|---|---|---|---|
| Removable prosthesis | 758 | 450 | 308 |
| Nicotinism | 365 | 201 | 164 |
| Poor oral hygiene | 308 | 180 | 128 |
| Dry mouth | 248 | 164 | 84 |
| 24/7 use of removable dentures | 170 | 122 | 48 |
| General Predisposing Factor | Total | Female | Male |
|---|---|---|---|
| Decompensated diabetes | 172 | 107 | 65 |
| Systemic glucocorticosteroid therapy/inhaled steroid therapy | 82 | 53 | 29 |
| Antibiotic therapy in the last 6 months | 59 | 39 | 20 |
| Systemic immunosuppressive therapy | 54 | 38 | 16 |
| Radiotherapy to the head and neck area | 53 | 28 | 25 |
| Systemic chemotherapy | 35 | 18 | 17 |
| AIDS/HIV | 8 | 4 | 4 |
| Other diseases and conditions with disease deficits (neutropenia) | 5 | 2 | 3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tkaczyk, M.; Kuśka-Kielbratowska, A.; Fiegler-Rudol, J.; Niemczyk, W.; Mertas, A.; Skaba, D.; Wiench, R. The Prevalence and Drug Susceptibility of Candida Species and an Analysis of Risk Factors for Oral Candidiasis—A Retrospective Study. Antibiotics 2025, 14, 876. https://doi.org/10.3390/antibiotics14090876
Tkaczyk M, Kuśka-Kielbratowska A, Fiegler-Rudol J, Niemczyk W, Mertas A, Skaba D, Wiench R. The Prevalence and Drug Susceptibility of Candida Species and an Analysis of Risk Factors for Oral Candidiasis—A Retrospective Study. Antibiotics. 2025; 14(9):876. https://doi.org/10.3390/antibiotics14090876
Chicago/Turabian StyleTkaczyk, Marcin, Anna Kuśka-Kielbratowska, Jakub Fiegler-Rudol, Wojciech Niemczyk, Anna Mertas, Dariusz Skaba, and Rafał Wiench. 2025. "The Prevalence and Drug Susceptibility of Candida Species and an Analysis of Risk Factors for Oral Candidiasis—A Retrospective Study" Antibiotics 14, no. 9: 876. https://doi.org/10.3390/antibiotics14090876
APA StyleTkaczyk, M., Kuśka-Kielbratowska, A., Fiegler-Rudol, J., Niemczyk, W., Mertas, A., Skaba, D., & Wiench, R. (2025). The Prevalence and Drug Susceptibility of Candida Species and an Analysis of Risk Factors for Oral Candidiasis—A Retrospective Study. Antibiotics, 14(9), 876. https://doi.org/10.3390/antibiotics14090876






