Wearable Electrocardiography for Physical Activity Monitoring: Definition of Validation Protocol and Automatic Classification
Abstract
1. Introduction
2. Materials and Methods
- 4 tests in a resting condition;
- 1 test after walking on a treadmill for 2 min at 3 km/h (with 0-slope);
- 1 test after walking on a treadmill for 2 min at 8 km/h (with 0-slope);
- 1 test after walking on a treadmill for 2 min at 10 km/h (with 0-slope).
2.1. Acquisition Devices
2.2. Data Analysis for Metrological Characterization
- Distribution of deviations: it is expected that measurement differences assume a Gaussian-like distribution. Both mean (µ) and standard deviation (σ) values of such experimental measurement differences are computed, being associated with measurement accuracy and precision, respectively;
- Analysis of agreement, which is conducted through the Bland–Altman plot [36]. This graph plots the measurement differences with respect to the expected measurement value (i.e., the mean value between test and reference measurements). In this way, it is possible to derive the confidence interval at 95% (CI95%) of the levels of agreement (meaningful of the statistical confidence of the measurement), as well as the bias (the mean value of the measurement differences—substantially coincident with the µ value obtained from the distribution of deviations);
- Correlation between the test and reference signals and computation of the related Pearson’s correlation coefficient (ρ). Specifically, the strength of the linear correlation is evaluated as strong for ρ > 0.7, moderate for 0.3 < ρ < 0.7, and low for ρ < 0.3 [37].
2.3. ML-Based Classification
- Support vector machine (SVM) [38]: it is based on statistical learning frameworks. It sets a model assigning an item to a category in a non-probabilistic classification setting, maximizing the gap between different categories;
- Random forest (RF) [39]: it realizes different decision trees in the training phase. It corrects overfitting issues and usually outperforms decision trees;
- Simple logistic (SL) [40]: it builds a linear logistic regression model based on the determination of the relationship between two variables, fitting a logistic curve to binary data;
- Decision table (DT) [41]: it can be used for a numeric prediction starting from decision trees. It is based on if–then rules and is more compact than decision trees;
- Naïve Bayes (NB) [42]: it assumes that a feature value does not depend on the others and that there are no correlations among them. It uses the method of maximum likelihood, with or without Bayesian probability methods.
- Mean value and standard deviation of HR;
- The root mean square of successive inter-beat intervals (RMSSD);
- The percentage of successive RR intervals (i.e., IBIs, intended as differences between two consequent R peaks) differing for a value greater than 50 ms (pNN50).
- Input layer;
- Bidirectional-LSTM (BiLSTM) with 100 hidden units;
- 9 fully connected layer;
- Softmax;
- Output layer.
3. Results
3.1. Measurement Accuracy and Precision
3.2. Physical Activity Level Classification
4. Discussion
4.1. Measurement Accuracy and Precision
4.2. Physical Activity Level Classification
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Vijayan, V.; Connolly, J.P.; Condell, J.; McKelvey, N.; Gardiner, P. Review of Wearable Devices and Data Collection Considerations for Connected Health. Sensors 2021, 21, 5589. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Li, Y.; Zhang, S.; Shahabi, F.; Xia, S.; Deng, Y.; Alshurafa, N. Deep Learning in Human Activity Recognition with Wearable Sensors: A Review on Advances. Sensors 2022, 22, 1476. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.-W.; O’Brien, M.K.; Horin, A.P.; Koch, L.L.M.; Lee, J.Y.; Xu, S.; Zee, P.C.; Arora, V.M.; Jayaraman, A. Sleep Monitoring during Acute Stroke Rehabilitation: Toward Automated Measurement Using Multimodal Wireless Sensors. Sensors 2022, 22, 6190. [Google Scholar] [CrossRef] [PubMed]
- Santucci, F.; Presti, D.L.; Massaroni, C.; Schena, E.; Setola, R. Precordial Vibrations: A Review of Wearable Systems, Signal Processing Techniques, and Main Applications. Sensors 2022, 22, 5805. [Google Scholar] [CrossRef]
- Cosoli, G.; Mansi, S.A.; Arnesano, M. Combined use of wearable devices and Machine Learning for the measurement of thermal sensation in indoor environments. In Proceedings of the 2022 IEEE International Workshop on Metrology for Living Environment (MetroLivEn), Cosenza, Italy, 25–27 May 2022; pp. 1–6. [Google Scholar] [CrossRef]
- Carey, L.; Stanwell, P.; Terry, D.P.; McIntosh, A.S.; Caswell, S.V.; Iverson, G.L.; Gardner, A.J. Verifying Head Impacts Recorded by a Wearable Sensor using Video Footage in Rugby League: A Preliminary Study. Sports Med. Open 2019, 5, 9. [Google Scholar] [CrossRef]
- Di Paolo, S.; Zaffagnini, S.; Pizza, N.; Grassi, A.; Bragonzoni, L. Poor Motor Coordination Elicits Altered Lower Limb Biomechanics in Young Football (Soccer) Players: Implications for Injury Prevention through Wearable Sensors. Sensors 2021, 21, 4371. [Google Scholar] [CrossRef]
- Cosoli, G.; Antognoli, L.; Veroli, V.; Scalise, L. Accuracy and Precision of Wearable Devices for Real-Time Monitoring of Swimming Athletes. Sensors 2022, 22, 4726. [Google Scholar] [CrossRef]
- Wu, M.; Wang, R.; Hu, Y.; Fan, M.; Wang, Y.; Li, Y.; Wu, S. Invisible experience to real-time assessment in elite tennis athlete training: Sport-specific movement classification based on wearable MEMS sensor data. Proc. Inst. Mech. Eng. Part P J. Sports Eng. Technol. 2021. [Google Scholar] [CrossRef]
- Adams, J.L.; Dinesh, K.; Snyder, C.W.; Xiong, M.; Tarolli, C.G.; Sharma, S.; Dorsey, E.R.; Sharma, G. A real-world study of wearable sensors in Parkinson’s disease. NPJ Parkinsons Dis. 2021, 7, 106. [Google Scholar] [CrossRef]
- Mughal, H.; Javed, A.R.; Rizwan, M.; Almadhor, A.S.; Kryvinska, N. Parkinson’s Disease Management via Wearable Sensors: A Systematic Review. IEEE Access 2022, 10, 35219–35237. [Google Scholar] [CrossRef]
- Wilson, R.; Vangala, S.; Elashoff, D.; Safari, T.; Smith, B. Using Wearable Sensor Technology to Measure Motion Complexity in Infants at High Familial Risk for Autism Spectrum Disorder. Sensors 2021, 21, 616. [Google Scholar] [CrossRef] [PubMed]
- Hinde, K.; White, G.; Armstrong, N. Wearable Devices Suitable for Monitoring Twenty Four Hour Heart Rate Variability in Military Populations. Sensors 2021, 21, 1061. [Google Scholar] [CrossRef] [PubMed]
- Mohapatra, P.; Premkumar, P.S.; Sivaprakasam, M. A Yellow–Orange Wavelength-Based Short-Term Heart Rate Variability Measurement Scheme for Wrist-Based Wearables. IEEE Trans. Instrum. Meas. 2018, 67, 1091–1101. [Google Scholar] [CrossRef]
- Rahman, M.; Morshed, B.I. Extraction of Respiration Rate from Wrist ECG Signals. In Proceedings of the 2021 IEEE 12th Annual Ubiquitous Computing, Electronics & Mobile Communication Conference (UEMCON), New York, NY, USA, 1–4 December 2021; pp. 565–570. [Google Scholar] [CrossRef]
- Singh, P.; Kaur, G.; Kaur, D. Infant Monitoring System Using Wearable Sensors Based on Blood Oxygen Saturation: A Review. In Intelligent, Secure, and Dependable Systems in Distributed and Cloud Environments; Springer: Cham, Switzerland, 2017; pp. 162–168. [Google Scholar]
- Cvetkovic, B.; Milic, R.; Lustrek, M. Estimating Energy Expenditure with Multiple Models Using Different Wearable Sensors. IEEE J. Biomed. Health Informatics 2016, 20, 1081–1087. [Google Scholar] [CrossRef]
- Arakawa, T. Recent Research and Developing Trends of Wearable Sensors for Detecting Blood Pressure. Sensors 2018, 18, 2772. [Google Scholar] [CrossRef]
- Poli, A.; Cosoli, G.; Iadarola, G.; Spinsante, S.; Scalise, L. Feasibility of Blood Pressure Measurement through Wearable Devices: Analysis of Smartwatches Performance. In Proceedings of the 2022 IEEE International Symposium on Medical Measurements and Applications (MeMeA), Messina, Italy, 22–24 June 2022; pp. 1–6. [Google Scholar] [CrossRef]
- Prigent, G.; Aminian, K.; Rodrigues, T.; Vesin, J.-M.; Millet, G.P.; Falbriard, M.; Meyer, F.; Paraschiv-Ionescu, A. Indirect Estimation of Breathing Rate from Heart Rate Monitoring System during Running. Sensors 2021, 21, 5651. [Google Scholar] [CrossRef] [PubMed]
- Cosoli, G.; Antognoli, L.; Scalise, L. Indirect Estimation of Breathing Rate through Wearable Devices. In Proceedings of the 2022 IEEE International Symposium on Medical Measurements and Applications (MeMeA), Messina, Italy, 22–24 June 2022; pp. 1–6. [Google Scholar] [CrossRef]
- Dong, Q. Leakage Prediction in Machine Learning Models When Using Data from Sports Wearable Sensors. Comput. Intell. Neurosci. 2022, 2022, 5314671. [Google Scholar] [CrossRef] [PubMed]
- Kunze, K.N.; Orr, M.; Krebs, V.; Bhandari, M.; Piuzzi, N.S. Potential benefits, unintended consequences, and future roles of artificial intelligence in orthopaedic surgery research. Bone Jt. Open 2022, 3, 93–97. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Yan, W.; Lan, K.; Ma, C.; Wu, D.; Wu, A.; Yang, Z.; Wang, J.; Zang, Y.; Yan, M.; et al. Assessing Electrocardiogram and Respiratory Signal Quality of a Wearable Device (SensEcho): Semisupervised Machine Learning-Based Validation Study. JMIR mHealth uHealth 2021, 9, e25415. [Google Scholar] [CrossRef]
- Ebrahimi, Z.; Loni, M.; Daneshtalab, M.; Gharehbaghi, A. A review on deep learning methods for ECG arrhythmia classification. Expert Syst. Appl. X 2020, 7, 100033. [Google Scholar] [CrossRef]
- Rana, A.; Kim, K.K. ECG Heartbeat Classification Using a Single Layer LSTM Model. In Proceedings of the 2019 International SoC Design Conference (ISOCC), Jeju, Republic of Korea, 6–9 October 2019; pp. 267–268. [Google Scholar] [CrossRef]
- Mekruksavanich, S.; Jitpattanakul, A. Smartwatch-based Human Activity Recognition Using Hybrid LSTM Network. In Proceedings of the 2020 IEEE SENSORS, Rotterdam, The Netherlands, 25–28 October 2020; pp. 1–4. [Google Scholar] [CrossRef]
- Amor, J.D.; James, C.J. Validation of a Commercial Android Smartwatch as an Activity Monitoring Platform. IEEE J. Biomed. Health Informatics 2018, 22, 968–978. [Google Scholar] [CrossRef] [PubMed]
- Kheirkhahan, M.; Nair, S.; Davoudi, A.; Rashidi, P.; Wanigatunga, A.A.; Corbett, D.B.; Mendoza, T.; Manini, T.M.; Ranka, S. A smartwatch-based framework for real-time and online assessment and mobility monitoring. J. Biomed. Informatics 2019, 89, 29–40. [Google Scholar] [CrossRef] [PubMed]
- Izmailova, E.S.; McLean, I.L.; Hather, G.; Merberg, D.; Homsy, J.; Cantor, M.; Volfson, D.; Bhatia, G.; Perakslis, E.D.; Benko, C.; et al. Continuous Monitoring Using a Wearable Device Detects Activity-Induced Heart Rate Changes after Administration of Amphetamine. Clin. Transl. Sci. 2019, 12, 677–686. [Google Scholar] [CrossRef] [PubMed]
- Moraes, C.; Cambras, T.; Diez-Noguera, A.; Schimitt, R.; Dantas, G.; Levandovski, R.; Hidalgo, M.P. A new chronobiological approach to discriminate between acute and chronic depression using peripheral temperature, rest-activity, and light exposure parameters. BMC Psychiatry 2013, 13, 77. [Google Scholar] [CrossRef]
- WMA Declaration of Helsinki—Ethical Principles for Medical Research Involving Human Subjects—WMA—The World Medical Association. Available online: https://www.wma.net/policies-post/wma-declaration-of-helsinki-ethical-principles-for-medical-research-involving-human-subjects/ (accessed on 9 December 2020).
- Samsung Galaxy Watch 3|Samsung UK. Available online: https://www.samsung.com/uk/watches/galaxy-watch/galaxy-watch3-45mm-mystic-black-lte-sm-r845fzkaeua/ (accessed on 11 November 2022).
- Zephyr BioHarness BTLE ECHO Module (Gen 3)|HaB Direct. Available online: https://www.habdirect.co.uk/product/zephyr-bioharness-btle-echo-module-gen-3/ (accessed on 11 November 2022).
- BioHarness 3.0 User Manual. 2012. Available online: www.zephyanywhere.com (accessed on 28 December 2022).
- Altman, D.G.; Bland, J.M. Measurement in Medicine: The Analysis of Method Comparison Studies. J. R. Stat. Soc. Ser. D Stat. 1983, 32, 307–317. Available online: https://www.jstor.org/stable/2987937 (accessed on 2 March 2022). [CrossRef]
- Akoglu, H. User’s guide to correlation coefficients. Turk. J. Emerg. Med. 2018, 18, 91–93. [Google Scholar] [CrossRef]
- Ifenthaler, D.; Widanapathirana, C. Development and Validation of a Learning Analytics Framework: Two Case Studies Using Support Vector Machines. Technol. Knowl. Learn. 2014, 19, 221–240. [Google Scholar] [CrossRef]
- Balyan, A.K.; Ahuja, S.; Lilhore, U.K.; Sharma, S.K.; Manoharan, P.; Algarni, A.D.; Elmannai, H.; Raahemifar, K. A Hybrid Intrusion Detection Model Using EGA-PSO and Improved Random Forest Method. Sensors 2022, 22, 5986. [Google Scholar] [CrossRef]
- Kirasich, K.; Smith, T.; Sadler, B. Random Forest vs. Logistic Regression: Binary Classification for Heterogeneous Datasets. SMU Data Sci. Rev. 2018, 1, 3. Available online: https://scholar.smu.edu/datasciencereview/vol1/iss3/9 (accessed on 27 December 2022).
- Huysmans, J.; Dejaeger, K.; Mues, C.; Vanthienen, J.; Baesens, B. An empirical evaluation of the comprehensibility of decision table, tree and rule based predictive models. Decis. Support Syst. 2011, 51, 141–154. [Google Scholar] [CrossRef]
- Jiang, L.; Zhang, L.; Li, C.; Wu, J. A Correlation-Based Feature Weighting Filter for Naive Bayes. IEEE Trans. Knowl. Data Eng. 2019, 31, 201–213. [Google Scholar] [CrossRef]
- Cook, D.J. Activity learning: Discovering, Recognizing, and Predicting Human Behavior from Sensor Data. Available online: https://www.wiley.com/en-gb/Activity+Learning%3A+Discovering%2C+Recognizing%2C+and+Predicting+Human+Behavior+from+Sensor+Data-p-9781118893760 (accessed on 14 February 2022).
- Hall, M.; Frank, E.; Holmes, G.; Pfahringer, B.; Reutemann, P.; Witten, I.H. The WEKA data mining software: An update. SIGKDD Explor. Newsl. 2009, 11, 10–18. [Google Scholar] [CrossRef]
- Stavropoulos, T.G.; Papastergiou, A.; Mpaltadoros, L.; Nikolopoulos, S.; Kompatsiaris, I. IoT Wearable Sensors and Devices in Elderly Care: A Literature Review. Sensors 2020, 20, 2826. [Google Scholar] [CrossRef] [PubMed]
- Mamdiwar, S.D.; R, A.; Shakruwala, Z.; Chadha, U.; Srinivasan, K.; Chang, C.-Y. Recent Advances on IoT-Assisted Wearable Sensor Systems for Healthcare Monitoring. Biosensors 2021, 11, 372. [Google Scholar] [CrossRef] [PubMed]
- Majumder, S.; Mondal, T.; Deen, M.J. Wearable Sensors for Remote Health Monitoring. Sensors 2017, 17, 130. [Google Scholar] [CrossRef] [PubMed]
- Pillai, S.; Upadhyay, A.; Sayson, D.; Nguyen, B.H.; Tran, S.D. Advances in Medical Wearable Biosensors: Design, Fabrication and Materials Strategies in Healthcare Monitoring. Molecules 2022, 27, 165. [Google Scholar] [CrossRef] [PubMed]
- E4 Wristband|Real-Time Physiological Signals|Wearable PPG, EDA, Temperature, Motion Sensors. Available online: https://www.empatica.com/en-eu/research/e4/ (accessed on 1 December 2020).
- Cosoli, G.; Poli, A.; Antognoli, L.; Spinsante, S.; Scalise, L. What is my heart rate right now? Comparing data from different devices. In Proceedings of the 2022 IEEE International Instrumentation and Measurement Technology Conference (I2MTC), Ottawa, ON, Canada, 16–19 May 2022. [Google Scholar]
- Sequeira, N.; D’Souza, D.; Angaran, P.; Aves, T.; Dorian, P. Common wearable devices demonstrate variable accuracy in measuring heart rate during supraventricular tachycardia. Heart Rhythm. 2020, 17, 854–859. [Google Scholar] [CrossRef]
- Erdaş, Ç.B.; Güney, S. Human Activity Recognition by Using Different Deep Learning Approaches for Wearable Sensors. Neural Process. Lett. 2021, 53, 1795–1809. [Google Scholar] [CrossRef]
- Cosoli, G.; Poli, A.; Scalise, L.; Spinsante, S. Measurement of multimodal physiological signals for stimulation detection by wearable devices. Measurement 2021, 184, 109966. [Google Scholar] [CrossRef]
- Bijalwan, V.; Semwal, V.B.; Gupta, V. Wearable sensor-based pattern mining for human activity recognition: Deep learning approach. Ind. Robot. Int. J. Robot. Res. Appl. 2022, 49, 21–33. [Google Scholar] [CrossRef]
- Bleser, G.; Steffen, D.; Reiss, A.; Weber, M.; Hendeby, G.; Fradet, L. Personalized physical activity monitoring using wearable sensors. In Smart Health; Lecture Notes in Computer Science; Springer: Cham, Switzerland, 2015; Volume 8700, pp. 99–124. [Google Scholar] [CrossRef]
- Seshadri, D.R.; Li, R.T.; Voos, J.E.; Rowbottom, J.R.; Alfes, C.M.; Zorman, C.A.; Drummond, C.K. Wearable sensors for monitoring the internal and external workload of the athlete. NPJ Digit. Med. 2019, 2, 71. [Google Scholar] [CrossRef] [PubMed]
- Ranieri, C.M.; MacLeod, S.; Dragone, M.; Vargas, P.A.; Romero, R.A.F. Activity Recognition for Ambient Assisted Living with Videos, Inertial Units and Ambient Sensors. Sensors 2021, 21, 768. [Google Scholar] [CrossRef] [PubMed]
- Donati, M.; Olivelli, M.; Giovannini, R.; Fanucci, L. RT-PROFASY: Enhancing the Well-being, Safety and Productivity of Workers by Exploiting Wearable Sensors and Artificial Intelligence. In Proceedings of the 2022 IEEE International Workshop on Metrology for Industry 4.0 & IoT (MetroInd4.0&IoT), Trento, Italy, 7–9 June 2022; pp. 69–74. [Google Scholar] [CrossRef]
- Victory, A.; Letkiewicz, A.; Cochran, A.L. Digital solutions for shaping mood and behavior among individuals with mood disorders. Curr. Opin. Syst. Biol. 2020, 21, 25–31. [Google Scholar] [CrossRef]
- Taj-Eldin, M.; Ryan, C.; O’Flynn, B.; Galvin, P. A Review of Wearable Solutions for Physiological and Emotional Monitoring for Use by People with Autism Spectrum Disorder and Their Caregivers. Sensors 2018, 18, 4271. [Google Scholar] [CrossRef]
- Buchman, A.S.; Dawe, R.J.; Leurgans, S.E.; Curran, T.A.; Truty, T.; Yu, L.; Barnes, L.L.; Hausdorff, J.M.; Bennett, D.A. Different Combinations of Mobility Metrics Derived from a Wearable Sensor Are Associated with Distinct Health Outcomes in Older Adults. Journals Gerontol. Ser. A 2012, 75, 1176–1183. [Google Scholar] [CrossRef] [PubMed]
- Madrid-Navarro, C.J.; Sevilla, F.E.; Mínguez-Castellanos, A.; Campos, M.; Ruiz-Abellán, F.; Madrid, J.A.; Rol, M.A. Multidimensional Circadian Monitoring by Wearable Biosensors in Parkinson’s Disease. Front. Neurol. 2018, 9, 157. [Google Scholar] [CrossRef] [PubMed]
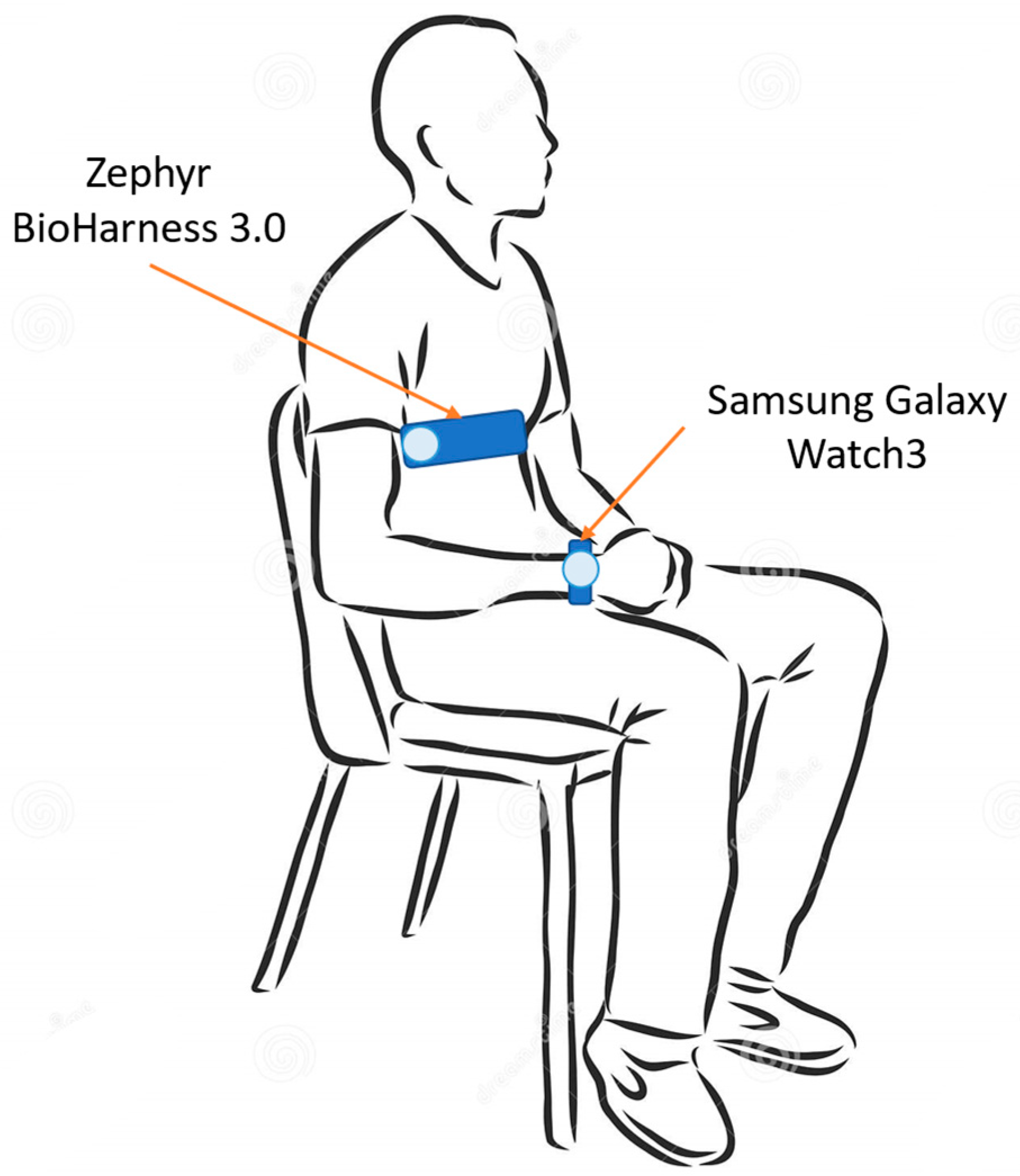
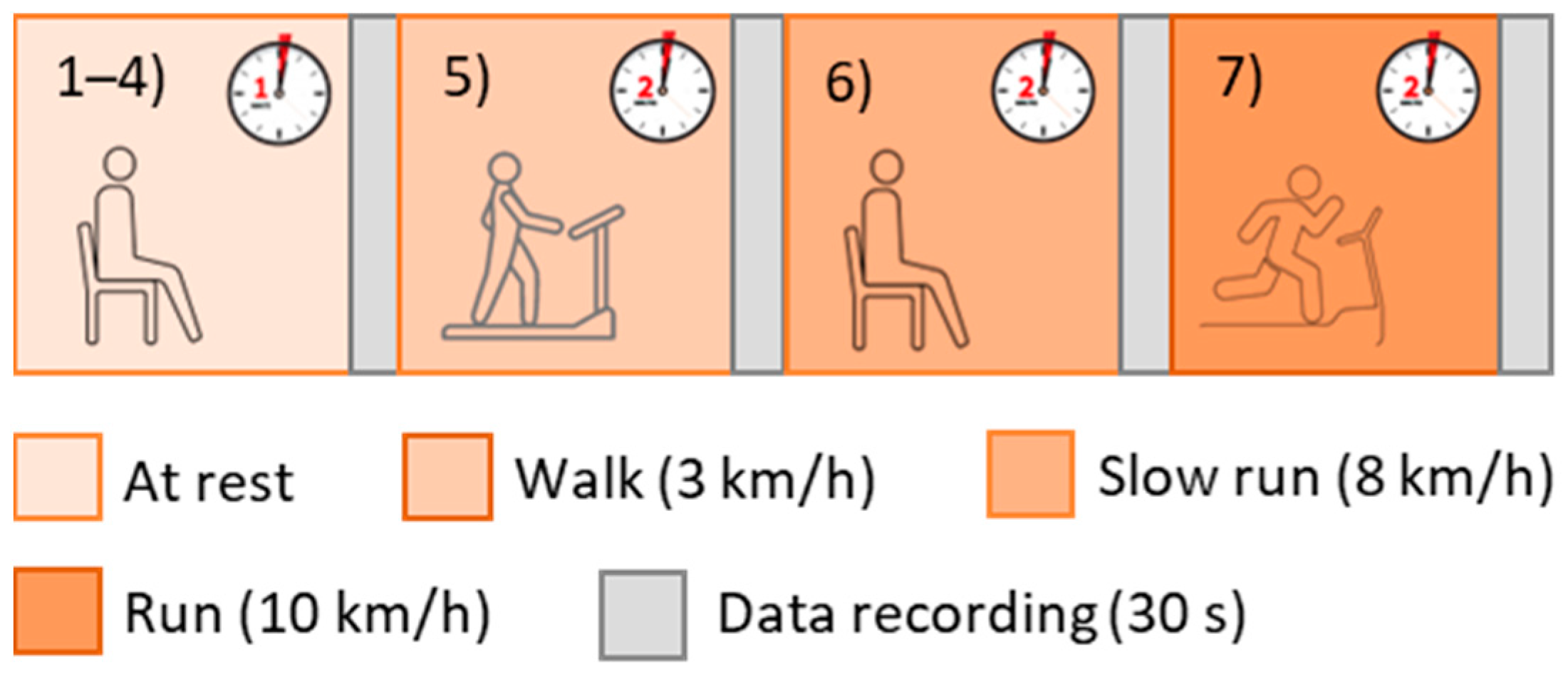

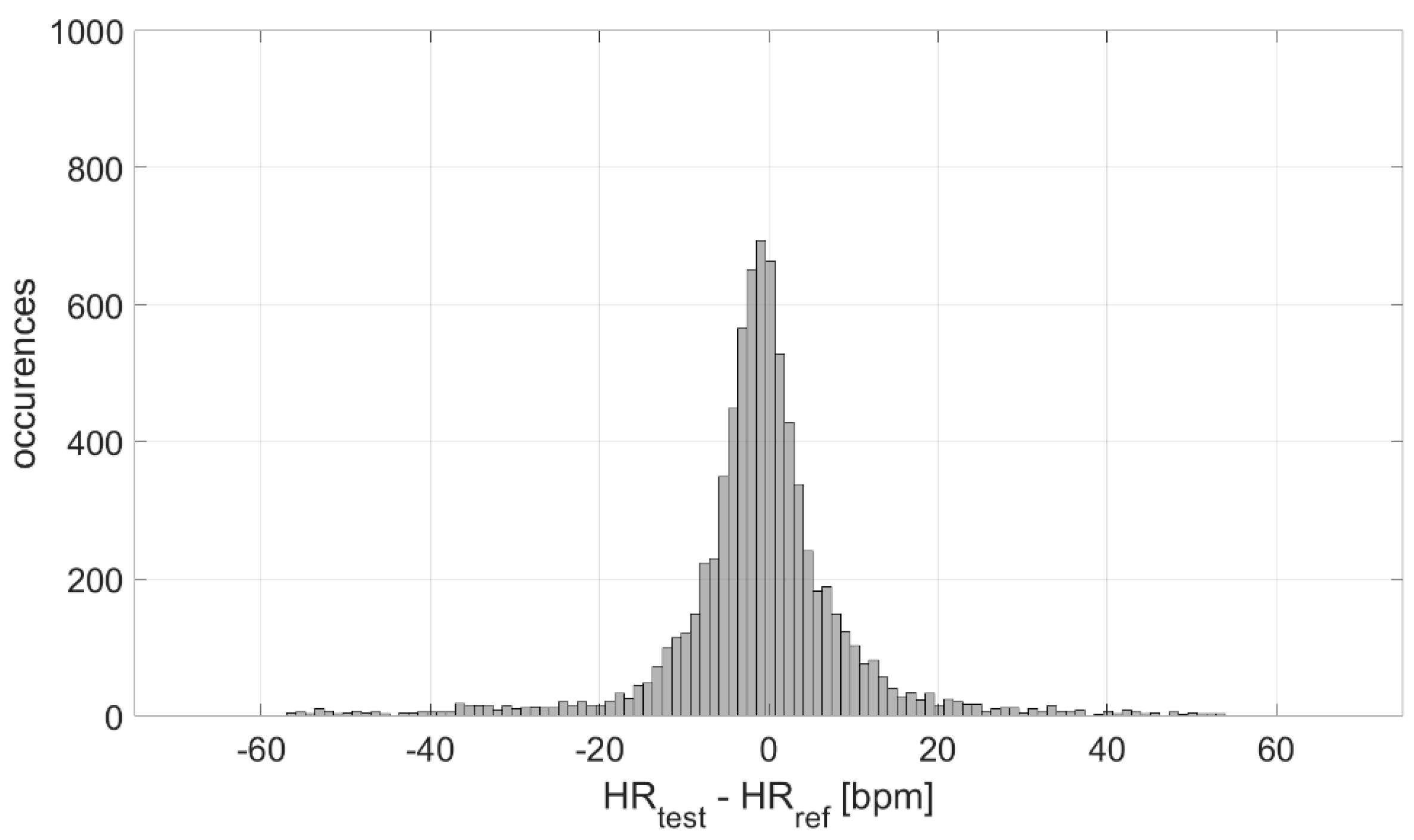


| Mean | Standard Deviation | Confidence Interval at 95% | |
|---|---|---|---|
| Age | 22 years | 3 years | (21, 23) years |
| Weight | 64 kg | 9 kg | (61, 67) kg |
| Height | 1.69 m | 0.07 m | (1.67, 1.71) m |
| BMI | 22.5 kg/m2 | 2.3 kg/m2 | (21.6, 23.3) kg/m2 |
| Model | Accuracy | Recall | Precision | F-Measure | ROC Area | TP | TN | FP | FN |
|---|---|---|---|---|---|---|---|---|---|
| SVM | 0.78 | 0.78 | 0.83 | 0.78 | 0.80 | 26 | 23 | 12 | 1 |
| RF | 0.81 | 0.80 | 0.81 | 0.80 | 0.84 | 24 | 26 | 9 | 3 |
| SL | 0.70 | 0.70 | 0.70 | 0.70 | 0.79 | 19 | 25 | 10 | 8 |
| DT | 0.66 | 0.66 | 0.67 | 0.64 | 0.80 | 11 | 30 | 5 | 16 |
| NB | 0.76 | 0.76 | 0.82 | 0.76 | 0.76 | 27 | 21 | 14 | 1 |
| LSTM | 0.79 | 0.62 | 0.73 | 0.67 | 0.84 | 1180 | 442 | 276 | 166 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cosoli, G.; Antognoli, L.; Scalise, L. Wearable Electrocardiography for Physical Activity Monitoring: Definition of Validation Protocol and Automatic Classification. Biosensors 2023, 13, 154. https://doi.org/10.3390/bios13020154
Cosoli G, Antognoli L, Scalise L. Wearable Electrocardiography for Physical Activity Monitoring: Definition of Validation Protocol and Automatic Classification. Biosensors. 2023; 13(2):154. https://doi.org/10.3390/bios13020154
Chicago/Turabian StyleCosoli, Gloria, Luca Antognoli, and Lorenzo Scalise. 2023. "Wearable Electrocardiography for Physical Activity Monitoring: Definition of Validation Protocol and Automatic Classification" Biosensors 13, no. 2: 154. https://doi.org/10.3390/bios13020154
APA StyleCosoli, G., Antognoli, L., & Scalise, L. (2023). Wearable Electrocardiography for Physical Activity Monitoring: Definition of Validation Protocol and Automatic Classification. Biosensors, 13(2), 154. https://doi.org/10.3390/bios13020154







