Trends in Antituberculosis Drug Resistance and Associated Factors: A 31-Year Observational Study at a Tertiary Hospital in Barcelona
Abstract
1. Introduction
2. Results
2.1. Global TB Cases Distribution by Period
2.2. TB Cases Demographic Data
2.3. 1st Line TB Drug Resistance Data
2.4. 2nd Line TB Drug Resistance Data
3. Discussion
4. Materials and Methods
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| TB | Tuberculosis |
| MDR | Multidrug-resistant |
| XDR | Extensively drug-resistant |
| SEPAR | Spanish society of pulmonary diseases |
| SEIMC | Spanish society of infectious diseases and clinical microbiology |
| IQR | Interquartile range |
| E | Ethambutol |
| H | Isoniazid |
| Z | Pyrazynamide |
| R | Rifampicin |
| S | Streptomycin |
| PR | Prevalence ratio |
| aPR | Adjusted prevalence ratio |
| CI | Confidence interval |
| NS | Statistical non-significant |
| HCB | Hospital Clínic of Barcelona |
| DST | Drug susceptibility test |
| FLDST | First-line Drug Susceptibility Testing |
| SLDST | Second-line Drug Susceptibility Testing |
| OR | Odds ratios |
| ORa | Adjusted odds ratios |
| CI | Confidence intervals |
References
- World Health Organization. Global Tuberculosis Report 2021; WHO: Geneva, Switzerland, 2021. [Google Scholar]
- World Health Organization. Global Tuberculosis Report 2024; WHO: Geneva, Switzerland, 2024. [Google Scholar]
- Rodrigo, T.; Millet, J.P.; Bravo, M.; Tabernero, E.M.; Caylà, J.A.; Rincón, I.V.; Pla, E.D.; Remacha, M.C.M.; Ibáñez, A.F.; Durán, J.G.; et al. Evolution of Tuberculosis in Spain: Will the objectives of the End Tuberculosis Strategy be Achieved in Western European Countries? Enferm. Infecc. Microbiol. Clin. 2025, in press. [Google Scholar] [CrossRef]
- Alcaide, F.; Santín, M. Multidrug-resistant tuberculosis. Enferm. Infecc. Microbiol. Clin. 2008, 26 (Suppl. S13), 54–60. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Montalva, A.; Caminero, J.A.; Guna, M.R.; Sanz, T.R.; Rabuñal, R.; Millet, J.P.; Gullón-Blanco, J.A.; Anibarro, L.; Perez-Mendoza, G.; Medina, J.F.; et al. Clinical Practice Guidelines on the Management of Resistant Tuberculosis of the Spanish Society of Pulmonology and Thoracic Surgery (SEPAR) and the Spanish Society of Infectious Diseases and Clinical Microbiology (SEIMC). Arch. Bronconeumol. 2024, 60, 759–767. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. WHO Consolidated Operational Handbook on Tuberculosis. Module 4: Treatment and Care; World Health Organization: Geneva, Switzerland, 2025. [Google Scholar]
- Martín-Casabona, N.; Alcaide, F.; Coll, P.; González, J.; José, M.; Salvadó, M.; Caylà, J.A.; Grupo de Trabajo sobre Resistencias en Tuberculosis. Drug resistance of Mycobacterium tuberculosis. Multicenter study in the Barcelona area. Med. Clin. 2000, 115, 493–498. [Google Scholar] [CrossRef]
- Royo, M.M.; Morán, F.G.; Muñoz, R.M.; Serrano, F.P.; Alfaro, P.P.; Pena, A.A.; Benito, M.A. Evolution of resistance to Mycobacterium tuberculosis in the province of Castellón. Years 1992–1998. Arch. Bronchopneumol. 2000, 36, 551–556. [Google Scholar] [CrossRef] [PubMed]
- Perez del Molino Bernal, M.L.; Túñez, V.; Cruz-Ferro, E.; Fernández-Villar, A.; Vázquez-Gallardo, R.; Díaz-Cabanela, D.; Anibarro, L.; Galego de Estudio de Resistencias Grupo. Study of Mycobacterium tuberculosis drug resistance in the region of Galicia, Spain. Int. J. Tuberc. Lung Dis. 2005, 9, 1230–1235. [Google Scholar] [PubMed]
- Alba Alvarez, L.M.; Garcia Garcia, J.M.; Pérez Hernández, M.D.; Martínez González, S.; Palacios Gutiérrez, J.J. Usefulness of phenotypic and genotypic methods in the study of Mycobacterium tuberculosis resistance to first-line anti-tuberculous drugs. Arch. Bronchopneumol. 2017, 53, 192–198. [Google Scholar] [CrossRef]
- Blanquer, R.; Rodrigo, T.; Casals, M.; Manzano, J.R.; García-García, J.M.; Calpe, J.L.; Valencia, E.; Pascual, T.; Mir, I.; Jiménez, M.Á.; et al. Resistance to first-line antituberculous drugs in Spain during 2010–2011. RETUBES Studio. Arch. Bronconeumol. 2015, 51, 24–30. [Google Scholar] [CrossRef]
- Gegia, M.; Winters, N.; Benedetti, A.; van Soolingen, D.; Menzies, D. Treatment of isoniazid-resistant tuberculosis with first-line drugs: A systematic review and meta-analysis. Lancet Infect. Dis. 2017, 17, 223–234. [Google Scholar] [CrossRef]
- Guenther, G.; Van Leth, F.; Alexandru, S.; Altet, N.; Avsar, K.; Bang, D.; Barbuta, R.; Bothamley, G.; Ciobanu, A.; Crudu, V. Multidrug-Resistant Tuberculosis in Europe, 2010–2011. Emerg. Infect Dis. 2015, 21, 409–416. [Google Scholar] [CrossRef] [PubMed]
- González-García, A.; Fortún, J.; Navas, E.E.; Martín-Dávila, P.; Tato, M.; Gómez-Mampaso, E.; Moreno, S. The changing epidemiology of tuberculosis in a Spanish tertiary hospital (1995–2013). Medicine 2017, 96, e7219. [Google Scholar] [CrossRef]
- Calderón Hernáiz, R.; Ruiz Giardín, J.M.; Cabello Clotet, N.; Canora Lebrato, J. Tuberculosis and immigration in an area of southwest Madrid. Int. J. Tuberc. Lung Dis. 2016, 20, 530–535. [Google Scholar] [CrossRef] [PubMed]
- Millet, J.P.; Orcau, A.; López-Muley, C.; Molero, J.; Artigas, A.; Avellanés, I.; Clos, R.; Collado, L.; de Andrés, A.; Forns, M.L.; et al. La Tuberculosi A Barcelona. Informe 2022; Agència de Salut Pública de Barcelona: Barcelona, Spain, 2024; Available online: https://www.aspb.cat/wp-content/uploads/2020/10/ASPB-Tuberculosi-Barcelona-2022-cast_241106.pdf (accessed on 15 March 2025).
- Caylà, J.A.; Orcau, A. Control of tuberculosis in large cities in developed countries: An organizational problem. BMC Med. 2011, 9, 127. [Google Scholar] [CrossRef]
- Minion, J.; Gallant, V.; Wolfe, J.; Jamieson, F.; Long, R. Multidrug and Extensively Drug-resistant Tuberculosis in Canada 1997–2008: Demographic and Disease Characteristics. PLoS ONE 2013, 8, e53466. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Faustini, A.; Hall, A.J.; Perucci, C.A. Risk factors for multidrug resistant tuberculosis in Europe: A systematic review. Thorax 2006, 61, 158–163. [Google Scholar] [CrossRef]
- Suchindran, S.; Brouwer, E.S.; Van Rie, A. Is HIV infection a risk factor for multi-drug resistant tuberculosis? A systematic review. PLoS ONE 2009, 4, e5561. [Google Scholar] [CrossRef]
- World Health Organization. Technical Manual for Drug Susceptibility Testing of Medicines Used in the Treatment of Tuberculosis; WHO: Geneva, Switzerland, 2018. [Google Scholar]
- Wang, W.; Zhou, H.; Cai, L.; Yang, T. Association between the rifampicin resistance mutations and rifabutin susceptibility in Mycobacterium tuberculosis: A meta-analysis. J. Glob. Antimicrob. Resist. 2025, 40, 53–61. [Google Scholar] [CrossRef]
- Brossier, F.; Veziris, N.; Truffot-Pernot, C.; Jarlier, V.; Sougakoff, W. Molecular investigation of resistance to the antituberculous drug ethionamide in multidrug-resistant clinical isolates of Mycobacterium tuberculosis. Antimicrob. Agents Chemother. 2011, 55, 355–360. [Google Scholar] [CrossRef] [PubMed]
- Sarin, R.; Bhalla, M.; Kumar, G.; Singh, A.; Myneedu, V.P.; Singhal, R. Correlation of inhA mutations and ethionamide susceptibility: Experience from national reference center for tuberculosis. Lung India 2021, 38, 520–523. [Google Scholar] [CrossRef]
- Chen, Y.; Yuan, Z.; Shen, X.; Wu, J.; Wu, Z.; Xu, B. Resistance to Second-Line Antituberculosis Drugs and Delay in Drug Susceptibility Testing among Multidrug-Resistant Tuberculosis Patients in Shanghai. Biomed. Res. Int. 2016, 2016, 2628913. [Google Scholar] [CrossRef]
- Mase, S.R.; Chorba, T. Treatment of Drug-Resistant Tuberculosis. Clin. Chest Med. 2019, 40, 775–795. [Google Scholar] [CrossRef] [PubMed]
- Garrigó, M.; Aragón, L.M.; Alcaide, F.; Borrell, S.; Cardenosa, E.; Galán, J.J.; Gonzalez-Martín, J.; Martin-Casabona, N.; Moreno, C.; Salvado, M.; et al. Multicenter laboratory evaluation of the MB/BacT Mycobacterium detection system and the BACTEC MGIT 960 system in comparison with the BACTEC 460TB system for susceptibility testing of Mycobacterium tuberculosis. J. Clin. Microbiol. 2007, 45, 1766–1770. [Google Scholar] [CrossRef] [PubMed]
- Rüsch-Gerdes, S.; Pfyffer, G.E.; Casal, M.; Chadwick, M.; Siddiqi, S. Multicenter laboratory validation of the BACTEC MGIT 960 technique for testing susceptibilities of Mycobacterium tuberculosis to classical second-line drugs and newer antimicrobials. J. Clin. Microbiol. 2006, 44, 688–692. [Google Scholar] [CrossRef] [PubMed]


| Total (N = 2448) | 1991–2000 (N = 1301) | 2001–2022 (N = 1147) | p-Value | ||||
|---|---|---|---|---|---|---|---|
| Gender (male), N (%) | 1715 | 70.1 | 956 | 73.4 | 759 | 66.1 | <0.001 1 |
| Age (median [IQR]), years | 38 | 29–55 | 36 | 29–53 | 40 | 29–58 | <0.001 2 |
| Age distribution, N (%) | |||||||
| <18 | 42 | 1.7 | 20 | 1.5 | 22 | 1.9 | 0.007 1 |
| 18–29 | 610 | 24.9 | 339 | 26.1 | 271 | 23.6 | |
| 30–49 | 1055 | 43.1 | 586 | 45 | 469 | 40.9 | |
| >49 | 741 | 30.3 | 356 | 27.4 | 385 | 33.6 | |
| Origin, N (%) | |||||||
| Spain | 1765 | 72.1 | 1130 | 86.9 | 635 | 55.4 | <0.001 1 |
| Rest of Europe | 64 | 2.6 | 11 | 0.8 | 53 | 4.6 | |
| Africa–Maghreb | 136 | 5.6 | 46 | 3.5 | 90 | 7.8 | |
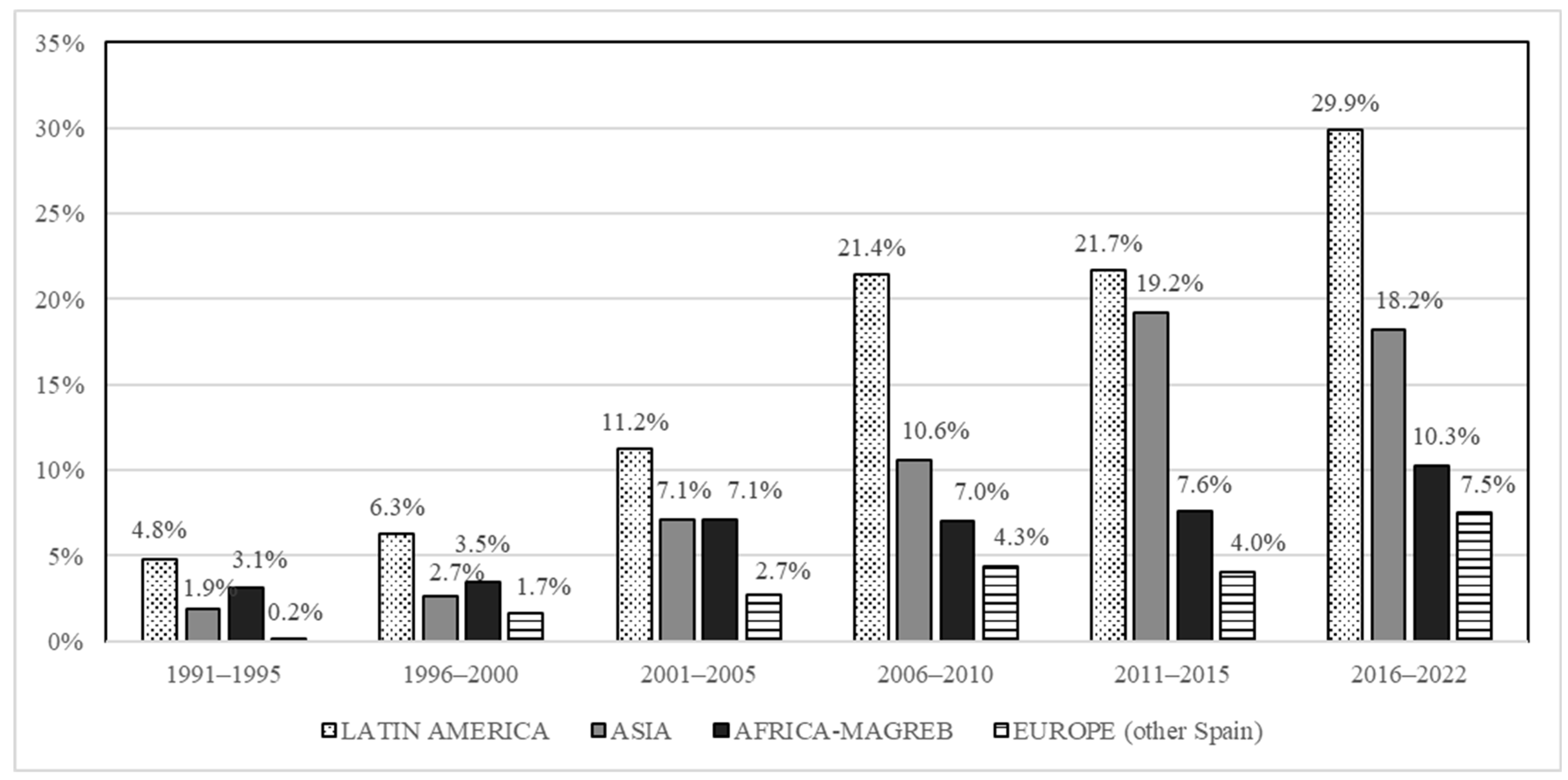
| Latin America | 306 | 12.5 | 77 | 5.9 | 229 | 20 | |
| Asia | 177 | 7.2 | 37 | 2.8 | 140 | 11.2 | |
| Social situation, N (%) | |||||||
| Homeless people | 190 | 7.8 | 87 | 6.7 | 103 | 9 | <0.021 1 |
| Prison | 257 | 11.4 | 223 | 17.1 | 34 | 3 | <0.001 1 |
| HIV infection, N (%) | 605 | 24.7 | 450 | 34.6 | 155 | 13.5 | <0.001 1 |
| Other clinical disorders, N (%) | |||||||
| Kidney failure | 69 | 2.8 | 34 | 2.6 | 35 | 3.1 | 0.29 1 |
| Immunosuppression | 108 | 4.4 | 29 | 2.2 | 79 | 6.9 | <0.001 1 |
| Diabetes | 148 | 6.0 | 66 | 5.1 | 82 | 7.1 | 0.020 1 |
| Alcohol consuming | 426 | 17.4 | 259 | 19.9 | 167 | 14.6 | <0.001 1 |
| Tuberculosis clinical location, N (%) | 0.883 1 | ||||||
| Pulmonary | 1960 | 80.1 | 1045 | 80.3 | 915 | 79.8 | 0.38 1 |
| Extra-pulmonary | 488 | 19.9 | 256 | 19.7 | 232 | 20.2 | 0.38 1 |
| Bacteriology, N (%) | <0.001 | ||||||
| Culture +/smear − | 1194 | 48.8 | 556 | 42.7 | 638 | 55.6 | |
| Culture +/smear + | 1254 | 51.2 | 745 | 57.3 | 509 | 44.4 | |
| New tuberculosis cases, N (%) | 2189 | 89.4 | 1110 | 85.3 | 1079 | 94.1 | <0.001 1 |
| Non-susceptible tuberculosis, N (%) | 299 | 12.2 | 112 | 8.6 | 187 | 16.3 | <0.001 1 |
| Outcome, N (%) | <0.001 1 | ||||||
| Treatment completed | 1537 | 62.8 | 740 | 56.9 | 797 | 69.5 | |
| Dead | 279 | 11.4 | 182 | 14 | 97 | 8.5 | |
| Off Barcelona 3 | 425 | 17.4 | 311 | 23.9 | 114 | 9.9 | |
| Lost to follow-up | 196 | 8 | 65 | 5 | 121 | 11.4 | |
| Another 4 | 11 | 0.4 | 3 | 0.2 | 8 | 0.7 | |
| Total (N = 2448) | 1991–2000 (N = 1301) | 2001–2022 (N = 1147) | p-Value | ||||
|---|---|---|---|---|---|---|---|
| N | % | N | % | N | % | ||
| New tuberculosis cases | 2189 | 89.4 | 1110 | 85.3 | 1079 | 94.1 | |
| Treated tuberculosis cases | 259 | 10.6 | 191 | 14.7 | 68 | 5.9 | <0.001 |
| Total cases | |||||||
| Susceptible | 2149 | 87.8 | 1189 | 91.4 | 960 | 83.7 | |
| Resistant | 299 | 12.2 | 112 | 8.6 | 187 | 16.3 | <0.001 |
| New cases | |||||||
| Susceptible | 1944 | 88.8 | 1035 | 93.2 | 909 | 84.2 | |
| Resistant | 245 | 11.2 | 75 | 6.8 | 170 | 15.8 | <0.001 |
| Treated cases | |||||||
| Susceptible | 205 | 79.2 | 154 | 80.6 | 51 | 75 | |
| Resistant | 54 | 20.8 | 37 | 19.4 | 17 | 25 | 0.42 |
| Resistance 1 | 414 | 16.9 | 168 | 12.9 | 246 | 21.4 | <0.001 |
| H | 190 | 7.8 | 94 | 7.2 | 96 | 8.4 | |
| R | 59 | 2.4 | 36 | 2.8 | 23 | 2 | |
| E | 24 | 1 | 7 | 0.5 | 17 | 1.5 | |
| Z | 58 | 2.4 | 22 | 1.7 | 36 | 3.1 | |
| S | 83 | 3.4 | 9 | 0.7 | 74 | 6.5 | |
| Monoresistance | 208 | 8.5 | 71 | 5.5 | 137 | 11.9 | <0.001 |
| H | 102 | 4.2 | 53 | 4.1 | 49 | 4.3 | |
| R | 3 | 0.1 | 0 | - | 3 | 0.1 | |
| E | 8 | 0.3 | 0 | - | 8 | 0.7 | |
| Z | 29 | 1.2 | 8 | 0.6 | 21 | 1.8 | |
| S | 66 | 2.7 | 10 | 0.8 | 56 | 4.9 | |
| Multiresistance | 56 | 2.3 | 36 | 2.8 | 20 | 1.7 | 0.498 |
| H + R | 23 | 0.9 | 16 | 1.2 | 7 | 0.6 | |
| H + R + E | 2 | 0.1 | 2 | 0.2 | 0 | - | |
| H + R + Z | 9 | 0.4 | 8 | 0.6 | 1 | 0.1 | |
| H + R + S | 6 | 0.2 | 4 | 0.3 | 2 | 0.2 | |
| H + R + E + Z | 3 | 0.1 | 2 | 0.2 | 1 | 0.1 | |
| H + R + E + S | 3 | 0.1 | 1 | 0.1 | 2 | 0.2 | |
| H + R + Z + S | 5 | 0.2 | 2 | 0.2 | 3 | 0.3 | |
| H + R + E + Z + S | 5 | 0.2 | 0 | 0.0 | 5 | 0.4 | |
| Polyresistance | 35 | 1.4 | 5 | 0.4 | 30 | 2.6 | 0.201 |
| H + E | 1 | 0.1 | 1 | 0.1 | 0 | 0.0 | |
| H + Z | 3 | 0.1 | 1 | 0.1 | 2 | 0.2 | |
| H + S | 25 | 1.0 | 3 | 0.2 | 22 | 1.9 | |
| H + E + S | 2 | 0.1 | 0 | - | 2 | 0.2 | |
| H + Z + S | 1 | 0.1 | 0 | - | 1 | 0.1 | |
| S + Z | 3 | 0.1 | 0 | - | 3 | 0.1 | |
| Any Kind of Resistance 1 | Multiresistance 1 | |||
|---|---|---|---|---|
| Bivariate | Multivariate | Bivariate | Multivariate | |
| OR (95% CI) | aOR (95% CI) | OR (95% CI) | aOR (95% CI) | |
| Period | ||||
| 1990–2000 | 1 | − | 1 | 1 |
| 2001–2022 | 1.63 (1.28–2.09) | NS | 0.53 (0.30–0.94) | 0.40 (0.23–0.69) |
| Gender | ||||
| Man | 1 | − | 1 | − |
| Woman | 1.15 (0.89–1.47) | NS | 1.34 (0.99–1.81) | NS |
| Age (years) | ||||
| 1.00 (0.99–1.01) | NS | 1.00 (0.99–1.01) | NS | |
| Origin | ||||
| Spain | 1 | 1 | 1 | 1 |
| Rest of Europe | 3.45 (2.48–4.80) | 3.46 (2.42–4.95) | 3.00 (1.89–4.75) | 3.14 (1.94–5.07) |
| Africa–Maghreb | 3.33 (2.18–5.09) | 3.45 (2.28–5.21) | 3.70 (2.22–6.17) | 3.76 (2.25–6.29) |
| Latin America | 2.59 (1.90–3.51) | 2.66 (1.92–4.59) | 2.72 (1.87–3.96) | 2.69 (1.82–3.97) |
| Asia | 2.81 (1.82–4.33) | 2.97 (2.51–7.92) | 2.91 (1.69–4.98) | 2.89 (1.68–4.98) |
| Tuberculosis case | ||||
| New | 1 | 1 | 1 | 1 |
| Previously treated | 2.09 (1.59–2.75) | 2.56 (1.96–3.35) | 1.41 (0.94–2.13) | 1.79 (1.19–2.68) |
| Location | ||||
| Pulmonary | 1 | − | 1 | − |
| Extra-pulmonary | 0.94 (0.70–1.27) | NS | 1.16 (0.82–1.64) | NS |
| Bacteriology | ||||
| Culture +/smear − | 1 | − | 1 | − |
| Culture +/smear + | 1.05 (0.83–1.33) | NS | 0.91 (0.68–1.22) | NS |
| HIV infection | ||||
| Negative | 1 | − | − | |
| Positive | 0.85 (0.64–1.13) | NS | 0.77 (0.54–1.10) | NS |
| Unknown | 0.74 (0.51–1.06) | NS | 0.65 (0.41–1.04) | NS |
| Homeless people | ||||
| Yes | 0.79 (0.46–1.34) | NS | 0.80 (0.42–1.54) | NS |
| No | 1 | - | 1 | - |
| Prison | ||||
| Yes | 0.55 (0.34–0.90) | 0.57 (0.35–0.94) | 0.30 (0.13–0.67) | 0.33 (0.15–0.74) |
| No | 1 | 1 | 1 | 1 |
| Alcohol consuming | ||||
| Yes | 0.72 (0.51–1.02) | NS | 0.73 (0.48–1.12) | NS |
| No | 1 | - | 1 | - |
| Immunosuppression | ||||
| Yes | 0.77 (0.60–0.99) | NS | 0.77 (0.56–1.04) | NS |
| No | 1 | - | 1 | - |
| Distribution According First-Line Drug Resistance | ||||||||
|---|---|---|---|---|---|---|---|---|
| Second-Line Drug | Monoresistant | Polyresistant | Multiresistant | |||||
| Tested 1 N | Resistant 1 (%) | R/C 2 | % 2 | R/C 2 | % 2 | R/C 2 | % 2 | |
| Rifabutin | 99 | 14 (14.1) | 1/66 | 1.5 | 0/20 | 0 | 13/13 | 100 |
| Ethionamide | 200 | 49 (24.5) | 21/124 | 16.9 | 10/33 | 30.3 | 18/43 | 41.9 |
| Ofloxacin | 202 | 8 (4) | 5/125 | 4 | 0/33 | 0 | 3/44 | 6.8 |
| Amikacin | 202 | 2 (1) | 0/125 | 0 | 0/33 | 0 | 2/44 | 4.5 |
| Capreomycin | 198 | 2 (1) | 1/123 | 0.8 | 0/33 | 0 | 1/42 | 2.4 |
| Cycloserine | 201 | 26 (12.9) | 18/125 | 14.4 | 0/33 | 0 | 8/43 | 18.6 |
| Kanamycin | 203 | 2 (1) | 0/125 | 0 | 0/33 | 0 | 2/45 | 4.4 |
| Linezolid | 57 | 0 (0) | 0/41 | 0 | 0/6 | 0 | 0/10 | 0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Espasa, M.; Pagán, B.; Fernández-Pittol, M.; Orcau, Á.; Tudó, G.; García, F.; Martínez, J.-A.; Soler, N.; Horvath-Ruiz, L.; San-Nicolás, L.; et al. Trends in Antituberculosis Drug Resistance and Associated Factors: A 31-Year Observational Study at a Tertiary Hospital in Barcelona. Antibiotics 2025, 14, 875. https://doi.org/10.3390/antibiotics14090875
Espasa M, Pagán B, Fernández-Pittol M, Orcau Á, Tudó G, García F, Martínez J-A, Soler N, Horvath-Ruiz L, San-Nicolás L, et al. Trends in Antituberculosis Drug Resistance and Associated Factors: A 31-Year Observational Study at a Tertiary Hospital in Barcelona. Antibiotics. 2025; 14(9):875. https://doi.org/10.3390/antibiotics14090875
Chicago/Turabian StyleEspasa, Mateu, Belén Pagán, Mariana Fernández-Pittol, Ángels Orcau, Griselda Tudó, Felipe García, Jose-Antonio Martínez, Néstor Soler, Laura Horvath-Ruiz, Lorena San-Nicolás, and et al. 2025. "Trends in Antituberculosis Drug Resistance and Associated Factors: A 31-Year Observational Study at a Tertiary Hospital in Barcelona" Antibiotics 14, no. 9: 875. https://doi.org/10.3390/antibiotics14090875
APA StyleEspasa, M., Pagán, B., Fernández-Pittol, M., Orcau, Á., Tudó, G., García, F., Martínez, J.-A., Soler, N., Horvath-Ruiz, L., San-Nicolás, L., Martínez, D., Casals-Pascual, C., Vila, J., Millet, J.-P., Caylà, J. A., & Gonzalez-Martin, J. (2025). Trends in Antituberculosis Drug Resistance and Associated Factors: A 31-Year Observational Study at a Tertiary Hospital in Barcelona. Antibiotics, 14(9), 875. https://doi.org/10.3390/antibiotics14090875









