Overview and Evaluation of Existing Guidelines for Rational Antimicrobial Use in Small-Animal Veterinary Practice in Europe
Abstract
1. Introduction
2. Materials and Methods
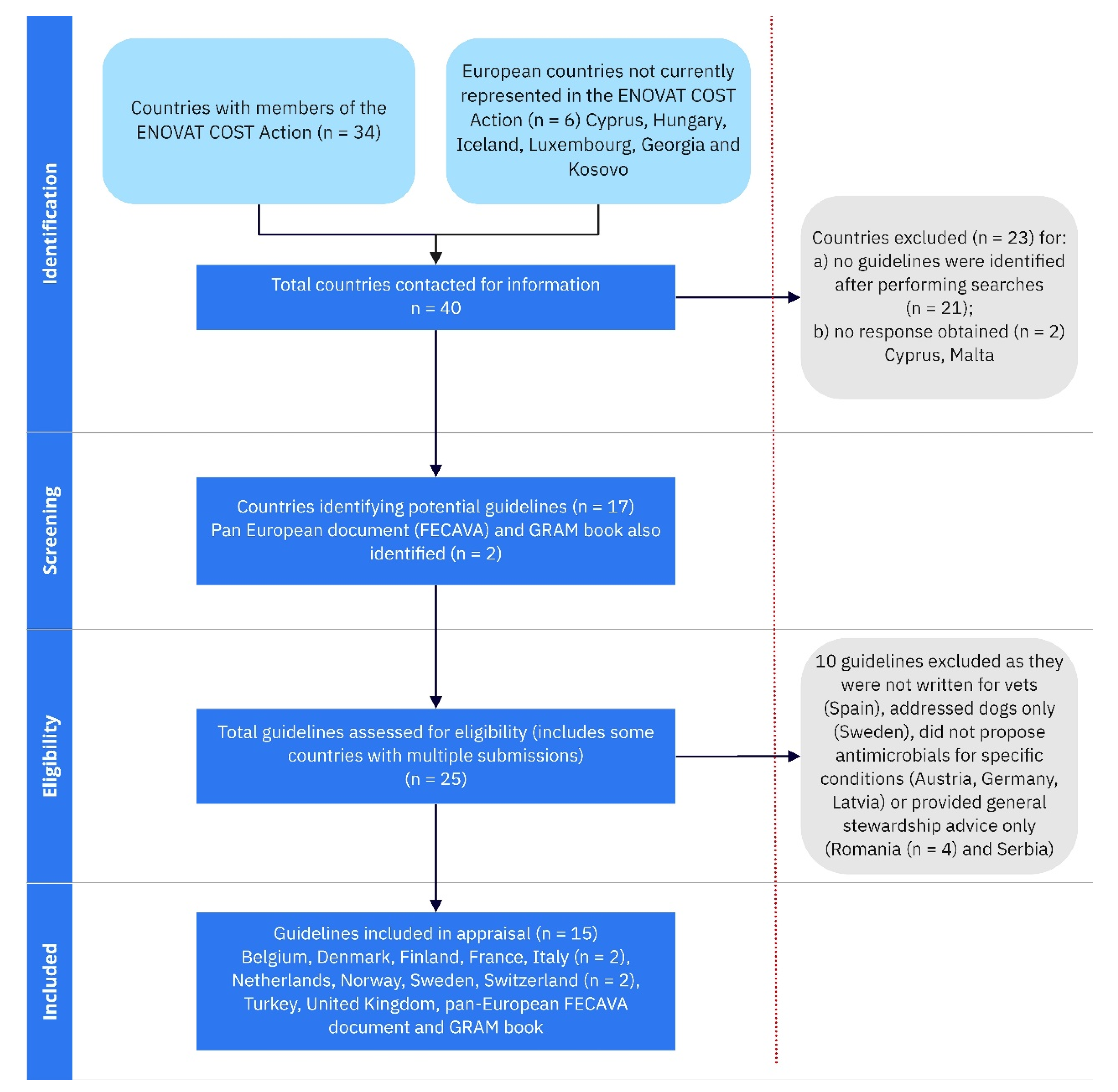
2.1. Identification of Potential Guideline Documents
2.2. Evaluation of Identified Antimicrobial Stewardship Guidelines
3. Results
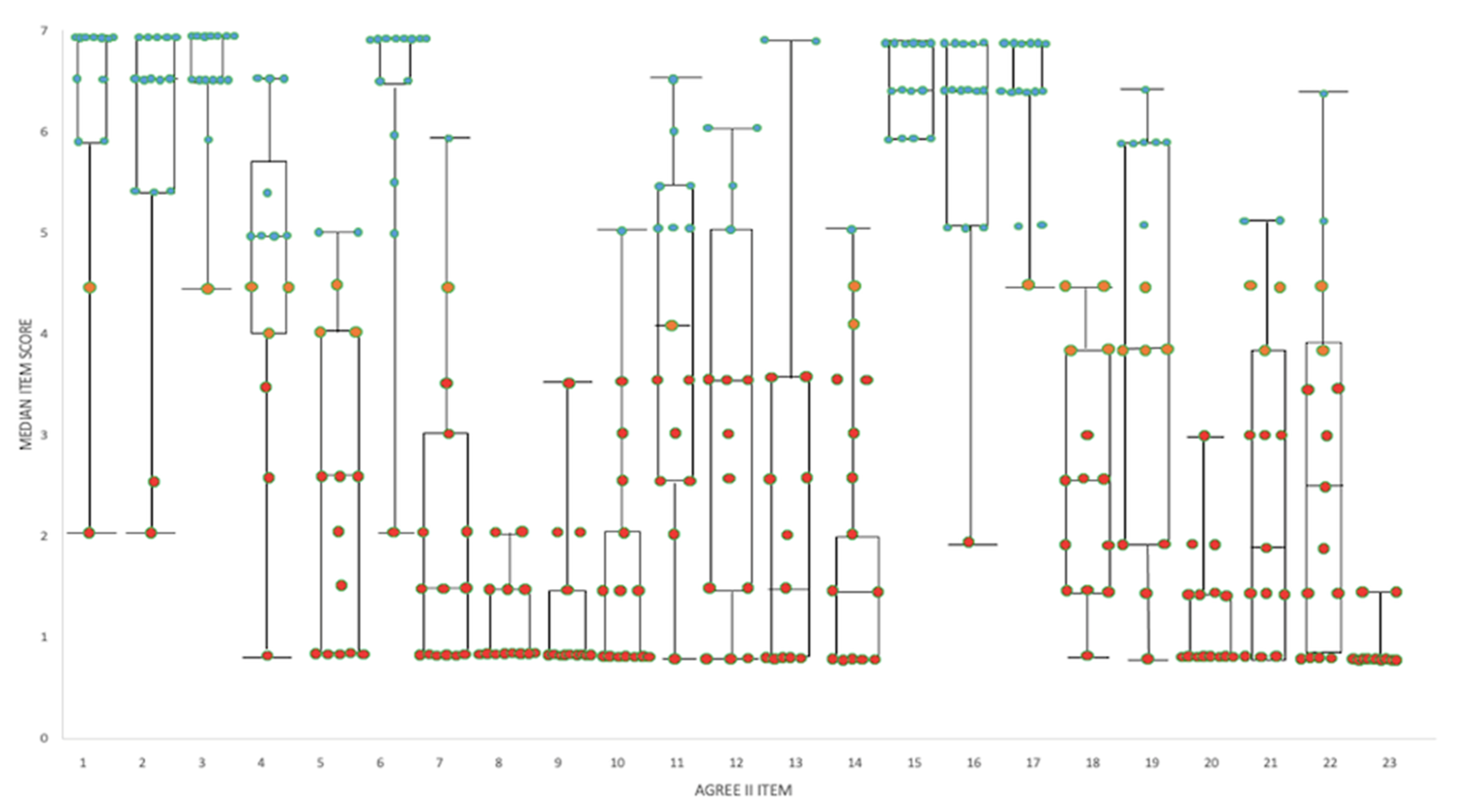
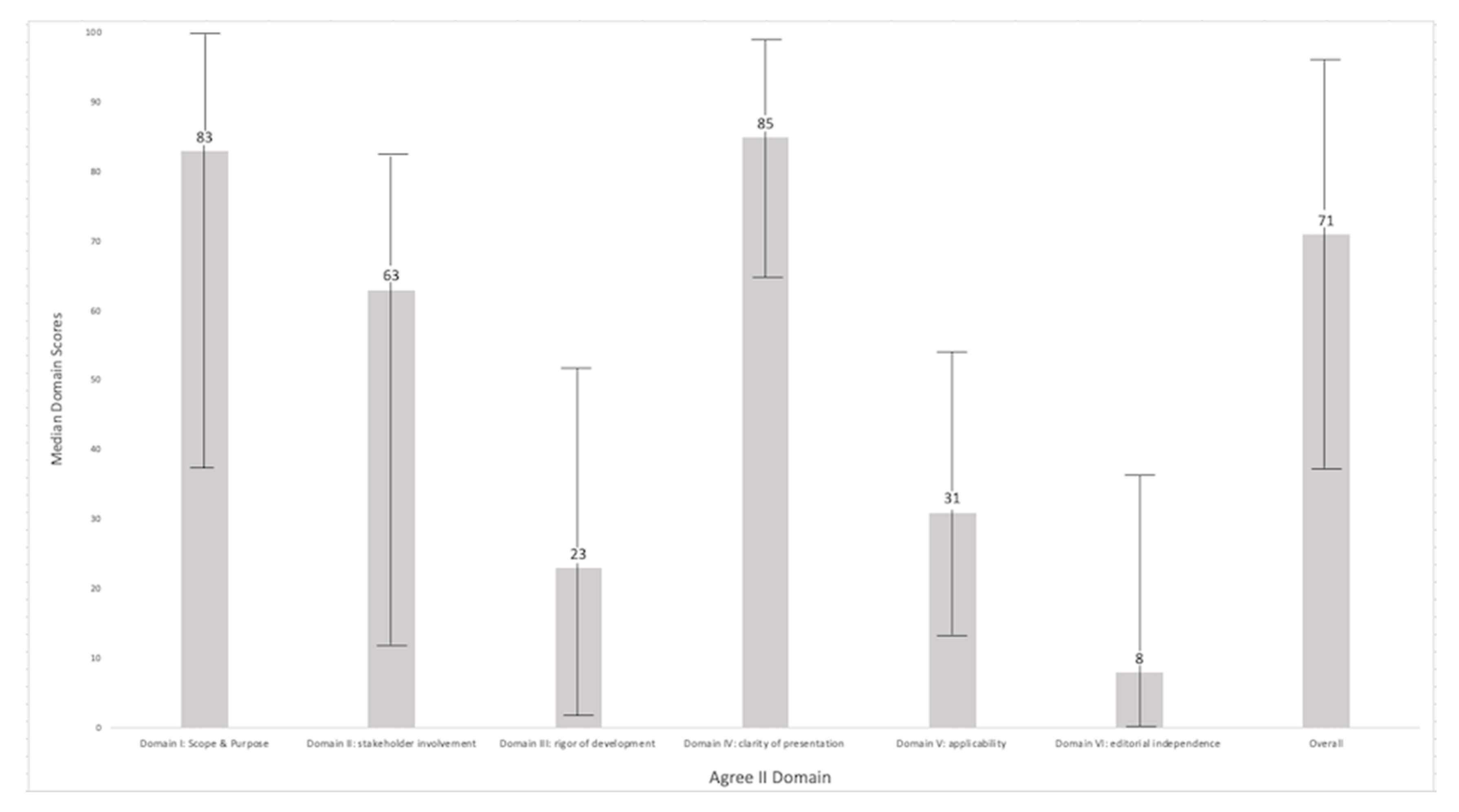
AGREE II Analysis
4. Discussion
5. Practical Impact of Our Findings and Recommendations
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cassini, A.; Diaz Högberg, L.; Plachouras, D.; Quattrocchi, A.; Hoxha, A.; Simonsen, G.S.; Colomb-Cotinat, M.; Kretzschmar, M.E.; Devleesschauwer, B.; Cecchini, M.; et al. Attributable deaths and disability-adjusted life-years caused by infections with antibiotic-resistant bacteria in the EU and the European Economic Area in 2015: A population-level modelling analysis. Lancet Infect. Dis. 2019, 19, 56–66. [Google Scholar] [CrossRef]
- O’Neill, J. Tackling Drug-Resistant Infections Globally: Final Report and Recommendations. In The Review on Antimicrobial Resistance; HM Government: London, UK, 2016. [Google Scholar]
- Klein, E.Y.; Van Boeckel, T.P.; Martinez, E.M.; Pant, S.; Gandra, S.; Levin, S.A.; Goosens, H.; Laxaminarayan, R. Global increase and geographic convergence in antibiotic consumption between 2000 and 2015. Proc. Natl. Acad. Sci. USA 2018, 115, E3463–E3470. [Google Scholar] [CrossRef] [PubMed]
- Woolhouse, M.; Ward, M.; van Bunnik, B.; Farrar, J. Antimicrobial resistance in humans, livestock and the wider environment. Philos. Trans. R. Soc. Lond. Ser. B Biol. Sci. 2015, 370, 20140083. [Google Scholar] [CrossRef]
- European Medicines Agency; European Surveillance of Veterinary Antimicrobial Consumption. Sales of Veterinary Antimicrobial Agents in 31 European Countries in 2018; EMA/24309/2020; European Medicines Agency: Amsterdam, The Netherlands, 2020. [Google Scholar]
- Borck Høg, B.; Bager, F.; Korsgaard, H.B.; Ellis-Iversen, J.; Pedersen, K.; Jensen, L.B.; Hendriksen, R.S.; Bortolaia, V.; Rhod Larsen, A.; Petersen, A.; et al. DANMAP 2017—Use of Antimicrobial Agents and Occurrence of Antimicrobial Resistance in Bacteria from Food Animals, Food and Humans in Denmark; Statens Serum Institut, National Veterinary Institute, Technical University of Denmark: Copenhagen, Denmark; National Food Institute, Technical University of Denmark: Copenhagen, Denmark, 2018; Available online: https://backend.orbit.dtu.dk/ws/files/161713656/Rapport_DANMAP_2017.pdf (accessed on 30 March 2021).
- Ma, G.C.; Worthing, K.A.; Gottlieb, T.; Ward, M.P.; Norris, J.M. Molecular characterization of community-associated methicillin-resistant Staphylococcus aureus from pet dogs. Zoonoses Public Health 2019, 67, 222–230. [Google Scholar] [CrossRef]
- Morris, D.O.; Loeffler, A.; Davis, M.F.; Guardabassi, L.; Weese, J.S. Recommendations for approaches to meticillin-resistant staphylococcal infections of small animals: Diagnosis, therapeutic considerations and preventative measures: Clinical Consensus Guidelines of the World Association for Veterinary Dermatology. Vet. Dermatol. 2017, 28, 304-e69. [Google Scholar] [CrossRef]
- Iseppi, R.; Di Cerbo, A.; Messi, P.; Sabia, C. Antibiotic Resistance and Virulence Traits in Vancomycin-Resistant Enterococci (VRE) and Extended-Spectrum β-Lactamase/AmpC-producing (ESBL/AmpC) Enterobacteriaceae from Humans and Pets. Antibiotics 2020, 9, 152. [Google Scholar] [CrossRef] [PubMed]
- Wada, Y.; Irekeola, A.A.; EA, R.E.N.S.; Yusof, W.; Lih Huey, L.; Ladan Muhammad, S.; Harun, A.; Yean, C.Y.; Zaidah, A.R. Prevalence of Vancomycin-Resistant Enterococcus (VRE) in Companion Animals: The First Meta-Analysis and Systematic Review. Antibiotics 2021, 10, 138. [Google Scholar] [CrossRef]
- Salgado-Caxito, M.; Benavides, J.A.; Adell, A.D.; Paes, A.C.; Moreno-Switt, A.I. Global prevalence and molecular characterization of extended-spectrum β-lactamase producing-Escherichia coli in dogs and cats—A scoping review and meta-analysis. One Health 2021, 12, 100236. [Google Scholar] [CrossRef]
- Pomba, C.; Endimiani, A.; Rossano, A.; Saial, D.; Couto, N.; Perreten, V. First report of OXA-23-mediated carbapenem resistance in sequence type 2 multidrug-resistant Acinetobacter baumannii associated with urinary tract infection in a cat. Antimicrob. Agents Chemother. 2014, 58, 1267–1268. [Google Scholar] [CrossRef]
- Reynolds, M.E.; Phan, H.T.T.; George, S.; Hubbard, A.T.M.; Stoesser, N.; Maciuca, I.E.; Crook, D.W.; Timofte, D. Occurrence and characterization of Escherichia coli ST410 co-harbouring blaNDM-5, blaCMY-42 and blaTEM-190 in a dog from the UK. J. Antimicrob. Chemother. 2019, 74, 1207–1211. [Google Scholar] [CrossRef] [PubMed]
- Davey, P.; Marwick, C.A.; Scott, C.L.; Charani, E.; McNeil, K.; Brown, E.; Gould, I.M.; Ramsay, C.R.; Michie, S. Interventions to improve antibiotic prescribing practices for hospital inpatients. Cochrane Database Syst. Rev. 2017, 2, CD003543. [Google Scholar] [CrossRef]
- Dellit, T.H.; Owens, R.C.; McGowan, J.E., Jr.; Gerding, D.N.; Weinstein, R.A.; Burke, J.P.; Huskins, W.C.; Paterson, D.L.; Fishman, N.O.; Carpenter, P.J.; et al. Infectious Diseases Society of America and the Society for Healthcare Epidemiology of America guidelines for developing an institutional program to enhance antimicrobial stewardship. Clin. Infect. Dis. 2007, 44, 159–177. [Google Scholar] [CrossRef] [PubMed]
- Guardabassi, L.; Prescott, J.F. Antimicrobial Stewardship in Small Animal Veterinary Practice. Vet. Clin. N. Am. Small Anim. Pract. 2015, 45, 361–376. [Google Scholar] [CrossRef] [PubMed]
- Allerton, F.; Jeffery, N. Prescription rebellion: Reduction of antibiotic use by small animal veterinarians. J. Small Anim. Pract. 2020, 61, 148–155. [Google Scholar] [CrossRef] [PubMed]
- Mevius, D.; Heederik, D.J. Reduction of antibiotic use in animals “let’s go Dutch”. J. Für Verbrauch. Und Lebensm. 2014, 9, 177–181. [Google Scholar] [CrossRef]
- Björkman, I.; Röing, M.; Sternberg Lewerin, S.; Stålsby Lundborg, C.; Eriksen, J. Animal Production with Restrictive Use of Antibiotics to Contain Antimicrobial Resistance in Sweden—A Qualitative Study. Front. Vet. Sci. 2021, 7, 619030. [Google Scholar] [CrossRef]
- More, S.J. European perspectives on efforts to reduce antimicrobial usage in food animal production. Ir. Vet. J. 2020, 73, 2. [Google Scholar] [CrossRef]
- Lloyd, D.H.; Page, S.W. Antimicrobial Stewardship in Veterinary Medicine. In Antimicrobial Resistance in Bacteria from Livestock and Companion Animals; Schwarz, S., Cavaco, L.M., Shen, J., Eds.; Wiley: Hoboken, NJ, USA, 2018. [Google Scholar]
- European Parliament, Council of the European Union. Regulation (EU) 2019/6 of the European Parliament and of the Council of 11 December 2018 on veterinary medicinal products and repealing Directive 2001/82/EC (Text with EEA relevance) PE/45/2018/REV/1 OJ L 4, 7.1.2019, p43–167. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX%3A32019R0006&qid=1616378125595 (accessed on 1 March 2021).
- Teale, C.J.; Moulin, G. Prudent use guidelines: A review of existing veterinary guidelines. Rev. Sci. Tech. 2012, 31, 343–354. [Google Scholar] [CrossRef]
- AGREE Next Steps Consortium. The AGREE II Instrument 2017. Available online: http://www.agreetrust.org (accessed on 24 February 2020).
- Hardefeldt, L.; Crabb, H.; Bailey, K.; Johnstone, T.; Gilkerson, J.R.; Billman-Jacobe, H.; Browning, G.F. Appraisal of the Australian Veterinary Prescribing Guidelines for antimicrobial prophylaxis for surgery in dogs and cats. Aust. Vet. J. 2019, 97, 316–322. [Google Scholar] [CrossRef]
- Global Repository of Available Guidelines for Responsible Use of Antimicrobials in Animal Health. Available online: http://www.worldvet.org/news.php?item=417 (accessed on 1 March 2021).
- WHO Handbook for Guideline Development. Available online: https://apps.who.int/iris/bitstream/handle/10665/75146/9789241548441_eng.pdf (accessed on 24 February 2020).
- Cicchetti, D.V. Guidelines, criteria, and rules of thumb for evaluating normed and standardized assessment instruments in psychology. Psychol. Assess. 1994, 6, 284–290. [Google Scholar] [CrossRef]
- Formularium AMCRA (Antimicrobial Consumption and Resistance in Animals). Available online: https://formularium.amcra.be (accessed on 1 March 2021).
- Antibiotic Use Guidelines for Companion Animal Practice (2nd Edition). Available online: https://www.ddd.dk/media/2175/assembled_final.pdf (accessed on 1 March 2021).
- Mikrobilääkkeiden Käyttösuositukset Eläinten Tärkeimpiin Tulehdus- ja Tartuntatauteihin. Available online: https://www.ruokavirasto.fi/globalassets/viljelijat/elaintenpito/elainten-laakitseminen/hallittu_laakekekaytto/mikrobilaakekaytonperiaatteet/mikrobilaakkeiden_kayttosuositukset_en.pdf (accessed on 1 March 2021).
- Guide De Bonnes Pratiques Filière Animaux De Compagnie Fiches De Recommandations Pour Un Bon Usage Des Antibiotiques. Available online: https://www.veterinaire.fr/fileadmin/user_upload/images/CRO/Languedoc-Roussillon/actualites/AB_afvac-fiches-antibiotiques-nov16.pdf (accessed on 1 March 2021).
- Linee Guida. Uso Prudente Dell’antibiotico Negli Animali da Compagnia. Available online: https://www.alimenti-salute.it/sites/default/files/Linee%20Guida%20PETs%202018.pdf (accessed on 1 March 2021).
- ANMVI. Linee Guida sul Corretto uso Degli Antibioticinella Clinica del Cane e del Gatto, 1st ed.; Edizioni Veterinarie; ANMVI: Cremona, Italy, 2018. [Google Scholar]
- Formularium Gezelschapsdieren Hond, kat en Konijn. Available online: https://www.knmvd.nl/app/uploads/sites/4/2018/09/Formularium-Hond-en-Kat.pdf (accessed on 1 March 2021).
- Terapianbefaling: Bruk av Antibakterielle Midler til Hund og Katt. Available online: https://legemiddelverket.no/Documents/Veterinærmedisin/Terapianbefalinger/Terapianbefaling_Antibakterielle%20midler%20hund%20og%20katt_2014_N.pdf (accessed on 1 March 2021).
- Guidelines for the Clinical Use of Antibiotics in the Treatment of Dogs and Cats. Available online: https://www.svf.se/media/ahwpbt52/policy-ab-english-10b.pdf (accessed on 1 March 2021).
- Therapieleitfaden für Tierärztinnen und Tierärzte—Hunde und Katzen. Available online: https://docplayer.org/128033855-Therapieleitfaden-fuer-tieraerztinnen-und-tieraerzte.html (accessed on 1 March 2021).
- AntibioticScout.ch. Available online: https://www.vetpharm.uzh.ch/abscout/ (accessed on 1 March 2021).
- Güneş Tıp Kitabevi. Veteriner Hekimlikte Antibiyotikler (Pratik Bilgiler Rehberi), 2nd ed.; Yarsan, E., Ed.; Güneş Tıp Kitabevi: Ankara, Turkey, 2017. [Google Scholar]
- PROTECT ME Poster. Available online: https://www.bsavalibrary.com/docserver/fulltext/10.22233/9781910443644/bsava_samsoc_protectmeposter_011118web.pdf?expires=1615760031&id=id&accname=guest&checksum=5DC3FBEB23A81F11F3997F55B04CFFCC (accessed on 1 March 2021).
- Federation of European Companion Animal Veterinary Associations Recommendations for Appropriate Antimicrobial Therapy. Available online: https://www.fecava.org/wp-content/uploads/2020/01/FECAVA-Recommendations-for-Appropriate-Antimicrobial-ENGLISH-1.pdf (accessed on 1 March 2021).
- Guidance for the Rational Use of Antimicrobials (GRAM) Book. CEVA Sante Animale. 2016. Available online: https://www.ava.com.au/siteassets/advocacy/gram-book---guidance-for-the-rational-use-of-antimicrobials.pdf (accessed on 1 March 2021).
- Hillier, A.; Lloyd, D.H.; Weese, J.S.; Blondeau, J.M.; Boothe, D.; Breitschwerdt, E.; Guardabassi, L.; Papich, M.G.; Rankin, S.; Turnidge, J.D.; et al. Guidelines for the diagnosis and antimicrobial therapy of canine superficial bacterial folliculitis (Antimicrobial Guidelines Working Group of the International Society for Companion Animal Infectious Diseases). Vet. Dermatol. 2014, 25, 163-e43. [Google Scholar] [CrossRef]
- Weese, J.; Giguère, S.; Guardabassi, L.; Morley, P.; Papich, M.; Ricciuto, D.; Sykes, J. ACVIM Consensus Statement on Therapeutic Antimicrobial Use in Animals and Antimicrobial Resistance. J. Vet. Intern. Med. 2015, 29, 487–498. [Google Scholar] [CrossRef]
- Lappin, M.R.; Blondeau, J.; Boothe, D.; Breitschwerdt, E.B.; Guardabassi, L.; Lloyd, D.H.; Papich, M.G.; Rankin, S.C.; Sykes, J.E.; Turnidge, J.; et al. Antimicrobial use guidelines for treatment of respiratory tract disease in dogs and cats: Antimicrobial Guidelines Working Group of the International Society for Companion Animal Infectious Diseases. J. Vet. Intern. Med. 2017, 31, 279–294. [Google Scholar] [CrossRef] [PubMed]
- Weese, J.S.; Blondeau, J.; Boothe, D.; Guardabassi, L.G.; Gumley, N.; Papich, M.; Jessen, L.R.; Lappin, M.; Rankin, S.; Westropp, J.L.; et al. International Society for Companion Animal Infectious Diseases (ISCAID) guidelines for the diagnosis and management of bacterial urinary tract infections in dogs and cats. Vet. J. 2019, 247, 8–25. [Google Scholar] [CrossRef]
- Greenhalgh, T.; Gill, P. Pressure to prescribe involves a complex interplay of factors. BMJ 1997, 315, 1482. [Google Scholar] [CrossRef]
- Hopman, N.E.M.; Hulscher, M.; Graveland, H.; Speksnijder, D.C.; Wagenaar, J.A.; Broens, E.M. Factors influencing antimicrobial prescribing by Dutch companion animal veterinarians: A qualitative study. Prev. Vet. Med. 2018, 158, 106–113. [Google Scholar] [CrossRef] [PubMed]
- Zhuo, A.; Labbate, M.; Norris, J.M.; Gilbert, G.L.; Ward, M.P.; Bajorek, B.V.; Degeling, C.; Rowbotham, S.J.; Dawson, A.; Nguyen, K.A.; et al. Opportunities and challenges to improving antibiotic prescribing practices through a One Health approach: Results of a comparative survey of doctors, dentists and veterinarians in Australia. BMJ Open 2018, 8, e020439. [Google Scholar] [CrossRef]
- Timofte, D.; Broens, E.M.; Guardabassi, L.; Pomba, C.; Allerton, F.; Ikonomopoulos, J.; Overesch, G.; Damborg, P.; European Network for Optimisation of Veterinary Antimicrobial Treatment (ENOVAT); ESCMID Study Group for Veterinary Microbiology (ESGVM); et al. Driving laboratory standardisation of bacterial culture and antimicrobial susceptibility testing in veterinary clinical microbiology in Europe and beyond. J. Clin. Microbiol. 2020. [Google Scholar] [CrossRef]
- Guardabassi, L.; Apley, M.; Olsen, J.E.; Toutain, P.-L.; Weese, S. Optimization of Antimicrobial Treatment to Minimize Resistance Selection. Microbiol. Spectr. 2018, 6. [Google Scholar] [CrossRef]
- Burke, S.; Black, V.; Sánchez-Vizcaíno, F.; Radford, A.; Hibbert, A.; Tasker, S. Use of cefovecin in a UK population of cats attending first opinion practices as recorded in electronic health records. J. Feline Med. Surg. 2017, 19, 687–692. [Google Scholar] [CrossRef] [PubMed]
- Escher, M.; Vanni, M.; Intorre, L.; Caprioli, A.; Tognetti, R.; Scavia, G. Use of antimicrobials in companion animal practice: A retrospective study in a veterinary teaching hospital in Italy. J. Antimicrob. Chemother 2011, 66, 920–927. [Google Scholar] [CrossRef] [PubMed]
- De Briyne, N.; Atkinson, J.; Pokludová, L.; Borriello, S.P.; Price, S. Factors influencing antibiotic prescribing habits and use of sensitivity testing amongst veterinarians in Europe. Vet. Rec. 2013, 173, 475. [Google Scholar] [CrossRef] [PubMed]
- Varadi, L.; Luo, J.L.; Hibbs, D.E.; Perry, J.D.; Anderson, R.J.; Orenga, S.; Groundwater, P.W. Methods for the detection and identification of pathogenic bacteria: Past, present, and future. Chem. Soc. Rev. 2017, 46, 4818–4832. [Google Scholar] [CrossRef]
- Buckland, E.L.; O’Neill, D.; Summers, J.; Mateus, A.; Church, D.; Redmond, L.; Brodbelt, D. Characterisation of antimicrobial usage in cats and dogs attending UK primary care companion animal veterinary practices. Vet. Rec. 2016, 179, 489. [Google Scholar] [CrossRef]
- Norris, J.M.; Zhuo, A.; Govendir, M.; Rowbotham, S.J.; Labbate, M.; Degeling, C.; Gilbert, G.L.; Dominey-Howes, D.; Ward, M.P. Factors influencing the behaviour and perceptions of Australian veterinarians towards antibiotic use and antimicrobial resistance. PLoS ONE 2019, 14, e0223534. [Google Scholar]
- Singleton, D.A.; Sánchez-Vizcaínoa, F.; Dawon, S.; Jones, P.H.; Noble, P.J.M.; Pinchbeck, G.L.; Williams, N.J.; Radford, A.D. Patterns of anti-microbial agent prescription in a sentinel population of canine and feline veterinary practices in the United Kingdom. Vet. J. 2017, 224, 18–24. [Google Scholar] [CrossRef]
- Robinson, N.J.; Dean, R.S.; Cobb, M.; Brennan, M.L. Factors influencing common diagnoses made during first-opinion small-animal consultations in the United Kingdom. Prev. Vet. Med. 2016, 131, 87–94. [Google Scholar] [CrossRef]
- Summers, J.F.; Hendricks, A.; Brodbelt, D.C. Prescribing practices of primary-care veterinary practitioners in dogs diagnosed with bacterial pyoderma. BMC Vet. Res. 2014, 10, 240. [Google Scholar] [CrossRef]
- Magalhães-Sant’Ana, M. Resisting the urge to prescribe vancomycin. In Pract. 2017, 39, 294–295. [Google Scholar] [CrossRef]
- McCulloch, S. Comments on the dilemma in the June issue: Resisting the urge to prescribe vancomycin. In Pract. 2017, 39, 343. [Google Scholar] [CrossRef]
- WHO. Critically Important Antimicrobials for Human Medicine—6th Review; 6th Revision 2019; World Health Organisation: Geneva, Switzerland, 2019. [Google Scholar]
- EMA/AMEG. Categorisation of Antibiotics in the European Union. EMA/CVMP/CHMP/682198/2017. 2019. Available online: https://www.ema.europa.eu/en/documents/report/categorisation-antibiotics-european-union-answer-request-european-commission-updating-scientific_en.pdf (accessed on 24 February 2020).
- Spellberg, B. The New Antimicrobial Mantra—“Shorter Is Better”. JAMA Intern. Med. 2016, 176, 1254–1255. [Google Scholar] [CrossRef]
- Spellberg, B.; Rice, L.B. Duration of Antimicrobial Therapy: Shorter is Better. Ann. Intern. Med. 2019, 171, 210–211. [Google Scholar] [CrossRef] [PubMed]
- Weese, J.S.; Blondeau, J.M.; Boothe, D.; Breitschwerdt, E.B.; Guardabassi, L.; Hillier, A.; Lloyd, D.H.; Papich, M.G.; Rankin, S.C.; Turnidge, J.D.; et al. Antimicrobial use guidelines for treatment of urinary tract disease in dogs and cats: Antimicrobial guidelines working group of the international society for companion animal infectious diseases. Vet. Med. Int. 2011, 2011, 263768. [Google Scholar] [CrossRef] [PubMed]
- Pouwels, K.B.; Hopkins, S.; Llewelyn, M.J.; Walker, A.S.; McNulty, C.A.M.; Robotham, J.V. Duration of antibiotic treatment for common infections in English primary care: Cross sectional analysis and comparison with guidelines. BMJ 2019, 364, 1440. [Google Scholar] [CrossRef]
- Viitanen, S.J.; Lappalainen, A.K.; Christensen, M.B.; Sankari, S.; Rajamäki, M.M. The Utility of Acute-Phase Proteins in the Assessment of Treatment Response in Dogs with Bacterial Pneumonia. J. Vet. Intern. Med. 2017, 31, 124–133. [Google Scholar] [CrossRef] [PubMed]
- Sinclair, D.; Isba, R.; Kredo, T.; Zani, B.; Smith, H.; Garner, P. World Health Organization Guideline Development: An Evaluation. PLoS ONE 2013, 8, e63715. [Google Scholar] [CrossRef]
- WHO. Guidelines for WHO Guidelines; WHO Press, World Health Organization: Geneva, Switzerland, 2003; Available online: http://whqlibdoc.who.int/hq/2003/EIP_GPE_EQC_2003_1.pdf (accessed on 1 March 2021).
- Guyatt, G.H.; Oxman, A.D.; Vist, G.E.; Kunz, R.; Falck-Ytter, Y.; Alonso-Coello, P.; Schünemann, H.J. GRADE: An emerging consensus on rating quality of evidence and strength of recommendations. BMJ 2008, 336, 924–926. [Google Scholar] [CrossRef] [PubMed]
- Guyatt, G.; Oxman, A.D.; Akl, E.A.; Kunz, R.; Vist, G.; Jan, B.; Norris, S.; Falck-Ytter, Y.; Glasziou, P.; DeBeer, H.; et al. GRADE guidelines: 1. Introduction-GRADE evidence profiles and summary of findings tables. J. Clin. Epidemiol. 2011, 64, 383–394. [Google Scholar] [CrossRef]
- Brouwers, M.C.; Kho, M.E.; Browman, G.P.; Burgers, J.S.; Cluzeau, F.; Feder, G.; Fervers, B.; Graham, I.D.; Grimshaw, J.; Hanna, S.E.; et al. AGREE II: Advancing guideline development, reporting and evaluation in health care. Can. Med Assoc. J. 2010, 182, E839–E842. [Google Scholar] [CrossRef]
- Petkovic, J.; Riddle, A.; Akl, E.A.; Khabsa, J.; Lytvyn, L.; Atwere, P.; Campbell, P.; Chalkidou, K.; Chang, S.M.; Crowe, S.; et al. Protocol for the development of guidance for stakeholder engagement in health and healthcare guideline development and implementation. Syst. Rev. 2020, 9, 21. [Google Scholar] [CrossRef]
- Wathne, J.S.; Kleppe, L.K.S.; Harthug, S.; Blix, H.S.; Nilsen, R.M.; Charani, E.; The Bergen Intervenion Teams; Smith, I. The effect of antibiotic stewardship interventions with stakeholder involvement in hospital settings: A multicentre, cluster randomized controlled intervention study. Antimicrob. Resist. Infect. Control. 2018, 7, 109. [Google Scholar] [CrossRef]
- Rees, G.M.; Bard, A.; Reyher, K.K. Designing a National Veterinary Prescribing Champion Programme for Welsh Veterinary Practices: The Arwain Vet Cymru Project. Antibiotics 2021, 10, 253. [Google Scholar] [CrossRef] [PubMed]
- Browman, G.P.; Somerfield, M.R.; Lyman, G.H.; Brouwers, M.C. When is good, good enough? Methodological pragmatism for sustainable guideline development. Implement. Sci. 2015, 10, 28. [Google Scholar] [CrossRef]
- Landgren, F.T.; Harvey, K.J.; Moulds, R.F.W.; Mashford, M.L.; Guthrie, B.; Hemming, M. Changing antibiotic prescribing by educational marketing. Med. J. Aust. 1988, 149, 595–599. [Google Scholar] [CrossRef]
- Donisi, V.; Sibani, M.; Carrara, E.; Del Piccolo, L.; Rimondini, M.; Mazzaferri, F.; Bovo, C.; Tacconelli, E. Emotional, cognitive and social factors of antimicrobial prescribing: Can antimicrobial stewardship intervention be effective without addressing psycho-social factors? J. Antimicrob. Chemother. 2019, 74, 2844–2847. [Google Scholar] [CrossRef] [PubMed]
- Hardefeldt, L.Y.; Gilkerson, J.R.; Billman-Jacobe, H.; Stevenson, M.A.; Thursky, K.; Bailey, K.E.; Browning, G.F. Barriers to and enablers of implementing antimicrobial stewardship programs in veterinary practices. J. Vet. Intern. Med. 2018, 32, 1092–1099. [Google Scholar] [CrossRef] [PubMed]
- Lam, T.T.; Dang, D.A.; Tran, H.H.; Van Do, D.; Le, H.; Negin, J.; Jan, S.; Marks, G.B.; Nguyen, T.A.; Fox, G.J.; et al. What are the most effective community-based antimicrobial stewardship interventions in low- and middle-income countries? A narrative review. J. Antimicrob. Chemother. 2021, dkaa556. [Google Scholar] [CrossRef]
- Drekonja, D.; Filice, G.A.; Greer, N.; Olson, A.; MacDonald, R.; Rutks, I.; Wilt, T.J. Antimicrobial Stewardship in Outpatient Settings: A Systematic Review. Infect. Control. Hosp. Epidemiol. 2015, 36, 142–152. [Google Scholar] [CrossRef]
- Wagner, B.; Filice, G.A.; Drekonja, D.; Greer, N.; MacDonald, R.; Rutks, I.; Butler, M.; Wilt, T.J. Antimicrobial Stewardship Programs in Inpatient Hospital Settings: A Systematic Review. Infect. Control. Hosp. Epidemiol. 2015, 35, 1209–1228. [Google Scholar] [CrossRef]
- Lightman, J.; Battersby, I. Preliminary findings of a survey of UK small animal general practitioners to describe antibiotic use. In BSAVA Congress Proceedings 2018; BSAVA: Birmingham, UK, 2018; pp. 417–418. [Google Scholar]
- Lutz, B.; Lehner, C.; Schmitt, K.; Willi, B.; Schüpbach, G.; Mevissen, M.; Peter, R.; Müntener, C.; Naegeli, H.; Schuller, S. Antimicrobial prescriptions and adherence to prudent use guidelines for selected canine diseases in Switzerland in 2016. Vet. Rec. Open 2020, 7, e000370. [Google Scholar] [CrossRef]
- Weese, J.S. Investigation of antimicrobial use and the impact of antimicrobial use guidelines in a small animal veterinary teaching hospital: 1995–2004. J. Am. Vet. Med. Assoc. 2006, 228, 553–558. [Google Scholar] [CrossRef]
- Jessen, L.R.; Sørensen, T.M.; Lilja, Z.L.; Kristensen, M.; Hald, T.; Damborg, P. Cross-sectional survey on the use and impact of the Danish national antibiotic use guidelines for companion animal practice. Acta Vet. Scand. 2017, 59, 81. [Google Scholar] [CrossRef]
- Hopman, N.E.M.; Portengen, L.; Hulscher, M.E.J.L.; Heederik, D.J.J.; Verheij, T.J.M.; Wagenaar, J.A.; Prins, j.; Bosje, T.; Schipper, L.; van Geijlswijk, I.M.; et al. Implementation and evaluation of an antimicrobial stewardship programme in companion animal clinics: A stepped-wedge design intervention study. PLoS ONE 2019, 14, e0225124. [Google Scholar] [CrossRef] [PubMed]
- Sarrazin, S.; Vandael, F.; Van Cleven, A.; De Graef, E.; De Rooster, H.; Dewulf, J. De impact van advies omtrent het gebruik van antimicrobiële middelen op het voorschrijfgedrag in veertien Vlaamse praktijken voor kleine huisdieren. (The impact of antimicrobial use guidelines on prescription habits in fourteen Flemish small animal practices). Vlaams Diergeneeskundig Tijdschrift 2017, 86, 173. [Google Scholar]
- UK-VARSS. Veterinary Antibiotic Resistance and Sales Surveillance Report (UK-VARSS 2019); Veterinary Medicines Directorate: New Haw, UK, 2020. [Google Scholar]
- Overview of Farm-Level AMU Monitoring Systems. Available online: https://aacting.org/monitoring-systems (accessed on 1 March 2021).
| Country | Last Updated | Antimicrobial Stewardship Guidelines (ASGs) |
|---|---|---|
| Belgium | 2020 [29] | Formularium Antimicrobial Consumption and Resistance in Animals (AMCRA) |
| Denmark | 2018 [30] | Antibiotic Use Guidelines for Companion Animal Practice (2nd Edition) * |
| Finland | 2018 [31] | Mikrobilääkkeiden käyttösuositukset eläinten tärkeimpiin tulehdus- ja tartuntatauteihin * |
| France | 2017 [32] | Guide De Bonnes Pratiques Filière Animaux De Compagnie Fiches De Recommandations Pour Un Bon Usage Des Antibiotiques 2017 |
| Italy | 2017 [33] | Linee guida. Uso prudente dell’antibiotico negli animali da compagnia |
| 2017 [34] | Linee Guida sul corretto uso degli antibioticinella clinica del cane e del gatto | |
| Netherlands | 2017 [35] | Formularium gezelschapsdieren hond, kat en konijn |
| Norway | 2014 [36] | Terapianbefaling: Bruk av antibakterielle midler til hund og katt |
| Sweden | 2010 [37] | Guidelines for the clinical use of antibiotics in the treatment of dogs and cats * |
| Switzerland | 2019 [38] | Therapieleitfaden für Tierärztinnen und Tierärzte—Hunde und Katzen |
| 2020 [39] | AntibioticScout.ch | |
| Turkey | 2017 [40] | Veteriner Hekimlikte Antibiyotikler (Pratik Bilgiler Rehberi) 2nd Edition |
| United Kingdom | 2018 [41] | PROTECT ME poster * |
| FECAVA | 2018 [42] | FECAVA Recommendations for Appropriate Antimicrobial Therapy * |
| GRAM book | 2016 [43] | Guidance for the rational use of antimicrobials * |
| Recommendation | Number of ASGs (n = 15) | Percentage of ASGs (%) |
|---|---|---|
| Antimicrobials are not indicated for management of: | ||
| Acute diarrhea | 15 | 100 |
| Clean/elective surgical procedures | 13 | 87 |
| Feline lower urinary tract disease | 11 | 73 |
| subclinical bacteriuria | 8 | 53 |
| Non-antimicrobial therapeutic options described | 14 | 93 |
| Use topical medication instead of systemic medication where appropriate | 15 | 100 |
| Select narrow over broad-spectrum antimicrobials or encourage de-escalation to a narrower spectrum | 13 | 87 |
| Avoid certain antimicrobials reserved for human use only, e.g., vancomycin or carbapenems | 12 | 80 |
| Mention highest priority critically important antimicrobials (HPCIAs) | 10 | 66 |
| Tier antimicrobial suggestions (first line, second line) | 13 | 87 |
| Promote use of diagnostic techniques (cytology/culture) to identify putative bacteria | 15 | 100 |
| List common pathogens found in specific conditions | 14 | 93 |
| Monitor local antimicrobial resistance patterns | 5 | 33 |
| Audit/monitor individual/practice AMU | 8 | 53 |
| AGREE II Item | ICC | 95% Confidence Interval | Correlation | ||
|---|---|---|---|---|---|
| 1 | The overall objective(s) of the guideline is (are) specifically described | 0.64 | 0.21 | 0.86 | Good |
| 2 | The clinical question(s) covered by the guideline is (are) specifically described | 0.28 | −0.57 | 0.73 | Poor |
| 3 | The patients to whom the guideline is meant to apply are specifically described | 0.52 | −0.04 | 0.82 | Fair |
| 4 | The guideline development group includes individuals from all the relevant professional groups | 0.71 | 0.36 | 0.89 | Good |
| 5 | The patients’ views and preferences have been sought | 0.16 | −0.82 | 0.68 | Poor |
| 6 | The target users of the guideline are clearly defined | 0.57 | 0.06 | 0.84 | Fair |
| 7 | Systematic methods were used to search for evidence | 0.42 | 0.26 | 0.78 | Fair |
| 8 | The criteria for selecting the evidence are clearly described | −0.25 | −1.9 | 0.54 | Poor |
| 9 | The strengths and limitations of the body of evidence are clearly described | 0.41 | −0.28 | 0.78 | Fair |
| 10 | The methods for formulating the recommendations are clearly described | 0.56 | 0.03 | 0.83 | Fair |
| 11 | The health benefits, side effects, and risks have been considered in formulating the recommendations | 0.31 | −0.49 | 0.74 | Poor |
| 12 | There is an explicit link between the recommendations and the supporting evidence | 0.58 | 0.08 | 0.84 | Fair |
| 13 | The guideline has been externally reviewed by experts prior to its publication | 0.70 | 0.34 | 0.89 | Good |
| 14 | A procedure for updating the guideline is provided | 0.42 | −0.24 | 0.72 | Fair |
| 15 | The recommendations are specific and unambiguous | 0.52 | −0.4 | 0.84 | Fair |
| 16 | The different options for management of the condition are clearly presented | 0.54 | −0.01 | 0.83 | Fair |
| 17 | Key recommendations are easily identifiable | 0.15 | −0.85 | 0.68 | Poor |
| 18 | The guideline is supported with tools for application | 0.39 | −0.42 | 0.78 | Poor |
| 19 | The potential organizational barriers in applying the recommendations have been discussed | 0.42 | −0.25 | 0.78 | Fair |
| 20 | The potential cost implications of applying the recommendations have been considered | 0.02 | −1.14 | 0.63 | Poor |
| 21 | The guideline presents key review criteria for monitoring and/or audit purposes | 0.12 | −0.91 | 0.67 | Poor |
| 22 | The guideline is editorially independent from the funding body | 0.42 | −0.37 | 0.79 | Fair |
| 23 | Conflicts of interest of guideline development members have been recorded | 0.58 | 0.01 | 0.85 | Fair |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Allerton, F.; Prior, C.; Bagcigil, A.F.; Broens, E.; Callens, B.; Damborg, P.; Dewulf, J.; Filippitzi, M.-E.; Carmo, L.P.; Gómez-Raja, J.; et al. Overview and Evaluation of Existing Guidelines for Rational Antimicrobial Use in Small-Animal Veterinary Practice in Europe. Antibiotics 2021, 10, 409. https://doi.org/10.3390/antibiotics10040409
Allerton F, Prior C, Bagcigil AF, Broens E, Callens B, Damborg P, Dewulf J, Filippitzi M-E, Carmo LP, Gómez-Raja J, et al. Overview and Evaluation of Existing Guidelines for Rational Antimicrobial Use in Small-Animal Veterinary Practice in Europe. Antibiotics. 2021; 10(4):409. https://doi.org/10.3390/antibiotics10040409
Chicago/Turabian StyleAllerton, Fergus, Cameron Prior, Arzu Funda Bagcigil, Els Broens, Bénédicte Callens, Peter Damborg, Jeroen Dewulf, Maria-Eleni Filippitzi, Luís Pedro Carmo, Jonathan Gómez-Raja, and et al. 2021. "Overview and Evaluation of Existing Guidelines for Rational Antimicrobial Use in Small-Animal Veterinary Practice in Europe" Antibiotics 10, no. 4: 409. https://doi.org/10.3390/antibiotics10040409
APA StyleAllerton, F., Prior, C., Bagcigil, A. F., Broens, E., Callens, B., Damborg, P., Dewulf, J., Filippitzi, M.-E., Carmo, L. P., Gómez-Raja, J., Harpaz, E., Mateus, A., Nolff, M., Phythian, C. J., Timofte, D., Zendri, F., & Jessen, L. R. (2021). Overview and Evaluation of Existing Guidelines for Rational Antimicrobial Use in Small-Animal Veterinary Practice in Europe. Antibiotics, 10(4), 409. https://doi.org/10.3390/antibiotics10040409







