Is Antibiotic Prophylaxis Necessary before Dental Implant Procedures in Patients with Orthopaedic Prostheses? A Systematic Review
Abstract
1. Introduction
2. Materials and Methods
2.1. Focused Question
2.2. Eligibility Criteria
2.2.1. Inclusion Criteria
2.2.2. Exclusion Criteria
2.3. Information Sources and Search Strategy
2.4. Study Records
2.5. Risk of Bias
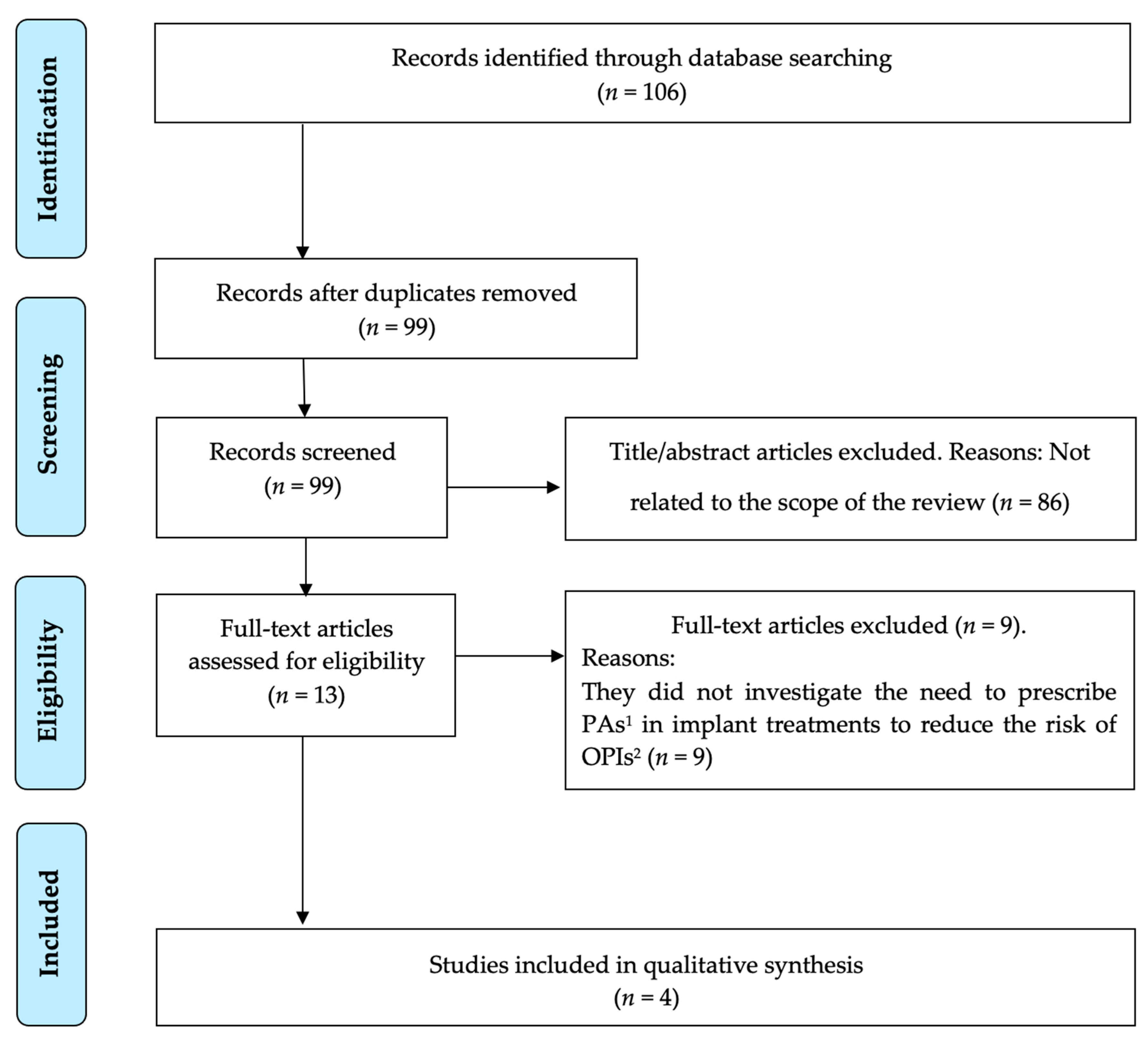
3. Results
3.1. Study Selection
3.2. Study Characteristics
3.3. Risk of Bias within Studies
4. Discussion
Strengths and Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bloom, D.E. 7 Billion and Counting. Science 2011, 333, 562–569. [Google Scholar] [CrossRef] [PubMed]
- Christensen, K.; Doblhammer, G.; Rau, R.; Vaupel, J.W. Ageing populations: The challenges ahead. Lancet 2009, 374, 1196–1208. [Google Scholar] [CrossRef]
- Zimmerli, W.; Sendi, P. Antibiotics for Prevention of Periprosthetic Joint Infection Following Dentistry: Time to Focus on data. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2010, 50, 17–19. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lübbeke, A.J.; Silman, A.J.; Barea, C.; Prieto-Alhambra, D.; Carr, A.J. Mapping existing hip and knee replacement registries in Europe. Health Policy 2018, 122, 548–557. [Google Scholar] [CrossRef] [PubMed]
- Watters, W., 3rd; Rethman, M.P.; Hanson, N.B.; Abt, E.; Anderson, P.A.; Carroll, K.C.; Futrell, H.C.; Garvin, K.; Glenn, S.O.; Hellstein, J.; et al. Prevention of Orthopaedic Implant Infection in Patients Undergoing Dental Procedures. J. Am. Acad. Orthop. Surg. 2013, 21, 180–189. [Google Scholar] [CrossRef]
- Kassebaum, N.J.; Smith, A.G.C.; Bernabé, E.; Fleming, T.D.; Reynolds, A.E.; Vos, T.; Murray, C.J.L.; Marcenes, W.; GBD 2015 Oral Health Collaborators. Global, Regional, and National Prevalence, Incidence, and Disability-Adjusted Life Years for Oral Conditions for 195 Countries, 1990–2015: A Systematic Analysis for the Global Burden of Diseases, Injuries, and Risk Factors. J. Dent. Res. 2017, 96, 380–387. [Google Scholar] [CrossRef]
- Rademacher, W.M.H.; Walenkamp, G.H.I.M.; Moojen, D.J.F.; Hendriks, J.G.E.; Goedendorp, T.A.; Rozema, F.R. Antibiotic prophylaxis is not indicated prior to dental procedures for prevention of periprosthetic joint infections. Acta Orthop. 2017, 88, 568–574. [Google Scholar] [CrossRef]
- Skaar, D.D.; Park, T.; Swiontkowski, M.F.; Kuntz, K.M. Cost-effectiveness of antibiotic prophylaxis for dental patients with prosthetic joints: Comparisons of antibiotic regimens for patients with total hip arthroplasty. J. Am. Dent. Assoc. 2015, 146, 830–839. [Google Scholar] [CrossRef]
- Moreira, A.I.; Mendes, L.; Pereira, J.A. Is there scientific evidence to support antibiotic prophylaxis in patients with periodontal disease as a means to decrease the risk of prosthetic joint infections? A systematic review. Int. Orthop. 2020, 44, 231–236. [Google Scholar] [CrossRef]
- Zimmerli, W.; Zak, O.; Vosbeck, K. Experimental Hematogenous Infection of Subcutaneously Implanted Foreign Bodies. Scand. J. Infect. Dis. 1985, 17, 303–310. [Google Scholar] [CrossRef]
- Blomgren, G. Hematogenous Infection of Total Joint Replacement: An Experimental Study in the Rabbit. Acta Orthop. Scand. Suppl. 1981, 187, 1–64. [Google Scholar] [CrossRef]
- Colterjohn, T.; de Beer, J.; Petruccelli, D.; Zabtia, N.; Winemaker, M. Antibiotic Prophylaxis for Dental Procedures at Risk of Causing Bacteremia Among Post-Total Joint Arthroplasty Patients: A Survey of Canadian Orthopaedic Surgeons and Dental Surgeons. J. Arthroplast. 2014, 29, 1091–1097. [Google Scholar] [CrossRef]
- McNally, C.M.; Visvanathan, R.; Liberali, S.; Adams, R.J. Antibiotic prophylaxis for dental treatment after prosthetic joint replacement: Exploring the orthopaedic surgeon’s opinion. Arthroplast. Today 2016, 2, 123–126. [Google Scholar] [CrossRef] [PubMed]
- Alam, M.; Bastakoti, B. Therapeutic Guidelines: Antibiotic. Version 15. Aust. Prescr. 2015, 38, 137. [Google Scholar] [CrossRef]
- Committee, J.F. British National Formulary 67; BMJ Group and Pharmaceutical Press: London, UK, 2014. [Google Scholar]
- Canadian Dental Association. New CDA position statement on dental patients with orthopedic implants. J. Can. Dent. Assoc. 2013, 79, d126. [Google Scholar]
- New Zealand Dental Association. Code of Practice: Antibiotic Prophylaxis for Patients with Prosthetic Joint Replacements Undergoing Dental Treatment; New Zealand Dental Association Inc.: Auckland, New Zealand, 2018. [Google Scholar]
- Salgado-Peralvo, A.-O.; Velasco-Ortega, E.; Peña-Cardelles, J.-F.; Kewalramani, N.; Monsalve-Guil, L.; Jiménez-Guerra, Á.; Ortiz-García, I.; Moreno-Muñoz, J.; Núñez-Márquez, E.; Cabanillas-Balsera, D. Clinical Practice Guideline for the Prescription of Preventive Antibiotics in Oral Implantology; Sociedad Española de Implantes: Madrid, Spain, 2021. [Google Scholar]
- Cassini, A.; Hogberg, L.D.; Plachouras, D.; Quattrocchi, A.; Hoxha, A.; Simonsen, G.S.; Colomb-Cotinat, M.; Kretzschmar, M.E.; Devleesschauwer, B.; Cecchini, M.; et al. Attributable deaths and disability-adjusted life-years caused by infections with antibiotic-resistant bacteria in the EU and the European Economic Area in 2015: A population-level modelling analysis. Lancet Infect. Dis. 2019, 19, 56–66. [Google Scholar] [CrossRef]
- O’Neill, J. Tackling Drug-Resistant Infections Globally: Final Report and Recommendations. Review on Antimicrobial Resistance; Government of the United Kingdom: London, UK, 2016. [Google Scholar]
- Salgado-Peralvo, A.-O.; Mateos-Moreno, M.-V.; Velasco-Ortega, E.; Peña-Cardelles, J.-F.; Kewalramani, N. Preventive antibiotic therapy in bone augmentation procedures in oral implantology: A systematic review. J. Stomatol. Oral Maxillofac. Surg. 2021, 22. [Google Scholar] [CrossRef]
- Rodríguez-Sánchez, F.; Arteagoitia, I.; Teughels, W.; Rodríguez-Andrés, C.; Quirynen, M. Antibiotic dosage prescribed in oral implant surgery: A meta-analysis of cross-sectional surveys. PLoS ONE 2020, 15, e0236981. [Google Scholar] [CrossRef]
- Romandini, M.; De Tullio, I.; Congedi, F.; Kalemaj, Z.; D’Ambrosio, M.; Laforí, A.; Quaranta, C.; Buti, J.; Perfetti, G. Antibiotic prophylaxis at dental implant placement: Which is the best protocol? A systematic review and network meta-analysis. J. Clin. Periodontol. 2019, 46, 382–395. [Google Scholar] [CrossRef] [PubMed]
- Salgado-Peralvo, A.-O.; Peña-Cardelles, J.-F.; Kewalramani, N.; Ortiz-García, I.; Jiménez-Guerra, Á.; Uribarri, A.; Velasco-Ortega, E.; Moreno-Muñoz, J.; Núñez-Márquez, E.; Monsalve-Guil, L. Is Penicillin Allergy a Risk Factor for Early Dental Implant Failure? A Systematic Review. Antibiotics 2021, 10, 1227. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; The PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA Statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef]
- Joanna Briggs Institute. Checklist for Systematic Reviews and Research Syntheses; The Joanna Briggs Institute: Adelaide, Australia, 2017. [Google Scholar]
- Sollecito, T.P.; Abt, E.; Lockhart, P.B.; Truelove, E.; Paumier, T.M.; Tracy, S.L.; Tampi, M.; Beltrán-Aguilar, E.D.; Frantsve-Hawley, J. The use of prophylactic antibiotics prior to dental procedures in patients with prosthetic joints: Evidence-based clinical practice guideline for dental practitioners—a report of the American Dental Association Council on Scientific Affairs. J. Am. Dent. Assoc. 2015, 146, 11–16.e8. [Google Scholar] [CrossRef] [PubMed]
- Canadian Agency for Drugs and Technologies in Health. Antibiotic Prophylaxis in Patients with Orthopedic Implants Undergoing Dental Procedures: A Review of Clinical Effectiveness, Safety, and Guidelines; Canadian Agency for Drugs and Technologies in Health: Kanata, ON, Canda, 2016. [Google Scholar]
- Selik, R.M.; Mokotoff, E.D.; Branson, B.; Owen, S.M.; Whitmore, S.; Hall, H.I. Control and Prevention Revised surveillance case definition for HIV infection—United States, 2014. MMWR. Recomm. Rep. 2014, 63, 1–10. [Google Scholar]
- Berbari, E.F.; Osmon, D.R.; Carr, A.; Hanssen, A.D.; Baddour, L.M.; Greene, D.; Kupp, L.I.; Baughan, L.W.; Harmsen, W.S.; Mandrekar, J.N.; et al. Dental Procedures as Risk Factors for Prosthetic Hip or Knee Infection: A Hospital-Based Prospective Case-Control Study. Clin. Infect. Dis. an Off. Publ. Infect. Dis. Soc. Am. 2010, 50, 8–16. [Google Scholar] [CrossRef]
- Canadian Dental Association; Canadian Orthopedic Association; Association of Medical Microbiology and Infectious Disease. Dental Patients with Total Joint Replacement; The Canadian Dental Association: Ottawa, ON, Canda.
- American Dental Association; American Academy of Orthopaedic Surgeons Antibiotic prophylaxis for dental patients with total joint replacements. J. Am. Dent. Assoc. 2003, 134, 895–899. [CrossRef] [PubMed]
- American Academy of Orthopaedic Surgeons. Information Statement 1014: Antibiotic Prophylaxis for Bacteriemia in Patients with Joint Replacements; American Academy of Orthopaedic Surgeons: Rosemont, IL, USA, 2009. [Google Scholar]
- Skaar, D.D.; O’Connor, H.; Hodges, J.S.; Michalowicz, B.S. Dental procedures and subsequent prosthetic joint infections: Findings from the Medicare Current Beneficiary Survey. J. Am. Dent. Assoc. 2011, 142, 1343–1351. [Google Scholar] [CrossRef]
- Stévignon, T.; Mouton, A.; Meyssonnier, V.; Kerroumi, Y.; Yazigi, A.; Aubert, T.; Lhotellier, L.; Le Strat, V.; Passeron, D.; Graff, W.; et al. Haematogenous prosthetic knee infections: Prospective cohort study of 58 patients. Orthop. Traumatol. Surg. Res. 2019, 105, 647–651. [Google Scholar] [CrossRef]
- Barrere, S.; Reina, N.; Peters, O.A.; Rapp, L.; Vergnes, J.-N.; Maret, D. Dental assessment prior to orthopedic surgery: A systematic review. Orthop. Traumatol. Surg. Res. 2019, 105, 761–772. [Google Scholar] [CrossRef]
- American Dental Association. Management of Patients with Prosthetic Joints Undergoing Dental Procedures; American Dental Association: Chicago, IL, USA.
- Kao, F.-C.; Hsu, Y.-C.; Chen, W.-H.; Lin, J.-N.; Lo, Y.-Y.; Tu, Y.-K. Prosthetic Joint Infection Following Invasive Dental Procedures and Antibiotic Prophylaxis in Patients with Hip or Knee Arthroplasty. Infect. Control. Hosp. Epidemiol. 2017, 38, 154–161. [Google Scholar] [CrossRef]
- Piñeiro, A.; Tomás, I.; Blanco, J.; Alvarez, M.; Seoane, J.; Diz, P. Bacteraemia following dental implants’ placement. Clin. Oral Implant. Res. 2010, 21, 913–918. [Google Scholar] [CrossRef]
- Roberts, G.J.; Simmons, N.B.; Longhurst, P.; Hewitt, P.B. Bacteraemia following local anaesthetic injections in children. Br. Dent. J. 1998, 185, 295–298. [Google Scholar] [CrossRef] [PubMed]
- Lockhart, P.B. An analysis of bacteremias during dental extractions. A double-blind, placebo-controlled study of chlorhexidine. Arch. Intern. Med. 1996, 156, 513–520. [Google Scholar] [CrossRef]
- Barbosa, M.; Carmona, I.T.; Amaral, B.; Limeres, J.; Alvarez, M.; Cerqueira, C.; Diz, P. General anesthesia increases the risk of bacteremia following dental extractions. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2010, 110, 706–712. [Google Scholar] [CrossRef]
- Noori, N.; Myerson, C.; Charlton, T.; Thordarson, D. Is Antibiotic Prophylaxis Necessary Before Dental Procedures in Patients Post Total Ankle Arthroplasty? Foot Ankle Int. 2018, 40, 237–241. [Google Scholar] [CrossRef] [PubMed]
- Segura-Egea, J.J.; Gould, K.; Şen, B.H.; Jonasson, P.; Cotti, E.; Mazzoni, A.; Sunay, H.; Tjäderhane, L.; Dummer, P.M.H. European Society of Endodontology position statement: The use of antibiotics in endodontics. Int. Endod. J. 2018, 51, 20–25. [Google Scholar] [CrossRef]
- Renvert, S.; Aghazadeh, A.; Hallström, H.; Persson, G.R. Factors related to peri-implantitis—A retrospective study. Clin. Oral Implant. Res. 2014, 25, 522–529. [Google Scholar] [CrossRef] [PubMed]
- Nagy, R.; Szabo, K.; Szucs, A.; Ruszin, T.; Joob-Fancsaly, A. Impact of rheumatoid arthritis in oral surgery and implantology treatment based on literature. Fogorv. Szle. 2017, 110, 3–6. [Google Scholar]
- Arteagoitia, I.; Rodriguez Andrés, C.; Ramos, E. Does chlorhexidine reduce bacteremia following tooth extraction? A systematic review and meta-analysis. PLoS ONE 2018, 13, e0195592. [Google Scholar] [CrossRef] [PubMed]
- Canullo, L.; Laino, L.; Longo, F.; Filetici, P.; D’Onofrio, I.; Troiano, G. Does Chlorhexidine Prevent Complications in Extractive, Periodontal, and Implant Surgery? A Systematic Review and Meta-analysis with Trial Sequential Analysis. Int. J. Oral Maxillofac. Implant. 2020, 35, 1149–1158. [Google Scholar] [CrossRef]
- Tomás, I.; Alvarez, M.; Limeres, J.; Tomás, M.; Medina, J.; Otero, J.L.; Diz, P. Effect of a Chlorhexidine Mouthwash on the Risk of Postextraction Bacteremia. Infect. Control. Hosp. Epidemiol. 2007, 28, 577–582. [Google Scholar] [CrossRef]
- Solderer, A.; Kaufmann, M.; Hofer, D.; Wiedemeier, D.; Attin, T.; Schmidlin, P.R. Efficacy of chlorhexidine rinses after periodontal or implant surgery: A systematic review. Clin. Oral Investig. 2019, 23, 21–32. [Google Scholar] [CrossRef] [PubMed]
| Component | Description |
|---|---|
| P (problem/population) | Patients with OPs 1 that have had a dental implant treatment |
| I (intervention) | PAs 2 on the day of surgery and/or extended postoperatively |
| C (comparison) | Not prescribing PAsPrescribing a placeboOther antibiotics or antibiotic regimensSame antibiotic with different dosage/duration |
| O (outcome) | Risk of infection from OPsSafety (for example, benefits for the patient, OPIs 3 prevention, resistance to antimicrobials) |
| PICO question | In patients with OPs who are about to undergo an implant procedure, does the prescription of PAs decrease the risk of infection of OPs versus not taking them? |
| Questions | Rademacher et al. [7] (2017) | CADTH [28] (2016) | Sollecito et al. [27] (2013) | Watters et al. [5] (2013) |
|---|---|---|---|---|
| 1. Is the review question clearly and explicitly stated? |  |  |  |  |
| 2. Were the inclusion criteria appropriate for the review question? |  |  |  |  |
| 3. Was the search strategy appropriate? |  |  |  |  |
| 4. Were the sources and resources used to search for studies adequate? |  |  |  |  |
| 5. Were the criteria for appraising studies appropriate? |  |  |  |  |
| 6. Was critical appraisal conducted by two or more reviewers independently? |  |  |  |  |
| 7. Were there methods to minimize errors in data extraction? |  |  |  |  |
| 8. Were the methods used to combine studies appropriate? |  |  |  |  |
| 9. Was the likelihood of publication bias assessed? |  |  |  |  |
| 10. Were policy and/or practice recommendations supported by the reported data? |  |  |  |  |
| 11. Were the specific directives for new research appropriate? |  |  |  |  |
 —Yes;
—Yes;  —No;
—No;  —Unclear;
—Unclear;  —Not applicable.
—Not applicable.Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Salgado-Peralvo, A.-O.; Peña-Cardelles, J.-F.; Kewalramani, N.; Garcia-Sanchez, A.; Mateos-Moreno, M.-V.; Velasco-Ortega, E.; Ortiz-García, I.; Jiménez-Guerra, Á.; Végh, D.; Pedrinaci, I.; et al. Is Antibiotic Prophylaxis Necessary before Dental Implant Procedures in Patients with Orthopaedic Prostheses? A Systematic Review. Antibiotics 2022, 11, 93. https://doi.org/10.3390/antibiotics11010093
Salgado-Peralvo A-O, Peña-Cardelles J-F, Kewalramani N, Garcia-Sanchez A, Mateos-Moreno M-V, Velasco-Ortega E, Ortiz-García I, Jiménez-Guerra Á, Végh D, Pedrinaci I, et al. Is Antibiotic Prophylaxis Necessary before Dental Implant Procedures in Patients with Orthopaedic Prostheses? A Systematic Review. Antibiotics. 2022; 11(1):93. https://doi.org/10.3390/antibiotics11010093
Chicago/Turabian StyleSalgado-Peralvo, Angel-Orión, Juan-Francisco Peña-Cardelles, Naresh Kewalramani, Alvaro Garcia-Sanchez, María-Victoria Mateos-Moreno, Eugenio Velasco-Ortega, Iván Ortiz-García, Álvaro Jiménez-Guerra, Dániel Végh, Ignacio Pedrinaci, and et al. 2022. "Is Antibiotic Prophylaxis Necessary before Dental Implant Procedures in Patients with Orthopaedic Prostheses? A Systematic Review" Antibiotics 11, no. 1: 93. https://doi.org/10.3390/antibiotics11010093
APA StyleSalgado-Peralvo, A.-O., Peña-Cardelles, J.-F., Kewalramani, N., Garcia-Sanchez, A., Mateos-Moreno, M.-V., Velasco-Ortega, E., Ortiz-García, I., Jiménez-Guerra, Á., Végh, D., Pedrinaci, I., & Monsalve-Guil, L. (2022). Is Antibiotic Prophylaxis Necessary before Dental Implant Procedures in Patients with Orthopaedic Prostheses? A Systematic Review. Antibiotics, 11(1), 93. https://doi.org/10.3390/antibiotics11010093










