Biosensors for Non-Invasive Detection of Celiac Disease Biomarkers in Body Fluids
Abstract
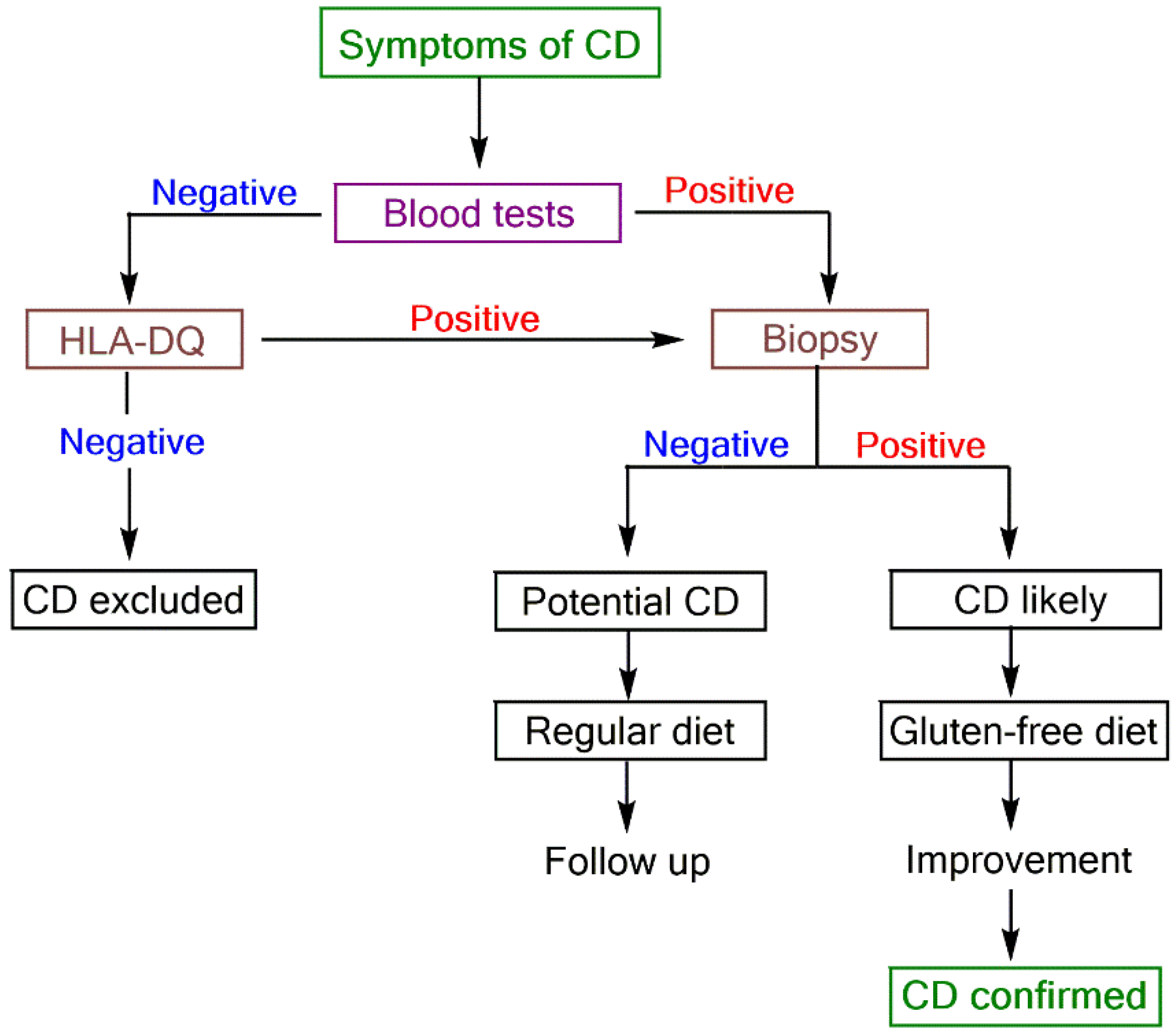
1. Introduction
2. Biomarkers of CD
3. Biosensors for CD Detection
3.1. Detection of Anti-Gliadin Antibody (AGA)
3.2. Detection of Antibody against Deamidated Gliadin Peptides (DGPA)
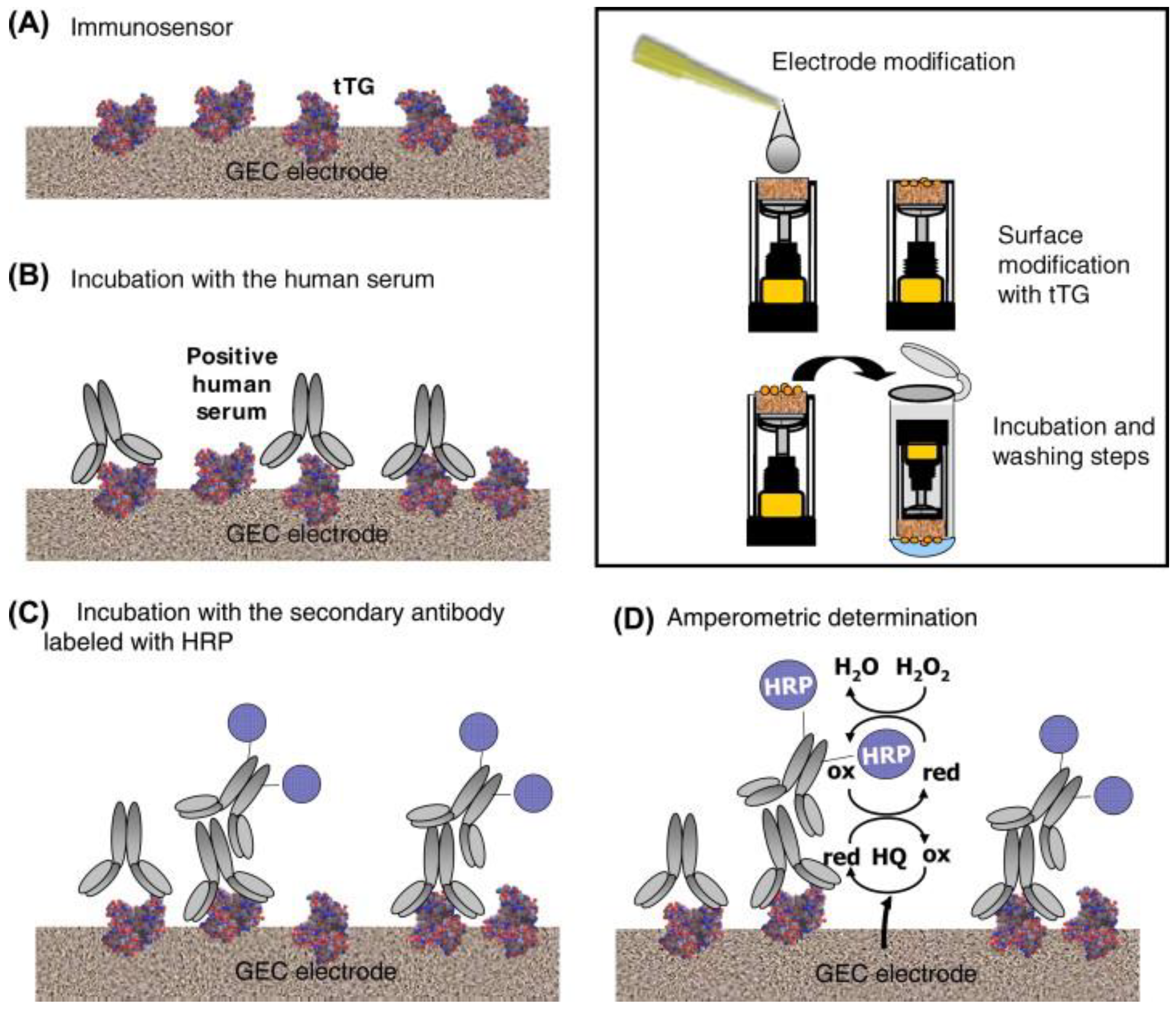
3.3. Detection of Anti-Transglutaminase Antibody (TGA)
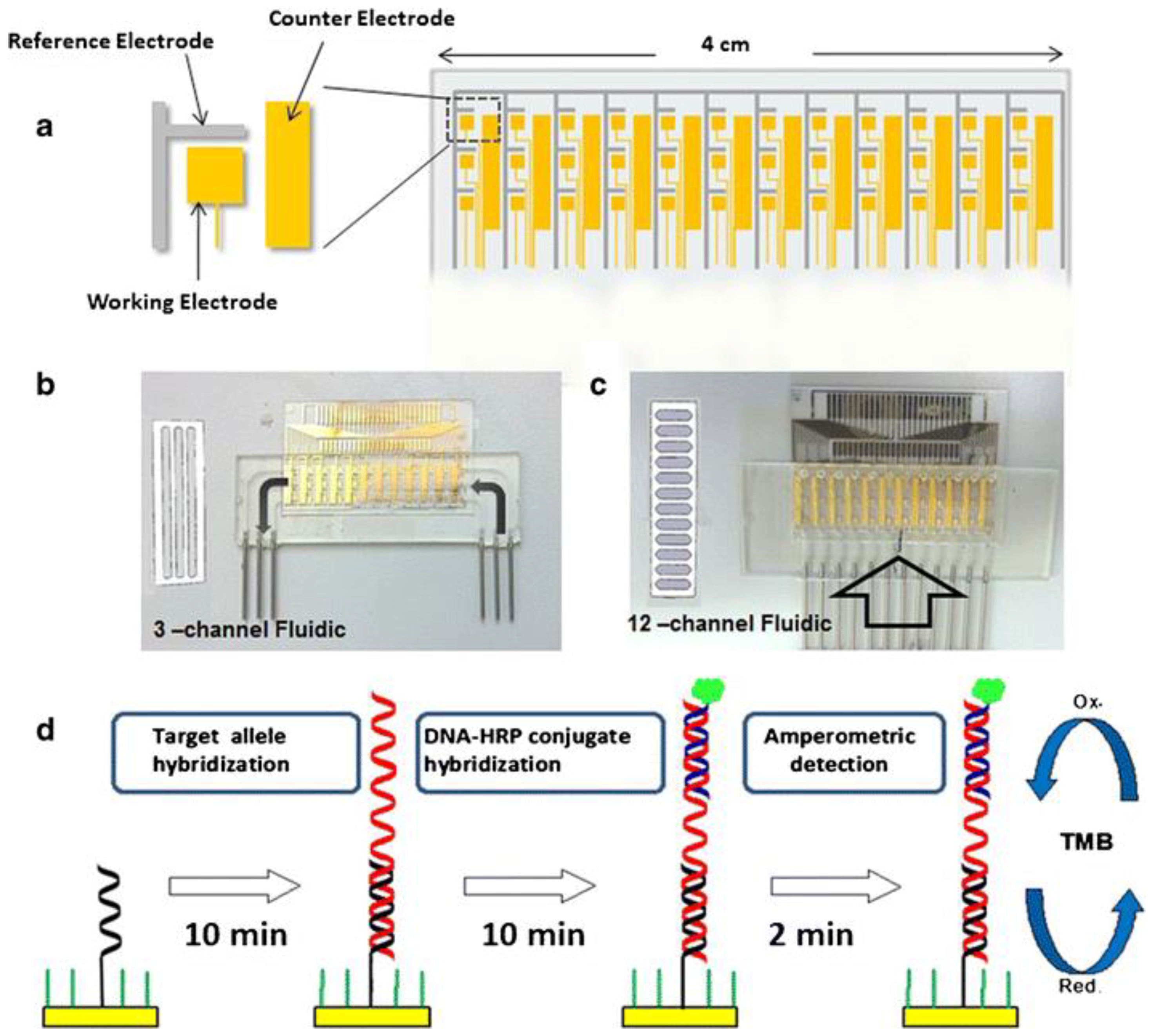
3.4. Detection of HLA-DQ2/DQ8 Alleles
4. Conclusions and Outlook
Acknowledgments
Conflicts of Interest
References
- Green, P.H.R.; Cellier, C. Celiac disease. N. Engl. J. Med. 2007, 357, 1731–1743. [Google Scholar] [CrossRef] [PubMed]
- Ludvigsson, J.F.; Rubio-Tapia, A.; van Dyke, C.T.; Melton III, L.J.; Zinsmeister, A.R.; Lahr, B.D.; Murray, J.A. Increasing incidence of celiac disease in a North American Population. Am. J. Gastroenterol. 2013, 108, 818–824. [Google Scholar] [CrossRef] [PubMed]
- Mustalahti, K.; Catassi, C.; Reunanen, A.; Fabiani, E.; Heier, M.; McMillan, S.; Murray, L.; Metzger, M.H.; Gasparin, M.; Bravi, E.; et al. The prevalence of celiac disease in Europe: Results of a centralized, international mass screening project. Ann. Med. 2010, 42, 587–595. [Google Scholar] [CrossRef] [PubMed]
- García Nieto, V.M. A History of Celiac Disease. In Celiac Disease and Non-Celiac Gluten Sensitvity; Rodrigo, L., Peña, A.S., Eds.; OmniaScience: Barcelona, Spain, 2014; pp. 45–59. ISBN 978-84-942118-2-9. [Google Scholar]
- Koehler, P.; Wieser, H.; Konitzer, K. Celiac Disease—A Complex Disorder. In Celiac Disease and Gluten: Multidisciplinary Challenges and Opportunities; Koehler, P., Weiser, H., Konitzer, K., Eds.; Academic Press, Elsevier Inc.: Waltham, MA, USA, 2014; pp. 1–96. ISBN 978-0-12-420220-7. [Google Scholar]
- Parra-Blanco, A.; Agüero, C.; Cimmino, D.; González, N.; Ibáñez, P.; Pedreira, S. The Role of Endoscopy in Celiac Disease and its Complications: Advances in Imaging Techniques and Computerization. In Celiac Disease and Non-Celiac Gluten Sensitivity; Rodrigo, L., Peña, A.S., Eds.; OmniaScience: Barcelona, Spain, 2014; pp. 171–202. ISBN 978-84-942118-2-9. [Google Scholar]
- Fernandez-Jimenez, N.; Plaza-Izurieta, L.; Bilbao, J.R. Genetic Markers in Celiac Disease. In Celiac Disease and Non-Celiac Gluten Sensitivity; Rodrigo, L., Peña, A.S., Eds.; OmniaScience: Barcelona, Spain, 2014; pp. 103–121. ISBN 978-84-942118-2-9. [Google Scholar]
- Farré, C. The Role of Serology in Celiac Disease Screening, Diagnosis and Follow-Up. In Celiac Disease and Non-Celiac Gluten Sensitivity; Rodrigo, L., Peña, A.S., Eds.; OmniaScience: Barcelona, Spain, 2014; pp. 151–169. ISBN 978-84-942118-2-9. [Google Scholar]
- Buergin-Wolff, A.; Hernandez, R.; Just, M.; Stoner, E. Immunofluorescent antibodies against gliadin: A screening test for coeliac disease. Helv. Paediatr. Acta 1976, 31, 375–380. [Google Scholar]
- Butler, J. Enzyme-Linked Immunosorbent Assay. J. Immunoass. Immunochem. 2000, 21, 165–209. [Google Scholar] [CrossRef] [PubMed]
- O´Farrelly, C.; Kelly, J.; Hekkens, W.; Bradley, B.; Thompson, A.; Feighery, C.; Weir, D.G. Alpha gliadin antibody levels: A serological test for coeliac disease. Br. Med. J. 1983, 286, 2007–2010. [Google Scholar] [CrossRef]
- Scherf, K.A.; Ciccocioppo, R.; Pohanka, M.; Rimarova, K.; Opatrilova, R.; Rodrigo, L.; Kruzliak, P. Biosensors for the Diagnosis of Celiac Disease: Current Status and Future Perspectives. Mol. Biotechnol. 2016, 58, 381–392. [Google Scholar] [CrossRef] [PubMed]
- Grieshaber, D.; MacKenzie, R.; Vörös, J.; Reimhult, E. Electrochemical Biosensors—Sensor Principles and Architectures. Sensors 2008, 8, 1400–1458. [Google Scholar] [CrossRef] [PubMed]
- Balkenhohl, T.; Lisdat, F. Screen-printed electrodes as impedimetric immunosensors for the detection of anti-transglutaminase antibodies in human sera. Anal. Chim. Acta 2007, 597, 50–57. [Google Scholar] [CrossRef] [PubMed]
- Vives-Pi, M.; Takasawa, S.; Pujol-Autonell, I.; Planas, R.; Cabre, E.; Ojanguren, I.; Montraveta, M.; Santos, A.L.; Ruiz-Ortiz, E. Biomarkers for Diagnosis and Monitoring of Celiac Disease. Clin. Gastroenterol. 2013, 47, 308–313. [Google Scholar] [CrossRef] [PubMed]
- Leffler, D.A.; Schuppan, D. Update on serologic testing in celiac disease. Am. J. Gastroenterol. 2010, 105, 2520–2524. [Google Scholar] [CrossRef] [PubMed]
- Hill, I.D. What are the sensitivity and specificity of serologic tests for celiac disease? Do sensitivity and specificity vary in different populations? Gastroenterol. 2005, 128, S25–S32. [Google Scholar] [CrossRef]
- Planas, R.; Pujol-Autonell, I.; Ruiz, E.; Montraveta, M.; Cabre, E.; Lucas-Martin, A.; Pujol-Borrell, R.; Martinez-Caceres, E.; Vives-Pi, M. Regenerating gene Iα is a biomarker for diagnostic and monitoring of celiac disease: A preliminary study. Transl. Res. 2011, 158, 140–145. [Google Scholar] [CrossRef] [PubMed]
- Jabri, B.; Sollid, L.M. Tissue-mediated control of immunopathology in coeliac disease. Nat. Rev. Immunol. 2009, 9, 858–870. [Google Scholar] [CrossRef] [PubMed]
- Lebwohl, B.; Rubio-Tapia, A.; Assiri, A.; Newland, C.; Guandalini, S. Diagnosis of celiac disease. Gastrointest. Endosc. Clin. N. Am. 2012, 22, 661–677. [Google Scholar] [CrossRef] [PubMed]
- Pasinszki, T.; Krebsz, M.; Tung, T.T.; Losic, D. Carbon Nanomaterial Based Biosensors for Non-Invasive Detection of Cancer and Disease Biomarkers for Clinical Diagnosis. Sensors 2017, 17, 1919. [Google Scholar] [CrossRef] [PubMed]
- Rosales-Rivera, L.C.; Acero-Sánchez, J.L.; Lozano-Sánchez, P.; Katakis, I.; O’Sullivan, C.K. Electrochemical immunosensor detection of antigliadin antibodies from real human serum. Biosens. Bioelectron. 2011, 26, 4471–4476. [Google Scholar] [CrossRef] [PubMed]
- Ortiz, M.; Fragoso, A.; O’Sullivan, C.K. Detection of Antigliadin Autoantibodies in Celiac Patient Samples Using a Cyclodextrin-Based Supramolecular Biosensor. Anal. Chem. 2011, 83, 2931–2938. [Google Scholar] [CrossRef] [PubMed]
- Wajs, E.; Fernández, N.; Fragoso, A. Supramolecular biosensors based on electropolymerised pyrrole–cyclodextrin modified surfaces for antibody detection. Analyst 2016, 141, 3274–3279. [Google Scholar] [CrossRef] [PubMed]
- Corres, J.M.; Matias, I.R.; Goicoechea, J.; Arregui, F.J.; Viegas, D.; Araújo, F.M.; Santos, J.L. Experimental Results of Antigliadin Antibodies Detection using Long Period Fiber Grating. In Proceedings of the 19th International Conference on Optical Fibre Sensors, Perth, WA, Australia, 14–18 April 2008. [Google Scholar]
- Corres, J.M.; Matias, I.R.; Bravo, J.; Arregui, F.J. Tapered optical fiber biosensor for the detection of anti-gliadin antibodies. Sens. Actuators B Chem. 2008, 135, 166–171. [Google Scholar] [CrossRef]
- Tung, T.T.; Nine, M.J.; Krebsz, M.; Pasinszki, T.; Coghlan, C.J.; Tran, D.N.H.; Losic, D. Recent Advances in Sensing Applications of Graphene Assemblies and Their Composites. Adv. Funct. Mater. 2017, 27, 1702891. [Google Scholar] [CrossRef]
- Socorro, A.B.; Del Villar, I.; Corres, J.M.; Arregui, F.J.; Matias, I.R. Spectral width reduction in lossy mode resonance-based sensors by means of tapered optical fibre structures. Sens. Actuators B Chem. 2014, 200, 53–60. [Google Scholar] [CrossRef]
- Neves, M.M.P.S.; González-García, M.G.; Nouws, H.P.A.; Costa-García, A. An electrochemical deamidated gliadin antibody immunosensor for celiac disease clinical diagnosis. Analyst 2013, 138, 1956–1958. [Google Scholar] [CrossRef] [PubMed]
- Pividori, M.I.; Lermo, A.; Bonanni, A.; Alegret, S.; del Valle, M. Electrochemical immunosensor for the diagnosis of celiac disease. Anal. Biochem. 2009, 388, 229–234. [Google Scholar] [CrossRef] [PubMed]
- Dulay, S.; Lozano-Sánchez, P.; Iwuoha, E.; Katakis, I.; O’Sullivan, C.K. Electrochemical detection of celiac disease-related anti-tissue transglutaminase antibodies using thiol based surface chemistry. Biosens. Bioelectron. 2011, 26, 3852–3856. [Google Scholar] [CrossRef] [PubMed]
- Neves, M.M.P.S.; González-García, M.B.; Nouws, H.P.A.; Costa-García, A. Celiac disease detection using a transglutaminase electrochemical immunosensor fabricated on nanohybrid screen-printed carbon electrodes. Biosens. Bioelectron. 2012, 31, 95–100. [Google Scholar] [CrossRef] [PubMed]
- Kergaravat, S.V.; Beltramino, L.; Garnero, N.; Trotta, L.; Wagener, M.; Pividori, M.I.; Hernandez, S.R. Electrochemical magneto immunosensor for the detection of anti-TG2 antibody in celiac disease. Biosens. Bioelectron. 2013, 48, 203–209. [Google Scholar] [CrossRef] [PubMed]
- Martín-Yerga, D.; González-García, M.B.; Costa-García, A. Electrochemical immunosensor for anti-tissue transglutaminase antibodies based on the in situ detection of quantum dots. Talanta 2014, 130, 598–602. [Google Scholar] [CrossRef] [PubMed]
- Singh, K.V.; Bhura, D.K.; Nandamuri, G.; Whited, A.M.; Evans, D.; King, J.; Solanki, R. Nanoparticle-Enhanced Sensitivity of a Nanogap-Interdigitated Electrode Array Impedimetric Biosensor. Langmuir 2011, 27, 13931–13939. [Google Scholar] [CrossRef] [PubMed]
- West, N.; Baker, P.; Waryo, T.; Ngece, F.R.; Iwuoha, E.I.; O’Sullivan, C.; Katakis, I. Highly sensitive gold-overoxidized polypyrrole nanocomposite immunosensor for antitransglutaminase antibody. J. Bioact. Compat. Polym. 2013, 28, 167–177. [Google Scholar] [CrossRef]
- Wilson, L.; vam Wyk, J.; Rassie, C.; Ross, N.; Sunday, C.; Makelane, H.R.; Bilibana, M.; Waryo, T.; Mapolie, S.; Baker, P.G.; et al. Electrochemical Immunosensor Based on the Interactions Between Polypyrrole and Cobalt (II) Salicylaldiimine Dendrimer. Int. J. Electrochem. Sci. 2015, 10, 3207–3222. [Google Scholar]
- Martín-Yerga, D.; Costa-García, A. Towards a blocking-free electrochemical immunosensing strategy for anti-transglutaminase antibodies using screen-printed electrodes. Bioelectrochem. 2015, 105, 88–94. [Google Scholar] [CrossRef] [PubMed]
- Martín-Yerga, D.; Fanjul-Bolado, P.; Hernández-Santos, D.; Costa-García, A. Enhanced detection of quantum dots by the magnetohydrodynamic effect for electrochemical biosensing. Analyst 2017, 142, 1591–1600. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Kaushal, A.; Kumar, A.; Kumar, D. Ultrasensitive transglutaminase based nanosensor for early detection of celiac disease in human. Int. J. Biol. Macromol. 2017, 105, 905–911. [Google Scholar] [CrossRef] [PubMed]
- Rosales-Rivera, L.C.; Dulay, S.; Lozano-Sánchez, P.; Katakis, I.; Acero-Sánchez, J.L.; O’Sullivan, C.K. Disulfide-modified antigen for detection of celiac disease-associated anti-tissue transglutaminase autoantibodies. Anal. Bioanal. Chem. 2017, 409, 3799–3806. [Google Scholar] [CrossRef] [PubMed]
- Habtamu, H.B.; Sentic, M.; Silvestrini, M.; De Leo, L.; Not, T.; Arbault, S.; Manojlovic, D.; Sojic, N.; Ugo, P. A Sensitive Electrochemiluminescence Immunosensor for Celiac Disease Diagnosis Based on Nanoelectrode Ensembles. Anal. Chem. 2015, 87, 12080–12087. [Google Scholar] [CrossRef] [PubMed]
- Cennamo, N.; Varriale, A.; Pennacchio, A.; Staiano, M.; Massarotti, D.; Zeni, L.; D’Auria, S. An innovative plastic optical fiber-based biosensor for new bio/applications. The case of celiac disease. Sens. Actuators B Chem. 2013, 176, 1008–1014. [Google Scholar] [CrossRef]
- Manfredi, A.; Mattarozzi, M.; Giannetto, M.; Careri, M. Piezoelectric immunosensor based on antibody recognition of immobilized open-tissue transglutaminase: An innovative perspective on diagnostic devices for celiac disease. Sens. Actuators B Chem. 2014, 201, 300–307. [Google Scholar] [CrossRef]
- Joda, H.; Beni, V.; Alakulppi, N.; Partanen, J.; Lind, K.; Strömbom, L.; Latta, D.; Höth, J.; Katakis, I.; O’Sullivan, C.K. Medium-high resolution electrochemical genotyping of HLA-DQ2/DQ8 for detection of predisposition to coeliac disease. Anal. Bioanal. Chem. 2014, 406, 2757–2769. [Google Scholar] [CrossRef] [PubMed]
- Joda, H.; Beni, V.; Willems, A.; Frank, R.; Höth, J.; Lind, K.; Strömbom, L.; Katakis, I.; O´Sullivan, C.K. Modified primers for rapid and direct electrochemical analysis of coeliac disease associated HLA alleles. Biosens. Bioelectron. 2015, 73, 64–70. [Google Scholar] [CrossRef] [PubMed]
- Beni, V.; Zewdu, T.; Joda, H.; Katakis, I.; O’Sullivan, C.K. Gold nanoparticle fluorescent molecular beacon for low-resolution DQ2 gene HLA typing. Anal. Bioanal. Chem. 2012, 402, 1001–1009. [Google Scholar] [CrossRef] [PubMed]
| Sensor Platform//Label | Analyte | Instrumental Technique 1 | Linearity Range | LOD | Ref. |
|---|---|---|---|---|---|
| Au/DT2/anti-gliadin//anti-IgG-HRP | AGA | AMP | 0–1000 ng/mL | 46 ng/mL | [22] |
| Au/CDPSH/ADA-CMC-GLI//anti-IgG-HRP | AGA | AMP | 0–750 ng/mL | 20 ng/mL | [23] |
| Au/PPy/CD/ADA-CMC-GLI//anti-IgG-HRP | AGA | AMP | 0–10 µg/mL | 33 ng/mL | [24] |
| Au/PPy/GLI//anti-IgG-HRP | AGA | AMP | 0–10 µg/mL | 135 ng/mL | [24] |
| Au/CD/ADA-CMC-GLI//anti-IgG-HRP | AGA | AMP | 0–10 µg/mL | 240 ng/mL | [24] |
| Au/C3SAM/GLI//anti-IgG-HRP | AGA | AMP | 0–10 µg/mL | 250 ng/mL | [24] |
| LPFG/(PAH/SM-30)14/gliadin | AGA | EWS | n.a. | 5 ppm | [25] |
| OF/(gliadin/PSS)n | AGA | EWS | n.a. | 1 ppm | [26] |
| OF/(PAH/PAA)(PAH/PSS)3(gliadin/PSS)2(gliadin) | AGA | LMR | n.a. | 5 ppm | [28] |
| SPCE/MWCNT/AuNP/DPGx4//anti-IgG-AP | DGPA | CV | n.a. | n.a. | [29] |
| SPGE/PSS/TG//anti-Ig-HRP | TGA | EIS | n.a. | n.a. | [14] |
| GEC/tTG//anti-Ig-HRP | TGA | AMP | n.a. | n.a. | [30] |
| Au/DT2/tTG//anti-IgG-HRP | TGA | AMP | 0–10,000 ng/mL | 390 ng/mL | [31] |
| SPCE/MWCNT/AuNP/tTG//anti-Ig-AP | TGA | CV | 0–40 U/mL | n.a. | [32] |
| SPE/MB-TG2//anti-IgA-HRP | TGA | SWV | n.a. | n.a. | [33] |
| SPCE/tTG//anti-IgG-QD | TGA | DPV | 0–40 U/mL | 2.2 U/mL | [34] |
| IDE/cysteamine/tTG//Pr-A-AuNP | TGA | EIS | 0.03–30 nM | n.a. | [35] |
| GCE/OPPy/AuNP/tTG | TGA | EIS | 1–100 ng/mL | 5.2 ng/mL | [36] |
| Pt/Ppy-Co(II)SDD/tTG | TGA | EIS | 0.2–1.8 µg/mL | 201 ng/mL | [37] |
| SPCE/tTG//anti-IgA-BT/QD-STV | TGA | DPV | 3–100 U/mL | 2.4 U/mL | [38] |
| SPCE/tTG//anti-IgA-BT/QD-STV | TGA | DPV | 3–40 U/mL | 1.0 U/mL | [39] |
| MWCNT-AuNP/GQD/PAMAM/tTG | TGA | DPV | n.a. | 20 fg/mL | [40] |
| Au/s-tTG//anti-Ig-HRP | TGA | AMP | 0.26–6.9 μg/mL | 260 ng/mL | [41] |
| Au/PC/tTG//anti-IgG-SA-Ru | TGA | ECL | 1.5–10,000 ng/mL | 0.5 ng/mL | [42] |
| POF/Au/tTG | TGA | SPR | 30–3000 nM | n.a. | [43] |
| Au/MUA/open-tTG//anti-IgG-AuNPs | TGA | QCM | 1.3–12 µg/mL | 1.3 μg/mL | [44] |
| Au/ssDNA//ssDNA-HRP | HLA | AMP | 1–50 nM | 231 pM | [45] |
| Au/ssDNA//ssDNA-HRP | HLA | AMP | n.a. | n.a. | [46] |
| AuNP/MB | HLA | FS | 0–10 nM | 0.5 nM | [47] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pasinszki, T.; Krebsz, M. Biosensors for Non-Invasive Detection of Celiac Disease Biomarkers in Body Fluids. Biosensors 2018, 8, 55. https://doi.org/10.3390/bios8020055
Pasinszki T, Krebsz M. Biosensors for Non-Invasive Detection of Celiac Disease Biomarkers in Body Fluids. Biosensors. 2018; 8(2):55. https://doi.org/10.3390/bios8020055
Chicago/Turabian StylePasinszki, Tibor, and Melinda Krebsz. 2018. "Biosensors for Non-Invasive Detection of Celiac Disease Biomarkers in Body Fluids" Biosensors 8, no. 2: 55. https://doi.org/10.3390/bios8020055
APA StylePasinszki, T., & Krebsz, M. (2018). Biosensors for Non-Invasive Detection of Celiac Disease Biomarkers in Body Fluids. Biosensors, 8(2), 55. https://doi.org/10.3390/bios8020055






