The Role of Statins on Helicobacter pylori Eradication: Results from the European Registry on the Management of H. pylori (Hp-EuReg)
Abstract
:1. Introduction
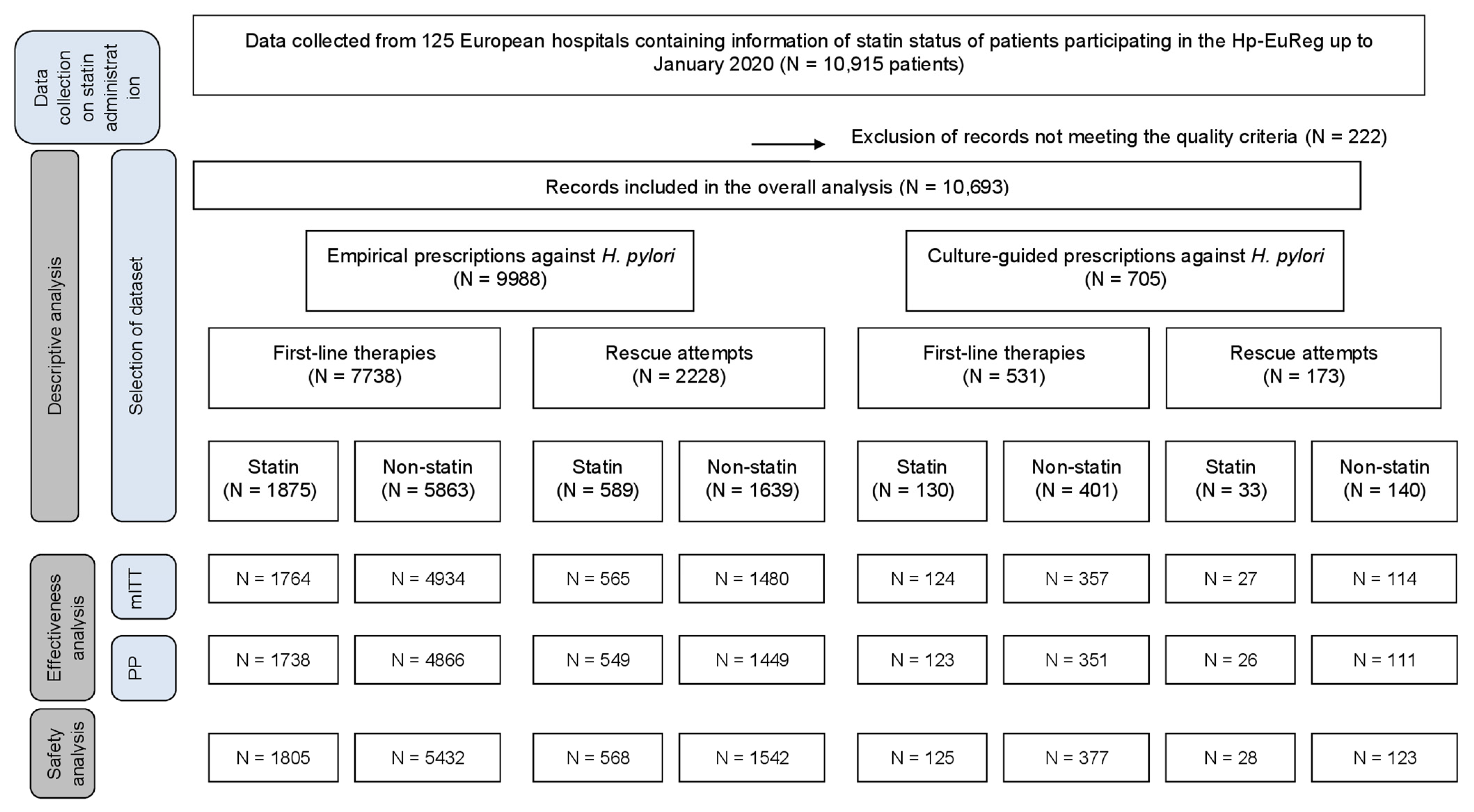
2. Results
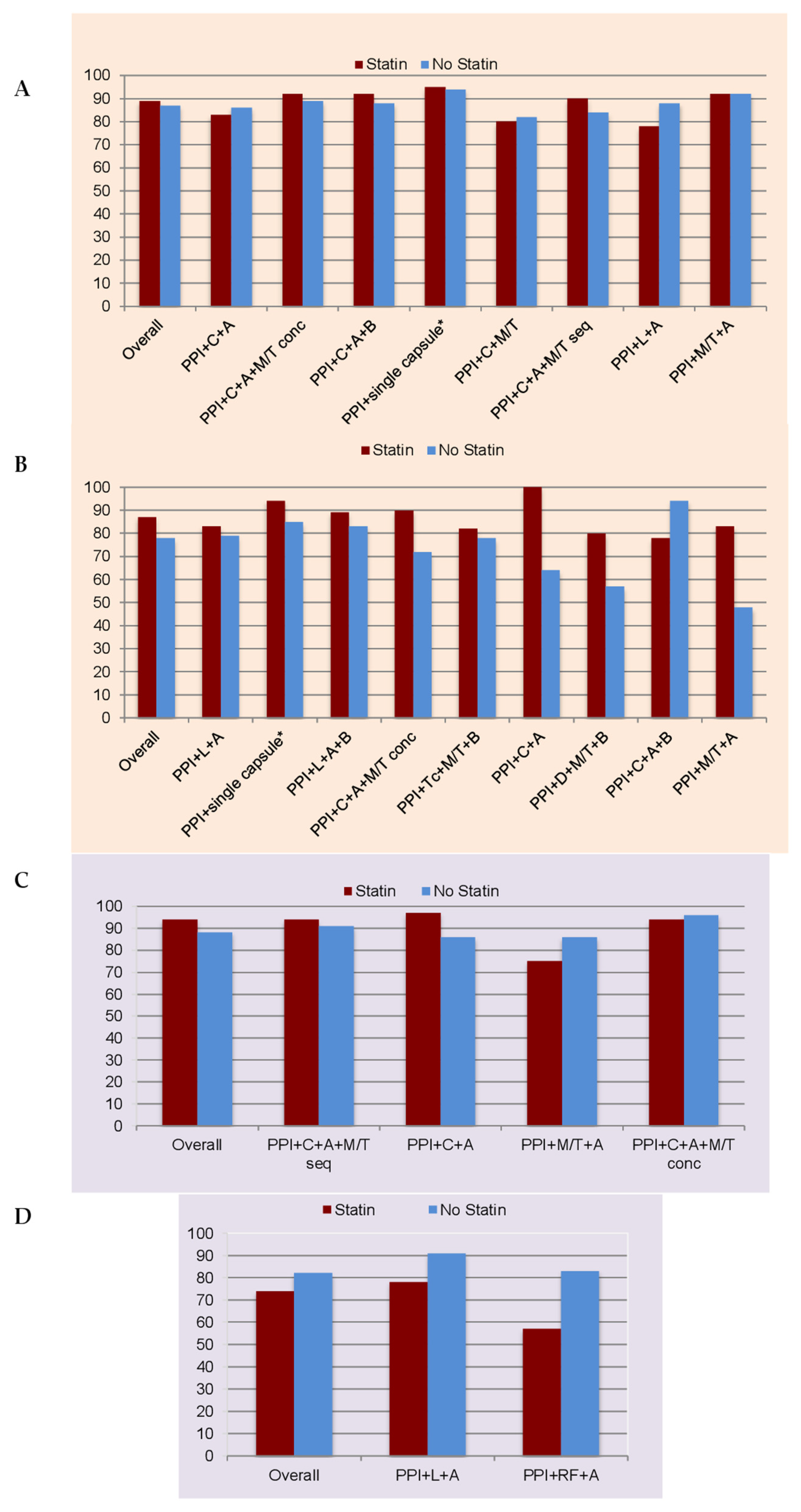
2.1. Empirical Prescription
2.1.1. First-Line Therapy
2.1.2. Rescue Therapies
2.2. Culture-Guided Prescriptions
2.2.1. First-Line Therapy
2.2.2. Rescue Therapies
3. Discussion
4. Materials and Methods
4.1. Variables
4.2. Effectiveness Analysis
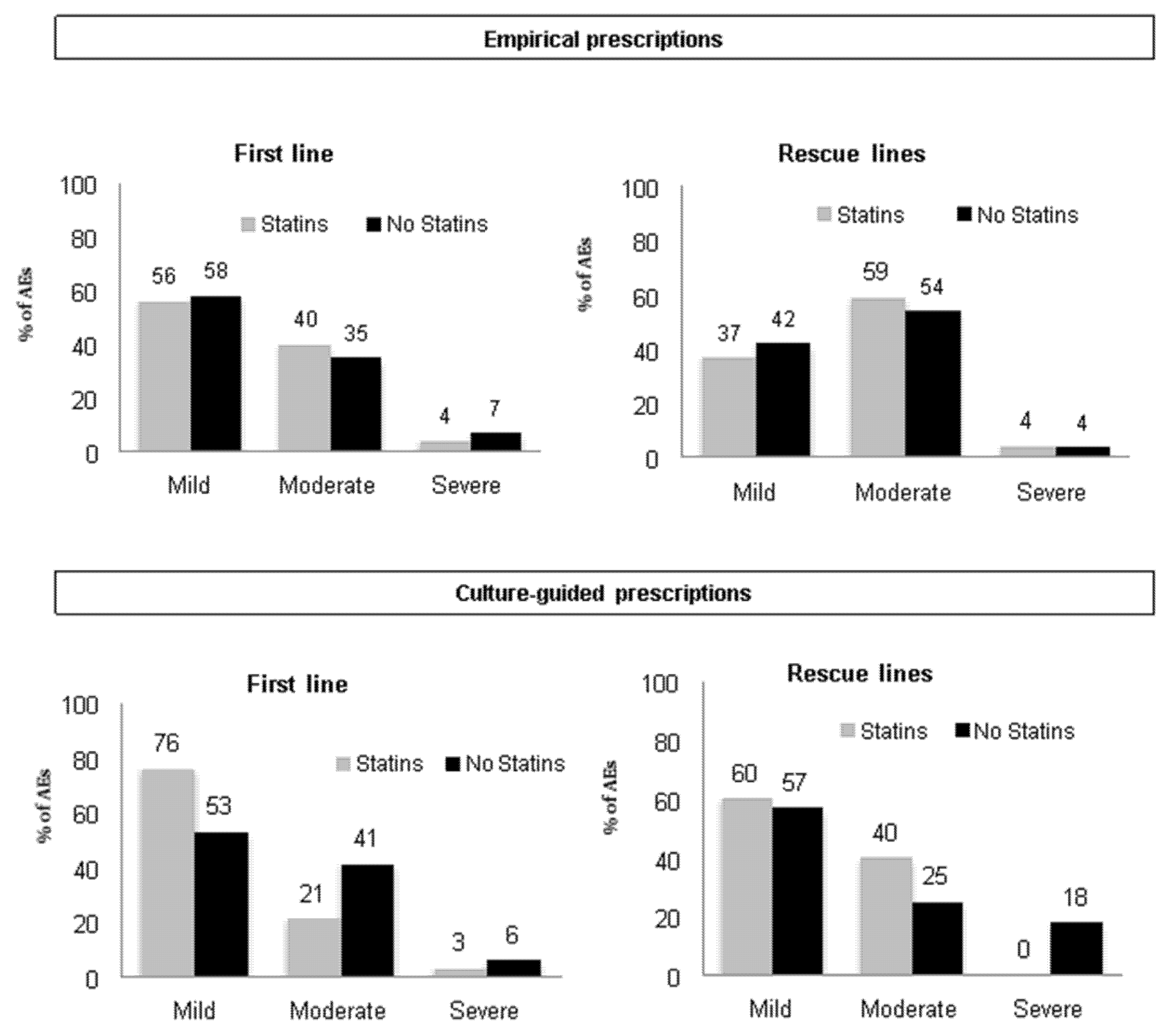
4.3. Safety Analysis
4.4. Statistical Analyses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Malfertheiner, P.; Megraud, F.; O’Morain, C.A.; Gisbert, J.P.; Kuipers, E.J.; Axon, A.; Bazzoli, F.; Gasbarrini, A.; Atherton, J.; Graham, D.J.; et al. Management of Helicobacter pylori infection-the Maastricht V/Florence Consensus Report. Gut 2017, 66, 6–30. [Google Scholar] [CrossRef] [Green Version]
- Crowe, S.E. Helicobacter pylori Infection. N. Engl. J. Med. 2019, 380, 1158–1165. [Google Scholar] [CrossRef]
- Pellicano, R.R.D.; Fagoonee, S.; Astegiano, M.; Saracco, G.M.; Mégraud, M. A 2016 panorama of Helicobacter pylori infection: Key messages for clinicians. Panminerva Med. 2016, 58, 304–317. [Google Scholar]
- Graham, D.Y.; Lu, H.; Yamaoka, Y. Therapy for Helicobacter pylori infection can be improved: Sequential therapy and beyond. Drugs 2008, 68, 725–736. [Google Scholar] [CrossRef] [PubMed]
- Fallone, C.A.; Moss, S.F.; Malfertheiner, P. Reconciliation of Recent Helicobacter pylori Treatment Guidelines in a Time of Increasing Resistance to Antibiotics. Gastroenterology 2019, 157, 44–53. [Google Scholar] [CrossRef] [PubMed]
- Megraud, F.; Coenen, S.; Versporten, A.; Kist, M.; Lopez-Brea, M.; Hirschl, A.; Andersen, L.P.; Goossens, H.; Glupczynski, Y. Helicobacter pylori resistance to antibiotics in Europe and its relationship to antibiotic consumption. Gut 2013, 62, 34–42. [Google Scholar] [CrossRef]
- Nyssen, O.P.; Bordin, D.; Tepes, B.; Perez-Aisa, A.; Vaira, D.; Caldas, M.; Bujanda, L.; Castro-Fernadez, M.; Lerang, F.; Leja, M.; et al. European Registry on Helicobacter pylori management (Hp-EuReg): Patterns and trends in first-line empirical eradication prescription and outcomes of 5 years and 21,533 patients. Gut 2021, 70, 40–54. [Google Scholar] [CrossRef] [PubMed]
- Caldas, M.; Pérez-Aisa, M.; Castro-Fernández, M.; Bujanda, L.; Lucendo, A.J.; Rodrigo, L.; Huguet, J.M.; Pérez-Lasala, J.; Molina-Infante, J.; Barrio, J.; et al. European Registry on Helicobacter pylori Management: Effectiveness of First and Second-Line Treatment in Spain. Antibiotics 2020, 10, 13. [Google Scholar] [CrossRef] [PubMed]
- Gisbert, J.P.; McNicholl, A.G. Optimization strategies aimed to increase the efficacy of, H. pylori eradication therapies. Helicobacter 2017, 22, e12392. [Google Scholar] [CrossRef]
- Li, C.; Shi, Y.; Suo, B.; Tian, X.; Zhou, L.; Song, Z. PPI-amoxicillin dual therapy four times daily is superior to guidelines recommended regimens in the Helicobacter pylori eradication therapy within Asia: A systematic review and meta-analysis. Helicobacter 2021, e12816. [Google Scholar] [CrossRef]
- Georgopoulos, S.; Papastergiou, V. An update on current and advancing pharmacotherapy options for the treatment of, H. pylori infection. Expert Opin. Pharmacother. 2020, 22, 729–741. [Google Scholar] [CrossRef]
- Grundy, S.M.; Stone, N.J.; Bailey, A.L.; Beam, C.; Birtcher, K.K.; Blumenthal, R.S.; Braun, L.T.; De Ferranti, S.; Faiella-Tommasino, J.; Forman, D.E.; et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA Guideline on the Management of Blood Cholesterol. J. Am. Coll. Cardiol. 2019, 73, e285–e350. [Google Scholar] [CrossRef]
- Cholesterol Treatment Trialists’ (CTT) Collaboration; Baigent, C.; Blackwell, L.; Emberson, J.; Holland, L.E.; Reith, C.; Bhala, N.; Peto, R.; Barnes, E.H.; Keech, A.; et al. Efficacy and safety of more intensive lowering of LDL cholesterol: A meta-analysis of data from 170,000 participants in 26 randomised trials. Lancet 2010, 376, 1670–1681. [Google Scholar]
- Dehnavi, S.; Sohrabi, N.; Sadeghi, M.; Lansberg, P.; Banach, M.; Al-Rasadi, K.; Johnston, T.P.; Sahebkar, A. Statins and autoimmunity: State-of-the-art. Pharmacol. Ther. 2020, 214, 107614. [Google Scholar] [CrossRef] [PubMed]
- Pinal-Fernandez, I.; Casal-Dominguez, M.; Mammen, A.L. Statins: Pros and cons. Med. Clínica 2018, 150, 398–402. [Google Scholar] [CrossRef]
- Albert, M.A.; Danielson, E.; Rifai, M.N.; Ridker, P.M. Effect of statin therapy on C-reactive protein levels: The pravastatin inflammation/CRP evaluation (PRINCE): A randomized trial and cohort study. JAMA 2001, 286, 64–70. [Google Scholar] [CrossRef]
- Deshpande, A.; Pasupuleti, V.; Rothberg, M.B. Statin Therapy and Mortality from Sepsis: A Meta-analysis of Randomized Trials. Am. J. Med. 2015, 128, 410–417.e1. [Google Scholar] [CrossRef] [PubMed]
- Wan, Y.-D.; Sun, T.-W.; Kan, Q.-C.; Guan, F.-X.; Zhang, S.-G. Effect of statin therapy on mortality from infection and sepsis: A meta-analysis of randomized and observational studies. Crit. Care 2014, 18, R71. [Google Scholar] [CrossRef] [Green Version]
- Cheung, K.-S.; Chan, E.W.; Wong, A.Y.S.; Chen, L.; Seto, W.-K.; Wong, I.C.K.; Leung, W.K. Statins were associated with a reduced gastric cancer risk in patients with eradicated Helicobacter pylori infection: A territory-wide propensity score matched study. Cancer Epidemiol. Biomark. Prev. 2020, 29, 493–499. [Google Scholar] [CrossRef] [PubMed]
- Chiu, H.-F.; Ho, S.-C.; Chang, C.-C.; Wu, T.-N.; Yang, C.-Y. Statins Are Associated With a Reduced Risk of Gastric Cancer: A Population-Based Case–Control Study. Am. J. Gastroenterol. 2011, 106, 2098–2103. [Google Scholar] [CrossRef]
- Pantea, M.; Negovan, A.; Voidăzan, S.; Macarie, M.; Mocan, S.; Bataga, S.M. Statins and gastroduodenal endoscopic lesions. Medicine 2018, 97, e13579. [Google Scholar] [CrossRef]
- Lin, C.-J.; Liao, W.-C.; Lin, H.-J.; Hsu, Y.-M.; Lin, C.-L.; Chen, Y.-A.; Feng, C.-L.; Chen, A.-J.; Kao, M.-C.; Lai, C.-H.; et al. Statins Attenuate Helicobacter pylori CagA Translocation and Reduce Incidence of Gastric Cancer: In Vitro and Population-Based Case-Control Studies. PLoS ONE 2016, 11, e0146432. [Google Scholar] [CrossRef]
- Ribaldone, D.G.; Vernero, M.; Caviglia, G.P. Targeting IL-10, ZO-1 gene expression and IL-6/STAT-3 trans-signalling by a combination of atorvastatin and mesalazine to enhance anti-inflammatory effects and attenuate progression of oxazolone-induced colitis. Fundam. Clin. Pharmacol. 2020, 35, 141–142. [Google Scholar] [CrossRef]
- Yamato, M.; Watanabe, T.; Higuchi, K.; Taira, K.; Tanigawa, T.; Shiba, M.; Tominaga, K.; Fujiwara, Y.; Oshitani, N.; Takeuchi, K.; et al. Anti-Inflammatory Effects of Pravastatin on Helicobacter Pylori-Induced Gastritis in Mice. Dig. Dis. Sci. 2007, 52, 2833–2839. [Google Scholar] [CrossRef] [PubMed]
- Nseir, W.K.J.; Tatour, I.; Haiek, S.; Samara, M.; Assy, N. Long-Term Statin Therapy Affects the Severity of Chronic Gastritis. Helicobacter 2010, 15, 510–515. [Google Scholar] [CrossRef]
- Nseir, W.; Diab, H.; Mahamid, M.; Abu-Elheja, O.; Samara, M.; Abid, A.; Mograbi, J. Randomised clinical trial: Simvastatin as adjuvant therapy improves significantly the Helicobacter pylori eradication rate—A placebo-controlled study. Aliment. Pharmacol. Ther. 2012, 36, 231–238. [Google Scholar] [CrossRef]
- Liao, W.-C.; Huang, M.-Z.; Wang, M.L.; Lin, C.-J.; Lu, T.-L.; Lo, H.-R.; Pan, Y.-J.; Sun, T.-C.; Kao, M.-C.; Lin, H.-J.; et al. Statin Decreases Helicobacter pylori Burden in Macrophages by Promoting Autophagy. Front. Cell. Infect. Microbiol. 2017, 6, 203. [Google Scholar] [CrossRef] [Green Version]
- Lai, C.-H.; Chang, Y.-C.; Du, S.-Y.; Wang, H.-J.; Kuo, C.-H.; Fang, S.-H.; Fu, H.-W.; Lin, H.-H.; Chiang, A.-S.; Wang, W.-C. Cholesterol Depletion Reduces Helicobacter pylori CagA Translocation and CagA-Induced Responses in AGS Cells. Infect. Immun. 2008, 76, 3293–3303. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McNicholl, A.G.; O’Morain, C.A.; Megraud, F.; Gisbert, J.P. Protocol of the European Registry on the management of Helicobacter pylori infection (Hp-EuReg). Helicobacter 2019, 24, e12630. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.Q.; Hunt, R.H. Treatment after failure: The problem of “non-responders”. Gut 1999, 45, I40–I44. [Google Scholar] [CrossRef] [Green Version]
- Molina-Infante, J.; Lucendo, A.J.; Angueira, T.; Rodriguez-Tellez, M.; Perez-Aisa, A.; Balboa, A.; Barrio, J.; Martin-Noguerol, E.; Gomez-Rodriguez, J.; Botargues-Bote, J.M.; et al. Optimised empiric triple and concomitant therapy for Helicobacter pylori eradication in clinical practice: The OPTRICON study. Aliment. Pharmacol. Ther. 2015, 41, 581–589. [Google Scholar] [CrossRef] [PubMed]
- Graham, D.Y.; Lu, H.; Dore, M.P. Relative potency of proton-pump inhibitors, Helicobacter pylori therapy cure rates, and meaning of double-dose PPI. Helicobacter 2019, 24, e12554. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McNicholl, A.G.; Bordin, D.S.; Lucendo, A.; Fadeenko, G.; Fernandez, M.C.; Вoйнoван, И.; Zakharova, N.V.; Sarsenbaeva, A.S.; Bujanda, L.; Perez-Aisa, A.; et al. Combination of Bismuth and Standard Triple Therapy Eradicates Helicobacter pylori Infection in More than 90% of Patients. Clin. Gastroenterol. Hepatol. 2020, 18, 89–98. [Google Scholar] [CrossRef]
- Nyssen, O.P.; Perez-Aisa, A.; Castro-Fernandez, M.; Pellicano, R.; Huguet, J.M.; Rodeigo, L.; Ortun, J.; Gomez-Rodriguez, B.J.; Pinto, R.M.; Areia, M.; et al. European Registry on Helicobacter pylori management: Single-capsule bismuth quadruple therapy is effective in real-world clinical practice. United Eur. Gastroenterol. J. 2021, 9, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Hassan, A.M.; Shawky, M.A.E.-G.; Mohammed, A.Q.; Haridy, M.A.; Eid, K.A.-E.-A. Simvastatin improves the eradication rate of Helicobacter pylori: Upper Egypt experience. Infect. Drug Resist. 2019, 12, 1529–1534. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McNicholl, A.G.; Gisbert, J.P. Commentary: Comparators in, H. pylori eradication—stating the ethics of statins. Aliment. Pharmacol. Ther. 2012, 36, 400–401. [Google Scholar] [CrossRef]
- Parsi, A.; Biuseh, M.; Mohammadi, S.; Hajiani, E.; Hashemi, S.J.; Saeidian, H.; Biuseh, M. Study of probiotic and simvastatin additive effect on Helicobacter pylori eradication rate. Int. J. Gastrointest. Interv. 2019, 8, 160–164. [Google Scholar] [CrossRef] [Green Version]
- Sarkeshikian, S.S.; Ghadir, M.R.; Alemi, F.; Jalali, S.M.; Hormati, A.; Mohammadbeigi, A. Atorvastatin in combination with conventional antimicrobial treatment of Helicobacter pylori eradication: A randomized controlled clinical trial. J. Gastroenterol. Hepatol. 2019, 35, 71–75. [Google Scholar] [CrossRef]
- Dormuth, C.R.; Filion, K.; Paterson, J.M.; James, M.T.; Teare, G.F.; Raymond, C.B.; Rahme, E.; Tamim, H.; Lipscombe, L.; for the Canadian Network for Observational Drug Effect Studies (CNODES) Investigators. Higher potency statins and the risk of new diabetes: Multicentre, observational study of administrative databases. BMJ 2014, 348, g3244. [Google Scholar] [CrossRef] [Green Version]
- Harris, P.A.; Taylor, R.; Thielke, R.; Gonzalez, N.; Conde, J.G. Research electronic data capture (REDCap)—A metadata-driven methodology and workflow process for providing translational research informatics support. J. Biomed. Inform 2009, 42, 377–381. [Google Scholar] [CrossRef] [Green Version]
- McNicholl, A.G.; Gisbert, J.P. Research to the N-Power: The Strengths of Networked Clinical Collaboration in Spain. Am. J. Gastroenterol. 2017, 112, 1761–1764. [Google Scholar] [CrossRef] [PubMed]
- Kirchheiner, J.; Glatt, S.; Fuhr, U.; Klotz, U.; Meineke, I.; Seufferlein, T.; Brockmöller, J. Relative potency of proton-pump inhibitors-comparison of effects on intragastric pH. Eur. J. Clin. Pharmacol. 2009, 65, 19–31. [Google Scholar] [CrossRef] [PubMed]
| Statin Users, N (%) | Non-Statin Users, N (%) | p-Value | ||
|---|---|---|---|---|
| Patients evaluated | 2635 (25) | 8058 (75) | ||
| Age (years) +/−SD | 63 ± 10 | 53 ± 15 | <0.001 | |
| Gender | Females | 1518 (58) | 5093 (63) | <0.001 |
| Males | 1114 (42) | 2962 (37) | ||
| Penicillin allergy | Yes | 141 (5) | 347 (4) | 0.026 |
| No | 2494 (95) | 7711 (96) | ||
| Indication | No ulcer | 1982 (75.3) | 6526 (81) | <0.001 |
| Ulcer disease | 649 (25) | 1511 (19) | ||
| Line of eradication | First line | 2005 (76) | 6264 (78) | 0.097 |
| Rescue lines | 622 (24) | 1779 (22) | ||
| Prescription | Empirical | 2472 (94) | 7516 (93) | 0.332 |
| Culture-guided | 163 (6) | 542 (7) | ||
| PPI dose | Low | 949 (37) | 3220 (41) | 0.002 |
| Standard | 626 (24) | 1742 (22) | ||
| High | 1006 (39) | 2914 (37) | ||
| Length (days) | 7 | 219 (9) | 941 (12) | 0.002 |
| 10 | 1425 (55) | 4195 (53) | ||
| 14 | 929 (36) | 2719 (35) | ||
| Compliance (≥90%) | Yes | 2476 (98) | 7262 (98) | 0.502 |
| No | 54 (2) | 176 (2) | ||
| Adverse events | Yes | 594 (24) | 1703 (23) | 0.460 |
| No | 1934 (76) | 5772 (77) | ||
| Effectiveness | mITT | 2190 (88) | 5874 (85) | <0.001 |
| PP | 2160 (89) | 5818 (86) | 0.001 |
| Simvastatin N (%) | Atorvastatin N (%) | Rosuvastatin N (%) | Other N (%) | p-Value | ||
|---|---|---|---|---|---|---|
| Patients evaluated | 155 (45) | 134 (39) | 37 (11) | 17 (5) | ||
| Age (years) +/−SD | 63 ± 10 | 63 ± 10 | 66 ± 8 | 63 ± 13 | 0.492 | |
| Gender | Females | 86 (55.5) | 69 (51.5) | 19 (51) | 10 (59) | 0.869 |
| Males | 69 (44.5) | 65 (48.5) | 18 (49) | 7 (41) | ||
| Penicillin allergy | Yes | 7 (4.5) | 4 (3) | 0 (0) | 1 (6) | 0.532 |
| No | 148 (95.5) | 130 (97) | 37 (100) | 16 (94) | ||
| Indication | No ulcer | 119 (77) | 91 (68) | 32 (86.5) | 14 (82) | 0.070 |
| Ulcer disease | 35 (23) | 43 (32) | 5 (13.5) | 3 (18) | ||
| Treatment attempt | First | 120 (77) | 107 (81) | 30 (81) | 14 (82) | 0.862 |
| Rescue | 35 (23) | 25 (19) | 7 (19) | 3 (18) | ||
| Prescription | Empirical | 135 (87) | 108 (81) | 35 (95) | 15 (88) | 0.142 |
| Culture-guided | 20 (13) | 26 (19) | 2 (5) | 2 (12) | ||
| PPI dose | Low | 75 (49) | 80 (62) | 29 (78) | 10 (59) | 0.026 |
| Standard | 43 (28) | 23 (18) | 2 (5) | 4 (23) | ||
| High | 36 (23) | 26 (20) | 6 (16) | 3 (18) | ||
| Length (days) | 7 | 17 (11) | 17 (13) | 5 (13) | 2 (12) | 0.278 |
| 10 | 107 (70) | 90 (69) | 31 (84) | 11 (65) | ||
| 14 | 28 (18) | 23 (18) | 1 (3) | 4 (23) | ||
| Compliance (≥90%) | Yes | 148 (98) | 128 (98.5) | 37 (100) | 16 (100) | 0.789 |
| No | 3 (2) | 2 (1.5) | 0 (0) | 0 (0) | ||
| Adverse events | Yes | 18 (12) | 21 (16) | 5 (13.5) | 2 (12.5) | 0.751 |
| No | 136 (88) | 109 (84) | 32 (86.5) | 14 (87.5) | ||
| Effectiveness | mITT | 122 (84) | 112 (91) | 29 (81) | 16 (100) | 0.086 |
| PP | 122 (85) | 111 (92) | 29 (81) | 16 (100) | 0.074 |
| N (%) | mITT Effectiveness | PP Effectiveness | Adverse Events | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| N Total (%) | 95% CI | p-Value | N Total (%) | 95% CI | p-Value | N Total (%) | 95% CI | p-Value | ||
| Overall empirical therapies | ||||||||||
| Statins | 2472 (25) | 2330 (88) | 87–89 | <0.001 | 2288 (88.5) | 87–90 | 0.001 | 2375 (24) | 22–25 | 0.542 |
| No S | 7516 (75) | 6414 (85) | 84–86 | 6315 (86) | 85–87 | 6975 (23) | 22–24 | |||
| Overall culture-guided therapies | ||||||||||
| Statins | 163 (23) | 151 (90) | 84–94 | 0.266 | 149 (91) | 85–95 | 0.330 | 153 (22) | 16–30 | 0.589 |
| No S | 542 (77) | 471 (87) | 83–90 | 462 (88) | 84–91 | 500 (20) | 17–24 | |||
| Empirical Prescription N = 9988 | Culture-Guided Prescription N = 705 | ||||||
|---|---|---|---|---|---|---|---|
| OR | 95% CI | p-Value | OR | 95% CI | p-Value | ||
| Gender | Female | 1 | NS | ||||
| Male | 1.218 | 1.1–1.4 | 0.004 | ||||
| Indication | No ulcer | 1 | NS | ||||
| Ulcer disease | 1.272 | 1.1–1.5 | 0.005 | ||||
| Treatment attempt | First | 1 | 1 | ||||
| Rescue | 0.522 | 0.45–0.60 | <0.001 | 0.480 | 0.29–0.81 | 0.006 | |
| Length (days) | 7 | 1 | NS | ||||
| 10 | 1.375 | 1.1–1.7 | 0.001 | ||||
| 14 | 1.424 | 1.1–1.8 | 0.002 | ||||
| PPI dose | Low | 1 | NS | ||||
| Standard | 1.492 | 1.3–1.8 | <0.001 | ||||
| High | 2.026 | 1.7–2.4 | <0.001 | ||||
| Compliance | No | 1 | 1 | ||||
| Yes | 5.224 | 3.6–7.6 | <0.001 | 12.716 | 3.6–45 | <0.001 | |
| Statin use | No | 1 | NS | ||||
| Yes | 1.269 | 1.1–1.5 | 0.002 | ||||
| N (R) | mITT Effectiveness | PP Effectiveness | Adverse Events | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| N (%) | 95% CI | p-Value | N Total (%) | 95% CI | p-Value | N Total (%) | 95% CI | p-Value | |||
| Overall | Statins | 1875 | 1764 (88.5) | 87–90 | 0.254 | 1738 (89) | 87–90 | 0.388 | 1805 (20) | 18–22 | 0.197 |
| No S | 5863 | 4934 (87) | 86–88 | 4866 (88) | 87–89 | 5432 (21) | 20–23 | ||||
| PPI + C + A | Statins | 605 (32) | 541 (83) | 79–86 | 0.063 | 531 (83) | 79–86 | 0.048 | 567 (15) | 12–18 | 0.792 |
| No S | 2259 (39) | 1779 (86) | 84–87 | 1753 (86) | 85–88 | 1985 (15) | 14–17 | ||||
| PPI + C + A + M/T conc | Statins | 541 (29) | 532 (91.5) | 89–94 | 0.056 | 526 (92) | 89–94 | 0.099 | 537 (19) | 16–22 | 0.004 |
| No S | 1253 (21) | 1207 (88.5) | 87–90 | 1189 (89) | 87–91 | 1229 (25) | 23–28 | ||||
| PPI + C + A + B | Statins | 186 (10) | 182 (92) | 87–96 | 0.131 | 177 (92) | 87–96 | 0.180 | 181 (30) | 24–38 | 0.094 |
| No S | 840 (14) | 610 (88) | 86–91 | 603 (89) | 86–91 | 824 (24) | 22–28 | ||||
| PPI + single capsule * | Statins | 291 (16) | 279 (95) | 92–98 | 0.540 | 275 (96) | 93–98 | 0.700 | 283 (26) | 21–32 | 0.067 |
| No S | 636 (11) | 601 (94) | 92–96 | 590 (95) | 93–97 | 612 (32) | 29–36 | ||||
| PPI + C+ M/T | Statins | 99 (5) | 93 (80) | 70–87 | 0.660 | 93 (80) | 70–87 | 0.657 | 94 (19) | 12–29 | 0.852 |
| No S | 441 (8) | 384 (81.5) | 77–85 | 382 (82) | 77–85 | 395 (20) | 16–24 | ||||
| PPI + C + A + M/T seq | Statins | 45 (2) | 41 (90) | 77–97 | 0.566 | 40 (90) | 76–97 | 0.566 | 42 (7) | 2–20 | 0.066 |
| No S | 79 (1) | 70 (84) | 74–92 | 70 (84) | 74–92 | 73 (22) | 13–33 | ||||
| PPI + L + A | Statins | 24 (1) | 23 (78) | 56–93 | 0.243 | 23 (78) | 56–93 | 0.254 | 24 (29) | 13–51 | 0.429 |
| No S | 79 (1) | 75 (88) | 78–94 | 74 (88) | 78–94 | 75 (21) | 13–32 | ||||
| PPI + M/T + A | Statins | 13 (1) | 13 (92) | 64–100 | 1 | 13 (92) | 64–100 | 1 | 13 (31) | 9–61 | 0.733 |
| No S | 55 (1) | 47 (91.5) | 80–98 | 47 (91.5) | 80–98 | 55 (25.5) | 15–39 | ||||
| Overall | PPI + C+A | PPI + C+M/T | PPI + C+A + M/T Conc | PPI + C+A + B | PPI + Single Capsule * | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| OR | 95% CI | p-Value | OR | 95% CI | p-Value | OR | 95% CI | p-Value | OR | 95% CI | p-Value | OR | 95% CI | p-Value | OR | 95% CI | p-Value | ||
| Gender | Female | 1 | NS | NS | NS | NS | |||||||||||||
| Male | 1.367 | 1.2–1.6 | <0.001 | 1.547 | 1.2–1.9 | 0.001 | |||||||||||||
| Length (days) | 7 | 1 | NS | NS | NS | NS | |||||||||||||
| 10 | 1.328 | 1.1–1.6 | 0.008 | 0.343 | 0.15–0.78 | 0.011 | |||||||||||||
| 14 | 1.286 | 1–1.7 | 0.047 | 0.261 | 0.12–0.58 | 0.001 | |||||||||||||
| PPI dose | Low | 1 | 1 | 1 | NS | ||||||||||||||
| Standard | 1.619 | 1.3–2 | <0.001 | 1.843 | 1.4–2.4 | <0.001 | 3.698 | 1.2–12 | 0.026 | 1.381 | 0.92–2.1 | 0.122 | 7.451 | 3.0–18 | <0.001 | ||||
| High | 1.982 | 1.6–2.4 | <0.001 | 2.644 | 1.9–3.6 | <0.001 | 2.518 | 1.4–4.7 | 0.003 | 1.827 | 1.3–2.6 | 0.001 | 2.451 | 1.5–4.0 | <0.001 | ||||
| Compliance | No | 1 | NS | 1 | NS | 1 | |||||||||||||
| Yes | 6.197 | 4–9.7 | <0.001 | 5.613 | 2.6–12.1 | <0.001 | 10.136 | 4.4–24 | <0.001 | 29.0 | 9.6–88 | <0.001 | |||||||
| Statin use | No | NS | NS | NS | NS | NS | |||||||||||||
| Yes | 0.763 | 0.59–0.99 | 0.046 | ||||||||||||||||
| N (R) | mITT Effectiveness | PP Effectiveness | Adverse Events | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| N (%) | 95% CI | p-Value | N (%) | 95% CI | p-Value | N (%) | 95% CI | p-Value | |||
| Overall | Statins No S | 589 1639 | 565 (87) 1480 (78) | 84–90 76–80 | <0.001 | 549 (88) 1449 (78) | 85–91 76–80 | <0.001 | 568 (35) 1542 (28) | 31–39 26–31 | 0.003 |
| PPI + L + A | Statins No S | 177 (30) 545 (33) | 172 (83) 504 (79) | 76–88 75–82 | 0.263 | 169 (83) 494 (79) | 76–88 75–83 | 0.275 | 175 (27) 521 (25) | 21–35 22–29 | 0.584 |
| PPI + single capsule * | Statins No S | 170 (29) 328 (20) | 164 (94) 298 (85) | 89–97 81–89 | 0.006 | 159 (95) 291 (86) | 90–98 81–89 | 0.002 | 165 (42) 310 (39) | 35–50 34–45 | 0.473 |
| PPI + L + A + B | Statins No S | 65 (11) 178 (11) | 63 (89) 166 (83) | 78–95 77–89 | 0.280 | 59 (91.5) 164 (83.5) | 81–97 77–89 | 0.133 | 62 (39) 168 (34.5) | 27–52 27–42 | 0.556 |
| PPI + C + A + M/T conc | Statins | 31 (5) | 31 (90) | 74–98 | 0.043 | 30 (90) | 74–98 | 0.107 | 31 (29) | 14–48 | 0.253 |
| No S | 76 (5) | 71 (72) | 60–82 | 68 (73.5) | 61–84 | 74 (19) | 11–30 | ||||
| PPI + M/T + Tc + B | Statins | 17 (3) | 17 (82) | 57–96 | 1 | 17 (82) | 57–96 | 1 | 17 (35) | 14–62 | 0.933 |
| No S | 88 (5) | 83 (78) | 68–87 | 82 (78) | 68–86 | 88 (36) | 26–47 | ||||
| PPI + C + A | Statins | 10 (2) | 10 (100) | NA | 0.024 | 10 (100) | NA | 0.024 | 10 (20) | 3–56 | 1 |
| No S | 53 (3) | 44 (64) | 48–78 | 44 (64) | 48–78 | 52 (23) | 13–37 | ||||
| PPI + M/T + D + B | Statins | 15 (3) | 15 (80) | 52–96 | 0.134 | 14 (79) | 49–95 | 0.205 | 14 (29) | 8–58 | 0.728 |
| No S | 44 (3) | 42 (57) | 41–72 | 41 (56) | 40–72 | 43 (23) | 12–39 | ||||
| PPI + C + A + B | Statins | 10 (2) | 9 (78) | 40–97 | 0.188 | 9 (78) | 40–97 | 0.111 | 9 (56) | 21–86 | 0.043 |
| No S | 40 (2) | 34 (94) | 80–99 | 33 (97) | 84–100 | 36 (19) | 8–36 | ||||
| PPI + M/T + A | Statins | 6 (1) | 6 (83) | 36–100 | 0.187 | 6 (83) | 36–100 | 0.187 | 6 (0) | 0 | 1 |
| No S | 35 (2) | 29 (48) | 30–68 | 29 (48) | 30–68 | 31 (6.5) | 1–21 | ||||
| Overall | PPI + L+A | PPI + Single Capsule * | PPI + L+A + B | PPI + C+A + M/T Conc | PPI + M/T + Tc + B | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| OR | 95% CI | p-Value | OR | 95% CI | p-Value | OR | 95% CI | p-Value | OR | 95% CI | p-Value | OR | 95% CI | p-Value | OR | 95% CI | p-Value | ||
| Age (years) | 18–30 | 1 | NS | NS | NS | NS | NS | ||||||||||||
| 31–50 | 0.413 | 0.20–0.84 | 0.015 | ||||||||||||||||
| 51–70 | 0.357 | 0.18–0.72 | 0.004 | ||||||||||||||||
| >70 | 0.442 | 0.20–0.96 | 0.039 | ||||||||||||||||
| Diagnosis | No ulcer | 1 | NS | NS | NS | NS | NS | ||||||||||||
| Ulcer disease | 1.520 | 1.1–2.1 | 0.008 | ||||||||||||||||
| Length (days) | 7 | 1 | NS | NS | NS | 1 | |||||||||||||
| 10 | 2.425 | 1.4–4.2 | 0.001 | 1 | 1.246 | 0.26–6.0 | 0.784 | ||||||||||||
| 14 | 2.914 | 1.6–5.2 | <0.001 | 2.818 | 1.6–5.0 | <0.001 | 5.640 | 1.0–31 | 0.046 | ||||||||||
| PPI dose | Low | 1 | 1 | NS | 1 | NS | NS | ||||||||||||
| Standard | 1.204 | 0.89–1.6 | 0.230 | 1.283 | 0.80–2.1 | 0.296 | 2.778 | 0.43–18 | 0.284 | ||||||||||
| High | 1.995 | 1.5–2.7 | <0.001 | 1.921 | 1.1–3.4 | 0.024 | 3.822 | 1.2–12 | 0.025 | ||||||||||
| Compliance | No | 1 | NS | 1 | 1 | NS | NS | ||||||||||||
| Yes | 3.937 | 2.0–7.6 | <0.001 | 4.442 | 1.3–15.8 | 0.021 | 6.880 | 1.3–36 | 0.022 | ||||||||||
| Statin use | No | 1 | NS | 1 | NS | 1 | NS | ||||||||||||
| Yes | 1.896 | 1.4–2.6 | <0.001 | 2.752 | 1.3–5.7 | 0.006 | 3.660 | 0.99–13 | 0.050 | ||||||||||
| N (R) | mITT Effectiveness | PP Effectiveness | Adverse Events | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| N (%) | 95% CI | p-Value | N (%) | 95% CI | p-Value | N (%) | 95% CI | p-Value | |||
| First-line (N = 531) | |||||||||||
| Overall | Statins | 130 | 124 (93.5) | 88–97 | 0.095 | 123 (93.5) | 88–97 | 0.163 | 125 (21) | 14–29 | 0.871 |
| No S | 401 | 357 (88) | 84–91 | 351 (89) | 85–92 | 377 (22) | 18–26 | ||||
| PPI + C + A + M/T seq | Statins | 54 (42) | 53 (94) | 84–99 | 0.557 | 52 (94) | 84–99 | 0.559 | 53 (23) | 12–36 | 0.794 |
| No S | 143 (36) | 123 (91) | 85.96 | 122 (90) | 84–95 | 135 (24) | 18–33 | ||||
| PPI + C + A | Statins | 40 (31) | 36 (97) | 86–100 | 0.074 | 36 (97) | 86–100 | 0.189 | 37 (8) | 2–22 | 0.903 |
| No S | 131 (33) | 117 (85.5) | 78–91 | 113 (88.5) | 81–94 | 120 (8) | 4–14 | ||||
| PPI + M/T + A | Statins | 4 (3) | 4 (75) | 19–99 | 0.496 | 4 (75) | 19–99 | 0.496 | 4 (50) | 7–93 | 0.229 |
| No S | 44 (11) | 42 (86) | 72–95 | 42 (86) | 72–95 | 43 (21) | 10–36 | ||||
| PPI + C + A + M/T conc | Statins | 16 (12) | 16 (94) | 70–100 | 1 | 16 (94) | 70–100 | 1 | 16 (38) | 15–65 | 0.950 |
| No S | 26 (7) | 25 (96) | 80–100 | 24 (96) | 79–100 | 26 (39) | 20–59 | ||||
| Rescue lines (N = 173) | |||||||||||
| Overall | Statins | 33 | 27 (74) | 54–89 | 0.379 | 26 (77) | 56–91 | 0.479 | 28 (29) | 13–49 | 0.130 |
| No S | 140 | 114 (82) | 73–88 | 111 (83) | 75–89 | 123 (16) | 10–24 | ||||
| PPI + L + A | Statins | 10 (30) | 9 (78) | 40–97 | 0.299 | 9 (78) | 40–97 | 0.299 | 8 (25) | 3–65 | 0.578 |
| No S | 37 (26) | 32 (91) | 75–98 | 32 (91) | 75–98 | 33 (12) | 3–28 | ||||
| PPI + RF + A | Statins | 7 (22) | 7 (57) | 18–90 | 0.130 | 7 (57) | 18–90 | 0.143 | 7 (57) | 18–90 | 0.018 |
| No S | 36 (26) | 30 (83) | 65–94 | 29 (83) | 64–94 | 34 (12) | 3–28 | ||||
| First-Line | Rescue Therapies | |||||
|---|---|---|---|---|---|---|
| OR | 95% CI | p-Value | OR | 95% CI | p-Value | |
| Compliance | ||||||
| No | 1 | NS | ||||
| Yes | 12.4 | 2.7–57 | 0.001 | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Caldas, M.; Pérez-Aisa, Á.; Tepes, B.; Keco-Huerga, A.; Bujanda, L.; Lucendo, A.J.; Rodrigo, L.; Vaira, D.; Fernández-Salazar, L.; Huguet, J.M.; et al. The Role of Statins on Helicobacter pylori Eradication: Results from the European Registry on the Management of H. pylori (Hp-EuReg). Antibiotics 2021, 10, 965. https://doi.org/10.3390/antibiotics10080965
Caldas M, Pérez-Aisa Á, Tepes B, Keco-Huerga A, Bujanda L, Lucendo AJ, Rodrigo L, Vaira D, Fernández-Salazar L, Huguet JM, et al. The Role of Statins on Helicobacter pylori Eradication: Results from the European Registry on the Management of H. pylori (Hp-EuReg). Antibiotics. 2021; 10(8):965. https://doi.org/10.3390/antibiotics10080965
Chicago/Turabian StyleCaldas, María, Ángeles Pérez-Aisa, Bojan Tepes, Alma Keco-Huerga, Luis Bujanda, Alfredo J. Lucendo, Luis Rodrigo, Dino Vaira, Luis Fernández-Salazar, Jose M. Huguet, and et al. 2021. "The Role of Statins on Helicobacter pylori Eradication: Results from the European Registry on the Management of H. pylori (Hp-EuReg)" Antibiotics 10, no. 8: 965. https://doi.org/10.3390/antibiotics10080965
APA StyleCaldas, M., Pérez-Aisa, Á., Tepes, B., Keco-Huerga, A., Bujanda, L., Lucendo, A. J., Rodrigo, L., Vaira, D., Fernández-Salazar, L., Huguet, J. M., Pérez-Lasala, J., Jurecic, N. B., Fadeenko, G., Barrio, J., Areia, M., Ortuño, J., Pellicano, R., Leja, M., Molina-Infante, J., ... on behalf of the Hp-EuReg Investigators. (2021). The Role of Statins on Helicobacter pylori Eradication: Results from the European Registry on the Management of H. pylori (Hp-EuReg). Antibiotics, 10(8), 965. https://doi.org/10.3390/antibiotics10080965










