Antibacterial and Antifungal Management in Relation to the Clinical Characteristics of Elderly Patients with Infective Endocarditis: A Retrospective Analysis
Abstract
1. Introduction
2. Results
2.1. General Characteristics of the Study Participants
2.2. Endocarditis Features
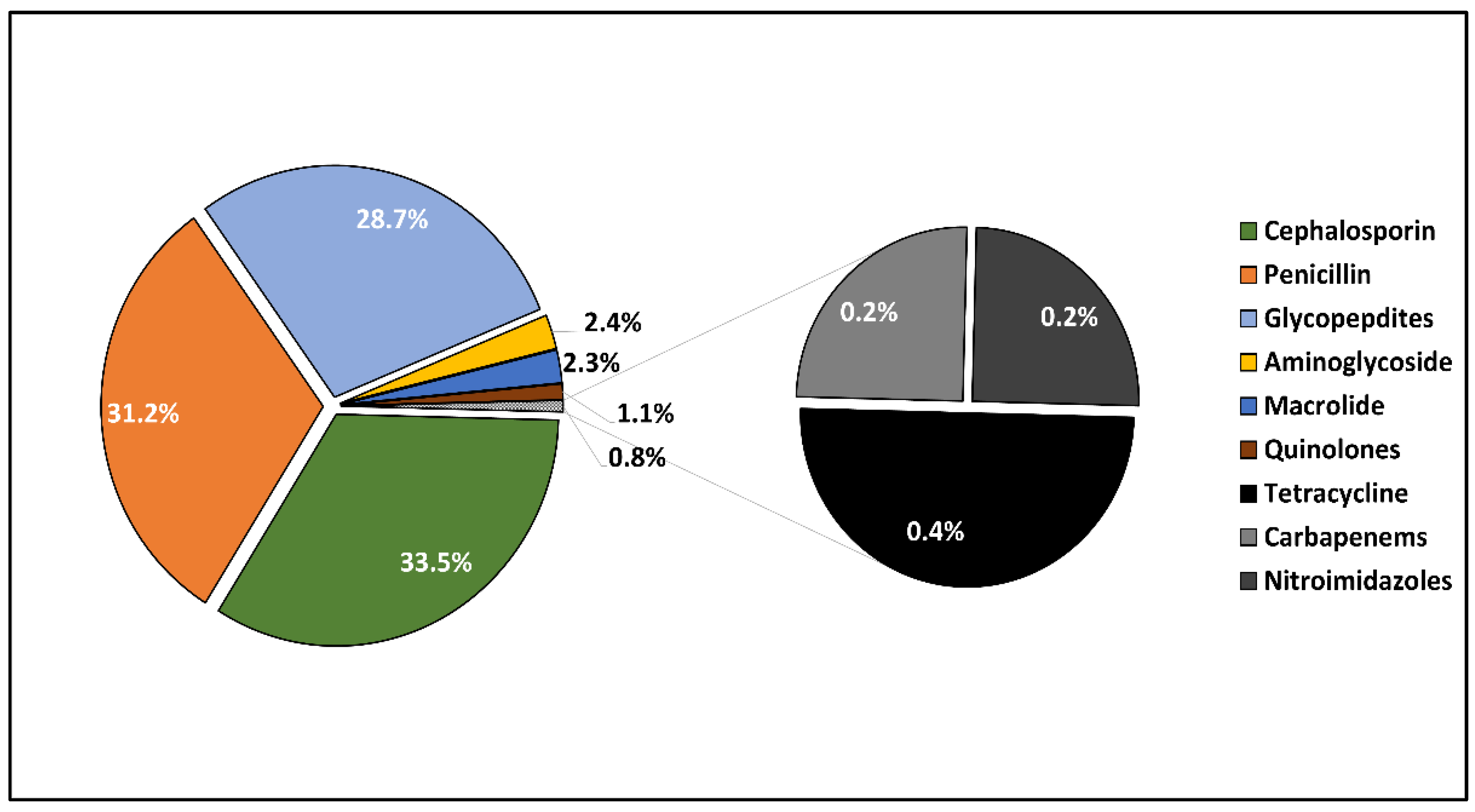
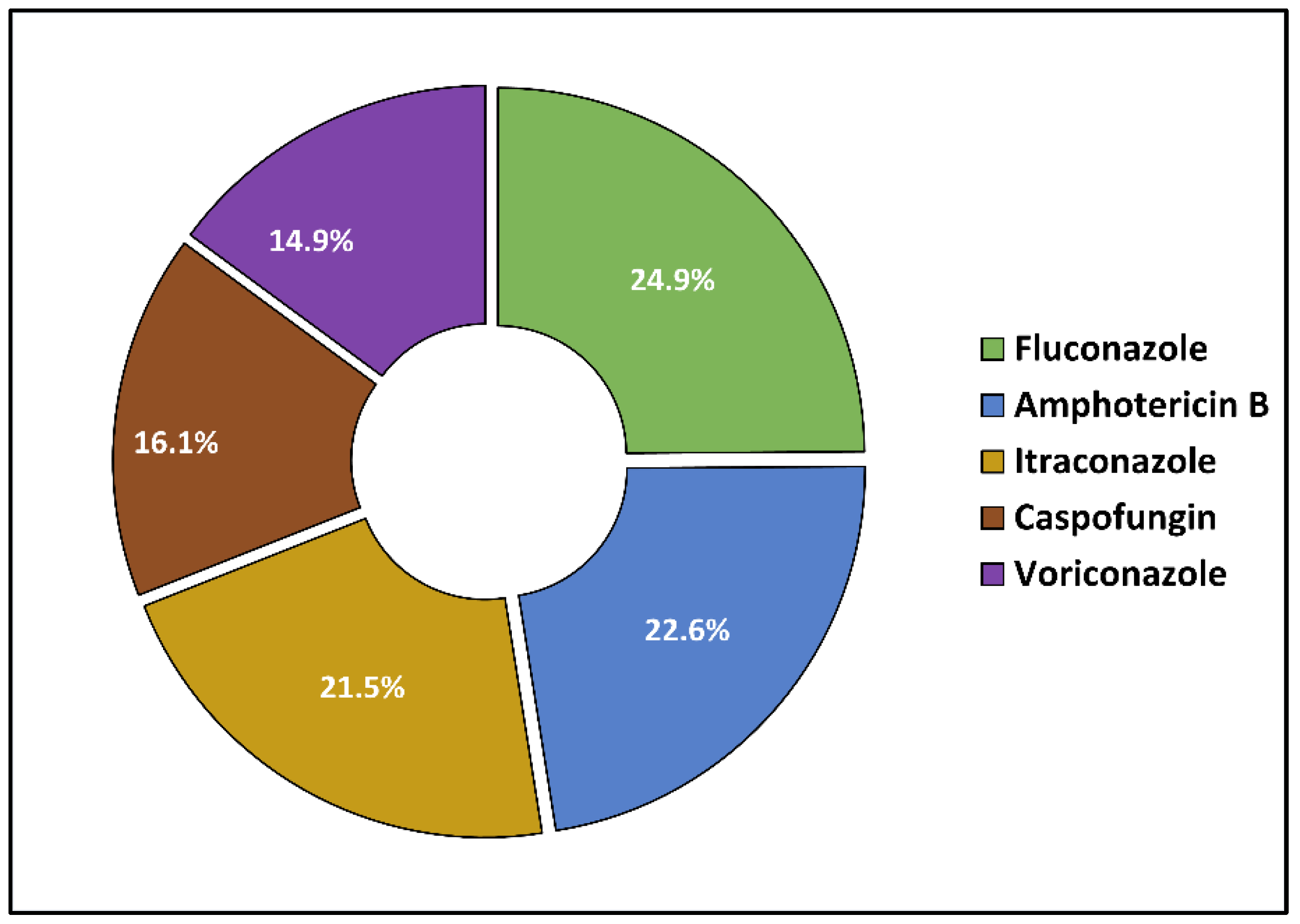
2.3. Microbal Identification and Antibacterial and Antifungal Management
2.4. Risk Factor Analysis
3. Discussion
4. Materials and Methods
4.1. Study Design and Ethical Considerations
4.2. Inclusion Criteria and Study Variables
4.3. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Prendki, V. Management of elderly patients with infective endocarditis. Clin. Microbiol. Infect. 2019, 25, 1169–1170. [Google Scholar] [CrossRef] [PubMed]
- United Nations Department of Economic and Social Affairs Population Division. World Population Ageing 2017 e Highlights (ST/ESA/SER.A/397). 2017. Available online: http://www.un.org/en/development/desa/population/publications/pdf/ageing/WPA2017_Highlights.pdf (accessed on 20 May 2022).
- El Chakhtoura, N.G.; Bonomo, R.A.; Jump, R.L.P. Influence of Aging and Environment on Presentation of Infection in Older Adults. Infect. Dis. Clin. N. Am. 2017, 31, 593–608. [Google Scholar] [CrossRef] [PubMed]
- Marincu, I.; Bratosin, F.; Vidican, I.; Bostanaru, A.C.; Frent, S.; Cerbu, B.; Turaiche, M.; Tirnea, L.; Timircan, M. Predictive Value of Comorbid Conditions for COVID-19 Mortality. J. Clin. Med. 2021, 10, 2652. [Google Scholar] [CrossRef] [PubMed]
- Solomon, D.H. Geriatric assessment: Methods for clinical decision making. JAMA 1988, 259, 2450–2452. [Google Scholar] [CrossRef] [PubMed]
- Keynan, Y.; Rubinstein, E. Pathophysiology of infective endocarditis. Curr. Infect. Dis. Rep. 2013, 15, 342–346. [Google Scholar] [CrossRef]
- Pant, S.; Patel, N.J.; Deshmukh, A.; Golwala, H.; Patel, N.; Badheka, A.; Hirsch, G.A.; Mehta, J.L. Trends in infective endocarditis incidence, microbiology, and valve replacement in the United States from 2000 to 2011. J. Am. Coll. Cardiol. 2015, 65, 2070–2076. [Google Scholar] [CrossRef]
- Ambrosioni, J.; Hernandez-Meneses, M.; Téllez, A.; Pericàs, J.; Falces, C.; Tolosana, J.M.; Vidal, B.; Almela, M.; Quintana, E.; Llopis, J.; et al. The changing epidemiology of infective endocarditis in the twenty-first century. Curr. Infect. Dis. Rep. 2017, 19, 21. [Google Scholar] [CrossRef]
- Dumache, R.; Enache, A.; Barbarii, L.; Constantinescu, C.; Pascalau, A.; Jinca, C.; Arghirescu, S. Chimerism Monitoring by Short Tandem Repeat (STR) Markers in Allogeneic Stem Cell Transplantation. Clin. Lab. 2018, 64, 1535–1543. [Google Scholar] [CrossRef]
- Citu, C.; Burlea, B.; Gorun, F.; Motoc, A.; Gorun, O.M.; Malita, D.; Ratiu, A.; Margan, R.; Grigoras, M.L.; Bratosin, F.; et al. Predictive Value of Blood Coagulation Parameters in Poor Outcomes in COVID-19 Patients: A Retrospective Observational Study in Romania. J. Clin. Med. 2022, 11, 2831. [Google Scholar] [CrossRef]
- Murdoch, D.R.; Corey, G.R.; Hoen, B.; Miró, J.M.; Fowler, V.G.; Bayer, A.S.; Karchmer, A.W.; Olaison, L.; Pappas, P.A.; Moreillon, P.; et al. Clinical presentation, etiology, and outcome of infective endocarditis in the 21st century: The International Collaboration on Endocarditis–Prospective Cohort Study. Arch. Intern. Med. 2009, 169, 463–473. [Google Scholar] [CrossRef]
- Citu, I.M.; Citu, C.; Gorun, F.; Neamtu, R.; Motoc, A.; Burlea, B.; Rosca, O.; Bratosin, F.; Hosin, S.; Manolescu, D.; et al. Using the NYHA Classification as Forecasting Tool for Hospital Readmission and Mortality in Heart Failure Patients with COVID-19. J. Clin. Med. 2022, 11, 1382. [Google Scholar] [CrossRef] [PubMed]
- Kreitmann, L.; Montaigne, D.; Launay, D.; Morell-Dubois, S.; Maillard, H.; Lambert, M.; Hachulla, E.; Sobanski, V. Clinical Characteristics and Outcome of Patients with Infective Endocarditis Diagnosed in a Department of Internal Medicine. J. Clin. Med. 2020, 9, 864. [Google Scholar] [CrossRef] [PubMed]
- Holland, T.L.; Baddour, L.M.; Bayer, A.S.; Hoen, B.; Miro, J.M.; Fowler, V.G. Infective endocarditis. Nat. Rev. Dis. Prim. 2016, 2, 16059. [Google Scholar] [CrossRef] [PubMed]
- Pilut, C.N.; Citu, C.; Gorun, F.; Bratosin, F.; Gorun, O.M.; Burlea, B.; Citu, I.M.; Grigoras, M.L.; Manolescu, D.; Gluhovschi, A. The Utility of Laboratory Parameters for Cardiac Inflammation in Heart Failure Patients Hospitalized with SARS-CoV-2 Infection. Diagnostics 2022, 12, 824. [Google Scholar] [CrossRef] [PubMed]
- Cabell, C.H.; Abrutyn, E. Progress toward a global understanding of infective endocarditis: Early lessons from the International Collaboration on Endocarditis investigation. Infect. Dis. Clin. N. Am. 2002, 16, 255–272. [Google Scholar] [CrossRef]
- Ngiam, J.N.; Liong, T.S.; Sim, M.Y.; Chew, N.W.S.; Sia, C.-H.; Chan, S.P.; Lim, T.W.; Yeo, T.-C.; Tambyah, P.A.; Loh, P.H.; et al. Risk Factors for Mortality in Cardiac Implantable Electronic Device (CIED) Infections: A Systematic Review and Meta-Analysis. J. Clin. Med. 2022, 11, 3063. [Google Scholar] [CrossRef]
- Bogdan, I.; Citu, C.; Bratosin, F.; Malita, D.; Romosan, I.; Gurban, C.V.; Bota, A.V.; Turaiche, M.; Bratu, M.L.; Pilut, C.N.; et al. The Impact of Multiplex PCR in Diagnosing and Managing Bacterial Infections in COVID-19 Patients Self-Medicated with Antibiotics. Antibiotics 2022, 11, 437. [Google Scholar] [CrossRef]
- Durante-Mangoni, E.; Bradley, S.; Selton-Suty, C.; Tripodi, M.F.; Barsic, B.; Bouza, E.; Cabell, C.H.; de Oliveira Ramos, A.I.; Fowler, V.; Hoen, B.; et al. Current Features of Infective Endocarditis in Elderly Patients: Results of the International Collaboration on Endocarditis Prospective Cohort Study. Arch. Intern. Med. 2008, 168, 2095–2103. [Google Scholar] [CrossRef]
- Nkomo, V.T.; Gardin, J.M.; Skelton, T.N.; Gottdiener, J.S.; Scott, C.G.; Enriquez-Sarano, M. Burden of valvular heart diseases: A population-based study. Lancet 2006, 368, 1005–1011. [Google Scholar] [CrossRef]
- Bea, C.; Vela, S.; García-Blas, S.; Perez-Rivera, J.-A.; Díez-Villanueva, P.; de Gracia, A.I.; Fuertes, E.; Oltra, M.R.; Ferrer, A.; Belmonte, A.; et al. Infective Endocarditis in the Elderly: Challenges and Strategies. J. Cardiovasc. Dev. Dis. 2022, 9, 192. [Google Scholar] [CrossRef]
- Gopalakrishnan, P.P.; Shukla, S.K.; Tak, T. Infective endocarditis: Rationale for revised guidelines for antibiotic prophylaxis. Clin. Med. Res. 2009, 7, 63–68. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Janszky, I.; Gémes, K.; Ahnve, S.; Asgeirsson, H.; Möller, J. Invasive Procedures Associated with the Development of Infective Endocarditis. J. Am. Coll. Cardiol. 2018, 71, 2744–2752. [Google Scholar] [CrossRef] [PubMed]
- Vahanian, A. The growing burden of infective endocarditis in the elderly. Eur. Heart J. 2003, 24, 1539–1540. [Google Scholar] [CrossRef]
- Gregoratos, G. Infective endocarditis in the elderly: Diagnosis and management. Am. J. Geriatr. Cardiol. 2003, 12, 183–189. [Google Scholar] [CrossRef] [PubMed]
- Salem, M.; Friedrich, C.; Saad, M.; Frank, D.; Salem, M.; Puehler, T.; Schoettler, J.; Schoeneich, F.; Cremer, J.; Haneya, A. Active Infective Native and Prosthetic Valve Endocarditis: Short- and Long-Term Outcomes of Patients after Surgical Treatment. J. Clin. Med. 2021, 10, 1868. [Google Scholar] [CrossRef]
- Khalil, H.; Soufi, S. Prosthetic Valve Endocarditis. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2022. Available online: https://www.ncbi.nlm.nih.gov/books/NBK567731/ (accessed on 20 May 2022).
- Evangelista, A.; Gonzalez-Alujas, M.T. Echocardiography in infective endocarditis. Heart 2004, 90, 614–617. [Google Scholar] [CrossRef]
- Sunder, S.; Grammatico-Guillon, L.; Lemaignen, A.; Lacasse, M.; Gaborit, C.; Boutoille, D.; Tattevin, P.; Denes, E.; Guimard, T.; Dupont, M.; et al. Incidence, characteristics, and mortality of infective endocarditis in France in 2011. PLoS ONE 2019, 14, e0223857. [Google Scholar] [CrossRef]
- Dhawan, V.K. Infective endocarditis in elderly patients. Clin. Infect. Dis. 2002, 34, 806–812. [Google Scholar] [CrossRef]
- Paul, G.; Ochs, L.; Hohmann, C.; Baldus, S.; Michels, G.; Meyer-Schwickerath, C.; Fätkenheuer, G.; Mader, N.; Wahlers, T.; Weber, C.; et al. Surgical Procedure Time and Mortality in Patients with Infective Endocarditis Caused by Staphylococcus aureus or Streptococcus Species. J. Clin. Med. 2022, 11, 2538. [Google Scholar] [CrossRef]
- Babeș, E.E.; Lucuța, D.A.; Petcheși, C.D.; Zaha, A.A.; Ilyes, C.; Jurca, A.D.; Vesa, C.M.; Zaha, D.C.; Babeș, V.V. Clinical Features and Outcome of Infective Endocarditis in a University Hospital in Romania. Medicina 2021, 57, 158. [Google Scholar] [CrossRef]
- Molnar, A.; Kovacs, E.; Rancea, R.; Mateas, F.; Beyer, R.; Sacui, D.; Trifan, C.; Turturica, S.; Manea, A. Long-Term Therapeutic Results After Surgically Treated Infective Endocarditis—A Single Centre Experience. Struct. Heart 2019, 3, 50. [Google Scholar] [CrossRef]
- Bin Abdulhak, A.A.; Baddour, L.M.; Erwin, P.J.; Hoen, B.; Chu, V.H.; Mensah, G.A.; Tleyjeh, I.M. Global and Regional Burden of Infective Endocarditis, 1990-2010: A Systematic Review of the Literature. Glob. Heart 2014, 9, 131–143. [Google Scholar] [CrossRef] [PubMed]
- Katzenellenbogen, J.M.; Ralph, A.P.; Wyber, R.; Carapetis, J.R. Rheumatic heart disease: Infectious disease origin, chronic care approach. BMC Health Serv. Res. 2017, 17, 793. [Google Scholar] [CrossRef] [PubMed]
- Rostagno, C. Heart valve disease in elderly. World J. Cardiol. 2019, 11, 71–83. [Google Scholar] [CrossRef] [PubMed]
- Lomas, J.M.; Martínez-Marcos, F.J.; Plata, A.; Ivanova, R.; Gálvez, J.; Ruiz, J.; Reguera, J.M.; Noureddine, M.; de la Torre, J.; de Alarcón, A. Healthcare-associated infective endocarditis: An undesirable effect of healthcare universalization. Clin. Microbiol. Infect. 2010, 16, 1683–1690. [Google Scholar] [CrossRef]
- Daly, C.G. Antibiotic prophylaxis for dental procedures. Aust. Prescr. 2017, 40, 184–188. [Google Scholar] [CrossRef]
- Math, R.S.; Sharma, G.; Kothari, S.S.; Kalaivani, M.; Saxena, A.; Kumar, A.S.; Bahl, V.K. Prospective study of infective endocarditis from a developing country. Am. Heart J. 2011, 162, 633–638. [Google Scholar] [CrossRef]
- Naber, C.K.; Erbel, R. Diagnosis of culture negative endocarditis: Novel strategies to prove the suspect guilty. Heart 2003, 89, 241–243. [Google Scholar] [CrossRef]
- Habib, G.; Erba, P.A.; Iung, B.; Donal, E.; Cosyns, B.; Laroche, C.; Popescu, B.A.; Prendergast, B.; Tornos, P.; Sadeghpour, A.; et al. EURO-ENDO Investigators. Clinical presentation, aeti-ology and outcome of infective endocarditis. Results of the ESC-EORP EURO-ENDO (European infective endocarditis) registry: A prospective cohort study. Eur. Heart J. 2019, 40, 3222–3232. [Google Scholar] [CrossRef]
- Sunil, M.; Hieu, H.Q.; Arjan Singh, R.S.; Ponnampalavanar, S.; Siew, K.S.W.; Loch, A. Evolving trends in infective endocarditis in a developing country: A consequence of medical progress? Ann. Clin. Microbiol. Antimicrob. 2019, 18, 43. [Google Scholar] [CrossRef]
- Ghosh, S.; Sahoo, R.; Nath, R.K.; Duggal, N.; Gadpayle, A.K. A Study of Clinical, Microbiological, and Echocardiographic Profile of Patients of Infective Endocarditis. Int. Sch. Res. Not. 2014, 2014, 340601. [Google Scholar] [CrossRef] [PubMed]
- Chambers, H.F.; Korzeniowski, O.M.; Sande, M.A. Staphylococcus aureus endocarditis: Clinical manifestations in addicts and nonaddicts. Medicine 1983, 62, 170–177. [Google Scholar] [CrossRef] [PubMed]
- Barrau, K.; Boulamery, A.; Imbert, G.; Casalta, J.P.; Habib, G.; Messana, T.; Bonnet, J.L.; Rubinstein, E.; Raoult, D. Causative organisms of infective endocarditis according to host status. Clin. Microbiol. Infect. 2004, 10, 302–308. [Google Scholar] [CrossRef] [PubMed]
- Sekar, P.; Johnson, J.R.; Thurn, J.R.; Drekonja, D.M.; Morrison, V.A.; Chandrashekhar, Y.; Adabag, S.; Kuskowski, M.A.; Filice, G.A. Comparative Sensitivity of Transthoracic and Transesophageal Echocardiography in Diagnosis of Infective Endocarditis Among Veterans with Staphylococcus aureus Bacteremia. Open Forum Infect. Dis. 2017, 4, ofx035. [Google Scholar] [CrossRef]
- Bussani, R.; DE-Giorgio, F.; Pesel, G.; Zandonà, L.; Sinagra, G.; Grassi, S.; Baldi, A.; Abbate, A.; Silvestri, F. Overview and Comparison of Infectious Endocarditis and Non-infectious Endocarditis: A Review of 814 Autoptic Cases. Vivo 2019, 33, 1565–1572. [Google Scholar] [CrossRef]
- Yallowitz, A.W.; Decker, L.C. Infectious Endocarditis. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2022. Available online: https://www.ncbi.nlm.nih.gov/books/NBK557641 (accessed on 20 May 2022).
- Veve, M.P.; McCurry, E.D.; Cooksey, G.E.; Shorman, M.A. Epidemiology and outcomes of non-HACEK infective endocarditis in the southeast United States. PLoS ONE 2020, 15, e0230199. [Google Scholar] [CrossRef]
- D’Agostino, D.; Bottalico, L.; Santacroce, L. Infective endocarditis: What is changed in epidemiology and prophylaxis. Acta Med. Mediterr. 2012, 28, 311–319. [Google Scholar]
- Cresti, A.; Chiavarelli, M.; Scalese, M.; Nencioni, C.; Valentini, S.; Guerrini, F.; D’Aiello, I.; Picchi, A.; De Sensi, F.; Habib, G. Epidemiological and mortality trends in infective endocarditis, a 17-year population-based prospective study. Cardiovasc. Diagn. Ther. 2017, 7, 27–35. [Google Scholar] [CrossRef]
- Badiee, P.; Amirghofran, A.A.; Ghazi Nour, M.; Shafa, M.; Nemati, M.H. Incidence and outcome of documented fungal endocarditis. Int. Cardiovasc. Res. J. 2014, 8, 152–155. [Google Scholar]
- Demir, T.; Ergenoglu, M.U.; Ekinci, A.; Tanrikulu, N.; Sahin, M.; Demirsoy, E. Aspergillus flavus endocarditis of the native mitral valve in a bone marrow transplant patient. Am. J. Case Rep. 2015, 16, 25–30. [Google Scholar]
- Yuan, S.M. Fungal Endocarditis. Braz. J. Cardiovasc. Surg. 2016, 31, 252–255. [Google Scholar] [CrossRef] [PubMed]
- Forestier, E.; Fraisse, T.; Roubaud-Baudron, C.; Selton-Suty, C.; Pagani, L. Managing infective endocarditis in the elderly: New issues for an old disease. Clin. Interv. Aging 2016, 11, 1199–1206. [Google Scholar] [CrossRef] [PubMed]
- Fowler, V.G.; Boucher, H.W.; Corey, G.R.; Abrutyn, E.; Karchmer, A.W.; Rupp, M.E.; Levine, D.P.; Chambers, H.F.; Tally, F.P.; Vigliani, G.A.; et al. Daptomycin versus standard therapy for bacteremia and endocarditis caused by Staphylococcus aureus. N. Engl. J. Med. 2006, 355, 653–665. [Google Scholar] [CrossRef]
- Khan, O.; Shafi, A.M.; Timmis, A. International guideline changes and the incidence of infective endocarditis: A systematic review. Open Heart 2016, 3, e000498. [Google Scholar] [CrossRef] [PubMed]
| Variables * | <65 Years (n = 78) | >65 Years (n = 131) | p-Value |
|---|---|---|---|
| Age (mean ± SD) | 59.6 ± 7.2 | 67.5 ± 11.8 | <0.001 |
| Sex | 0.651 | ||
| Men | 43 (55.1%) | 68 (51.9%) | |
| Women | 35 (44.9%) | 63 (48.1%) | |
| BMI | |||
| Underweight (<18.5 kg/m2) | 5 (6.4%) | 7 (5.3%) | 0.051 |
| Normal weight (18.5–25.0 kg/m2) | 40 (51.3%) | 46 (35.1%) | |
| Overweight (>25.0 kg/m2) | 33 (42.3%) | 59.5 (59.5%) | |
| Tobacco and alcohol use | |||
| Smoking | 28 (35.9%) | 40 (30.5%) | 0.423 |
| Chronic alcohol consumption | 3 (3.8%) | 6 (4.6%) | 0.800 |
| Comorbidities | |||
| Cardiac | 33 (42.3%) | 61 (46.6%) | 0.549 |
| Metabolic | 13 (16.7%) | 29 (22.1%) | 0.339 |
| Cerebrovascular | 21 (26.9%) | 48 (36.6%) | 0.148 |
| Digestive & liver | 14 (17.9%) | 34 (26.0%) | 0.183 |
| Kidney disease | 6 (7.7%) | 14 (10.7%) | 0.476 |
| Cancer | 7 (9.0%) | 18 (13.7%) | 0.304 |
| Variables * | <65 Years (n = 78) | >65 Years (n = 131) | p-Value |
|---|---|---|---|
| Days from symptom onset until treatment (median, IQR) | 3 (2) | 2 (2) | 0.622 |
| Presence of vegetations | 61 (78.2%) | 109 (83.2%) | 0.369 |
| Presence of cardiac abscess | 14 (17.9%) | 28 (21.4%) | 0.550 |
| Localization on native valves (n = 140) | 59 (75.6%) | 81 (61.8%) | 0.040 |
| Aortic | 11 (18.6%) | 33 (40.7%) | 0.005 |
| Aortic and tricuspid | 16 (27.1%) | 12 (14.8%) | 0.072 |
| Mitral | 20 (33.9%) | 24 (29.6%) | 0.591 |
| Mitral and aortic | 12 (20.3%) | 12 (14.8%) | 0.391 |
| Localization on prosthetic valves (n = 69) | 19 (24.4%) | 50 (38.2%) | 0.040 |
| Aortic biologic valve | 3 (15.8%) | 6 (12.0%) | 0.676 |
| Aortic mechanical valve | 3 (15.8%) | 7 (14.0%) | 0.850 |
| Mitral biologic valve | 4 (21.1%) | 12 (24.0%) | 0.795 |
| Mitral mechanical valve | 5 (26.3%) | 15 (30.0%) | 0.763 |
| Mitral and aortic biologic valve | 2 (10.5%) | 6 (12.0%) | 0.864 |
| Mitral and aortic mechanical valve | 2 (10.5%) | 4 (8.0%) | 0.739 |
| Etiology | |||
| Peripheral/central vein catheter | 27 (34.6%) | 51 (38.9%) | 0.532 |
| Hemodialysis | 6 (7.7%) | 11 8.4%) | 0.856 |
| Cardiac surgery | 3 (3.8%) | 7 (5.3%) | 0.623 |
| Angiography | 5 (6.4%) | 9 (6.9%) | 0.897 |
| Vascular surgery | 8 (10.3%) | 4 (3.1%) | 0.030 |
| Gastrointestinal | 7 (9.0%) | 12 (9.2%) | 0.963 |
| Maxillo-facial interventions | 9 (11.5%) | 16 (12.2%) | 0.884 |
| Ear-nose-throat interventions | 13 (16.7%) | 21 (16.0%) | 0.904 |
| Variables * | <65 Years (n = 78) | >65 Years (n = 131) | p-Value |
|---|---|---|---|
| Surgical repair (n = 140) | 33 (42.3%) | 75 (57.3%) | 0.036 |
| Type of surgery performed | 0.044 | ||
| Aortic valve replacement | 6 (18.2%) | 20 (26.7%) | |
| Mitral valve replacement | 11 (33.3%) | 13 (17.3%) | |
| Double valve replacement | 4 (12.1%) | 24 (32.0%) | |
| Mitral valve repair | 12 (36.4%) | 18 (24.0%) | |
| Complications | |||
| Heart failure on admission | 0.005 | ||
| Yes | 25 (32.1%) | 68 (51.9%) | |
| No | 53 (67.9%) | 63 (48.1%) | |
| Severity of valvular regurgitation | 0.010 | ||
| Mild | 49 (62.8%) | 54 (41.2%) | |
| Moderate | 22 (28.2%) | 59 (45.0%) | |
| Severe | 7 (9.0%) | 18 (13.7%) | |
| Oxygen supplementation | |||
| Yes | 38 (48.7%) | 86 (65.6%) | 0.015 |
| No | 40 (51.3%) | 45 (34.4%) | |
| Cardiogenic shock | 7 (9.0%) | 19 (14.5%) | 0.241 |
| Valvular leak | 7 (9.0%) | 25 (19.1%) | 0.049 |
| Stroke | 8 (10.3%) | 19 (14.5%) | 0.375 |
| Atrioventricular block | 18 (23.1%) | 49 (37.4%) | 0.031 |
| Kidney failure | 14 (17.9%) | 47 (35.9%) | 0.005 |
| Mediastinitis | 8 (10.3%) | 27 (20.6%) | 0.052 |
| Systemic sepsis | 24 (30.8%) | 63 (48.1%) | 0.014 |
| Outcomes | |||
| ICU admission | 30 (38.5%) | 79 (60.3%) | 0.002 |
| Days in the ICU (mean ± SD) | 5.9 ± 2.2 | 7.7 ± 4.0 | <0.001 t |
| Days between symptom onset and death (mean ± SD) | 8.2 ± 6.6 | 4.7 ± 6.0 | <0.001 t |
| Days between symptom onset and ICU admission (mean ± SD) | 5.8 ± 4.1 | 3.0 ± 5.3 | <0.001 t |
| Mortality | 21 (26.9%) | 53 (40.5%) | 0.047 |
| Days until discharge (mean ± SD) | 13.8 ± 4.3 | 19.4 ± 7.1 | <0.001 t |
| Variables * | <65 Years (n = 78) | >65 Years (n = 131) | p-Value |
|---|---|---|---|
| Tests performed for infection identification | 0.579 | ||
| Culture | 46 (59.0%) | 77 (58.8%) | |
| PCR | 47 (28.2%) | 31 (23.7%) | |
| Culture and PCR | 72 (12.8%) | 23 (17.6%) | |
| Testing | |||
| Positive samples | 48 (61.5%) | 77 (58.8%) | |
| False negative result | 30 (38.5%) | 54 (41.2%) | |
| Identification | 0.542 | ||
| Bacterial | 69 (88.5%) | 112 (85.5%) | |
| Fungal | 9 (11.5%) | 19 (14.5%) | |
| Pathogens involved | 0.319 | ||
| Staphylococcus aureus | 22 (28.2%) | 38 (29.0%) | |
| CoNs | 7 (9.0%) | 19 (14.5%) | |
| Streptococcus spp. | 20 (25.6%) | 20 (15.3%) | |
| Escherichia coli | 3 (3.8%) | 9 (6.9%) | |
| Enterococcus faecalis | 12 (15.4%) | 17 (13.0%) | |
| Other gram-negative bacilli | 5 (6.4%) | 9 (6.9%) | |
| Candida spp. | 6 (7.7%) | 13 (9.9%) | |
| Aspergillus spp. | 3 (3.8%) | 6 (4.6%) | |
| Severe treatment complications | 6 (7.7%) | 23 (17.6%) | 0.046 |
| Treatment regimen type | 0.417 | ||
| Monotherapy | 45 (57.7%) | 68 (51.9%) | |
| Combined | 33 (42.3%) | 63 (48.1%) | |
| Multidrug resistance | 0.758 | ||
| Yes | 24 (30.8%) | 43 (32.8%) | |
| No | 54 (69.2%) | 88 (67.2%) | |
| Number of pathogens identified | 0.566 | ||
| Monoinfection | 73 (93.6%) | 125 (95.4%) | |
| Two pathogens | 5 (6.4%) | 6 (4.6%) |
| Variables * | <65 Years (n = 78) | >65 Years (n = 131) | p-Value |
|---|---|---|---|
| Antibiotics | |||
| Acute immune reactions | 5 (6.4%) | 6 (4.6%) | 0.566 |
| Delayed reactions | 4 (5.1%) | 4 (3.1%) | 0.449 |
| Nephrotoxicity | 4 (5.1%) | 18 (13.7%) | 0.049 |
| Neurotoxicity | 2 (2.6%) | 9 (6.9%) | 0.177 |
| Liver injury | 3 (3.8%) | 16 (12.2%) | 0.041 |
| Enterocolitis | 11 (14.1%) | 38 (29.0%) | 0.013 |
| Falls and delirium | 1 (1.3%) | 16 (12.2%) | 0.005 |
| Antifungals | |||
| Acute immune reactions | 1 (1.3%) | 3 (2.3%) | 0.606 |
| Delayed reactions | 0 (0.0%) | 2 (1.5%) | 0.272 |
| Nephrotoxicity | 2 (2.6%) | 11 (8.4%) | 0.091 |
| Neurotoxicity | 1 (1.3%) | 4 (3.1%) | 0.417 |
| Liver injury | 2 (2.6%) | 9 (6.9%) | 0.532 |
| Enterocolitis | 14 (5.1%) | 10 (7.6%) | 0.483 |
| Falls and delirium | 0 (0.0%) | 7 (20.6%) | 0.037 |
| Factors * | <65 Years or (95% CI) | p-Value | >65 Years or (95% CI) | p-Value |
|---|---|---|---|---|
| Heart failure at admission | 3.15 (2.87–5.21) | <0.001 | 4.07 (3.44–6.90) | <0.001 |
| Septic shock | 3.08 (2.66–6.09) | <0.001 | 6.19 (4.15–8.28) | <0.001 |
| Treatment nephrotoxicity | 1.66 (1.07–2.34) | 0.001 | 3.14 (2.36–4.03) | 0.001 |
| Severe treatment complications | 3.39 (2.25–5.11) | <0.001 | 4.65 (3.82–6.21) | <0.001 |
| Antibiotic resistance | 2.61 (1.71–4.06) | <0.001 | 3.24 (2.09–5.52) | <0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Budea, C.M.; Pricop, M.; Bratosin, F.; Bogdan, I.; Saenger, M.; Ciorica, O.; Braescu, L.; Domuta, E.M.; Grigoras, M.L.; Citu, C.; et al. Antibacterial and Antifungal Management in Relation to the Clinical Characteristics of Elderly Patients with Infective Endocarditis: A Retrospective Analysis. Antibiotics 2022, 11, 956. https://doi.org/10.3390/antibiotics11070956
Budea CM, Pricop M, Bratosin F, Bogdan I, Saenger M, Ciorica O, Braescu L, Domuta EM, Grigoras ML, Citu C, et al. Antibacterial and Antifungal Management in Relation to the Clinical Characteristics of Elderly Patients with Infective Endocarditis: A Retrospective Analysis. Antibiotics. 2022; 11(7):956. https://doi.org/10.3390/antibiotics11070956
Chicago/Turabian StyleBudea, Camelia Melania, Marius Pricop, Felix Bratosin, Iulia Bogdan, Miriam Saenger, Ovidiu Ciorica, Laurentiu Braescu, Eugenia Maria Domuta, Mirela Loredana Grigoras, Cosmin Citu, and et al. 2022. "Antibacterial and Antifungal Management in Relation to the Clinical Characteristics of Elderly Patients with Infective Endocarditis: A Retrospective Analysis" Antibiotics 11, no. 7: 956. https://doi.org/10.3390/antibiotics11070956
APA StyleBudea, C. M., Pricop, M., Bratosin, F., Bogdan, I., Saenger, M., Ciorica, O., Braescu, L., Domuta, E. M., Grigoras, M. L., Citu, C., Diaconu, M. M., & Marincu, I. (2022). Antibacterial and Antifungal Management in Relation to the Clinical Characteristics of Elderly Patients with Infective Endocarditis: A Retrospective Analysis. Antibiotics, 11(7), 956. https://doi.org/10.3390/antibiotics11070956








