Longitudinal Study of Therapeutic Adherence in a Cystic Fibrosis Unit: Identifying Potential Factors Associated with Medication Possession Ratio
Abstract
1. Introduction
2. Results
2.1. Demographic and Clinical Characteristics
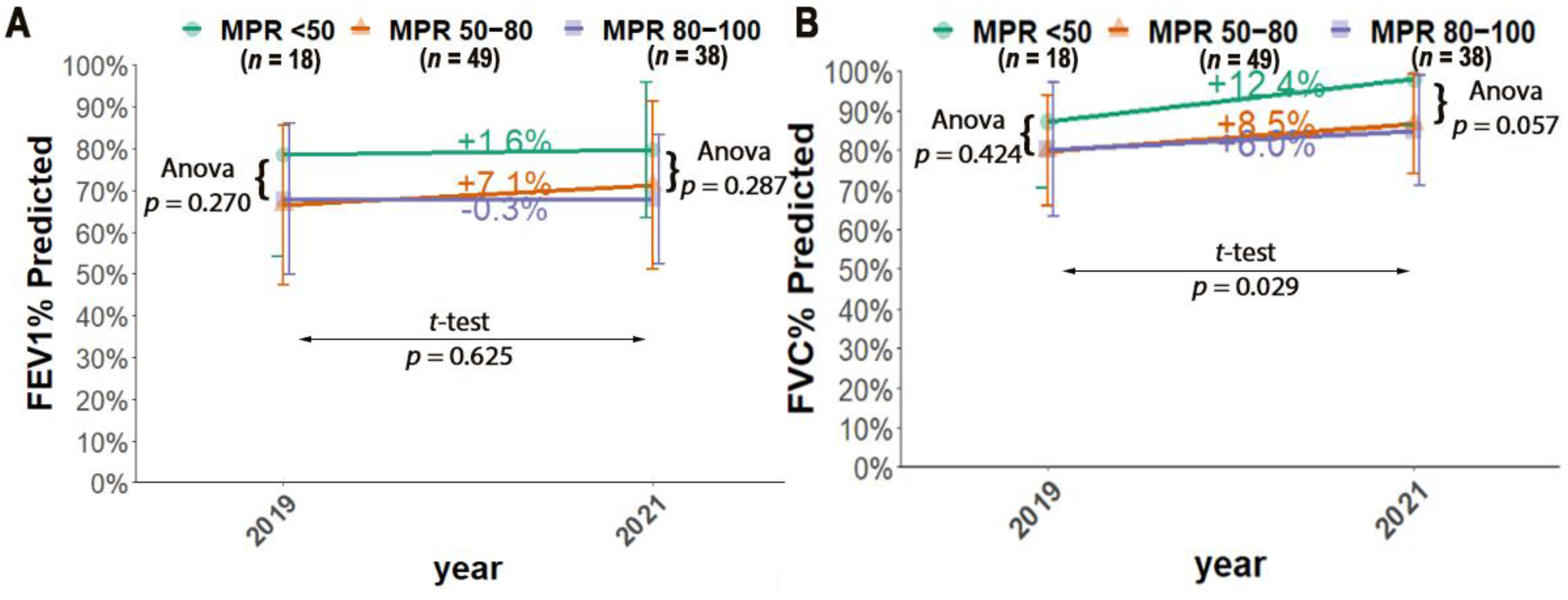
2.2. The Relationship between Compliance and Lung Function
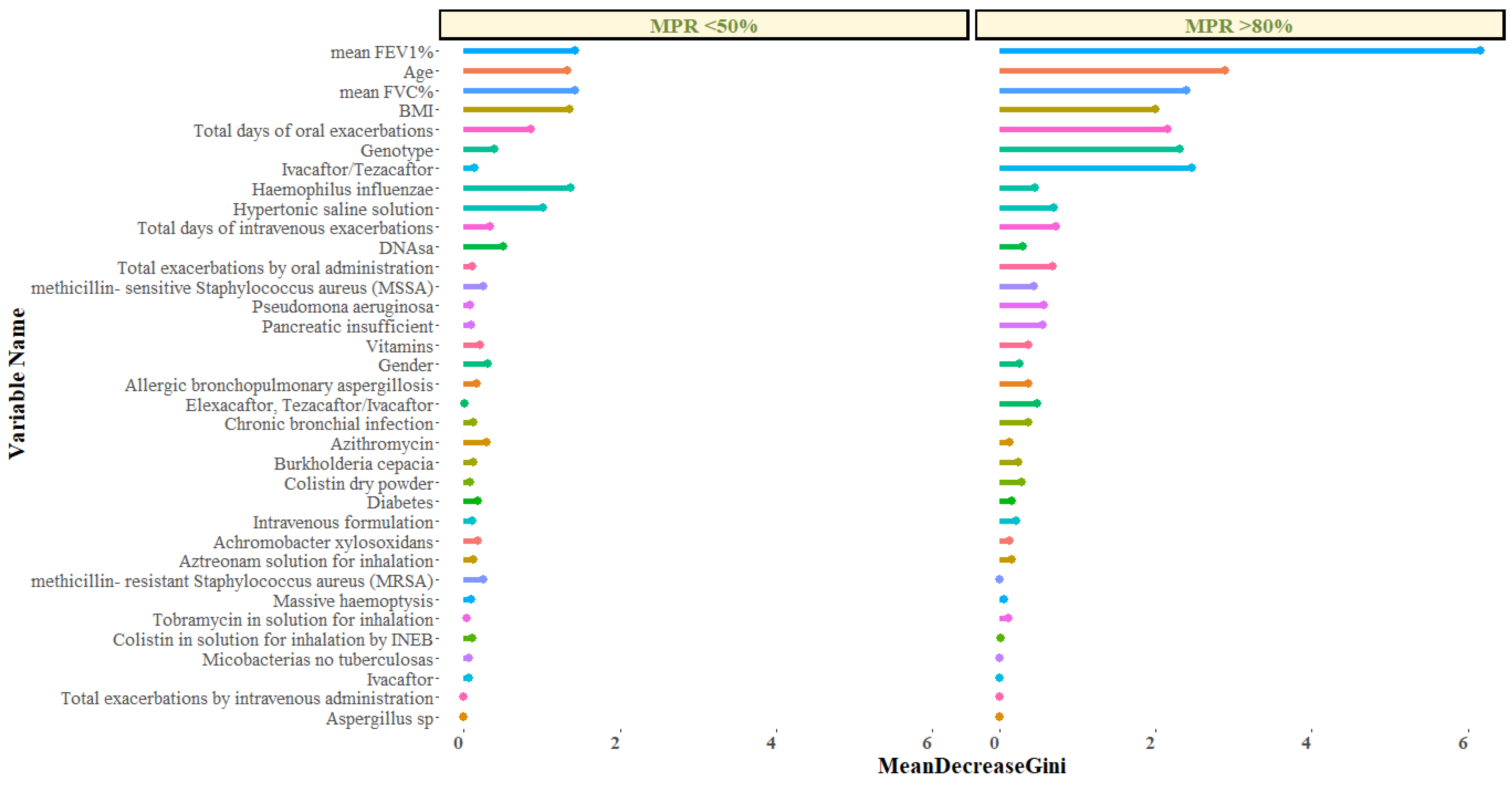
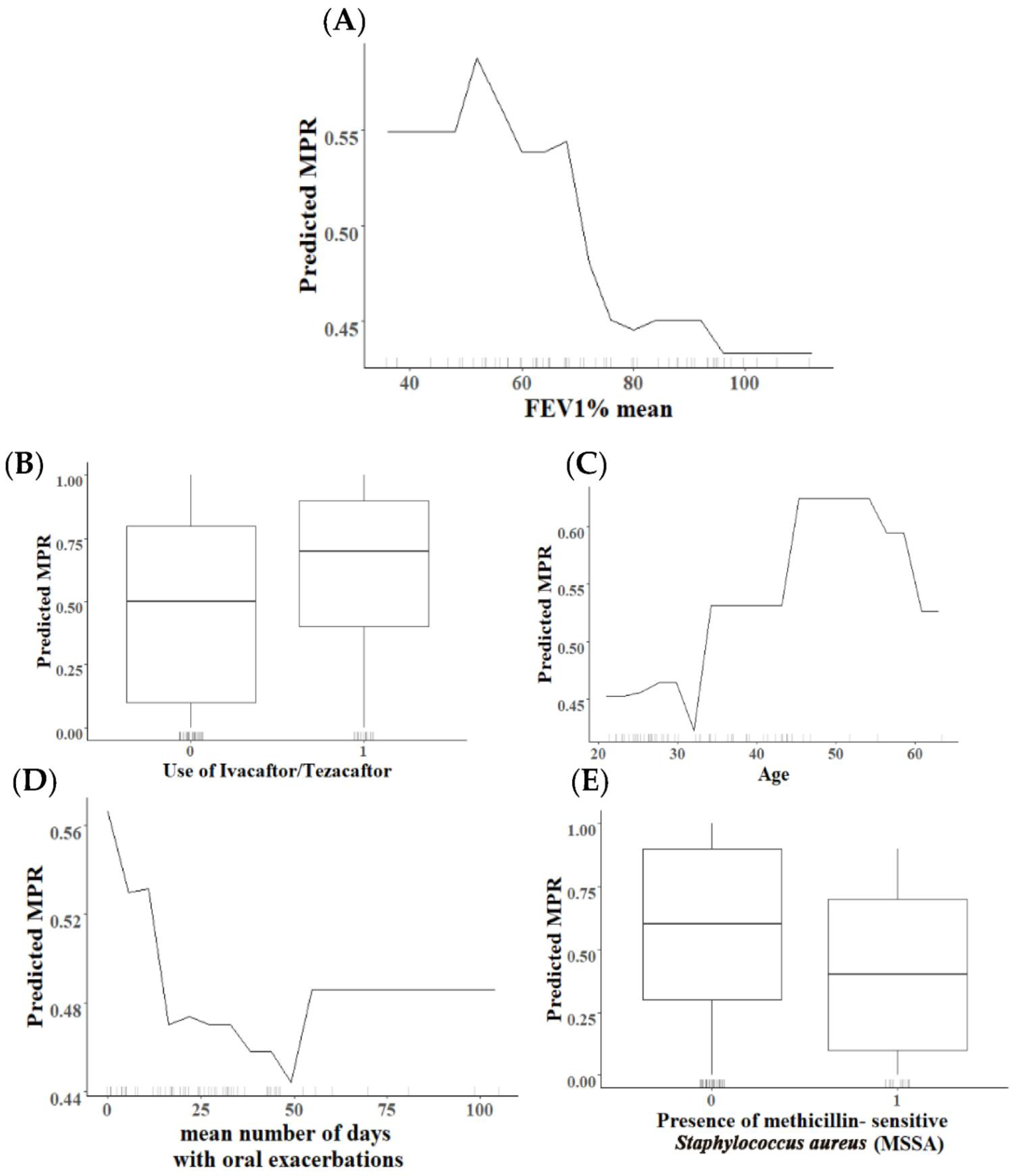
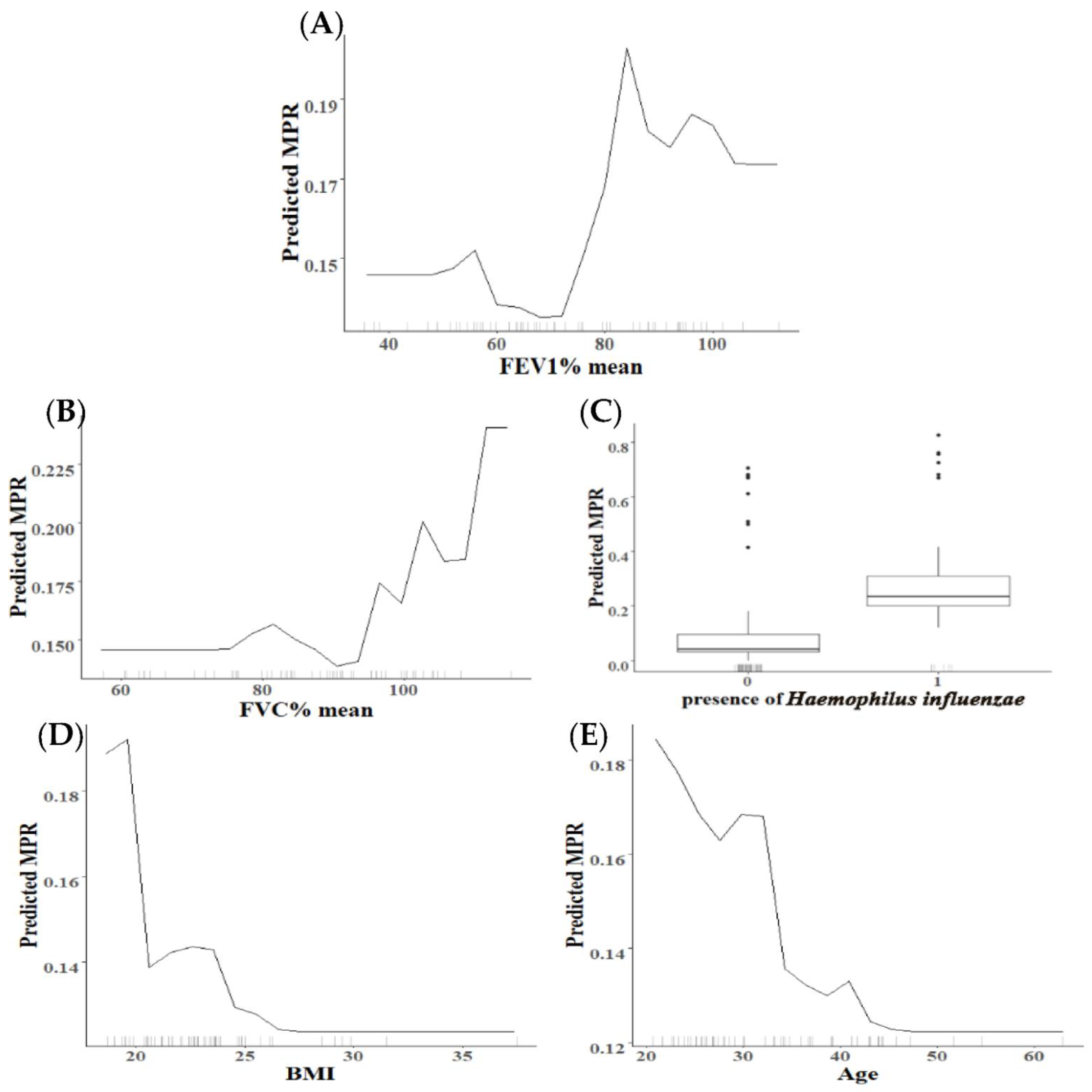
2.3. Analysis of Variables with Higher Relevance in the Percentage of MPR
3. Discussion
4. Materials and Methods
4.1. Patient Recruitment
4.2. AT Assessment and Collection of Clinical Variables
4.3. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sawicki, G.S.; Sellers, D.E.; Robinson, W.M. High treatment burden in adults with cystic fibrosis: Challenges to disease self-management. J. Cyst. Fibros. 2009, 8, 91–96. [Google Scholar] [CrossRef] [PubMed]
- Narayanan, S.; Mainz, J.G.; Gala, S.; Tabori, H.; Grossoehme, D. Adherence to therapies in cystic fibrosis: A targeted literature review. Expert Rev. Respir. Med. 2016, 11, 129–145. [Google Scholar] [CrossRef] [PubMed]
- Moreno, R.G.; García-Clemente, M.; Diab-Cáceres, L.; Martínez-Vergara, A.; Martínez-García, M.; Gómez-Punter, R. Treatment of Pulmonary Disease of Cystic Fibrosis: A Comprehensive Review. Antibiotics 2021, 10, 486. [Google Scholar] [CrossRef] [PubMed]
- Taylor-Cousar, J.L.; Munck, A.; McKone, E.F.; Van Der Ent, C.K.; Moeller, A.; Simard, C.; Wang, L.T.; Ingenito, E.P.; McKee, C.; Lu, Y.; et al. Tezacaftor–Ivacaftor in Patients with Cystic Fibrosis Homozygous for Phe508del. N. Engl. J. Med. 2017, 377, 2013–2023. [Google Scholar] [CrossRef] [PubMed]
- Rowe, S.M.; Daines, C.; Ringshausen, F.C.; Kerem, E.; Wilson, J.; Tullis, E.; Nair, N.; Simard, C.; Han, L.; Ingenito, E.P.; et al. Tezacaftor–Ivacaftor in Residual-Function Heterozygotes with Cystic Fibrosis. N. Engl. J. Med. 2017, 377, 2024–2035. [Google Scholar] [CrossRef]
- Flume, P.A.; Biner, R.F.; Downey, D.G.; Brown, C.; Jain, M.; Fischer, R.; De Boeck, K.; Sawicki, G.S.; Chang, P.; Paz-Diaz, H.; et al. Long-term safety and efficacy of tezacaftor–ivacaftor in individuals with cystic fibrosis aged 12 years or older who are homozygous or heterozygous for Phe508del CFTR (EXTEND): An open-label extension study. Lancet Respir. Med. 2021, 9, 733–746. [Google Scholar] [CrossRef]
- Graeber, S.Y.; Vitzthum, C.; Pallenberg, S.T.; Naehrlich, L.; Stahl, M.; Rohrbach, A.; Drescher, M.; Minso, R.; Ringshausen, F.C.; Rueckes-Nilges, C.; et al. Effects of Elexacaftor/Tezacaftor/Ivacaftor Therapy on CFTR Function in Patients with Cystic Fibrosis and One or Two F508del Alleles. Am. J. Respir. Crit. Care Med. 2022, 205, 540–549. [Google Scholar] [CrossRef]
- Carnovale, V.; Iacotucci, P.; Terlizzi, V.; Colangelo, C.; Ferrillo, L.; Pepe, A.; Francalanci, M.; Taccetti, G.; Buonaurio, S.; Celardo, A.; et al. Elexacaftor/Tezacaftor/Ivacaftor in Patients with Cystic Fibrosis Homozygous for the F508del Mutation and Advanced Lung Disease: A 48-Week Observational Study. J. Clin. Med. 2022, 11, 1021. [Google Scholar] [CrossRef]
- World Health Organization. Adherence to Long-Term Therapies: Evidence for Action. Available online: https://www.who.int/chp/knowledge/publications/adherence_report/en/ (accessed on 8 June 2021).
- Quittner, A.L.; Zhang, J.; Marynchenko, M.; Chopra, P.A.; Signorovitch, J.; Yushkina, Y.; Riekert, K.A. Pulmonary Medication Adherence and Health-care Use in Cystic Fibrosis. Chest 2014, 146, 142–151. [Google Scholar] [CrossRef]
- Eakin, M.N.; Riekert, K.A. The impact of medication adherence on lung health outcomes in cystic fibrosis. Curr. Opin. Pulm. Med. 2013, 19, 687–691. [Google Scholar] [CrossRef]
- Conway, S.P.; Pond, M.N.; Hamnett, T.; Watson, A. Compliance with treatment in adult patients with cystic fibrosis. Thorax 1996, 51, 29–33. [Google Scholar] [CrossRef] [PubMed]
- Abbott, J.; Dodd, M.; Bilton, D.; Webb, A.K. Treatment compliance in adults with cystic fibrosis. Thorax 1994, 49, 115–120. [Google Scholar] [CrossRef] [PubMed]
- Pagès-Puigdemont, N.; Valverde-Merino, M.I. Métodos para medir la adhesión terapeútica. Ars Pham. 2018, 59, 163–172. [Google Scholar]
- Modi, A.C.; Lim, C.S.; Yu, N.; Geller, D.; Wagner, M.H.; Quittner, A.L. A multi-method assessment of treatment adherence for chil-dren with cystic fibrosis. J. Cyst. Fibros. 2006, 5, 177–185. [Google Scholar] [CrossRef] [PubMed]
- Quittner, A.L.; Saez-Flores, E.; Barton, J.D. The psychological burden of cystic fibrosis. Curr. Opin. Pulm. Med. 2016, 22, 187–191. [Google Scholar] [CrossRef]
- Liu, M.; Xu, X.; Tao, Y.; Wang, X. An improved random forest method based on RELIEFF for medical diagnosis. In Proceedings of the 2017 IEEE International Conference on Computational Science and Engineering (CSE) and IEEE International Confer-ence on Embedded and Ubiquitous Computing (EUC), Guangzhou, China, 21–24 July 2017; Volume 1, pp. 44–49. [Google Scholar]
- Strobl, C.; Boulesteix, A.L.; Zeileis, A.; Hothorn, T. Bias in random forest variable importance measures: Illustrations, sources and a solution. BMC Bioinformat. 2007, 8, 25. [Google Scholar] [CrossRef]
- Luan, J.; Zhang, C.; Xu, B.; Xue, Y.; Ren, Y. The predictive performances of random forest models with limited sample size and different species traits. Fish. Res. 2020, 227, 105534. [Google Scholar] [CrossRef]
- Grömping, U. Variable Importance Assessment in Regression: Linear Regression versus Random Forest. Am. Stat. 2009, 63, 308–319. [Google Scholar] [CrossRef]
- Grekousis, G.; Feng, Z.; Marakakis, I.; Lu, Y.; Wang, R. Ranking the importance of demographic, socioeconomic, and underlying health factors on US COVID-19 deaths: A geographical random forest approach. Health Place 2022, 74, 102744. [Google Scholar] [CrossRef]
- Report of the WHO-China Joint Mission on Coronavirus Disease 2019 (COVID-19). 2020. Available online: https://www.who.int/docs/default-source/coronaviruse/who-china-joint-mission-on-covid-19-final-report.pdf (accessed on 9 March 2020).
- Ahn, D.-G.; Shin, H.-J.; Kim, M.-H.; Lee, S.; Kim, H.-S.; Myoung, J.; Kim, B.-T.; Kim, S.-J. Current Status of Epidemiology, Diagnosis, Therapeutics, and Vaccines for Novel Coronavirus Disease 2019 (COVID-19). J. Microbiol. Biotechnol. 2020, 30, 313–324. [Google Scholar] [CrossRef]
- Mehta, Z.; Kamal, K.M.; Miller, R.; Covvey, J.R.; Giannetti, V. Adherence to cystic fibrosis transmembrane conductance regulator (CFTR) modulators: Analysis of a national specialty pharmacy database. J. Drug Assess. 2021, 10, 62–67. [Google Scholar] [CrossRef] [PubMed]
- Calle, M.L.; Urrea, V. Letter to the Editor: Stability of Random Forest importance measures. Briefings Bioinform. 2010, 12, 86–89. [Google Scholar] [CrossRef] [PubMed]
- Manika, K.; Hatziagorou, E.; Msc, S.C.K.; Kyrvasili, S.S.; Sourla, E.; Kouroukli, E.; Sionidou, M.; Papadaki, E.; Tsanakas, J. Adherence to inhaled therapies over 4 years in people with cystic fibrosis. Pediatr. Pulmonol. 2022, 57, 956–964. [Google Scholar] [CrossRef] [PubMed]
- Salmon, P.; Young, B. A new paradigm for clinical communication: Critical review of literature in cancer care. Med. Educ. 2016, 51, 258–268. [Google Scholar] [CrossRef] [PubMed]
- Eakin, M.N.; Bilderback, A.; Boyle, M.P.; Mogayzel, P.J.; Riekert, K.A. Longitudinal association between medication adherence and lung health in people with cystic fibrosis. J. Cyst. Fibros. 2011, 10, 258–264. [Google Scholar] [CrossRef]
- Ball, R.; Southern, K.W.; McCormack, P.; Duff, A.J.; Brownlee, K.G.; McNamara, P.S. Adherence to nebulised therapies in adolescents with cystic fibrosis is best on week-days during school term-time. J. Cyst. Fibros. 2013, 12, 440–444. [Google Scholar] [CrossRef]
- Snider, B.; McBean, E.A.; Yawney, J.; Gadsden, S.A.; Patel, B. Identification of variable importance for predictions of mortality from COVID-19 using AI models for Ontario, Canada. Front. Public Health 2021, 9, 675766. [Google Scholar] [CrossRef]
- Soogun, A.O.; Kharsany, A.; Zewotir, T.; North, D.; Ogunsakin, R.E. Identifying Potential Factors Associated with High HIV viral load in KwaZulu-Natal, South Africa using Multiple Correspondence Analysis and Random Forest Analysis. BMC Med. Res. Methodol. 2022, 22, 174. [Google Scholar] [CrossRef]
- Machado, G.; Mendoza, M.R.; Corbellini, L.G. What variables are important in predicting bovine viral diarrhea virus? A random forest approach. Vet. Res. 2015, 46, 85. [Google Scholar] [CrossRef]
- Abraham, O.; Buechel, B.M.; Gay, S.; Szela, B.L.; Decker, C.A.; Braun, A.T. Using A Work System Framework to Investigate Pharmacists’ Roles in Cystic Fibrosis Management. J. Pediatr. Pharmacol. Ther. 2022, 27, 270–276. [Google Scholar] [CrossRef]
- Zobell, J.T.; Schwab, E.; Collingridge, D.S.; Ball, C.; Nohavec, R.; Asfour, F. Impact of pharmacy services on cystic fibrosis medication adherence. Pediatr. Pulmonol. 2017, 52, 1006–1012. [Google Scholar] [CrossRef] [PubMed]
- Abraham, O.; Li, J.S.; Monangai, K.E.; Feathers, A.M.; Weiner, D. The pharmacist’s role in supporting people living with cystic fibrosis. J. Am. Pharm. Assoc. 2018, 58, 246–249. [Google Scholar] [CrossRef] [PubMed]
- Sánchez Azofra, A.; Calvo-García, A.; Ruiz Garcia, A.; Girón Moreno, R.; Ibañez Zurriaga, A.; Orzaiz, A.; Sanz, P.; Bermúdez, A.; Baladron, M. Cystic fibrosis outpatient treatment andmedical costs: A retrospective análisis. Ofil·Ilaphar 2020, 30, 283–290. [Google Scholar]
- Muther Emily, F. Understanding Treatment Adherence in Cystic Fibrosis: Challenges and Opportunities. In Cystic Fibrosis: A Multi-Organ System Approach: A Multi-Organ System Approach; Davis, S., Rosenfeld, M., Chmiel, J., Eds.; Humana: Cham, Switzerland, 2020; pp. 449–463. [Google Scholar] [CrossRef]
- Mayer-Hamblett, N.; Nichols, D.P.; Odem-Davis, K.; Riekert, K.A.; Sawicki, G.S.; Donaldson, S.H.; Ratjen, F.; Konstan, M.W.; Simon, N.; Rosenbluth, D.B.; et al. Evaluating the Impact of Stopping Chronic Therapies after Modulator Drug Therapy in Cystic Fibrosis: The SIMPLIFY Clinical Trial Study Design. Ann. Am. Thorac. Soc. 2021, 18, 1397–1405. [Google Scholar] [CrossRef] [PubMed]
- CF STORM; A Trial to Assess Change in Respiratory Function for People with Cystic Fibrosis on Triple Combination Therapy (Kaftrio™) after Rationalisation of Nebulised Mucoactive Therapies. Available online: https://www.cfstorm.org.uk (accessed on 27 September 2022).
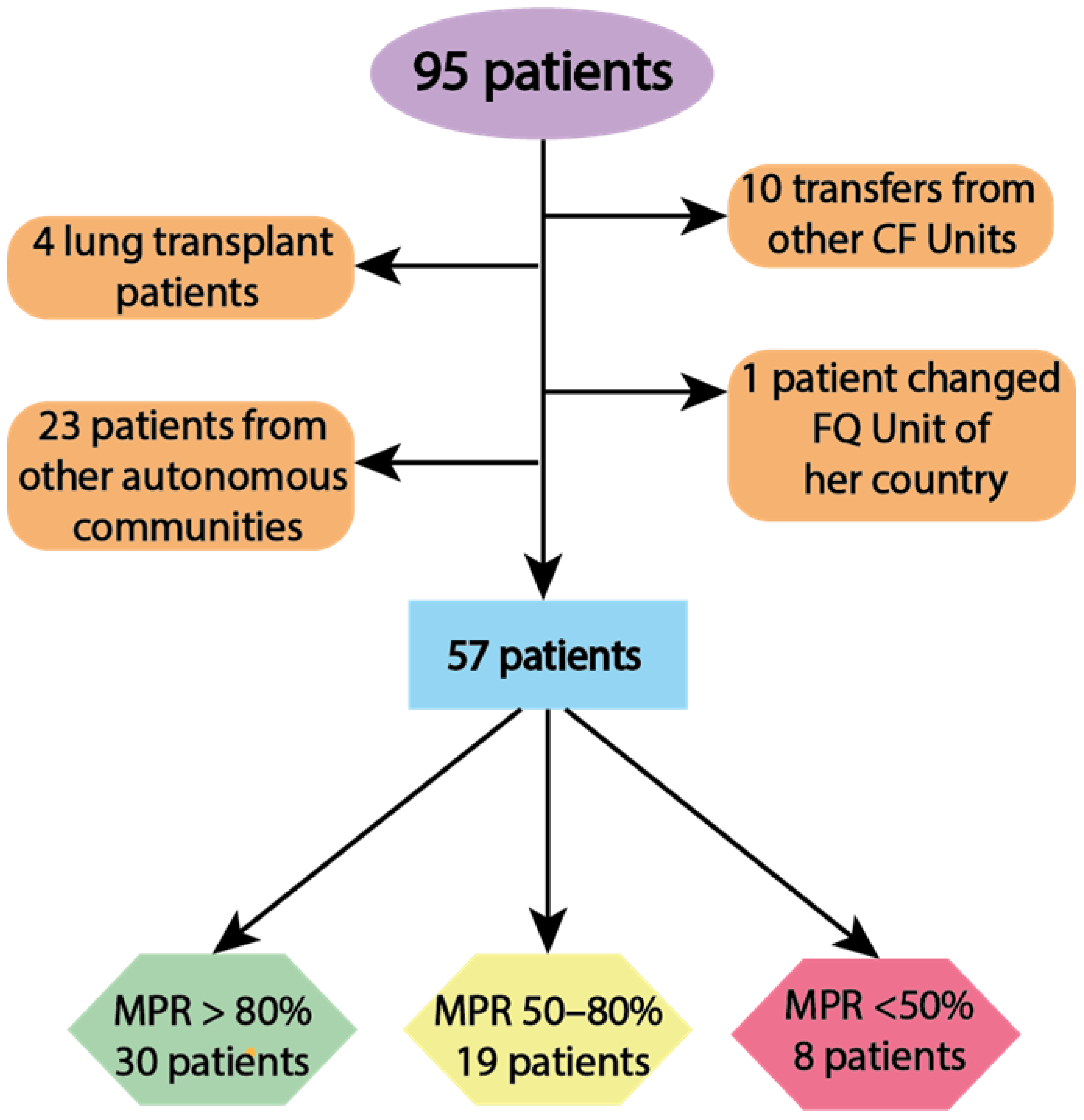
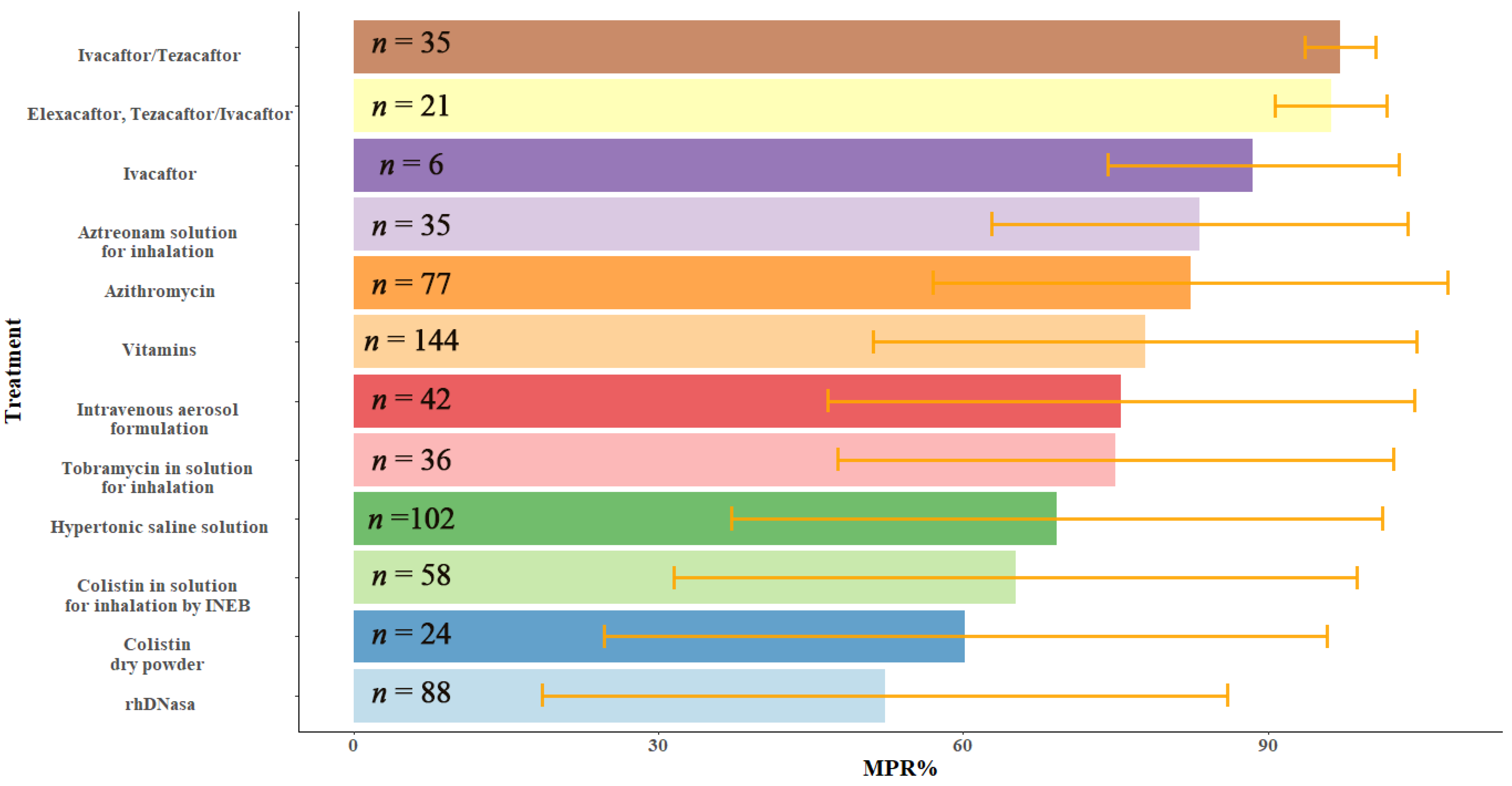
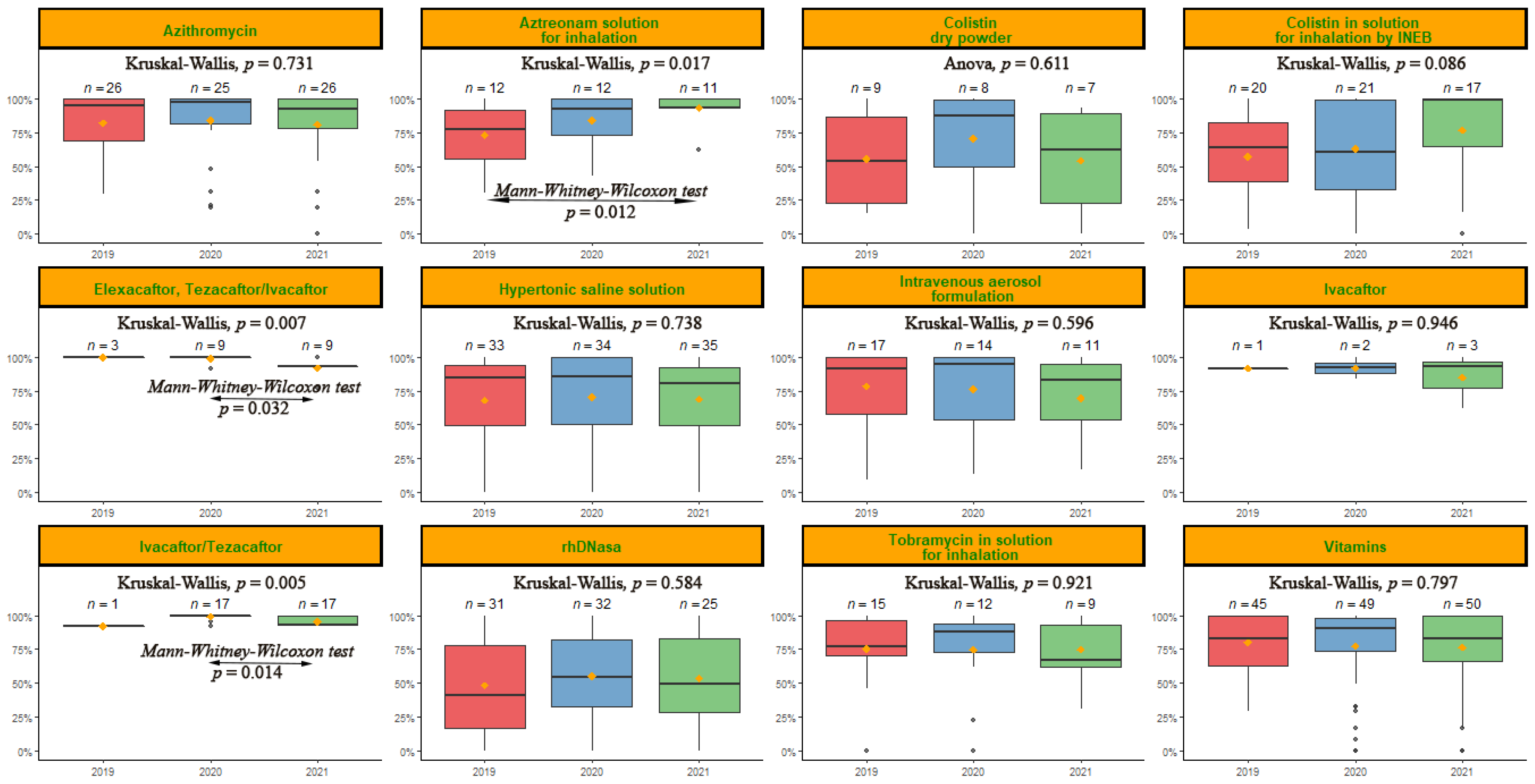
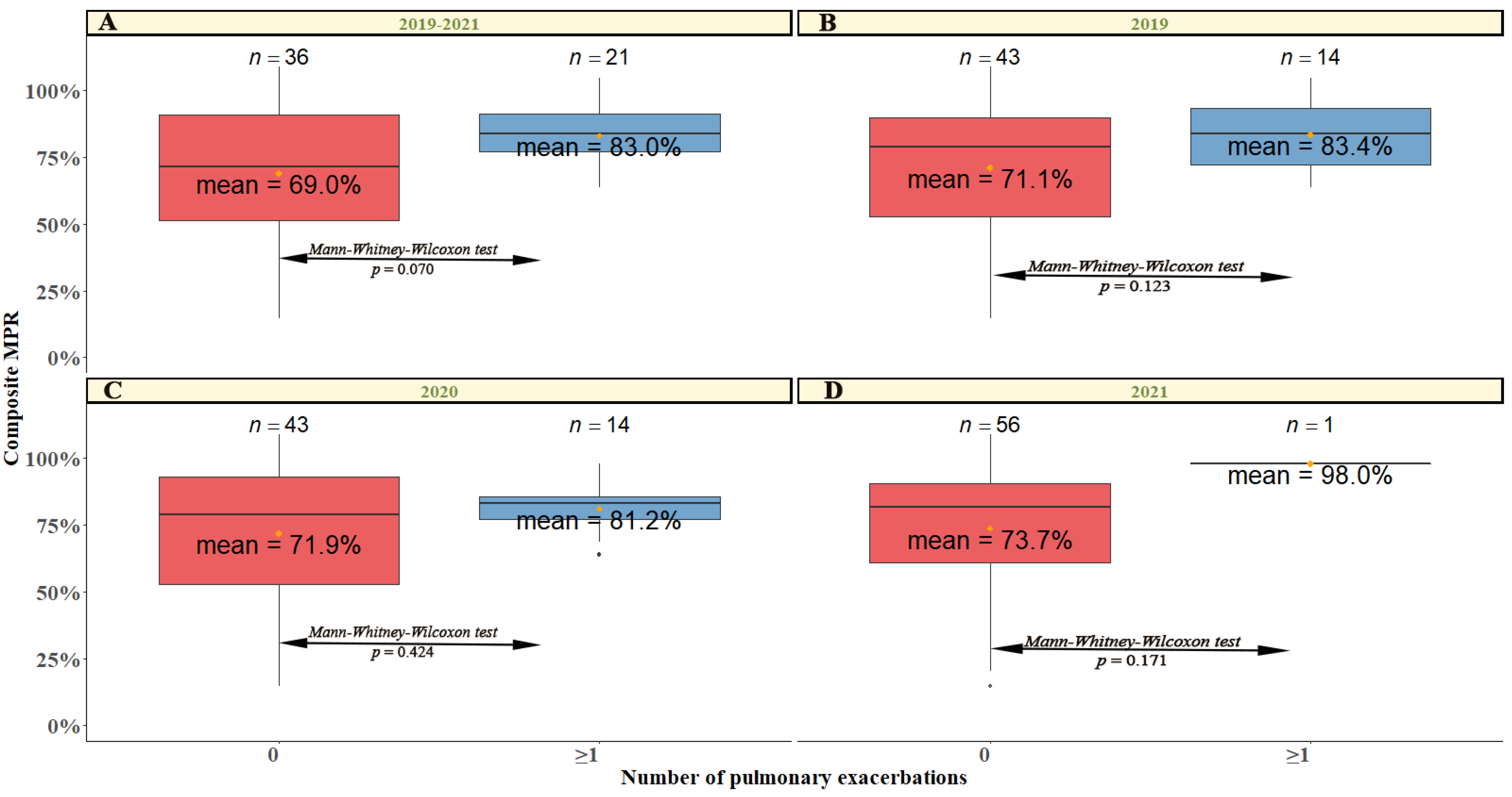
| Demographic and Clinical Characteristics | Total (n = 57) | MPR < 50 (n = 8) | MPR 50–80 (n = 19) | MPR > 80 (n = 30) | p-Value |
|---|---|---|---|---|---|
| Age, (mean ± SD) | 33.9 ± 9.4 | 28.5 ± 6.3 | 32.0 ± 10.5 | 36.5 ± 8.7 | 0.030 *4 |
| 20–30 yrs., n (%) | 25 (43.9%) | 6 (75.0%) | 11 (57.9%) | 8 (26.7%) | 0.017 * |
| 30–40 yrs., n (%) | 16 (28.1%) | 1 (12.5%) | 4 (21.1%) | 11 (36.7%) | 0.421 |
| ≥40 yrs., n (%) | 16 (28.1%) | 1 (12.5%) | 4 (21.1%) | 11 (36.7%) | 0.421 |
| Gender, n (%) | |||||
| Women | 34 (59.6%) | 4 (50.0%) | 11 (57.9%) | 19 (63.3%) | 0.810 |
| BMI, (mean ± SD) | 23.2 ± 3.4 | 22.1 ± 1.7 | 23.7 ± 4.5 | 23.2 ± 2.9 | 0.786 |
| Genotype, n (%) | |||||
| F508del/F508del | 20 (35.1%) | 1 (12.5%) | 5 (26.3%) | 14 (46.7%) | 0.145 |
| F508del/other | 23 (40.4%) | 4 (50.0%) | 11 (57.9%) | 8 (26.7%) | 0.072 |
| other/other | 14 (24.6%) | 3 (37.5%) | 3 (15.8%) | 8 (26.7%) | 0.429 |
| Pancreatic insufficient, n (%) | 46 (80.7%) | 6 (75.0%) | 15 (78.9%) | 25 (83.3%) | 0.727 |
| Diabetes, n (%) | 24 (42.1%) | 4 (50.0%) | 8 (42.1%) | 12 (40.0%) | 0.933 |
| Allergic bronchopulmonary aspergillosis, n (%) | 14 (24.6%) | 2 (25.0%) | 3 (15.8%) | 9 (30.0%) | 0.529 |
| Massive hemoptysis, n (%) | 9 (15.8%) | 1 (12.5%) | 2 (10.5%) | 6 (20.0%) | 0.783 |
| Lung function, (mean ± SD) | |||||
| FEV1 1 % | |||||
| 2019 | 71.0 ± 20.3 | 80.5 (16.8) | 75.4 (23.7) | 65.7 (17.8) | 0.099 |
| 2020 | 72.9 ± 18.5 | 84.1 (15.7) | 76.4 (22.2) | 67.6 (15.1) | 0.042 |
| 2021 3 | 71.8 ± 18.7 | 82.0 (10.2) | 76.4 (22.5) | 66.9 (16.1) | 0.059 |
| Total | 72.2 ± 18.9 | 81.7 (15.1) | 76.4 (22.5) | 66.9 (16.1) | 0.069 |
| FVC% 2 | |||||
| 2019 | 82.7 ± 15.6 | 90.0 (13.0) | 86.1 (14.7) | 78.5 (15.9) | 0.094 |
| 2020 | 88.1 ± 14.0 | 99.3 (11.6) | 90.6 (14.1) | 83.5 (12.7) | 0.009 * |
| 2021 1 | 87.9 ± 13.6 | 96.3 (12.9) | 91.1 (13.8) | 84.2 (12.7) | 0.059 |
| Total | 86.5 ± 13.9 | 95.0 (11.6) | 89.6 (13.8) | 82.2 (13.3) | 0.031 |
| Bacteria presence, n (%) | |||||
| Chronic bronchial infection | 48 (84.2%) | 8 (100%) | 16 (84.2%) | 24 (80.0%) | 0.593 |
| Pseudomonas aeruginosa | 22 (38.6%) | 1 (12.5%) | 6 (31.6%) | 15 (50.0%) | 0.377 |
| methicillin-sensitive Staphylococcus aureus (MSSA) | 16 (28.1%) | 4 (50.0%) | 7 (36.8%) | 5 (16.7%) | 0.511 |
| methicillin-resistant Staphylococcus aureus (MRSA) | 10 (17.5%) | 2 (25.0%) | 3 (15.8%) | 5 (16.7%) | 0.951 |
| Achromobacter xylosoxidans | 6 (10.5%) | 1 (12.5%) | 3 (15.8%) | 2 (6.7%) | 0.921 |
| Burkholderia cepacia | 5 (8.8%) | 1 (12.5%) | 1 (5.26%) | 3 (10.0%) | 0.915 |
| Aspergillus sp | 1 (1.8%) | 0 (0.0%) | 0 (0.0%) | 1 (3.3%) | 0.806 |
| Haemophilus influenzae | 6 (10.5%) | 4 (50.0%) | 0 (0.0%) | 2 (6.7%) | 0.004 * |
| Stenotrophomonas maltophilia | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) | 1.000 |
| Mycobacteria non tuberculous | 1 (1.8%) | 0 (0.0%) | 0 (0.0%) | 1 (3.33%) | 0.806 |
| Year | Oral Administration | Intravenous Administration | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Total (n = 57) | MPR < 50 (n = 8) | MPR 50–80 (n = 19) | MPR > 80 (n = 30) | p-Value | Total (n = 57) | MPR < 50 (n = 8) | MPR 50–80 (n = 19) | MPR > 80 (n = 30) | p-Value | ||
| Exacerbations (mean ± SD) | 2019 | 2.1 ± 1.9 | 1.6 ± 1.2 | 2.2 ± 1.7 | 2.1 ± 2.1 | 0.763 | 0.4 ± 0.8 | 0.0 ± 0.0 | 0.4 ± 0.9 | 0.5 ± 0.9 | 0.220 |
| 2020 | 1.8 ± 1.6 | 1.5 ± 0.9 | 1.4 ± 1.6 | 2.1 ± 1.7 | 0.228 | 0.2 ± 0.4 | 0.0 ± 0.0 | 0.3 ± 0.5 | 0.3 ± 0.5 | 0.217 | |
| 2021 | 0.9 ± 1.0 | 0.9 ± 1.2 | 0.9 ± 0.9 | 0.8 ± 1.0 | 0.858 | 0.0 ± 0.1 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.2 | 0.648 | |
| Total | 1.5 ± 1.1 | 1.4 ± 0.7 | 1.5 ± 1.1 | 1.7 ± 1.3 | 0.71 | 0.2 ± 0.3 | 0.0 ± 0.0 | 0.2 ± 0.3 | 0.3 0.4 | 0.072 | |
| ≥1 exacerbation, n (%) | 2019 | 43 (75.4%) | 6 (75.0%) | 15 (78.9%) | 22 (73.3%) | 0.905 | 14 (24.6%) | 0 (0.0%) | 5 (26.3%) | 9 (30.0%) | 0.211 |
| 2020 | 43 (75.4%) | 7 (87.5%) | 12 (63.2%) | 24 (80.0%) | 0.285 | 14 (24.6%) | 0 (0.0%) | 5 (26.3%) | 9 (30.0%) | 0.211 | |
| 2021 | 31 (54.4%) | 3 (37.5%) | 12 (63.2%) | 16 (53.3%) | 0.467 | 1 (1.8%) | 0 (0.0%) | 0 (0.0%) | 1 (3.3%) | 0.633 | |
| Total | 42 (73.7%) | 7 (87.5%) | 14 (73.7%) | 21 (70.0%) | 0.607 | 4 (7.0%) | 0 (0.0%) | 1 (5.3%) | 3 (10.0%) | 0.576 | |
| Days 2 (mean ± SD) | 2019 | 47.9 ± 32.5 | 33.7 (10.1) | 48.5 (24.4) | 51.1 (39.9) | 0.548 | 26.6 ± 14.01 | NA 3 ± NA | 26.6 ± 17.4 | 26.6±12.9 | 0.838 |
| 2020 | 42.2 ± 31.0 | 30.0 (18.4) | 37.9 (23.8) | 47.9 (36.2) | 0.452 | 16.2 ± 4.7 | NA ± NA | 18.2 ± 3.8 | 15.1±5.0 | 0.246 | |
| 2021 | 29.5 ± 18.6 | 35.0 (18.5) | 21.6 (11.3) | 34.3 (21.8) | 0.137 | 14.0 ± ND | NA ± NA | NA ± NA | 14.0 ± NA | NA | |
| Total | 30.8 ± 22.4 | 22.9 (12.4) | 28.9 (16.1) | 34.2 (27.1) | 0.568 | 9.7 ± 5.5 | NA ± NA | 10.7 ± 4.2 | 9.3 ± 6.2 | 0.275 | |
| Variable | MPR > 80 | MPR < 50 | ||
|---|---|---|---|---|
| MDA | MDG | MDA | MDG | |
| Gender | −1.1 | 0.5 | 0.3 | 0.3 |
| Age | 0.5 | 3.0 | −0.1 | 1.3 |
| Genotype | 2.8 | 1.9 | −2.1 | 0.4 |
| Pancreatic insufficient | 0.0 | 0.1 | −0.1 | 0.1 |
| Diabetes | 1.6 | 0.5 | −1.6 | 0.2 |
| Allergic bronchopulmonary aspergillosis | 0.1 | 0.5 | 0.4 | 0.2 |
| Massive haemoptysis | −1.1 | 0.6 | −2.1 | 0.1 |
| BMI | 0.3 | 1.8 | −0.9 | 1.4 |
| Chronic bronchial infection | 0.1 | 0.3 | 1.9 | 0.1 |
| Pseudomonas aeruginosa | 0.0 | 0.1 | −0.1 | 0.1 |
| methicillin-sensitive Staphylococcus aureus (MSSA) | 1.1 | 2.5 | 0.2 | 0.3 |
| methicillin-resistant Staphylococcus aureus (MRSA) | 0.0 | 0.0 | 0.0 | 0.3 |
| Achromobacter xylosoxidans | 0.0 | 0.3 | −0.2 | 0.2 |
| Burkholderia cepacia | 0.0 | 0.0 | −0.1 | 0.1 |
| Aspergillus sp | 0.0 | 0.0 | 0.0 | 0.0 |
| Haemophilus influenzae | 0.0 | 0.0 | 5.7 | 1.4 |
| Mycobacteria non tuberculous | 0.0 | 0.3 | 0.0 | 0.1 |
| Total exacerbations by intravenous administration | 0.0 | 0.1 | 0.0 | 0.0 |
| Total exacerbations by oral administration | −1.1 | 0.7 | 0.1 | 0.1 |
| Hypertonic saline solution | 1.1 | 0.2 | 3.7 | 1.0 |
| DNAsa | −0.9 | 0.3 | 3.3 | 0.5 |
| Azithromycin | 0.0 | 0.4 | 1.7 | 0.3 |
| Aztreonam solution for inhalation | 1.1 | 0.4 | 0.1 | 0.1 |
| Tobramycin in solution for inhalation | 1.5 | 0.3 | −0.8 | 0.0 |
| Colistin dry powder | −1.6 | 0.6 | 0.6 | 0.1 |
| Colistin in solution for inhalation by INEB | 0.0 | 0.0 | 0.8 | 0.1 |
| Ivacaftor | 0.0 | 0.0 | 0.0 | 0.1 |
| Ivacaftor/Tezacaftor | −0.2 | 3.0 | 1.7 | 0.1 |
| Elexacaftor, Tezacaftor/Ivacaftor | 0.0 | 0.0 | 1.0 | 0.0 |
| Vitamins | 0.0 | 0.0 | 0.9 | 0.2 |
| Intravenous formulation | 0.0 | 0.1 | −0.8 | 0.1 |
| Total FVC% | 1.6 | 2.1 | 2.3 | 1.4 |
| TotalFEV1% | 1.6 | 3.3 | 2.0 | 1.4 |
| Total days of intravenous exacerbations | 0.8 | 1.0 | 0.2 | 0.3 |
| Total days of oral exacerbations | 0.9 | 3.0 | 0.6 | 0.9 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Girón, R.M.; Peláez, A.; Ibáñez, A.; Martínez-Besteiro, E.; Gómez-Punter, R.M.; Martínez-Vergara, A.; Ancochea, J.; Morell, A. Longitudinal Study of Therapeutic Adherence in a Cystic Fibrosis Unit: Identifying Potential Factors Associated with Medication Possession Ratio. Antibiotics 2022, 11, 1637. https://doi.org/10.3390/antibiotics11111637
Girón RM, Peláez A, Ibáñez A, Martínez-Besteiro E, Gómez-Punter RM, Martínez-Vergara A, Ancochea J, Morell A. Longitudinal Study of Therapeutic Adherence in a Cystic Fibrosis Unit: Identifying Potential Factors Associated with Medication Possession Ratio. Antibiotics. 2022; 11(11):1637. https://doi.org/10.3390/antibiotics11111637
Chicago/Turabian StyleGirón, Rosa Mª, Adrián Peláez, Amparo Ibáñez, Elisa Martínez-Besteiro, Rosa Mar Gómez-Punter, Adrián Martínez-Vergara, Julio Ancochea, and Alberto Morell. 2022. "Longitudinal Study of Therapeutic Adherence in a Cystic Fibrosis Unit: Identifying Potential Factors Associated with Medication Possession Ratio" Antibiotics 11, no. 11: 1637. https://doi.org/10.3390/antibiotics11111637
APA StyleGirón, R. M., Peláez, A., Ibáñez, A., Martínez-Besteiro, E., Gómez-Punter, R. M., Martínez-Vergara, A., Ancochea, J., & Morell, A. (2022). Longitudinal Study of Therapeutic Adherence in a Cystic Fibrosis Unit: Identifying Potential Factors Associated with Medication Possession Ratio. Antibiotics, 11(11), 1637. https://doi.org/10.3390/antibiotics11111637






