Sustainable Sensing with Paper Microfluidics: Applications in Health, Environment, and Food Safety
Abstract
1. Introduction
2. Fundamentals of Paper Microfluidics
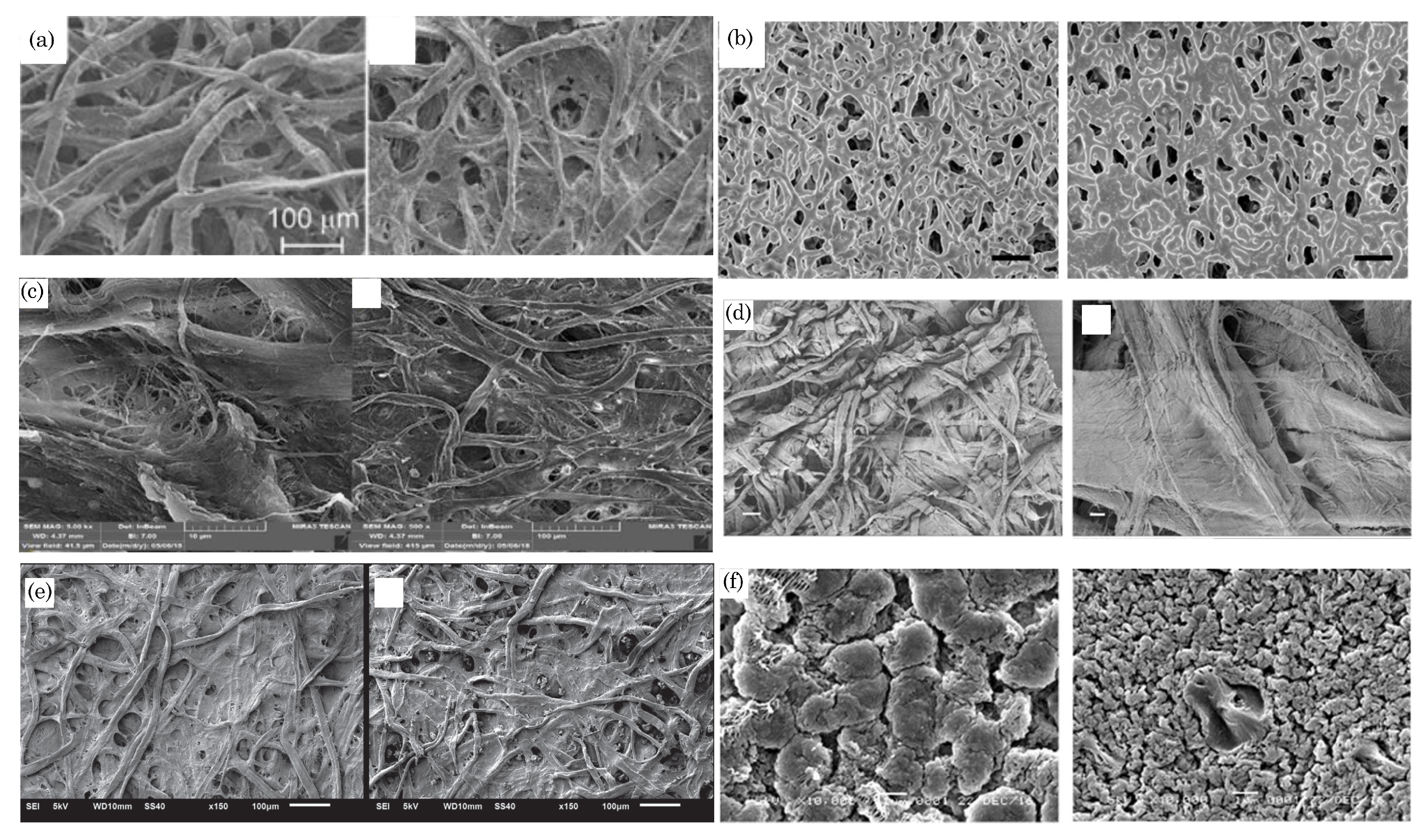
2.1. Paper Types and Their Characteristics
2.2. Paper Selection Factors
2.3. Principles of Fluid Transport in Paper
2.3.1. Classical Lucas–Washburn Equation (Capillary Flow)
2.3.2. Darcy’s Law for Fluid Flow
2.4. Dimensionless Numbers for Fluid Transport
2.4.1. Capillary Number (Ca)
2.4.2. Reynolds Number (Re)
2.4.3. Weber Number (We)
2.4.4. Schmidt Number (Sc)
2.4.5. Péclet Number ()
3. Classifications of Paper-Based Assays
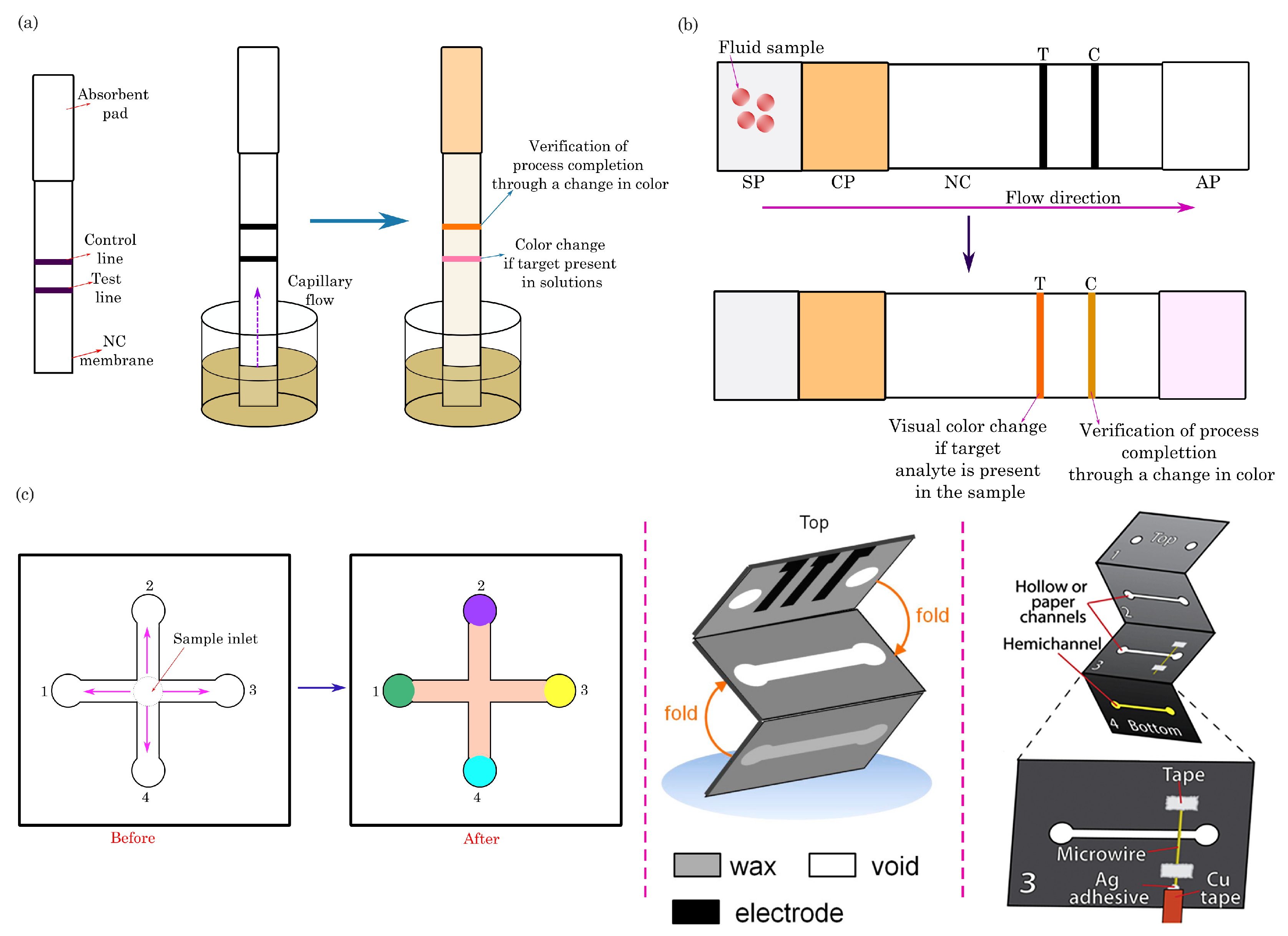
3.1. Dipstick Assays
3.2. Lateral-Flow Assays
3.3. Microfluidic Paper-Based Analytical Devices (PADs)
4. Fabrication Techniques for Paper-Based Devices
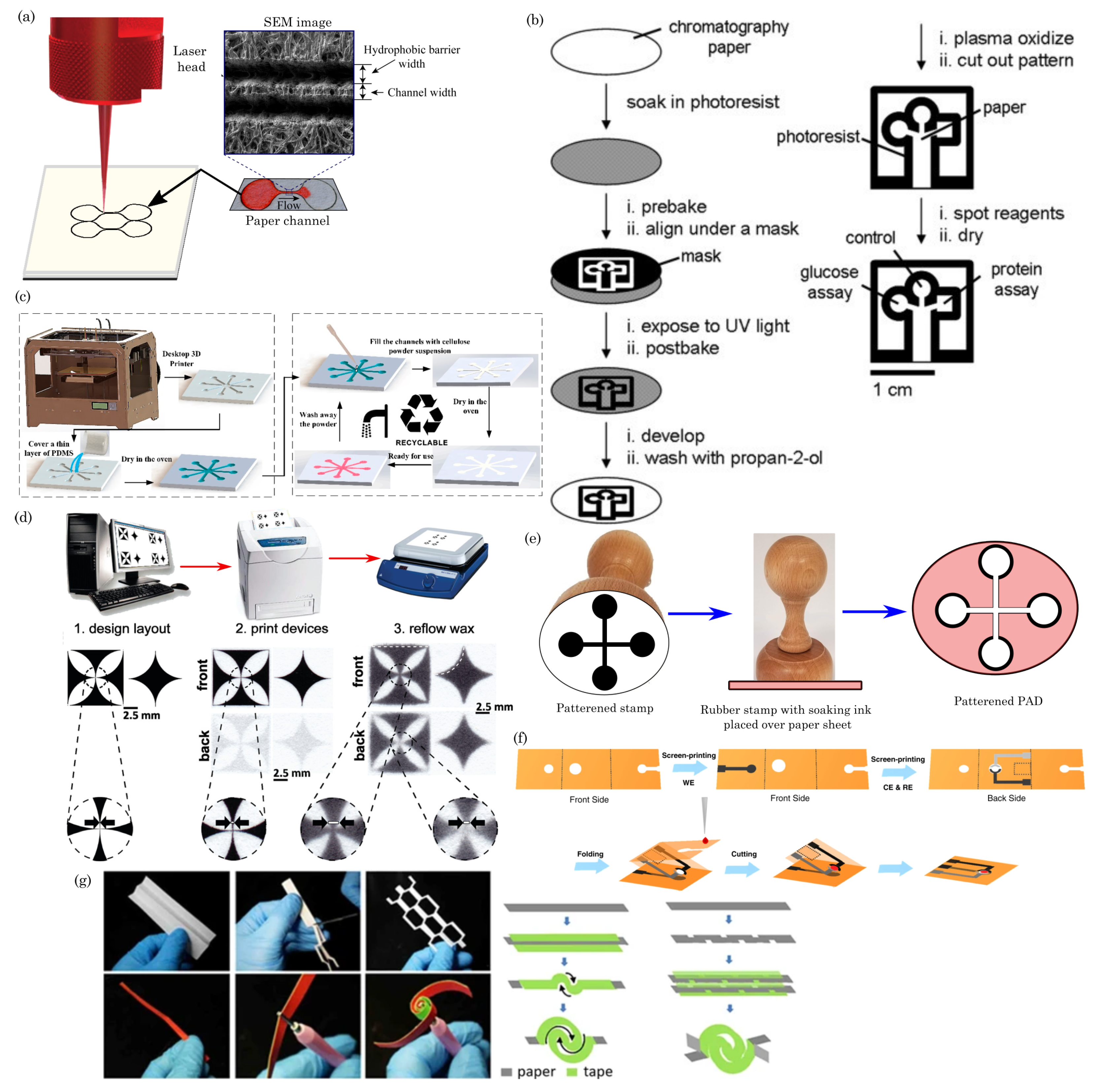
4.1. Blade Cutting/Plotting
4.2. Laser Cutting
4.3. Photolithography
4.4. 3D Printing
4.5. Screen Printing
4.6. Wax Printing
4.7. Inkjet Printing
4.8. Embossing
4.9. Origami, Quilling, and Kirigami
5. Detection Techniques
5.1. Colorimetric Sensing
5.2. Electrochemical Sensing
5.3. Fluorescence
5.4. Chemiluminescence
5.5. Electrochemiluminescence
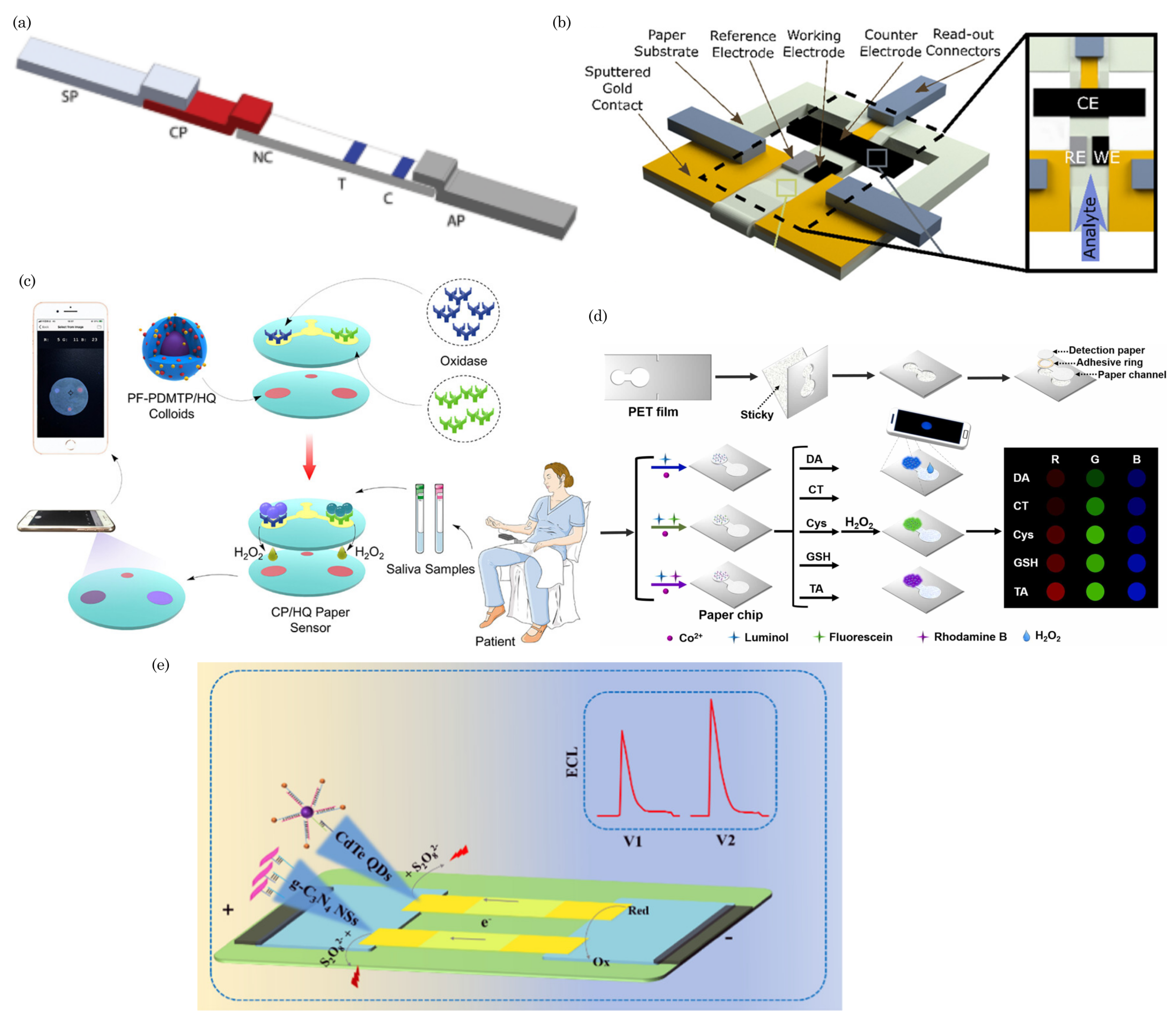
5.6. Surface-Enhanced Raman Spectroscopy (SERS)
6. Signal Readout Approach
6.1. Qualitative
6.2. Quantitative
7. Applications in Health Sensing
7.1. Diagnostic Assays for Infectious Diseases and Others Analytes
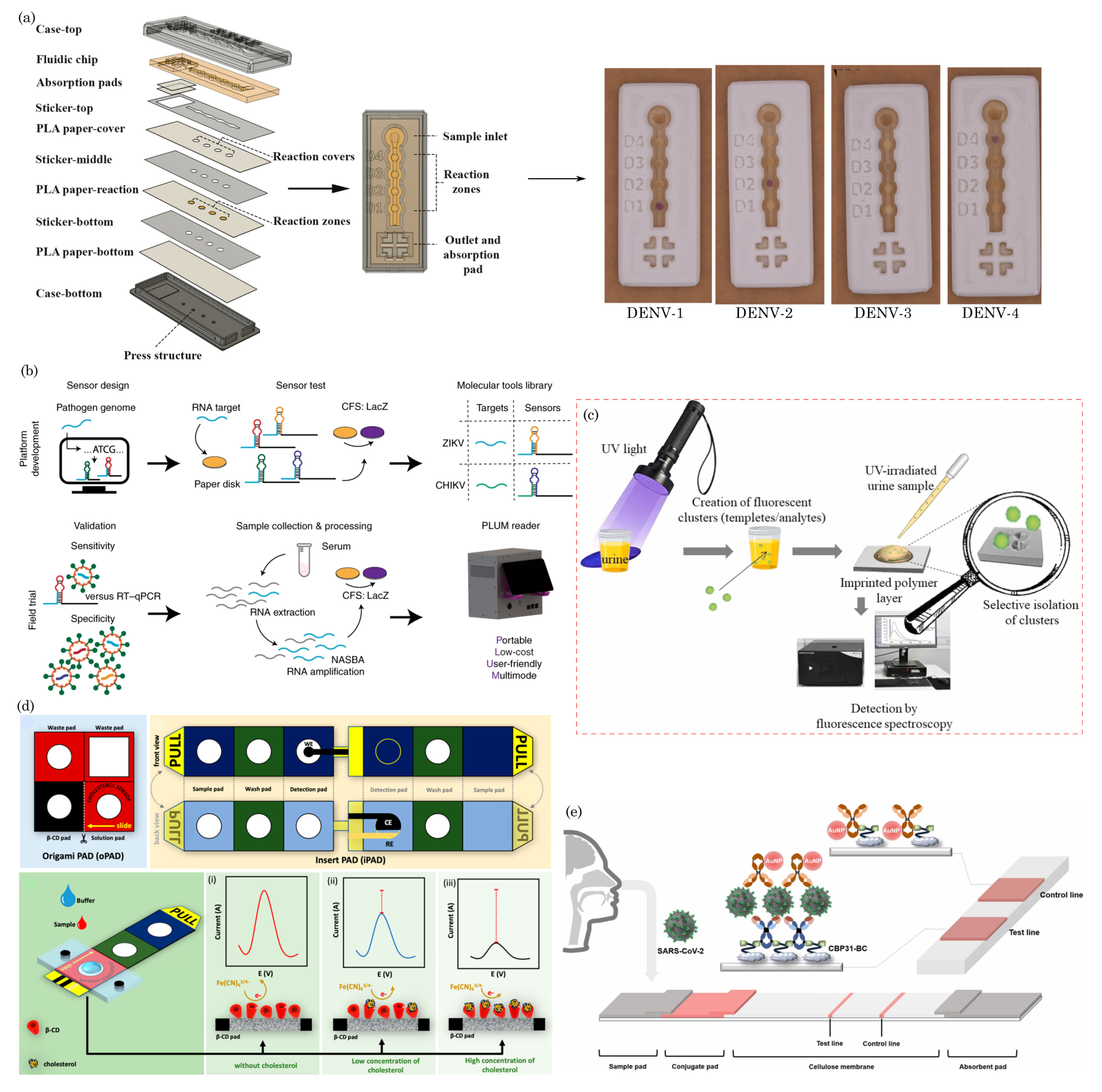
7.2. Wearable and Portable Health-Monitoring Devices
7.3. Animal Health Screening
8. Environmental Monitoring/Sensing
8.1. Detection of Soil Contaminants

8.2. Water Quality Monitoring
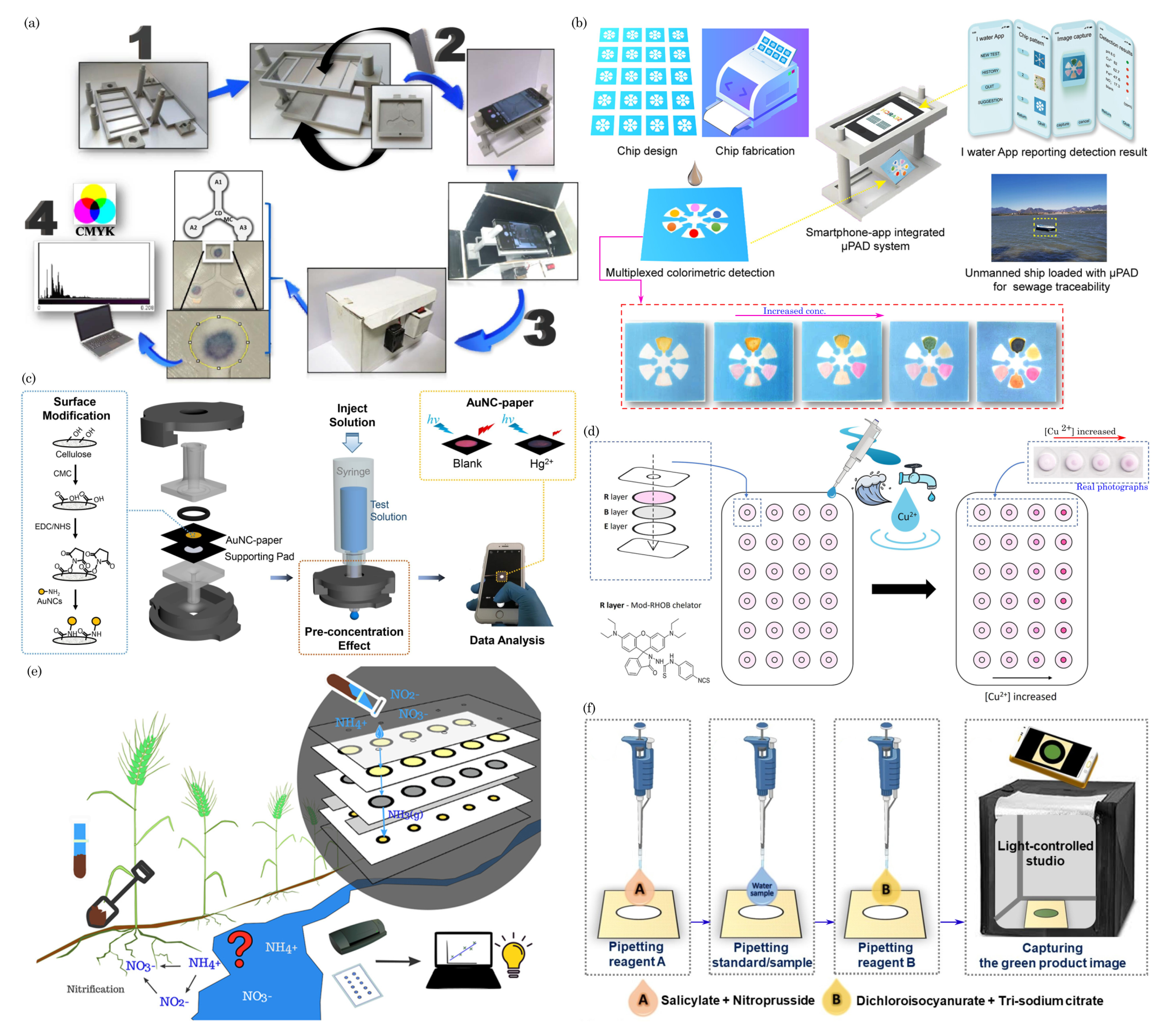
8.3. Air Quality Monitoring/Gas Sensing
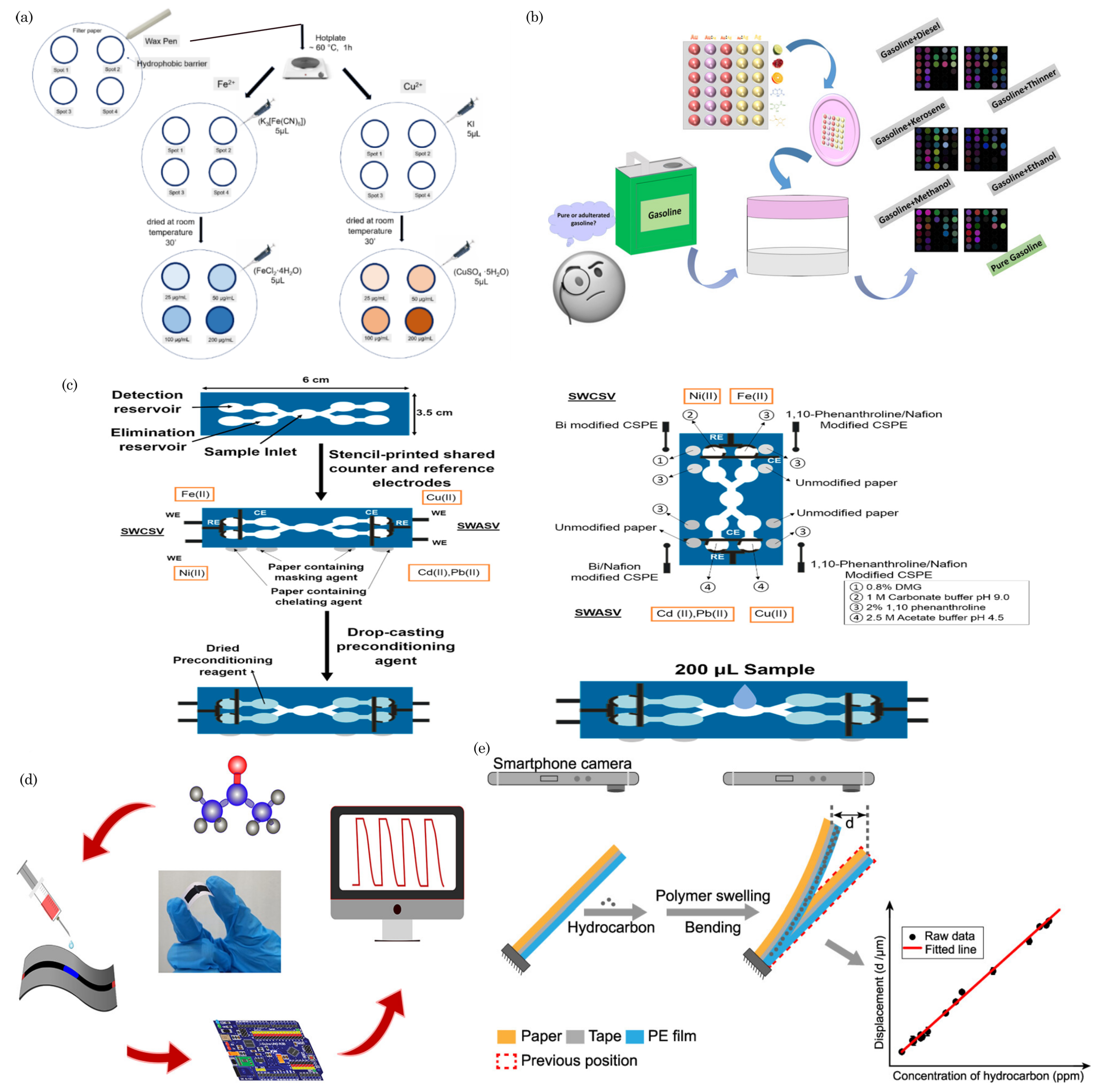
9. Food Safety
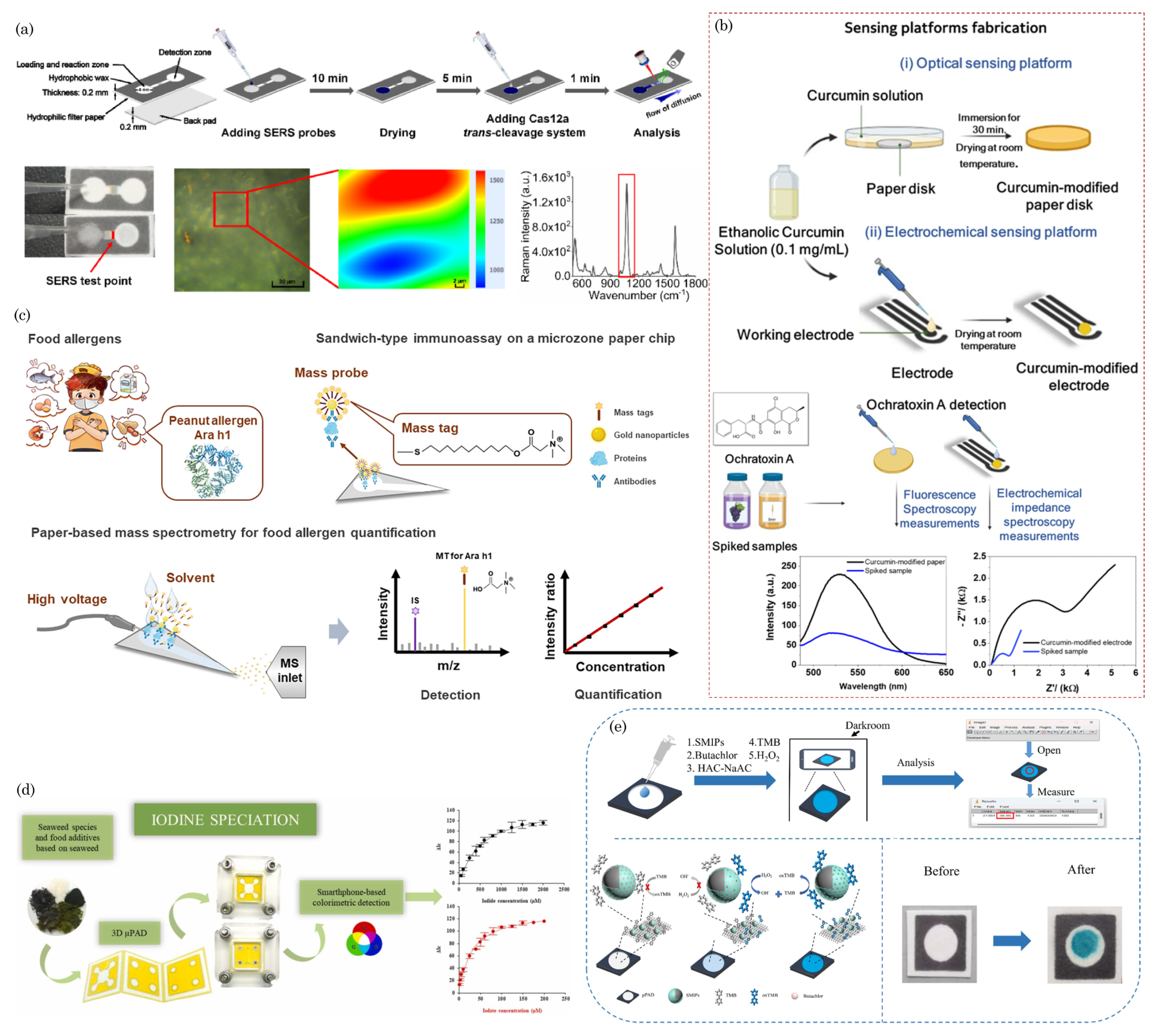
10. Biodegradability and Sustainability
10.1. Environmental Impact of Traditional Microfluidic Devices
10.2. Advantages of Biodegradable Paper Microfluidics
11. Challenges and Future Perspectives
11.1. Current Challenges in Paper Microfluidics
11.2. Prospects and Potential Innovations
12. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Peeling, R.W.; Holmes, K.K.; Mabey, D.; Ronald, A. Rapid tests for sexually transmitted infections (STIs): The way forward. Sex. Transm. Dis. 2006, 82, v1–v6. [Google Scholar] [CrossRef] [PubMed]
- Kosack, C.S.; Page, A.L.; Klatser, P.R. A guide to aid the selection of diagnostic tests. Bull. World Health Organ. 2017, 95, 639–645. [Google Scholar] [CrossRef] [PubMed]
- Martinez, A.W.; Phillips, S.T.; Whitesides, G.M.; Carrilho, E. Diagnostics for the developing world: Microfluidic paper-based analytical devices. Anal. Chem. 2010, 82, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Vicente, A.T.; Araújo, A.; Gaspar, D.; Santos, L.; Marques, A.C.; Mendes, M.J.; Pereira, L.; Fortunato, E.; Martins, R. Optoelectronics and bio devices on paper powered by solar cells. In Nanostructured Solar Cells; IntechOpen: London, UK, 2017. [Google Scholar] [CrossRef]
- Nontawong, N.; Ngaosri, P.; Chunta, S.; Jarujamrus, P.; Nacapricha, D.; Lieberzeit, P.A.; Amatatongchai, M. Smart sensor for assessment of oxidative/nitrative stress biomarkers using a dual-imprinted electrochemical paper-based analytical device. Anal. Chim. Acta 2022, 1191, 339363. [Google Scholar] [CrossRef] [PubMed]
- Patella, B.; Parisi, A.; Moukri, N.; Gitto, F.; Busacca, A.; Aiello, G.; Russo, M.; O’Riordan, A.; Inguanta, R. Phosphate ions detection by using an electrochemical sensor based on laser-scribed graphene oxide on paper. Electrochim. Acta 2023, 461, 142600. [Google Scholar] [CrossRef]
- Lu, Y.; Shi, W.; Qin, J.; Lin, B. Fabrication and characterization of paper-based microfluidics prepared in nitrocellulose membrane by wax printing. Anal. Chem. 2010, 82, 329–335. [Google Scholar] [CrossRef] [PubMed]
- Tang, R.H.; Li, M.; Liu, L.N.; Zhang, S.F.; Alam, N.; You, M.; Ni, Y.H.; Li, Z.D. Chitosan-modified nitrocellulose membrane for paper-based point-of-care testing. Cellulose 2020, 27, 3835–3846. [Google Scholar] [CrossRef]
- Tang, R.; Xie, M.Y.; Li, M.; Cao, L.; Feng, S.; Li, Z.; Xu, F. Nitrocellulose membrane for paper-based biosensor. Appl. Mater. Today 2022, 26, 101305. [Google Scholar] [CrossRef]
- Tang, R.; Xie, M.; Yan, X.; Qian, L.; Giesy, J.P.; Xie, Y. A nitrocellulose/cotton fiber hybrid composite membrane for paper-based biosensor. Cellulose 2023, 30, 6457–6469. [Google Scholar] [CrossRef]
- Boonyasit, Y.; Chailapakul, O.; Laiwattanapaisal, W. A multiplexed three-dimensional paper-based electrochemical impedance device for simultaneous label-free affinity sensing of total and glycated haemoglobin: The potential of using a specific single-frequency value for analysis. Anal. Chim. Acta 2016, 936, 1–11. [Google Scholar] [CrossRef]
- Boonyasit, Y.; Chailapakul, O.; Laiwattanapaisal, W. A folding affinity paper-based electrochemical impedance device for cardiovascular risk assessment. Biosens. Bioelectron. 2019, 130, 389–396. [Google Scholar] [CrossRef] [PubMed]
- Arduini, F.; Cinti, S.; Caratelli, V.; Amendola, L.; Palleschi, G.; Moscone, D. Origami multiple paper-based electrochemical biosensors for pesticide detection. Biosens. Bioelectron. 2019, 126, 346–354. [Google Scholar] [CrossRef] [PubMed]
- Mondal, B.P.; Das, S.; Ranjan, P.; Datta, A. Optimizing Output Performances in Stationery Papers–Based Hybrid Inorganic–Organic Flexible Thermoelectric Generators. Phys. Status Solidi A 2023, 220, 2300228. [Google Scholar] [CrossRef]
- Gong, S.; Schwalb, W.; Wang, Y.; Chen, Y.; Tang, Y.; Si, J.; Shirinzadeh, B.; Cheng, W. A wearable and highly sensitive pressure sensor with ultrathin gold nanowires. Nat. Commun. 2014, 5, 3132. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.; Madzivhandila, P.; Ntuli, L.; Bezuidenhout, P.; Zheng, H.; Land, K. Printed paper–based electrochemical sensors for low-cost point-of-need applications. Electrocatalysis 2019, 10, 342–351. [Google Scholar] [CrossRef]
- Lim, W.Y.; Goh, C.H.; Yap, K.Z.; Ramakrishnan, N. One-step fabrication of paper-based inkjet-printed graphene for breath monitor sensors. Biosensors 2023, 13, 209. [Google Scholar] [CrossRef]
- Camargo, J.R.; Andreotti, I.A.; Kalinke, C.; Henrique, J.M.; Bonacin, J.A.; Janegitz, B.C. Waterproof paper as a new substrate to construct a disposable sensor for the electrochemical determination of paracetamol and melatonin. Talanta 2020, 208, 120458. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Wright, C.; Dincel, O.; Chi, T.Y.; Kameoka, J. A low-cost paper glucose sensor with molecularly imprinted polyaniline electrode. Sensors 2020, 20, 1098. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Chiang, S.W.; Lin, W.; Yang, C.; Li, Z.; Liu, J.; Cui, X.; Kang, F.; Wong, C.P. Towards practical application of paper based printed circuits: Capillarity effectively enhances conductivity of the thermoplastic electrically conductive adhesives. Sci. Rep. 2014, 4, 6275. [Google Scholar] [CrossRef]
- Martins, G.V.; Riveiro, A.; Chiussi, S.; Sales, M. Flexible sensing devices integrating molecularly-imprinted polymers for the detection of 3-nitrotyrosine biomarker. Biosens. Bioelectron. X 2022, 10, 100107. [Google Scholar] [CrossRef]
- Qi, J.; Li, B.; Zhou, N.; Wang, X.; Deng, D.; Luo, L.; Chen, L. The strategy of antibody-free biomarker analysis by in-situ synthesized molecularly imprinted polymers on movable valve paper-based device. Biosens. Bioelectron. 2019, 142, 111533. [Google Scholar] [CrossRef] [PubMed]
- Chen, I.H.; You, M.W.; Tsai, J.H.; Chang, J.H.; Cheng, I.C.; Hsu, C.C.; Luo, S.C.; Chen, C.F.; Chen, J.Z. Feasibility study of dielectric barrier discharge jet-patterned perfluorodecyltrichlorosilane-coated paper for biochemical diagnosis. ECS J. Solid State Sci. Technol. 2021, 10, 037005. [Google Scholar] [CrossRef]
- Cao, C.X.; Yuan, J.; Cheng, J.P.; Han, B.H. Synthesis of porous polymer/tissue paper hybrid membranes for switchable oil/water separation. Sci. Rep. 2017, 7, 3101. [Google Scholar] [CrossRef] [PubMed]
- Arena, A.; Donato, N.; Saitta, G.; Bonavita, A.; Rizzo, G.; Neri, G. Flexible ethanol sensors on glossy paper substrates operating at room temperature. Sens. Actuators B Chem. 2010, 145, 488–494. [Google Scholar] [CrossRef]
- Jiang, L.; Nelson, G.W.; Kim, H.; Sim, I.; Han, S.O.; Foord, J.S. Cellulose-derived supercapacitors from the carbonisation of filter paper. ChemistryOpen 2015, 4, 586–589. [Google Scholar] [CrossRef] [PubMed]
- Lin, D.; Li, B.; Fu, L.; Qi, J.; Xia, C.; Zhang, Y.; Chen, J.; Choo, J.; Chen, L. A novel polymer-based nitrocellulose platform for implementing a multiplexed microfluidic paper-based enzyme-linked immunosorbent assay. Microsyst. Nanoeng. 2022, 8, 53. [Google Scholar] [CrossRef] [PubMed]
- Jabar, A.W.; AL-Bawi, Z.F.; Faris, R.A.; Wahhab, H.K. Plasmonic Nanoparticles Decorated Salty Paper Based on SERS Platform for Diagnostic low-Level Contamination: Lab on Paper. Iraqi J. Laser 2019, 18, 55–61. [Google Scholar]
- Fernandes, A.R.; Bernardo, R.A.; Carvalho, T.C.d.; Vaz, B.G.; Chaves, A.R. Graphene oxides coated paper as a substrate to paper spray ionization mass spectrometry for creatinine determination in urine samples. J. Braz. Chem. Soc. 2019, 30, 1074–1081. [Google Scholar] [CrossRef]
- Arahman, N.; Fahrina, A.; Amalia, S.; Sunarya, R.; Mulyati, S. Effect of PVP on the characteristic of modified membranes made from waste PET bottles for humic acid removal. F1000Research 2017, 6, 668. [Google Scholar] [CrossRef]
- Van der Westhuizen, J.; Du Plessis, J.P. An attempt to quantify fibre bed permeability utilizing the phase average Navier-Stokes equation. Compos. Part A Appl. Sci. Manuf. 1996, 27, 263–269. [Google Scholar] [CrossRef]
- Nabovati, A.; Llewellin, E.W.; Sousa, A.C. A general model for the permeability of fibrous porous media based on fluid flow simulations using the lattice Boltzmann method. Compos. Part A Appl. Sci. Manuf. 2009, 40, 860–869. [Google Scholar] [CrossRef]
- Choi, J.R.; Liu, Z.; Hu, J.; Tang, R.; Gong, Y.; Feng, S.; Ren, H.; Wen, T.; Yang, H.; Qu, Z.; et al. Polydimethylsiloxane-paper hybrid lateral flow assay for highly sensitive point-of-care nucleic acid testing. Anal. Chem. 2016, 88, 6254–6264. [Google Scholar] [CrossRef] [PubMed]
- Parolo, C.; Medina-Sánchez, M.; De La Escosura-Muñiz, A.; Merkoçi, A. Simple paper architecture modifications lead to enhanced sensitivity in nanoparticle based lateral flow immunoassays. Lab Chip 2013, 13, 386–390. [Google Scholar] [CrossRef] [PubMed]
- Garnier, G.; Then, W.L. Paper microfluidics: Applications and perspectives. In Proceedings of the 15th Fundamental Research Symposium, Cambridge, UK, 8–13 September 2013; Advances in Pulp and Paper Research. Fundamental Research Committee: Manchester, UK, 2013; pp. 541–583. [Google Scholar] [CrossRef]
- Hong, S.; Kim, W. Dynamics of water imbibition through paper channels with wax boundaries. Microfluid. Nanofluid. 2015, 19, 845–853. [Google Scholar] [CrossRef]
- Conrath, M.; Fries, N.; Zhang, M.; Dreyer, M.E. Radial capillary transport from an infinite reservoir. Transp. Porous Med. 2010, 84, 109–132. [Google Scholar] [CrossRef]
- Mendez, S.; Fenton, E.M.; Gallegos, G.R.; Petsev, D.N.; Sibbett, S.S.; Stone, H.A.; Zhang, Y.; López, G.P. Imbibition in porous membranes of complex shape: Quasi-stationary flow in thin rectangular segments. Langmuir 2010, 26, 1380–1385. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Hagen, J.A.; Papautsky, I. Paper pump for passive and programmable transport. Biomicrofluidics 2013, 7, 014107. [Google Scholar] [CrossRef]
- Shou, D.; Ye, L.; Fan, J.; Fu, K.; Mei, M.; Wang, H.; Chen, Q. Geometry-induced asymmetric capillary flow. Langmuir 2014, 30, 5448–5454. [Google Scholar] [CrossRef] [PubMed]
- Elizalde, E.; Urteaga, R.; Berli, C.L. Rational design of capillary-driven flows for paper-based microfluidics. Lab Chip 2015, 15, 2173–2180. [Google Scholar] [CrossRef]
- Kumar, S.; Bhushan, P.; Bhattacharya, S. Fluid Transport Mechanisms in Paper-Based Microfluidic Devices. In Paper Microfluidics Theory and Applications; Springer: Singapore, 2019; Chapter 2; pp. 7–28. [Google Scholar]
- Zhong, Z.; Wu, R.; Wang, Z.; Tan, H. An investigation of paper based microfluidic devices for size based separation and extraction applications. J. Chromatogr. B 2015, 1000, 41–48. [Google Scholar] [CrossRef]
- Garra, R.; Salusti, E. Application of the nonlocal Darcy law to the propagation of nonlinear thermoelastic waves in fluid saturated porous media. Phys. D Nonlinear Phenom. 2013, 250, 52–57. [Google Scholar] [CrossRef]
- Mambatta, A.K.; Jayarajan, J.; Rashme, V.L.; Harini, S.; Menon, S.; Kuppusamy, J. Reliability of dipstick assay in predicting urinary tract infection. J. Family Med. Prim. Care 2015, 4, 265–268. [Google Scholar] [CrossRef]
- Dadzie, I.; Quansah, E.; Puopelle Dakorah, M.; Abiade, V.; Takyi-Amuah, E.; Adusei, R. The effectiveness of dipstick for the detection of urinary tract infection. Can. J. Infect. Dis. Med. Microbiol. 2019, 2019, 8642628. [Google Scholar] [CrossRef] [PubMed]
- Renault, C.; Anderson, M.J.; Crooks, R.M. Electrochemistry in hollow-channel paper analytical devices. J. Am. Chem. Soc. 2014, 136, 4616–4623. [Google Scholar] [CrossRef] [PubMed]
- Carrell, C.; Kava, A.; Nguyen, M.; Menger, R.; Munshi, Z.; Call, Z.; Nussbaum, M.; Henry, C. Beyond the lateral flow assay: A review of paper-based microfluidics. Microelectron. Eng. 2019, 206, 45–54. [Google Scholar] [CrossRef]
- Noviana, E.; Ozer, T.; Carrell, C.S.; Link, J.S.; McMahon, C.; Jang, I.; Henry, C.S. Microfluidic paper-based analytical devices: From design to applications. Chem. Rev. 2021, 121, 11835–11885. [Google Scholar] [CrossRef] [PubMed]
- Peto, T.; Affron, D.; Afrough, B.; Agasu, A.; Ainsworth, M.; Allanson, A.; Allen, K.; Allen, C.; Archer, L.; Ashbridge, N.; et al. COVID-19: Rapid antigen detection for SARS-CoV-2 by lateral flow assay: A national systematic evaluation of sensitivity and specificity for mass-testing. EClinicalMedicine 2021, 36, 100924. [Google Scholar] [CrossRef] [PubMed]
- Davies, B.; Araghi, M.; Moshe, M.; Gao, H.; Bennet, K.; Jenkins, J.; Atchison, C.; Darzi, A.; Ashby, D.; Riley, S.; et al. Acceptability, usability, and performance of lateral flow immunoassay tests for severe acute respiratory syndrome coronavirus 2 antibodies: REACT-2 study of self-testing in nonhealthcare key workers. Open Forum Infect. Dis. 2021, 8, ofab496. [Google Scholar] [CrossRef] [PubMed]
- Budd, J.; Miller, B.S.; Weckman, N.E.; Cherkaoui, D.; Huang, D.; Decruz, A.T.; Fongwen, N.; Han, G.R.; Broto, M.; Estcourt, C.S.; et al. Lateral flow test engineering and lessons learned from COVID-19. Nat. Rev. Bioeng. 2023, 1, 13–31. [Google Scholar] [CrossRef]
- Mahmud, M.A.; Blondeel, E.J.; Kaddoura, M.; MacDonald, B.D. Features in microfluidic paper-based devices made by laser cutting: How small can they be? Micromachines 2018, 9, 220. [Google Scholar] [CrossRef]
- Martinez, A.W.; Phillips, S.T.; Butte, M.J.; Whitesides, G.M. Patterned paper as a platform for inexpensive, low-volume, portable bioassays. Angew. Chem. 2007, 119, 1340–1342. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Gao, Q.; Wu, W.B.; Nie, J.; Fu, J.Z. 3D printed paper-based microfluidic analytical devices. Micromachines 2016, 7, 108. [Google Scholar] [CrossRef]
- Carrilho, E.; Martinez, A.W.; Whitesides, G.M. Understanding wax printing: A simple micropatterning process for paper-based microfluidics. Anal. Chem. 2009, 81, 7091–7095. [Google Scholar] [CrossRef]
- Wang, Y.; Sun, S.; Luo, J.; Xiong, Y.; Ming, T.; Liu, J.; Ma, Y.; Yan, S.; Yang, Y.; Yang, Z.; et al. Low sample volume origami-paper-based graphene-modified aptasensors for label-free electrochemical detection of cancer biomarker-EGFR. Microsyst. Nanoeng. 2020, 6, 32. [Google Scholar] [CrossRef]
- Gao, B.; Chi, J.; Liu, H.; Gu, Z. Vertical paper analytical devices fabricated using the principles of quilling and kirigami. Sci. Rep. 2017, 7, 7255. [Google Scholar] [CrossRef] [PubMed]
- Johnston, M. The Book of Paper Quilling: Techniques & Projects for Paper Filigree; Sterling Publishing Company, Inc.: New York, NY, USA, 1995. [Google Scholar]
- Fenton, E.M.; Mascarenas, M.R.; López, G.P.; Sibbett, S.S. Multiplex lateral-flow test strips fabricated by two-dimensional shaping. ACS Appl. Mater. Interfaces 2009, 1, 124–129. [Google Scholar] [CrossRef] [PubMed]
- Nie, J.; Zhang, Y.; Lin, L.; Zhou, C.; Li, S.; Zhang, L.; Li, J. Low-cost fabrication of paper-based microfluidic devices by one-step plotting. Anal. Chem. 2012, 84, 6331–6335. [Google Scholar] [CrossRef] [PubMed]
- Mahmud, M.A.; Blondeel, E.J.; Kaddoura, M.; MacDonald, B.D. Creating compact and microscale features in paper-based devices by laser cutting. Analyst 2016, 141, 6449–6454. [Google Scholar] [CrossRef]
- de Araujo, W.R.; Frasson, C.M.; Ameku, W.A.; Silva, J.R.; Angnes, L.; Paixão, T.R. Single-step reagentless laser scribing fabrication of electrochemical paper-based analytical devices. Angew. Chem. 2017, 129, 15309–15313. [Google Scholar] [CrossRef]
- Theillet, G.; Rubens, A.; Foucault, F.; Dalbon, P.; Rozand, C.; Leparc-Goffart, I.; Bedin, F. Laser-cut paper-based device for the detection of dengue non-structural NS1 protein and specific IgM in human samples. Arch. Virol. 2018, 163, 1757–1767. [Google Scholar] [CrossRef]
- Zhang, R.; Cai, S.; Wu, Q.; Zhu, Y.; Yin, X.; Xu, Y.; Yang, Y.; Chang, H. Laser cutting assisted fabrication of assembled solid-state supercapacitors based on polypyrrole coated paper. J. Electroanal. Chem. 2022, 919, 116522. [Google Scholar] [CrossRef]
- Li, X.; Su, D.; Gu, Y.; Zhang, J.; Li, S.; Xiao, Y.; He, J.; Wang, W.; Li, D. Laser fabrication of epidermal paper-based graphene sensors. Appl. Mater. Today 2024, 36, 102051. [Google Scholar] [CrossRef]
- Martinez, A.W.; Phillips, S.T.; Wiley, B.J.; Gupta, M.; Whitesides, G.M. FLASH: A rapid method for prototyping paper-based microfluidic devices. Lab Chip 2008, 8, 2146–2150. [Google Scholar] [CrossRef] [PubMed]
- Kakoti, A.; Siddiqui, M.F.; Goswami, P. A low cost design and fabrication method for developing a leak proof paper based microfluidic device with customized test zone. Biomicrofluidics 2015, 9, 026502. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Shi, Z.Z. Microfluidic paper-based analytical devices fabricated by low-cost photolithography and embossing of Parafilm®. Lab Chip 2015, 15, 1642–1645. [Google Scholar] [CrossRef]
- Park, C.; Han, Y.D.; Kim, H.V.; Lee, J.; Yoon, H.C.; Park, S. Double-sided 3D printing on paper towards mass production of three-dimensional paper-based microfluidic analytical devices (3D-μPADs). Lab Chip 2018, 18, 1533–1538. [Google Scholar] [CrossRef] [PubMed]
- Fu, X.; Xia, B.; Ji, B.; Lei, S.; Zhou, Y. Flow controllable three-dimensional paper-based microfluidic analytical devices fabricated by 3D printing technology. Anal. Chim. Acta 2019, 1065, 64–70. [Google Scholar] [CrossRef]
- He, Y.; Wu, W.b.; Fu, J.z. Rapid fabrication of paper-based microfluidic analytical devices with desktop stereolithography 3D printer. RSC Adv. 2015, 5, 2694–2701. [Google Scholar] [CrossRef]
- Puneeth, S.; Salve, M.; Akshatha, R.; Goel, S. Realization of microfluidic paper-based analytical devices using a 3-D printer: Characterization and optimization. IEEE Trans. Device Mater. Reliab. 2019, 19, 529–536. [Google Scholar] [CrossRef]
- Chiang, C.K.; Kurniawan, A.; Kao, C.Y.; Wang, M.J. Single step and mask-free 3D wax printing of microfluidic paper-based analytical devices for glucose and nitrite assays. Talanta 2019, 194, 837–845. [Google Scholar] [CrossRef]
- Silva-Neto, H.A.; Duarte-Junior, G.F.; Rocha, D.S.; Bedioui, F.; Varenne, A.; Coltro, W.K. Recycling 3D Printed Residues for the Development of Disposable Paper-Based Electrochemical Sensors. ACS Appl. Mater. Interfaces 2023, 15, 14111–14121. [Google Scholar] [CrossRef] [PubMed]
- Zaki, M.F.; Wu, Y.H.; Chen, P.C.; Chen, P.S. Determination of psychoactive substances in one microliter plasma using a novel 3D printing microfluidic paper-based column coupled to liquid chromatography-mass spectrometry. Sens. Actuators B Chem. 2023, 393, 134243. [Google Scholar] [CrossRef]
- Dungchai, W.; Chailapakul, O.; Henry, C.S. A low-cost, simple, and rapid fabrication method for paper-based microfluidics using wax screen-printing. Analyst 2011, 136, 77–82. [Google Scholar] [CrossRef] [PubMed]
- Sameenoi, Y.; Nongkai, P.N.; Nouanthavong, S.; Henry, C.S.; Nacapricha, D. One-step polymer screen-printing for microfluidic paper-based analytical device (μPAD) fabrication. Analyst 2014, 139, 6580–6588. [Google Scholar] [CrossRef] [PubMed]
- Lamas-Ardisana, P.J.; Casuso, P.; Fernandez-Gauna, I.; Martínez-Paredes, G.; Jubete, E.; Añorga, L.; Cabañero, G.; Grande, H.J. Disposable electrochemical paper-based devices fully fabricated by screen-printing technique. Electrochem. Commun. 2017, 75, 25–28. [Google Scholar] [CrossRef]
- Tasaengtong, B.; Sameenoi, Y. A one-step polymer screen-printing method for fabrication of microfluidic cloth-based analytical devices. Microchem. J. 2020, 158, 105078. [Google Scholar] [CrossRef]
- Mettakoonpitak, J.; Khongsoun, K.; Wongwan, N.; Kaewbutdee, S.; Siripinyanond, A.; Kuharuk, A.; Henry, C.S. Simple biodegradable plastic screen-printing for microfluidic paper-based analytical devices. Sens. Actuators B Chem. 2021, 331, 129463. [Google Scholar] [CrossRef]
- Xiong, C.; Li, M.; Han, Q.; Zhao, W.; Dai, L.; Ni, Y. Screen printing fabricating patterned and customized full paper-based energy storage devices with excellent photothermal, self-healing, high energy density and good electromagnetic shielding performances. J. Mater. Sci. Technol. 2022, 97, 190–200. [Google Scholar] [CrossRef]
- Oliveira, A.E.; Pereira, A.C.; de Resende, M.A. Fabrication of Low-cost Screen-printed Electrode in Paper Using Conductive Inks of Graphite and Silver/Silver Chloride. Electroanalysis 2023, 35, e202200093. [Google Scholar] [CrossRef]
- Liu, J.; Kong, X.; Wang, H.; Zhang, Y.; Fan, Y. Roll-to-roll wax transfer for rapid and batch fabrication of paper-based microfluidics. Microfluid. Nanofluidics 2020, 24, 6. [Google Scholar] [CrossRef]
- Walia, S.; Bhatnagar, I.; Liu, J.; Mitra, S.K.; Asthana, A. A novel method for fabrication of paper-based microfluidic devices using BSA-ink. Int. J. Biol. Macromol. 2021, 193, 1617–1622. [Google Scholar] [CrossRef] [PubMed]
- Espinosa, A.; Diaz, J.; Vazquez, E.; Acosta, L.; Santiago, A.; Cunci, L. Fabrication of paper-based microfluidic devices using a 3D printer and a commercially-available wax filament. Talanta Open 2022, 6, 100142. [Google Scholar] [CrossRef] [PubMed]
- Tran, B.T.; Rijiravanich, P.; Puttaraksa, N.; Surareungchai, W. Wax gates in laminated microfluidic paper-based immunosensors. Microchem. J. 2022, 178, 107343. [Google Scholar] [CrossRef]
- Brito-Pereira, R.; Ribeiro, C.; Costa, P.; Correia, V.; Cardoso, V.F.; Lanceros-Mendez, S. Graphene Based Printable Conductive Wax for Low-Power Thermal Actuation in Microfluidic Paper-Based Analytical Devices. Adv. Mater. Technol. 2023, 8, 2300051. [Google Scholar] [CrossRef]
- Monju, T.; Hirakawa, M.; Kuboyama, S.; Saiki, R.; Ishida, A. A fabrication technique for paper-based analytical devices via two-sided patterning with thermal-transfer printer and laminator. Sens. Actuators B Chem. 2023, 375, 132886. [Google Scholar] [CrossRef]
- Yamada, K.; Henares, T.G.; Suzuki, K.; Citterio, D. Paper-based inkjet-printed microfluidic analytical devices. Angew. Chem. Int. Ed. 2015, 54, 5294–5310. [Google Scholar] [CrossRef]
- Paschoalino, W.J.; Kogikoski Jr, S.; Barragan, J.T.; Giarola, J.F.; Cantelli, L.; Rabelo, T.M.; Pessanha, T.M.; Kubota, L.T. Emerging considerations for the future development of electrochemical paper-based analytical devices. ChemElectroChem 2019, 6, 10–30. [Google Scholar] [CrossRef]
- Ahmad, M.; Costa Angeli, M.A.; Ibba, P.; Vasquez, S.; Shkodra, B.; Lugli, P.; Petti, L. Paper-Based Printed Antenna: Investigation of Process-Induced and Climatic-Induced Performance Variability. Adv. Eng. Mater. 2023, 25, 2201703. [Google Scholar] [CrossRef]
- Galliani, M.; Ferrari, L.M.; Bouet, G.; Eglin, D.; Ismailova, E. Tailoring inkjet-printed PEDOT: PSS composition toward green, wearable device fabrication. APL Bioeng. 2023, 7, 016101. [Google Scholar] [CrossRef]
- Le, N.N.; Dinh, D.M.T.; Lam, P.H.; Le, A.V.T.; Le, M.T.; Pham, M.D.; Dang, D.M.T.; Dang, C.M. Fabrication of microfluidic paper-based channels by inkjet printing process for analytical applications. Adv. Nat. Sci. Nanosci. Nanotechnol. 2023, 14, 015015. [Google Scholar] [CrossRef]
- Ray, A.; Mohan, J.M.; Amreen, K.; Dubey, S.K.; Javed, A.; Ponnalagu, R.; Goel, S. Ink-jet-printed CuO nanoparticle-enhanced miniaturized paper-based electrochemical platform for hypochlorite sensing. Appl. Nanosci. 2023, 13, 1855–1861. [Google Scholar] [CrossRef]
- Silvestri, A.; Vázquez-Díaz, S.; Misia, G.; Poletti, F.; López-Domene, R.; Pavlov, V.; Zanardi, C.; Cortajarena, A.L.; Prato, M. An Electroactive and Self-Assembling Bio-Ink, based on Protein-Stabilized Nanoclusters and Graphene, for the Manufacture of Fully Inkjet-Printed Paper-Based Analytical Devices. Small 2023, 19, 2300163. [Google Scholar] [CrossRef] [PubMed]
- de Tarso Garcia, P.; Cardoso, T.M.G.; Garcia, C.D.; Carrilho, E.; Coltro, W.K.T. A handheld stamping process to fabricate microfluidic paper-based analytical devices with chemically modified surface for clinical assays. RSC Adv. 2014, 4, 37637–37644. [Google Scholar] [CrossRef]
- He, Y.; Wu, Y.; Xiao, X.; Fu, J.; Xue, G. A low-cost and rapid microfluidic paper-based analytical device fabrication method: Flash foam stamp lithography. RSC Adv. 2014, 4, 63860–63865. [Google Scholar] [CrossRef]
- Guan, Y.; Sun, B. Detection and extraction of heavy metal ions using paper-based analytical devices fabricated via atom stamp printing. Microsyst. Nanoeng. 2020, 6, 14. [Google Scholar] [CrossRef]
- de Araujo, T.A.; de Moraes, N.C.; Petroni, J.M.; Ferreira, V.S.; Lucca, B.G. Simple, fast, and instrumentless fabrication of paper analytical devices by novel contact stamping method based on acrylic varnish and 3D printing. Microchim. Acta 2021, 188, 437. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Lee, C.; Jeon, K.; Lee, J.Y.; Kim, Y.J.; Lee, J.G.; Kim, H.; Cho, M.; Kim, D.N. Harnessing a paper-folding mechanism for reconfigurable DNA origami. Nature 2023, 619, 78–86. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.A.; Yeh, W.S.; Tsai, T.T.; Chen, C.F. Three-dimensional origami paper-based device for portable immunoassay applications. Lab Chip 2019, 19, 598–607. [Google Scholar] [CrossRef] [PubMed]
- Tian, T.; An, Y.; Wu, Y.; Song, Y.; Zhu, Z.; Yang, C. Integrated distance-based origami paper analytical device for one-step visualized analysis. ACS Appl. Mater. Interfaces 2017, 9, 30480–30487. [Google Scholar] [CrossRef]
- Ding, J.; Li, B.; Chen, L.; Qin, W. A three-dimensional Origami paper-based device for potentiometric biosensing. Angew. Chem. Int. Ed. 2016, 55, 13033–13037. [Google Scholar] [CrossRef]
- Scida, K.; Li, B.; Ellington, A.D.; Crooks, R.M. DNA detection using origami paper analytical devices. Anal. Chem. 2013, 85, 9713–9720. [Google Scholar] [CrossRef]
- Liu, H.; Xiang, Y.; Lu, Y.; Crooks, R.M. Aptamer-based origami paper analytical device for electrochemical detection of adenosine. Angew. Chem. 2012, 124, 7031–7034. [Google Scholar] [CrossRef]
- Liu, X.; Sun, J.; Tong, Y.; Zhang, M.; Wang, X.; Guo, S.; Han, X.; Zhao, X.; Tang, Q.; Liu, Y. Calligraphy and Kirigami/Origami-Inspired All-Paper Touch–Temperature Sensor with Stimulus Discriminability. ACS Appl. Mater. Interfaces 2023, 15, 1726–1735. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Li, Y.; Wang, X.; Yu, H. Origami paper-based stretchable humidity sensor for textile-attachable wearable electronics. ACS Appl. Mater. Interfaces 2022, 14, 36227–36237. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Bhushan, P.; Krishna, V.; Bhattacharya, S. Tapered lateral flow immunoassay based point-of-care diagnostic device for ultrasensitive colorimetric detection of dengue NS1. Biomicrofluidics 2018, 12, 034104. [Google Scholar] [CrossRef] [PubMed]
- Assi, N.; Rypar, T.; Macka, M.; Adam, V.; Vaculovicova, M. Microfluidic paper-based fluorescence sensor for L-homocysteine using a molecularly imprinted polymer and in situ-formed fluorescent quantum dots. Talanta 2023, 255, 124185. [Google Scholar] [CrossRef]
- Goncharov, A.; Joung, H.A.; Ghosh, R.; Han, G.R.; Ballard, Z.S.; Maloney, Q.; Bell, A.; Aung, C.T.Z.; Garner, O.B.; Carlo, D.D.; et al. Deep Learning-Enabled Multiplexed Point-of-Care Sensor using a Paper-Based Fluorescence Vertical Flow Assay. Small 2023, 19, 2300617. [Google Scholar] [CrossRef]
- Ren, Y.; Cao, L.; Zhang, X.; Jiao, R.; Ou, D.; Wang, Y.; Zhang, D.; Shen, Y.; Ling, N.; Ye, Y. A novel fluorescence resonance energy transfer (FRET)-based paper sensor with smartphone for quantitative detection of Vibrio parahaemolyticus. Food Control 2023, 145, 109412. [Google Scholar] [CrossRef]
- Tong, X.; Cai, G.; Xie, L.; Wang, T.; Zhu, Y.; Peng, Y.; Tong, C.; Shi, S.; Guo, Y. Threaded 3D microfluidic paper analytical device-based ratiometric fluorescent sensor for background-free and visual detection of organophosphorus pesticides. Biosens. Bioelectron. 2023, 222, 114981. [Google Scholar] [CrossRef]
- Yuan, M.; Li, C.; Zheng, Y.; Cao, H.; Ye, T.; Wu, X.; Hao, L.; Yin, F.; Yu, J.; Xu, F. A portable multi-channel fluorescent paper-based microfluidic chip based on smartphone imaging for simultaneous detection of four heavy metals. Talanta 2024, 266, 125112. [Google Scholar] [CrossRef]
- Liu, W.; Guo, Y.; Li, H.; Zhao, M.; Lai, Z.; Li, B. A paper-based chemiluminescence device for the determination of ofloxacin. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2015, 137, 1298–1303. [Google Scholar] [CrossRef]
- Liu, D.; Ju, C.; Han, C.; Shi, R.; Chen, X.; Duan, D.; Yan, J.; Yan, X. Nanozyme chemiluminescence paper test for rapid and sensitive detection of SARS-CoV-2 antigen. Biosens. Bioelectron. 2021, 173, 112817. [Google Scholar] [CrossRef] [PubMed]
- Al Yahyai, I.; Hassanzadeh, J.; Al-Lawati, H.A. A novel and selective multi-emission chemiluminescence system for the quantification of deltamethrin in food samples. Sens. Actuators B Chem. 2021, 327, 128927. [Google Scholar] [CrossRef]
- Delaney, J.L.; Hogan, C.F.; Tian, J.; Shen, W. Electrogenerated chemiluminescence detection in paper-based microfluidic sensors. Anal. Chem. 2011, 83, 1300–1306. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Zhou, X.; Liu, W.; Yang, X.; Xing, D. Paper-based bipolar electrode electrochemiluminescence switch for label-free and sensitive genetic detection of pathogenic bacteria. Anal. Chem. 2016, 88, 10191–10197. [Google Scholar] [CrossRef] [PubMed]
- Valentine, C.J.; Takagishi, K.; Umezu, S.; Daly, R.; De Volder, M. Paper-based electrochemical sensors using paper as a scaffold to create porous carbon nanotube electrodes. ACS Appl. Mater. Interfaces 2020, 12, 30680–30685. [Google Scholar] [CrossRef] [PubMed]
- He, Z.; Huang, J.; Shen, W.; Lei, X.; Zhang, Y.; Zhu, L.; Shen, X.; Zhang, D.; Yu, D.; Zhou, M. A Paper-Based Fluorescent Sensor for Rapid Early Screening of Oral Squamous Cell Carcinoma. ACS Appl. Mater. Interfaces 2023, 15, 24913–24922. [Google Scholar] [CrossRef]
- Li, Z.; Zhu, M.; Li, F.; Li, Z.; Zhao, A.; Haghighatbin, M.A.; Cui, H. Microfluidic paper chip based multicolor chemiluminescence sensing strategy for discrimination of antioxidants. Sens. Actuators B Chem. 2023, 393, 134166. [Google Scholar] [CrossRef]
- Wang, F.; Liu, Y.; Fu, C.; Li, N.; Du, M.; Zhang, L.; Ge, S.; Yu, J. Paper-based bipolar electrode electrochemiluminescence platform for detection of multiple miRNAs. Anal. Chem. 2020, 93, 1702–1708. [Google Scholar] [CrossRef]
- Tay, L.L.; Poirier, S.; Ghaemi, A.; Hulse, J.; Wang, S. Paper-based surface-enhanced Raman spectroscopy sensors for field applications. J. Raman Spectrosc. 2021, 52, 563–572. [Google Scholar] [CrossRef]
- Eskandari, V.; Sahbafar, H.; Karooby, E.; Heris, M.H.; Mehmandoust, S.; Razmjoue, D.; Hadi, A. Surface-Enhanced Raman scattering (SERS) filter paper substrates decorated with silver nanoparticles for the detection of molecular vibrations of Acyclovir drug. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2023, 298, 122762. [Google Scholar] [CrossRef] [PubMed]
- Cate, D.M.; Adkins, J.A.; Mettakoonpitak, J.; Henry, C.S. Recent developments in paper-based microfluidic devices. Anal. Chem. 2015, 87, 19–41. [Google Scholar] [CrossRef] [PubMed]
- Magro, L.; Escadafal, C.; Garneret, P.; Jacquelin, B.; Kwasiborski, A.; Manuguerra, J.C.; Monti, F.; Sakuntabhai, A.; Vanhomwegen, J.; Lafaye, P.; et al. Paper microfluidics for nucleic acid amplification testing (NAAT) of infectious diseases. Lab Chip 2017, 17, 2347–2371. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Macdonald, J. Multiplexed lateral flow biosensors: Technological advances for radically improving point-of-care diagnoses. Biosens. Bioelectron. 2016, 83, 177–192. [Google Scholar] [CrossRef]
- Arias-Alpízar, K.; Sánchez-Cano, A.; Prat-Trunas, J.; de la Serna Serna, E.; Alonso, O.; Sulleiro, E.; Sánchez-Montalvá, A.; Diéguez, A.; Baldrich, E. Malaria quantitative POC testing using magnetic particles, a paper microfluidic device and a hand-held fluorescence reader. Biosens. Bioelectron. 2022, 215, 114513. [Google Scholar] [CrossRef] [PubMed]
- Prat-Trunas, J.; Arias-Alpizar, K.; Álvarez-Carulla, A.; Orio-Tejada, J.; Molina, I.; Sánchez-Montalvá, A.; Colomer-Farrarons, J.; Del Campo, F.; Miribel-Català, P.L.; Baldrich, E. Paper-based microfluidic electro-analytical device (PMED) for magneto-assay automation: Towards generic point-of-care diagnostic devices. Biosens. Bioelectron. 2024, 246, 115875. [Google Scholar] [CrossRef] [PubMed]
- Biswas, P.; Mukunthan Sulochana, G.N.; Banuprasad, T.N.; Goyal, P.; Modak, D.; Ghosh, A.K.; Chakraborty, S. All-Serotype Dengue Virus Detection through Multilayered Origami-Based Paper/Polymer Microfluidics. ACS Sens. 2022, 7, 3720–3729. [Google Scholar] [CrossRef] [PubMed]
- Le, T.N.; Hsiao, W.W.W.; Cheng, Y.Y.; Lee, C.C.; Huynh, T.T.; Pham, D.M.; Chen, M.; Jen, M.W.; Chang, H.C.; Chiang, W.H. Spin-Enhanced Lateral Flow Immunoassay for High-Sensitivity Detection of Nonstructural Protein NS1 Serotypes of the Dengue Virus. Anal. Chem. 2022, 94, 17819–17826. [Google Scholar] [CrossRef]
- Trakoolwilaiwan, T.; Takeuchi, Y.; Leung, T.S.; Sebek, M.; Storozhuk, L.; Nguyen, L.; Thanh, N.T.K. Development of a thermochromic lateral flow assay to improve sensitivity for dengue virus serotype 2 NS1 detection. Nanoscale 2023, 15, 12915–12925. [Google Scholar] [CrossRef]
- Kaarj, K.; Akarapipad, P.; Yoon, J.Y. Simpler, faster, and sensitive Zika virus assay using smartphone detection of loop-mediated isothermal amplification on paper microfluidic chips. Sci. Rep. 2018, 8, 12438. [Google Scholar] [CrossRef]
- Seok, Y.; Batule, B.S.; Kim, M.G. Lab-on-paper for all-in-one molecular diagnostics (LAMDA) of zika, dengue, and chikungunya virus from human serum. Biosens. Bioelectron. 2020, 165, 112400. [Google Scholar] [CrossRef]
- Suvanasuthi, R.; Chimnaronk, S.; Promptmas, C. 3D printed hydrophobic barriers in a paper-based biosensor for point-of-care detection of dengue virus serotypes. Talanta 2022, 237, 122962. [Google Scholar] [CrossRef] [PubMed]
- Karlikow, M.; da Silva, S.J.R.; Guo, Y.; Cicek, S.; Krokovsky, L.; Homme, P.; Xiong, Y.; Xu, T.; Calderón-Peláez, M.A.; Camacho-Ortega, S.; et al. Field validation of the performance of paper-based tests for the detection of the Zika and chikungunya viruses in serum samples. Nat. Biomed. Eng. 2022, 6, 246–256. [Google Scholar] [CrossRef] [PubMed]
- Gong, H.; Tang, L.; Chen, C.; Chen, F.; Cai, C. Portable paper-based molecularly imprinted sensor for visual real-time detection of influenza virus H5N1. Chem. Eng. J. 2023, 477, 146990. [Google Scholar] [CrossRef]
- Tavakoli, H.; Hirth, E.; Luo, M.; Timilsina, S.S.; Dou, M.; Dominguez, D.C.; Li, X. A microfluidic fully paper-based analytical device integrated with loop-mediated isothermal amplification and nano-biosensors for rapid, sensitive, and specific quantitative detection of infectious diseases. Lab Chip 2022, 22, 4693–4704. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.R.; Hu, J.; Tang, R.; Gong, Y.; Feng, S.; Ren, H.; Wen, T.; Li, X.; Abas, W.A.B.W.; Pingguan-Murphy, B.; et al. An integrated paper-based sample-to-answer biosensor for nucleic acid testing at the point of care. Lab Chip 2016, 16, 611–621. [Google Scholar] [CrossRef] [PubMed]
- Tang, R.; Yang, H.; Gong, Y.; You, M.; Liu, Z.; Choi, J.R.; Wen, T.; Qu, Z.; Mei, Q.; Xu, F. A fully disposable and integrated paper-based device for nucleic acid extraction, amplification and detection. Lab Chip 2017, 17, 1270–1279. [Google Scholar] [CrossRef]
- Natarajan, S.; Su, F.; Jayaraj, J.; Shah, M.I.I.; Huang, Y. A paper microfluidics-based fluorescent lateral flow immunoassay for point-of-care diagnostics of non-communicable diseases. Analyst 2019, 144, 6291–6303. [Google Scholar] [CrossRef] [PubMed]
- Shalaby, A.A.; Tsao, C.W.; Ishida, A.; Maeki, M.; Tokeshi, M. Microfluidic paper-based analytical devices for cancer diagnosis. Sens. Actuators B Chem. 2023, 379, 133243. [Google Scholar] [CrossRef]
- Sana, T.; Sharma, P.; Khanuja, M.; Narang, J. Detection of Uterine Cancer Biomarker EGFR through an Aptasensor Utilizing a Carbon Electrode Modified with Silver Nanowires. Mater. Chem. Phys. 2024, 319, 129412. [Google Scholar] [CrossRef]
- Gutiérrez-Capitán, M.; Sanchís, A.; Carvalho, E.O.; Baldi, A.; Vilaplana, L.; Cardoso, V.F.; Calleja, Á.; Wei, M.; de la Rica, R.; Hoyo, J.; et al. Engineering a Point-of-Care Paper-Microfluidic Electrochemical Device Applied to the Multiplexed Quantitative Detection of Biomarkers in Sputum. ACS Sens. 2023, 8, 3032–3042. [Google Scholar] [CrossRef] [PubMed]
- Rohrman, B.A.; Richards-Kortum, R.R. A paper and plastic device for performing recombinase polymerase amplification of HIV DNA. Lab Chip 2012, 12, 3082–3088. [Google Scholar] [CrossRef] [PubMed]
- Fu, X.; Cheng, Z.; Yu, J.; Choo, P.; Chen, L.; Choo, J. A SERS-based lateral flow assay biosensor for highly sensitive detection of HIV-1 DNA. Biosens. Bioelectron. 2016, 78, 530–537. [Google Scholar] [CrossRef] [PubMed]
- Tsao, Y.T.; Yang, C.Y.; Wen, Y.C.; Chang, T.C.; Matsuura, K.; Chen, Y.; Cheng, C.M. Point-of-care semen analysis of patients with infertility via smartphone and colorimetric paper-based diagnostic device. Bioeng. Transl. Med. 2021, 6, e10176. [Google Scholar] [CrossRef] [PubMed]
- Sarabi, M.R.; Yigci, D.; Alseed, M.M.; Mathyk, B.A.; Ata, B.; Halicigil, C.; Tasoglu, S. Disposable paper-based microfluidics for fertility testing. iScience 2022, 25, 104986. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Bhushan, P.; Bhattacharya, S. Development of a paper-based analytical device for colorimetric detection of uric acid using gold nanoparticles–graphene oxide (AuNPs–GO) conjugates. Anal. Methods 2016, 8, 6965–6973. [Google Scholar] [CrossRef]
- Kumar, S.; Bhushan, P.; Bhattacharya, S. Facile synthesis of Au@ Ag–hemin decorated reduced graphene oxide sheets: A novel peroxidase mimetic for ultrasensitive colorimetric detection of hydrogen peroxide and glucose. RSC Adv. 2017, 7, 37568–37577. [Google Scholar] [CrossRef]
- Pérez-Rodríguez, M.; del Pilar Cañizares-Macías, M. A prototype microfluidic paper-based chromatic device for simultaneous determination of copper (II) and zinc (II) in urine. Talanta Open 2023, 7, 100178. [Google Scholar] [CrossRef]
- Bezdekova, J.; Plevova, M.; Nejdl, L.; Macka, M.; Masarik, M.; Pacik, D.; Adam, V.; Vaculovicova, M. Prostate cancer diagnosed and staged using UV-irradiated urine samples and a paper-based analytical device. Sens. Actuators B Chem. 2024, 403, 135146. [Google Scholar] [CrossRef]
- Chaiyo, S.; Kunpatee, K.; Kalcher, K.; Yakoh, A.; Pungjunun, K. 3D Paper-Based Device Integrated with a Battery-Less NFC Potentiostat for Nonenzymatic Detection of Cholesterol. ACS Meas. Sci. Au 2024. [Google Scholar] [CrossRef]
- Ozer, T.; Henry, C.S. Paper-based analytical devices for virus detection: Recent strategies for current and future pandemics. TrAC Trends Anal. Chem. 2021, 144, 116424. [Google Scholar] [CrossRef] [PubMed]
- Adrover-Jaume, C.; Alba-Patino, A.; Clemente, A.; Santopolo, G.; Vaquer, A.; Russell, S.M.; Baron, E.; Del Campo, M.D.M.G.; Ferrer, J.M.; Berman-Riu, M.; et al. Paper biosensors for detecting elevated IL-6 levels in blood and respiratory samples from COVID-19 patients. Sens. Actuators B Chem. 2021, 330, 129333. [Google Scholar] [CrossRef]
- Song, Q.; Sun, X.; Dai, Z.; Gao, Y.; Gong, X.; Zhou, B.; Wu, J.; Wen, W. Point-of-care testing detection methods for COVID-19. Lab Chip 2021, 21, 1634–1660. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Deng, R.; Wang, Y.; Wu, C.; Zhang, K.; Wang, C.; Gong, N.; Ledesma-Amaro, R.; Teng, X.; Yang, C.; et al. A paper-based assay for the colorimetric detection of SARS-CoV-2 variants at single-nucleotide resolution. Nat. Biomed. Eng. 2022, 6, 957–967. [Google Scholar] [CrossRef] [PubMed]
- Fabiani, L.; Mazzaracchio, V.; Moscone, D.; Fillo, S.; De Santis, R.; Monte, A.; Amatore, D.; Lista, F.; Arduini, F. based immunoassay based on 96-well wax-printed paper plate combined with magnetic beads and colorimetric smartphone-assisted measure for reliable detection of SARS-CoV-2 in saliva. Biosens. Bioelectron. 2022, 200, 113909. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Chen, Z.; Yang, H. Colorimetric detection of SARS-CoV-2 variants with paper-based analytical devices. MedComm–Biomater. Appl. 2023, 2, e35. [Google Scholar] [CrossRef]
- Sen, A.; Masetty, M.; Weerakoon, S.; Morris, C.; Yadav, J.S.; Apewokin, S.; Trannguyen, J.; Broom, M.; Priye, A. Paper-based loop-mediated isothermal amplification and CRISPR integrated platform for on-site nucleic acid testing of pathogens. Biosens. Bioelectron. 2024, 257, 116292. [Google Scholar] [CrossRef] [PubMed]
- Lee, A.S.; Kim, S.M.; Kim, K.R.; Park, C.; Lee, D.G.; Heo, H.R.; Cha, H.J.; Kim, C.S. A colorimetric lateral flow immunoassay based on oriented antibody immobilization for sensitive detection of SARS-CoV-2. Sens. Actuators B Chem. 2023, 379, 133245. [Google Scholar] [CrossRef]
- Jin, H.; Abu-Raya, Y.S.; Haick, H. Advanced materials for health monitoring with skin-based wearable devices. Adv. Healthc. Mater. 2017, 6, 1700024. [Google Scholar] [CrossRef]
- Xu, Y.; Fei, Q.; Page, M.; Zhao, G.; Ling, Y.; Stoll, S.B.; Yan, Z. Paper-based wearable electronics. iScience 2021, 24, 102736. [Google Scholar] [CrossRef]
- Deroco, P.B.; Wachholz Junior, D.; Kubota, L.T. Paper-based Wearable Electrochemical Sensors: A New Generation of Analytical Devices. Electroanalysis 2023, 35, e202200177. [Google Scholar] [CrossRef]
- Bandodkar, A.J.; Hung, V.W.; Jia, W.; Valdés-Ramírez, G.; Windmiller, J.R.; Martinez, A.G.; Ramírez, J.; Chan, G.; Kerman, K.; Wang, J. Tattoo-based potentiometric ion-selective sensors for epidermal pH monitoring. Analyst 2013, 138, 123–128. [Google Scholar] [CrossRef] [PubMed]
- Nassar, J.M.; Mishra, K.; Lau, K.; Aguirre-Pablo, A.A.; Hussain, M.M. Recyclable nonfunctionalized paper-based ultralow-cost wearable health monitoring system. Adv. Mater. Technol. 2017, 2, 1600228. [Google Scholar] [CrossRef]
- Pal, A.; Goswami, D.; Cuellar, H.E.; Castro, B.; Kuang, S.; Martinez, R.V. Early detection and monitoring of chronic wounds using low-cost, omniphobic paper-based smart bandages. Biosens. Bioelectron. 2018, 117, 696–705. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Zhao, G.; Zhu, L.; Fei, Q.; Zhang, Z.; Chen, Z.; An, F.; Chen, Y.; Ling, Y.; Guo, P.; et al. Pencil–paper on-skin electronics. Proc. Natl. Acad. Sci. USA 2020, 117, 18292–18301. [Google Scholar] [CrossRef]
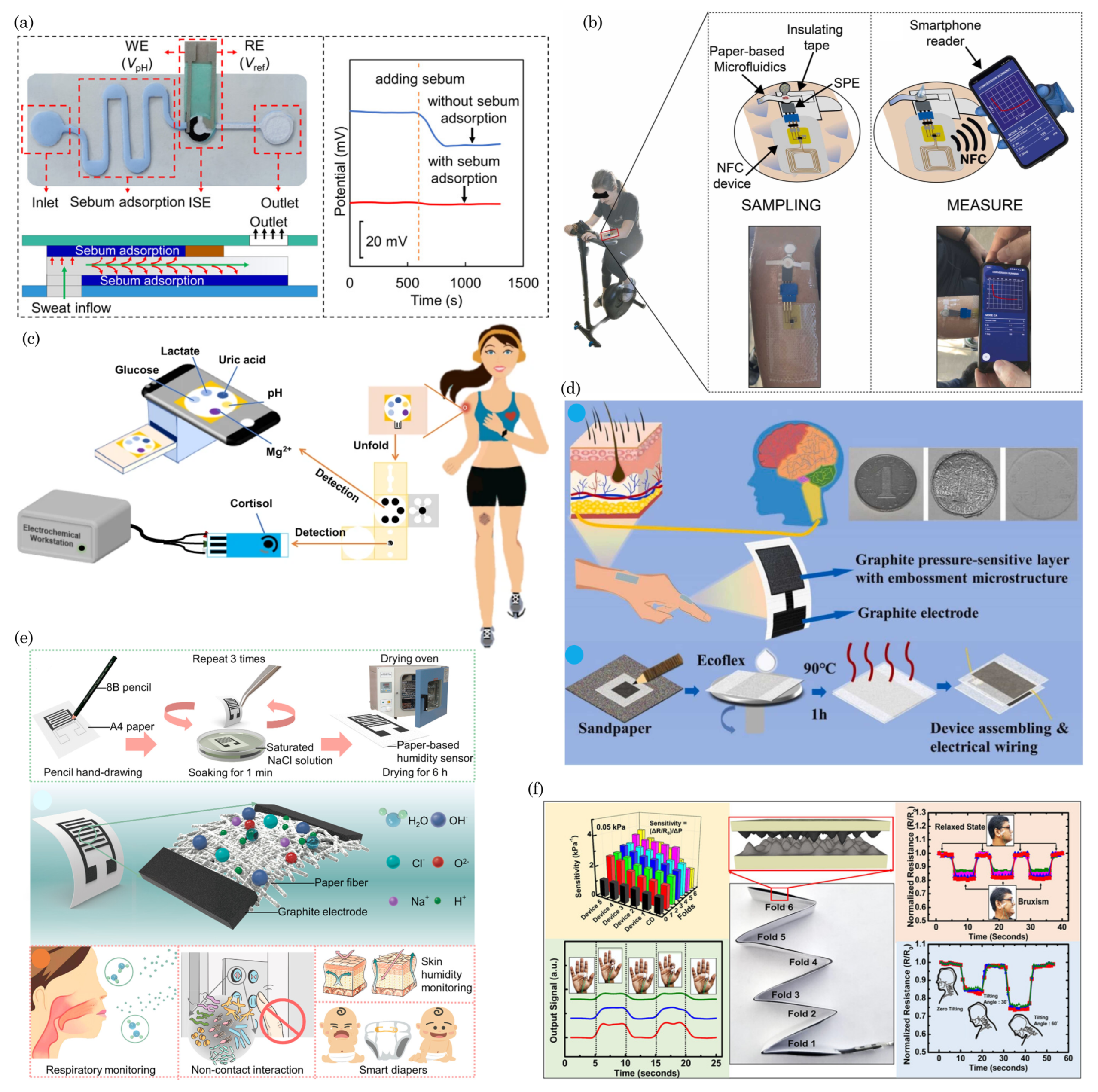
- Mogera, U.; Guo, H.; Namkoong, M.; Rahman, M.S.; Nguyen, T.; Tian, L. Wearable plasmonic paper–based microfluidics for continuous sweat analysis. Sci. Adv. 2022, 8, eabn1736. [Google Scholar] [CrossRef]
- Yang, M.; Sun, N.; Lai, X.; Wu, J.; Wu, L.; Zhao, X.; Feng, L. Paper-Based Sandwich-Structured Wearable Sensor with Sebum Filtering for Continuous Detection of Sweat pH. ACS Sens. 2023, 8, 176–186. [Google Scholar] [CrossRef]
- Fiore, L.; Mazzaracchio, V.; Serani, A.; Fabiani, G.; Fabiani, L.; Volpe, G.; Moscone, D.; Bianco, G.M.; Occhiuzzi, C.; Marrocco, G.; et al. Microfluidic paper-based wearable electrochemical biosensor for reliable cortisol detection in sweat. Sens. Actuators B Chem. 2023, 379, 133258. [Google Scholar] [CrossRef]
- Cheng, Y.; Feng, S.; Ning, Q.; Li, T.; Xu, H.; Sun, Q.; Cui, D.; Wang, K. Dual-signal readout paper-based wearable biosensor with a 3D origami structure for multiplexed analyte detection in sweat. Microsyst. Nanoeng. 2023, 9, 36. [Google Scholar] [CrossRef]
- Lai, Q.T.; Liang, H.Q.; Tang, X.G.; Zhang, D.; Roy, V.A.; Sun, Q.J. Printing Paper-Derived Ultralight and Highly Sensitive E-Skin for Health Monitoring and Information Encryption. J. Alloys Compd. 2024, 976, 173411. [Google Scholar] [CrossRef]
- Niu, G.; Wang, Z.; Xue, Y.; Yan, J.; Dutta, A.; Chen, X.; Wang, Y.; Liu, C.; Du, S.; Guo, L.; et al. Pencil-on-paper humidity sensor treated with NaCl solution for health monitoring and skin characterization. Nano Lett. 2023, 23, 1252–1260. [Google Scholar] [CrossRef]
- Karmakar, R.S.; Huang, J.F.; Chu, C.P.; Mai, M.H.; Chao, J.I.; Liao, Y.C.; Lu, Y.W. Origami-Inspired Conductive Paper-Based Folded Pressure Sensor with Interconnection Scaling at the Crease for Novel Wearable Applications. ACS Appl. Mater. Interfaces 2024, 16, 4231–4241. [Google Scholar] [CrossRef]
- Busin, V.; Burgess, S.; Shu, W. A hybrid paper-based microfluidic platform toward veterinary P-ELISA. Sens. Actuators B Chem. 2018, 273, 536–542. [Google Scholar] [CrossRef]
- Zhao, G.; Wang, H.; Hou, P.; He, C.; He, H. Rapid visual detection of Mycobacterium avium subsp. paratuberculosis by recombinase polymerase amplification combined with a lateral flow dipstick. J. Vet. Sci. 2018, 19, 242–250. [Google Scholar] [CrossRef]
- Yang, Z.; Xu, G.; Reboud, J.; Ali, S.A.; Kaur, G.; McGiven, J.; Boby, N.; Gupta, P.K.; Chaudhuri, P.; Cooper, J.M. Rapid veterinary diagnosis of bovine reproductive infectious diseases from semen using paper-origami DNA microfluidics. ACS Sens. 2018, 3, 403–409. [Google Scholar] [CrossRef]
- Huang, L.; Xiao, W.; Xu, T.; Chen, H.; Jin, Z.; Zhang, Z.; Song, Q.; Tang, Y. Miniaturized paper-based smartphone biosensor for differential diagnosis of wild-type pseudorabies virus infection versus vaccination immunization. Sens. Actuators B Chem. 2021, 327, 128893. [Google Scholar] [CrossRef]
- Feng, S.; Hua, M.Z.; Roopesh, M.; Lu, X. Rapid detection of three mycotoxins in animal feed materials using competitive ELISA-based origami microfluidic paper analytical device (μPAD). Anal. Bioanal. Chem. 2023, 415, 1943–1951. [Google Scholar] [CrossRef]
- Li, S.; Liu, Y.; Wang, Y.; Wang, M.; Liu, C.; Wang, Y. Rapid detection of Brucella spp. and elimination of carryover using multiple cross displacement amplification coupled with nanoparticles-based lateral flow biosensor. Front. Cell. Infect. Microbiol. 2019, 9, 78. [Google Scholar] [CrossRef]
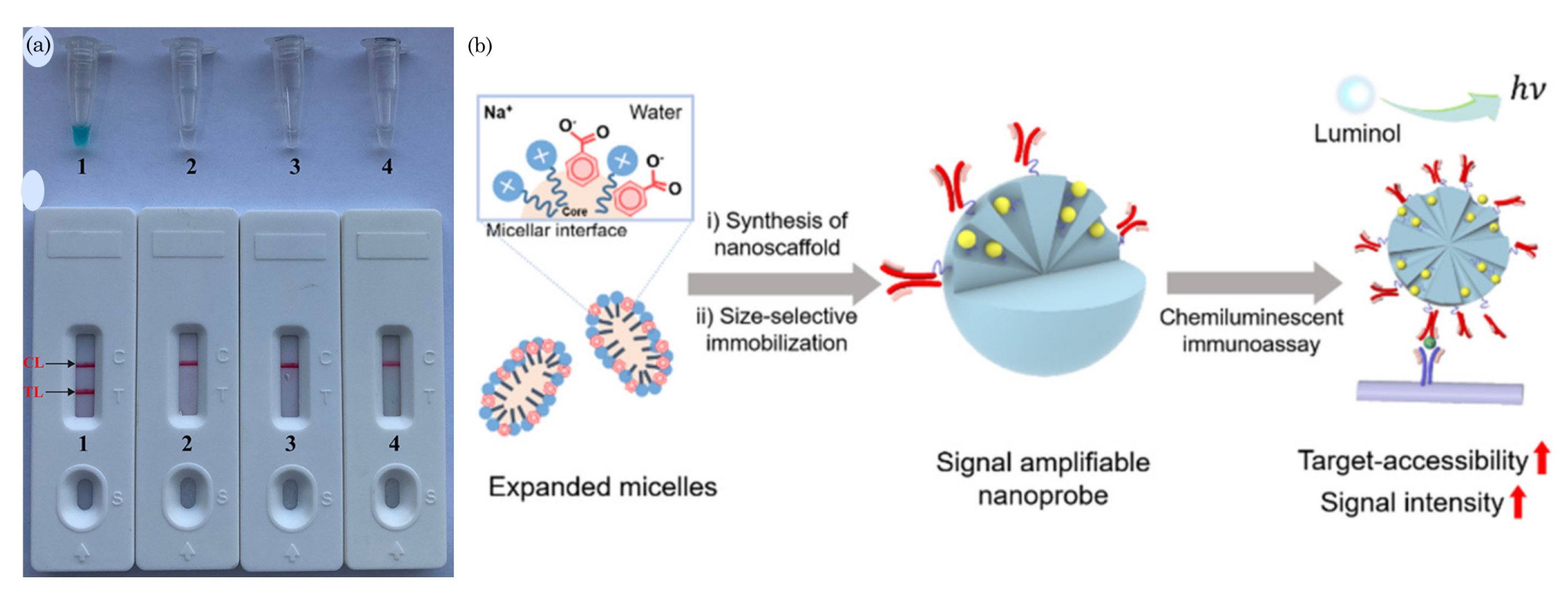
- Jung, H.; Park, S.H.; Lee, J.; Lee, B.; Park, J.; Seok, Y.; Choi, J.H.; Kim, M.G.; Song, C.S.; Lee, J. A size-selectively biomolecule-immobilized nanoprobe-based chemiluminescent lateral flow immunoassay for detection of avian-origin viruses. Anal. Chem. 2020, 93, 792–800. [Google Scholar] [CrossRef]
- Meredith, N.A.; Quinn, C.; Cate, D.M.; Reilly, T.H.; Volckens, J.; Henry, C.S. Paper-based analytical devices for environmental analysis. Analyst 2016, 141, 1874–1887. [Google Scholar] [CrossRef]
- Colozza, N.; Caratelli, V.; Moscone, D.; Arduini, F. Paper-based devices as new smart analytical tools for sustainable detection of environmental pollutants. Case Stud. Chem. Environ. Eng. 2021, 4, 100167. [Google Scholar] [CrossRef]
- Alahmad, W.; Cetinkaya, A.; Kaya, S.I.; Varanusupakul, P.; Ozkan, S.A. Electrochemical paper-based analytical devices for environmental analysis: Current trends and perspectives. Trends Environ. Anal. Chem. 2023, 40, e00220. [Google Scholar] [CrossRef]
- Suo, Z.; Liang, R.; Liu, R.; Wei, M.; He, B.; Jiang, L.; Sun, X.; Jin, H. A convenient paper-based fluorescent aptasensor for high-throughput detection of Pb2+ in multiple real samples (water-soil-food). Anal. Chim. Acta 2023, 1239, 340714. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Chang, W.; Zhang, H.; Cai, Z.; Yang, Y.; Zeng, C. Visual and Real-Time Monitoring of Cd2+ in Water, Rice, and Rice Soil with Test Paper Based on [2 + 2] Lanthanide Clusters. Inorg. Chem. 2023, 62, 6387–6396. [Google Scholar] [CrossRef]
- GB 2762-2022; National Food Safety Standard Maximum Levels of Contaminants in Foods. State Administration for Market Regulation, National Health Commission: Beijing, China, 2022. Available online: https://food.chemlinked.com/database/view/3048 (accessed on 12 May 2024).
- Wang, S.; Ge, L.; Li, L.; Yan, M.; Ge, S.; Yu, J. Molecularly imprinted polymer grafted paper-based multi-disk micro-disk plate for chemiluminescence detection of pesticide. Biosens. Bioelectron. 2013, 50, 262–268. [Google Scholar] [CrossRef] [PubMed]
- Ma, Z.; Li, Y.; Lu, C.; Li, M. On-site Screening Method for Bioavailability Assessment of the Organophosphorus Pesticide, Methyl Parathion, and Its Primary Metabolite in Soils by Paper Strip Biosensor. J. Hazard. Mater. 2023, 457, 131725. [Google Scholar] [CrossRef] [PubMed]
- Cioffi, A.; Mancini, M.; Gioia, V.; Cinti, S. Office paper-based electrochemical strips for organophosphorus pesticide monitoring in agricultural soil. Environ. Sci. Technol. 2021, 55, 8859–8865. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Wang, Z.; Huang, X.; Huang, Q.; Wen, Y.; Li, B.; Holmes, M.; Shi, J.; Zou, X. Uniform stain pattern of robust MOF-mediated probe for flexible paper-based colorimetric sensing toward environmental pesticide exposure. Chem. Eng. J. 2023, 451, 138928. [Google Scholar] [CrossRef]
- Ranveer, S.A.; Harshitha, C.; Dasriya, V.; Tehri, N.; Kumar, N.; Raghu, H. Assessment of developed paper strip based sensor with pesticide residues in different dairy environmental samples. Curr. Res. Food Sci. 2023, 6, 100416. [Google Scholar] [CrossRef]
- Caratelli, V.; Fegatelli, G.; Moscone, D.; Arduini, F. A paper-based electrochemical device for the detection of pesticides in aerosol phase inspired by nature: A flower-like origami biosensor for precision agriculture. Biosens. Bioelectron. 2022, 205, 114119. [Google Scholar] [CrossRef]
- Taudte, R.V.; Beavis, A.; Wilson-Wilde, L.; Roux, C.; Doble, P.; Blanes, L. A portable explosive detector based on fluorescence quenching of pyrene deposited on coloured wax-printed μPADs. Lab Chip 2013, 13, 4164–4172. [Google Scholar] [CrossRef]
- Wang, J.; Yang, L.; Liu, B.; Jiang, H.; Liu, R.; Yang, J.; Han, G.; Mei, Q.; Zhang, Z. Inkjet-printed silver nanoparticle paper detects airborne species from crystalline explosives and their ultratrace residues in open environment. Anal. Chem. 2014, 86, 3338–3345. [Google Scholar] [CrossRef]
- Ueland, M.; Blanes, L.; Taudte, R.V.; Stuart, B.H.; Cole, N.; Willis, P.; Roux, C.; Doble, P. Capillary-driven microfluidic paper-based analytical devices for Lab on a Chip screening of explosive residues in soil. J. Chromatogr. A 2016, 1436, 28–33. [Google Scholar] [CrossRef]
- de Araujo, W.R.; Cardoso, T.M.; da Rocha, R.G.; Santana, M.H.; Munoz, R.A.; Richter, E.M.; Paixão, T.R.; Coltro, W.K. Portable analytical platforms for forensic chemistry: A review. Anal. Chim. Acta 2018, 1034, 1–21. [Google Scholar] [CrossRef]
- Raucci, A.; Miglione, A.; Cimmino, W.; Cioffi, A.; Singh, S.; Spinelli, M.; Amoresano, A.; Musile, G.; Cinti, S. Technical Evaluation of a Paper-Based Electrochemical Strip to Measure Nitrite Ions in the Forensic Field. ACS Meas. Sci. Au 2023, 4, 136–143. [Google Scholar] [CrossRef]
- Hunter, P.R.; Zmirou-Navier, D.; Hartemann, P. Estimating the impact on health of poor reliability of drinking water interventions in developing countries. Sci. Total. Environ. 2009, 407, 2621–2624. [Google Scholar] [CrossRef]
- Adu-Manu, K.S.; Tapparello, C.; Heinzelman, W.; Katsriku, F.A.; Abdulai, J.D. Water quality monitoring using wireless sensor networks: Current trends and future research directions. ACM Trans. Sens. Netw. 2017, 13, 1–41. [Google Scholar] [CrossRef]
- Vikesland, P.J. Nanosensors for water quality monitoring. Nat. Nanotec. 2018, 13, 651–660. [Google Scholar] [CrossRef]
- Liu, F.; Nordin, A.; Li, F.; Voiculescu, I. A lab-on-chip cell-based biosensor for label-free sensing of water toxicants. Lab Chip 2014, 14, 1270–1280. [Google Scholar] [CrossRef]
- Liu, B.; Lei, Y.; Li, B. A batch-mode cube microbial fuel cell based “shock” biosensor for wastewater quality monitoring. Biosens. Bioelectron. 2014, 62, 308–314. [Google Scholar] [CrossRef]
- Sicard, C.; Glen, C.; Aubie, B.; Wallace, D.; Jahanshahi-Anbuhi, S.; Pennings, K.; Daigger, G.T.; Pelton, R.; Brennan, J.D.; Filipe, C.D. Tools for water quality monitoring and mapping using paper-based sensors and cell phones. Water Res. 2015, 70, 360–369. [Google Scholar] [CrossRef] [PubMed]
- İncel, A.; Akın, O.; Çağır, A.; Yıldız, Ü.H.; Demir, M.M. Smart phone assisted detection and quantification of cyanide in drinking water by paper based sensing platform. Sens. Actuators B Chem. 2017, 252, 886–893. [Google Scholar] [CrossRef]
- Cho, J.H.; Gao, Y.; Choi, S. A portable, single-use, paper-based microbial fuel cell sensor for rapid, on-site water quality monitoring. Sensors 2019, 19, 5452. [Google Scholar] [CrossRef]
- Cho, J.H.; Gao, Y.; Ryu, J.; Choi, S. Portable, disposable, paper-based microbial fuel cell sensor utilizing freeze-dried bacteria for in situ water quality monitoring. ACS Omega 2020, 5, 13940–13947. [Google Scholar] [CrossRef] [PubMed]
- Charbaji, A.; Heidari-Bafroui, H.; Anagnostopoulos, C.; Faghri, M. A new paper-based microfluidic device for improved detection of nitrate in water. Sensors 2020, 21, 102. [Google Scholar] [CrossRef] [PubMed]
- Jaballah, M.B.; Karrat, A.; Amine, A.; Dridi, C. Immobilization of diphenylcarbazide on paper-based analytical devices for the pre-concentration and detection of chromium VI in water samples. Talanta 2023, 265, 124889. [Google Scholar] [CrossRef] [PubMed]
- da Silva, V.A.O.P.; de Freitas, R.C.; de Oliveira, P.R.; Moreira, R.C.; Marcolino-Júnior, L.H.; Bergamini, M.F.; Coltro, W.K.; Janegitz, B.C. Microfluidic paper-based device integrated with smartphone for point-of-use colorimetric monitoring of water quality index. Measurement 2020, 164, 108085. [Google Scholar] [CrossRef]
- Xiong, X.; Guo, C.; Yan, G.; Han, B.; Wu, Z.; Chen, Y.; Xu, S.; Shao, P.; Song, H.; Xu, X.; et al. Simultaneous Cross-type Detection of Water Quality Indexes via a Smartphone-App Integrated Microfluidic Paper-Based Platform. ACS Omega 2022, 7, 44338–44345. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.H.; Chen, S.J.; Lee, J.E.; Chu, W.Y.; Yu, C.J.; Chang, C.C.; Chen, C.F. The detection of Mercury (II) ions using fluorescent gold nanoclusters on a portable paper-based device. Chem. Eng. J. 2022, 430, 133070. [Google Scholar] [CrossRef]
- Aguiar, J.I.; Ribeiro, S.O.; Leite, A.; Rangel, M.; Rangel, A.O.; Mesquita, R.B. Use of a rhodamine-based chelator in a microfluidic paper-based analytical device for the in-situ copper quantification in natural waters. Talanta 2024, 271, 125683. [Google Scholar] [CrossRef]
- Uhlikova, N.; Almeida, M.I.G.; McKelvie, I.D.; Kolev, S.D. Microfluidic paper-based analytical device for the speciation of inorganic nitrogen species. Talanta 2024, 271, 125671. [Google Scholar] [CrossRef] [PubMed]
- Thangjitsirisin, K.; Seeharaj, P.; Choengchan, N. Superhydrophobic eggshell for fabrication of hydrophobic barrier of paper-based analytical device for colorimetric determination of ammonium ion in water. Microchem. J. 2024, 200, 110464. [Google Scholar] [CrossRef]
- Sameenoi, Y.; Panymeesamer, P.; Supalakorn, N.; Koehler, K.; Chailapakul, O.; Henry, C.S.; Volckens, J. Microfluidic paper-based analytical device for aerosol oxidative activity. Environ. Sci. Technol. 2013, 47, 932–940. [Google Scholar] [CrossRef] [PubMed]
- Khachornsakkul, K.; Hung, K.H.; Chang, J.J.; Dungchai, W.; Chen, C.H. A rapid and highly sensitive paper-based colorimetric device for the on-site screening of ammonia gas. Analyst 2021, 146, 2919–2927. [Google Scholar] [CrossRef] [PubMed]
- Mettakoonpitak, J.; Sawatdichai, N.; Thepnuan, D.; Siripinyanond, A.; Henry, C.S.; Chantara, S. Microfluidic paper-based analytical devices for simultaneous detection of oxidative potential and copper in aerosol samples. Microchim. Acta 2023, 190, 241. [Google Scholar] [CrossRef] [PubMed]
- De Matteis, V.; Cascione, M.; Fella, G.; Mazzotta, L.; Rinaldi, R. Colorimetric paper-based device for hazardous compounds detection in air and water: A proof of concept. Sensors 2020, 20, 5502. [Google Scholar] [CrossRef] [PubMed]
- Bordbar, M.M.; Tashkhourian, J.; Hemmateenejad, B. Paper-Based Optical Nose Made with Bimetallic Nanoparticles for Monitoring Ignitable Liquids in Gasoline. ACS Appl. Mater. Interfaces 2022, 14, 8333–8342. [Google Scholar] [CrossRef] [PubMed]
- Arroyo, P.; Gómez-Suárez, J.; Suárez, J.I.; Lozano, J. Low-Cost Air Quality Measurement System Based on Electrochemical and PM Sensors with Cloud Connection. Sensors 2021, 21, 6228. [Google Scholar] [CrossRef]
- Li, G.; Yuan, H.; Mou, J.; Dai, E.; Zhang, H.; Li, Z.; Zhao, Y.; Dai, Y.; Zhang, X. Electrochemical detection of nitrate with carbon nanofibers and copper co-modified carbon fiber electrodes. Compos. Commun. 2022, 29, 101043. [Google Scholar] [CrossRef]
- Mettakoonpitak, J.; Volckens, J.; Henry, C.S. Janus electrochemical paper-based analytical devices for metals detection in aerosol samples. Anal. Chem. 2019, 92, 1439–1446. [Google Scholar] [CrossRef]
- Davis, D.; Narayanan, S.K.; Ajeev, A.; Nair, J.; Jeeji, J.; Vijayan, A.; Viyyur Kuttyadi, M.; Nelliparambil Sathian, A.; Arulraj, A.K. Flexible Paper-Based Room-Temperature Acetone Sensors with Ultrafast Regeneration. ACS Appl. Mater. Interfaces 2023, 15, 25734–25743. [Google Scholar] [CrossRef]
- Qin, X.; Wu, T.; Zhu, Y.; Shan, X.; Liu, C.; Tao, N. A paper based milli-cantilever sensor for detecting hydrocarbon gases via smartphone camera. Anal. Chem. 2020, 92, 8480–8486. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, J.; Zhao, Z.; Lian, K.; Yin, L.; Wang, J.; Man, S.; Liu, G.; Ma, L. SERS-based CRISPR/Cas assay on microfluidic paper analytical devices for supersensitive detection of pathogenic bacteria in foods. Biosens. Bioelectron. 2022, 207, 114167. [Google Scholar] [CrossRef]
- Lin, X.; Li, C.; Tong, X.; Duan, N.; Wang, Z.; Wu, S. A portable paper-based aptasensor for simultaneous visual detection of two mycotoxins in corn flour using dual-color upconversion nanoparticles and Cu-TCPP nanosheets. Food Chem. 2023, 404, 134750. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Wang, H.; Yu, Z.; Tang, X.; Zhou, M.; Guo, Y.; Xiong, B. A disposable immunosensor array using cellulose paper assembled chemiresistive biosensor for simultaneous monitoring of mycotoxins AFB1 and FB1. Talanta 2024, 276, 126145. [Google Scholar] [CrossRef] [PubMed]
- Xiang, J.; Qi, J.; Hu, D.; Wang, C.; Wang, L.; Wu, Y.; Chen, J.; Zhang, Z.; Wang, X.; Li, B.; et al. Molecularly imprinted metal-organic frameworks assisted cloth and paper hybrid microfluidic devices for visual detection of gonyautoxin. J. Hazard. Mater. 2024, 469, 133969. [Google Scholar] [CrossRef] [PubMed]
- dos Santos, D.M.; Migliorini, F.L.; Coatrini-Soares, A.; Soares, J.C.; Mattoso, L.H.; Oliveira, O.N.; Correa, D.S. Low-cost paper-based sensors modified with curcumin for the detection of ochratoxin a in beverages. Sens. Actuators Rep. 2024, 7, 100184. [Google Scholar] [CrossRef]
- Hua, M.Z.; Lu, X. Development of a microfluidic paper-based immunoassay for rapid detection of allergic protein in foods. ACS Sens. 2020, 5, 4048–4056. [Google Scholar] [CrossRef] [PubMed]
- Kunpatee, K.; Panphloi, M.; Charoenkitamorn, K.; Pimpitak, U.; Puthong, S.; Buakeaw, A.; Komolpis, K.; Sain, M.M.; Yakoh, A.; Chaiyo, S. Electrochemical lateral flow immunosensor with enhanced reproducibility for milk allergen detection. Sens. Actuators B Chem. 2024, 401, 135042. [Google Scholar] [CrossRef]
- Han, X.; Zhang, D.; Xie, M.; Yang, J.; Wang, Y.; Li, H.; Wang, S.; Pan, M. Microfluidic paper-based analysis device applying black phosphorus nanosheets@ MWCNTs-COOH: A portable and efficient strategy for detection of β-Lactoglobulin in dairy products. Food Chem. 2024, 446, 138844. [Google Scholar] [CrossRef]
- Ortiz-Gómez, I.; Ipatov, A.; Barreiro-Docío, E.; Salinas-Castillo, A.; de Orbe-Payá, I.; Capitán-Vallvey, L.F.; Prado, M. Microfluidic paper-based analytical aptasensor for fluorometric β-lactoglobulin determination. Microchem. J. 2024, 198, 110121. [Google Scholar] [CrossRef]
- He, Y.; Hua, M.Z.; Feng, S.; Lu, X. Development of a smartphone-integrated microfluidic paper-based optosensing platform coupled with molecular imprinting technique for in-situ determination of histamine in canned tuna. Food Chem. 2024, 451, 139446. [Google Scholar] [CrossRef]
- Pan, M.; Han, X.; Chen, S.; Yang, J.; Wang, Y.; Li, H.; Wang, S. Paper-based microfluidic device for selective detection of peanut allergen Ara h1 applying black phosphorus-Au nanocomposites for signal amplification. Talanta 2024, 267, 125188. [Google Scholar] [CrossRef] [PubMed]
- Lu, C.; Xu, S.; Wang, S.; Wang, T.; Wang, W.L.; Yang, C.; Zhang, Y. Facile and Ultrasensitive Food Allergen Quantification Using Microzone Paper-Based Mass Spectrometric Immunoassay. Anal. Chem. 2024, 96, 2387–2395. [Google Scholar] [CrossRef] [PubMed]
- Martins, T.S.; Machado, S.A.; Oliveira Jr, O.N.; Bott-Neto, J.L. Optimized paper-based electrochemical sensors treated in acidic media to detect carbendazim on the skin of apple and cabbage. Food Chem. 2023, 410, 135429. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Wang, S.; Yang, F.; Li, Z.; Huang, W. Visual inspection of acidic pH and bisulfite in white wine using a colorimetric and fluorescent probe. Food Chem. 2023, 408, 135200. [Google Scholar] [CrossRef] [PubMed]
- Placer, L.; Lavilla, I.; Pena-Pereira, F.; Bendicho, C. A 3D microfluidic paper-based analytical device with smartphone-assisted colorimetric detection for iodine speciation in seaweed samples. Sens. Actuators B Chem. 2023, 377, 133109. [Google Scholar] [CrossRef]
- Masoomi, S.; Sharifi, H.; Hemmateenejad, B. A paper-based optical tongue for characterization of Iranian honey: Identification of geographical/botanical origins and adulteration detection. Food Control 2024, 155, 110052. [Google Scholar] [CrossRef]
- Selvaraj, S.; Ravi Shankaran, D. Nano Enabled Plasmonic Strips For Colorimetric Detection Of Food Adulterants. ChemistrySelect 2023, 8, e202302027. [Google Scholar] [CrossRef]
- Andrade, L.M.; Romanholo, P.V.; Ananias, A.C.A.; Venancio, K.P.; Silva-Neto, H.A.; Coltro, W.K.; Sgobbi, L.F. Pocket test for instantaneous quantification of starch adulterant in milk using a counterfeit banknote detection pen. Food Chem. 2023, 405, 134844. [Google Scholar] [CrossRef]
- Dortez, S.; Crevillen, A.G.; Escarpa, A.; Cinti, S. Electroanalytical paper-based device for reliable detection and quantification of sugars in milk. Sens. Actuators B Chem. 2024, 398, 134704. [Google Scholar] [CrossRef]
- Shalileh, F.; Sabahi, H.; Dadmehr, M.; Hosseini, M. Sensing approaches toward detection of urea adulteration in milk. Microchem. J. 2023, 193, 108990. [Google Scholar] [CrossRef]
- Wu, Y.; Zhang, L.; Zhang, D.; Yu, R. A surface molecularly imprinted microfluidic paper based device with smartphone assisted colorimetric detection for butachlor in mung bean. Food Chem. 2024, 435, 137659. [Google Scholar] [CrossRef] [PubMed]
- Qin, X.; Liu, J.; Zhang, Z.; Li, J.; Yuan, L.; Zhang, Z.; Chen, L. Microfluidic paper-based chips in rapid detection: Current status, challenges, and perspectives. TrAC Trends Anal. Chem. 2021, 143, 116371. [Google Scholar] [CrossRef]
- Gautam, N.; Verma, R.; Ram, R.; Singh, J.; Sarkar, A. Development of a biodegradable microfluidic paper-based device for blood-plasma separation integrated with non-enzymatic electrochemical detection of ascorbic acid. Talanta 2024, 266, 125019. [Google Scholar] [CrossRef]
- Yamada, K.; Shibata, H.; Suzuki, K.; Citterio, D. Toward practical application of paper-based microfluidics for medical diagnostics: State-of-the-art and challenges. Lab Chip 2017, 17, 1206–1249. [Google Scholar] [CrossRef]
| Fabrication Techniques | Equipment and Materials Requirements | Advantages | Limitations | Ref. |
|---|---|---|---|---|
| Blade cutting/plotting | X-Y plotter, knife | Provides sharp features, no chemical required | Limited to 2D designs | [60,61] |
| Laser cutting | Laser cutter | Precise, customizable designs, suitable for large-scale production, high resolution (∼60 m) | Requires specialized equipment and polymer films to protect the paper device from damage, may generate debris | [62,63,64,65,66] |
| Photolithography | UV light, heating plate, photomask, photoresists (positive/negative), mask aligner, chemicals, oxygen plasma | High resolution (∼200 m), well-established microfabrication technique | Equipment-intensive, may involve multiple complex steps and chances of channel contamination | [67,68,69] |
| 3D printing | 3D printer, inks | Allows for complex, customized designs | Limited resolution compared to traditional microfabrication | [70,71,72,73,74,75,76] |
| Screen printing | Mesh screen, hot plate, transparency film, wax | Low-cost, scalable for mass production | Resolution may vary, suitable for relatively simple designs, new screens are required for different patterns | [77,78,79,80,81,82,83] |
| Wax printing | Hot plate, wax printer, solid wax | Simple, rapid, cost-effective, and suitable for prototyping | Limited resolution (∼550 m), wax spread, limited channel height control, temperature sensitivity | [84,85,86,87,88,89] |
| Inkjet printing | Customized inkjet printer, hydrophobic ink, hot plate, and chemicals | Noncontact, suitable for rapid prototyping | Resolution may be lower than other techniques, requires multiple steps, and post-printing heating is required for some inks | [90,91,92,93,94,95,96] |
| Embossing | Embossing tools, adhesives, silane | Simple, flexible, suitable for rapid prototyping | Limited resolution, may affect paper integrity, susceptible to contamination | [97,98,99,100] |
| Origami and kirigami | Paper cutting and folding tools, adhesives | Foldable structures, flexible design, enhanced functionality, scalability | Precision challenges, design and assembly complexity, limited material compatibility | [101,102,103,104,105,106,107,108] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kumar, S.; Kaushal, J.B.; Lee, H.P. Sustainable Sensing with Paper Microfluidics: Applications in Health, Environment, and Food Safety. Biosensors 2024, 14, 300. https://doi.org/10.3390/bios14060300
Kumar S, Kaushal JB, Lee HP. Sustainable Sensing with Paper Microfluidics: Applications in Health, Environment, and Food Safety. Biosensors. 2024; 14(6):300. https://doi.org/10.3390/bios14060300
Chicago/Turabian StyleKumar, Sanjay, Jyoti Bala Kaushal, and Heow Pueh Lee. 2024. "Sustainable Sensing with Paper Microfluidics: Applications in Health, Environment, and Food Safety" Biosensors 14, no. 6: 300. https://doi.org/10.3390/bios14060300
APA StyleKumar, S., Kaushal, J. B., & Lee, H. P. (2024). Sustainable Sensing with Paper Microfluidics: Applications in Health, Environment, and Food Safety. Biosensors, 14(6), 300. https://doi.org/10.3390/bios14060300







