Prescription of Aminoglycosides in 23 French Neonatal Intensive Care Units
Abstract
:1. Introduction
2. Methods
2.1. Study Design
2.2. Characteristics of the CPOE/CDS System and Prospectively Recorded Data
2.3. The Key Intervention
2.4. Inclusion Criteria
2.5. Definitions
2.6. Statistical Analysis
3. Results
3.1. NICU Characteristics
3.2. Patients Characteristics
3.3. Characteristics of Aminoglycosides Prescription
3.3.1. Patient Exposure to Aminoglycosides
3.3.2. Exposure to Gentamicin and Amikacin Prescription
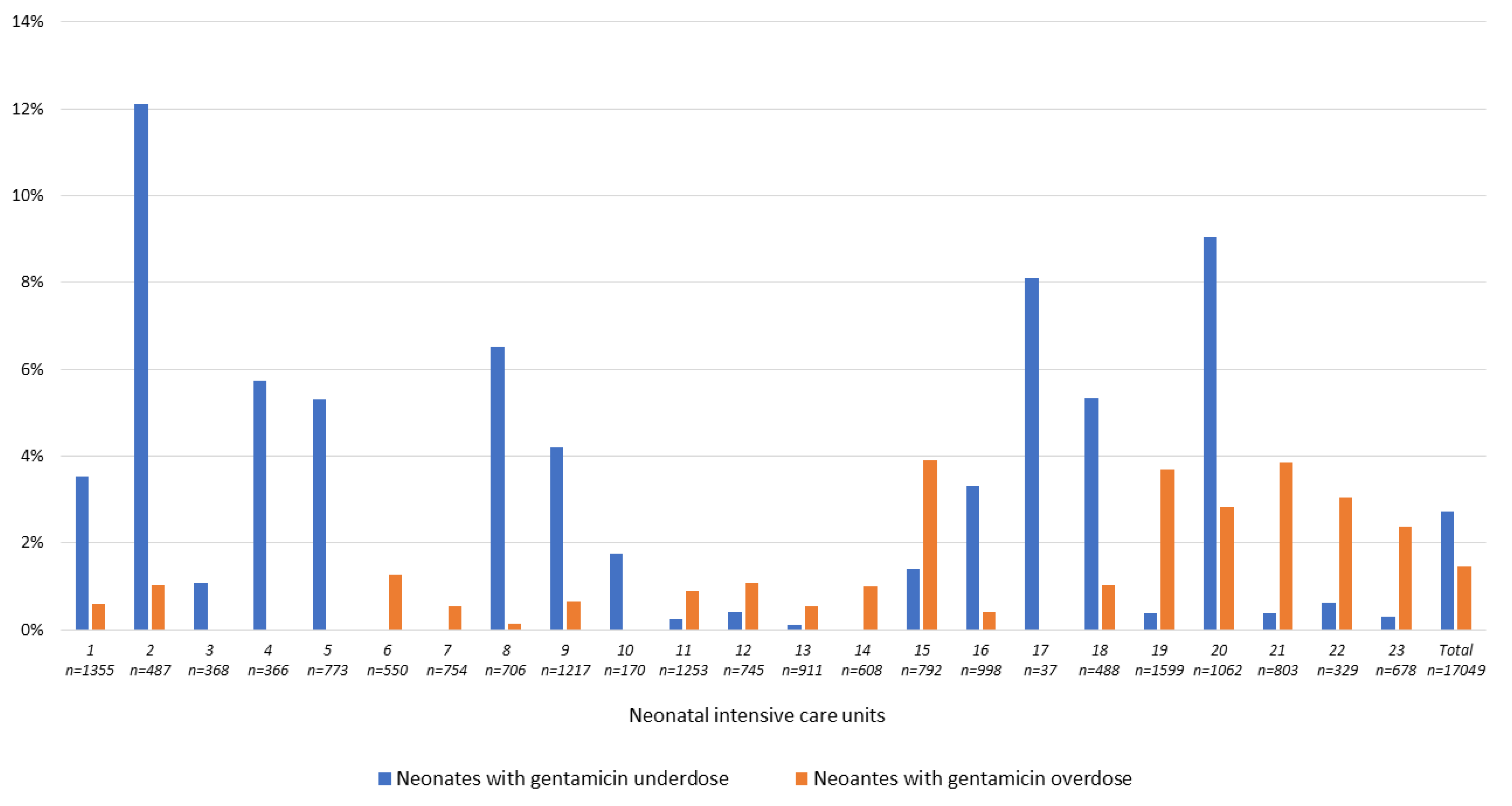
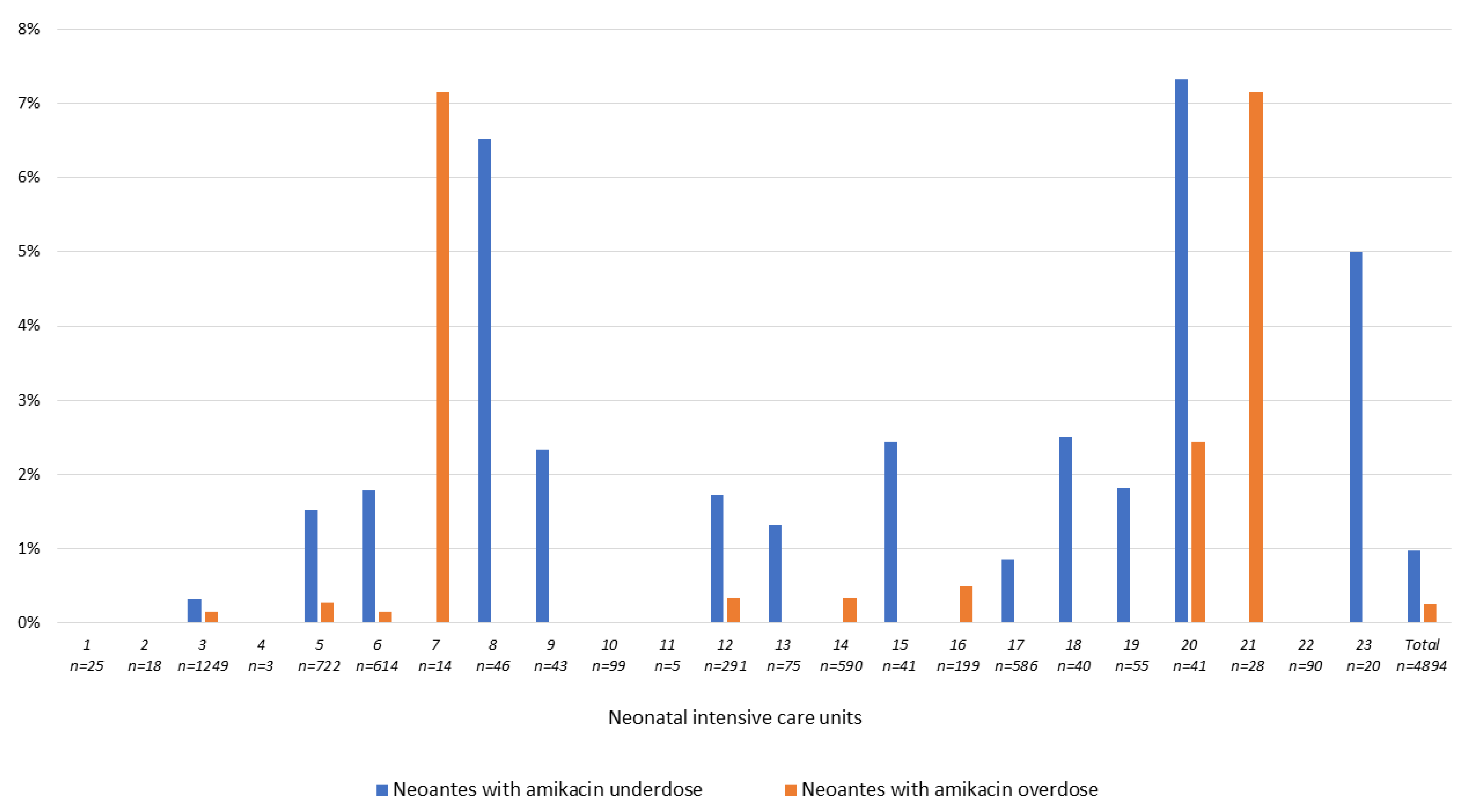
3.3.3. The Question of Aminoglycoside over-and under-Dosing
4. Discussion
Limits and Strengths
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Martin-Mons, S.; Lorrain, S.; Iacobelli, S.; Gouyon, B.; Gouyon, J.-B.; B-PEN Study Group. Antibiotics Prescription Over Three Years in a French Benchmarking Network of 23 Level 3 Neonatal Wards. Front. Pharmacol. 2021, 11, 585018. [Google Scholar] [CrossRef]
- Garrido, F.; Allegaert, K.; Arribas, C.; Villamor, E.; Raffaeli, G.; Paniagua, M.; Cavallaro, G.; on behalf of European Antibiotics Study Group. Variations in Antibiotic Use and Sepsis Management in Neonatal Intensive Care Units: A European Survey. Antibiotics 2021, 10, 1046. [Google Scholar] [CrossRef]
- Metsvaht, T.; Nellis, G.; Varendi, H.; Nunn, A.J.; Graham, S.; Rieutord, A.; Storme, T.; McElnay, J.; Mulla, H.; Turner, M.A.; et al. High variability in the dosing of commonly used antibiotics revealed by a Europe-wide point prevalence study: Implications for research and dissemination. BMC Pediatr. 2015, 15, 41. [Google Scholar] [CrossRef] [Green Version]
- Shahmoradi, L.; Safdari, R.; Ahmadi, H.; Zahmatkeshan, M. Clinical decision support systems-based interventions to improve medication outcomes: A systematic literature review on features and effects. Med. J. Islam. Repub. Iran. 2021, 35, 27. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, M.-N.R.; Mosel, C.; Grzeskowiak, L.E. Interventions to reduce medication errors in neonatal care: A systematic review. Ther. Adv. Drug Saf. 2017, 9, 123–155. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Devin, J.; Cleary, B.J.; Cullinan, S. The impact of health information technology on prescribing errors in hospitals: A systematic review and behaviour change technique analysis. Syst. Rev. 2020, 9, 275. [Google Scholar] [CrossRef] [PubMed]
- Rajar, P.; Saugstad, O.D.; Berild, D.; Dutta, A.; Greisen, G.; Lausten-Thomsen, U.; Mande, S.S.; Nangia, S.; Petersen, F.C.; Dahle, U.R.; et al. Antibiotic Stewardship in Premature Infants: A Systematic Review. Neonatology 2020, 117, 673–686. [Google Scholar] [CrossRef] [PubMed]
- Cantey, J.B.; Wozniak, P.S.; Pruszynski, J.; Sánchez, P.J. Reducing unnecessary antibiotic use in the neonatal intensive care unit (SCOUT): A prospective interrupted time-series study. Lancet Infect. Dis. 2016, 16, 1178–1184. [Google Scholar] [CrossRef]
- Lu, C.; Liu, Q.; Yuan, H.; Wang, L. Implementation of the smart use of antibiotics program to reduce unnecessary antibiotic use in a neonatal ICU: A prospective interrupted time-series study in a developing country. Crit. Care Med. 2019, 47, e1–e7. [Google Scholar] [CrossRef]
- Schulman, J.; Dimand, R.J.; Lee, H.C.; Duenas, G.V.; Bennett, M.V.; Gould, J.B. Neonatal Intensive Care Unit Antibiotic Use. Pediatrics 2015, 135, 826–833. [Google Scholar] [CrossRef] [Green Version]
- Schulman, J.; Profit, J.; Lee, H.C.; Dueñas, G.; Bennett, M.V.; Parucha, J.; Jocson, M.A.; Gould, J.B. Variations in Neonatal Antibiotic Use. Pediatrics 2018, 142, e20180115. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- O’Leary, E.N.; van Santen, K.L.; Edwards, E.M.; Braun, D.; Buus-Frank, M.E.; Edwards, J.R.; Guzman-Cottrill, J.A.; Horbar, J.D.; Lee, G.M.; Neuhauser, M.M.; et al. Using NHSN’s Antimicrobial Use Option to Monitor and Improve Antibiotic Stewardship in Neonates. Hosp. Pediatr. 2019, 9, 340–347. [Google Scholar] [CrossRef] [PubMed]
- Fanaroff, A.A.; Fanaroff, J.M. Advances in Neonatal Infections. Am. J. Perinatol. 2020, 37, S5–S9. [Google Scholar] [CrossRef]
- Klingenberg, C.; Kornelisse, R.F.; Buonocore, G.; Maier, R.F.; Stocker, M. Culture-Negative Early-Onset Neonatal Sepsis—At the Crossroad between Efficient Sepsis Care and Antimicrobial Stewardship. Front. Pediatr. 2018, 6, 285. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Akinboyo, I.C.; Gerber, J.S. Principles, policy and practice of antibiotic stewardship. Semin. Perinatol. 2020, 44, 151324. [Google Scholar] [CrossRef] [PubMed]
- Mukhopadhyay, S. Antibiotic stewardship in neonates and in neonatal intensive care units. Semin. Perinatol. 2020, 44, 151321. [Google Scholar] [CrossRef]
- Gouyon, B.; Iacobelli, S.; Saliba, E.; Quantin, C.; Pignolet, A.; Jacqz-Aigrain, E.; Gouyon, J.B. A Computer Prescribing Order Entry-Clinical Decision Support system designed for neonatal care: Results of the ‘preselected prescription’ concept at the bedside. J. Clin. Pharm. Ther. 2016, 42, 64–68. [Google Scholar] [CrossRef] [PubMed]
- Gouyon, B.; Martin-Mons, S.; Iacobelli, S.; Razafimahefa, H.; Kermorvant-Duchemin, E.; Brat, R.; Caeymaex, L.; Couringa, Y.; Alexandre, C.; Lafon, C.; et al. Characteristics of prescription in 29 Level 3 Neonatal Wards over a 2-year period (2017–2018). An inventory for future research. PLoS ONE 2019, 14, e0222667. [Google Scholar] [CrossRef] [PubMed]
- Allegaert, K.; Smits, A.; Van Donge, T.; Anker, J.V.D.; Sarafidis, K.; Levtchenko, E.; Mekahli, D. Renal Precision Medicine in Neonates and Acute Kidney Injury: How to Convert a Cloud of Creatinine Observations to Support Clinical Decisions. Front. Pediatr. 2020, 8, 366. [Google Scholar] [CrossRef]
- Liem, T.B.Y.; Slob, E.M.A.; Termote, J.U.M.; Wolfs, T.F.W.; Egberts, A.C.G.; Rademaker, C.M.A. Comparison of antibiotic dosing recommendations for neonatal sepsis from established reference sources. Int. J. Clin. Pharm. 2018, 40, 436–443. [Google Scholar] [CrossRef] [Green Version]
- Stark, A.; Smith, P.B.; Hornik, C.P.; Zimmerman, K.O.; Hornik, C.D.; Pradeep, S.; Clark, R.H.; Benjamin, D.K.; Laughon, M.; Greenberg, R.G. Medication Use in the Neonatal Intensive Care Unit and Changes from 2010 to 2018. J. Pediatr. 2021. [Google Scholar] [CrossRef] [PubMed]
- McMullan, B.J.; Mahony, M.; Java, L.; Mostaghim, M.; Plaister, M.; Wu, C.; White, S.; Al Yazidi, L.; Martin, E.; Bryant, P.; et al. Improving intravenous-to-oral antibiotic switch in children: A team-based audit and implementation approach. BMJ Open Qual. 2021, 10, e001120. [Google Scholar] [CrossRef]
- Ho, T.; Buus-Frank, M.E.; Edwards, E.M.; Morrow, K.A.; Ferrelli, K.; Srinivasan, A.; Pollock, D.A.; Dukhovny, D.; Zupancic, J.A.; Pursley, D.M.; et al. Adherence of Newborn-Specific Antibiotic Stewardship Programs to CDC Recommendations. Pediatrics 2018, 142, e20174322. [Google Scholar] [CrossRef] [Green Version]
- Alghamdi, A.A.; Keers, R.; Sutherland, A.; Ashcroft, D. Prevalence and Nature of Medication Errors and Preventable Adverse Drug Events in Paediatric and Neonatal Intensive Care Settings: A Systematic Review. Drug Saf. 2019, 42, 1423–1436. [Google Scholar] [CrossRef] [Green Version]
- Koeck, J.A.; Young, N.J.; Kontny, U.; Orlikowsky, T.; Bassler, D.; Eisert, A. Interventions to Reduce Medication Dispensing, Administration, and Monitoring Errors in Pediatric Professional Healthcare Settings: A Systematic Review. Front. Pediatr. 2021, 9, 633064. [Google Scholar] [CrossRef] [PubMed]
- Allegaert, K. Rational Use of Medicines in Neonates: Current Observations, Areas for Research and Perspectives. Healthcare 2018, 6, 115. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| Gestational Age (Weeks) | |||||
|---|---|---|---|---|---|
| (22–26) n = 1358 | (27–31) n = 3251 | (32–36) n = 4171 | ≥37 n = 6627 | Total n = 15,407 | |
| Time interval, n (%) | |||||
| 24 h | 39 (2.9) | 52 (1.6) | 1320 (31.6) | 6196 (93.5) | 7607 (49.4) |
| 36 h | 5 (0.4) | 1041 (32.0) | 2636 (63.2) | 393 (5.9) | 4075 (26.4) |
| 48 h | 1314 (96.8) | 2103 (64.7) | 83 (2.0) | 32 (0.5) | 3532 (22.9) |
| Gestational Age (Weeks) | |||||
|---|---|---|---|---|---|
| (22–26) n = 449 | (27–31) n = 1027 | (32–36) n = 1057 | ≥37 n = 1266 | Total n = 3799 | |
| Time interval, n (%) | |||||
| 24 h | 1 (0.2) | 16 (1.6) | 556 (52.6) | 1250 (98.7) | 1823 (48.0) |
| 30 h | 0 (0.0) | 37 (3.6) | 120 (11.4) | 0 (0.0) | 157 (4.1) |
| 36 h | 2 (0.4) | 239 (23.3) | 80 (7.6) | 1 (0.1) | 322 (8.5) |
| 48 h | 397 (88.4) | 712 (69.3) | 297 (28.1) | 5 (0.4) | 1411 (37.1) |
| 60 h | 46 (10.2) | 21 (2.0) | 0 (0.0) | 0 (0.0) | 67 (1.8) |
| Postnatal Age at First Prescription | ||||
|---|---|---|---|---|
| 1st Week n = 1358 | 2nd Week n = 81 | 3rd Week n = 29 | 4th Week n = 43 | |
| Time interval, n (%) | ||||
| 24 h | 39 (2.9) | 1 (1.2) | 1 (3.4) | 2 (4.7) |
| 36 h | 5 (0.4) | 14 (17.3) | 8 (27.6) | 15 (34.9) |
| 48 h | 1314 (96.8) | 66 (81.5) | 20 (69.0) | 26 (60.5) |
| Postnatal Age at First Prescription | ||||
|---|---|---|---|---|
| 1st Week n = 3251 | 2nd Week n = 228 | 3rd Week n = 99 | 4th Week n = 63 | |
| Time interval, n (%) | ||||
| 24 h | 52 (1.6) | 45 (19.7) | 25 (25.3) | 31 (49.2) |
| 36 h | 1041 (32.0) | 95 (41.7) | 37 (37.4) | 16 (25.4) |
| 48 h | 2103 (64.7) | 84 (36.8) | 36 (36.4) | 16 (25.4) |
| No n = 16,800 | Yes n = 249 | p-Value | OR * (CI 95%) | p-Value | |
|---|---|---|---|---|---|
| Year of admission, n (%) | <0.0001 | <0.0001 | |||
| 2017 | 3673 (21.9) | 87 (34.9) | 1 | ||
| 2018 | 4448 (26.5) | 58 (23.3) | 0.55 (0.39; 0.77) | ||
| 2019 | 4409 (26.2) | 56 (22.5) | 0.54 (0.38; 0.76) | ||
| 2020 | 4270 (25.4) | 48 (19.3) | 0.47 (0.33; 0.68) | ||
| Male, n (%) | 9706 (57.8) | 141 (56.6) | 0.72 | ||
| Birth weight (g), mean (±SD) | 2366 (1053) | 1688 (804) | <0.0001 | ||
| Birth weight Z-score, mean (±SD) | −0.2 (1.0) | −0.2 (0.9) | 0.61 | ||
| Intrauterine growth restriction (birth weight z-score < −1.28), n (%) | 2159 (12.9) | 23 (9.2) | 0.09 | ||
| Gestational age (weeks), n (%) | <0.0001 | <0.0001 | |||
| (22; 26) | 1484 (8.8) | 27 (10.8) | 1.45 (0.82; 2.56) | ||
| (27; 31) | 3535 (21.0) | 106 (42.6) | 3.02 (1.93; 4.73) | ||
| (32; 36) | 4394 (26.2) | 81 (32.5) | 2.60 (1.71; 3.97) | ||
| ≥37 | 7387 (44.0) | 35 (14.1) | 1 | ||
| Length of stay (days), mean (±SD) | 25.6 (33.5) | 42.9 (37.1) | <0.0001 | ||
| Length of stay (days), n (%) | <0.0001 | ||||
| (1; 4) | 4303 (25.6) | 14 (5.6) | 1 | ||
| (5; 11) | 4235 (25.2) | 32 (12.9) | 1.87 (0.99; 3.54) | ||
| (12; 35) | 4215 (25.1) | 91 (36.5) | 3.65 (2.02; 6.62) | ||
| ≥36 | 4047 (24.1) | 112 (45.0) | 3.30 (1.76; 6.19) | ||
| Amikacin exposure during hospitalization, n (%) | 707 (4.2) | 14 (5.6) | 0.27 | ||
| Neonates with at least one amikacin underdose, n (%) | 12 (0.1) | 0 (0.0) | 0.99 | ||
| Duration of gentamicin (days), n (%) | <0.0001 | <0.0001 | |||
| (1; 2) | 13,498 (80.3) | 150 (60.2) | 1 | ||
| (3; 4) | 2627 (15.6) | 55 (22.1) | 1.40 (1.01; 1.94) | ||
| ≥5 | 675 (4.0) | 44 (17.7) | 4.08 (2.77; 6.03) | ||
| Postnatal age at first prescription of gentamicin, mean (±SD) | 1.9 (4.6) | 3.0 (6.3) | 0.07 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martin-Mons, S.; Gouyon, B.; Lorrain, S.; Abasse, S.; Alexandre, C.; Binson, G.; Brat, R.; Caeymaex, L.; Couringa, Y.; Desbruyeres, C.; et al. Prescription of Aminoglycosides in 23 French Neonatal Intensive Care Units. Antibiotics 2021, 10, 1422. https://doi.org/10.3390/antibiotics10111422
Martin-Mons S, Gouyon B, Lorrain S, Abasse S, Alexandre C, Binson G, Brat R, Caeymaex L, Couringa Y, Desbruyeres C, et al. Prescription of Aminoglycosides in 23 French Neonatal Intensive Care Units. Antibiotics. 2021; 10(11):1422. https://doi.org/10.3390/antibiotics10111422
Chicago/Turabian StyleMartin-Mons, Séverine, Béatrice Gouyon, Séverine Lorrain, Soumeth Abasse, Cénéric Alexandre, Guillaume Binson, Roselyne Brat, Laurence Caeymaex, Yvan Couringa, Cécile Desbruyeres, and et al. 2021. "Prescription of Aminoglycosides in 23 French Neonatal Intensive Care Units" Antibiotics 10, no. 11: 1422. https://doi.org/10.3390/antibiotics10111422
APA StyleMartin-Mons, S., Gouyon, B., Lorrain, S., Abasse, S., Alexandre, C., Binson, G., Brat, R., Caeymaex, L., Couringa, Y., Desbruyeres, C., Meglio, M. D.-D., Escourrou, G., Flamein, F., Flechelles, O., Girard, O., Kermorvant-Duchemin, E., Lapillonne, A., Lafon, C., Di Maio, M., ... Gouyon, J.-B. (2021). Prescription of Aminoglycosides in 23 French Neonatal Intensive Care Units. Antibiotics, 10(11), 1422. https://doi.org/10.3390/antibiotics10111422







