Occurrence and Quantitative Microbial Risk Assessment of Methicillin-Resistant Staphylococcus aureus (MRSA) in a Sub-Catchment of the Yodo River Basin, Japan
Abstract
1. Introduction
2. Materials and Methods
2.1. Sampling
2.2. Microbials Analysis
2.3. Calculation of Mass Flux and the Hospital Effluent Contributions to the STP Influent and the River Water Environment in the Target Yodo River Basin
2.4. Quantitative Microbial Risk Assessment (QMRA) Analysis
2.5. Statistical Analysis
3. Results
3.1. Occurrence of S. aureus and MRSA in the Yodo River Basin
3.2. Mass Flux-Based Analysis of the Contributions of S. aureus and MRSA from the Hospital Effluent to the STP Influent, and from the STP Effluent to the Rivers
3.3. Quantitative Microbial Risk Assessment of S. aureus and MRSA in the River Environment
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AMRB | antimicrobial-resistant bacteria |
| CFU | colony-forming units |
| DALY | disability-adjusted life year |
| JANIS | Japan nosocomial infections surveillance |
| MRSA | methicillin-resistant Staphylococcus aureus |
| MSSA | methicillin-susceptible Staphylococcus aureus |
| N.D. | not detected |
| QMRA | quantitative microbial risk assessment |
| S. aureus | Staphylococcus aureus |
| SD | standard deviation |
| STP | sewage treatment plant |
| WHO | World Health Organization |
References
- Marston, H.D.; Dixon, D.M.; Knisely, J.M.; Palmore, T.N.; Fauci, A.S. Antimicrobial resistance. JAMA 2016, 316, 1193–1204. [Google Scholar] [CrossRef] [PubMed]
- Mulani, M.S.; Kamble, E.E.; Kumkar, S.N.; Tawre, M.S.; Pardesi, K.R. Emerging strategies to combat ESKAPE pathogens in the era of antimicrobial resistance: A review. Front. Microbiol. 2019, 10, 539. [Google Scholar] [CrossRef] [PubMed]
- Amarasiri, M.; Sano, D.; Suzuki, S. Understanding human health risks caused by antibiotic resistant bacteria (ARB) and antibiotic resistance genes (ARG) in water environments: Current knowledge and questions to be answered. Crit. Rev. Environ. Sci. Technol. 2020, 50, 2016–2059. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Global Action Plan on Antimicrobial Resistance; WHO: Geneva, Switzerland, 2015; pp. 1–19. [Google Scholar]
- World Health Organization (WHO). One Health. Available online: http://www.who.int/features/qa/one-health/en/ (accessed on 30 September 2022).
- van Bruggen, A.H.C.; Goss, E.M.; Havelaar, A.; van Diepeningen, A.D.; Finckh, M.R.; Morris, J.G. One Health—Cycling of diverse microbial communities as a connecting force for soil, plant, animal, human and ecosystem health. Sci. Total Environ. 2019, 664, 927–937. [Google Scholar] [CrossRef] [PubMed]
- Booton, R.D.; Meeyai, A.; Alhusein, N.; Buller, H.; Feil, E.; Lambert, H.; Mongkolsuk, S.; Pitchforth, E.; Reyher, K.K.; Sakcamduang, W.; et al. One health drivers of antibacterial resistance: Quantifying the relative impacts of human, animal and environmental use and transmission. One Health 2021, 12, 100220. [Google Scholar] [CrossRef] [PubMed]
- Domingo-Echaburu, S.; Dávalos, L.M.; Orive, G.; Lertxundi, U. Drug pollution & sustainable development goals. Sci. Total Environ. 2021, 800, 149412. [Google Scholar] [PubMed]
- Nathan, C.; Cars, O. Antibiotic resistance—problems, progress, and prospects. N. Engl. J. Med. 2014, 371, 1761–1763. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Antibiotic-Resistant “Priority Pathogens”—A Catalogue of 12 Families of Bacteria that Pose the Greatest Threat to Human Health. Available online: http://www.who.int/mediacentre/news/releases/2017/bacteria-antibiotics-needed/en/ (accessed on 30 September 2022).
- Cai, L.; Sun, J.; Yao, F.; Yuan, Y.; Zeng, M.; Zhang, Q.; Xie, Q.; Wang, S.; Wang, Z.; Jiao, X. Antimicrobial resistance bacteria and genes detected in hospital sewage provide valuable information in predicting clinical antimicrobial resistance. Sci. Total Environ. 2021, 795, 148815. [Google Scholar] [CrossRef]
- Loudermilk, E.M.; Kotay, S.M.; Barry, K.E.; Parikh, H.I.; Colosi, L.M.; Mathers, A.J. Tracking Klebsiella pneumoniae carbapenemase gene as an indicator of antimicrobial resistance dissemination from a hospital to surface water via a municipal wastewater treatment plant. Water Res. 2022, 213, 118151. [Google Scholar] [CrossRef]
- Arias, C.A.; Murray, B.E. Antibiotic-resistant bugs in the 21st century—A clinical super-challenge. N. Engl. J. Med. 2009, 360, 439–443. [Google Scholar] [CrossRef]
- G Goldstein, R.E.R.; Micallef, S.A.; Gibbs, S.G.; Davis, J.A.; He, X.; George, A.; Kleinfelter, L.M.; Schreiber, N.A.; Mukherjee, S.; Sapkota, A.; et al. Methicillin-resistant Staphylococcus aureus (MRSA) detected at four U.S. Wastewater treatment plants. Environ. Health Perspect. 2012, 120, 1551–1558. [Google Scholar] [CrossRef]
- López, A.; Rodríguez-Chueca, J.; Mosteo, R.; Gómez, J.; Rubio, E.; Goñi, P.; Ormad, M.P. How does urban wastewater treatment affect the microbial quality of treated wastewater? Proc. Safe. Environ. Protec. 2019, 130, 22–30. [Google Scholar] [CrossRef]
- Stefani, S.; Chung, D.R.; Lindsay, J.A.; Friedrich, A.W.; Kearns, A.M.; Westh, H.; MacKenzie, F.M. Meticillin-resistant Staphylococcus aureus (MRSA): Global epidemiology and harmonisation of typing methods. Int. J. Antimicrob. Age. 2012, 39, 273–282. [Google Scholar] [CrossRef] [PubMed]
- Lakhundi, S.; Zhang, K. Methicillin-resistant Staphylococcus aureus: Molecular characterization, evolution, and epidemiology. Clin. Microbiol. Rev. 2018, 31, e00020-18. [Google Scholar] [CrossRef] [PubMed]
- Lee, A.S.; de Lencastre, H.; Garau, J.; Kluytmans, J.; Malhotra-Kumar, S.; Peschel, A.; Harbarth, S. Methicillin-resistant Staphylococcus aureus. Nat. Rev. Dis. Prim. 2018, 4, 18033. [Google Scholar] [CrossRef]
- Garcia, A.B.; Vinuela-Prieto, J.M.; Lopez-Gonzalez, L.; Candel, F.J. Correlation between resistance mechanisms in Staphylococcus aureus and cell wall and septum thickening. Infect. Drug Resist. 2017, 10, 353–356. [Google Scholar] [CrossRef]
- Delaney, J.A.C.; Schneider-Lindner, V.; Brassard, P.; Suissa, S. Mortality after infection with methicillin-resistant Staphylococcus aureus (MRSA) diagnosed in the community. BMC Med. 2008, 6, 2. [Google Scholar] [CrossRef]
- Kumar, A.; Pal, D. Antibiotic resistance and wastewater: Correlation, impact and critical human health challenges. J. Environ. Chem. Eng. 2018, 6, 52–58. [Google Scholar] [CrossRef]
- Matsunaga, N.; Hayakawa, K. Estimating the impact of antimicrobial resistance. Lancet Glob. Health 2018, 6, e934–e935. [Google Scholar] [CrossRef]
- Tsuzuki, S.; Matsunaga, N.; Ohmagari, N. Road to comprehensive estimation of antimicrobial resistance (AMR) disease burden in Japan. Glob. Health Med. 2020, 2, 164–167. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Quantitative Microbial Risk Assessment, Application for Water Safety Management; WHO: Geneva, Switzerland, 2016; pp. 1–187. [Google Scholar]
- Owens, C.E.L.; Angles, M.L.; Cox, P.T.; Byleveld, P.M.; Osborne, N.J.; Rahman, M.B. Implementation of quantitative microbial risk assessment (QMRA) for public drinking water supplies: Systematic review. Water Res. 2020, 174, 115614. [Google Scholar] [CrossRef] [PubMed]
- Dada, A.C.; Gyawali, P. Quantitative microbial risk assessment (QMRA) of occupational exposure to SARS-CoV-2 in wastewater treatment plants. Sci. Total Environ. 2021, 763, 142989. [Google Scholar] [CrossRef] [PubMed]
- Schoen, M.E.; Jahne, M.A.; Garland, J.; Ramirez, L.; Lopatkin, A.J.; Hamilton, K.A. Quantitative microbial risk assessment of antimicrobial resistant and susceptible Staphylococcus aureus in reclaimed wastewaters. Environ. Sci. Technol. 2021, 55, 15246–15255. [Google Scholar] [CrossRef] [PubMed]
- Tyagi, N.; Gurian, P.L.; Kumar, A. Using qmra to understand possible exposure risks of SARS-CoV-2 from the water environment. Environ. Sci. Pollut. Res. 2022, 29, 7240–7253. [Google Scholar] [CrossRef] [PubMed]
- Lake Biwa-Yodo River Water Quality Preservation Organization, Japan; Lake Biwa-Yodo River Water Quality Preservation Organization (BYQ). Report on Water Environment in Biwa Lake-Yodo River System 2019; BYQ: Osaka, Japan, 2021; pp. 1–90. (In Japanese) [Google Scholar]
- Ministry of Health Labour and Welfare (Japan). Vital Statistics in Japan; Ministry of Health Labour and Welfare: Tokyo, Japan, 2021; pp. 1–102. [Google Scholar]
- Azuma, T.; Otomo, K.; Kunitou, M.; Shimizu, M.; Hosomaru, K.; Mikata, S.; Ishida, M.; Hisamatsu, K.; Yunoki, A.; Mino, Y.; et al. Environmental fate of pharmaceutical compounds and antimicrobial-resistant bacteria in hospital effluents, and contributions to pollutant loads in the surface waters in Japan. Sci. Total Environ. 2019, 657, 476–484. [Google Scholar] [CrossRef] [PubMed]
- Azuma, T.; Hayashi, T. Effects of natural sunlight on antimicrobial-resistant bacteria (AMRB) and antimicrobial-susceptible bacteria (AMSB) in wastewater and river water. Sci. Total Environ. 2021, 766, 142568. [Google Scholar] [CrossRef]
- Japan Meteorological Agency 2021. Weather Statistics. Available online: http://www.jma.go.jp/jma/index.html (accessed on 30 September 2022).
- Zheng, J.; Su, C.; Zhou, J.; Xu, L.; Qian, Y.; Chen, H. Effects and mechanisms of ultraviolet, chlorination, and ozone disinfection on antibiotic resistance genes in secondary effluents of municipal wastewater treatment plants. Chem. Eng. 2017, 317, 309–316. [Google Scholar] [CrossRef]
- Dunkin, N.; Weng, S.; Coulter, C.G.; Jacangelo, J.G.; Schwab, K.J. Impacts of virus processing on human norovirus GI and GII persistence during disinfection of municipal secondary wastewater effluent. Water Res. 2018, 134, 1–12. [Google Scholar] [CrossRef]
- Lamba, M.; Graham, D.W.; Ahammad, S.Z. Hospital wastewater releases of carbapenem-resistance pathogens and genes in urban India. Environ. Sci. Technol. 2017, 51, 13906–13912. [Google Scholar] [CrossRef]
- Haller, L.; Chen, H.; Ng, C.; Le, T.H.; Koh, T.H.; Barkham, T.; Sobsey, M.; Gin, K.Y.H. Occurrence and characteristics of extended-spectrum β-lactamase- and carbapenemase- producing bacteria from hospital effluents in Singapore. Sci. Total Environ. 2018, 615, 1119–1125. [Google Scholar] [CrossRef]
- Serna-Galvis, E.A.; Vélez-Peña, E.; Osorio-Vargas, P.; Jiménez, J.N.; Salazar-Ospina, L.; Guaca-González, Y.M.; Torres-Palma, R.A. Inactivation of carbapenem-resistant Klebsiella pneumoniae by photo-fenton: Residual effect, gene evolution and modifications with citric acid and persulfate. Water Res. 2019, 161, 354–363. [Google Scholar] [CrossRef] [PubMed]
- Sauter, D.; Stange, C.; Schumacher, V.; Tiehm, A.; Gnirss, R.; Wintgens, T. Impact of ozonation and biological post-treatment of municipal wastewater on microbiological quality parameters. Environ. Sci. Water Res. Technol. 2021, 7, 1643–1656. [Google Scholar] [CrossRef]
- Azuma, T.; Uchiyama, T.; Zhang, D.; Usui, M.; Hayashi, T. Distribution and characteristics of carbapenem-resistant and extended-spectrum β-lactamase (ESBL) producing Escherichia coli in hospital effluents, sewage treatment plants, and river water in an urban area of Japan. Sci. Total Environ. 2022, 839, 156232. [Google Scholar] [CrossRef] [PubMed]
- Sib, E.; Voigt, A.M.; Wilbring, G.; Schreiber, C.; Faerber, H.A.; Skutlarek, D.; Parcina, M.; Mahn, R.; Wolf, D.; Brossart, P.; et al. Antibiotic resistant bacteria and resistance genes in biofilms in clinical wastewater networks. Int. J. Hyg. Environ. Health 2019, 222, 655–662. [Google Scholar] [CrossRef] [PubMed]
- Schreiber, C.; Zacharias, N.; Essert, S.M.; Wasser, F.; Müller, H.; Sib, E.; Precht, T.; Parcina, M.; Bierbaum, G.; Schmithausen, R.M.; et al. Clinically relevant antibiotic-resistant bacteria in aquatic environments—An optimized culture-based approach. Sci. Total Environ. 2021, 750, 142265. [Google Scholar] [CrossRef]
- Manufacturer’s Protocol for ChromID® Chromogenic Media. Available online: http://www.biomerieux.fr/diagnostic-clinique/milieux-de-culture (accessed on 30 September 2022).
- Japan Sewage Works Association. Statistics of Sewerage (In Japanese); Japan Sewage Works Association: Tokyo, Japan, 2021. [Google Scholar]
- Oliveira, T.S.; Murphy, M.; Mendola, N.; Wong, V.; Carlson, D.; Waring, L. Characterization of pharmaceuticals and personal care products in hospital effluent and waste water influent/effluent by direct-injection LC-MS-MS. Sci. Total Environ. 2015, 518–519, 459–478. [Google Scholar] [CrossRef]
- Yao, L.; Chen, Z.Y.; Dou, W.Y.; Yao, Z.K.; Duan, X.C.; Chen, Z.F.; Zhang, L.J.; Nong, Y.J.; Zhao, J.L.; Ying, G.-G. Occurrence, removal and mass loads of antiviral drugs in seven wastewater treatment plants with various treatment processes. Water Res. 2021, 207, 117803. [Google Scholar] [CrossRef]
- Mishra, S.; Kneis, D.; Berendonk, T.U.; Aubeneau, A. Optimum positioning of wastewater treatment plants in a river network: A model-based approach to minimize microbial pollution. Sci. Total Environ. 2019, 691, 1310–1319. [Google Scholar] [CrossRef]
- Cheng, X.; Xu, J.; Smith, G.; Zhang, Y. Metagenomic insights into dissemination of antibiotic resistance across bacterial genera in wastewater treatment. Chemosphere 2021, 271, 129563. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention (CDC). Guidelines for Environmental Infection Control in Health-Care Facilities Recommendations of CDC and the Healthcare Infection Control Practices Advisory Committee (HICPAC); Centers for Disease Control and Prevention (CDC): Atlanta, GA, USA, 2013; pp. 1–241. [Google Scholar]
- Turner, N.A.; Sharma-Kuinkel, B.K.; Maskarinec, S.A.; Eichenberger, E.M.; Shah, P.P.; Carugati, M.; Holland, T.L.; Fowler, V.G. Methicillin-resistant Staphylococcus aureus: An overview of basic and clinical research. Nat. Rev. Microbiol. 2019, 17, 203–218. [Google Scholar] [CrossRef]
- Ojer-Usoz, E.; González, D.; García-Jalón, I.; Vitas, A.I. High dissemination of extended-spectrum β-lactamase-producing enterobacteriaceae in effluents from wastewater treatment plants. Water Res. 2014, 56, 37–47. [Google Scholar] [CrossRef] [PubMed]
- Hrenovic, J.; Ivankovic, T.; Ivekovic, D.; Repec, S.; Stipanicev, D.; Ganjto, M. The fate of carbapenem-resistant bacteria in a wastewater treatment plant. Water Res. 2017, 126, 232–239. [Google Scholar] [CrossRef] [PubMed]
- Azuma, T.; Hayashi, T. On-site chlorination responsible for effective disinfection of wastewater from hospital. Sci. Total Environ. 2021, 776, 145951. [Google Scholar] [CrossRef]
- Rizzo, L.; Manaia, C.; Merlin, C.; Schwartz, T.; Dagot, C.; Ploy, M.C.; Michael, I.; Fatta-Kassinos, D. Urban wastewater treatment plants as hotspots for antibiotic resistant bacteria and genes spread into the environment: A review. Sci. Total Environ. 2013, 447, 345–360. [Google Scholar] [CrossRef]
- Tiedeken, E.J.; Tahar, A.; McHugh, B.; Rowan, N.J. Monitoring, sources, receptors, and control measures for three european union watch list substances of emerging concern in receiving waters—A 20 year systematic review. Sci. Total Environ. 2017, 574, 1140–1163. [Google Scholar] [CrossRef] [PubMed]
- Gwenzi, W.; Musiyiwa, K.; Mangori, L. Sources, behaviour and health risks of antimicrobial resistance genes in wastewaters: A hotspot reservoir. J. Environ. Chem. Eng. 2020, 8, 102220. [Google Scholar] [CrossRef]
- Haas, C.N. Acceptable microbial risk. J. AWWA 1996, 88, 8. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Guidelines for Drinking-Water Quality, 4th ed.; WHO: Geneva, Switzerland, 2011; pp. 1–541. [Google Scholar]
- World Health Organization (WHO). Guidelines for Drinking-Water Quality, 4th ed.; Incorporating the 1st Addendum; WHO: Geneva, Switzerland, 2017; pp. 1–541. [Google Scholar]
- Chen, Y.Q.; Chen, C.; Zhang, X.J.; Zheng, Q.; Liu, Y.Y. Inactivation of resistant mycobacteria mucogenicum in water: Chlorine resistance and mechanism analysis. Biomed. Environ. Sci. 2012, 25, 230. [Google Scholar]
- Ministry of Health Labour and Welfare (Japan). Japan Nosocomial Infections Surveillance (JANIS), Nosocomial Infections Surveillance for Drug-Resistant Bacteria. Available online: https://janis.mhlw.go.jp/english/index.asp (accessed on 30 September 2022).
- Chau, K.K.; Barker, L.; Budgell, E.P.; Vihta, K.D.; Sims, N.; Kasprzyk-Hordern, B.; Harriss, E.; Crook, D.W.; Read, D.S.; Walker, A.S.; et al. Systematic review of wastewater surveillance of antimicrobial resistance in human populations. Environ. Int. 2022, 162, 107171. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention (CDC). Antibiotic resistance threats in the United States, 2019; Centers for Disease Control and Prevention (CDC): Atlanta, GA, USA, 2019; pp. 1–139. [Google Scholar]
- Choi, Y.; He, H.; Dodd, M.C.; Lee, Y. Degradation kinetics of antibiotic resistance gene meca of methicillin-resistant Staphylococcus aureus (MRSA) during water disinfection with chlorine, ozone, and ultraviolet light. Environ. Sci. Technol. 2021, 55, 2541–2552. [Google Scholar] [CrossRef]
- Wan, M.T.; Chou, C.C. Spreading of β-lactam resistance gene (mecA) and methicillin-resistant Staphylococcus aureus through municipal and swine slaughterhouse wastewaters. Water Res. 2014, 64, 288–295. [Google Scholar] [CrossRef] [PubMed]
- Weissbrodt, D.; Kovalova, L.; Ort, C.; Pazhepurackel, V.; Moser, R.; Hollender, J.; Siegrist, H.; McArdell, C.S. Mass flows of X-ray contrast media and cytostatics in hospital wastewater. Environ. Sci. Technol. 2009, 43, 4810–4817. [Google Scholar] [CrossRef] [PubMed]
- Santos, L.H.M.L.M.; Gros, M.; Rodriguez-Mozaz, S.; Delerue-Matos, C.; Pena, A.; Barceló, D.; Montenegro, M.C.B.S.M. Contribution of hospital effluents to the load of pharmaceuticals in urban wastewaters: Identification of ecologically relevant pharmaceuticals. Sci. Total Environ. 2013, 461–462, 302–316. [Google Scholar] [CrossRef] [PubMed]
- Aydin, S.; Aydin, M.E.; Ulvi, A.; Kilic, H. Antibiotics in hospital effluents: Occurrence, contribution to urban wastewater, removal in a wastewater treatment plant, and environmental risk assessment. Environ. Sci. Pollut. Res. 2019, 26, 544–558. [Google Scholar] [CrossRef] [PubMed]
- Afsa, S.; Hamden, K.; Lara Martin, P.A.; Mansour, H.B. Occurrence of 40 pharmaceutically active compounds in hospital and urban wastewaters and their contribution to mahdia coastal seawater contamination. Environ. Sci. Pollut. Res. 2020, 27, 1941–1955. [Google Scholar] [CrossRef]
- ter Laak, T.L.; van der Aa, M.; Houtman, C.J.; Stoks, P.G.; van Wezel, A.P. Relating environmental concentrations of pharmaceuticals to consumption: A mass balance approach for the river rhine. Environ. Int. 2010, 36, 403–409. [Google Scholar] [CrossRef]
- Azuma, T.; Nakada, N.; Yamashita, N.; Tanaka, H. Mass balance of anti-influenza drugs discharged into the yodo river system, Japan, under an influenza outbreak. Chemosphere 2013, 93, 1672–1677. [Google Scholar] [CrossRef]
- Yuan, X.; Hu, J.; Li, S.; Yu, M. Occurrence, fate, and mass balance of selected pharmaceutical and personal care products (PPCPs) in an urbanized river. Environ. Pollut. 2020, 266, 115340. [Google Scholar] [CrossRef]
- Adachi, F.; Sekizuka, T.; Yamato, M.; Fukuoka, K.; Yamaguchi, N.; Kuroda, M.; Kawahara, R. Characterization of fri carbapenemase-producing enterobacter spp. Isolated from a hospital and the environment in osaka, Japan. J. Antimicrob. Chemother. 2021, 76, 3061–3062. [Google Scholar] [CrossRef]
- Mackuľak, T.; Cverenkárová, K.; Vojs Staňová, A.; Fehér, M.; Tamáš, M.; Škulcová, A.B.; Gál, M.; Naumowicz, M.; Špalková, V.; Bírošová, L. Hospital wastewater—Source of specific micropollutants, antibiotic-resistant microorganisms, viruses, and their elimination. Antibiotics 2021, 10, 1070. [Google Scholar] [CrossRef]
- Verlicchi, P. Trends, new insights and perspectives in the treatment of hospital effluents. Curr. Opin. Environ. Sci. Health 2021, 19, 100217. [Google Scholar] [CrossRef] [PubMed]
- Baba, H.; Nishiyama, M.; Watanabe, T.; Kanamori, H. Review of antimicrobial resistance in wastewater in Japan: Current challenges and future perspectives. Antibiotics 2022, 11, 849. [Google Scholar] [CrossRef] [PubMed]
- Sekizuka, T.; Itokawa, K.; Tanaka, R.; Hashino, M.; Yatsu, K.; Kuroda, M. Metagenomic analysis of urban wastewater treatment plant effluents in Tokyo. Infect. Drug Resist. 2022, 15, 4763–4777. [Google Scholar] [CrossRef] [PubMed]
- Sekizuka, T.; Tanaka, R.; Hashino, M.; Yatsu, K.; Kuroda, M. Comprehensive genome and plasmidome analysis of antimicrobial resistant bacteria in wastewater treatment plant effluent of Tokyo. Antibiotics 2022, 11, 1283. [Google Scholar] [CrossRef]
- Auguet, O.; Pijuan, M.; Borrego, C.M.; Rodriguez-Mozaz, S.; Triadó-Margarit, X.; Giustina, S.V.D.; Gutierrez, O. Sewers as potential reservoirs of antibiotic resistance. Sci. Total Environ. 2017, 605–606, 1047–1054. [Google Scholar] [CrossRef]
- He, P.; Zhou, Y.; Shao, L.; Huang, J.; Yang, Z.; Lü, F. The discrepant mobility of antibiotic resistant genes: Evidence from their spatial distribution in sewage sludge flocs. Sci. Total Environ. 2019, 697, 134176. [Google Scholar] [CrossRef]
- Zhang, X.; Li, R. Variation and distribution of antibiotic resistance genes and their potential hosts in microbial electrolysis cells treating sewage sludge. Biores. Technol. 2020, 315, 123838. [Google Scholar] [CrossRef]
- Zhang, Y.; Pei, M.; Zhang, B.; He, Y.; Zhong, Y. Changes of antibiotic resistance genes and bacterial communities in the advanced biological wastewater treatment system under low selective pressure of tetracycline. Water Res. 2021, 207, 117834. [Google Scholar] [CrossRef]
- He, H.; Zhou, P.; Shimabuku, K.K.; Fang, X.; Li, S.; Lee, Y.; Dodd, M.C. Degradation and deactivation of bacterial antibiotic resistance genes during exposure to free chlorine, monochloramine, chlorine dioxide, ozone, ultraviolet light, and hydroxyl radical. Environ. Sci. Technol. 2019, 53, 2013–2026. [Google Scholar] [CrossRef]
- Azuma, T.; Usui, M.; Hayashi, T. Inactivation of antibiotic-resistant bacteria in wastewater by ozone-based advanced water treatment processes. Antibiotics 2022, 11, 210. [Google Scholar] [CrossRef]
- Azuma, T.; Katagiri, M.; Sekizuka, T.; Kuroda, M.; Watanabe, M. Inactivation of bacteria and residual antimicrobials in hospital wastewater by ozone treatment. Antibiotics 2022, 11, 862. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Zhang, Y.; Wu, J.; Wang, J.; Cai, Y. Fate of antibiotic resistance genes in reclaimed water reuse system with integrated membrane process. J. Hazard. Mater. 2020, 382, 121025. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Ma, X.; Liu, Y.; Yi, X.; Du, G.; Li, J. Fate of antibiotics, antibiotic-resistant bacteria, and cell-free antibiotic-resistant genes in full-scale membrane bioreactor wastewater treatment plants. Biores. Technol. 2020, 302, 122825. [Google Scholar] [CrossRef] [PubMed]
- Nihemaiti, M.; Yoon, Y.; He, H.; Dodd, M.C.; Croué, J.P.; Lee, Y. Degradation and deactivation of a plasmid-encoded extracellular antibiotic resistance gene during separate and combined exposures to UV254 and radicals. Water Res. 2020, 182, 115921. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.; Guo, X.; Li, S.; Li, F. A review of the bioelectrochemical system as an emerging versatile technology for reduction of antibiotic resistance genes. Environ. Int. 2021, 156, 106689. [Google Scholar] [CrossRef]
- Liu, Y.; Zhang, S.; Fang, H.; Wang, Q.; Jiang, S.; Zhang, C.; Qiu, P. Inactivation of antibiotic resistant bacterium Escherichia coli by electrochemical disinfection on molybdenum carbide electrode. Chemosphere 2022, 287, 132398. [Google Scholar] [CrossRef]
- Campo, N.; De Flora, C.; Maffettone, R.; Manoli, K.; Sarathy, S.; Santoro, D.; Gonzalez-Olmos, R.; Auset, M. Inactivation kinetics of antibiotic resistant Escherichia coli in secondary wastewater effluents by peracetic and performic acids. Water Res. 2020, 169, 115227. [Google Scholar] [CrossRef]
- Ping, Q.; Yan, T.; Wang, L.; Li, Y.; Lin, Y. Insight into using a novel ultraviolet/peracetic acid combination disinfection process to simultaneously remove antibiotics and antibiotic resistance genes in wastewater: Mechanism and comparison with conventional processes. Water Res. 2022, 210, 118019. [Google Scholar] [CrossRef]
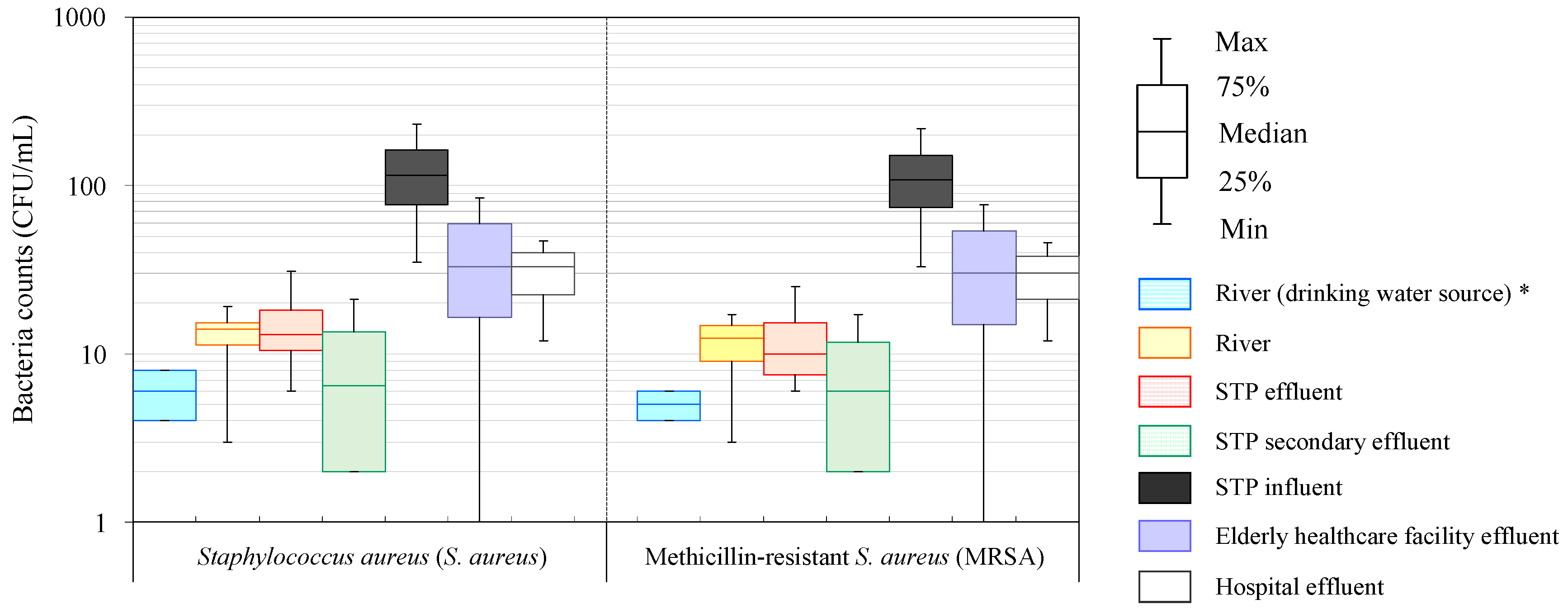

| Bacteria | Sample Type | Bacteria Counts (CFU/mL) | |||
|---|---|---|---|---|---|
| Mean (SD) | Median | Maximum | Minimum | ||
| Staphylococcus aureus (S. aureus) | River (drinking water source) * | 6 (2) | 6 | 8 | 4 |
| River | 13 (7) | 14 | 19 | 3 | |
| STP effluent | 16 (11) | 13 | 31 | 6 | |
| STP secondary effluent | 9 (9) | 7 | 21 | 2 | |
| STP influent | 124 (83) | 115 | 232 | 35 | |
| Elderly healthcare facility effluent | 39 (43) | 33 | 85 | N.D. | |
| Hospital effluent | 31 (18) | 33 | 47 | 12 | |
| Methicillin-resistant Staphylococcus aureus (MRSA) | River (drinking water source) * | 5 (1) | 5 | 6 | 4 |
| River | 11 (6) | 13 | 17 | 3 | |
| STP effluent | 13 (9) | 10 | 25 | 6 | |
| STP secondary effluent | 8 (7) | 6 | 17 | 2 | |
| STP influent | 117 (78) | 108 | 219 | 33 | |
| Elderly healthcare facility effluent | 36 (39) | 30 | 77 | N.D. | |
| Hospital effluent | 29 (17) | 30 | 46 | 12 | |
| Sample | Mean Mass Flux (CFU/day) | |
|---|---|---|
| Staphylococcus aureus (S. aureus) | Methicillin-Resistant Staphylococcus aureus (MRSA) | |
| River (drinking water source) | 1.1 × 1014 | 8.9 × 1013 |
| River | 3.5 × 1013 | 3.2 × 1013 |
| STP effluent | 3.0 × 1012 | 2.4 × 1012 |
| STP secondary effluent | 1.7 × 1012 | 1.5 × 1012 |
| STP influent | 2.4 × 1013 | 2.2 × 1013 |
| Hospital effluent | 1.4 × 1010 | 1.3 × 1010 |
| Bacteria | Contribution of Hospital Effluent (% of Total STP Influent) | Contribution of STP Effluent (% of Total River Water) | Contribution of STP Effluent (% of Total River Water (Drinking Water Source)) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Mean | Maximum | Minimum | Mean | Maximum | Minimum | Mean | Maximum | Minimum | |
| Staphylococcus aureus (S. aureus) | 0.1 | 0.2 | <0.1 | 14.8 | 24.7 | 4.9 | 4.0 | 6.8 | 2.1 |
| Methicillin-resistant Staphylococcus aureus (MRSA) | 0.1 | 0.2 | <0.1 | 14.4 | 25.4 | 3.9 | 4.0 | 7.4 | 2.1 |
| Risk Unit | Percentile | Total | Abundance of Methicillin-Resistant Staphylococcus aureus (MRSA) |
|---|---|---|---|
| Infection (ppy) | 50th | 4.6 × 10−3 | 98.9% |
| 95th | 4.8 × 10−3 | 98.9% | |
| DALYs (ppy) | 50th | 7.5 × 10−4 | 99.5% |
| 95th | 7.8 × 10−4 | 99.5% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Azuma, T.; Murakami, M.; Sonoda, Y.; Ozaki, A.; Hayashi, T. Occurrence and Quantitative Microbial Risk Assessment of Methicillin-Resistant Staphylococcus aureus (MRSA) in a Sub-Catchment of the Yodo River Basin, Japan. Antibiotics 2022, 11, 1355. https://doi.org/10.3390/antibiotics11101355
Azuma T, Murakami M, Sonoda Y, Ozaki A, Hayashi T. Occurrence and Quantitative Microbial Risk Assessment of Methicillin-Resistant Staphylococcus aureus (MRSA) in a Sub-Catchment of the Yodo River Basin, Japan. Antibiotics. 2022; 11(10):1355. https://doi.org/10.3390/antibiotics11101355
Chicago/Turabian StyleAzuma, Takashi, Michio Murakami, Yuki Sonoda, Akihiko Ozaki, and Tetsuya Hayashi. 2022. "Occurrence and Quantitative Microbial Risk Assessment of Methicillin-Resistant Staphylococcus aureus (MRSA) in a Sub-Catchment of the Yodo River Basin, Japan" Antibiotics 11, no. 10: 1355. https://doi.org/10.3390/antibiotics11101355
APA StyleAzuma, T., Murakami, M., Sonoda, Y., Ozaki, A., & Hayashi, T. (2022). Occurrence and Quantitative Microbial Risk Assessment of Methicillin-Resistant Staphylococcus aureus (MRSA) in a Sub-Catchment of the Yodo River Basin, Japan. Antibiotics, 11(10), 1355. https://doi.org/10.3390/antibiotics11101355






