Amperometric Biosensor Based on Zirconium Oxide/Polyethylene Glycol/Tyrosinase Composite Film for the Detection of Phenolic Compounds
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Apparatus
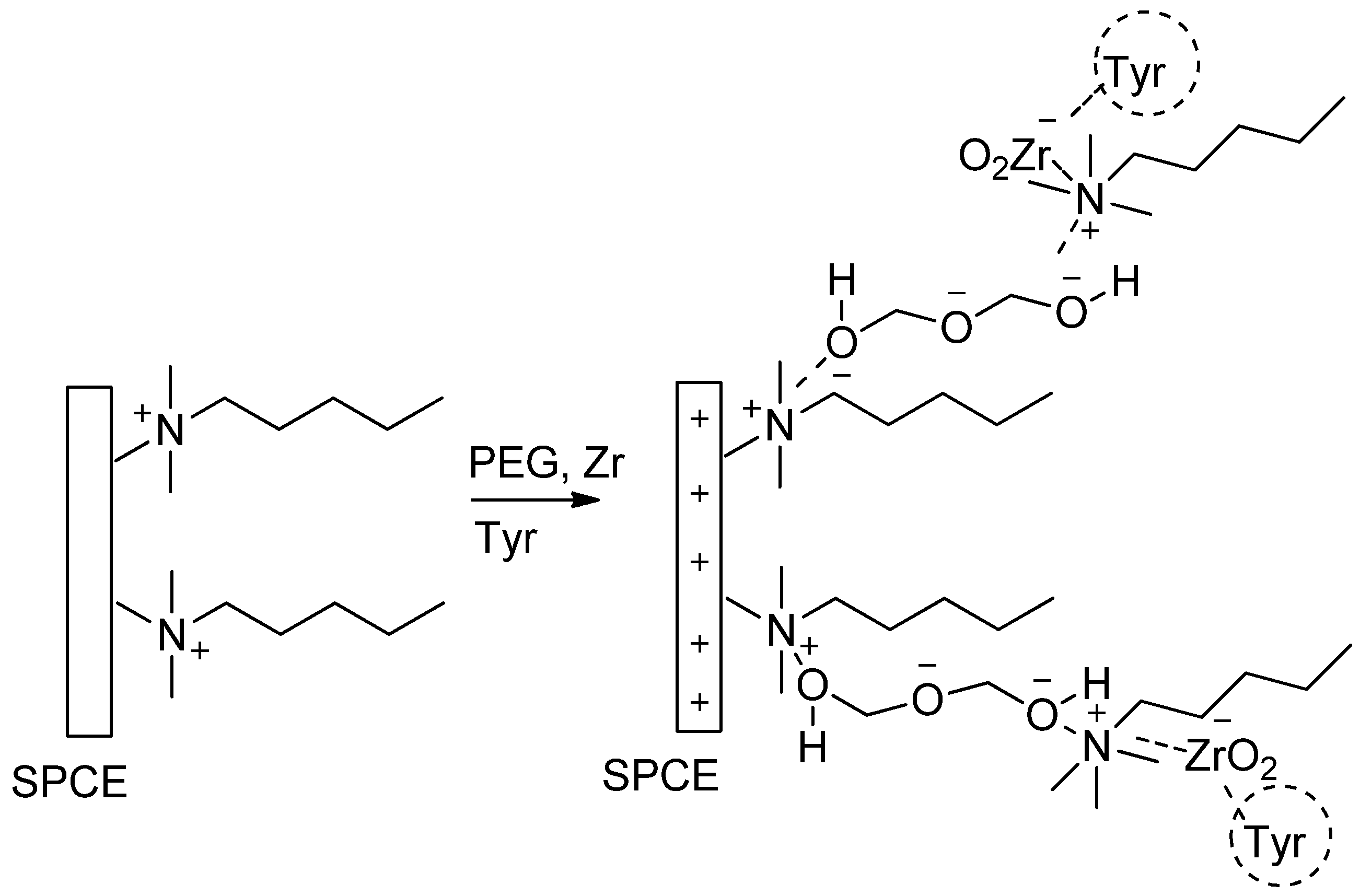
2.3. Preparation of Enzyme Electrode
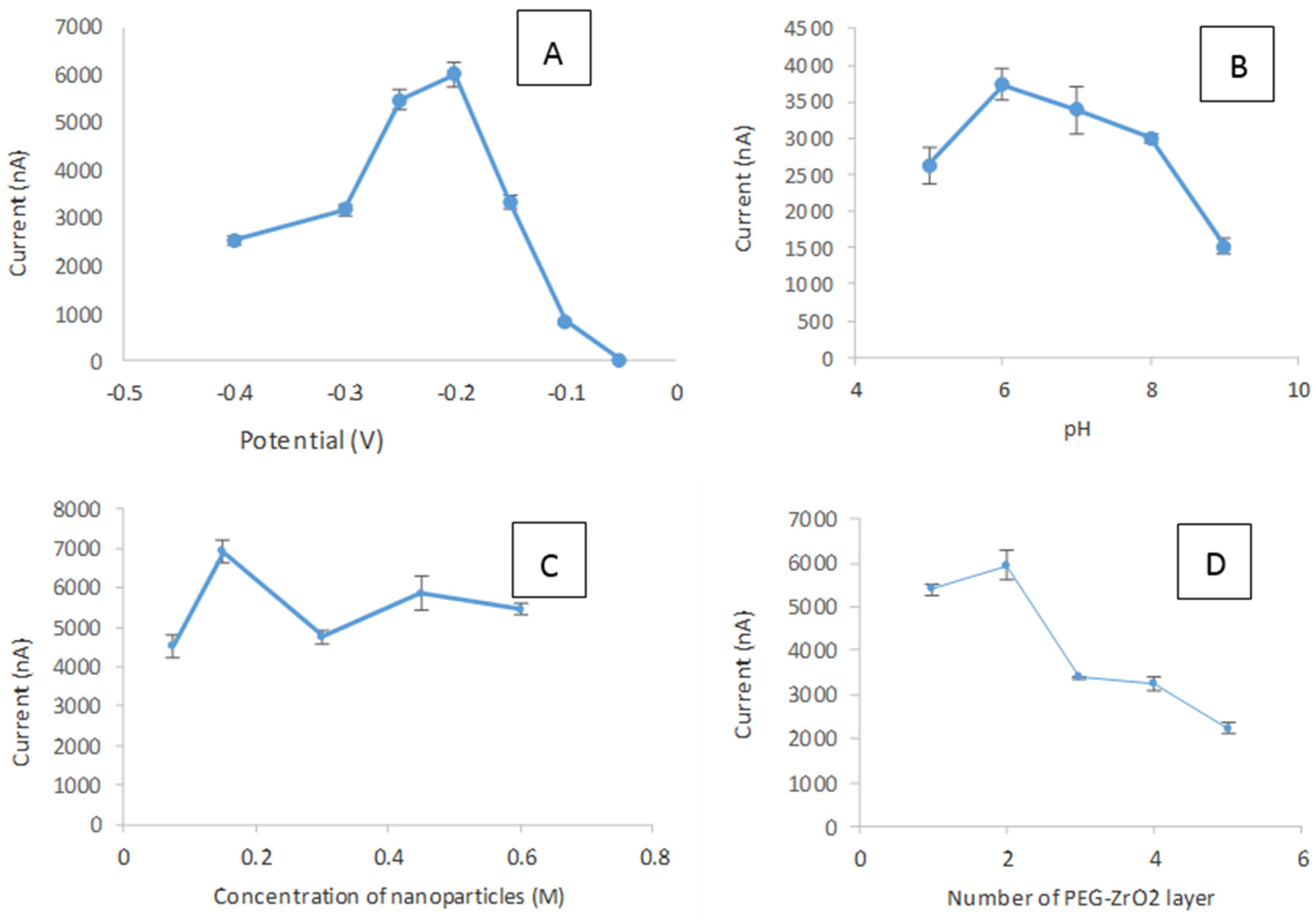
2.4. Optimization and Performance Studies of the Biosensor
3. Results and Discussion
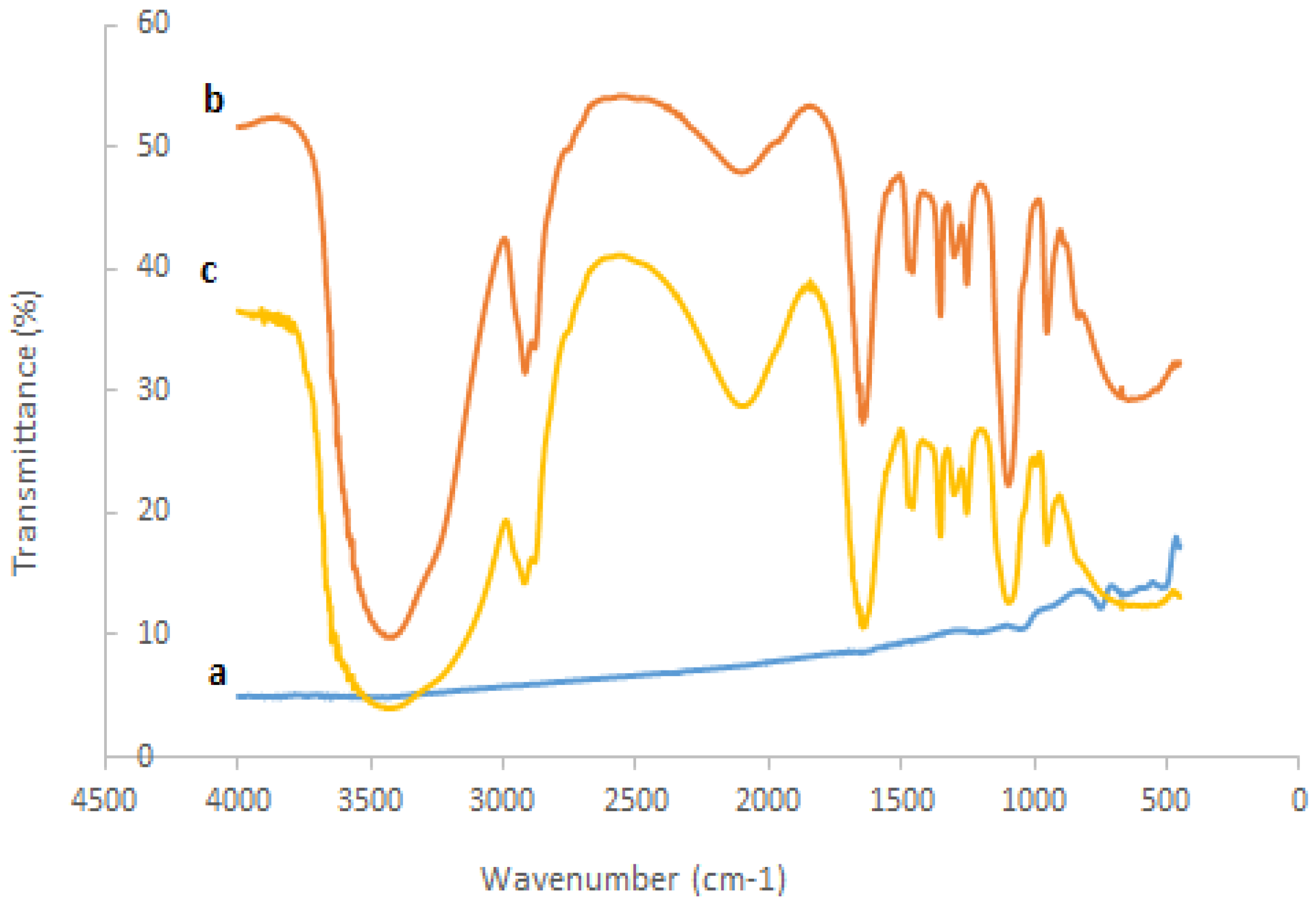
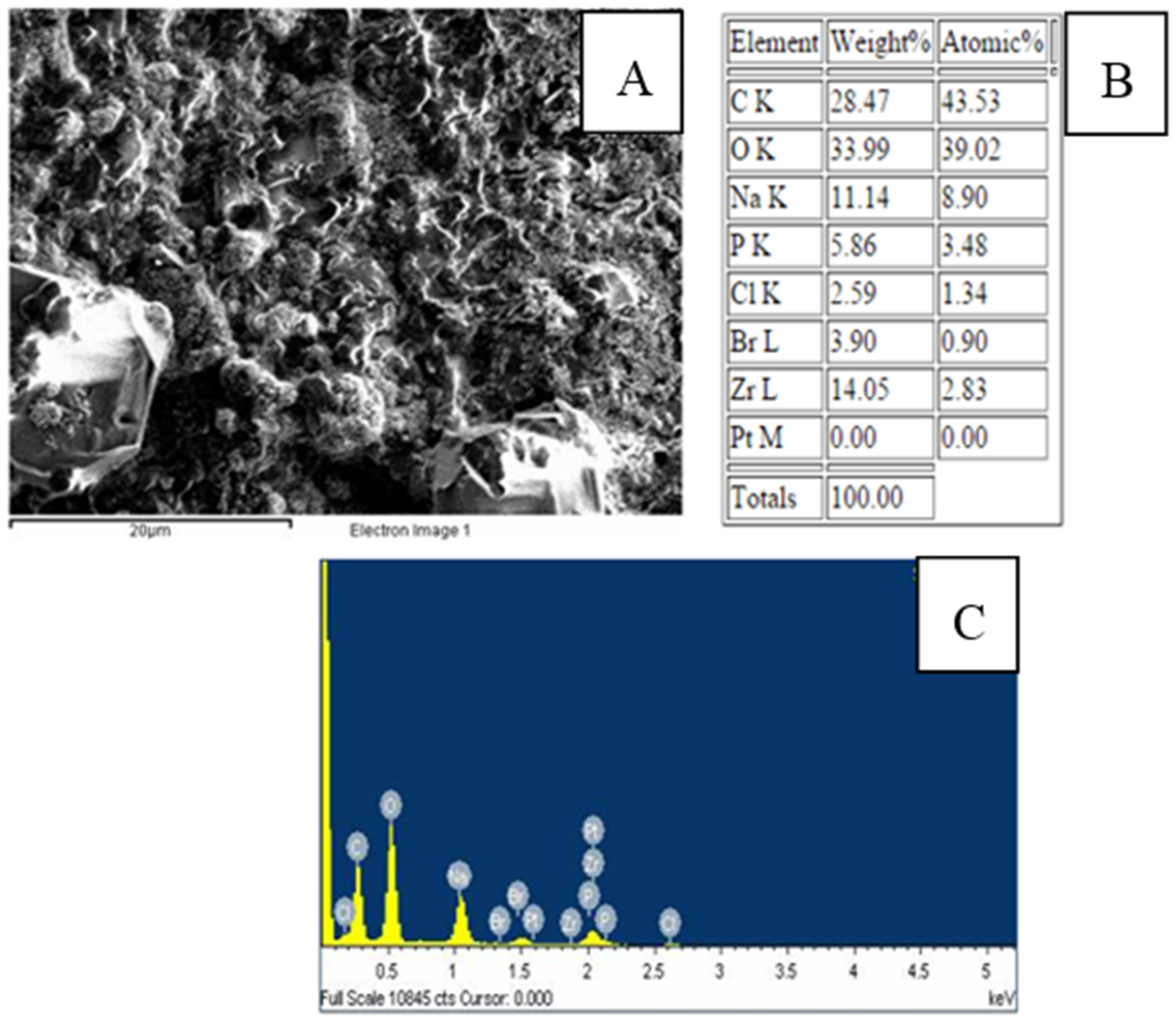
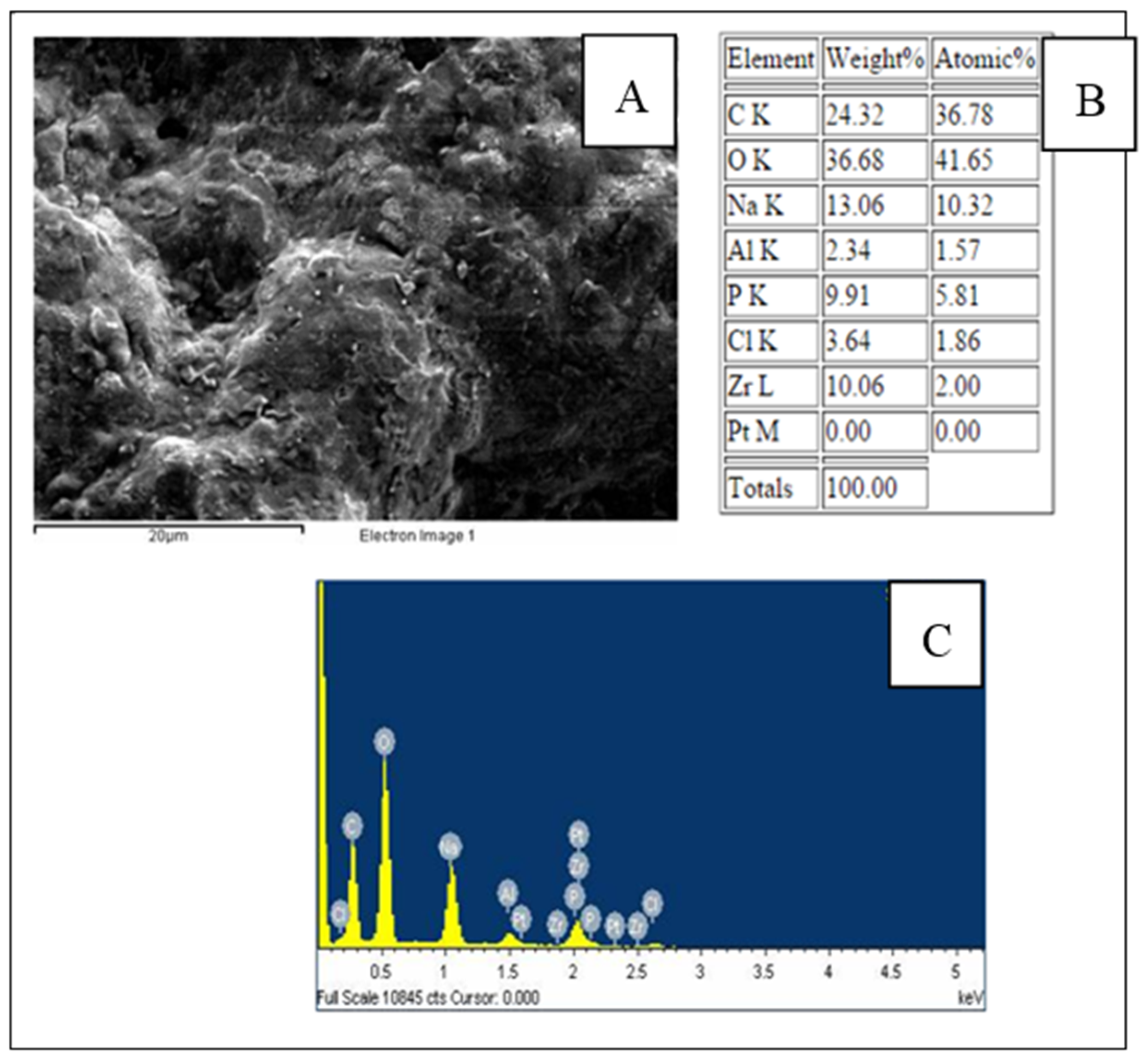
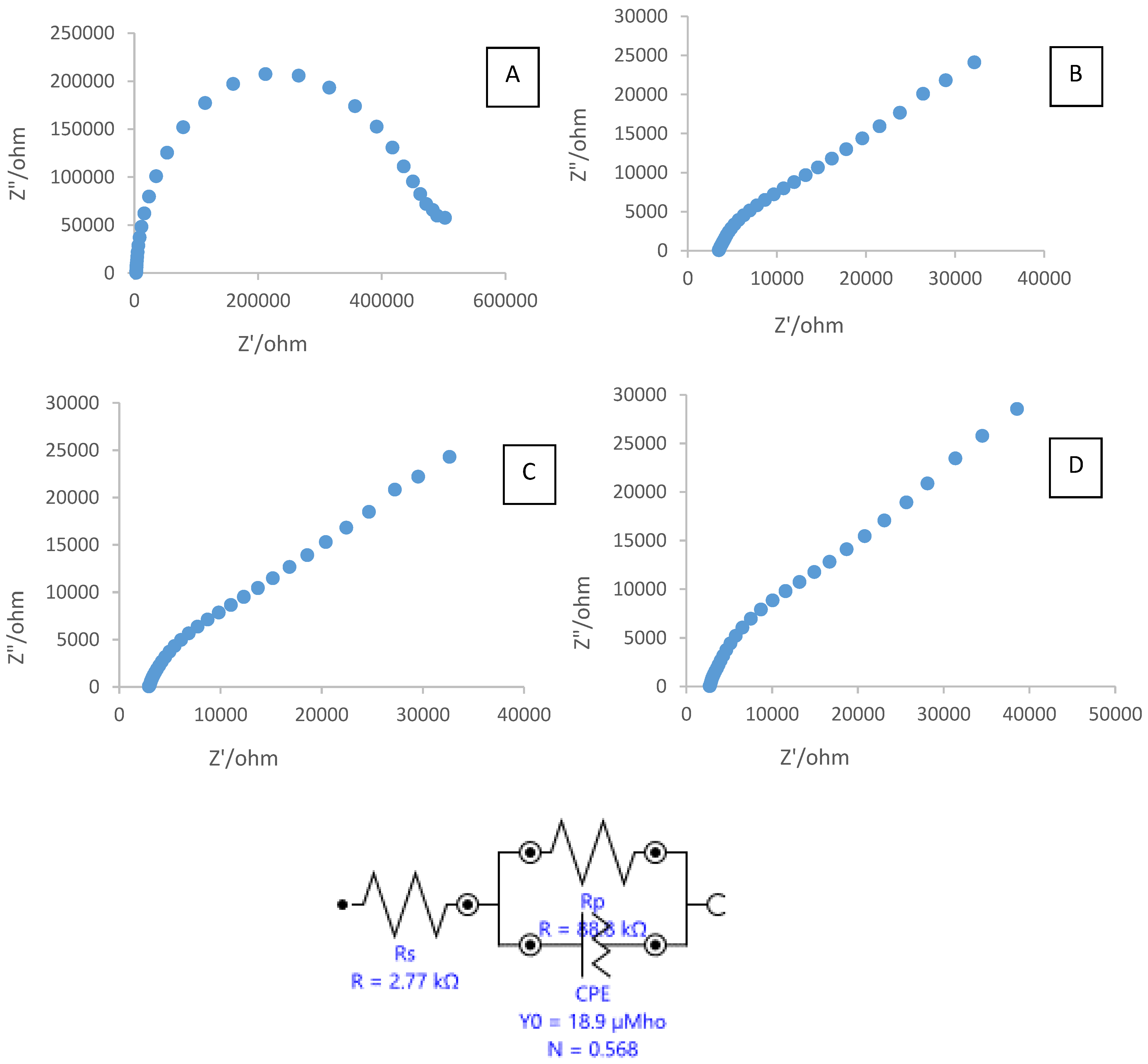
3.1. Characterization of the Modified SPCE
3.2. Electrochemical Studies
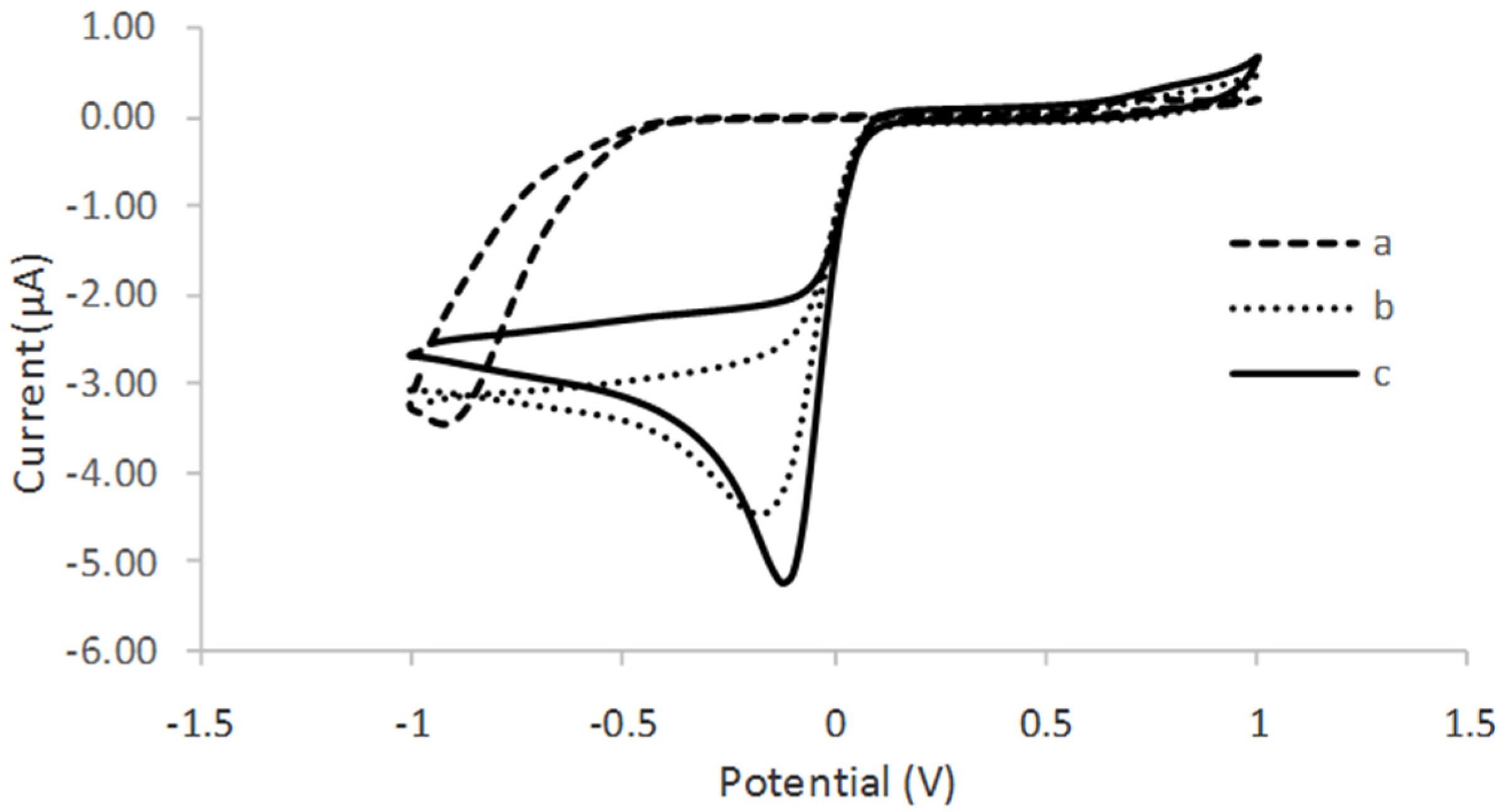
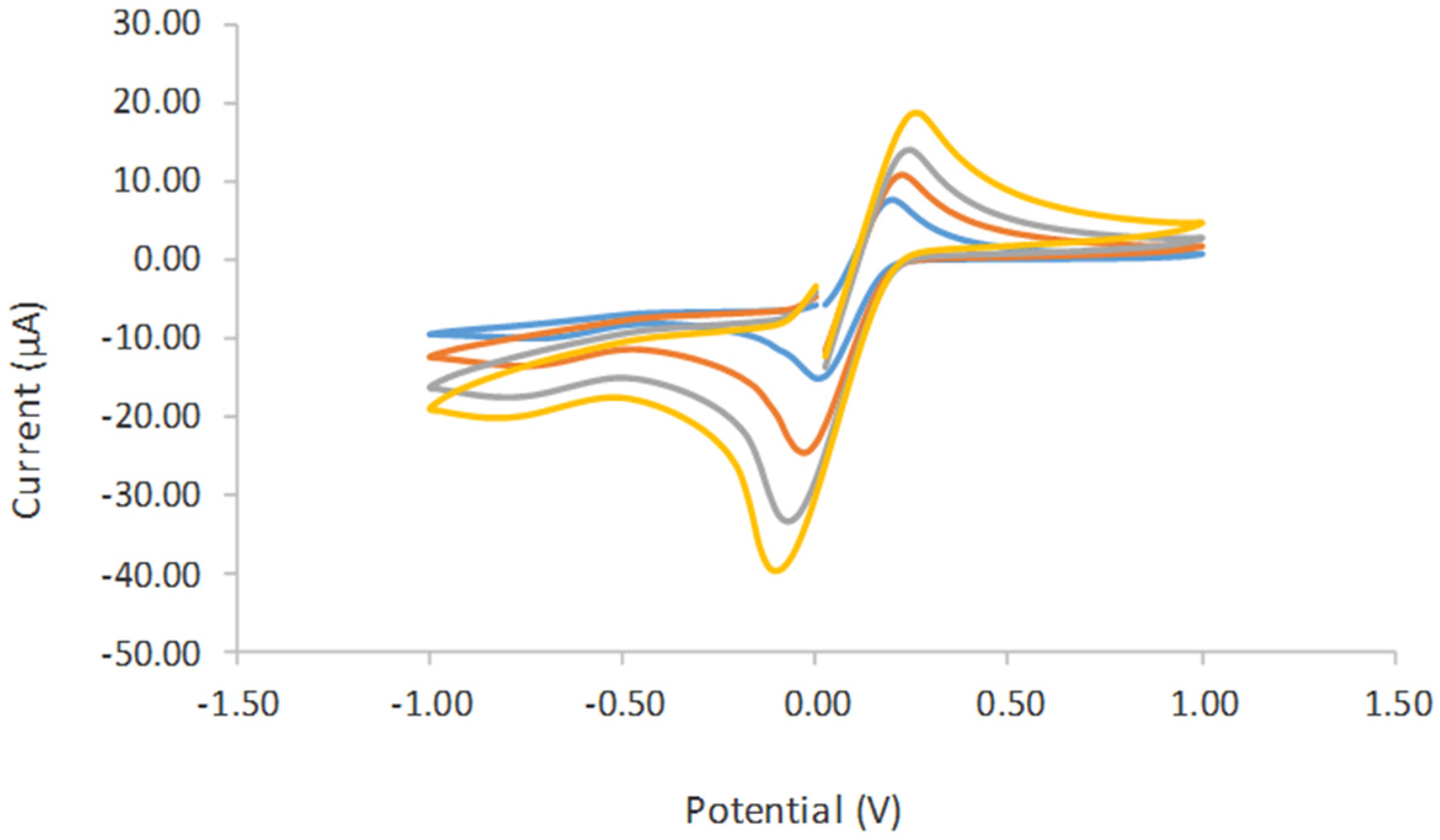
3.3. Cyclic Voltamogram
3.4. Interference Studies
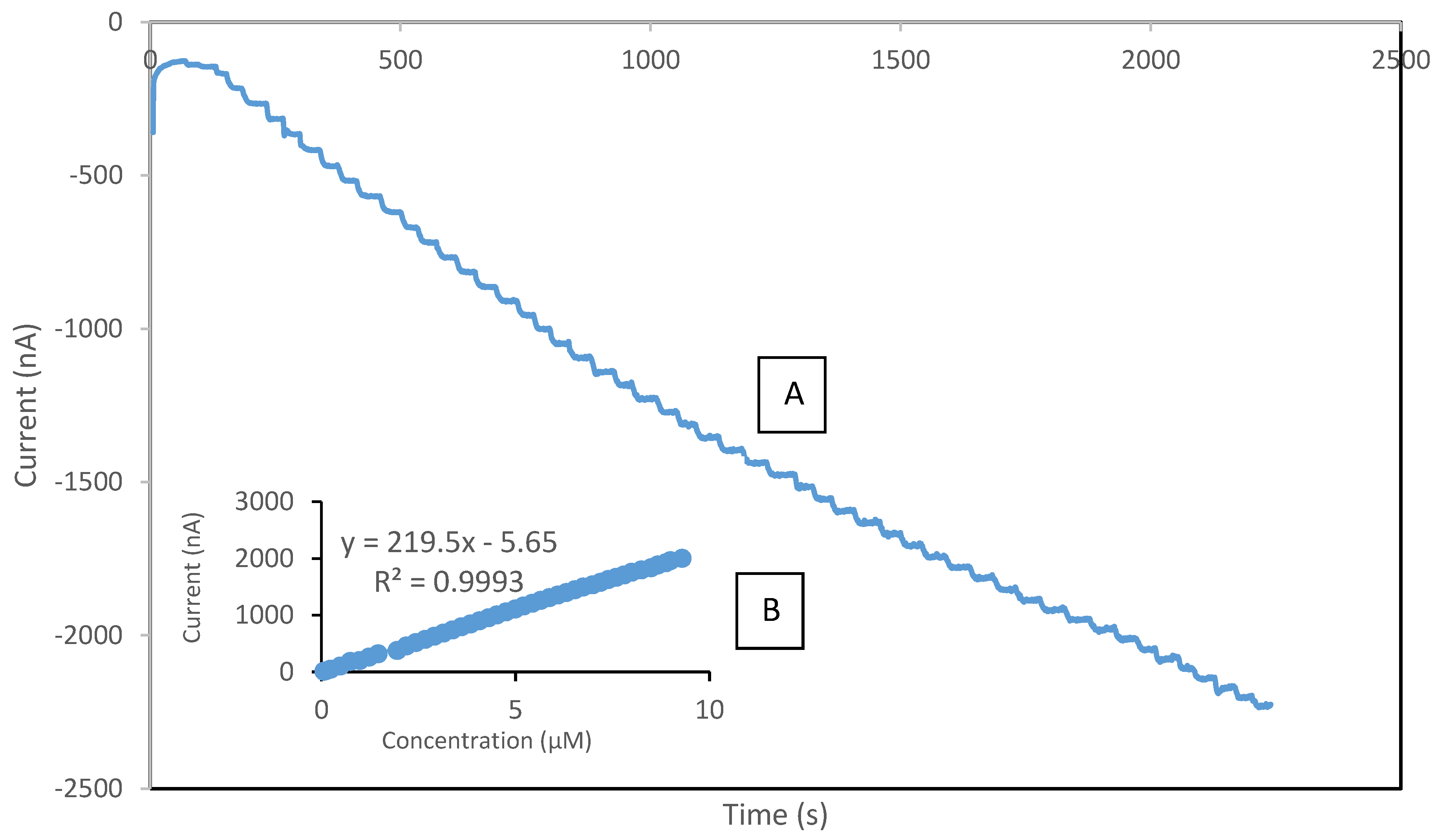
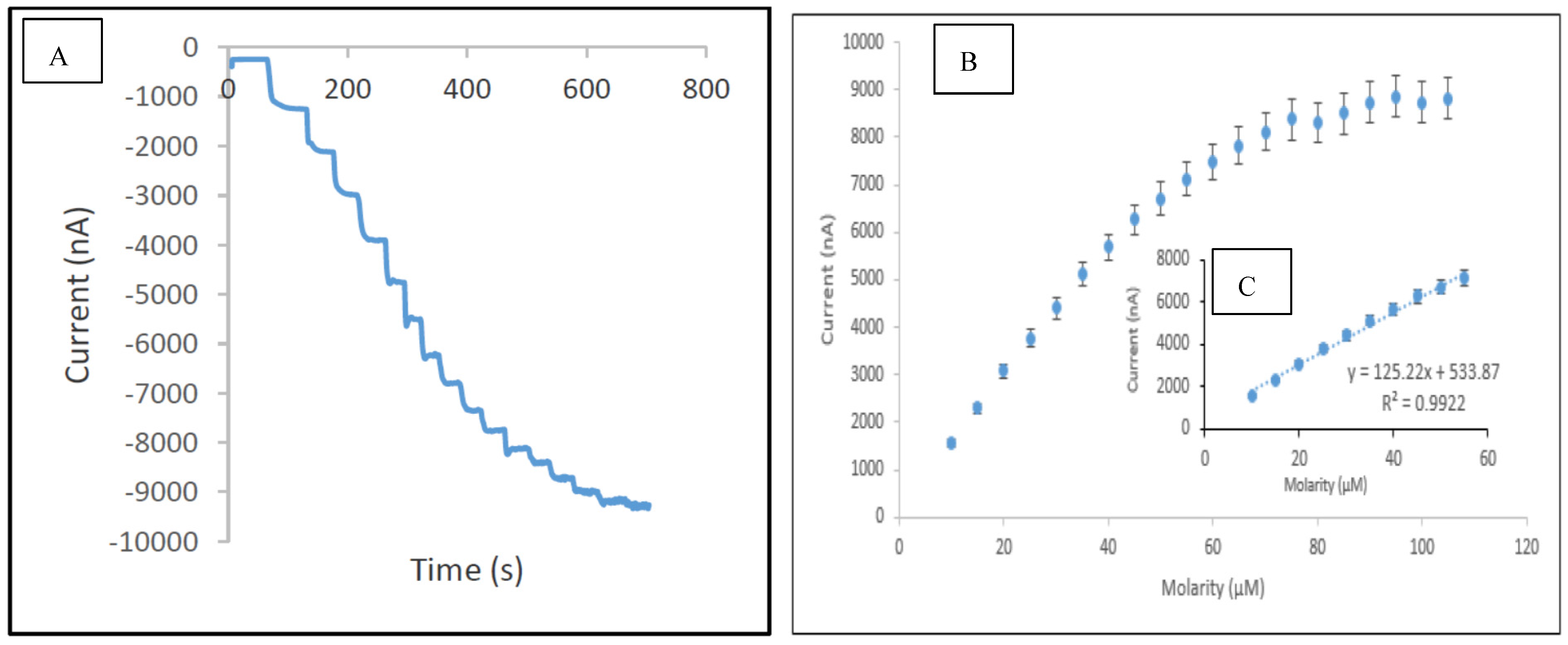
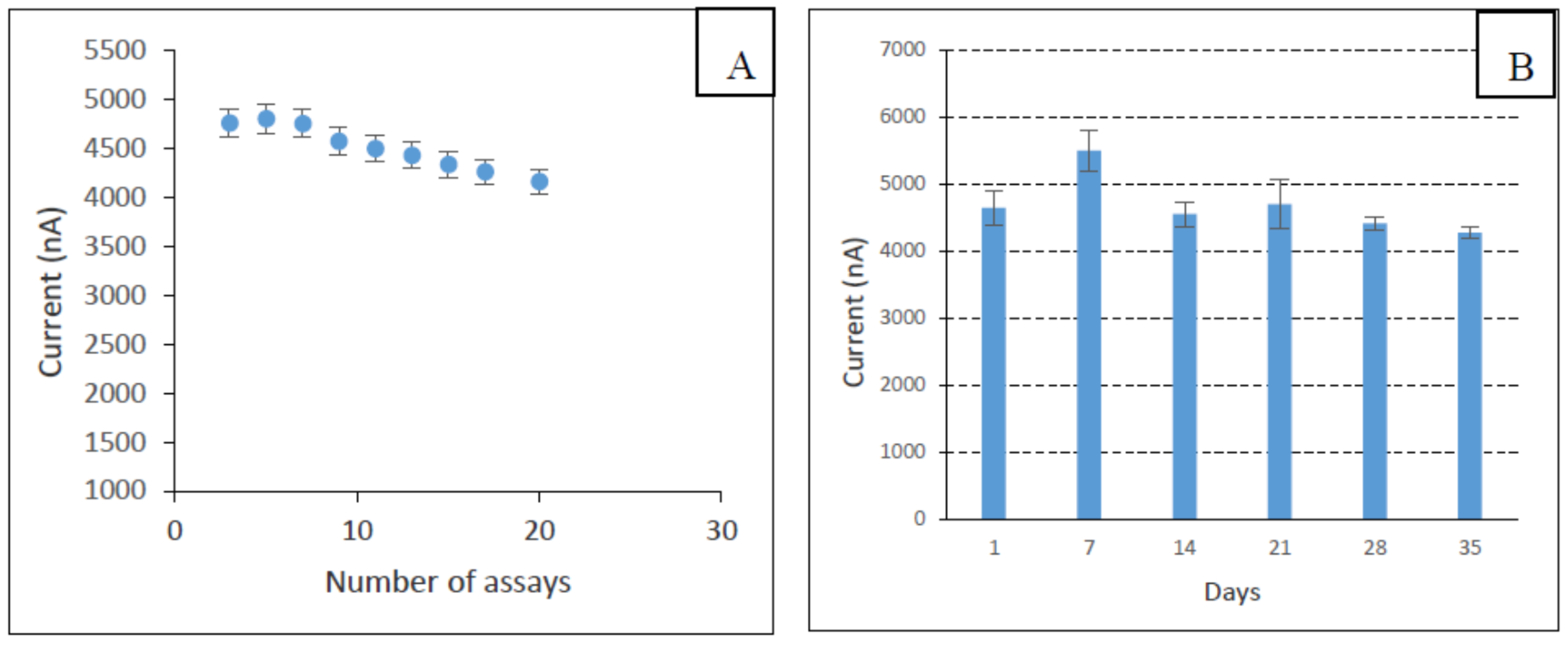
3.5. Analytical Performance of the Developed Biosensor
3.6. Selectivity Study
3.7. Analysis of Spiked Real Water Samples
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| MDPI | Multidisciplinary Digital Publishing Institute |
| DOAJ | Directory of open access journals |
| TLA | Three letter acronym |
| LD | linear dichroism |
References
- Michalowicz, J.; Duda, W. Phenols-sources and toxicity. Pol. J. Environ. Stud. 2007, 16, 347–362. [Google Scholar]
- Lavilla, I.; Gil, S.; Costas, M.; Bendicho, C. Dispersive liquid-liquid microextraction combined with microvolume spectrophotometry to turn green the 5530 APHA standard method for determining phenols in water and wastewater. Talanta 2012, 98, 197–202. [Google Scholar] [CrossRef] [PubMed]
- Santana, C.M.; Ferrera, Z.S.; Padron, M.E.T.; Rodrigiez, J.J.S. Methodologies for the extraction of phenolic compounds from environmental samples: New Approaches. Molecules 2009, 14, 298–320. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Z.; Tian, L.; Li, Z.; Jia, L.; Zhang, X.; Xia, M.; Hu, Y. Whole-cell method for phenol detection based on the color reaction of phenol with 4-aminoantipyrine catalyzed by CotA laccase on endospore surfaces. Biosens. Bioelectron. 2015, 69, 162–166. [Google Scholar] [CrossRef] [PubMed]
- Asan, A.; Isildak, I. Determination of major phenolic compounds in water by reversed-phase liquid chromatography after pre-column derivatization with benzoyl chloride. J. Chromatogr. A 2003, 988, 145–149. [Google Scholar] [CrossRef]
- Nassiri, M.; Zahedi, M.M.; Pourmortazavi, S.M.; Yousefzade, M. Optimization of dispersive liquid-liquid microextraction for preconcentration and spectrophotometric determination of phenols in Chabahar Bay seawater after derivatization with 4-aminoantipyridine. Mar. Pollut. Bull. 2014, 86, 512–517. [Google Scholar] [CrossRef] [PubMed]
- Ou, J.; Hu, L.; Hu, L.; Li, X.; Zou, H. Determining of phenolic compounds in river waterwith on-line coupling bisphenol A imprinted monolithic pre-column with high performance liquid chromatography. Talanta 2006, 69, 1001–1006. [Google Scholar] [CrossRef] [PubMed]
- Feng, Q.; Zhao, L.; Lin, J.M. Molecularly imprinted polymer as micro-solid phase extraction combined with high performance liquid chromatography to determine phenolic compounds in environmental water samples. Anal. Chim. Acta 2009, 650, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Shan, D.; Zhang, J.; Xue, H.G.; Zhang, Y.C.; Cosnier, S.; Ding, S.N. Polycrystalline bismuth oxide films for development of amperometric biosensor for phenolic compound. Biosens. Bioelectron. 2009, 24, 3671–3676. [Google Scholar] [CrossRef] [PubMed]
- Solanki, P.R.; Kaushik, A.; Chavdan, P.M.; Maheshwari, S.N.; Malhotra, B.D. Nanostructured zirconium oxide based genosensor for Escherichia coli detection. Electrochem. Commun. 2009, 11, 2272–2277. [Google Scholar] [CrossRef]
- Pundir, S.; Chauhan, N.; Narang, J.; Pundir, C.S. Amperometric choline biosensor based on multiwalled carbon nanotubes/zirconium oxide nanoparticles electrodeposited on glassy carbon electrode. Anal. Biochem. 2012, 427, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Cao, Y.; Chen, D.; Kong, J.; Deng, J. Amperometric biosensor based on nanoporous ZrO2 matrix. Anal. Chim. Acta 2003, 478, 59–66. [Google Scholar] [CrossRef]
- Zhao, G.; Feng, J.J.; Xu, J.J.; Chen, H.Y. Direct electrochemistry and electrocatalysis of heme proteins immobilized on self-assembled ZrO2 film. Electrochem. Commun. 2005, 7, 724–729. [Google Scholar] [CrossRef]
- Liu, S.; Dai, Z.; Chen, H.; Ju, H. Imobilization of hemoglobin on zirconium dioxide nanoparticles for preparation of a novel hydrogen peroxide biosensor. Biosens. Bioelectron. 2004, 19, 963–969. [Google Scholar] [CrossRef] [PubMed]
- Sumana, G.; Das, M.; Srivasta, S.; Malhotra, B.D. A novel urea biosensor based on zirconia. Thin Solid Films 2010, 519, 1187–1191. [Google Scholar] [CrossRef]
- Yang, Y.; Yang, H.; Yang, M.; Liu, Y.; Shen, G.; Yu, R. Amperometric glucose biosensor based on a surface treated nanoporous ZrO2/Chitosan composite film as immobilization matrix. Anal. Chim. Acta 2004, 525, 213–220. [Google Scholar] [CrossRef]
- Chang, M.Y.; Juang, R.S. Stability and reactivity of acid phosphatase immobilized on composite beads of chitosan and ZrO2 powders. Int. J. Biol. Macromol. 2007, 40, 224–231. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.F.; Liu, Z.M.; Liu, Y.L.; Yang, Y.H.; Shen, G.L.; Yu, R.Q. A mediator-free phenol biosensor based on immobilizing tyrosinase on ZnO nanoparticles. Anal. Biochem. 2006, 349, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Rawal, R.; Chawla, S.; Devender, S.; Pundi, C.S. An amperometric biosensor based on laccase immobilized onto Fe3O4NPs/cMWCNT/PANI/Au electrode for determination of phenolic content in tea leaves extract. Enzym. Microb. Technol. 2012, 51, 179–185. [Google Scholar] [CrossRef] [PubMed]
- Qiao, K.; Hu, N. Direct electron transfer and electrocatalysis of myoglobin loaded in layer-by-layer fils assembled with nonionic poly(ethylene glycol) and ZrO2 nanoparticles. Bioelectrochemistry 2009, 75, 71–76. [Google Scholar] [CrossRef] [PubMed]
- Arshak, K.; Velusamy, V.; Korostynska, O.; Stasiak, K.O.; Adley, C. Conducting polymers and their applications to biosensors: Emphasizing on foodborne pathogen detection. IEEE Sens. J. 2009, 9, 1942–1951. [Google Scholar] [CrossRef]
- Shi, C.; Dai, Y.; Liu, Q.; Xie, Y.; Xu, X. The FT-IR spectrometric analysis of the changes of polyphenol oxidase II secondary structure. J. Mol. Struct. 2003, 644, 139–144. [Google Scholar] [CrossRef]
- Apetrei, I.M.; Apetrei, C. The biocomposite screen-printed biosensor based on immobilization of tyrosinase onto the carboxyl functionalised carbon nanotube for assaying tyramine in fish products. J. Food Eng. 2015, 149, 1–8. [Google Scholar] [CrossRef]
- Lu, L.; Zhang, L.; Zhang, X.; Huan, S.; Shen, G.; Yu, R. A novel tyrosinase biosensor based on hydroxyapatite-chitosan nanocomposite for the detection of phenolic compounds. Anal. Chim. Acta 2010, 665, 146–151. [Google Scholar] [CrossRef] [PubMed]
- Apetrei, C.; Rodriguez-Mendez, M.L.; De Saja, J.A. Amperometrictyrosinase based biosensor using an electropolymerized phosphate-doped polypyrrole film as an immobilization support.Application for detection of phenolic compounds. Electrochim. Acta 2011, 56, 8919–8925. [Google Scholar] [CrossRef]
- Xue, H.; Shen, Z. A highly stable biosensor for phenols prepared by immobilizing polyphenol oxidase into polyaniline-polyacrylonitrile composite matrix. Talanta 2000, 57, 289. [Google Scholar] [CrossRef]
- Cheng, Y.; Liu, Y.; Huang, J.; Li, K.; Xian, Y.; Zhang, W.; Jin, L. Amperometrictyrosinase biosensor based on Fe3O4 nanoparticles-coated carbon nanotubes nanocomposite for rapid detection of coliforms. Electrochim. Acta 2009, 54, 2588–2594. [Google Scholar] [CrossRef]
- Liu, Z.; Wang, J.; Xie, D.; Chen, G. Polyaniline-coated Fe3O4 nanoparticles-carbon-nanotube composite and its application in electrochemical biosensing. Small Nano Micro 2008, 4, 462–466. [Google Scholar] [CrossRef] [PubMed]
- Vedrine, C.; Fabiano, S.; Tran-Minh, C. Amperometric tyrosinase based biosensor using an electrogeneratedpolythiophene film as an entrapment support. Talanta 2003, 59, 535–544. [Google Scholar] [CrossRef]
- Shan, D.; Cosnier, S.; Mousty, C. Layered double hydroxides: An attractives material for electrochemical biosensor design. Anal. Chem. 2003, 75, 3872–3879. [Google Scholar] [CrossRef] [PubMed]
- Han, E.; Shan, D.; Xue, H.G.; Cosnier, S. Hybrid Material based on chitosan and layered double hydroxides: Characterization and application to the design of amperometric phenol biosensor. Biomacromolecules 2007, 8, 971–975. [Google Scholar] [CrossRef] [PubMed]
- Fan, Q.; Shan, D.; Xue, H.G.; He, Y.Y.; Cosnier, S. Amperometric phenol biosensor based on laponite clay–chitosan nanocomposite matrix. Biosens. Bioelectron. 2007, 22, 816–821. [Google Scholar] [CrossRef] [PubMed]
- Song, W.; Li, D.W.; Li, Y.T.; Li, Y.; Long, Y.T. Disposable biosensor based on graphene oxide conjugated with tyrosinase assembled gold nanoparticles. Biosens. Bioelectron. 2011, 26, 3181–3186. [Google Scholar] [CrossRef] [PubMed]
| Interferent | Coexisting Level | Change of Current Response (%) |
|---|---|---|
| Ascorbic acid | 3 µM | −5.78 |
| Uric acid | 300 µM | −0.69 |
| H2O2 | 50 µM | 5.18 |
| Glucose | 1000 µM | 4.65 |
| Mg2+ | 40 µM | 2.88 |
| Ca2+ | 40 µM | 3.20 |
| Fe3+ | 40 µM | 1.14 |
| Composite System | Linear Range (µM) | Applied Potential (V) | Detection Limit (µM) | Response Time (s) | Reference |
|---|---|---|---|---|---|
| Zinc oxide/catechol | 0.15–65 | −0.2 | 0.05 | 10 | [18] |
| Iron oxide/coliform | 0.01–39 | −0.2 | 0.005 | 5 | [27] |
| Bismuth oxide/catechol | 0.01–8 | −0.2 | 0.05 | 8 | [9] |
| Graphene oxide-gold nanoparticle/catechol | 0.083–23 | −0.25 | 0.024 | 6 | [33] |
| Iron oxide/MWCNT/PANI Au/guaicol/polyphenol | 0.1-10 10–500 | −0.2 | 0.03 | 3 | [19] |
| Zirconium oxide | 0.075–10 10–55 | −0.2 | 0.034 | 10 | This work |
| Phenol Compound | Linear Range (µM) | Correlation Coefficient (R2) | Detection Limit (µM) | (µM) |
|---|---|---|---|---|
| Phenol | 0.5–9.5 10.0–55.0 | 0.9900 0.9993 | 0.0379 0.1003 | 61.42 193.33 |
| 4-chlorophenol | 1.5–7.0 | 0.9942 | 0.2372 | 3650.00 |
| p-kresol | 0.025–1.0 1.0–12.0 | 0.9999 0.9997 | 0.0175 0.5232 | 1.50 5.25 |
| River Water | Total Phenolic Content by Present Method (µM) Mean ± SD (n = 3) | Total Phenolic Content by Standard Spectrophotometric Method (µM) Mean ± SD (n = 3) | T-Test Value |
|---|---|---|---|
| River 1 | 0.5839 ± 0.0127 | 0.6675 ± 0.0092 | 1.5500 |
| River 2 | 0.5891 ± 0.0003 | 0.6242 ± 0.0139 | 1.2448 |
| River 3 | 0.5897 ± 0.0002 | 0.5833 ± 0.0416 | 0.0771 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ahmad, N.M.; Abdullah, J.; Yusof, N.A.; Ab Rashid, A.H.; Abd Rahman, S.; Hasan, M.R. Amperometric Biosensor Based on Zirconium Oxide/Polyethylene Glycol/Tyrosinase Composite Film for the Detection of Phenolic Compounds. Biosensors 2016, 6, 31. https://doi.org/10.3390/bios6030031
Ahmad NM, Abdullah J, Yusof NA, Ab Rashid AH, Abd Rahman S, Hasan MR. Amperometric Biosensor Based on Zirconium Oxide/Polyethylene Glycol/Tyrosinase Composite Film for the Detection of Phenolic Compounds. Biosensors. 2016; 6(3):31. https://doi.org/10.3390/bios6030031
Chicago/Turabian StyleAhmad, Nor Monica, Jaafar Abdullah, Nor Azah Yusof, Ahmad Hazri Ab Rashid, Samsulida Abd Rahman, and Md. Rakibul Hasan. 2016. "Amperometric Biosensor Based on Zirconium Oxide/Polyethylene Glycol/Tyrosinase Composite Film for the Detection of Phenolic Compounds" Biosensors 6, no. 3: 31. https://doi.org/10.3390/bios6030031
APA StyleAhmad, N. M., Abdullah, J., Yusof, N. A., Ab Rashid, A. H., Abd Rahman, S., & Hasan, M. R. (2016). Amperometric Biosensor Based on Zirconium Oxide/Polyethylene Glycol/Tyrosinase Composite Film for the Detection of Phenolic Compounds. Biosensors, 6(3), 31. https://doi.org/10.3390/bios6030031






