Highly Sensitive Detection of Chymotrypsin Based on Metal Organic Frameworks with Peptides Sensors
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemical Reagents and Instruments
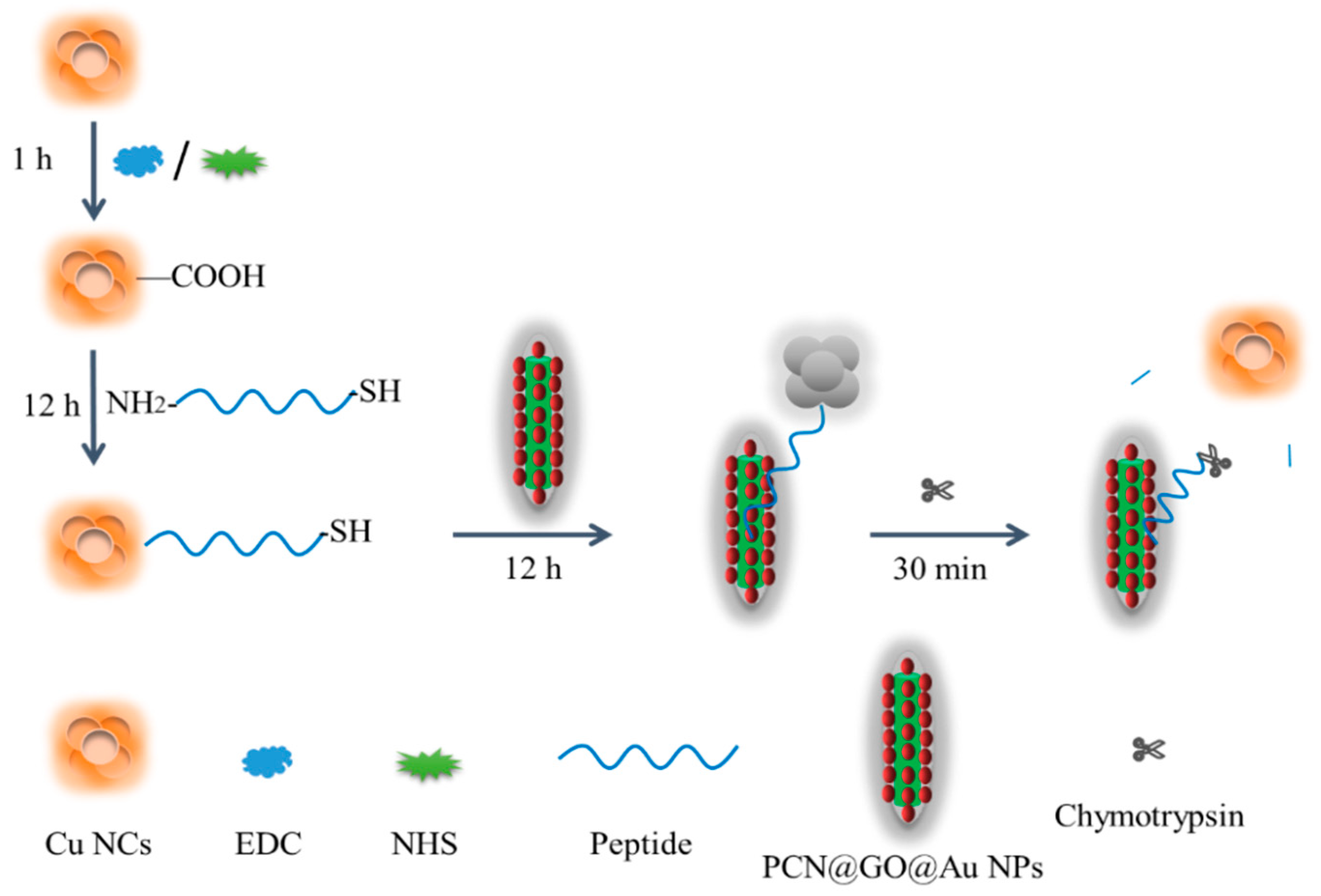
2.2. Methods
3. Results
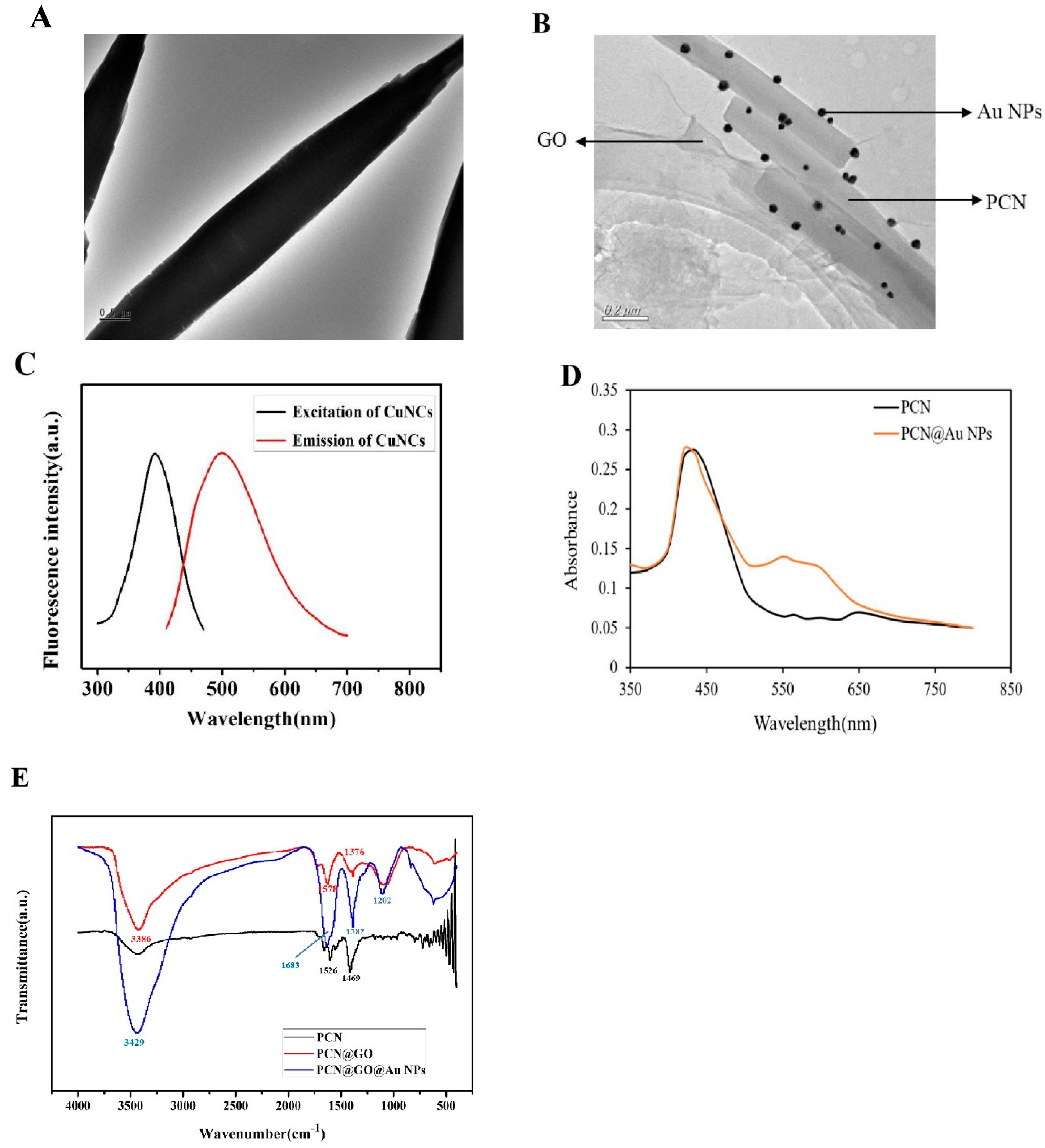
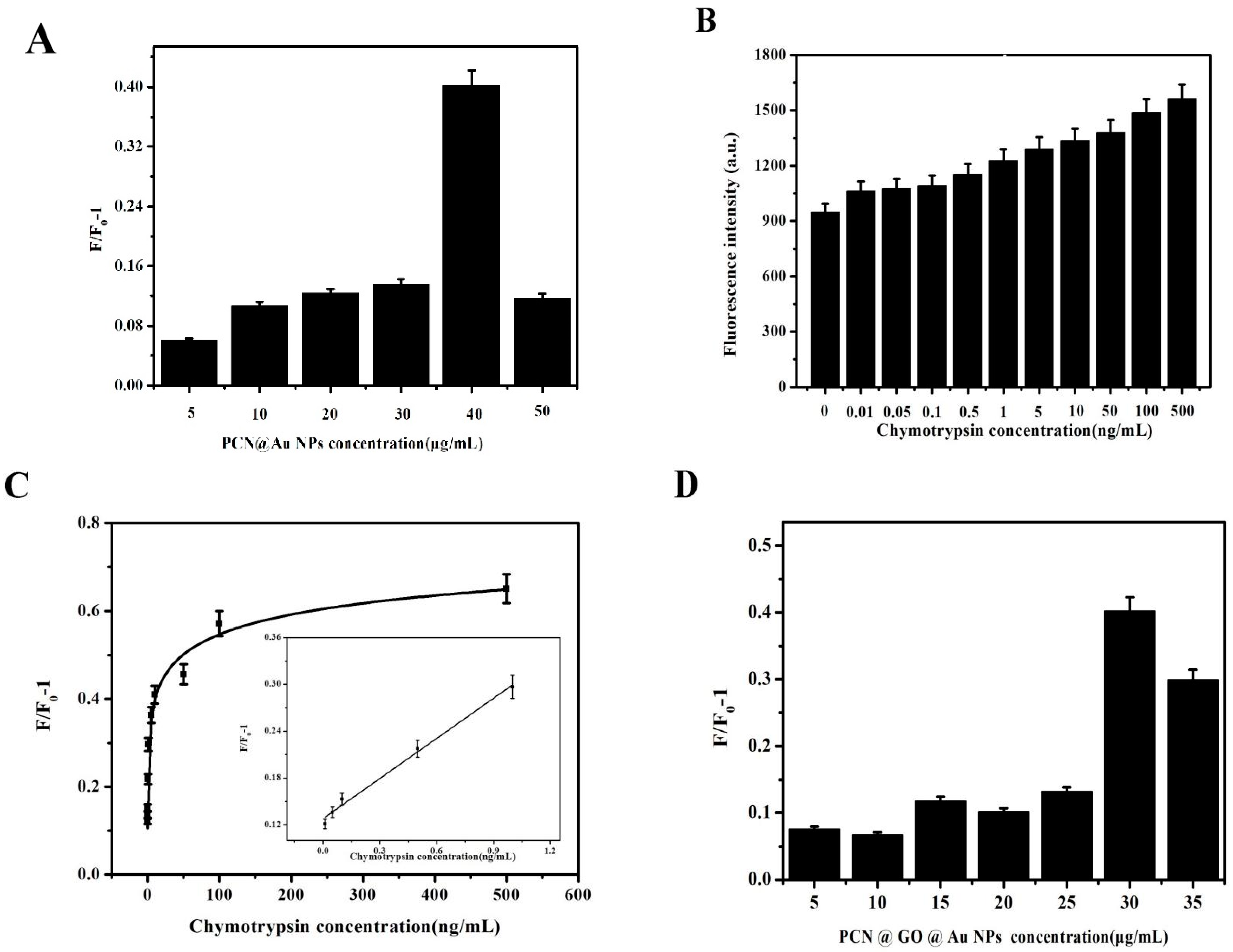
3.1. Preparation of PCN@AuNPs
3.2. Sensitive Detection Based on PCN@Au NPs
3.3. Preparation of PCN@GO@Au NPs
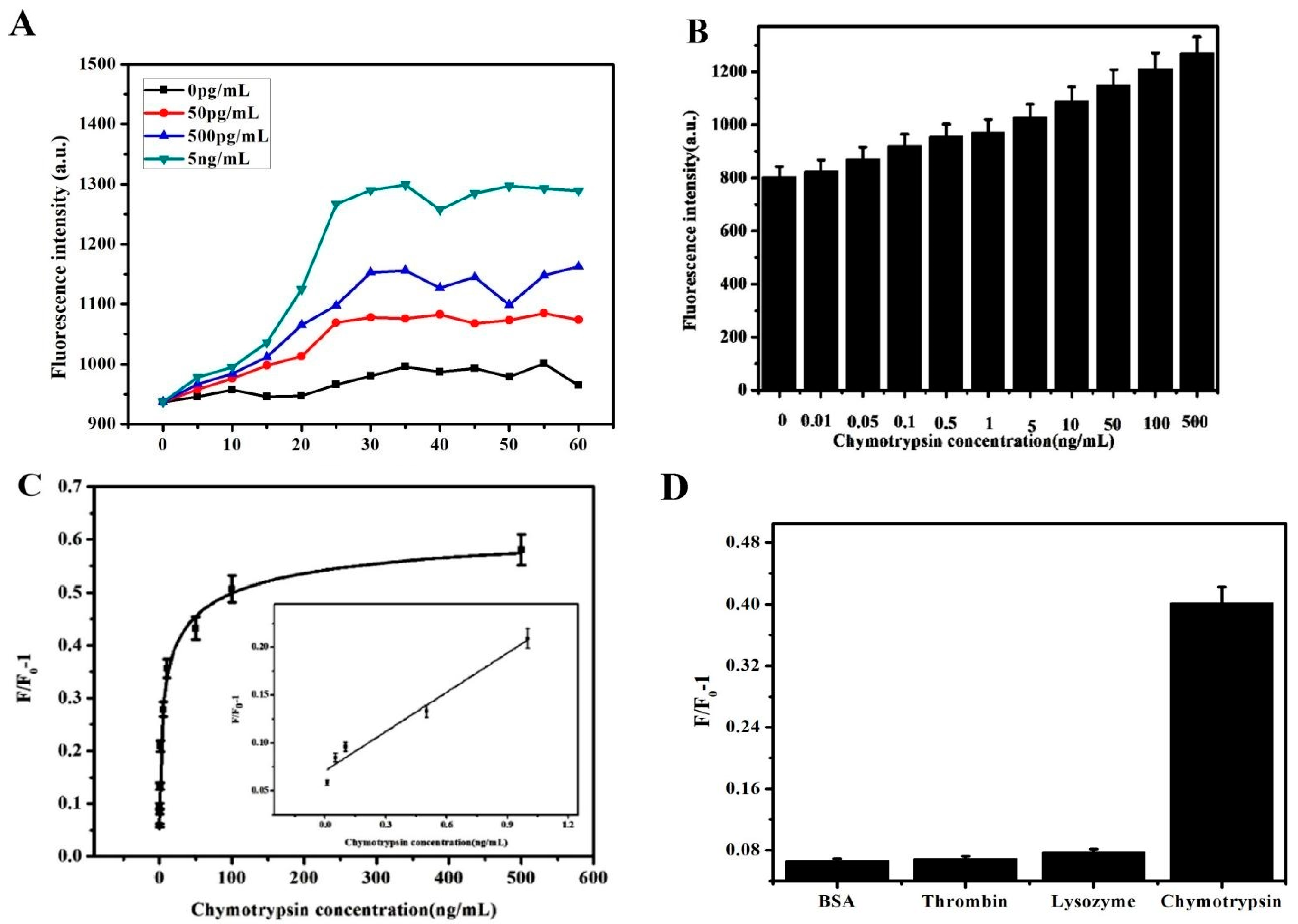
3.4. Kinetic Analysis Based on PCN@GO@AuNPs
3.5. Sensitive Detection Based on PCN@GO@Au NPs
3.6. Selective Analysis Based on PCN@GO@Au NPs
3.7. Application in Real Sample
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Oldziej, A.; Bolkun, L.; Galar, M.; Kalita, J.; Ostrowska, H.; Romaniuk, W.; Kloczko, J. Assessment of proteasome concentration and chymotrypsin-like activity in plasma of patients with newly diagnosed multiple myeloma. Leukemia Res. 2014, 38, 925–930. [Google Scholar] [CrossRef] [PubMed]
- Piovarci, I.; Hianik, T.; Ivanov, I.N. Detection of chymotrypsin by optical and acoustic methods. Biosensors 2021, 11, 63. [Google Scholar] [CrossRef] [PubMed]
- Zou, X.; Zhao, Y.; Lai, C.; Liang, Y.; Lin, W. A non-peptide probe for detecting chymotrypsin activity based on protection-deprotection strategy in living systems. J. Mater. Chem. B 2021, 9, 8417–8423. [Google Scholar] [CrossRef] [PubMed]
- John, G.; Lieb, I.I.; Peter, V.; Gastroenterology, D.J. Pancreatic function testing: Here to stay for the 21st century. World J. Gastroenterol. 2008, 14, 3149–3158. [Google Scholar]
- Zhang, Z.; Luo, L.; Zhu, L.; Ding, Y.; Deng, D.; Wang, Z.J. Aptamer-linked biosensor for thrombin based on AuNPs/thionine-graphene nanocomposite. Analyst 2013, 138, 5365–5370. [Google Scholar] [CrossRef]
- Mu, S.; Xu, Y.; Zhang, Y.; Guo, X.; Li, J.; Wang, Y.; Liu, X.; Zhang, H. A non-peptide NIR fluorescent probe for detection of chymotrypsin and its imaging application. J. Mater. Chem. B 2019, 7, 2974–2980. [Google Scholar] [CrossRef]
- He, L.; Pagneux, Q.; Larroulet, I.; Serrano, A.Y.; Szunerits, S.J.B. Bioelectronics. Label-free femtomolar cancer biomarker detection in human serum using graphene-coated surface plasmon resonance chips. Biosens. Bioelectron. 2017, 89, 606–611. [Google Scholar] [CrossRef]
- Zhu, S.Y.; Zhao, X.E.; Zhang, W.; Liu, Z.Y.; Qi, W.J.; Anjum, S.; Xu, G.B. Fluorescence detection of glutathione reductase activity based on deoxyribonucleic acid-templated silver nanoclusters. Anal. Chim. Acta 2013, 786, 111–115. [Google Scholar] [CrossRef] [PubMed]
- Chaiendoo, K.; Tuntulani, T.; Ngeontae, W. A paper-based ferrous ion sensor fabricated from an ion exchange polymeric membrane coated on a silver nanocluster-impregnated filter paper. Mater. Chem. Phys. 2017, 199, 272–279. [Google Scholar] [CrossRef]
- Zheng, Y.; Lai, L.; Liu, W.; Jiang, H.; Wang, X. Recent advances in biomedical applications of fluorescent gold nanoclusters. Adv. Colloid Interface Sci. 2017, 242, 1. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.; Petryayeva, E.; Algar, W.R. Quantum dot-based concentric FRET configuration for the parallel detection of protease activity and concentration. Anal. Chem. 2014, 86, 11181–11188. [Google Scholar] [CrossRef]
- Wang, X.H.; Geng, J.; Miyoshi, D.; Ren, J.S.; Sugimoto, N.; Qu, X.G. A rapid and sensitive “add-mix-measure” assay for multiple proteinases based on one gold nanoparticle-peptide-fluorophore conjugate. Biosens. Bioelectron. 2010, 26, 743–747. [Google Scholar] [CrossRef]
- Okorochenkova, Y.; Hlavac, J. Novel ratiometric xanthene-based probes for protease detection. Dyes Pigm. 2017, 143, 232–238. [Google Scholar] [CrossRef]
- Okorochenkova, Y.; Porubsky, M.; Benicka, S.; Hlavac, J. A novel three-fluorophore system as a ratiometric sensor for multiple protease detection. Chem. Commun. 2018, 54, 7589–7592. [Google Scholar] [CrossRef]
- Huang, X.L.; He, Z.M.; Guo, D.; Liu, Y.J.; Song, J.B.; Yung, B.C.; Lin, L.S.; Yu, G.C.; Zhu, J.J.; Xiong, Y.H.; et al. Three-in-one” nanohybrids as synergistic nanoquenchers to enhance no-wash fluorescence biosensors for ratiometric detection of cancer biomarkers. Theranostics 2018, 8, 3461–3473. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.S.; Yang, X.Y.; Niu, G.; Song, J.B.; Yang, H.H.; Chen, X.Y. Dual-enhanced photothermal conversion properties of reduced graphene oxide-coated gold superparticles for light-triggered acoustic and thermal theranostics. Nanoscale 2016, 8, 2116–2122. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Lou, Y.; Guo, C.; Jia, Q.; Song, Y.; Tian, J.Y.; Zhang, S.; Wang, M.; He, L.; Du, M. Metal–organic frameworks (MOFs) based chemosensors/biosensors for analysis of food contaminants. Trends Food Sci. Tech. 2021, 118, 569–588. [Google Scholar] [CrossRef]
- Zhao, Y.; Zeng, H.; Zhu, X.W.; Lu, W.; Li, D. Metal-organic frameworks as photoluminescent biosensing platforms: Mechanismsand applications. Chem. Soc. Rev. 2021, 50, 4484–4513. [Google Scholar] [CrossRef]
- Karmakar, A.; Samanta, P.; Dutta, S.; Ghosh, S.K. Fluorescent “turn-on” sensing based on metal-organic frameworks (MOFs). Chem. Asian J. 2019, 14, 4506–4519. [Google Scholar] [CrossRef]
- Wu, F.; Ye, J.H.; Cao, Y.L.; Wang, Z.Y.; Miao, T.T.; Shi, Q. Recent advances in fluorescence sensors based on DNA–MOF hybrids. Luminescence 2020, 35, 437–621. [Google Scholar] [CrossRef]
- Chen, R.; Chen, X.R.; Zhou, T.F.; Lin, T.; Leng, Y.K.; Huang, X.L.; Xiong, Y.H. Three-in-One “Multifunctional nanohybrids with colorimetric magnetic catalytic activities to enhance immunochromatographic diagnosis”. ACS Nano 2022, 16, 3351–3361. [Google Scholar] [CrossRef] [PubMed]
- Das, H.T.; Barai, P.; Dutta, S.; Das, N.; Das, P.; Roy, M.; Alauddin, M.; Barai, H.R. Polymer composites with quantum dots as potential electrode materials for supercapacitors application: A review. Polymers 2022, 14, 1053. [Google Scholar] [CrossRef]
- Li, X.G.; Zhang, F.; Gao, Y.; Zhou, Q.M.; Zhao, Y.; Li, Y.; Huo, J.Z.; Zhao, X.J. Facile synthesis of red emitting 3-aminophenylboronic acid functionalized copper nanoclusters for rapid, selective and highly sensitive detection of glycoproteins. Biosens. Bioelectron. 2016, 86, 270–276. [Google Scholar] [CrossRef]
- Liao, X.Q.; Li, R.Y.; Long, X.H.; Li, Z.J. Ultra sensitive and wide-range pH sensor based on the BSA-capped Cu nanoclusters fabricated by fast synthesis through the use of hydrogen peroxide additive. RSC Adv. 2015, 5, 48835–48841. [Google Scholar]
- Zheng, X.J.; Liang, R.P.; Li, Z.J.; Zhang, L.; Qiu, J.D. One-step, stabilizer-free and green synthesis of Cu nanoclusters as fluorescent probes for sensitive and selective detection of nitrite ions. Sens. Actuators B Chem. 2016, 230, 314. [Google Scholar] [CrossRef]
- Momeni, S.; Ahmadi, R.; Safavi, A.; Nabipour, I. Blue-emitting copper nanoparticles as a fluorescent probe for detection of cyanide ions. Talanta 2017, 175, 514. [Google Scholar] [CrossRef]
- Chen, L.Y.; Luque, R.; Li, Y.W. Controllable design of tunable nanostructures inside metal–organic frameworks. Chem. Soc. Rev. 2017, 46, 4614. [Google Scholar] [CrossRef]
- Volosskiy, B.; Niwa, K.; Chen, Y.; Zhao, Z.P.; Weiss, N.O.; Zhong, X.; Ding, M.N.; Lee, C.; Huang, Y.; Duan, X.F. Metal-organic framework templated synthesis of ultrathin, well-aligned metallic nanowires. ACS Nano 2015, 9, 3044–3049. [Google Scholar] [CrossRef]
- Yang, Q.; Xu, Q.; Jiang, H.L. Metal–organic frameworks meet metal nanoparticles: Synergistic effect for enhanced catalysis. Chem. Soc. Rev. 2017, 46, 4774–4808. [Google Scholar] [CrossRef] [PubMed]
- He, C.B.; Liu, D.M.; Lin, W.B. Nanomedicine applications of hybrid nanomaterials built from metal-ligand coordination bonds: Nanoscale metal-organic frameworks and nanoscale coordination polymers. Chem. Rev. 2015, 115, 11079–11108. [Google Scholar] [CrossRef]
- Bu, X.N.; Fu, Y.X.; Jiang, X.W.; Jin, H.; Gui, R.J. Self-assembly of DNA-templated copper nanoclusters and carbon dots for ratiometric fluorometric and visual determination of arginine and acetaminophen with a logic-gate operation. Microchim. Acta 2020, 187, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Bu, X.N.; Fu, Y.X.; Jin, H.; Gui, R.J. Specific enzymatic synthesis of 2,3-diaminophenazine and copper nanoclusters used for dual-emission ratiometric and naked-eye visual fluorescence sensing of choline. New J. Chem. 2018, 42, 17323–17330. [Google Scholar] [CrossRef]
- Jiang, X.W.; Jin, H.; Sun, Y.J.; Sun, Z.J.; Gui, R.J. Assembly of black phosphorus quantum dots-doped MOF and silver nanoclusters as a versatile enzyme-catalyzed biosensor for solution, flexible substrate and latent fingerprint visual detection of baicalin. Biosens. Bioelectron. 2020, 152, 112012. [Google Scholar] [CrossRef] [PubMed]
- Borse, S.; Murthy, Z.V.P.; Park, T.-J.; Kailasa, S.K. Pepsin mediated synthesis of blue fluorescent copper nanoclusters for sensing of flutamide and chloramphenicol drugs. Microchem. J. 2021, 164, 105947. [Google Scholar] [CrossRef]
- Bhamore, J.R.; Jha, S.; Mungara, A.K.; Singhal, R.K.; Sonkeshariya, D.; Kailasa, S.K. One-step green synthetic approach for the preparation of multicolor emitting copper nanoclusters and their applications in chemical species sensing and bioimaging. Biosens. Bioelectron. 2016, 80, 243–248. [Google Scholar] [CrossRef]
- Bhamore, J.R.; Deshmukh, B.; Haran, V.; Jha, S.; Singhal, R.K.; Lenka, N.; Kailasa, S.K.; Murthy, Z.V.P. One-step eco-friendly approach for the fabrication of synergistically engineered fluorescent copper nanoclusters: Sensing of Hg2+ ion and cellular uptake and bioimaging properties. New J. Chem. 2018, 42, 1510–1520. [Google Scholar] [CrossRef]
- Wang, Y.; Cui, M.; Jiao, M.X.; Luo, X.L. Antifouling and ultrasensitive biosensing interface based on self-assembled peptide and aptamer on macroporous gold for electrochemical detection of immunoglobulin E in serum. Anal. Bioanal. Chem. 2018, 410, 5871–5878. [Google Scholar] [CrossRef]
- Wang, M.K.; Wang, S.; Su, D.D.; Su, X.G. Copper nanoclusters/polydopamine nanospheres based fluorescence aptasensor for protein kinase activity determination. Anal. Chim. Acta 2018, 1035, 184–191. [Google Scholar] [CrossRef]
- Qian, H.S.; Huang, Y.; Duan, X.L.; Wei, X.T.; Fan, Y.P.; Gan, D.L.; Yue, S.J.; Cheng, W.; Chen, T.M. Fiber optic surface plasmon resonance biosensor for detection of PDGF-BB in serum based on self-assembled aptamer and antifouling peptide monolayer. Biosens. Bioelectron. 2019, 140, 111350. [Google Scholar] [CrossRef]
- Sun, H.; Panicker, R.C.; Shao, Q.Y. Activity based fingerprinting of proteases using FRET peptides. Biopolymers 2010, 88, 141–149. [Google Scholar] [CrossRef]
- Bhunia, S.K.; Jana, N.R. Peptide-functionalized colloidal graphene via interdigited bilayer coating and fluorescence turn-on detection of enzyme. ACS Appl. Mater. Interfaces 2011, 3, 3335–3341. [Google Scholar] [CrossRef]
- Miao, H.; Wang, L.; Zhuo, Y.; Zhou, Z.N.; Yang, X.M. Bioelectronics. Label-free fluorimetric detection of CEA using carbon dots derived from tomato juice. Biosens. Bioelectron. 2016, 86, 83–89. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.J.; Wei, Z.K.; Luo, X.D.; Wan, Q.; Qiu, R.L.; Wang, S.Z. An ultrasensitive homogeneous aptasensor for carcinoembryonic antigen based on upconversion fluorescence resonance energy transfer. Talanta 2019, 195, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Cao, J.; Jiang, X.; Pan, Z.; Fu, N. A sensitive ratiometric fluorescence probe for chymotrypsin activity and inhibitor screening. Sens. Actuators B Chem. 2018, 273, 204–210. [Google Scholar] [CrossRef]
- Li, S.Q.; Fu, Y.W.; Ma, X.J.; Zhang, Y.D. Label-free fluorometric detection of chymotrypsin activity using graphene oxide/nucleic-acid-stabilized silver nanoclusters hybrid materials. Biosens. Bioelectron. 2017, 88, 210–216. [Google Scholar] [CrossRef] [PubMed]
| Method | Analyst | Detection Limit | Reference |
|---|---|---|---|
| Fluorescence detection | AuNPs/peptide | 0.095 ng mL−1 | [12] |
| Fluorescence detection | Fluorophore/peptide | 10 ng mL−1 | [40] |
| Fluorescence detection | Colloidal GO | 0.0475 ng mL−1 | [41] |
| Fluorescence detection | carbon dots | 0.3 ng mL−1 | [42] |
| Fluorescence detection | GO/up-conversion nanoparticles | 7.9 pg mL−1 | [43] |
| Fluorescence detection | Ratiometric fluorescence probe | 8.4 ng mL−1 | [44] |
| Fluorescence detection | GO/dC12-AgNCs | 3 ng mL−1 | [45] |
| Fluorescence detection | PCN@GO@AuNPs/CuNCs | 3.91 pg mL−1 | This study |
| Samples | Added (pg mL−1) | Obtained (pg mL−1) | Recovery (%) | RSD (%) |
|---|---|---|---|---|
| 1 | 0 | 0 | -- | -- |
| 2 | 10 | 11.004 | 110.04 | 1.92 |
| 3 | 50 | 48.012 | 96.02 | 2.43 |
| 4 | 100 | 103.524 | 103.52 | 1.41 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, L.; Liu, C.; Gao, L. Highly Sensitive Detection of Chymotrypsin Based on Metal Organic Frameworks with Peptides Sensors. Biosensors 2023, 13, 263. https://doi.org/10.3390/bios13020263
Liu L, Liu C, Gao L. Highly Sensitive Detection of Chymotrypsin Based on Metal Organic Frameworks with Peptides Sensors. Biosensors. 2023; 13(2):263. https://doi.org/10.3390/bios13020263
Chicago/Turabian StyleLiu, Lei, Cheng Liu, and Li Gao. 2023. "Highly Sensitive Detection of Chymotrypsin Based on Metal Organic Frameworks with Peptides Sensors" Biosensors 13, no. 2: 263. https://doi.org/10.3390/bios13020263
APA StyleLiu, L., Liu, C., & Gao, L. (2023). Highly Sensitive Detection of Chymotrypsin Based on Metal Organic Frameworks with Peptides Sensors. Biosensors, 13(2), 263. https://doi.org/10.3390/bios13020263




