Solution-Induced Degradation of the Silicon Nanobelt Field-Effect Transistor Biosensors
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Materials
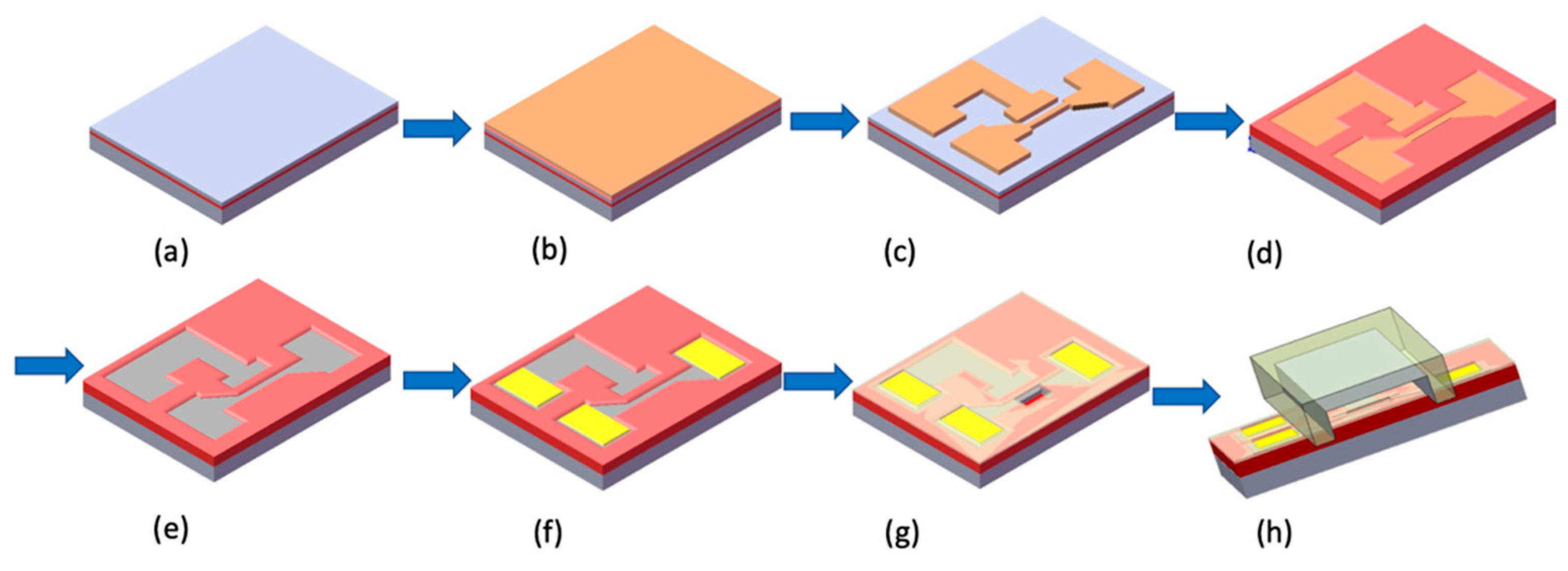
2.2. Fabrication of the Silicon NBFET
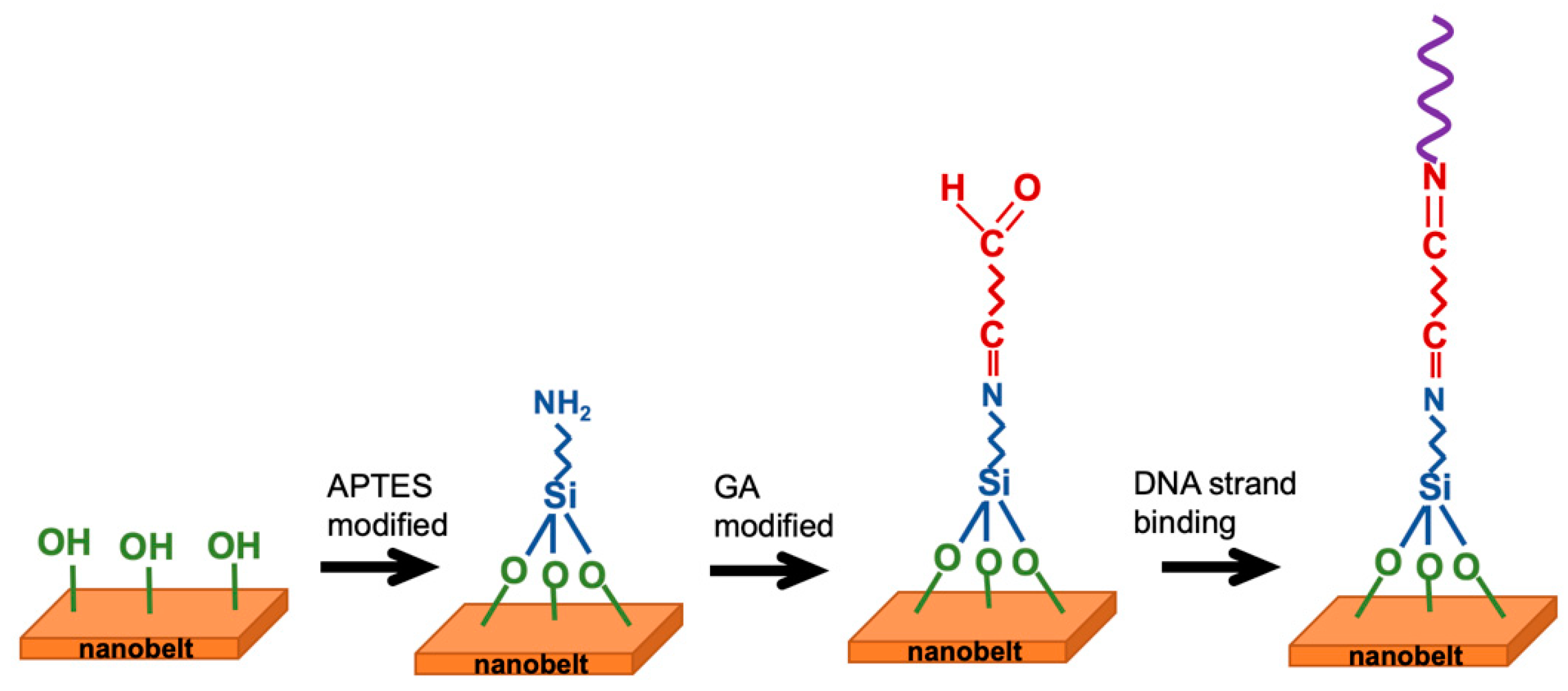
2.3. Surface Functionalization and Biografting
2.4. Fabrication of Microfluidic Channel
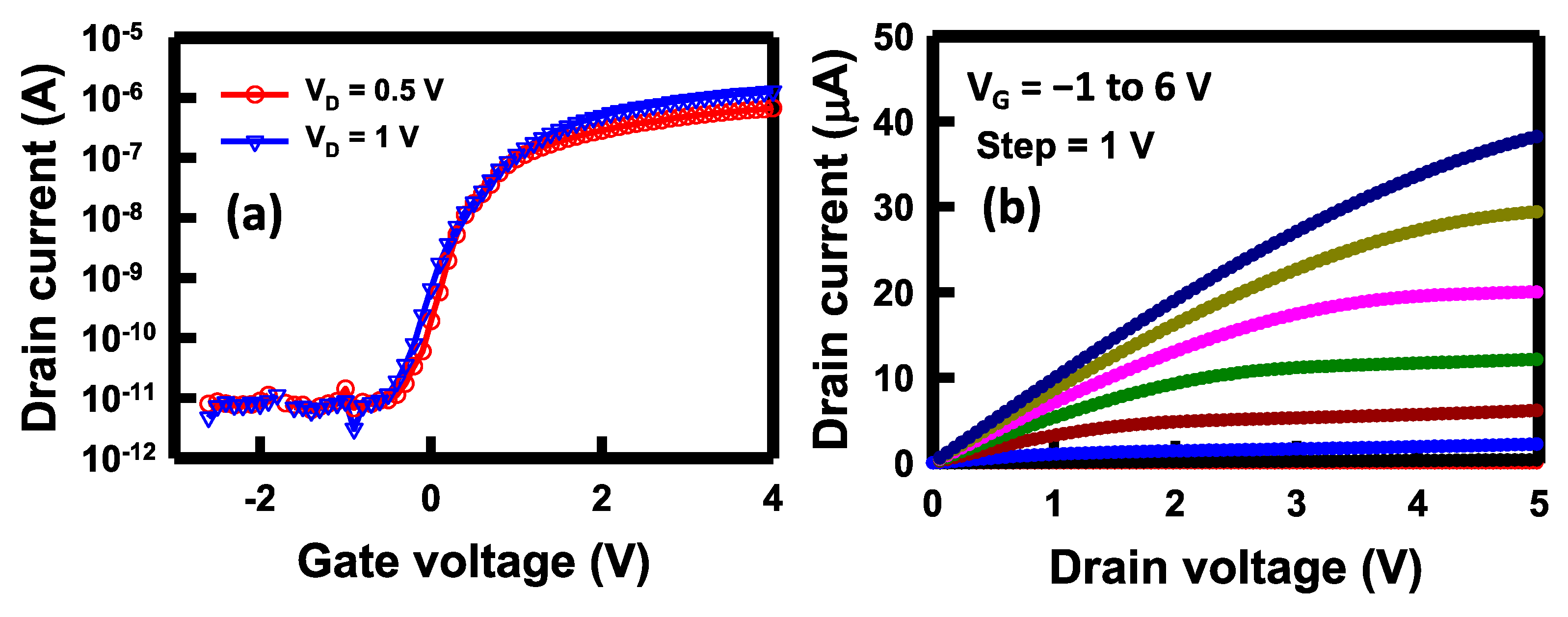
2.5. Device Measurement and Analysis
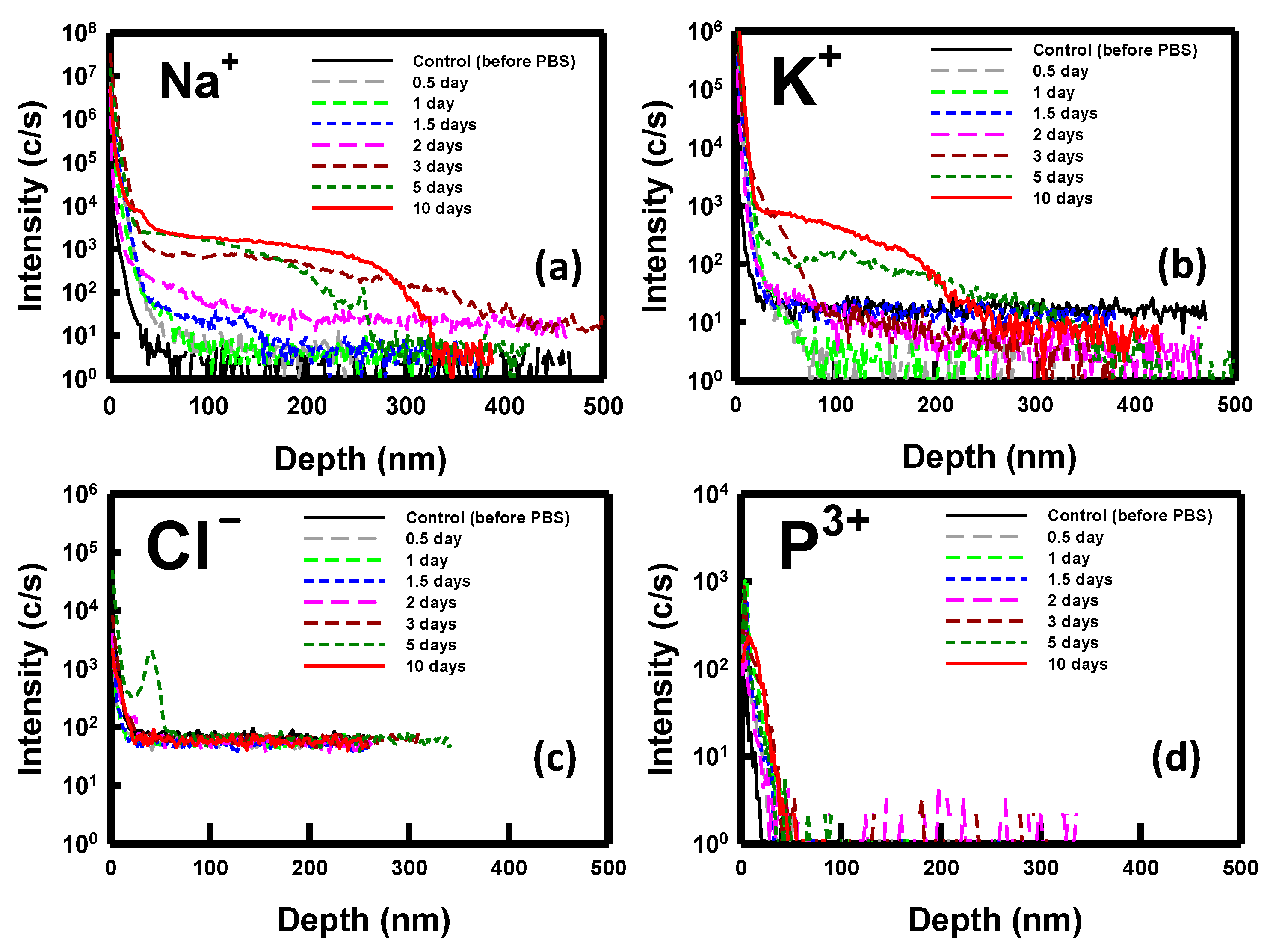
3. Results and Discussions
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
List of Acronyms
| APTES | 3-amino-propyl-triethoxy-silane |
| AFM | atomic force microscope |
| C2H5OH | ethanol |
| DI | deionized |
| FET | field-effect transistor |
| FITC | fluorescein isothiocyanate |
| FESEM | field-emission scanning electron microscope |
| GA | glutaraldehyde |
| ID–VG | drain current versus gate voltage |
| ID–VD | drain current versus drain voltage |
| LOD | limit of detection |
| NBFET | nanobelt field-effect transistor |
| PBS | phosphate-buffered saline |
| PDMS | polydimethylsiloxane |
| SOI | silicon-on-insulator |
| SiO2 | silicon oxide |
| Si3N4 | silicon nitride |
| SIMS | secondary ion mass spectroscopy |
| SS | subthreshold swing |
| TSRI | Taiwan Semiconductor Research Institute |
| TEM | tunneling electron microscope |
| Vt | threshold voltage |
References
- Huang, C.W.; Lin, C.; Nguyen, M.K.; Hussain, A.; Bui, X.T.; Ngo, H.H. A Review of Biosensor for Environmental Monitoring: Principle, Application, and Corresponding Achievement of Sustainable Development Goals. Bioengineered 2023, 14, 58. [Google Scholar] [CrossRef]
- Zhang, S.P.; Huang, Y.T.; Ren, H.Z.; Chen, Y.J.; Yan, S.S.; Dai, H.; Lv, L. Facile and Portable Multimodal Sensing Platform Driven by Photothermal-Controlled Release System for Biomarker Detection. Biosens. Bioelectron. 2023, 235, 115413. [Google Scholar] [CrossRef]
- Quazi, S. Application of Biosensors in Cancers, an Overview. Front. Bioeng. Biotechnol. 2023, 11, 1193493. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.C. Polycrystalline Silicon Nanowire Field Effect Transistor Biosensors for SARS-CoV-2 Detection. J. Electrochem. Soc. 2022, 169, 077514. [Google Scholar] [CrossRef]
- Shetti, N.P.; Bukkitgar, S.D.; Reddy, K.R.; Reddy, C.V.; Aminabhavi, T.M. Zno-Based Nanostructured Electrodes for Electrochemical Sensors and Biosensors in Biomedical Applications. Biosens. Bioelectron. 2019, 141, 111417. [Google Scholar] [CrossRef]
- Ding, M.L.; Zhang, S.L.; Wang, J.E.; Ding, Y.; Ding, C.F. Ultrasensitive Ratiometric Electrochemiluminescence Sensor with an Efficient Antifouling and Antibacterial Interface of Psbma@Sio-Mxene for Oxytetracycline Trace Detection in the Marine Environment. Anal. Chem. 2023, 95, 16327. [Google Scholar] [CrossRef]
- Sukjee, W.; Sangma, C.; Lieberzeit, P.A.; Ketsuwan, K.; Thepparit, C.; Chailapakul, O.; Ngamrojanavanich, N. Ev71 Virus Induced Silver Nanoparticles Self-Assembly in Polymer Composites with an Application as Virus Biosensor. Sens. Actuators B-Chem. 2023, 393, 134324. [Google Scholar] [CrossRef]
- Parichenko, A.; Choi, W.; Shin, S.; Schlecht, M.; Gutierrez, R.; Akbar, T.F.; Werner, C.; Lee, J.S.; Ibarlucea, B.; Cuniberti, G. Hydrogel-Gated Silicon Nanotransistors for SARS-CoV-2 Antigen Detection in Physiological Ionic Strength. Adv. Mater. Interfaces 2023, 10, 2300391. [Google Scholar] [CrossRef]
- Hayashi, H.; Fujita, M.; Kuroiwa, S.; Ohashi, K.; Okada, M.; Shibasaki, F.; Osaka, T.; Momma, T. Semiconductor-Based Biosensor Exploiting Competitive Adsorption with Charged Pseudo-Target Molecules for Monitoring 5-Fluorouracil Concentration in Human Serum. Sens. Actuators B-Chem. 2023, 395, 134495. [Google Scholar] [CrossRef]
- Ren, Q.Q.; Jiang, L.Y.; Ma, S.H.; Li, T.; Zhu, Y.; Qiu, R.; Xing, Y.; Yin, F.; Li, Z.G.; Ye, X.Y.; et al. Multi-Body Biomarker Entrapment System: An All-Encompassing Tool for Ultrasensitive Disease Diagnosis and Epidemic Screening. Adv. Mater. 2023, 35, e2304119. [Google Scholar] [CrossRef]
- Shariati, M.; Sadeghi, M.; Khoshkhoo, S.M.; Azimi, N. Materialization of a Novel Decorated Nanowire Biosensor Platform Based on Field Effect Transistor under Electrochemical Gate Modulation. J. Electrochem. Soc. 2023, 170, 077502. [Google Scholar] [CrossRef]
- Xing, J.; Dong, Q.Y.; Ding, Q.; Yang, X.; Yuan, R.; Yuan, Y.L. Photoactive Conjugated Microporous Polymer/Carbon Nanotube Coupled with T-Junction Recycling Dual-Strand Displacement Amplification for Sensing N-Gene of COVID-19. Sens. Actuators B-Chem. 2023, 376, 132974. [Google Scholar] [CrossRef]
- Monavari, S.M.; Marsusi, F.; Memarian, N.; Qasemnazhand, M. Carbon Nanotubes and Nanobelts as Potential Materials for Biosensor. Sci. Rep. 2023, 13, 3118. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharyya, I.M.; Cohen, S.; Shalabny, A.; Bashouti, M.; Akabayov, B.; Shalev, G. Specific and Label-Free Immunosensing of Protein-Protein Interactions with Silicon-Based Immunofets. Biosens. Bioelectron. 2019, 132, 143. [Google Scholar] [CrossRef] [PubMed]
- Manimekala, T.; Sivasubramanian, R.; Dharmalingam, G. Nanomaterial-Based Biosensors Using Field-Effect Transistors: A Review. J. Electron. Mater. 2022, 51, 1950. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Kikuchi, N.; Ohshima, N.; Kajisa, T.; Sakata, T.; Izumi, T.; Sone, H. Design and Fabrication of Silicon Nanowire-Based Biosensors with Integration of Critical Factors: Toward Ultrasensitive Specific Detection of Biomolecules. ACS Appl. Mater. Interfaces 2020, 12, 51808. [Google Scholar] [CrossRef] [PubMed]
- Kazemi, N.; Abdolrazzaghi, M.; Light, P.E.; Musilek, P. In-Human Testing of a Non-Invasive Continuous Low-Energy Microwave Glucose Sensor with Advanced Machine Learning Capabilities. Biosens. Bioelectron. 2023, 241, 115668. [Google Scholar] [CrossRef]
- Das, S.; Saxena, K.; Tinguely, J.C.; Pal, A.; Wickramasinghe, N.L.; Khezri, A.; Dubey, V.; Ahmad, A.; Perumal, V.; Ahmad, R.; et al. Sers Nanowire Chip and Machine Learning-Enabled Classification of Wild-Type and Antibiotic-Resistant Bacteria at Species and Strain Levels. ACS Appl. Mater. Interfaces 2023, 15, 24047. [Google Scholar] [CrossRef]
- Ouhibi, A.; Raouafi, A.; Lorrain, N.; Guendouz, M.; Raouafi, N.; Moadhen, A. Functionalized Sers Substrate Based on Silicon Nanowires for Rapid Detection of Prostate Specific Antigen. Sens. Actuators B-Chem. 2021, 330, 129352. [Google Scholar] [CrossRef]
- Pradhan, S.; Albin, S.; Heise, R.L.; Yadavalli, V.K. Portable, Disposable, Biomimetic Electrochemical Sensors for Analyte Detection in a Single Drop of Whole Blood. Chemosensors 2022, 10, 263. [Google Scholar] [CrossRef]
- Das, N.; Roychaudhuri, C. Reliability Study of Nanoporous Silicon Oxide Impedance Biosensor for Virus Detection: Influence of Surface Roughness. IEEE Trans. Device Mater. Reliab. 2015, 15, 402. [Google Scholar] [CrossRef]
- Elgiddawy, N.; Ren, S.W.; Yassar, A.; Louis-Joseph, A.; Sauriat-Dorizon, H.; El Rouby, W.M.A.; El-Gendy, A.O.; Farghali, A.A.; Korri-Youssoufi, H. Dispersible Conjugated Polymer Nanoparticles as Biointerface Materials for Label-Free Bacteria Detection. ACS Appl. Mater. Interfaces 2020, 12, 39979. [Google Scholar] [CrossRef]
- Tian, M.; Wang, J.H.; Li, C.H.; Wang, Z.X.; Liu, G.F.; Lv, E.G.; Zhao, X.F.; Li, Z.; Cao, D.Y.; Liu, H.L.; et al. Qualitative and Quantitative Detection of Microcystin-Lr Based on Sers-Fet Dual-Mode Biosensor. Biosens. Bioelectron. 2022, 212, 114434. [Google Scholar] [CrossRef] [PubMed]
- Kenaan, A.; Li, K.Z.; Barth, I.; Johnson, S.; Song, J.; Krauss, T.F. Guided Mode Resonance Sensor for the Parallel Detection of Multiple Protein Biomarkers in Human Urine with High Sensitivity. Biosens. Bioelectron. 2020, 153, 112047. [Google Scholar] [CrossRef] [PubMed]






Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lin, J.-C.; Zhou, Z.-Y.; Cheng, Y.-C.; Chang, I.-N.; Lin, C.-E.; Wu, C.-C. Solution-Induced Degradation of the Silicon Nanobelt Field-Effect Transistor Biosensors. Biosensors 2024, 14, 65. https://doi.org/10.3390/bios14020065
Lin J-C, Zhou Z-Y, Cheng Y-C, Chang I-N, Lin C-E, Wu C-C. Solution-Induced Degradation of the Silicon Nanobelt Field-Effect Transistor Biosensors. Biosensors. 2024; 14(2):65. https://doi.org/10.3390/bios14020065
Chicago/Turabian StyleLin, Jung-Chih, Zhao-Yu Zhou, Yi-Ching Cheng, I-Nan Chang, Chu-En Lin, and Chi-Chang Wu. 2024. "Solution-Induced Degradation of the Silicon Nanobelt Field-Effect Transistor Biosensors" Biosensors 14, no. 2: 65. https://doi.org/10.3390/bios14020065
APA StyleLin, J.-C., Zhou, Z.-Y., Cheng, Y.-C., Chang, I.-N., Lin, C.-E., & Wu, C.-C. (2024). Solution-Induced Degradation of the Silicon Nanobelt Field-Effect Transistor Biosensors. Biosensors, 14(2), 65. https://doi.org/10.3390/bios14020065





