Point-of-Care Compatibility of Ultra-Sensitive Detection Techniques for the Cardiac Biomarker Troponin I—Challenges and Potential Value
Abstract
1. Introduction
2. Complexity of cTnI Detection
3. Biomarker Recognition
3.1. Immunosensors
3.2. Alternative Biorecognition Elements
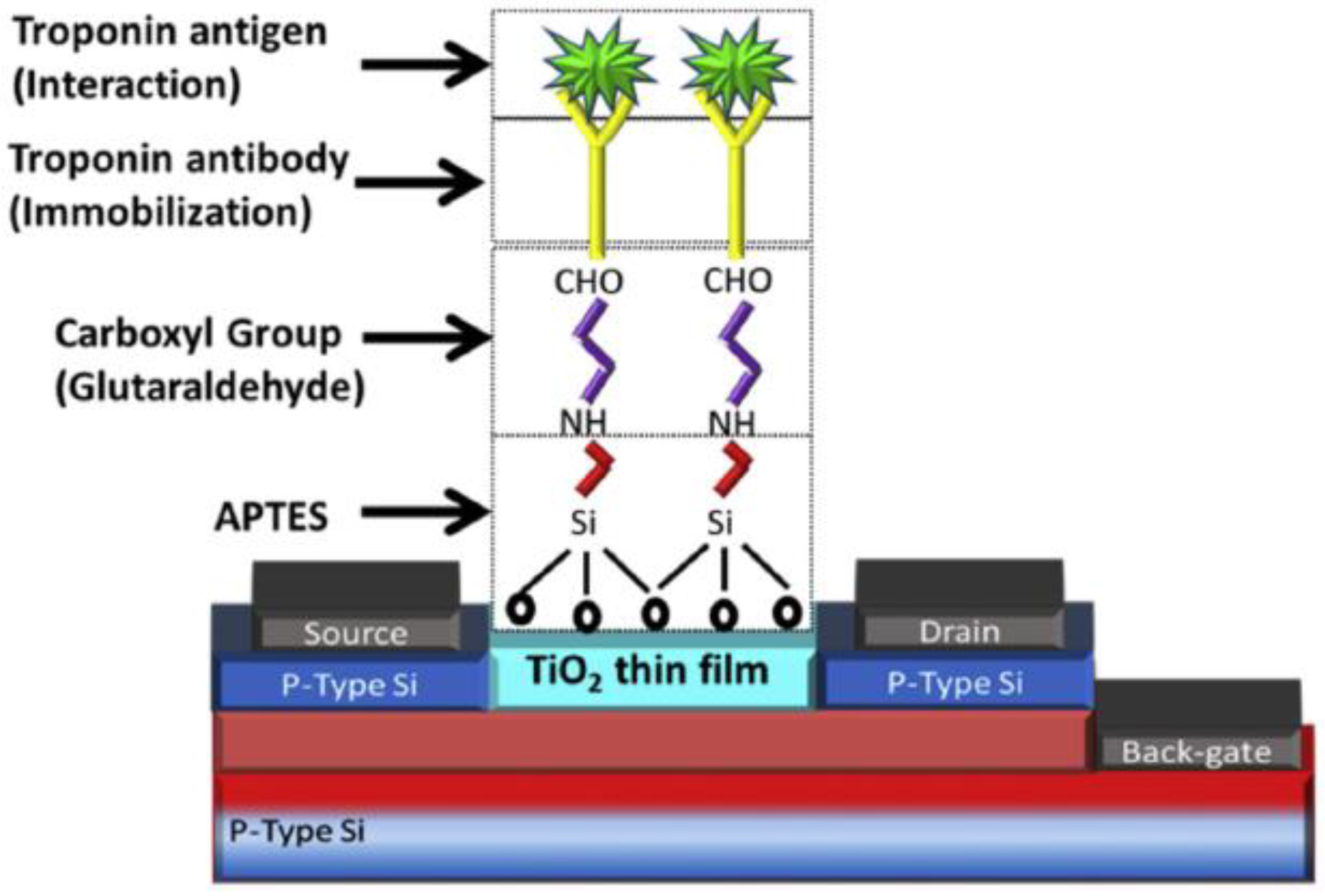
4. Immobilisation Strategies
5. Incorporation of Nanomaterials
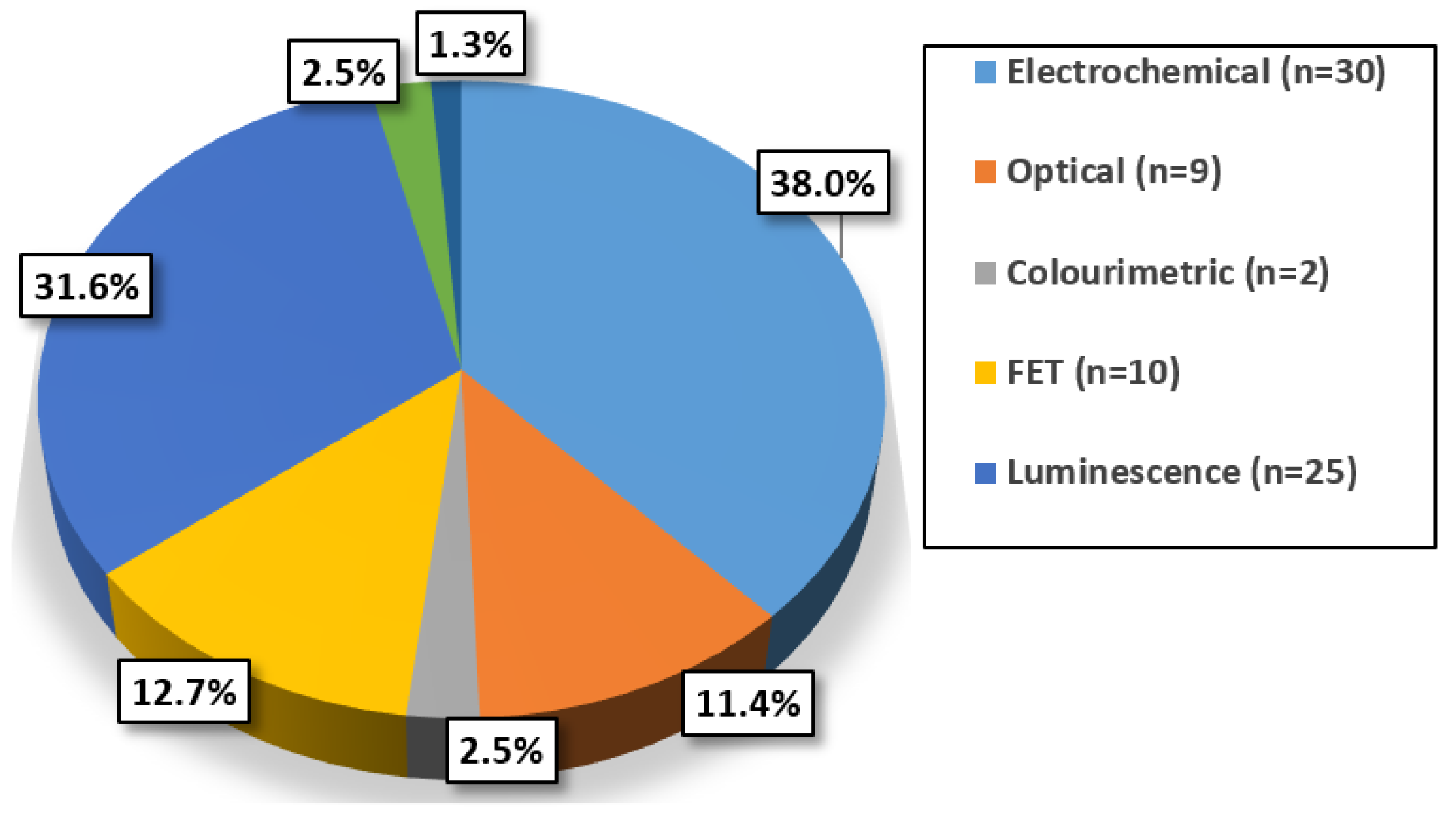
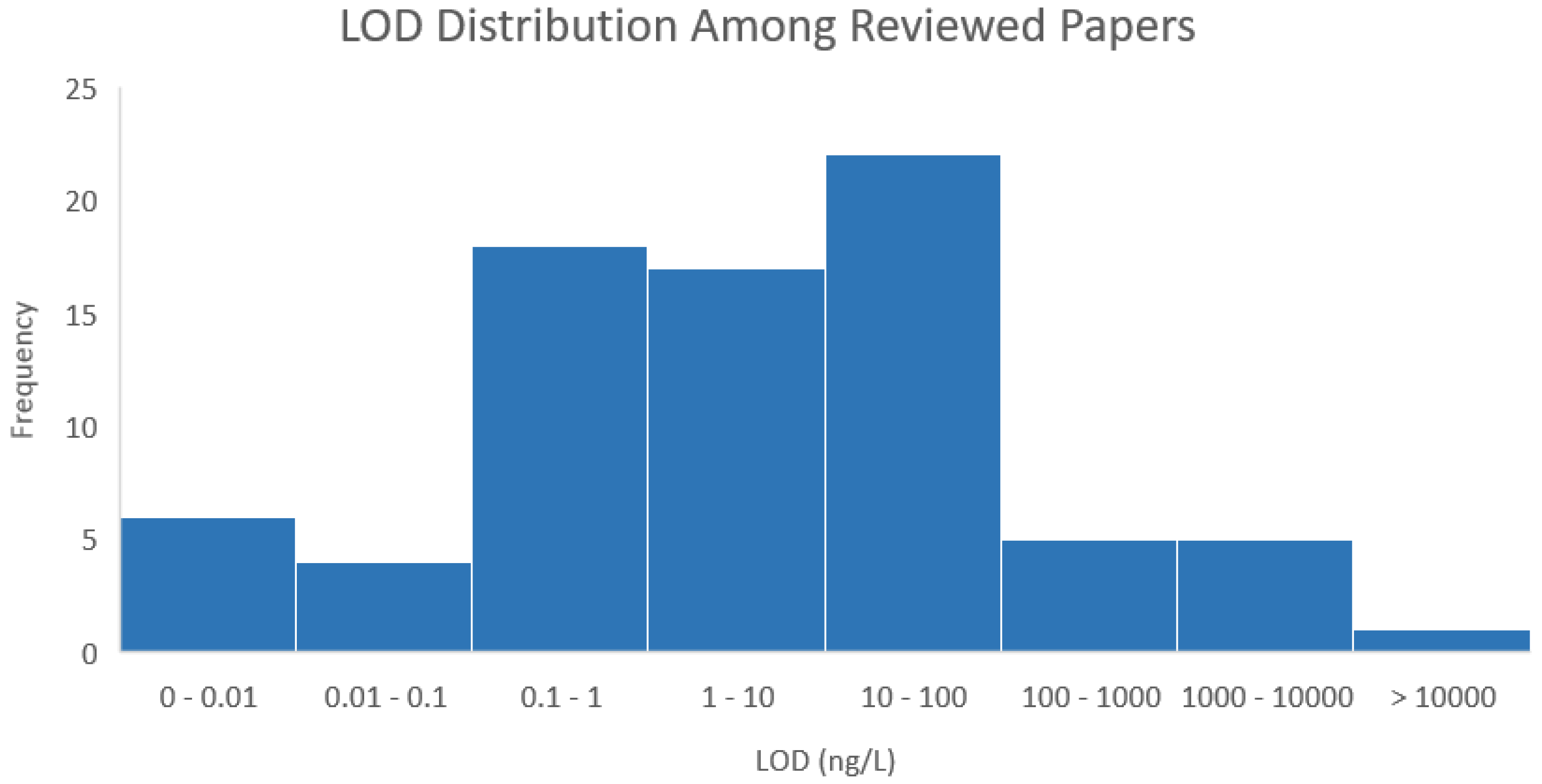
6. Evaluating cTnI Detection Techniques
7. Impact of Labels
Reducing the Response Time
8. Transition from Assay to POC Platform
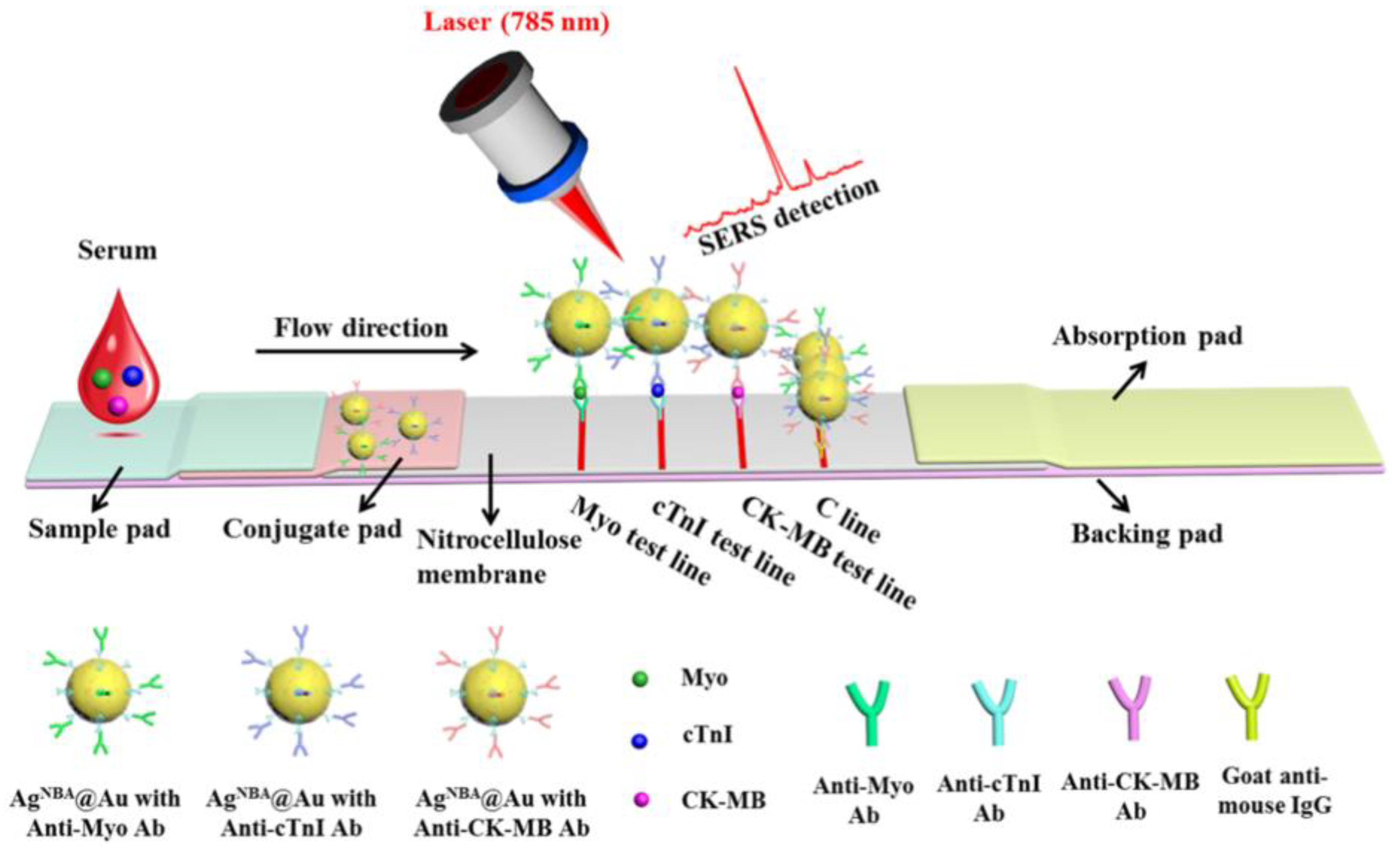
9. Multiplexing
10. POC Compatible Techniques
11. Conclusions
12. Reprinting Figures
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Price, C.P.; St John, A.; Hicks, J.M. Point-of-Care Testing: What, Why, When, and Where? In Point-of-Care Testing, 2nd ed.; AACC Press: Washington, DC, USA, 2004; pp. 3–30. [Google Scholar]
- McDonnell, B.; Hearty, S.; Leonard, P.; O’Kennedy, R. Cardiac biomarkers and the case for point-of-care testing. Clin. Biochem. 2009, 42, 549–561. [Google Scholar] [CrossRef] [PubMed]
- Vasan, R.S. Biomarkers of cardiovascular disease: Molecular basis and practical considerations. Circulation 2006, 113, 2335–2362. [Google Scholar] [CrossRef] [PubMed]
- Nikolova, A.P.; Hitzeman, T.C.; Baum, R.; Caldaruse, A.-M.; Agvanian, S.; Xie, Y.; Geft, D.R.; Chang, D.H.; Moriguchi, J.D.; Hage, A.; et al. Association of a Novel Diagnostic Biomarker, the Plasma Cardiac Bridging Integrator 1 Score, With Heart Failure with Preserved Ejection Fraction and Cardiovascular Hospitalization. JAMA Cardiol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Mair, J.; Jaffe, A.; Apple, F.; Lindahl, B. Cardiac Biomarkers. Dis. Markers 2015, 2015. [Google Scholar] [CrossRef] [PubMed]
- Hasić, S.; Kiseljaković, E.; Jadrić, R.; Radovanović, J.; Winterhalter-Jadrić, M. Cardiac troponin I: The gold standard in acute myocardial infarction diagnosis. Bosn. J. Basic Med. Sci. 2003, 3, 41–44. [Google Scholar] [CrossRef] [PubMed]
- Roffi, M.; Patrono, C.; Zamorano, J.L. CardioPulse: What’s new in the 2015 European Society of Cardiology Guidelines for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation. Eur. Heart J. 2016, 37, 206–207. [Google Scholar] [CrossRef] [PubMed]
- Twerenbold, R.; Boeddinghaus, J.; Nestelberger, T.; Wildi, K.; Rubini Gimenez, M.; Badertscher, P.; Mueller, C. Clinical Use of High-Sensitivity Cardiac Troponin in Patients with Suspected Myocardial Infarction. J. Am. Coll. Cardiol. 2017, 70, 996–1012. [Google Scholar] [CrossRef] [PubMed]
- Januzzi, J.L.; Sharma, U.; Zakroysky, P.; Truong, Q.A.; Woodard, P.K.; Pope, J.H.; Hauser, T.; Mayrhofer, T.; Nagurney, J.T.; Schoenfeld, D.; et al. Sensitive troponin assays in patients with suspected acute coronary syndrome: Results from the multi-center Rule out Myocardial Infarction Using Computer Assisted Tomography (ROMICAT) II Trial. Am. Heart J. 2015, 169, 572–578.e1. [Google Scholar] [CrossRef] [PubMed]
- IFCC C-CB. High-Sensitivity* Cardiac Troponin I and T Assay Analytical Characteristics Designated by Manufacturer; IFCC Committee on Clinical Applications of Cardiac Bio-Markers (C-CB): Milano, Italy, 2018. [Google Scholar]
- Christenson, R.H.; Jacobs, E.; Uettwiller-Geiger, D.; Estey, M.P.; Lewandrowski, K.; Koshy, T.I.; Kupfer, K.; Li, Y.; Wesenberg, J.C. Comparison of 13 Commercially Available Cardiac Troponin Assays in a Multicenter North American Study. J. Appl. Lab. Med. 2017, 1, 544–561. [Google Scholar] [CrossRef]
- IFCC C-CB. Point of Care Cardiac Troponin I and T Assay Analytical Characteristics Designated by Manufacturer; IFCC Committee on Clinical Applications of Cardiac Bio-Markers (C-CB): Milano, Italy, 2018. [Google Scholar]
- Apple, F.S.; Ler, R.; Murakami, M.M. Determination of 19 cardiac troponin I and T assay 99th percentile values from a common presumably healthy population. Clin. Chem. 2012, 58, 1574–1581. [Google Scholar] [CrossRef] [PubMed]
- Apple, F.S.; Collinson, P.O.; IFCC Task Force on Clinical Applications of Cardiac Biomarkers. Analytical Characteristics of High-Sensitivity Cardiac Troponin Assays. Clin. Chem. 2012, 58, 54–61. [Google Scholar] [CrossRef] [PubMed]
- Jarolim, P. High sensitivity cardiac troponin assays in the clinical laboratories. Clin. Chem. Lab. Med. 2015, 53, 635–652. [Google Scholar] [CrossRef] [PubMed]
- Apple, F.S. Counterpoint: Standardization of Cardiac Troponin I Assays Will Not Occur in My Lifetime. Clin. Chem. 2012, 58, 169–171. [Google Scholar] [CrossRef] [PubMed]
- Wu, A.H.B.; Christenson, R.H. Analytical and assay issues for use of cardiac troponin testing for risk stratification in primary care. Clin. Biochem. 2013, 46, 969–978. [Google Scholar] [CrossRef] [PubMed]
- Thygesen, K.; Alpert, J.S.; Jaffe, A.S.; Simoons, M.L.; Chaitman, B.R.; White, H.D. Third Universal Definition of Myocardial Infarction. Circulation 2012, 126, 2020–2035. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.M.; Thambiah, S.C.; Zahari Sham, S.Y.; Omar, R.; Hambali, Z.; Samsudin, I.N. Determination of the 99th percentile upper reference limit for high-sensitivity cardiac troponin I in Malaysian population. Malays. J. Pathol. 2017, 39, 135–140. [Google Scholar] [PubMed]
- Franzini, M.; Lorenzoni, V.; Masotti, S.; Prontera, C.; Chiappino, D.; Latta, D.D.; Daves, M.; Deluggi, I.; Zuin, M.; Ferrigno, L.; et al. The calculation of the cardiac troponin T 99th percentile of the reference population is affected by age, gender, and population selection: A multicenter study in Italy. Clin. Chim. Acta 2015, 438, 376–381. [Google Scholar] [CrossRef] [PubMed]
- Sheng, Q.; Qiao, X.; Zhou, M.; Zheng, J. Recent progress in electrochemical sensing of cardiac troponin by using nanomaterial-induced signal amplification. Microchim. Acta 2017, 184, 1573–1585. [Google Scholar] [CrossRef]
- Shanmugam, N.R.; Muthukumar, S.; Prasad, S. A review on ZnO-based electrical biosensors for cardiac biomarker detection. Future Sci. OA 2017, 3, FSO196. [Google Scholar] [CrossRef] [PubMed]
- Qureshi, A.; Gurbuz, Y.; Niazi, J.H. Biosensors for cardiac biomarkers detection: A review. Sens. Actuators B Chem. 2012, 171–172, 62–76. [Google Scholar] [CrossRef]
- Thygesen, K.; Mair, J.; Katus, H.; Plebani, M.; Venge, P.; Collinson, P.; Lindahl, B.; Giannitsis, E.; Hasin, Y.; Galvani, M.; et al. Recommendations for the use of cardiac troponin measurement in acute cardiac care. Eur. Heart J. 2010, 31, 2197–2204. [Google Scholar] [CrossRef] [PubMed]
- Christenson, R.H.; Bunk, D.M.; Schimmel, H.; Tate, J.R.; IFCC Working Group on Standardization of Troponin I. Point: Put simply, standardization of cardiac troponin I is complicated. Clin. Chem. 2012, 58, 165–168. [Google Scholar] [CrossRef] [PubMed]
- Apple, F.S.; Sharkey, S.W.; Falahati, A.; Murakami, M.; Mitha, N.; Christensen, D. Assessment of left ventricular function using serum cardiac troponin I measurements following myocardial infarction. Clin. Chim. Acta Int. J. Clin. Chem. 1998, 272, 59–67. [Google Scholar] [CrossRef]
- Hickman, P.E.; Potter, J.M.; Aroney, C.; Koerbin, G.; Southcott, E.; Wu, A.H.B.; Roberts, M.S. Cardiac troponin may be released by ischemia alone, without necrosis. Clin. Chim. Acta 2010, 411, 318–323. [Google Scholar] [CrossRef] [PubMed]
- Pettersson, K.; Eriksson, S.; Wittfooth, S.; Engström, E.; Nieminen, M.; Sinisalo, J. Autoantibodies to Cardiac Troponin Associate with Higher Initial Concentrations and Longer Release of Troponin I in Acute Coronary Syndrome Patients. Clin. Chem. 2009, 55, 938–945. [Google Scholar] [CrossRef] [PubMed]
- Eriksson, S.; Halenius, H.; Pulkki, K.; Hellman, J.; Pettersson, K. Negative interference in cardiac troponin I immunoassays by circulating troponin autoantibodies. Clin. Chem. 2005, 51, 839–847. [Google Scholar] [CrossRef] [PubMed]
- Eriksson, S.; Junikka, M.; Pettersson, K. An interfering component in cardiac troponin I immunoassays-Its nature and inhibiting effect on the binding of antibodies against different epitopes. Clin. Biochem. 2004, 37, 472–480. [Google Scholar] [CrossRef] [PubMed]
- Adamczyk, M.; Brashear, R.J.; Mattingly, P.G. Circulating cardiac troponin-I autoantibodies in human plasma and serum. Ann. N. Y. Acad. Sci. 2009, 1173, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Knoblock, R.J.; Lehman, C.M.; Smith, R.A.; Apple, F.S.; Roberts, W.L. False-positive AxSYM cardiac troponin I results in a 53-Year-Old woman. Arch. Pathol. Lab. Med. 2002, 126, 606–609. [Google Scholar] [PubMed]
- Al-Awadhi, A.M.; Olusi, S.; Hasan, E.A.; Abdullah, A. Serum concentrations of cardiac troponin-I in patients with rheumatoid arthritis, systemic lupus erythematosus, primary Sjogren’s syndrome and Graves’ disease. Singapore Med. J. 2007, 48, 847–849. [Google Scholar] [PubMed]
- Lam, Q.; Black, M.; Youdell, O.; Spilsbury, H.; Schneider, H.-G. Performance Evaluation and Subsequent Clinical Experience with the Abbott Automated Architect STAT Troponin-I Assay. Clin. Chem. 2006, 52, 298–300. [Google Scholar] [CrossRef] [PubMed]
- IFCC WG-TNI. Analytical Characteristics of Commercial and Research Cardiac Troponin I and T Assays Declared by the Manufacturer; IFCC Committee on Clinical Applications of Cardiac Bio-Markers (C-CB): Milano, Italy, December 2010. [Google Scholar]
- IFCC WG-TNI. Analytical Characteristics of Commercial Cardiac Troponin I and T Assays Declared by the Manufacturer; IFCC Committee on Clinical Applications of Cardiac Bio-Markers (C-CB): Milano, Italy, November 2014. [Google Scholar]
- Seo, S.-M.; Kim, S.-W.; Park, J.-N.; Cho, J.-H.; Kim, H.-S.; Paek, S.-H. A fluorescent immunosensor for high-sensitivity cardiac troponin I using a spatially-controlled polymeric, nano-scale tracer to prevent quenching. Biosens. Bioelectron. 2016, 83, 19–26. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.D.; Kim, H.-S.; Park, Y.M.; Chun, H.J.; Kim, J.-H.; Yoon, H.C. Retroreflective Janus Microparticle as a Nonspectroscopic Optical Immunosensing Probe. ACS Appl. Mater. Interfaces 2016, 8, 10767–10774. [Google Scholar] [CrossRef] [PubMed]
- Savukoski, T.; Engström, E.; Engblom, J.; Ristiniemi, N.; Wittfooth, S.; Lindahl, B.; Eggers, K.M.; Venge, P.; Pettersson, K. Troponin-specific autoantibody interference in different cardiac troponin I assay configurations. Clin. Chem. 2012, 58, 1040–1048. [Google Scholar] [CrossRef] [PubMed]
- Chambers, J.P.; Arulanandam, B.P.; Matta, L.L.; Weis, A.; Valdes, J.J. Biosensor recognition elements. Curr. Issues Mol. Biol. 2008, 10, 1–12. [Google Scholar] [PubMed]
- Perumal, V.; Hashim, U. Advances in biosensors: Principle, architecture and applications. J. Appl. Biomed. 2014, 12, 1–15. [Google Scholar] [CrossRef]
- Howard, C.G.; Kaser, M.R. Making and Using Antibodies A Practical Handbook, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2013. [Google Scholar]
- Lipman, N.S.; Jackson, L.R.; Trudel, L.J.; Weis-Garcia, F. Monoclonal Versus Polyclonal Antibodies: Distinguishing Characteristics, Applications, and Information Resources. ILAR J. 2005, 46, 258–268. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, Z.A.; Yeap, S.K.; Ali, A.M.; Ho, W.Y.; Alitheen, N.B.M.; Hamid, M. scFv antibody: Principles and clinical application. Clin. Dev. Immunol. 2012, 2012, 980250. [Google Scholar] [CrossRef] [PubMed]
- Shen, Z.; Stryker, G.A.; Mernaugh, R.L.; Yu, L.; Yan, H.; Zeng, X. Single-Chain Fragment Variable Antibody Piezoimmunosensors. Anal. Chem. 2005, 77, 797–805. [Google Scholar] [CrossRef] [PubMed]
- Hearty, S.; O’Kennedy, R. Exploiting recombinant antibodies in point-of-care (POC) diagnostics: The combinatorial advantage. Bioeng. Bugs 2011, 2, 182–186. [Google Scholar] [CrossRef] [PubMed]
- Hyytiä, H.; Järvenpää, M.-L.; Ristiniemi, N.; Lövgren, T.; Pettersson, K. A comparison of capture antibody fragments in cardiac troponin I immunoassay. Clin. Biochem. 2013, 46, 963–968. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y. Fabrication of an Immunosensor for Cardiac Troponin I Determination. Int. J. Electrochem. Sci. 2017, 7931–7940. [Google Scholar] [CrossRef]
- Ham, W.S.; Kim, M.K.; Gim, J.S.; Lee, J.S.; Wu, J.H.; Lee, K.B.; Kim, Y.K. Microstructure and Magnetic Properties of LaSrMnO Nanoparticles and Their Application to Cardiac Immunoassay. IEEE Trans. Magn. 2015, 51, 5101304. [Google Scholar] [CrossRef]
- Sirkka, N.; Lyytikäinen, A.; Savukoski, T.; Soukka, T. Upconverting nanophosphors as reporters in a highly sensitive heterogeneous immunoassay for cardiac troponin I. Anal. Chim. Acta 2016, 925, 82–87. [Google Scholar] [CrossRef] [PubMed]
- Delle, L.E.; Pachauri, V.; Sharma, S.; Shaforost, O.; Ma, H.; Adabi, M.; Lilischkis, R.; Wagner, P.; Thoelen, R.; Klein, N.; et al. ScFv-modified graphene-coated IDE-arrays for “label-free” screening of cardiovascular disease biomarkers in physiological saline. Biosens. Bioelectron. 2018, 102, 574–581. [Google Scholar] [CrossRef] [PubMed]
- O’Reilly, E.J.; Conroy, P.J.; Hearty, S.; Keyes, T.E.; O’Kennedy, R.; Forster, R.J.; Dennany, L. Electrochemiluminescence platform for the detection of C-Reactive proteins: Application of recombinant antibody technology to cardiac biomarker detection. RSC Adv. 2015, 5, 67874–67877. [Google Scholar] [CrossRef]
- Spain, E.; Gilgunn, S.; Sharma, S.; Adamson, K.; Carthy, E.; O’Kennedy, R.; Forster, R.J. Detection of prostate specific antigen based on electrocatalytic platinum nanoparticles conjugated to a recombinant scFv antibody. Biosens. Bioelectron. 2016, 77, 759–766. [Google Scholar] [CrossRef] [PubMed]
- Lakhin, A.V.; Tarantul, V.Z.; Gening, L.V. Aptamers: Problems, Solutions and Prospects. Acta Nat. 2013, 5, 34–43. [Google Scholar] [PubMed]
- Ku, T.-H.; Zhang, T.; Luo, H.; Yen, T.M.; Chen, P.-W.; Han, Y.; Lo, Y.-H. Nucleic Acid Aptamers: An Emerging Tool for Biotechnology and Biomedical Sensing. Sensors 2015, 15, 16281–16313. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Cao, Z.; Lu, Y. Functional nucleic acid sensors. Chem. Rev. 2009, 109, 1948–1998. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Zhao, Y.; Sun, L.; Qi, H.; Gao, Q.; Zhang, C. Electrogenerated chemiluminescence biosensor array for the detection of multiple AMI biomarkers. Sens. Actuators B Chem. 2018, 257, 60–67. [Google Scholar] [CrossRef]
- Liu, X.; Liu, H.; Li, M.; Qi, H.; Gao, Q.; Zhang, C. Highly Sensitive Electrochemiluminescence Assay for Cardiac Troponin I and Adenosine Triphosphate by using Supersandwich Amplification and Bifunctional Aptamer. Chemelectrochem 2017, 4, 1708–1713. [Google Scholar] [CrossRef]
- Xia, F.; White, R.J.; Zuo, X.; Patterson, A.; Xiao, Y.; Kang, D.; Gong, X.; Plaxco, K.W.; Heeger, A.J. An Electrochemical Supersandwich Assay for Sensitive and Selective DNA Detection in Complex Matrices. J. Am. Chem. Soc. 2010, 132, 14346–14348. [Google Scholar] [CrossRef] [PubMed]
- Vasapollo, G.; Sole, R.D.; Mergola, L.; Lazzoi, M.R.; Scardino, A.; Scorrano, S.; Mele, G. Molecularly Imprinted Polymers: Present and Future Prospective. Int. J. Mol. Sci. 2011, 12, 5908–5945. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Shen, X.-L.; Wang, H.-S.; Tao, J.; Huang, J.-Z.; Zeng, Q.; Wang, L.-S. MIPs-graphene nanoplatelets-MWCNTs modified glassy carbon electrode for the determination of cardiac troponin I. Anal. Biochem. 2017, 520, 9–15. [Google Scholar] [CrossRef] [PubMed]
- Liébana, S.; Drago, G.A. Bioconjugation and stabilisation of biomolecules in biosensors. Essays Biochem. 2016, 60, 59–68. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Byrne, H.; O’Kennedy, R.J. Antibodies and antibody-derived analytical biosensors. In Biosensor Technologies for Detection of Biomolecules; Estrela, P., Ed.; Portland Press Ltd.: London, UK, 2016; Volume 60, pp. 9–18. [Google Scholar]
- Mohamad, N.R.; Marzuki, N.H.C.; Buang, N.A.; Huyop, F.; Wahab, R.A. An overview of technologies for immobilization of enzymes and surface analysis techniques for immobilized enzymes. Biotechnol. Biotechnol. Equip. 2015, 29, 205–220. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Liang, L.-L.; Xiao, P.; Sun, L.-P.; Huang, Y.-Y.; Ran, Y.; Jin, L.; Guan, B.-O. A label-free cardiac biomarker immunosensor based on phase-shifted microfiber Bragg grating. Biosens. Bioelectron. 2018, 100, 155–160. [Google Scholar] [CrossRef] [PubMed]
- Adzhri, R.; Md Arshad, M.K.; Gopinath, S.C.B.; Ruslinda, A.R.; Fathil, M.F.M.; Ibau, C.; M. Nuzaihan, M.N. Enhanced sensitivity mediated ambipolar conduction with p-type TiO2 anatase transducer for biomarker capturing. Sens. Actuators Phys. 2017, 259, 57–67. [Google Scholar] [CrossRef]
- Gunda, N.S.K.; Singh, M.; Norman, L.; Kaur, K.; Mitra, S.K. Optimization and characterization of biomolecule immobilization on silicon substrates using (3-Aminopropyl)Triethoxysilane (APTES) and glutaraldehyde linker. Appl. Surf. Sci. 2014, 305, 522–530. [Google Scholar] [CrossRef]
- Fathil, M.F.M.; Md Arshad, M.K.; Ruslinda, A.R.; Gopinath, S.C.B.; Nuzaihan M.N., M.; Adzhri, R.; Hashim, U.; Lam, H.Y. Substrate-gate coupling in ZnO-FET biosensor for cardiac troponin I detection. Sens. Actuators B Chem. 2017, 242, 1142–1154. [Google Scholar] [CrossRef]
- Lee, J.-K.; Wang, I.-S.; Huang, C.-H.; Chen, Y.-F.; Huang, N.-T.; Lin, C.-T. Pre-Clinical Tests of an Integrated CMOS Biomolecular Sensor for Cardiac Diseases Diagnosis. Sensors 2017, 17. [Google Scholar] [CrossRef] [PubMed]
- Shen, S.-H.; Wang, I.-S.; Cheng, H.; Lin, C.-T. An enhancement of high-k/oxide stacked dielectric structure for silicon-based multi-nanowire biosensor in cardiac troponin I detection. Sens. Actuators B Chem. 2015, 218, 303–309. [Google Scholar] [CrossRef]
- Zhou, W.; Li, K.; Wei, Y.; Hao, P.; Chi, M.; Liu, Y.; Wu, Y. Ultrasensitive label-free optical microfiber coupler biosensor for detection of cardiac troponin I based on interference turning point effect. Biosens. Bioelectron. 2018, 106, 99–104. [Google Scholar] [CrossRef] [PubMed]
- Pierce Biotechnology. NHS and Sulfo-NHS; Thermo Fisher Scientific Inc.: Waltham, MA, USA, 2011. [Google Scholar]
- Hermanson, G.T. Chapter 11—(Strept)avidin–Biotin Systems. In Bioconjugate Techniques, 3rd ed.; Academic Press: Boston, MA, USA, 2013; pp. 465–505. [Google Scholar] [CrossRef]
- Han, K.N.; Le, T.-H.; Pham, X.-H.; Huynh-Nguyen, B.-C.; Kim, J.H.; Ko, E.; Kwon, H.T.; Seong, G.H. Immunoassay of cardiac biomarkers using a photodiode array biochip. Sens. Actuators B Chem. 2015, 207, 470–476. [Google Scholar] [CrossRef]
- Peluso, P.; Wilson, D.S.; Do, D.; Tran, H.; Venkatasubbaiah, M.; Quincy, D.; Heidecker, B.; Poindexter, K.; Tolani, N.; Phelan, M.; et al. Optimizing antibody immobilization strategies for the construction of protein microarrays. Anal. Biochem. 2003, 312, 113–124. [Google Scholar] [CrossRef]
- Vijayendran, R.A.; Leckband, D.E. A Quantitative Assessment of Heterogeneity for Surface-Immobilized Proteins. Anal. Chem. 2001, 73, 471–480. [Google Scholar] [CrossRef] [PubMed]
- Tate, J.; Ward, G. Interferences in Immunoassay. Clin. Biochem. Rev. 2004, 25, 105–120. [Google Scholar] [PubMed]
- Dupin, M.; Fortin, T.; Larue-Triolet, A.; Surault, I.; Beaulieu, C.; Gouel-Chéron, A.; Allaouchiche, B.; Asehnoune, K.; Roquilly, A.; Venet, F.; et al. Impact of Serum and Plasma Matrices on the Titration of Human Inflammatory Biomarkers Using Analytically Validated SRM Assays. J. Proteome Res. 2016, 15, 2366–2378. [Google Scholar] [CrossRef] [PubMed]
- Murosaki, T.; Ahmed, N.; Ping Gong, J. Antifouling properties of hydrogels. Sci. Technol. Adv. Mater. 2012, 12. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Cui, M.; Jiao, M.; Luo, X. Antifouling and ultrasensitive biosensing interface based on self-assembled peptide and aptamer on macroporous gold for electrochemical detection of immunoglobulin E in serum. Anal. Bioanal. Chem. 2018, 410, 5871–5878. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Wang, H.; Wang, Y.; Gui, T.; Wang, K.; Gao, C. Creation of antifouling microarrays by photopolymerization of zwitterionic compounds for protein assay and cell patterning. Biosens. Bioelectron. 2018, 102, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Chapman, J.; Regan, F. Nanofunctionalized Superhydrophobic Antifouling Coatings for Environmental Sensor Applications—Advancing Deployment with Answers from Nature. Adv. Eng. Mater. 2012, 14, B175–B184. [Google Scholar] [CrossRef]
- Noel, J.; Teizer, W.; Hwang, W. Antifouling Self-assembled Monolayers on Microelectrodes for Patterning Biomolecules. J. Vis. Exp. JoVE 2009. [Google Scholar] [CrossRef] [PubMed]
- Dhawan, S.; Sadanandan, S.; Haridas, V.; Voelcker, N.H.; Prieto-Simon, B. Novel peptidylated surfaces for interference-free electrochemical detection of cardiac troponin I. Biosens. Bioelectron. 2018, 99, 486–492. [Google Scholar] [CrossRef] [PubMed]
- Diware, M.S.; Cho, H.M.; Chegal, W.; Cho, Y.J.; Kim, D.S.; Won O, S.; Kim, K.-S.; Paek, S.-H. Ultrasensitive, label-free detection of cardiac biomarkers with optical SIS sensor. Biosens. Bioelectron. 2017, 87, 242–248. [Google Scholar] [CrossRef] [PubMed]
- Fan, D.; Bao, C.; Khan, M.S.; Wang, C.; Zhang, Y.; Liu, Q.; Zhang, X.; Wei, Q. A novel label-free photoelectrochemical sensor based on N,S-GQDs and CdS co-sensitized hierarchical Zn2SnO4 cube for detection of cardiac troponin I. Biosens. Bioelectron. 2018, 106, 14–20. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Xiong, C.; Wang, H.; Yuan, R.; Chai, Y. A sensitive electrochemiluminescence immunosensor for cardiac troponin I detection based on dual quenching of the self-enhanced Ru(II) complex by folic acid and in Situ generated oxygen. Sens. Actuators B Chem. 2017, 241, 765–772. [Google Scholar] [CrossRef]
- Rezaei, Z.; Ranjbar, B. Ultra-sensitive, rapid gold nanoparticle-quantum dot plexcitonic self-assembled aptamer-based nanobiosensor for the detection of human cardiac troponin I. Eng. Life Sci. 2017, 17, 165–174. [Google Scholar] [CrossRef]
- Liang, W.; Fan, C.; Zhuo, Y.; Zheng, Y.; Xiong, C.; Chai, Y.; Yuan, R. Multiparameter Analysis-Based Electrochemiluminescent Assay for Simultaneous Detection of Multiple Biomarker Proteins on a Single Interface. Anal. Chem. 2016, 88, 4940–4948. [Google Scholar] [CrossRef] [PubMed]
- Pan, M.; Gu, Y.; Yun, Y.; Li, M.; Jin, X.; Wang, S. Nanomaterials for Electrochemical Immunosensing. Sensors 2017, 17, 1041. [Google Scholar] [CrossRef]
- Thomas, G.K. Tuning functional properties: From nanoscale building blocks to hybrid nanomaterials. Platin. Jubil. Spec. Publ. 2010, 53–66. [Google Scholar]
- Bhatia, S. Natural Polymer Drug Delivery Systems: Nanoparticles, Plants, and Algae; Springer International Publishing: Berlin, Germany, 2016. [Google Scholar]
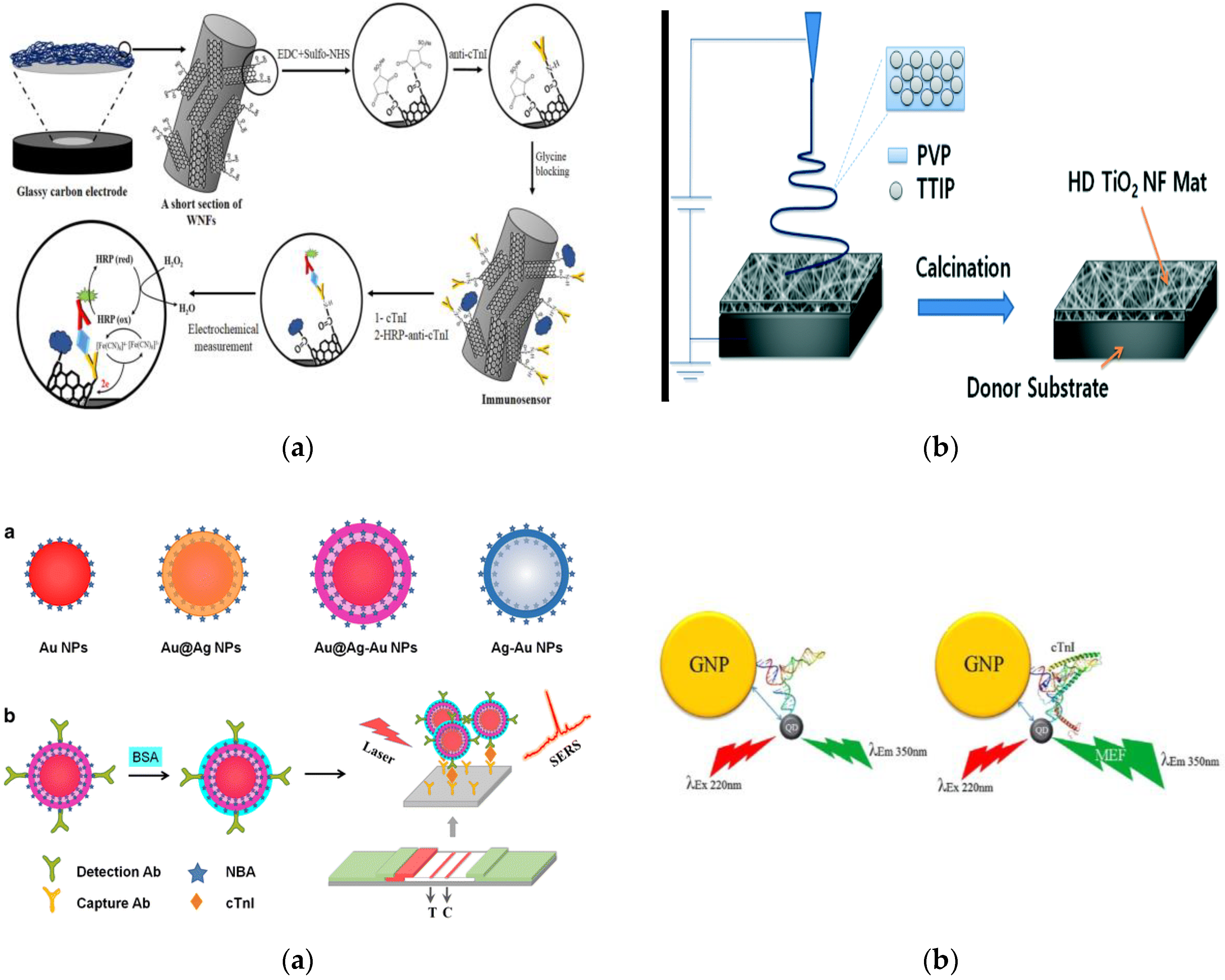
- Rezaei, B.; Shoushtari, A.M.; Rabiee, M.; Uzun, L.; Mak, W.C.; Turner, A.P.F. An electrochemical immunosensor for cardiac troponin I using electrospun carboxylated multi-walled carbon nanotube-whiskered nanofibres. Talanta 2018, 182, 178–186. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.S.; Sunkara, V.; Han, J.-R.; Park, Y.-S.; Cho, Y.-K. Electrospun TiO2 nanofiber integrated lab-on-a-disc for ultrasensitive protein detection from whole blood. Lab. Chip 2015, 15, 478–485. [Google Scholar] [CrossRef] [PubMed]
- Tan, C.M.; Arshad, M.K.M.; Fathil, M.F.M.; Adzhri, R.; Nuzaihan M.N., M.; Ruslinda, A.R.; Ibau, C.; Hashim, U. Interdigitated Electrodes Integrated with Zinc Oxide Nanoparticles for Cardiac Troponin I Biomarker Detection. In Proceedings of the 2016 IEEE International Conference on Semiconductor Electronics (ICSE), Kuala Lumpur, Malaysia, 17–19 August 2016; pp. 220–230. [Google Scholar] [CrossRef]
- Wu, Q.; Li, S.; Sun, Y.; Wang, J. Hollow gold nanoparticle-enhanced SPR based sandwich immunoassay for human cardiac troponin I. Microchim. Acta 2017, 184, 2395–2402. [Google Scholar] [CrossRef]
- Bai, T.; Wang, M.; Cao, M.; Zhang, J.; Zhang, K.; Zhou, P.; Liu, Z.; Liu, Y.; Guo, Z.; Lu, X. Functionalized Au@Ag-Au nanoparticles as an optical and SERS dual probe for lateral flow sensing. Anal. Bioanal. Chem. 2018, 410, 2291–2303. [Google Scholar] [CrossRef] [PubMed]
- LeRu, E.C.; Etchegoin, P.G. Principles of Surface-Enhanced Raman Spectroscopy: And Related Plasmonic Effects; Elsevier Science Bv: Amsterdam, The Netherlands, 2009. [Google Scholar]
- Li, J.-F.; Zhang, Y.-J.; Ding, S.-Y.; Panneerselvam, R.; Tian, Z.-Q. Core-Shell Nanoparticle-Enhanced Raman Spectroscopy. Chem. Rev. 2017, 117, 5002–5069. [Google Scholar] [CrossRef] [PubMed]
- Saha, K.; Agasti, S.S.; Kim, C.; Li, X.; Rotello, V.M. Gold Nanoparticles in Chemical and Biological Sensing. Chem. Rev. 2012, 112, 2739–2779. [Google Scholar] [CrossRef] [PubMed]
- Tram, D.T.N.; Wang, H.; Sugiarto, S.; Li, T.; Ang, W.H.; Lee, C.; Pastorin, G. Advances in Nanomaterials and Their Applications in Point of Care (POC) Devices for the Diagnosis of Infectious Diseases. Biotechnol. Adv. 2016, 34, 1275–1288. [Google Scholar] [CrossRef] [PubMed]
- Mirzaii-Dizgah, I.; Riahi, E. Salivary troponin I as an indicator of myocardial infarction. Indian J. Med. Res. 2013, 138, 861–865. [Google Scholar] [PubMed]
- Punyadeera, C. New frontiers in heart failure detection: Saliva testing. BMJ Innov. 2016, 2, 106–108. [Google Scholar] [CrossRef]
- Tan, Y.; Wang, Y.; Li, M.; Ye, X.; Wu, T.; Li, C. Enhanced photoelectrochemical immunosensing of cardiac troponin I based on energy transfer between N-acetyl-L-cysteine capped CdAgTe quantum dots and dodecahedral Au nanoparticles. Biosens. Bioelectron. 2017, 91, 741–746. [Google Scholar] [CrossRef] [PubMed]
- Zang, R.; He, Y.; Yuan, R.; Chai, Y. An ultrasensitive electrochemiluminescence immunosensor based on zeolitic imidazolate frameworks encapsulating spherical graphite crystals. J. Electroanal. Chem. 2016, 781, 284–288. [Google Scholar] [CrossRef]
- Valizadeh, B.; Nguyen, T.N.; Stylianou, K.C. Shape engineering of metal-organic frameworks. Polyhedron 2018, 145, 1–15. [Google Scholar] [CrossRef]
- Li, J.; Wang, X.; Zhao, G.; Chen, C.; Chai, Z.; Alsaedi, A.; Hayat, T.; Wang, X. Metal-organic framework-based materials: Superior adsorbents for the capture of toxic and radioactive metal ions. Chem. Soc. Rev. 2018, 47, 2322–2356. [Google Scholar] [CrossRef] [PubMed]
- Kreno, L.E.; Leong, K.; Farha, O.K.; Allendorf, M.; Van Duyne, R.P.; Hupp, J.T. Metal–Organic Framework Materials as Chemical Sensors. Chem. Rev. 2012, 112, 1105–1125. [Google Scholar] [CrossRef] [PubMed]
- Hu, Z.; Deibert, B.J.; Li, J. Luminescent Metal–organic Frameworks for Chemical Sensing and Explosive Detection. Chem. Soc. Rev. 2014, 43, 5815–5840. [Google Scholar] [CrossRef] [PubMed]
- Abbasi, E.; Aval, S.F.; Akbarzadeh, A.; Milani, M.; Nasrabadi, H.T.; Joo, S.W.; Hanifehpour, Y.; Nejati-Koshki, K.; Pashaei-Asl, R. Dendrimers: Synthesis, applications, and properties. Nanoscale Res. Lett. 2014, 9, 247. [Google Scholar] [CrossRef] [PubMed]
- Bhatnagar, D.; Kaur, I.; Kumar, A. Ultrasensitive cardiac troponin I antibody based nanohybrid sensor for rapid detection of human heart attack. Int. J. Biol. Macromol. 2017, 95, 505–510. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.-C.; Zhao, M.; Yu, Y.-Q.; Lei, Y.-M.; Chai, Y.-Q.; Yuan, R.; Zhuo, Y. Three-dimensional nano-network composed of Pt nanoparticles functionalized Mn-doped CeO2 and hemin/G-quadruplex as electrocatalysts for cardiovascular biomarker detection. Sens. Actuators B Chem. 2017, 246, 1–8. [Google Scholar] [CrossRef]
- El Harrad, L.; Bourais, I.; Mohammadi, H.; Amine, A. Recent Advances in Electrochemical Biosensors Based on Enzyme Inhibition for Clinical and Pharmaceutical Applications. Sensors 2018, 18. [Google Scholar] [CrossRef] [PubMed]
- Bansod, B.; Kumar, T.; Thakur, R.; Rana, S.; Singh, I. A review on various electrochemical techniques for heavy metal ions detection with different sensing platforms. Biosens. Bioelectron. 2017, 94, 443–455. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Kumar, S.; Augustine, S.; Malhotra, B.D. Protein functionalized nanostructured zirconia based electrochemical immunosensor for cardiac troponin I detection. J. Mater. Res. 2017, 32, 2966–2972. [Google Scholar] [CrossRef]
- Singal, S.; Srivastava, A.K.; Dhakate, S.; Biradara, A.M.; Rajesh. Electroactive graphene-multi-walled carbon nanotube hybrid supported impedimetric immunosensor for the detection of human cardiac troponin-I. RSC Adv. 2015, 5, 74994–75003. [Google Scholar] [CrossRef]
- Lahtinen, S.; Lyytikäinen, A.; Sirkka, N.; Päkkilä, H.; Soukka, T. Improving the sensitivity of immunoassays by reducing non-specific binding of poly(acrylic acid) coated upconverting nanoparticles by adding free poly(acrylic acid). Microchim. Acta 2018, 185, 220. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Qiu, H.; Prasad, P.N.; Chen, X. Upconversion Nanoparticles: Design, Nanochemistry, and Applications in Theranostics. Chem. Rev. 2014, 114, 5161–5214. [Google Scholar] [CrossRef] [PubMed]
- Apple, F.S. Cardiac troponin assays. Cardiovasc. Toxicol. 2001, 1, 93. [Google Scholar] [CrossRef]
- Gong, X.; Zhang, B.; Piao, J.; Zhao, Q.; Gao, W.; Peng, W.; Kang, Q.; Zhou, D.; Shu, G.; Chang, J. High sensitive and multiple detection of acute myocardial infarction biomarkers based on a dual-readout immunochromatography test strip. Nanomed. Nanotechnol. Biol. Med. 2018, 14, 1257–1266. [Google Scholar] [CrossRef] [PubMed]
- Matta, D.P.; Tripathy, S.; Vanjari, S.R.K.; Sharma, C.S.; Singh, S.G. An ultrasensitive label free nanobiosensor platform for the detection of cardiac biomarkers. Biomed. Microdevices 2016, 18, 111. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Yu, Y.-Q.; Peng, L.-Z.; Lei, Y.-M.; Chai, Y.-Q.; Yuan, R.; Zhuo, Y. Strong Electrochemiluminescence from MOF Accelerator Enriched Quantum Dots for Enhanced Sensing of Trace CTnI. Anal. Chem. 2018, 90, 3995–4002. [Google Scholar] [CrossRef] [PubMed]
- Jarolim, P.; Patel, P.P.; Conrad, M.J.; Chang, L.; Melenovsky, V.; Wilson, D.H. Fully Automated Ultrasensitive Digital Immunoassay for Cardiac Troponin I Based on Single Molecule Array Technology. Clin. Chem. 2015, 61, 1283–1291. [Google Scholar] [CrossRef] [PubMed]
- Wilson, D.H.; Rissin, D.M.; Kan, C.W.; Fournier, D.R.; Piech, T.; Campbell, T.G.; Meyer, R.E.; Fishburn, M.W.; Cabrera, C.; Patel, P.P.; et al. The Simoa HD-1 Analyzer: A Novel Fully Automated Digital Immunoassay Analyzer with Single-Molecule Sensitivity and Multiplexing. J. Lab. Autom. 2016, 21, 533–547. [Google Scholar] [CrossRef] [PubMed]
- Shanmugam, N.R.; Muthukumar, S.; Prasad, S. Surface modification of ZnO nanostructured electrodes with thiol and phosphonic acid moieties for biosensing applications. Anal. Methods 2017, 9, 5525–5533. [Google Scholar] [CrossRef]
- Qin, X.; Sui, Y.; Xu, A.; Liu, L.; Li, Y.; Tan, Y.; Chen, C.; Xie, Q. Ultrasensitive immunoassay of proteins based on in-situ enzymatic formation of quantum dots and microliter-droplet anodic stripping voltammetry. J. Electroanal. Chem. 2018, 811, 121–127. [Google Scholar] [CrossRef]
- Qin, X.; Gu, C.; Wang, M.; Dong, Y.; Nie, X.; Li, M.; Zhu, Z.; Yang, D.; Shao, Y. Triethanolamine-Modified Gold Nanoparticles Synthesized by a One-Pot Method and Their Application in Electrochemiluminescent Immunoassy. Anal. Chem. 2018, 90, 2826–2832. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Park, G.; Chakkarapani, S.K.; Kang, S.H. Ultra-sensitive plasmonic nanometal scattering immunosensor based on optical control in the evanescent field layer. Biosens. Bioelectron. 2015, 63, 444–449. [Google Scholar] [CrossRef] [PubMed]
- Syahir, A.; Usui, K.; Tomizaki, K.; Kajikawa, K.; Mihara, H. Label and Label-Free Detection Techniques for Protein Microarrays. Microarrays 2015, 4, 228–244. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.-J.; Cho, H.Y.; Kim, B.K.; Huh, C.; Chung, K.H.; Ahn, C.-G.; Kim, Y.J.; Kim, A. Highly sensitive detection of cardiac troponin I in human serum using gold nanoparticle-based enhanced sandwich immunoassay. Sens. Actuators B Chem. 2015, 221, 537–543. [Google Scholar] [CrossRef]
- Homola, J.; Yee, S.S.; Gauglitz, G. Surface plasmon resonance sensors: Review. Sens. Actuators B Chem. 1999, 54, 3–15. [Google Scholar] [CrossRef]
- Wu, Q.; Sun, Y.; Zhang, D.; Li, S.; Zhang, Y.; Ma, P.; Yu, Y.; Wang, X.; Song, D. Ultrasensitive magnetic field-assisted surface plasmon resonance immunoassay for human cardiac troponin I. Biosens. Bioelectron. 2017, 96, 288–293. [Google Scholar] [CrossRef] [PubMed]
- Jo, H.; Her, J.; Lee, H.; Shim, Y.-B.; Ban, C. Highly sensitive amperometric detection of cardiac troponin I using sandwich aptamers and screen-printed carbon electrodes. Talanta 2017, 165, 442–448. [Google Scholar] [CrossRef] [PubMed]
- Bhatnagar, D.; Kumar, V.; Kumar, A.; Kaur, I. Graphene quantum dots FRET based sensor for early detection of heart attack in human. Biosens. Bioelectron. 2016, 79, 495–499. [Google Scholar] [CrossRef] [PubMed]
- Shu, J.; Tang, D. Current Advances in Quantum-Dots-Based Photoelectrochemical Immunoassays. Chem. Asian J. 2017, 12, 2780–2789. [Google Scholar] [CrossRef] [PubMed]
- Akter, R.; Jeong, B.; Lee, Y.-M.; Choi, J.-S.; Rahman, M.A. Femtomolar detection of cardiac troponin I using a novel label-free and reagent-free dendrimer enhanced impedimetric immunosensor. Biosens. Bioelectron. 2017, 91, 637–643. [Google Scholar] [CrossRef] [PubMed]
- Qiao, X.; Li, K.; Xu, J.; Cheng, N.; Sheng, Q.; Cao, W.; Yue, T.; Zheng, J. Novel electrochemical sensing platform for ultrasensitive detection of cardiac troponin I based on aptamer-MoS2 nanoconjugates. Biosens. Bioelectron. 2018, 113, 142–147. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Lu, X.; Yang, Y.; Zhai, Y.; Zhang, J.; Li, L. A novel fluorescent aptasensor for the highly sensitive and selective detection of cardiac troponin I based on a graphene oxide platform. Anal. Bioanal. Chem. 2018, 410, 4285–4291. [Google Scholar] [CrossRef] [PubMed]
- Zhao, W.-W.; Chen, R.; Dai, P.-P.; Li, X.-L.; Xu, J.-J.; Chen, H.-Y. A General Strategy for Photoelectrochemical Immunoassay Using an Enzyme Label Combined with a CdS Quantum Dot/TiO2 Nanoparticle Composite Electrode. Anal. Chem. 2014, 86, 11513–11516. [Google Scholar] [CrossRef] [PubMed]
- Zhang, N.; Ma, Z.-Y.; Ruan, Y.-F.; Zhao, W.-W.; Xu, J.-J.; Chen, H.-Y. Simultaneous Photoelectrochemical Immunoassay of Dual Cardiac Markers Using Specific Enzyme Tags: A Proof of Principle for Multiplexed Bioanalysis. Anal. Chem. 2016, 88, 1990–1994. [Google Scholar] [CrossRef] [PubMed]
- Fan, G.-C.; Zhu, H.; Du, D.; Zhang, J.-R.; Zhu, J.-J.; Lin, Y. Enhanced Photoelectrochemical Immunosensing Platform Based on CdSeTe@CdS:Mn Core–Shell Quantum Dots-Sensitized TiO2 Amplified by CuS Nanocrystals Conjugated Signal Antibodies. Anal. Chem. 2016, 88, 3392–3399. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.-Y.; Chang, C.-L.; Wang, Y.-N.; Fu, L.-M. Microfluidic Mixing: A Review. Int. J. Mol. Sci. 2011, 12, 3263–3287. [Google Scholar] [CrossRef] [PubMed]
- Cai, G.; Xue, L.; Zhang, H.; Lin, J. A Review on Micromixers. Micromachines 2017, 8, 274. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.C.; Lee, H.; Lim, J.; Park, Y.J. An effective electrical sensing scheme using AC electrothermal flow on a biosensor platform based on a carbon nanotube network. Appl. Phys. Lett. 2016, 109, 223701. [Google Scholar] [CrossRef]
- Sharma, A.; Han, C.-H.; Jang, J. Rapid electrical immunoassay of the cardiac biomarker troponin I through dielectrophoretic concentration using imbedded electrodes. Biosens. Bioelectron. 2016, 82, 78–84. [Google Scholar] [CrossRef] [PubMed]
- St John, A.; Price, C.P. Existing and Emerging Technologies for Point-of-Care Testing. Clin. Biochem. Rev. 2014, 35, 155–167. [Google Scholar] [PubMed]
- St John, A.; Price, C.P. Economic Evidence and Point-of-Care Testing. Clin. Biochem. Rev. 2013, 34, 61–74. [Google Scholar] [PubMed]
- Singh, N.; Ali, M.A.; Ra, P.; Sharma, A.; Malhotra, B.D.; John, R. Microporous Nanocomposite Enabled Microfluidic Biochip for Cardiac Biomarker Detection. ACS Appl. Mater. Interfaces 2017, 9, 33576–33588. [Google Scholar] [CrossRef] [PubMed]
- Minitha, C.R.; Anithaa, V.S.; Subramaniam, V.; Rajendra Kumar, R.T. Impact of Oxygen Functional Groups on Reduced Graphene Oxide-Based Sensors for Ammonia and Toluene Detection at Room Temperature. ACS Omega 2018, 3, 4105–4112. [Google Scholar] [CrossRef]
- Rodenko, O.; Eriksson, S.; Tidemand-Lichtenberg, P.; Troldborg, C.P.; Fodgaard, H.; van Os, S.; Pedersen, C. High-sensitivity detection of cardiac troponin I with UV LED excitation for use in point-of-care immunoassay. Biomed. Opt. Express 2017, 8, 3749–3762. [Google Scholar] [CrossRef] [PubMed]
- Sarangadharan, I.; Regmi, A.; Chen, Y.-W.; Hsu, C.-P.; Chen, P.; Chang, W.-H.; Lee, G.-Y.; Chyi, J.-I.; Shiesh, S.-C.; Lee, G.-B.; et al. High sensitivity cardiac troponin I detection in physiological environment using AlGaN/GaN High Electron Mobility Transistor (HEMT) Biosensors. Biosens. Bioelectron. 2018, 100, 282–289. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.R.; Chun, H.J.; Han, Y.D.; Lee, K.W.; Hong, D.K.; Lee, K.N.; Yoon, H.C. A Time-Resolved Fluorescence Immunosensing Platform for Highly Sensitive Detection of Cardiac Troponin I. In Proceedings of the 19th International Conference on Solid-State Sensors, Actuators and Microsystems (TRANSDUCERS), Kaohsiung, Taiwan, 18–22 June 2017; pp. 1640–1643. [Google Scholar] [CrossRef]
- Ravkilde, J.; Nissen, H.; Hørder, M.; Thygesen, K. Independent prognostic value of serum creatine kinase isoenzyme MB mass, cardiac troponin T and myosin light chain levels in suspected acute myocardial infarction. Analysis of 28 months of follow-up in 196 patients. J. Am. Coll. Cardiol. 1995, 25, 574–581. [Google Scholar] [CrossRef]
- Rahman, M.M.; Alam, M.M.; Jahan, N.A.; Shila, J.S.; Arslam, M.I. Prognostic Role of Multiple Cardiac Biomarkers in Newly Diagnosed Acute Coronary Syndrome Patients. Mymensingh Med. J. MMJ 2016, 25, 326–333. [Google Scholar] [PubMed]
- Montague, C.; Kircher, T. Myoglobin in the early evaluation of acute chest pain. Am. J. Clin. Pathol. 1995, 104, 472–476. [Google Scholar] [CrossRef] [PubMed]
- Tucker, J.F.; Collins, R.A.; Anderson, A.J.; Hauser, J.; Kalas, J.; Apple, F.S. Early diagnostic efficiency of cardiac troponin I and troponin T for acute myocardial infarction. Acad. Emerg. Med. Off. J. Soc. Acad. Emerg. Med. 1997, 4, 13–21. [Google Scholar] [CrossRef]
- Zethelius, B.; Berglund, L.; Sundström, J.; Ingelsson, E.; Basu, S.; Larsson, A.; Venge, P.; Arnlöv, J. Use of multiple biomarkers to improve the prediction of death from cardiovascular causes. N. Engl. J. Med. 2008, 358, 2107–2116. [Google Scholar] [CrossRef] [PubMed]
- Hung, C.-L.; Chien, D.-K.; Shih, S.-C.; Chang, W.-H. The feasibility and diagnostic accuracy by multiple cardiac biomarkers in emergency chest pain patients: A clinical analysis to compare 290 Suspected acute coronary syndrome cases stratified by age and gender in Taiwan. BMC Cardiovasc. Disord. 2016, 16. [Google Scholar] [CrossRef] [PubMed]
- Shanmugam, N.R.; Muthukumar, S.; Chaudhry, S.; Anguiano, J.; Prasad, S. Ultrasensitive nanostructure sensor arrays on flexible substrates for multiplexed and simultaneous electrochemical detection of a panel of cardiac biomarkers. Biosens. Bioelectron. 2017, 89, 764–772. [Google Scholar] [CrossRef] [PubMed]
- Gustafsson, F.; Steensgaard-Hansen, F.; Badskjaer, J.; Poulsen, A.H.; Corell, P.; Hildebrandt, P. Diagnostic and prognostic performance of N-terminal ProBNP in primary care patients with suspected heart failure. J. Card. Fail. 2005, 11, S15–S20. [Google Scholar] [CrossRef] [PubMed]
- Sonawane, M.D.; Nimse, S.B.; Song, K.-S.; Kim, T. Multiplex detection of cardiac biomarkers. Anal. Methods 2017, 9, 3773–3776. [Google Scholar] [CrossRef]
- Weber, M.; Hamm, C. Role of B-type natriuretic peptide (BNP) and NT-proBNP in clinical routine. Heart 2006, 92, 843–849. [Google Scholar] [CrossRef] [PubMed]
- Song, K.-S.; Nimse, S.B.; Kim, J.; Kim, J.; Nguyen, V.-T.; Ta, V.-T.; Kim, T. 9G DNAChip: Microarray based on the multiple interactions of 9 consecutive guanines. Chem. Commun. Camb. Engl. 2011, 47, 7101–7103. [Google Scholar] [CrossRef] [PubMed]
- Niemeyer, C.M.; Boldt, L.; Ceyhan, B.; Blohm, D. DNA-Directed immobilization: Efficient, reversible, and site-selective surface binding of proteins by means of covalent DNA-streptavidin conjugates. Anal. Biochem. 1999, 268, 54–63. [Google Scholar] [CrossRef] [PubMed]
- Folsom, A.R.; Chambless, L.E.; Ballantyne, C.M.; Coresh, J.; Heiss, G.; Wu, K.K.; Boerwinkle, E.; Mosley, T.H.; Sorlie, P.; Diao, G.; et al. An assessment of incremental coronary risk prediction using C-reactive protein and other novel risk markers: The atherosclerosis risk in communities study. Arch. Intern. Med. 2006, 166, 1368–1373. [Google Scholar] [CrossRef] [PubMed]
- Nissen, S.E.; Tuzcu, E.M.; Schoenhagen, P.; Crowe, T.; Sasiela, W.J.; Tsai, J.; Orazem, J.; Magorien, R.D.; O’Shaughnessy, C.; Ganz, P.; et al. Statin therapy, LDL cholesterol, C-reactive protein, and coronary artery disease. N. Engl. J. Med. 2005, 352, 29–38. [Google Scholar] [CrossRef] [PubMed]
- Azuaje, F.J.; Dewey, F.E.; Brutsaert, D.L.; Devaux, Y.; Ashley, E.A.; Wagner, D.R. Systems-Based Approaches to Cardiovascular Biomarker Discovery. Circ. Genomic Precis. Med. 2012, 5, 360–367. [Google Scholar] [CrossRef] [PubMed]
- Meijers, W.C.; van der Velde, A.R.; Muller Kobold, A.C.; Dijck-Brouwer, J.; Wu, A.H.; Jaffe, A.; de Boer, R.A. Variability of biomarkers in patients with chronic heart failure and healthy controls. Eur. J. Heart Fail. 2017, 19, 357–365. [Google Scholar] [CrossRef] [PubMed]
- Melander, O.; Newton-Cheh, C.; Almgren, P.; Hedblad, B.; Berglund, G.; Engström, G.; Persson, M.; Smith, J.G.; Magnusson, M.; Christensson, A.; et al. Novel and conventional biomarkers for the prediction of incident cardiovascular events in the community. JAMA J. Am. Med. Assoc. 2009, 302, 49–57. [Google Scholar] [CrossRef] [PubMed]
- Straface, A.L.; Myers, J.H.; Kirchick, H.J.; Blick, K.E. A rapid point-of-care cardiac marker testing strategy facilitates the rapid diagnosis and management of chest pain patients in the emergency department. Am. J. Clin. Pathol. 2008, 129, 788–795. [Google Scholar] [CrossRef] [PubMed]
- Jaeger, C.; Wildi, K.; Twerenbold, R.; Reichlin, T.; Rubini Gimenez, M.; Neuhaus, J.-D.; Grimm, K.; Boeddinghaus, J.; Hillinger, P.; Nestelberger, T.; et al. One-hour rule-in and rule-out of acute myocardial infarction using high-sensitivity cardiac troponin I. Am. Heart J. 2016, 171, 92–102. [Google Scholar] [CrossRef] [PubMed]
- Sajid, M.; Kawde, A.-N.; Daud, M. Designs, formats and applications of lateral flow assay: A literature review. J. Saudi Chem. Soc. 2015, 19, 689–705. [Google Scholar] [CrossRef]
- O’Farrell, B. Lateral Flow Technology for Field-Based Applications—Basics and Advanced Developments. Top. Companion Anim. Med. 2015, 30, 139–147. [Google Scholar] [CrossRef] [PubMed]
- Cai, Y.; Kang, K.; Li, Q.; Wang, Y.; He, X. Rapid and Sensitive Detection of Cardiac Troponin I for Point-of-Care Tests Based on Red Fluorescent Microspheres. Mol. Basel Switz. 2018, 23. [Google Scholar] [CrossRef] [PubMed]
- Lou, D.; Fan, L.; Cui, Y.; Zhu, Y.; Gu, N.; Zhang, Y. Fluorescent Nanoprobes with Oriented Modified Antibodies to Improve Lateral Flow Immunoassay of Cardiac Troponin I. Anal. Chem. 2018, 90, 6502–6508. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Huang, L.; Liu, B.; Ni, H.; Sun, L.; Su, E.; Chen, H.; Gu, Z.; Zhao, X. Quantitative and ultrasensitive detection of multiplex cardiac biomarkers in lateral flow assay with core-shell SERS nanotags. Biosens. Bioelectron. 2018, 106, 204–211. [Google Scholar] [CrossRef] [PubMed]
- Amundson, B.E.; Apple, F.S. Cardiac Troponin Assays: A review of quantitative point-of-care devices and their efficacy in the diagnosis of myocardial infarction. Clin. Chem. Lab. Med. 2015, 53, 665–676. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Zapatero-Rodríguez, J.; Estrela, P.; O’Kennedy, R. Point-of-Care Diagnostics in Low Resource Settings: Present Status and Future Role of Microfluidics. Biosensors 2015, 5, 577–601. [Google Scholar] [CrossRef] [PubMed]
- Ainla, A.; Mousavi, M.P.S.; Tsaloglou, M.-N.; Redston, J.; Bell, J.G.; Fernandez-Abedul, M.T.; Whitesides, G.M. Open-Source Potentiostat for Wireless Electrochemical Detection with Smartphones. Anal. Chem. 2018, 90, 6240–6246. [Google Scholar] [CrossRef] [PubMed]
- Guner, H.; Ozgur, E.; Kokturk, G.; Celik, M.; Esen, E.; Topal, A.E.; Ayas, S.; Uludag, Y.; Elbuken, C.; Dana, A. A smartphone based surface plasmon resonance imaging (SPRi) platform for on-site biodetection. Sens. Actuators B Chem. 2017, 239, 571–577. [Google Scholar] [CrossRef]
- Sipova, H.; Piliarik, M.; Vala, M.; Chadt, K.; Adam, P.; Bockova, M.; Hegnerova, K.; Homola, J. Portable surface plasmon resonance biosensor for detection of nucleic acids. In Eurosensors Xxv; Kaltsas, G., Tsamis, C., Eds.; Elsevier Science Bv: Amsterdam, The Netherlands, 2011; Volume 25. [Google Scholar]
- Li, B.; Yang, S.; Akkus, O. A customized Raman system for point-of-care detection of arthropathic crystals in the synovial fluid. Analyst 2014, 139, 823–830. [Google Scholar] [CrossRef] [PubMed]
- Blacksberg, J.; Alerstam, E.; Maruyama, Y.; Cochrane, C.J.; Rossman, G.R. Miniaturized time-resolved Raman spectrometer for planetary science based on a fast single photon avalanche diode detector array. Appl. Opt. 2016, 55, 739–748. [Google Scholar] [CrossRef] [PubMed]
- Tang, M.; Zhou, Z.; Shangguan, L.; Zhao, F.; Liu, S. Electrochemiluminescent detection of cardiac troponin I by using soybean peroxidase labeled-antibody as signal amplifier. Talanta 2018, 180, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.R.; Han, Y.D.; Chun, H.J.; Lee, K.W.; Hong, D.-K.; Lee, K.-N.; Yoon, H.C. Encapsulation-Stabilized, Europium Containing Nanoparticle as a Probe for Time-Resolved luminescence Detection of Cardiac Troponin I. Biosensors-Basel 2017, 7, 48. [Google Scholar] [CrossRef] [PubMed]
- Xiong, M.; Wang, X.; Kong, Y.; Han, B. Development of Cardiac Troponin I Electrochemical Impedance Immunosensor. Int. J. Electrochem. Sci. 2017, 12, 4204–4214. [Google Scholar] [CrossRef]
- Li, Z.; Ma, K.; Cheng, Z.; Yan, C.; Liu, G. Fabrication of Electrochemical Immunosensor for Cardiac Biomarker Troponin I Determination and Its Potential for Acute Myocardial Infarction Diagnosis. Int. J. Electrochem. Sci. 2017, 2389–2399. [Google Scholar] [CrossRef]
- Kim, K.; Park, C.; Kwon, D.; Kim, D.; Meyyappan, M.; Jeon, S.; Lee, J.-S. Silicon nanowire biosensors for detection of cardiac troponin I (cTnI) with high sensitivity. Biosens. Bioelectron. 2016, 77, 695–701. [Google Scholar] [CrossRef] [PubMed]
- Chekin, F.; Vasilescu, A.; Jijie, R.; Singh, S.K.; Kurungot, S.; Iancu, M.; Badea, G.; Boukherroub, R.; Szunerits, S. Sensitive electrochemical detection of cardiac troponin I in serum and saliva by nitrogen-doped porous reduced graphene oxide electrode. Sens. Actuators B Chem. 2018, 262, 180–187. [Google Scholar] [CrossRef]
- Negahdary, M.; Behjati-Ardakani, M.; Sattarahmady, N.; Yadegari, H.; Heli, H. Electrochemical aptasensing of human cardiac troponin I based on an array of gold nanodumbbells-Applied to early detection of myocardial infarction. Sens. Actuators B Chem. 2017, 252, 62–71. [Google Scholar] [CrossRef]
- Yang, J.; Carey, P.; Ren, F.; Wang, Y.-L.; Good, M.L.; Jang, S.; Mastro, M.A.; Pearton, S.J. Rapid detection of cardiac troponin I using antibody-immobilized gate-pulsed AlGaN/GaN high electron mobility transistor structures. Appl. Phys. Lett. 2017, 111, 202104. [Google Scholar] [CrossRef]
- Sarangadharan, I.; Wang, S.-L.; Sukesan, R.; Chen, P.; Dai, T.-Y.; Pulikkathodi, A.K.; Hsu, C.-P.; Chiang, H.-H.K.; Liu, L.Y.-M.; Wang, Y.-L. Single Drop Whole Blood Diagnostics: Portable Biomedical Sensor for Cardiac Troponin I Detection. Anal. Chem. 2018, 90, 2867–2874. [Google Scholar] [CrossRef] [PubMed]
- Prakash, M.D.; Singh, S.G.; Sharma, C.S.; Krishna, V.S.R. Electrochemical Detection of Cardiac Biomarkers Utilizing Electrospun Multiwalled Carbon Nanotubes Embedded SU-8 Nanofibers. Electroanalysis 2017, 29, 380–386. [Google Scholar] [CrossRef]
- Kazemi, S.H.; Ghodsi, E.; Abdollahi, S.; Nadri, S. Porous graphene oxide nanostructure as an excellent scaffold for label-free electrochemical biosensor: Detection of cardiac troponin I. Mater. Sci. Eng. C Mater. Biol. Appl. 2016, 69, 447–452. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Jing, R.; Qi, H.; Gao, Q.; Zhang, C. Label-free electrochemical impedance peptide-based biosensor for the detection of cardiac troponin I incorporating gold nanoparticles modified carbon electrode. J. Electroanal. Chem. 2016, 781, 212–217. [Google Scholar] [CrossRef]
- Gupta, R.K.; Pandya, R.; Sieffert, T.; Meyyappan, M.; Koehne, J.E. Multiplexed electrochemical immunosensor for label-free detection of cardiac markers using a carbon nanofiber array chip. J. Electroanal. Chem. 2016, 773, 53–62. [Google Scholar] [CrossRef]
- Sandil, D.; Kumar, S.; Arora, K.; Srivastava, S.; Malhotra, B.D.; Sharma, S.C.; Puri, N.K. Biofunctionalized nanostructured tungsten trioxide based sensor for cardiac biomarker detection. Mater. Lett. 2017, 186, 202–205. [Google Scholar] [CrossRef]
- Singal, S.; Srivastava, A.K.; Biradar, A.M.; Mulchandani, A.; Rajesh. Pt nanoparticles-chemical vapor deposited graphene composite based immunosensor for the detection of human cardiac troponin I. Sens. Actuators B Chem. 2014, 205, 363–370. [Google Scholar] [CrossRef]
- Kim, S.-W.; Cho, I.-H.; Park, J.-N.; Seo, S.-M.; Paek, S.-H. A High-Performance Fluorescence Immunoassay Based on the Relaxation of Quenching, Exemplified by Detection of Cardiac Troponin I. Sensors 2016, 16, 669. [Google Scholar] [CrossRef] [PubMed]
- Singal, S.; Srivastava, A.K.; Rajesh. Electrochemical Impedance Analysis of Biofunctionalized Conducting Polymer-Modified Graphene-CNTs Nanocomposite for Protein Detection. Nano-Micro Lett. 2017, 9, 7. [Google Scholar] [CrossRef]
- Miao, L.; Zhang, L.; Jiao, L.; Tan, X.; Wei, Q.; Li, H. pH Readout enhanced ELISA for point-of-care testing of cardiac troponin I. Chin. Chem. Lett. 2017, 28, 1878–1880. [Google Scholar] [CrossRef]
- Jo, H.; Gu, H.; Jeon, W.; Youn, H.; Her, J.; Kim, S.-K.; Lee, J.; Shin, J.H.; Ban, C. Electrochemical Aptasensor of Cardiac Troponin I for the Early Diagnosis of Acute Myocardial Infarction. Anal. Chem. 2015, 87, 9869–9875. [Google Scholar] [CrossRef] [PubMed]
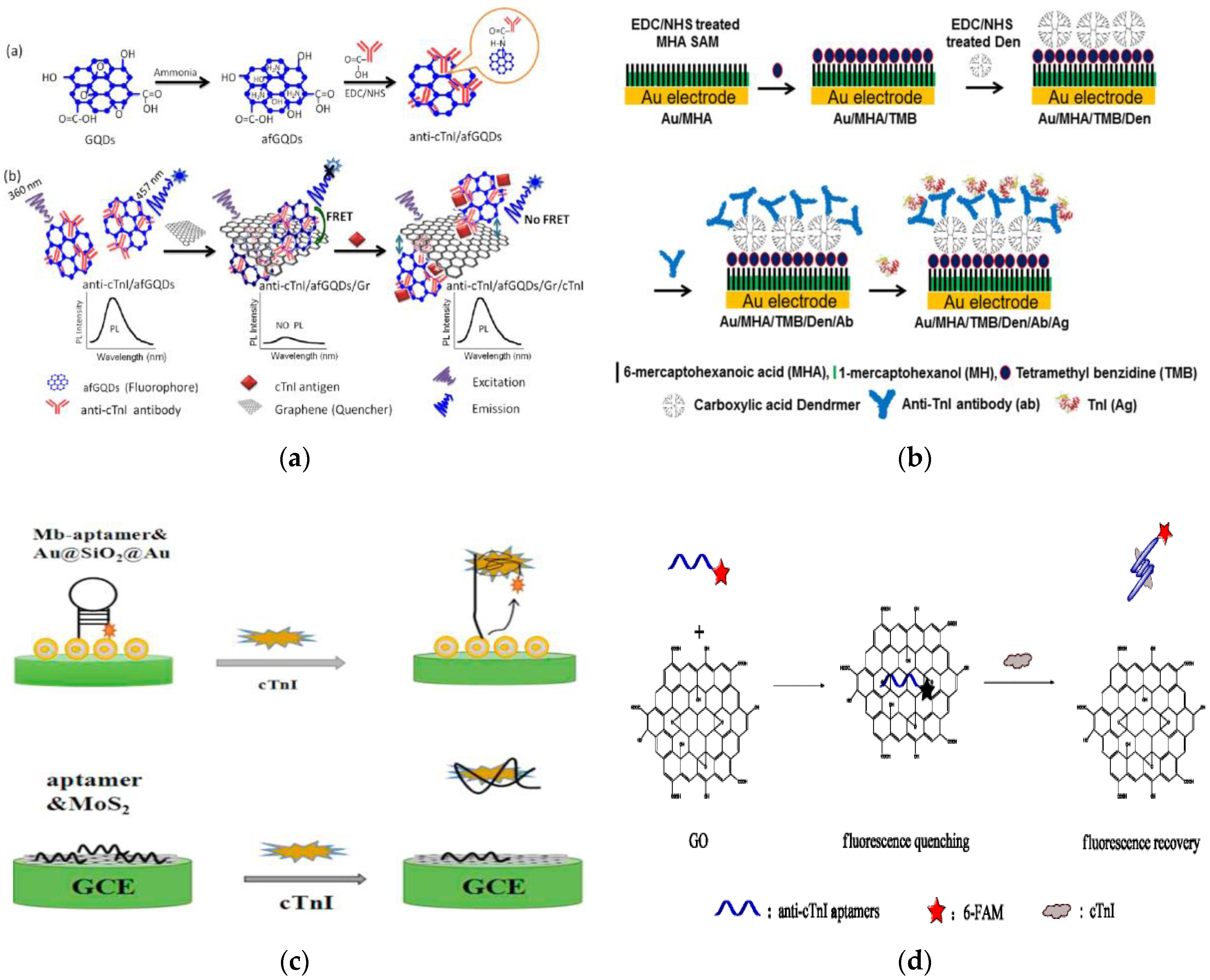

| Detection Approach | Biorecognition Element | Immobilisation Strategy | LOD |
|---|---|---|---|
| Optical [85] | Monoclonal Antibody | Ab/EDC/NHS/Dextran SAM/Tboc/Si Wafer | 5 ng/L |
| Photoelectrochemical [86] | Antibody * | Ab/EDC/NHS/TGA/CdS/N,S-GQDs/Zn2SnO4/ITO | 0.3 ng/L |
| Luminescence [87] | Monoclonal Antibody | Ab/Ru/EDC/NHS/PAMAM/AuNPs/Nafion/GCE | 12 pg/L |
| Electrochemical [61] | MIPs | MIPs/MWCNT/GS/GCE | 0.8 ng/L |
| Luminescence [88] | Aptamer | QD/EDC/NHS/L1/Aptamer/L2/DTT/AuNP | 7.2 μg/L |
| Luminescence [89] | Antibody* | Biotin-Ab/Streptavidin/Magnetic-NPs/GCP | 0.2 ng/L |
| Detection Method | Immobilisation Approach | Response Time (min) | LOD (ng/L) | Labels |
|---|---|---|---|---|
| Chemiresistance [121] | Ab/EDC/NHS/SU-8/MWCNT | 1 | 0.02 | None |
| Cyclic Voltammetry [111] | Ab/PAMAM/GQD/ATP/Au/SPE | 10 | 0.025 | None |
| Optical Microfiber Coupler [71] | Ab/EDC/NHS/PAA/PDDA/OMC | 10 | 0.002 | None |
| EIS & Mott-Schottky [125] | Ab/AUPA/ZnO/Au & Ab/DSP/ZnO/Au | 15 | 0.1 | None |
| Fluorescence [123] | Ab/Paramagnetic Beads | 45 | 0.01 | Biotinylated detection Ab/SβG |
| ECL [87] | Ru/EDC/NHS/PAMAM/AuNPs/Nafion/GCE | 115 | 0.012 | Detection Ab/PtCu3 NC/FA/GOD |
| ECL [122] | Ab/Au@TiO2/GCE | 120 | 0.00046 | Detection Ab/EDC/NHS/CdTe@IRMOF-3@CdTe |
| DPV—Anodic Stripping Voltammetry [126] | Ab/ABA/MWCNTs/GCE | 240 | 0.0011 | CdS/Detection Ab/ALP/AuNF |
| ECL [127] | Ab/TEOA@AuNP/GCE | - | 0.0055 | RuSiO2 |
| Total Internal Reflection [128] | Ab/Protein A/G/DSP/Au/Cr | - | 0.0000144 | Detection Ab/AgNP |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Regan, B.; O’Kennedy, R.; Collins, D. Point-of-Care Compatibility of Ultra-Sensitive Detection Techniques for the Cardiac Biomarker Troponin I—Challenges and Potential Value. Biosensors 2018, 8, 114. https://doi.org/10.3390/bios8040114
Regan B, O’Kennedy R, Collins D. Point-of-Care Compatibility of Ultra-Sensitive Detection Techniques for the Cardiac Biomarker Troponin I—Challenges and Potential Value. Biosensors. 2018; 8(4):114. https://doi.org/10.3390/bios8040114
Chicago/Turabian StyleRegan, Brian, Richard O’Kennedy, and David Collins. 2018. "Point-of-Care Compatibility of Ultra-Sensitive Detection Techniques for the Cardiac Biomarker Troponin I—Challenges and Potential Value" Biosensors 8, no. 4: 114. https://doi.org/10.3390/bios8040114
APA StyleRegan, B., O’Kennedy, R., & Collins, D. (2018). Point-of-Care Compatibility of Ultra-Sensitive Detection Techniques for the Cardiac Biomarker Troponin I—Challenges and Potential Value. Biosensors, 8(4), 114. https://doi.org/10.3390/bios8040114





