Electrochemical Biosensor for Rapid and Sensitive Detection of Magnetically Extracted Bacterial Pathogens
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagents and Materials
2.2. Apparatus
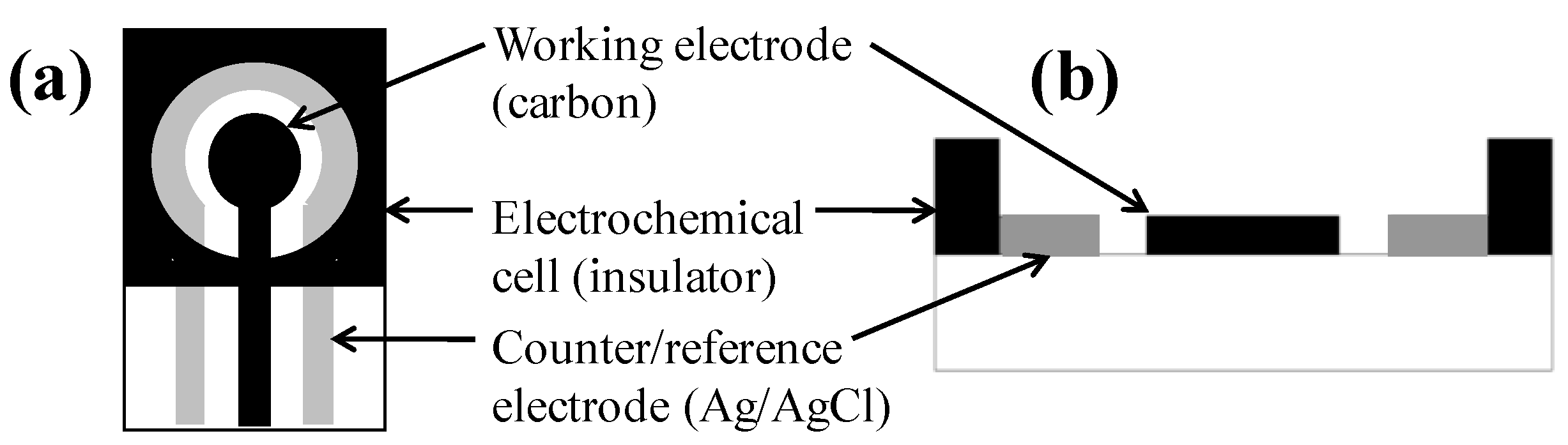
2.3. Preparation of Pure Polyaniline Nanoparticles
2.4. Preparation and Characterization of Core/Shell Nanoparticles
2.5. Electrochemical Characterization of Nanoparticles
2.6. Immunomagnetic Separation of Bacterial Target
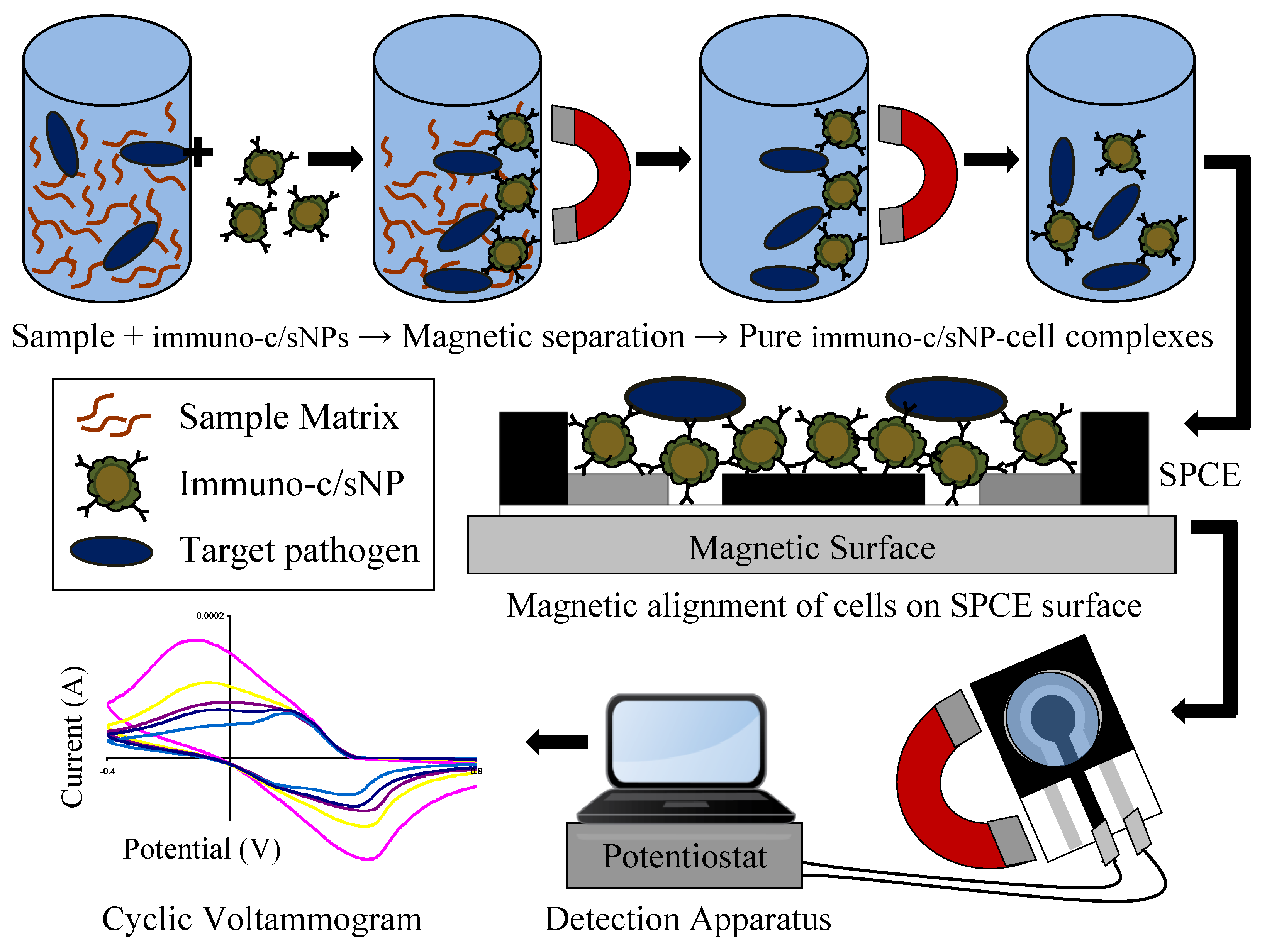
2.7. Electrochemical Detection of Bacterial Target
3. Results and Discussion
3.1. Preparation and Characterization of Core/Shell Nanoparticles
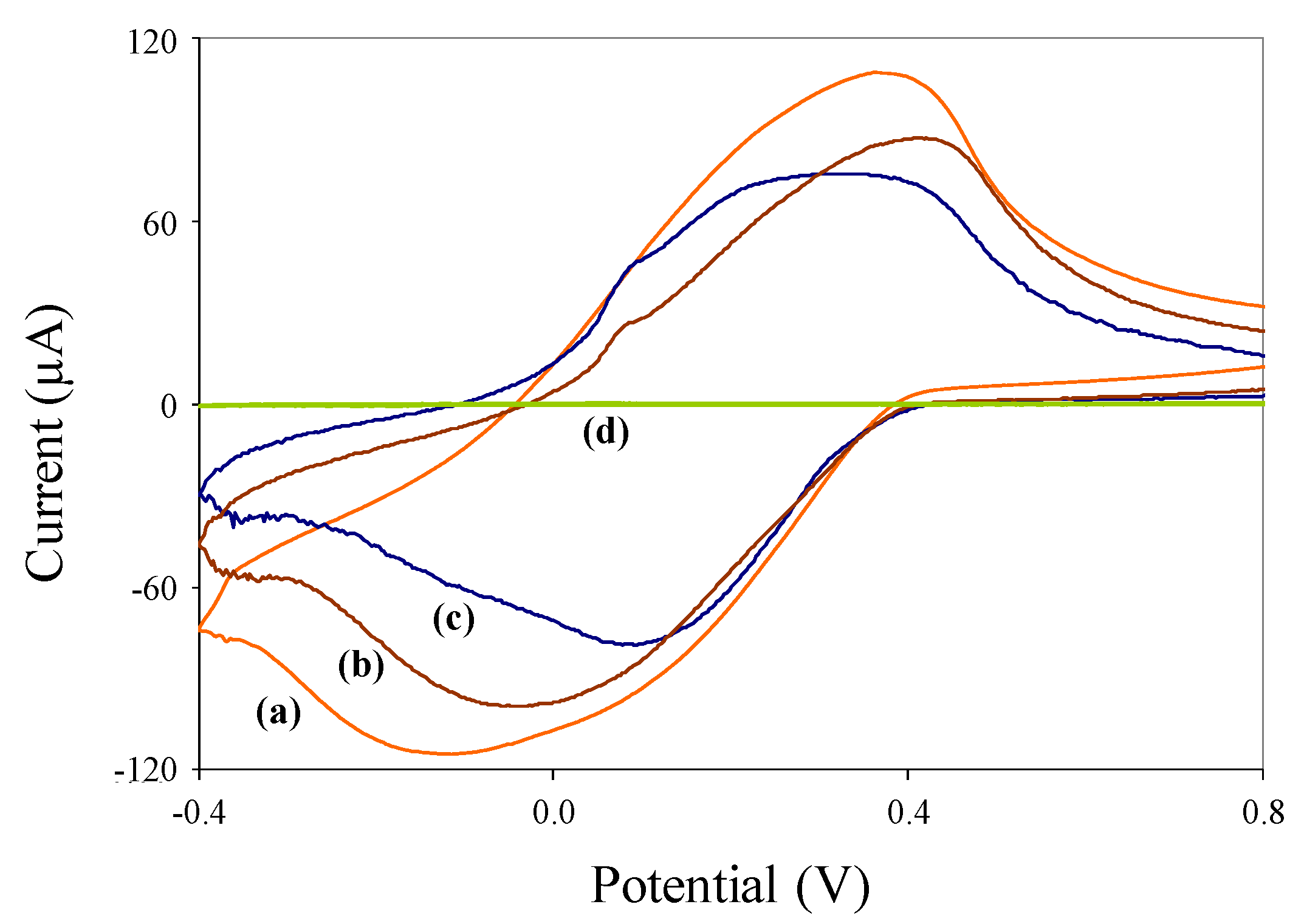
3.2. Electrochemical Characterization of Nanoparticles
3.3. Immunomagnetic Separation of Bacterial Target
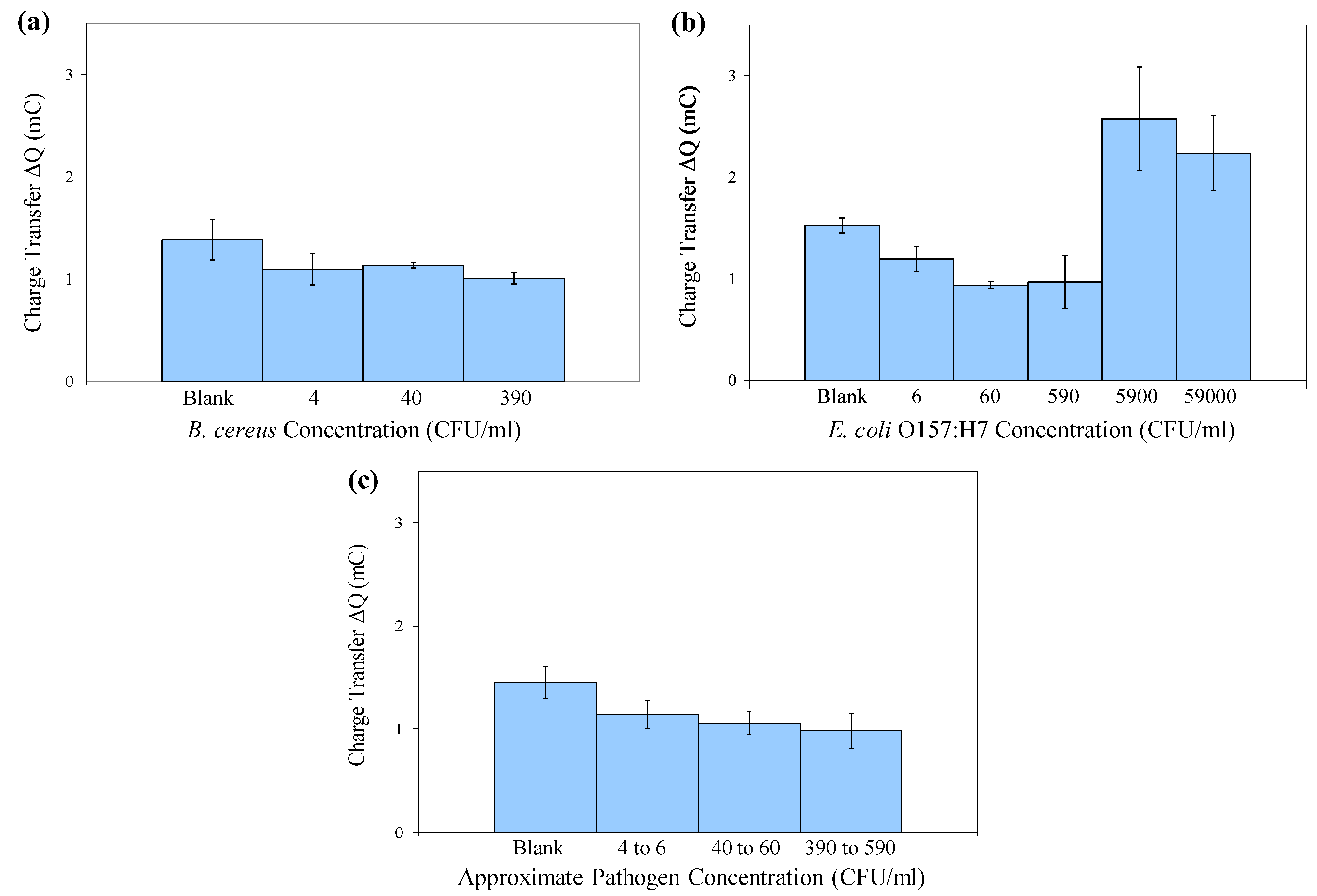
| Original Est. Viable Cell Concentration (CFU/mL) | Mean Captured Viable Cell Concentration (CFU/mL) | Mean Capture Efficiency (log( captured)/log(original)) ± S.D. | Mean Est. Viable Cell Number (CFU) on SPCE | Mean Charge Transfer ΔQ (mC) |
|---|---|---|---|---|
| ± S.D. ( n = 3) | ( n = 3) | ± S.D. ( n = 3) | ± S.D. ( n = 3) | |
| B. cereus | ||||
| 0 (Blank) | 0 | --- | 0 | 1.4 ± 0.20 |
| 4 × 100 | (0 ± 0) × 100 | (0 ± 0)% | 0 ± 0 | 1.1 ± 0.15 |
| 4 × 101 | (1 ± 1) × 101 | (48 ± 43)% | 1 ± 1 | 1.1 ± 0.03 |
| 3.9 × 102 | (1.2 ± 0.31) × 102 | (80 ± 4.4)% | 12 ± 3.1 | 1.0 ± 0.06 |
| E. coli O157:H7 | ||||
| 0 (Blank) | 0 | --- | 0 | 1.5 ± 0.07 |
| 6 × 100 | (7 ± 6) × 100 | (86 ± 75)% | 0.7 ± 0.6 | 1.2 ± 0.12 |
| 6 × 101 | (1 ± 0.6) × 101 | (62 ± 9.8)% | 1 ± 0.6 | 0.94 ± 0.03 |
| 5.9 × 102 | (1.7 ± 0.12) × 102 | (80 ± 1.1)% | 17 ± 1.2 | 0.97 ± 0.26 |
| 5.9 × 103 | (1.6 ± 0.81) × 103 | (84 ± 7.3)% | 160 ± 81 | 2.6 ± 0.51 |
| 5.9 × 104 | (1.3 ± 0.36) × 104 | (86 ± 2.4)% | 1,300 ± 360 | 2.2 ± 0.37 |
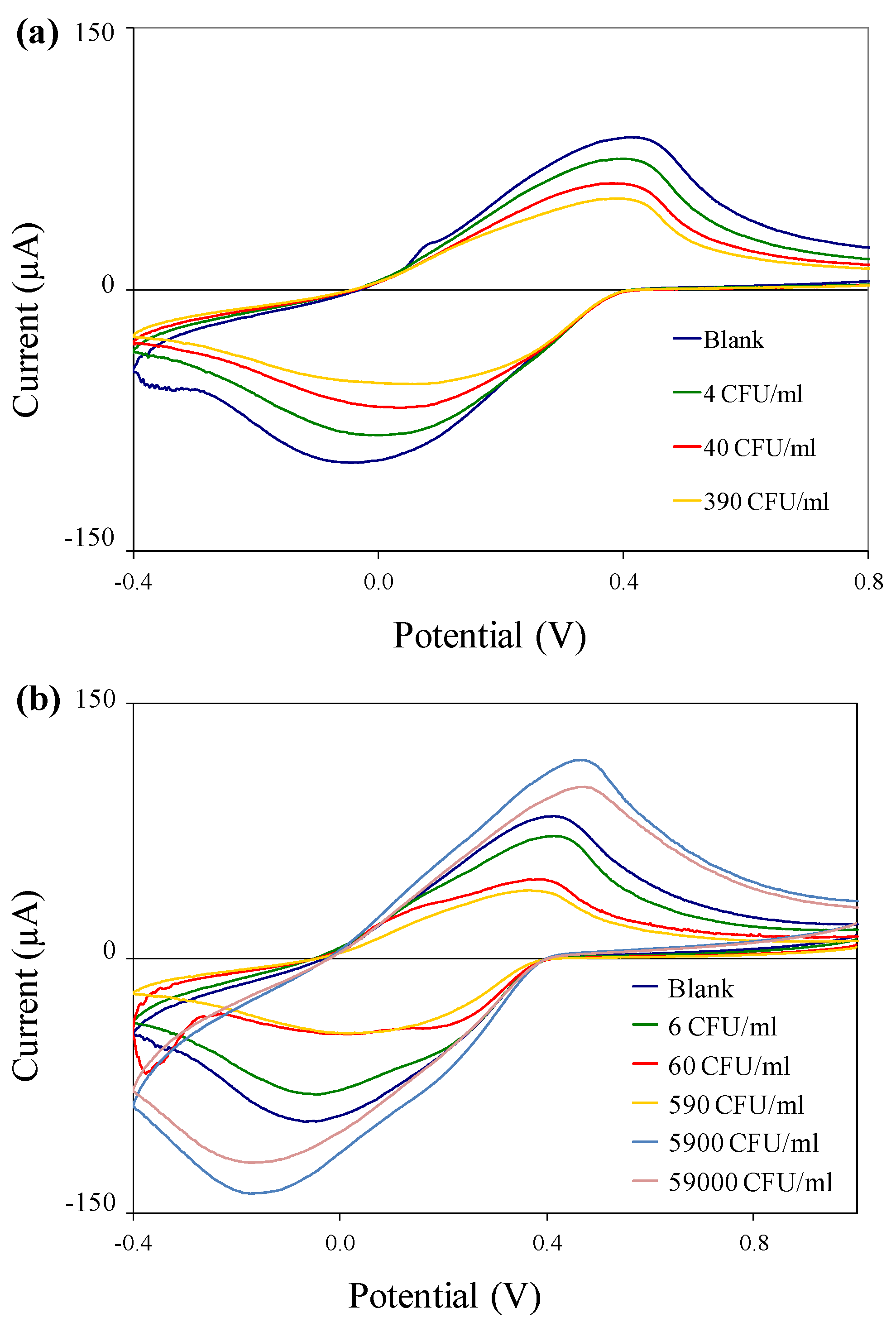
3.4. Electrochemical Detection of Bacterial Target

4. Conclusions
Acknowledgments
References
- Pal, S.; Setterington, E.B.; Alocilja, E.C. Electrically active magnetic nanoparticles for concentrating and detecting Bacillus anthracis spores in a direct-charge transfer biosensor. IEEE Sens. J. 2008, 8, 647–654. [Google Scholar]
- Pal, S.; Alocilja, E.C. Electrically Active Polyaniline Coated Magnetic (EAPM) nanoparticle as novel transducer in biosensor for detection of Bacillus anthracis spores in food samples. Biosens. Bioelectron. 2009, 24, 1437–1444. [Google Scholar]
- Bad Bug Book: Foodborne Pathogenic Micro-Organisms and Natural Toxins Handbook. Bacillus cereus and other Bacillus spp; U.S. Food and Drug Administration (FDA): Silver Spring, MD, USA. Available online: http://www.fda.gov/Food/FoodSafety/FoodborneIllness/FoodborneIllnessFoodbornePathogensNaturalToxins/BadBugBook/ucm070492.htm (accessed on 16 December 2011).
- Edwards, K.; Clancy, H.; Baeumner, A. Bacillus anthracis: Toxicology, epidemiology and current rapid-detection methods. Anal. Bioanal. Chem. 2006, 384, 73–84. [Google Scholar]
- Bioterrorism Agents/Diseases; Centers for Disease Control and Prevention (CDC): Atlanta, GA, USA. Available online: http://emergency.cdc.gov/agent/agentlist-category.asp (accessed on 16 December 2011).
- Biodefense and Emerging Infectious Diseases: NIAID Category A, B, and C Priority Pathogens; National Institute of Allergy and Infectious Diseases (NIAID): Bethesda, MD, USA. Available online: http://www.niaid.nih.gov/topics/BiodefenseRelated/Biodefense/Pages/CatA.aspx (accessed on 16 December 2011).
- Bacteriological Analytical Manual (BAM). Chapter 4A: Diarrheagenic Escherichia coli. U.S. Food and Drug Administration (FDA): Silver Spring, MD, USA. Available online: http://www.fda.gov/Food/ScienceResearch/LaboratoryMethods/BacteriologicalAnalyticalManualBAM/ucm070080.htm (accessed on 16 December 2011).
- Water Sanitation and Health: Guidelines for Drinking-Water Quality, 3rd ed; World Health Organization (WHO): Geneva, Switzerland. Available online: http://www.who.int/water_sanitation_health/dwq/gdwq3rev/en/index.html (accessed on 16 December 2011).
- E. coli Outbreak Investigations; Centers for Disease Control and Prevention (CDC): Atlanta, GA, USA. Available online: http://www.cdc.gov/ecoli/outbreaks.html (accessed on 16 December 2011).
- Escherichia coli O157:H7 and Other Shiga Toxin-Producing Escherichia coli (STEC); Centers for Disease Control and Prevention (CDC): Atlanta, GA, USA. Available online: http://www.cdc.gov/nczved/divisions/dfbmd/diseases/ecoli_o157h7/ (accessed on 16 December 2011).
- Jaffrezic-Renault, N.; Martelet, C.; Chevolot, Y.; Cloarec, J.-P. Biosensors and bio-bar code assays based on biofunctionalized magnetic microbeads. Sensors 2007, 7, 589–614. [Google Scholar] [CrossRef]
- Tu, S.-I.; Golden, M.; Cooke, P.; Paoli, G.; Gehring, A. Detection of Escherichia coli O157:H7 through the formation of sandwiched complexes with immunomagnetic and fluorescent beads. J. Rapid Meth. Aut. Mic. 2005, 13, 269–282. [Google Scholar] [CrossRef]
- Cheng, Y.; Liu, Y.; Huang, J.; Li, K.; Zhang, W.; Xian, Y.; Jin, L. Combining biofunctional magnetic nanoparticles and ATP bioluminescence for rapid detection of Escherichia coli. Talanta 2009, 77, 1332–1336. [Google Scholar] [CrossRef]
- Aslan, K.; Previte, M.J.R.; Zhang, Y.; Gallagher, T.; Baillie, L.; Geddes, C.D. Extraction and detection of DNA from Bacillus anthracis spores and the vegetative cells within 1 min. Anal. Chem. 2008, 80, 4125–4132. [Google Scholar]
- Liu, Y.J.; Yao, D.J.; Chang, H.Y.; Liu, C.M.; Chen, C. Magnetic bead-based DNA detection with multi-layers quantum dots labeling for rapid detection of Escherichia coli O157:H7. Biosens. Bioelectron. 2008, 24, 558–565. [Google Scholar]
- Ruan, C.; Wang, H.; Li, Y. A bienzyme electrochemical biosensor coupled with immunomagnetic separation for rapid detection of Escherichia coli O157:H7 in food samples. Trans. ASAE 2002, 45, 249–255. [Google Scholar]
- Gehring, A.G.; Brewster, J.D.; Irwin, P.L.; Tu, S.-I.; Van Houten, L.J. 1-Naphthyl phosphate as an enzymatic substrate for enzyme-linked immunomagnetic electrochemistry. J. Electroanal. Chem. 1999, 469, 27–33. [Google Scholar]
- Gehring, A.G.; Tu, S.-I. Enzyme-linked immunomagnetic electrochemical detection of live Escherichia coli O157:H7 in apple juice. J. Food Protect. 2005, 68, 146–149. [Google Scholar]
- Varshney, M.; Li, Y. Interdigitated array microelectrode based impedance biosensor coupled with magnetic nanoparticle-antibody conjugates for detection of Escherichia coli O157:H7 in food samples. Biosens. Bioelectron. 2007, 22, 2408–2414. [Google Scholar] [CrossRef]
- Varshney, M.; Li, Y.; Srinivasan, B.; Tung, S. A label-free, microfluidics and interdigitated array microelectrode-based impedance biosensor in combination with nanoparticles immunoseparation for detection of Escherichia coli O157:H7 in food samples. Sens. Actuat. B-Chem. 2007, 128, 99–107. [Google Scholar]
- Maalouf, R.; Hassen, W.; Fournier-Wirth, C.; Coste, J.; Jaffrezic-Renault, N. Comparison of two innovative approaches for bacterial detection: Paramagnetic nanoparticles and self-assembled multilayer processes. Microchim. Acta 2008, 163, 157–161. [Google Scholar] [CrossRef]
- Palchetti, I.; Mascini, M. Electroanalytical biosensors and their potential for food pathogen and toxin detection. Anal. Bioanal. Chem. 2008, 391, 455–471. [Google Scholar] [CrossRef]
- Daniels, J.S.; Pourmand, N. Label-free impedance biosensors: Opportunities and challenges. Electroanalysis 2007, 19, 1239–1257. [Google Scholar] [CrossRef]
- Obuchowska, A. Quantitation of bacteria through adsorption of intracellular biomolecules on carbon paste and screen-printed carbon electrodes and voltammetry of redox-active probes. Anal. Bioanal. Chem. 2008, 390, 1361–1371. [Google Scholar] [CrossRef]
- Shabani, A.; Zourob, M.; Allain, B.; Marquette, C.A.; Lawrence, M.F.; Mandeville, R. Bacteriophage-modified microarrays for the direct impedimetric detection of bacteria. Anal. Chem. 2008, 80, 9475–9482. [Google Scholar]
- Escamilla-Gómez, V.; Campuzano, S.; Pedrero, M.; Pingarrón, J.M. Gold screen-printed-based impedimetric immunobiosensors for direct and sensitive Escherichia coli quantisation. Biosens. Bioelectron. 2009, 24, 3365–3371. [Google Scholar]
- Gamella, M.; Campuzano, S.; Parrado, C.; Reviejo, A.J.; Pingarrón, J.M. Microorganisms recognition and quantification by lectin adsorptive affinity impedance. Talanta 2009, 78, 1303–1309. [Google Scholar]
- Susmel, S.; Guilbault, G.G.; O’Sullivan, C.K. Demonstration of labeless detection of food pathogens using electrochemical redox probe and screen printed gold electrodes. Biosens. Bioelectron. 2003, 18, 881–889. [Google Scholar] [CrossRef]
- Rahman, M.; Kumar, P.; Park, D.-S.; Shim, Y.-B. Electrochemical sensors based on organic conjugated polymers. Sensors 2008, 8, 118–141. [Google Scholar]
- Sarno, D.M.; Manohar, S.K.; MacDiarmid, A.G. Controlled interconversion of semiconducting and metallic forms of polyaniline nanofibers. Synth. Met. 2005, 148, 237–243. [Google Scholar]
- Prakash, R. Electrochemistry of polyaniline: Study of the pH effect and electrochromism. J. Appl. Polym. Sci. 2002, 83, 378–385. [Google Scholar] [CrossRef]
- Pal, S.; Alocilja, E.C.; Downes, F.P. Nanowire labeled direct-charge transfer biosensor for detecting Bacillus species. Biosens. Bioelectron. 2007, 22, 2329–2336. [Google Scholar]
- Laska, J.; Widlarz, J. One-step polymerization leading to conducting polyaniline. Synth. Met. 2003, 135–136, 263–264. [Google Scholar]
- Laska, J.; Widlarz, J. Water soluble polyaniline. Synth. Met. 2003, 135–13, 261–262. [Google Scholar]
- Sharma, R.; Lamba, S.; Annapoorni, S.; Sharma, P.; Inoue, A. Composition dependent magnetic properties of iron oxide-polyaniline nanoclusters. J. Appl. Phys. 2005, 97, 14311–14316. [Google Scholar]
- Liu, W.; Kumar, J.; Tripathy, S.; Senecal, K.J.; Samuelson, L. Enzymatically synthesized conducting polyaniline. J. Am. Chem. Soc. 1998, 121, 71–78. [Google Scholar]
- Varshney, M.; Yang, L.; Su, X.L.; Li, Y. Magnetic nanoparticle-antibody conjugates for the separation of Escherichia coli O157:H7 in ground beef. J. Food Protect. 2005, 68, 1804–1811. [Google Scholar]
- Widjojoatmodjo, M.N.; Fluit, A.C.; Torensma, R.; Verhoef, J. Comparison of immunomagnetic beads coated with protein A, protein G, or goat anti-mouse immunoglobulins. Applications in enzyme immunoassays and immunomagnetic separations. J. Immunol. Methods 1993, 165, 11–19. [Google Scholar] [CrossRef]
- Aybay, C. Differential binding characteristics of protein G and protein A for Fc fragments of papain-digested mouse IgG. Immunol. Lett. 2003, 85, 231–235. [Google Scholar] [CrossRef]
- Wang, Z.; Jin, G. Feasibility of protein A for the oriented immobilization of immunoglobulin on silicon surface for a biosensor with imaging ellipsometry. J. Biochem. Bioph. Meth. 2003, 57, 203–211. [Google Scholar]
- Marquis, R.E.; Carstensen, E.L. Electric conductivity and internal osmolality of intact bacterial cells. J. Bacteriol. 1973, 113, 1198–1206. [Google Scholar]
- Radke, S.M.; Alocilja, E.C. Design and fabrication of a microimpedance biosensor for bacterial detection. IEEE Sens. J. 2004, 4, 434–440. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Setterington, E.B.; Alocilja, E.C. Electrochemical Biosensor for Rapid and Sensitive Detection of Magnetically Extracted Bacterial Pathogens. Biosensors 2012, 2, 15-31. https://doi.org/10.3390/bios2010015
Setterington EB, Alocilja EC. Electrochemical Biosensor for Rapid and Sensitive Detection of Magnetically Extracted Bacterial Pathogens. Biosensors. 2012; 2(1):15-31. https://doi.org/10.3390/bios2010015
Chicago/Turabian StyleSetterington, Emma B., and Evangelyn C. Alocilja. 2012. "Electrochemical Biosensor for Rapid and Sensitive Detection of Magnetically Extracted Bacterial Pathogens" Biosensors 2, no. 1: 15-31. https://doi.org/10.3390/bios2010015
APA StyleSetterington, E. B., & Alocilja, E. C. (2012). Electrochemical Biosensor for Rapid and Sensitive Detection of Magnetically Extracted Bacterial Pathogens. Biosensors, 2(1), 15-31. https://doi.org/10.3390/bios2010015





