Poultry Litter Contamination by Escherichia coli Resistant to Critically Important Antimicrobials for Human and Animal Use and Risk for Public Health in Cameroon
Abstract
1. Introduction
2. Results
2.1. Farmers Demographic Surveyed in Centre, Littoral and West Region of Cameroon
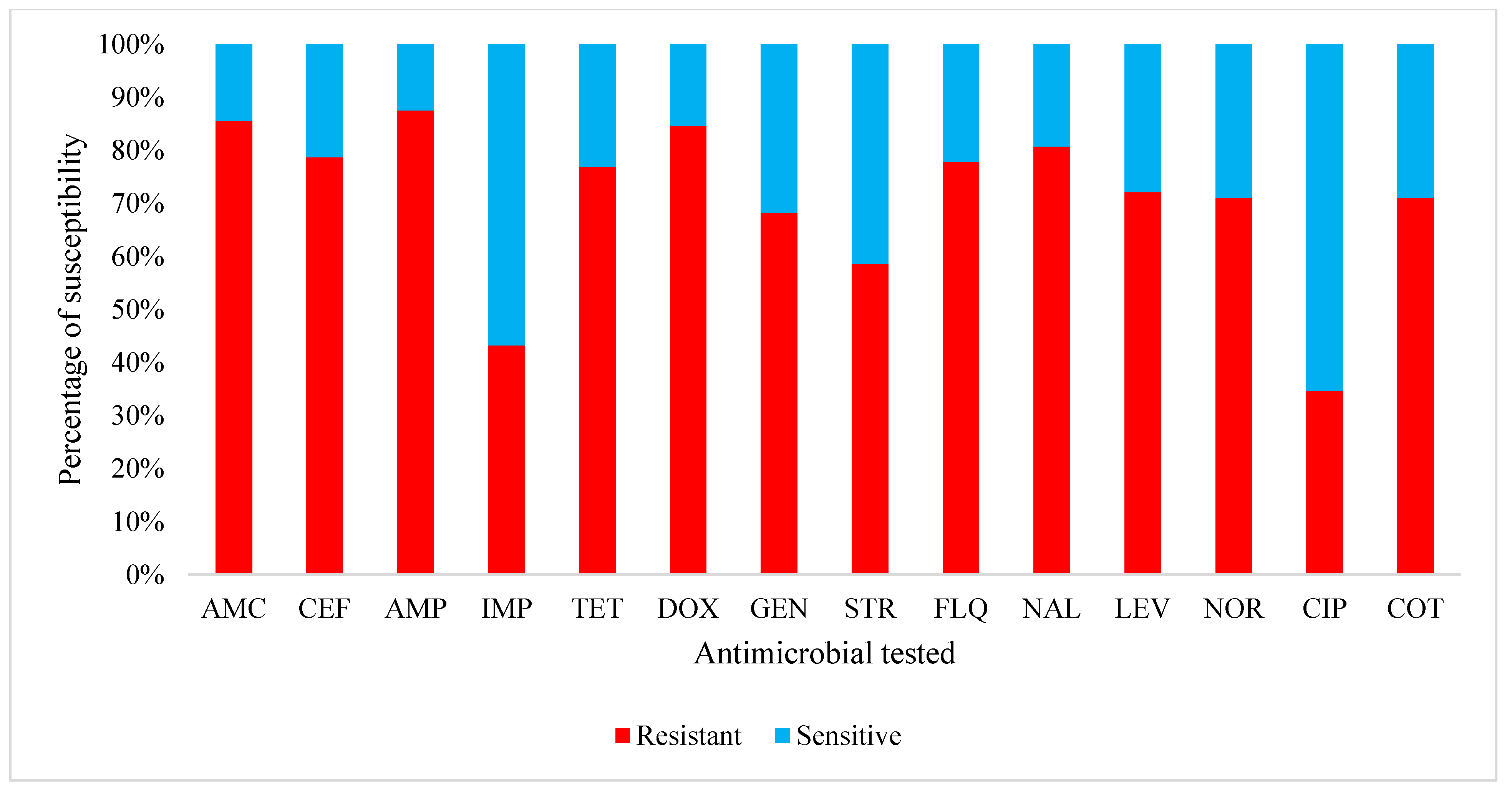
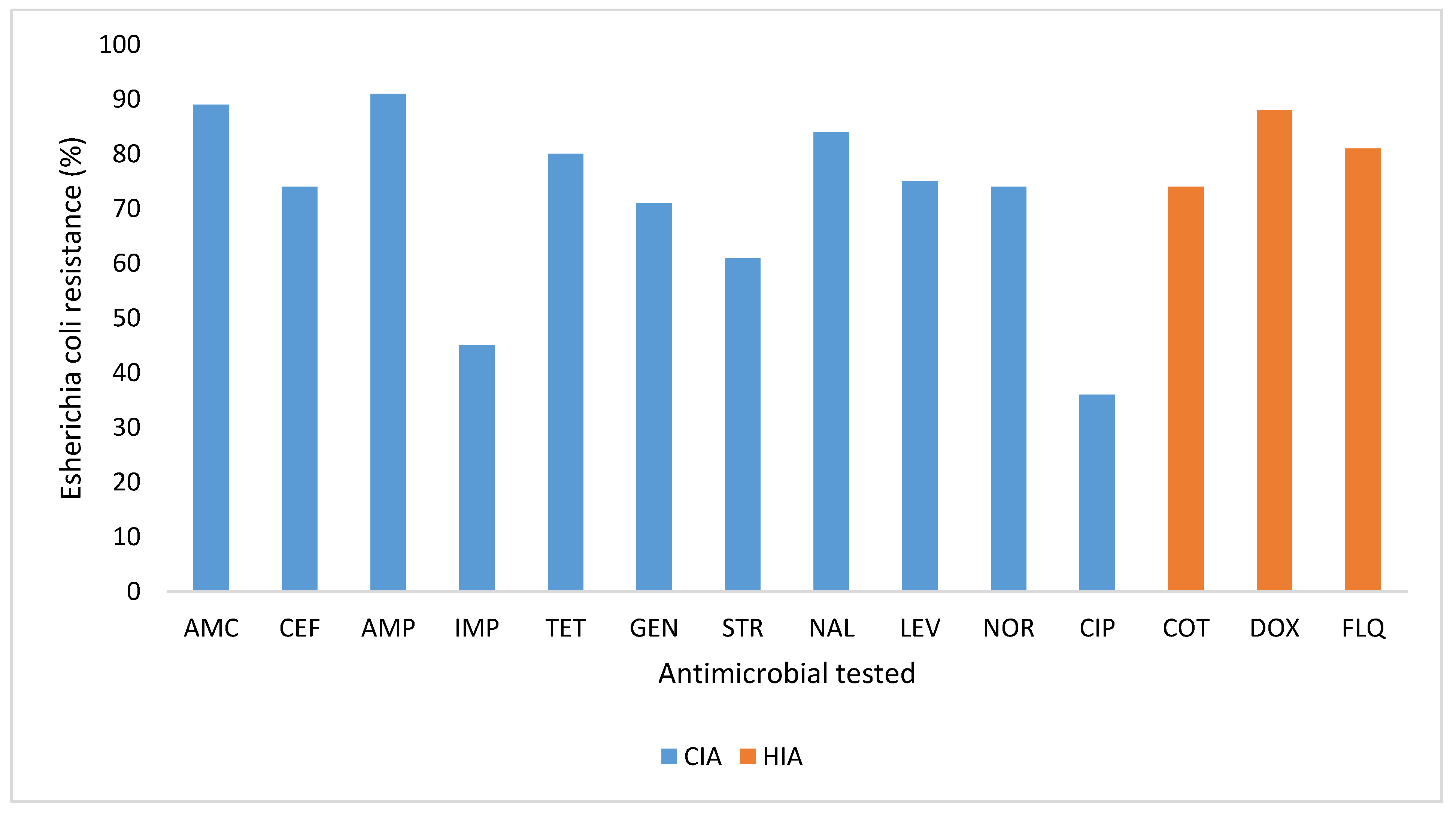
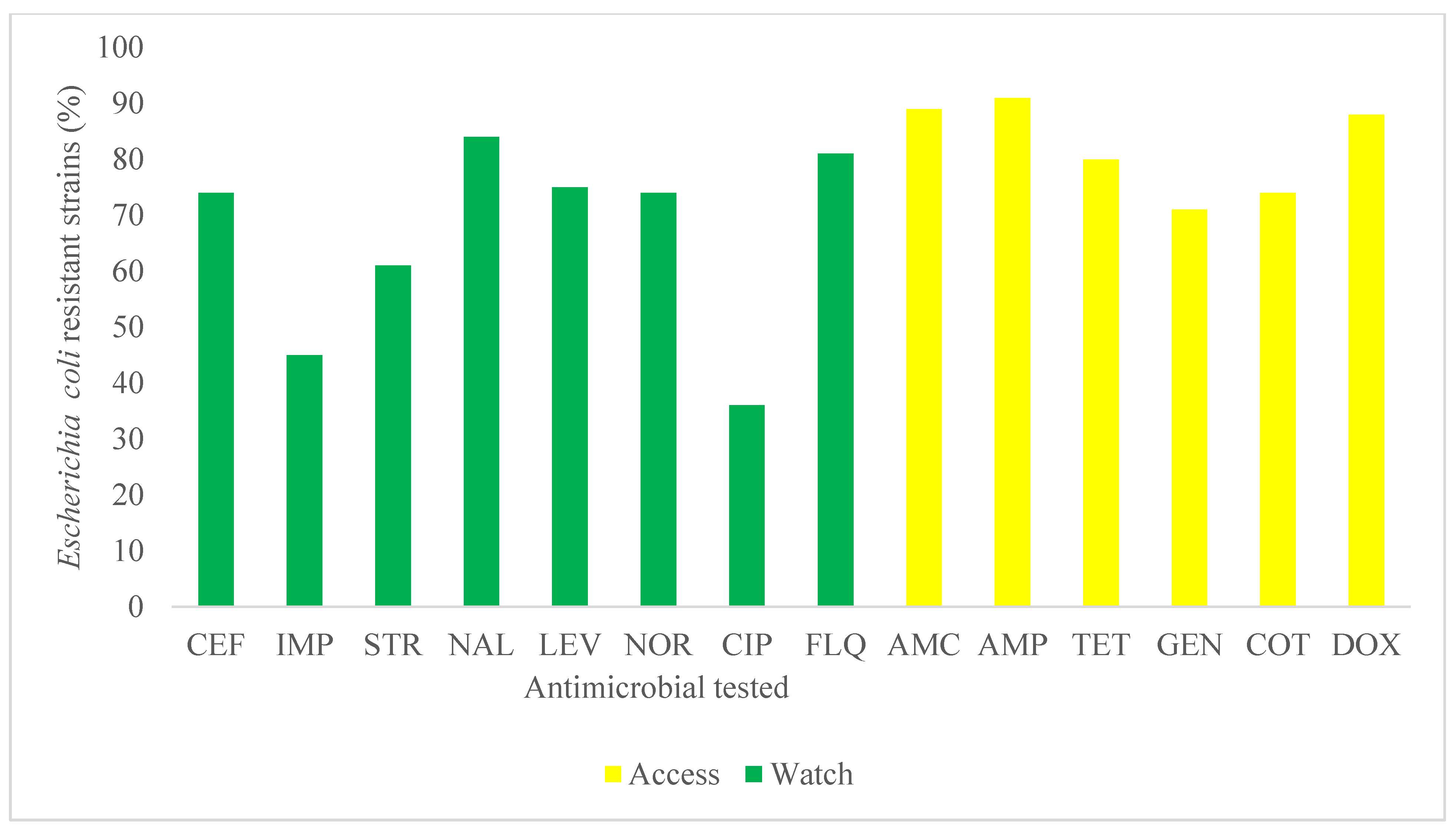
2.2. Prevalence of Antimicrobial Resistance and Phenotypic Resistance Profile of Escherichia coli
2.3. Risk Factors of Emergence and Diffusion of Resistant Germs from Poultry Litter
3. Discussion
4. Materials and Methods
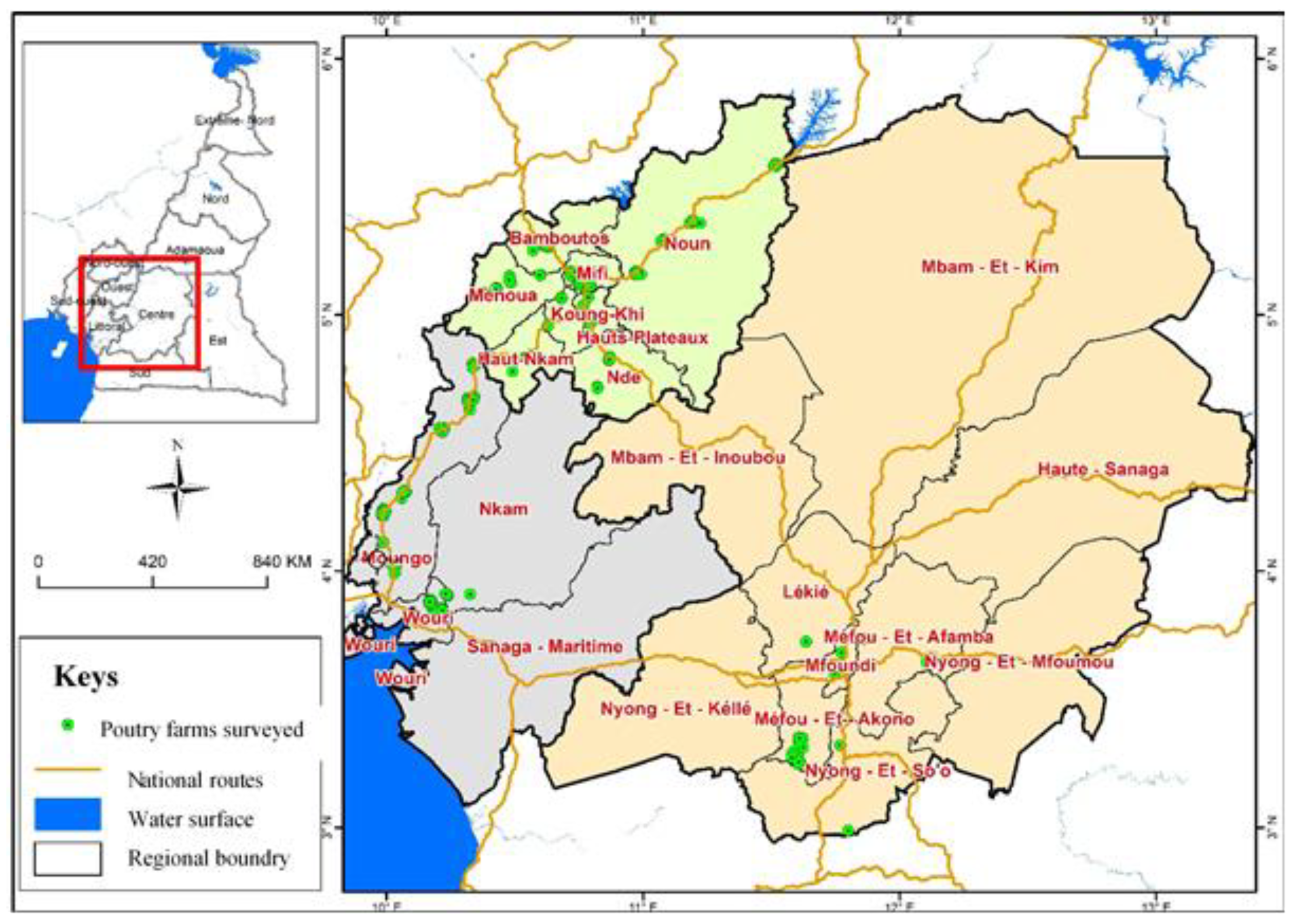
4.1. Study Site and Study Design
4.2. Sample Collection, Processing and Escherichia coli Isolation
4.3. Antimicrobial Susceptibility Test
4.4. Data Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Organization for Animal Health. Annual Report on Antimicrobial Agents Intended for the Use in Animals: Better Understanding of the Global Situation, Fourth Report. 2020. Available online: https://www.oie.int/fileadmin/Home/eng/Our_scientific_expertise/docs/pdf/A_Fourth_Annual_Report_AMU.pdf (accessed on 15 August 2019).
- Van Boeckel, P.T.; Brower, C.; Gilbert, M.; Grenfella, T.B.; Levina, A.S.; Robinsoni, T. Global trends in antimicrobial use in food animals. Proc. Nat. Acad. Sci. USA 2015, 112, 5649–5654. [Google Scholar] [CrossRef]
- Roskam, J.L.; Oude Lansink, G.M.; Saatkamp, W. The relation between technical farm performance and antimicrobial use of broiler farms. Poul. Sci. 2019. [Google Scholar] [CrossRef]
- Clifford, K.; Desai, D.; da Costa, P.C.; Meyer, H.; Klohe, K.; Winkler, A. Antimicrobial resistance in livestock and poor quality veterinary medicines. Bull. World Health Organ. 2018, 96, 662–664. [Google Scholar] [CrossRef]
- Holman, B.D.; Chenier, R.M. Impact of subtherapeutic administration of tylosin and chlortetracycline on antimicrobial resistance in farrow-to-finish swine. FEMS Microbiol. Ecol. 2013, 85, 81–83. [Google Scholar] [CrossRef]
- Ouedraogo, A.S.; Banuls, A.L.; Ouedraogo, R.; Godreuil, S. Emergence and spread of antibiotic resistance in West Africa: Contributing factors and threat assessment. Med. Sante Trop. 2017, 27, 147–154. [Google Scholar] [CrossRef]
- Tang, L.K.; Caffrey, P.N.; Nóbrega, B.D.; Cork, C.S.; Ronksley, E.P.; Barkema, W.H. Restricting the use of antibiotics in food-producing animals and its associations with antibiotic resistance in food-producing animals and human beings: A systematic review and meta-analysis. Lancet Planet. Health 2017, 1, 316–327. [Google Scholar] [CrossRef]
- FAO. Antimicrobial Resistance in the Environment: Summary Report of an FAO Meeting of Experts FAO Antimicrobial Resistance Working Group Report. 2018. Available online: http://www.fao.org/3/BU656en/bu656en.pdf (accessed on 16 August 2019).
- ECDC. Surveillance of Antimicrobial Resistance in Europe 2016; annual report of the European Antimicrobial Resistance Surveillance Network (EARS-NET); ECDC: Stockholm, Sweden, 2017. [Google Scholar]
- Awogbemi, J.; Adeyeye, M.; Akinkunmi, E.O. A Survey of Antmicrobial Agents Usage in Poultry Farms and Antbiotc Resistance in Escherichia Coli and Staphylococci Isolates from the Poultry in Ile-Ife. Nigeria. J. Infect. Dis. Epidemiol. 2018, 4, 047. [Google Scholar] [CrossRef]
- Kyakuwaire, M.; Olupot, G.; Amoding, A.; Nkedi-Kizza, P.; Basamba, A.T. How Safe is Chicken Litter for Land Application as an Organic Fertilizer?: A Review. Int. J. Environ. Res. Public Health 2019, 16, 3521. [Google Scholar] [CrossRef] [PubMed]
- Mathew, P.; Jaguga, C.; Mpundu, M.; Chand, J.S. Building knowledge and evidence base on antimicrobial resistance in Africa, through “One Health” based surveillance. Clin. Epidemiol. Glob. Health 2019. [CrossRef]
- Bhushan, C.; Khurana, A.; Sinha, R.; Nagaraju, M. Antibiotic Resistance in Poultry Environment: Spread of Resistance from Poultry Farm to Agricultural Field, New Delhi, India; Centre for Science and Environment: New Delhi, India, 2017. [Google Scholar]
- Furtula, V.; Farrell, E.G.; Diarrassouba, F.; Rempel, H.; Pritchard, J.; Diarra, M.S. Veterinary pharmaceuticals and antibiotic resistance of Escherichia coli isolates in poultry litter from commercial farms and controlled feeding trials. Poul. Sci. 2010, 89, 180–188. [Google Scholar] [CrossRef] [PubMed]
- Cookey, I.T.; Otokunefor, K. Poultry Environment as a Reservoir of Antimicrobial Resistant Bacteria: A Nigerian Story. Br. Microbiol. Res. J. 2017, 1, 1–11. [Google Scholar] [CrossRef]
- Eyasu, A.; Moges, F.; Alemu, A. Bacterial isolates from poultry litters and their antimicrobial susceptibility patterns in Gondar, Northwest Ethiopia. Int. J. Microbiol. Res. Rev. 2017, 6, 197–204. [Google Scholar]
- Vounba, P.; Yaghouba, K.; Ndiaye, C.; Arsenault, J.; Fairbrother, J.M.; Bada Alambedji, R. Molecular Characterization of Escherichia coli isolated from chickens with colibacillosis in Senegal. Foodborne Path. Dis. 2018. [Google Scholar] [CrossRef]
- Akoachere, T.J.-F.; Tanih, F.N.; Ndip, L.M.; Ndip, N.R. Phenotypic Characterization of Salmonella Typhimurium Isolates from Food-animals and Abattoir Drains in Buea, Cameroon. J. Health Popul. Nutr. 2009, 27, 612–618. [Google Scholar] [PubMed]
- Guetiya Wadoum, R.; Zambou, N.; Anyangwe, F.; Njimou, J.; Coman, M.; Verdenelli, M. Abusive use of antibiotics in poultry farming in Cameroon and the public health implications. Br. Poul. Sci. 2016, 57, 483–493. [Google Scholar] [CrossRef]
- Tatsadjieu, N.L.; Kemgang, S.T.; Mbofung, C.M. Impact de l’utilisation des antibiotiques sur la sensibilité des bactéries pathogènes de poules dans la ville de Ngaoundéré Cameroon. J. Exp. Biol. 2009, 5, 52–61. [Google Scholar]
- Wouafo, M.; Nzouankeu, A.; Kinfack, A.J.; Fonkoua, M.C.; Ejenguele, G. Prevalence and Antimicrobial Resistance of Salmonella Serotypes in Chickens from Retail Markets in Yaounde (Cameroon). Microbial Drug. Res. 2010, 16, 12. [Google Scholar] [CrossRef]
- Mouiche, M.M.M.; Moffo, F.; Akoachere, T.J.; Okah-Nnane, N.H.; Mapiefou, P.N.; Ngum Ndze, V.; Wade, A.; Djuikwo-Teukeng, F.; Toghoua, T.D.; Zambou, R.H.; et al. Antimicrobial resistance from a one health perspective in Cameroon: A systematic review and meta-analysis. BMC Publish Health 2019, 19, 1135. [Google Scholar] [CrossRef]
- Mccrea, B.A.; Macklin, K.S.; Norton, R.A.; Hess, J.B.; Bilgili, S.F. Recovery and genetic diversity of Escherichia coli isolates from deep litter, shallow litter, and surgical shoe covers. J. Appl. Poult. Res. 2008, 17, 237–242. [Google Scholar] [CrossRef]
- Velhner, M.; Milanov, D. Resistance to tetracycline in Escherichia coli and Staphylococcus aureus: Brief overview on mechanisms of resistance and epidemiology. Arch. Vet. Med. 2015, 8, 27–36. [Google Scholar] [CrossRef]
- Heuer, H.; Smalla, K. Manure and sulfadiazine synergistically increased bacterial antibiotic resistance in soil over at least two months. Environ. Microbiol. 2007, 9, 657–666. [Google Scholar] [CrossRef]
- Ngogang, M.P.; Ernest, T.; Kariuki, J.; Mouliom Mouiche, M.M.; Ngogang, J.; Wade, A.; van der Sande, M.A.B. Microbial Contamination of Chicken Litter Manure and Antimicrobial Resistance Threat in an Urban Area Setting in Cameroon. Antibiotics 2021, 10, 20. [Google Scholar] [CrossRef]
- Phiri, N.; Mainda, G.; Mukuma, M.; Sinyangwe, N.N.; Banda, J.L.; Kwenda, G.; Muligisa-Muonga1, E.; Flavien, N.B.; Mwansa, M.; Yamba, K.; et al. Antibiotic-resistant Salmonella species and Escherichia coli in broiler chickens from farms, abattoirs and open markets in selected districts of Zambia. bioRxiv 2020. [Google Scholar] [CrossRef]
- Adelowo, O.O.; Fagade, E.O.; Agers, Y. Antibiotic resistance and resistance genes in Escherichia coli from poultry farms, southwest Nigeria. J. Infect. Dev. Ctries. 2014, 8, 1103–1112. [Google Scholar] [CrossRef]
- Louokdom, S.J.; Fotsing Kwetché, P.; Djogang, Y.A.; Gamwo Dongmo, G.S.; Nankam Nguekap, L.W. Antibiotic susceptibility/resistance profile of bacteria from farm wastes: Findings in excreta from four poultries of West-Cameroon. World J. Adv. Healthc. Res. 2018, 2, 213–221. [Google Scholar]
- Fuh, N.J.; Owoseni, M.C.; Yami, A.L.; Utah, U.P.; Ekpiken, E.S.; Uchenna, M.O. Prevalence and Antibiotic Resistance of Escherichia coli O157: H7 Serotype from Chicken Droppings Produced by Free Ranged and Poultry Birds in Cross River, Nigeria. Am. J. Biomed. Life Sci. 2018, 6, 51–55. [Google Scholar]
- Abdalla, E.S.; Abia, L.K.A.; Amoako, G.D.; Perrett, K.; Bester, A.L.; Essack, Y.S. From Farm-to-Fork: E. Coli from an Intensive Pig Production System in South Africa Shows High Resistance to Critically Important Antibiotics for Human and Animal Use. Antibiotics 2021, 10, 178. [Google Scholar] [CrossRef]
- Thapa, D.B.; Chapagain, A. Antibiogram of Escherichia coli Isolated from Avian Colibacillosis in Chitwan District of Nepal. Int. J. Appl. Sci. Biotechnol. 2020, 8, 52–60. [Google Scholar] [CrossRef]
- Chantziaras, I. Epidemiology of Antimicrobial Resistance in Commensal E. coli: Focus on Selection and Spread of Fluoroquinolone Resistance in Broilers. Ph.D. Thesis, Ghent University, Ghent, Belgium, 2017. [Google Scholar]
- Mouiche, M.M.M.; Moffo, F.; Betsama, J.; Mapiefou, N.; Mbah, C.; Mpouam, S.; Ciake, S.; Feussom, J.M.; Kamnga, Z.; Awah-Ndukum, J.; et al. Challenges of antimicrobial consumption surveillance in food-producing animals in Sub-Saharan African countries: Patterns of antimicrobial imported in Cameroon from 2014 to 2019. J. Global Antimicrob. Res. 2020, 2, 771–778. [Google Scholar] [CrossRef] [PubMed]
- Moffo, F.; Mouiche, M.M.M.; Kochivi, L.F.; Dongmo, B.J.; Djomgang, K.H.; Tombe, P.; Mingoas, K.J.P.; Awah-Ndukum, J. Knowledge, attitudes, practices and risk perception of rural poultry farmers in Cameroon to antimicrobial use and resistance. Prev. Vet. Med. 2020, 182, 105087. [Google Scholar] [CrossRef] [PubMed]
- Mouiche, M.M.M.; Mazra, M.; Moffo, F.; Mpouam, S.E.; Ngoujegni, A.L.; Feussom, K.J.M.; Akoda, G.K.; Awah-Ndukum, J. Marché des médicaments vétérinaires au Cameroun: Reglémentation, Organisation et Circuits de Distribution. Bull. Anim. Prod. Afr. 2018, 66, 591–605. [Google Scholar]
- Têko-agbo, A.; Messomo Ndjana, F.; Walbadet, L.; Akoda, K.; Niang, M.; Abiola, F. Qualité des médicaments vétérinaires en circulation au Cameroun et au Sénégal. Dak. Med. 2009, 54, 226–234. [Google Scholar]
- Caruso, G. Antibiotic Resistance in Escherichia coli from Farm Livestock and Related Analytical Methods: A Review. J. AOAC Int. 2018, 101, 916–922. [Google Scholar] [CrossRef]
- Van Boeckel, T.P.; Pires, J.; Silvester, R.; Zhao, C.; Song, J.; Criscuolo, G.N. Global trends in antimicrobial resistance in animals in low—And middleincome countries. Science 2019, 365, eaaw1944. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Jiang, X. Microbiological safety of chicken litter or chicken litter-based organic fertilizers: A review. Agriculture 2014, 4, 1–29. [Google Scholar] [CrossRef]
- Kumar, K.; Thomson, A.; Singh, A.K.; Chander, Y.; Gupta, S.C. Enzyme-linked immunosorbent assay for ultratrace determination of antibiotics in aqueous samples. J. Environ. Qual. 2004, 33, 250–256. [Google Scholar] [CrossRef] [PubMed]
- Van Epps, A.; Blaney, L. Antibiotic residues in animal waste: Occurrence and degradation in conventional agricultural waste management practices. Curr. Pollut. Rep. 2016, 2, 135–155. [Google Scholar] [CrossRef]
- Boamah, V.; Agyare, C.; Odoi, H.; Dalsgaard, A. Practices and Factors Influencing the Use of Antibiotics in Selected Poultry Farms in Ghana. J. Antimicrob. Agents 2016, 2, 120. [Google Scholar] [CrossRef]
- Ozturk, Y.; Celik, S.; Sahin, E.; Acik, M.N.; Cetinkaya, B. Assessment of Farmers’ Knowledge, Attitudes and Practices on Antibiotics and Antimicrobial Resistance. Animals 2019, 9, 653. [Google Scholar] [CrossRef] [PubMed]
- INS. Annuaire Statistique du Cameroon: Recueil Des Series D’Informations Statistiques Sur Les Activités Economiques, Sociales, Politiques et Culturelles du Pays Jusqu’en 2013; INS: Yaounde, Cameroon, 2013. [Google Scholar]
- Kitching, R.P.; Taylor, N.M.; Thrusfield, M.V. Veterinary epidemiology: Vaccination strategies for foot-and-mouth disease. Nature 2007, 445, 412. [Google Scholar] [CrossRef] [PubMed]
- ISO. Microbiologie des Aliments—Méthode Horizontale Pour la Recherche Des Salmonella spp. Amendement 1: Annexe D: Recherche des Salmonella spp Dans les Matières Fécales des Animaux et Dans des Echantillons Environnementaux au Stade de la Production Primaire, 4th ed.; ISO: Geneva, Switzerland, 2007. [Google Scholar]
- EUCAST. Breakpoint Tables for Interpretation of MICs and Zone Diameters. Version 9.0 The European Committee on Antimicrobial Susceptibility Testing. 2019. Available online: http://www.eucast.org (accessed on 30 March 2021).
- WHO. Critically Important Antimicrobials for Human Medicine: Ranking of Medically Important Antimicrobials for Risk Management of Antimicrobial Resistance Due to Non-Human Use. 6th Revision. Available online: https://www.who.int/foodsafety/publications/antimicrobials-sixth/en/ (accessed on 14 August 2019).
- Hsia, Y.; Lee, R.B.; Versporten, A.; Yang, Y.; Bielicki, J.; Jackson, C. Use of the WHO Access, Watch, and Reserve classifcation to defne patterns of hospital antibiotic use (AWaRe): An analysis of paediatric survey data from 56 countries. Lancet Glob. Health 2019, 7, 861–871. [Google Scholar] [CrossRef]
| Regions | ||||
|---|---|---|---|---|
| Socio Professional Characteristics | West | Littoral | Centre | Total |
| n = 118 (%) | n = 70 (%) | n = 41 (%) | n = 229 (%) | |
| Gender | ||||
| Male | 90 (76.3) | 44 (62.9) | 26 (63.4) | 160 (69.9) |
| Female | 28 (23.7) | 26 (37.1) | 15 (36.6) | 69 (30.1) |
| Age range (Year) | ||||
| 20–29 | 3 (2.5) | 5 (7.1) | 7 (17.1) | 15 (6.6) |
| 30–39 | 35 (29.7) | 23 (32.9) | 12 (17.1) | 70 (30.6) |
| 40–49 | 65 (55.1) | 29 (41.4) | 9 (22.0) | 103 (45.0) |
| 50–59 | 15 (12.7) | 13 (18.5) | 13 (31.7) | 41 (17.9) |
| Educational level | ||||
| None | 2 (2.9) | 2 (2.9) | 0 (0.0) | 4 (1.7) |
| Primary | 12 (10.2) | 23 (32.9) | 3 (7.3) | 38 (16.6) |
| Secondary | 70 (59.3) | 29 (41.4) | 25 (61.0) | 124 (54.1) |
| Higher | 34 (28.8) | 15 (21.4) | 11 (26.8) | 60 (26.2) |
| Training in poultry farming | ||||
| Yes | 33 (27.7) | 9 (12.9) | 10 (24.4) | 52 (22.7) |
| No | 85 (72.0) | 61 (87.0) | 31 (75.6) | 177 (77.0) |
| Duration in poultry farming (month) | ||||
| 0–5 | 54 (45.8) | 22 (31.4) | 28 (68.3) | 104 (68.3) |
| 6–11 | 58 (49.2) | 28 (40.0) | 11 (26.8) | 97 (26.8) |
| 12–17 | 4 (3.4) | 16 (22.9) | 1 (2.4) | 21 (2.4) |
| ≥18 | 2 (1.7) | 4 (5.7) | 1 (2.4) | 7 (2.4) |
| Training on antimicrobial use | ||||
| Yes | 30 (25.4) | 2 (4.9) | 4 (5.7) | 36 (15.7) |
| No | 88 (74.6) | 39 (95.1) | 66 (94.3) | 193 (84.3) |
| Respect of vaccinal protocols | ||||
| No | 16 (13.6) | 1 (1.4) | 0 (0.0) | 17 (7.4) |
| Partially | 34 (28.8) | 1 (1.4) | 1 (2.4) | 36 (15.7) |
| Totally | 68 (57.6) | 68 (97.1) | 40 (97.0) | 176 (76.9) |
| Flock size | ||||
| <1000 | 43 (36.4) | 24 (34.3) | 17 (41.5) | 84 (36.7) |
| 1001–5000 | 68 (57.6) | 36 (51.4) | 18 (43.9) | 122 (53.3) |
| 5001–10,000 | 7 (5.9) | 10 (14.3) | 3 (14.6) | 23 (10.0) |
| Stocking density (number of chicken/m2) | ||||
| <6 | 24 (20.3) | 34 (48.6) | 2 (4.9) | 60 (26.2) |
| 9–6 | 46 (38.9) | 4 (5.7) | 25(60.9) | 75 (32.8) |
| >9 | 48 (40.7) | 32 (45.7) | 14 (34.2) | 94 (41.1) |
| Sanitary void lap (days) | ||||
| ≤14 | 11 (9.3) | 5 (7.1) | 2 (4.9) | 18 (7.9) |
| 14 | 16 (13.6) | 18 (25.7) | 3 (7.3) | 37 (16.2) |
| 14–30 | 42 (35.6) | 39 (55.7) | 36 (87.8) | 117 (51.1) |
| ≥30 | 49 (41.5) | 8 (11.4) | 0 (0.0) | 57 (24.9) |
| Frequency of disease | ||||
| Digestive | 36 (32.6) | 49 (44.6) | 26 (22.9) | 110 (48.0) |
| Respiratory | 19 (20.3) | 41 (44.3) | 33 (36.6) | 93 (40.6) |
| Nervous | 3 (2.2) | 1 (1.4) | 0 (0.0) | 4 (1.8) |
| Locomotory | 18 (15.5) | 0 (0.0) | 4 (9.7) | 22 (9.6) |
| Case of unsuccessful treatment | ||||
| Sale | 24 (20.3) | 13 (18.0) | 16 (39.0) | 53 (23.1) |
| Slaughter and consume | 93 (78.8) | 56 (80.0) | 25 (61) | 174 (76) |
| Quarantine | 1 (0.8) | 1 (1.4) | 2 (0.9) | 2 (0.9) |
| Management of death chicken | ||||
| Consummed | 37 (31.4) | 58 (82.9) | 18 (43.9) | 111 (49.3) |
| Dispose in the dustbin | 56 (47.5) | 8 (11.9) | 15 (36.6) | 79 (34.5) |
| Incinerate or bury | 25 (21.2) | 4 (5.7) | 8 (19.5) | 37 (16.2) |
| Litter management | ||||
| Sale to crops farmers | 49 (41.5) | 52 (74.3) | 22 (53.7) | 123 (53.7) |
| Spread on farms | 69 (58.5) | 18 (25.7) | 19 (46.3) | 106 (46.3) |
| Regions | ||||
|---|---|---|---|---|
| Factors | West | Littoral | Centre | Total |
| Purchase of antimicrobial | ||||
| Veterinary pharmacy | 86 (72.9%) | 50 (71.4%) | 39 (95.1%) | 175 (76.4%) |
| Parallel market | 32 (27.1%) | 20 (28.0%) | 2 (4.9%) | 54 (23.6%) |
| Reasons for AMU | ||||
| Preventive | 13 (11.0%) | 7 (10%) | 1 (2.4%) | 21 (9.2%) |
| Curative | 13 (11.0%) | 1 (2.4%) | 11 (15.7%) | 25 (10.9%) |
| Preventive and curative | 92 (77.9%) | 52 (74.3%) | 39 (95.1%) | 183 (79.9%) |
| Addition of antimicrobial in feed | ||||
| Yes | 28 (23.7%) | 16 (22.9%) | 2 (4.9%) | 46 (20.1%) |
| No | 90 (76.3%) | 54 (77.1%) | 39 (95.1%) | 183 (79.9%) |
| Management of expired drug | ||||
| Throw away in environment | 15 (56.8%) | 30 (42.9%) | 67 (36.6%) | 112 (48.9%) |
| Return to pharmacy | 14 (11.9%) | 8 (11.4%) | 0 (0.0%) | 22 (9.6%) |
| Administrated to animal | 26 (31.4%) | 32 (45.7%) | 37 (63.4%) | 95 (41.5%) |
| Most common AMU used in surveyed Farms | ||||
| Amoxicillin | 22 (18.6%) | 5 (7.1%) | 3 (7.3%) | 30 (13.1%) |
| Colistin | 18 (15.7%) | 11 (15.7%) | 26 (63.4%) | 55 (24.0%) |
| Doxycycline | 3 (2.5%) | 2 (2.9%) | 4 (9.8%) | 9 (3.9%) |
| Enrofloxacin | 0 (0.0%) | 2 (2.9%) | 1 (2.4%) | 3 (1.3%) |
| Flumequine | 0 (0.0%) | 2 (2.9%) | 1 (2.4%) | 3 (1.3%) |
| Norfloxacin | 0 (0.0%) | 14 (20.0%) | 0 (0.0%) | 14 (6.1%) |
| Oxytetracycline | 75 (50.2%) | 34 (48.6%) | 6 (14.6%) | 115 (50.2%) |
| Regions | Number of Sample Collected | Number of E. coli Isolated (%) | p-Value | Number of Strains Resistant and Prevalence of Resistance (%) 95% CI | p-Value |
|---|---|---|---|---|---|
| West Littoral Centre | 118 70 41 | 61 (51.7%) 20 (28.6%) 20 (48.8%) | 0.66 | 40 (65.6% (53.6–77.5)) 6 (30.0% (9.9–50.1)) 13 (65.0% (44.0–85.9)) | 0.0062 |
| Total | 229 | 101 (44.1%) | 59 (58.4% (48.8–68.0)) |
| Number of Antibiotic | Phenotypic Resistance Profile | Number of Isolates | MDRI |
|---|---|---|---|
| 1 | CEF | 5 | 0.07 |
| AMP | 3 | 0.07 | |
| 2 | CEF NAL | 2 | 0.14 |
| 5 | AMC CEF AMP TET DOX | 5 | 0.36 |
| 6 | AMC CEF AMP TET DOX NAL | 5 | 0.43 |
| 7 | AMC CEF AMP TET DOX NAL FLQ | 3 | 0.50 |
| AMC CEF AMP STR NOR NAL FLQ | 2 | 0.50 | |
| 8 | AMC CEF AMP DOX NAL NOR COT LEV | 5 | 0.57 |
| AMC CEF AMP DOX NAL NOR TET STR | 2 | 0.57 | |
| 9 | AMC CEF AMP TET DOX NAL FLQ COT STR | 7 | 0.64 |
| 10 | AMC CEF AMP TET DOX NAL FLQ COT STR LEV | 4 | 0.71 |
| 11 | AMC CEF AMP TET DOX NAL FLQ COT STR GEN NOR | 12 | 0.79 |
| 12 | AMC CEF AMP TET DOX NAL FLQ COT STR GEN NOR LEV | 2 | 0.86 |
| 13 | AMC CEF AMP TET DOX NAL FLQ COT STR GEN NOR LEV IMP | 1 | 0.93 |
| 14 | AMC CEF AMP TET DOX NAL FLQ COT STR GEN NOR LEV IMP CIP | 1 | 1 |
| Risk Factor | Category | Number of Samples Tested | Univariable | Multivariable | ||
|---|---|---|---|---|---|---|
| OR (95% CI) | p Value | OR (95% CI) | p Value | |||
| Region | West | 61 (60.4) | 2.19 (0.68–7.01) | 0.2 | 0.98 (0.21–4.69) | 0.98 |
| Littoral | 20 (19.8) | 0.23 (0.06–0.87) | 0.03 | 0.25 (0.06–1.13) | 0.07 | |
| Centre | 20 (19.8) | 1.0 | 1.0 | |||
| Training in poultry farming | No | 76 (75.2) | 0.39 (0.12–1.25) | 0.11 | 0.13 (0.03–0.64) | 0.01 |
| Yes | 25 (24.8) | 1.0 | 1.0 | |||
| Frequency of digestive tract infections | High | 55 (54.5) | 0.11 (0.04–0.40) | 0.001 | 0.10 (0.02–0.41) | 0.001 |
| Low | 46 (45.5) | 1.0 | 1.0 | |||
| Experience in poultry farming (months) | 0–5 | 45 (44.6) | 4.67 (0.89–27.40) | 0.06 | 11.66 (1.12–121.10) | 0.04 |
| 6–11 | 49 (48.5) | 3.02 (0.60–15.20) | 0.18 | 6.37 (0.66–61.66) | 0.11 | |
| 12–17 | 7 (6.9) | 1.0 | 1.0 | |||
| Respect of vaccinal protocol | No | 10 (9.9) | 5.52 (0.66–46.05) | 0.11 | 1.14 (0.10–13.67) | 0.92 |
| Partially | 20 (19.8) | 11.66 (1.48–92.14) | 0.02 | 5.11 (0.52–49.95) | 0.16 | |
| Totally | 71 (70.3) | 1.0 | 1.0 | |||
| WHO Classification | Antimicrobial Agents | Class | WHO-AWaRe | Disc Charges |
|---|---|---|---|---|
| Critically Important Antimicrobials | Ampicillin | Penicillin | Access | 10 µg |
| Amoxicilin/clavualanic acid | Penicillin | 20/10 µg | ||
| Gentamicin | Aminoglycoside | 10 µg | ||
| Ceftriaxone | Cephalosporine | Watch | 30 µg | |
| Ciprofloxacin | fluoroquinolone | 30 µg | ||
| Norfloxacin | fluoroquinolone | 10 µg | ||
| Streptomycin | Aminoglycoside | 10 µg | ||
| Flumequine | Quinolone | 30 µg | ||
| Imipenem | Carbamate | 10 µg | ||
| Levofloxacin | fluroquinolones | 5 µg | ||
| Nalidixic acid | fluoroquinolone | 30 µg | ||
| Highly Important antimicrobials | Tetracycline | Tetracycline | Access | 30 µg |
| Doxycycline | Tetracycline | 30 µg | ||
| Cotrimoxazole | Diaminopyrimidine/sulphamide | 25 µg |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moffo, F.; Mouiche, M.M.M.; Djomgang, H.K.; Tombe, P.; Wade, A.; Kochivi, F.L.; Dongmo, J.B.; Mbah, C.K.; Mapiefou, N.P.; Ngogang, M.P.; et al. Poultry Litter Contamination by Escherichia coli Resistant to Critically Important Antimicrobials for Human and Animal Use and Risk for Public Health in Cameroon. Antibiotics 2021, 10, 402. https://doi.org/10.3390/antibiotics10040402
Moffo F, Mouiche MMM, Djomgang HK, Tombe P, Wade A, Kochivi FL, Dongmo JB, Mbah CK, Mapiefou NP, Ngogang MP, et al. Poultry Litter Contamination by Escherichia coli Resistant to Critically Important Antimicrobials for Human and Animal Use and Risk for Public Health in Cameroon. Antibiotics. 2021; 10(4):402. https://doi.org/10.3390/antibiotics10040402
Chicago/Turabian StyleMoffo, Frédéric, Mohamed Moctar Mouliom Mouiche, Hervé Kapnang Djomgang, Patchely Tombe, Abel Wade, Fabrice Landjekpo Kochivi, Jarvis Bouna Dongmo, Cleophas Kahtita Mbah, Nabilah Pemi Mapiefou, Marie Paule Ngogang, and et al. 2021. "Poultry Litter Contamination by Escherichia coli Resistant to Critically Important Antimicrobials for Human and Animal Use and Risk for Public Health in Cameroon" Antibiotics 10, no. 4: 402. https://doi.org/10.3390/antibiotics10040402
APA StyleMoffo, F., Mouiche, M. M. M., Djomgang, H. K., Tombe, P., Wade, A., Kochivi, F. L., Dongmo, J. B., Mbah, C. K., Mapiefou, N. P., Ngogang, M. P., & Awah-Ndukum, J. (2021). Poultry Litter Contamination by Escherichia coli Resistant to Critically Important Antimicrobials for Human and Animal Use and Risk for Public Health in Cameroon. Antibiotics, 10(4), 402. https://doi.org/10.3390/antibiotics10040402






