Two Biosensors for the Determination of Interleukin-6 in Blood Plasma by Array SPRi
Abstract
:1. Introduction
2. Methods
2.1. Materials
2.1.1. Reagents
2.1.2. Biological Samples
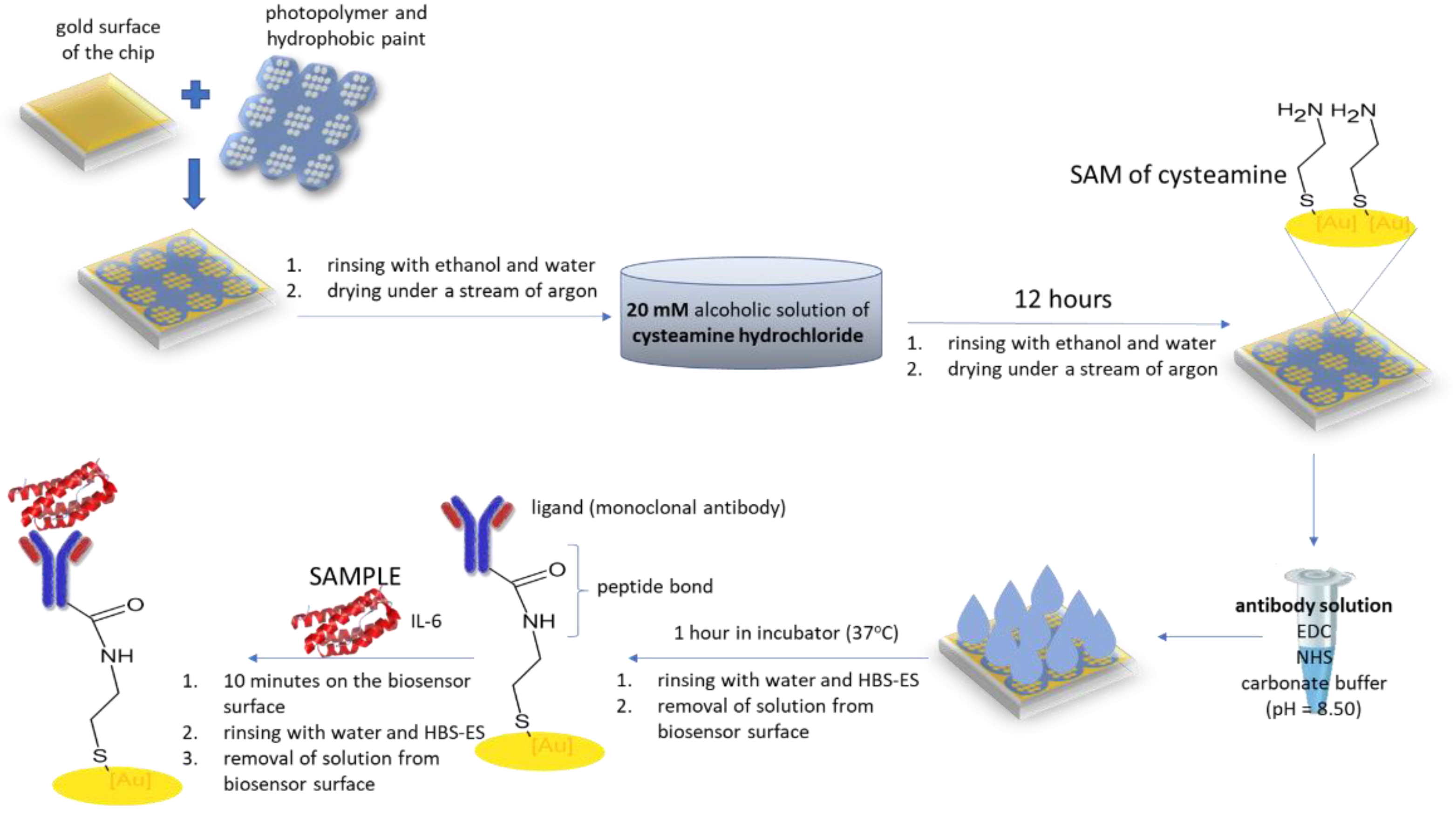
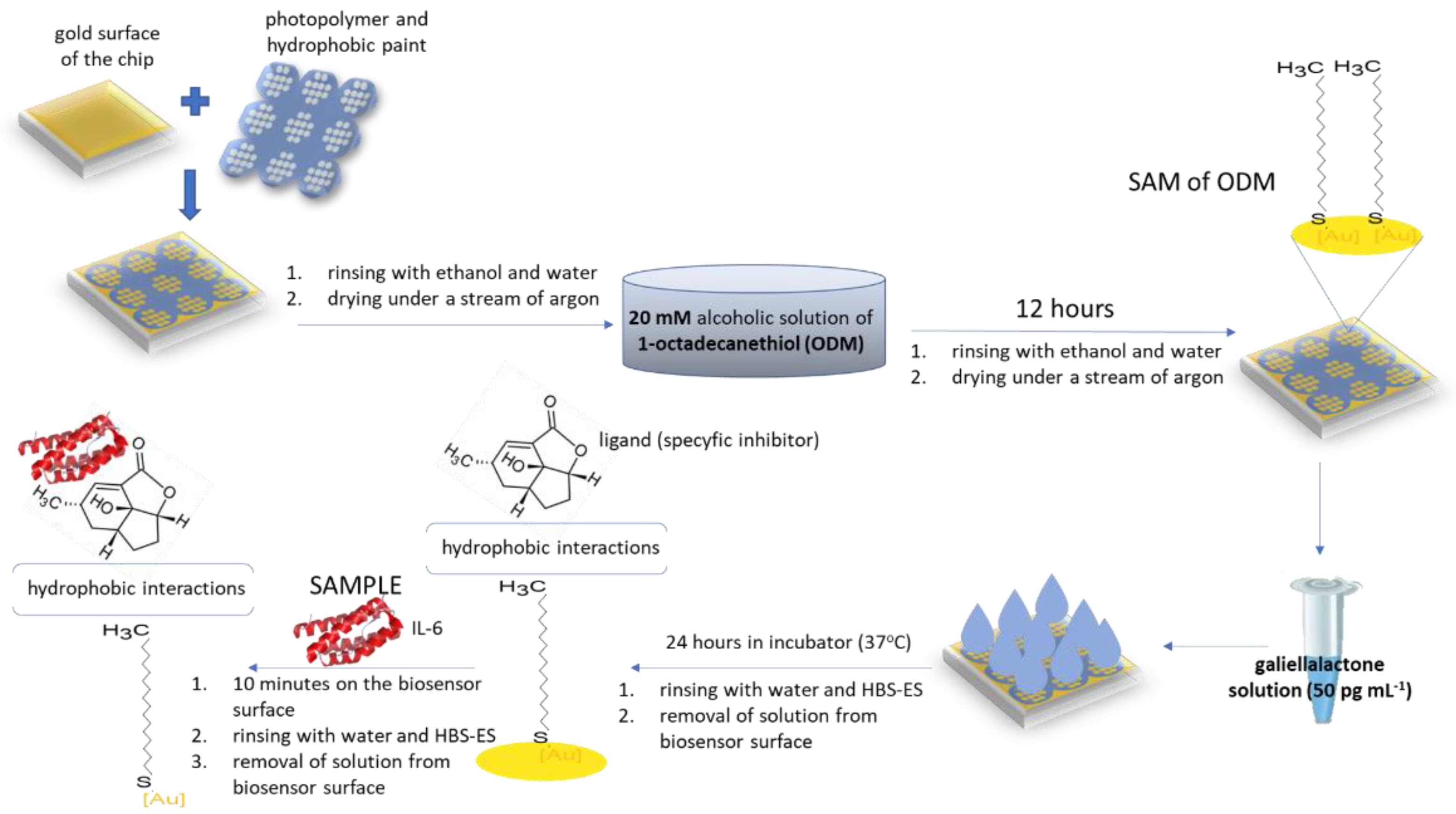
2.2. Biosensor Preparation
2.2.1. Chip Preparation
2.2.2. Antibody Immobilization
2.2.3. Immobilization of Galiellalactone
2.3. SPRi Measurements
2.4. Standard Method Used for Validation of the Developed Biosensor
3. Results
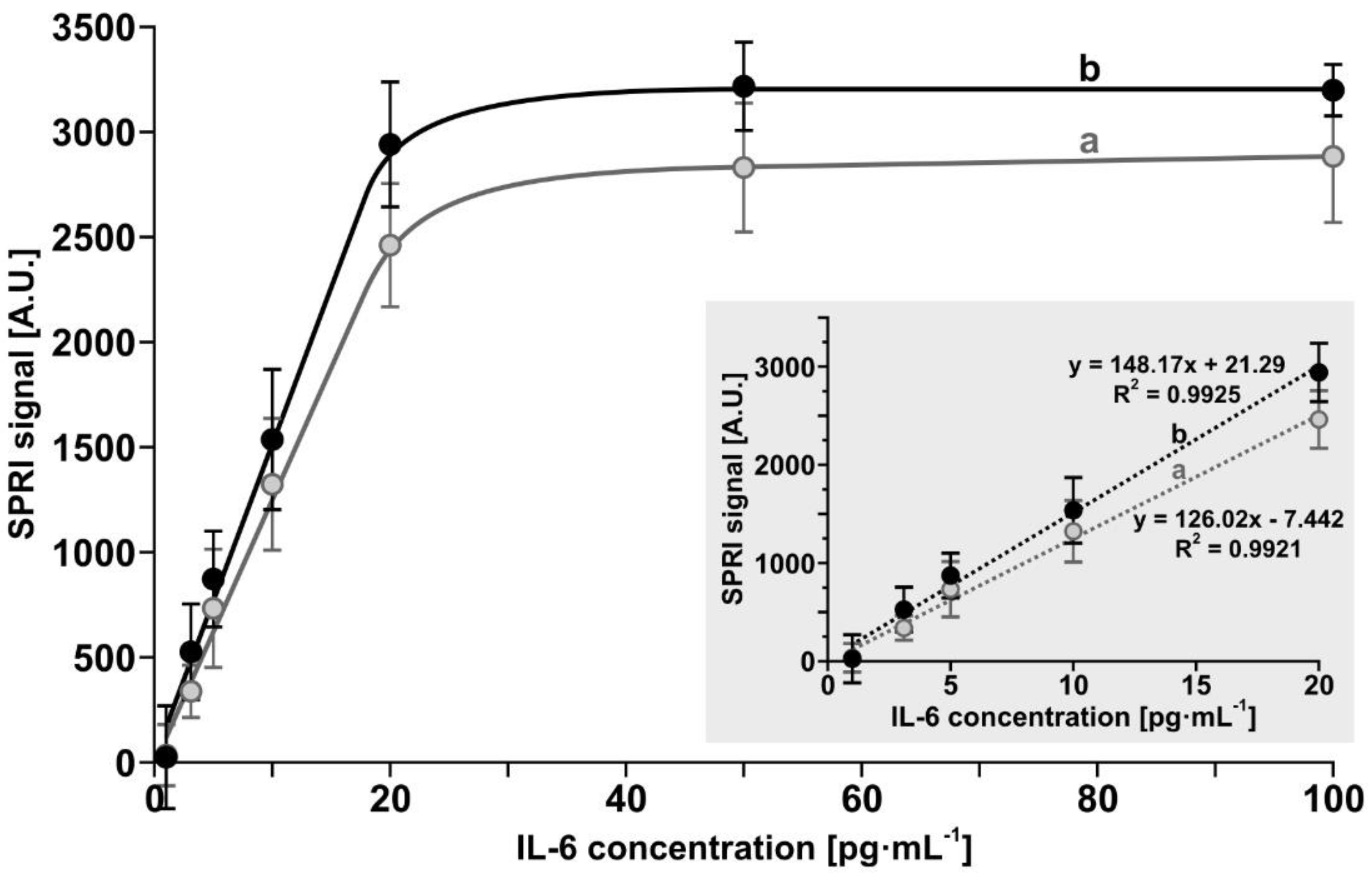
3.1. Calibration Curves for Both Biosensors
3.2. Selectivity of the Biosensor with Mouse Monoclonal Anti-IL-6 Antibody as Receptor
3.3. Selectivity of the Biosensor with the Inhibitor Galiellalactone as Receptor
3.4. Analytical Characteristics of the Developed Biosensors
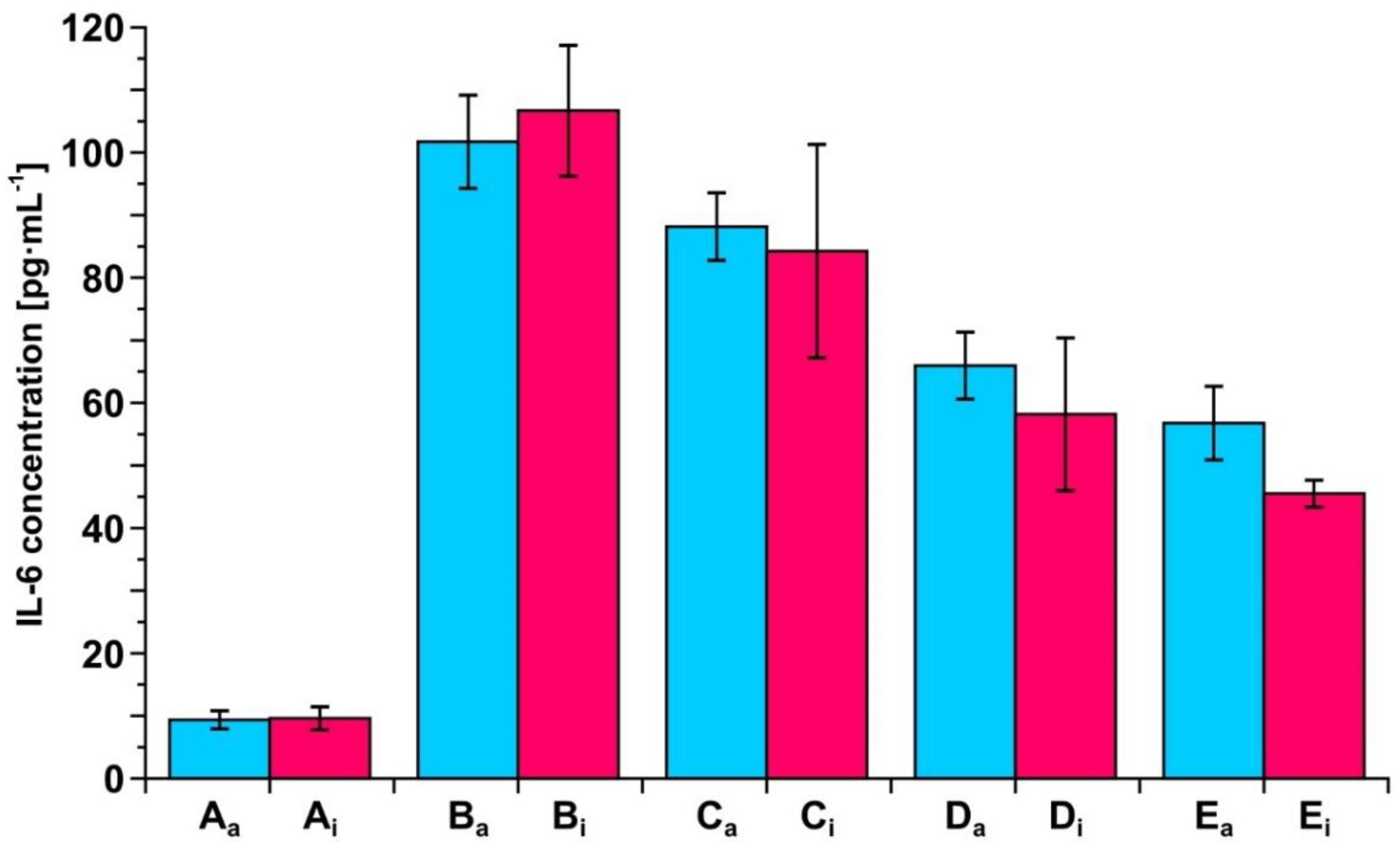
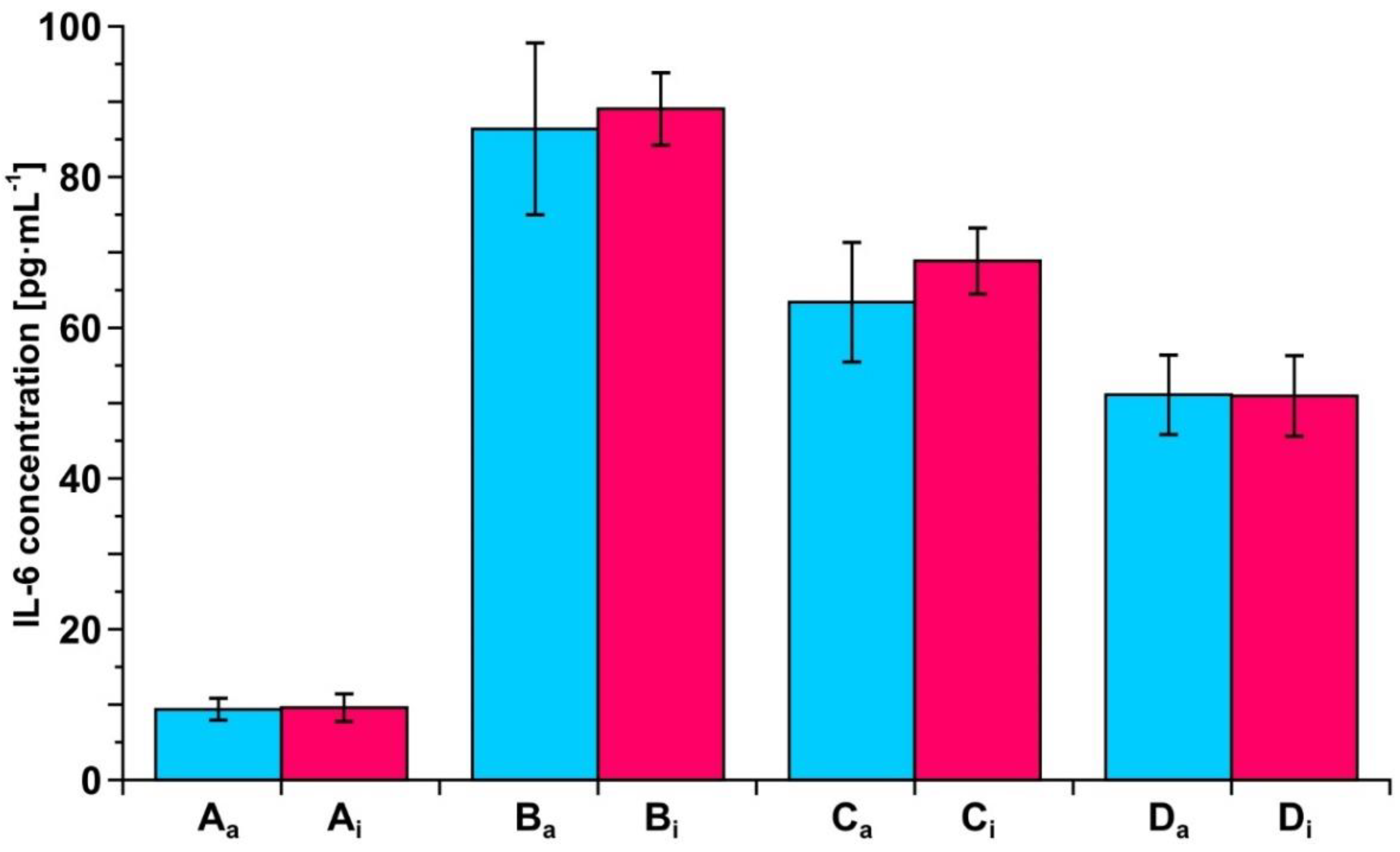
3.5. Determination of IL-6 Concentration in Biological Samples by the Two Biosensors
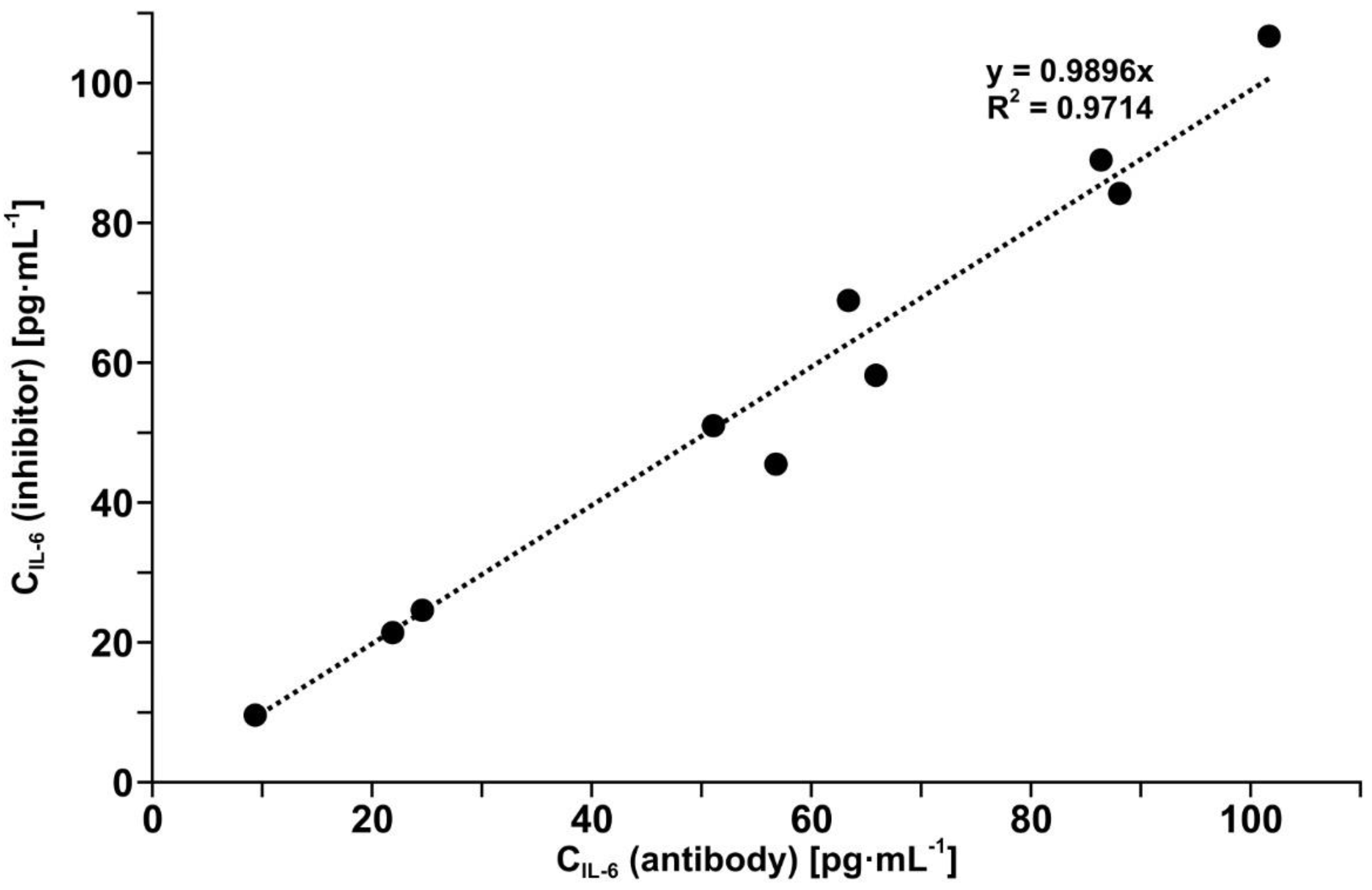
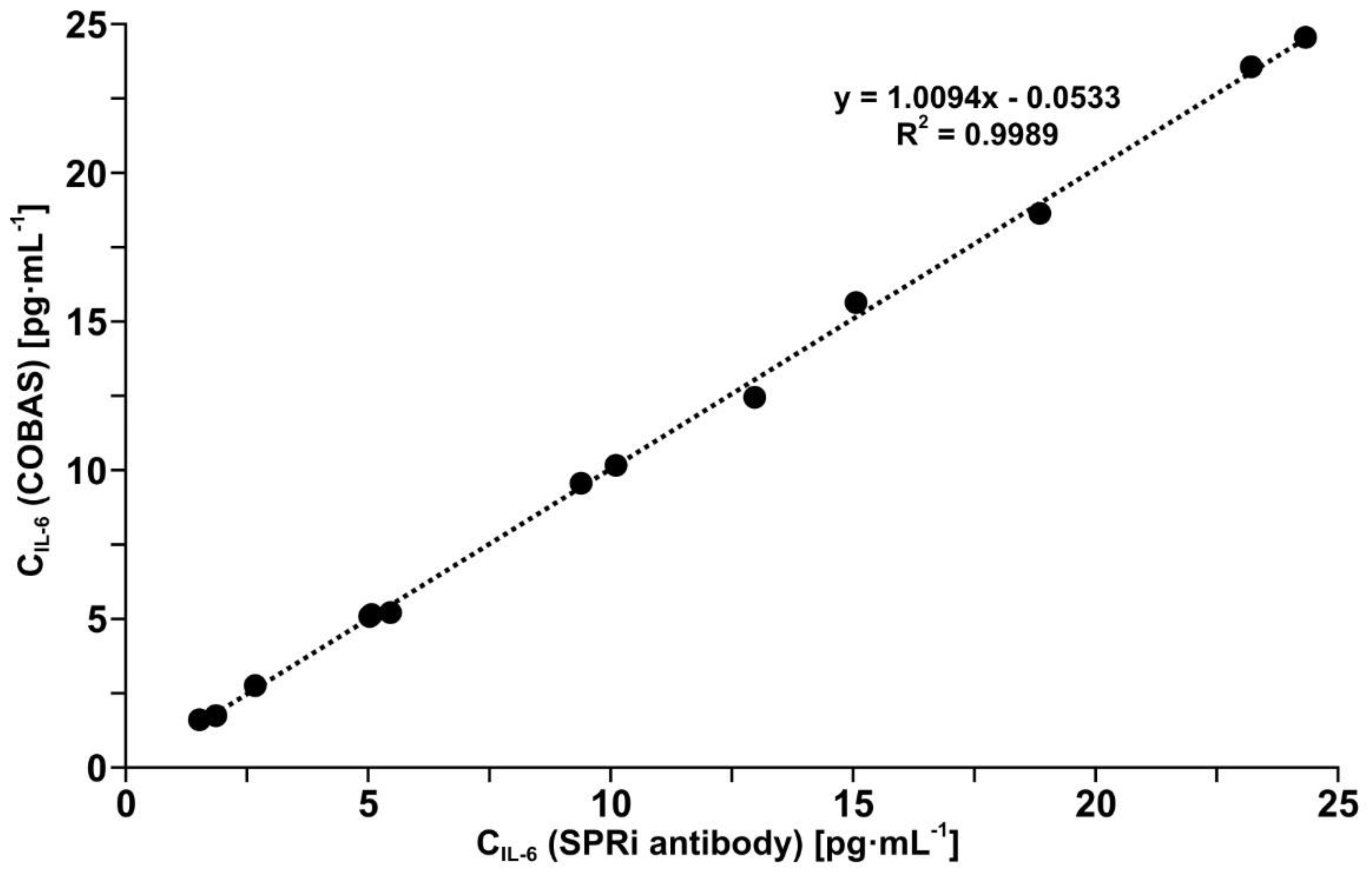
3.6. Validation of the Developed Biosensor with Mouse Monoclonal Anti-IL-6 Antibody as Receptor by Comparison with a Standard Method
3.7. Example of Clinical Application of the Developed Biosensors—Resection of Ovarian Tumor and Endometrial Cyst
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Liu, C.; Yang, Y.; Wu, Y. Recent Advances in Exosomal Protein Detection Via Liquid Biopsy Biosensors for Cancer Screening, Diagnosis, and Prognosis. AAPS J. 2018, 20, 41. [Google Scholar] [CrossRef] [PubMed]
- Springer, T.; Ermini, M.L.; Špačková, B.; Jablonků, J.; Homola, J. Enhancing sensitivity of surface plasmon resonance biosensors by functionalized gold nanoparticles: Size matters. Anal. Chem. 2014, 86, 10350–10356. [Google Scholar] [CrossRef] [PubMed]
- Springer, T.; Chadtová Song, X.; Ermini, M.L.; Lamačová, J.; Homola, J. Functional gold nanoparticles for optical affinity biosensing. Anal. Bioanal. Chem. 2017, 409, 4087–4097. [Google Scholar] [CrossRef] [PubMed]
- Ermini, M.L.; Chadtová Song, X.; Špringer, T.; Homola, J. Peptide functionalization of gold nanoparticles for the detection of carcinoembryonic antigen in blood plasma via SPR-based biosensor. Front. Chem. 2019, 7, 40. [Google Scholar] [CrossRef] [Green Version]
- Li, R.; Feng, F.; Chen, Z.Z.; Bai, Y.F.; Guo, F.F.; Wu, F.Y.; Zhou, G. Sensitive detection of carcinoembryonic antigen using surface plasmon resonance biosensor with gold nanoparticles signal amplification. Talanta 2015, 140, 143–149. [Google Scholar] [CrossRef]
- Pandey, P.S.; Raghuwanshi, S.K.; Kumar, S. Recent Advances in Two-Dimensional Materials-Based Kretschmann Configuration for SPR Sensors: A Review. IEEE Sens. J. 2022, 22, 15. [Google Scholar] [CrossRef]
- Chauhan, M.; Singh, V.K. Review on recent experimental SPR/LSPR based fiber optic analyte sensors. Opt. Fiber Technol. 2021, 64, 102580. [Google Scholar] [CrossRef]
- Szymanska, B.; Lukaszewski, Z.; Hermanowicz-Szamatowicz, K.; Gorodkiewicz, E. A biosensor for determination of the circulating biomarker CA125/MUC16 by Surface Plasmon Resonance Imaging. Talanta 2020, 206, 120–187. [Google Scholar] [CrossRef]
- Szymanska, B.; Lukaszewski, Z.; Zelazowska-Rutkowska, B.; Hermanowicz-Szamatowicz, K.; Gorodkiewicz, E. An SPRi Biosensor for Determination of the Ovarian Cancer Marker HE4 in Human Plasma. Sensors 2021, 21, 3567. [Google Scholar] [CrossRef]
- Szymanska, B.; Lukaszewski, Z.; Hermanowicz-Szamatowicz, K.; Gorodkiewicz, E. An immunosensor for the determination of carcinoembryonic antigen by Surface Plasmon Resonance Imaging. Anal. Biochem. 2020, 609, e113964. [Google Scholar] [CrossRef]
- Sankiewicz, A.; Romanowicz, L.; Laudanski, P.; Zelazowska-Rutkowska, B.; Puzan, B.; Cylwik, B.; Gorodkiewicz, E. SPR imaging biosensor for determination of laminin-5 as a potential cancer marker in biological material. Anal. Bioanal. Chem. 2016, 408, 5269–5276. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sankiewicz, A.; Lukaszewski, Z.; Trojanowska, K.; Gorodkiewicz, E. Determination of collagen type IV by Surface Plasmon Resonance Imaging using a specific biosensor. Anal. Biochem. 2016, 515, 40–46. [Google Scholar] [CrossRef] [PubMed]
- Sankiewicz, A.; Romanowicz, L.; Pyc, M.; Hermanowicz, A.; Gorodkiewicz, E. SPR imaging biosensor for the quantitation of fibronectin concentration in blood samples. J. Pharm. Biomed. Anal. 2018, 150, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Sankiewicz, A.; Hermanowicz, A.; Grycz, A.; Lukaszewski, Z.; Gorodkiewicz, E. An SPR imaging immunosensor for leptin determination in blood plasma. Anal. Methods 2021, 13, 642. [Google Scholar] [CrossRef]
- Tokarzewicz, A.; Romanowicz, L.; Sveklo, I.; Gorodkiewicz, E. The development of a matrix metalloproteinase-1 biosensor based on the surface plasmon resonance imaging technique. Anal. Methods 2016, 8, 6428. [Google Scholar] [CrossRef]
- Tokarzewicz, A.; Romanowicz, L.; Matuszczak, E.; Hermanowicz, A.; Gorodkiewicz, E. SPRI biosensors for quantitative determination of matrix metalloproteinase-2. Anal. Methods 2017, 9, 2407–2414. [Google Scholar] [CrossRef]
- Sankiewicz, A.; Guszcz, T.; Mena-Hortelano, R.; Zukowski, K.; Gorodkiewicz, E. Podoplanin serum and urine concentration in transitional bladder cancer. Cancer Biomark. 2016, 16, 343–350. [Google Scholar] [CrossRef]
- Guszcz, T.; Szymańska, B.; Kozlowski, R.; Lukaszewski, Z.; Laskowski, P.; Gorodkiewicz, E. Plasma aromatase as a sensitive and selective potential biomarker of bladder cancer and its role in tumorigenesis. Oncol. Lett. 2020, 19, 562–568. [Google Scholar] [CrossRef] [Green Version]
- Matuszczak, E.; Tylicka, M.; Debek, W.; Sankiewicz, A.; Gorodkiewicz, E.; Hermanowicz, A. Overexpression of ubiquitin carboxyl-terminal hydrolase L1 (UCHL1) in serum of children after thermal injury. Adv. Med. Sci. 2017, 62, 83–86. [Google Scholar] [CrossRef]
- Weremijewicz, A.; Matuszczak, E.; Sankiewicz, A.; Tylicka, M.; Komarowska, M.; Tokarzewicz, A.; Debek, W.; Gorodkiewicz, E.; Hermanowicz, A. Matrix metalloproteinase-2 and its correlation with basal membrane components laminin-5 and collagen type IV in burn patients measured with Surface Plasmon Resonance Imaging (SPRI) biosensors. Burns 2018, 44, 931–940. [Google Scholar] [CrossRef]
- Matuszczak, E.; Tylicka, M.; Hermanowicz, A.; Debek, W.; Sankiewicz, A.; Gorodkiewicz, E. Application of SPR Imaging Biosensor for the Measurement of 20S Proteasomes in Blood Plasma of Children with Thermal Injury. Ann. Clin. Lab. Sci. 2016, 46, 247–251. [Google Scholar]
- Brabek, J.; Jakubek, M.; Vellieux, F.; Novotný, J.; Kolá, M.; Lacina, J.; Szabo, P.; Strnadová, K.; Rösel, D.; Dvoránková, B.; et al. Interleukin-6: Molecule in the Intersection of Cancer, Ageing and COVID-19. Int. J. Mol. Sci. 2020, 21, 7937. [Google Scholar] [CrossRef] [PubMed]
- Amer, H.; Kartikasari, A.E.R.; Plebanski, M. Elevated Interleukin-6 Levels in the Circulation and Peritoneal Fluid of Patients with Ovarian Cancer as a Potential Diagnostic Biomarker: A Systematic Review and Meta-Analysis. J. Pers. Med. 2021, 11, 1335. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Li, J.; Liu, W.; Zhang, X.; Xue, L. The diagnostic value of interleukin 6 as a biomarker for gastric cancer: A meta-analysis and systematic review. Medicine 2021, 100, e27945. [Google Scholar] [CrossRef]
- Xu, J.; Ye, Y.; Zhang, H.; Szmitkowski, M.; Mäkinen, M.J.; Li, P. Diagnostic and prognostic value of serum interleukin-6 in colorectal cancer. Medicine 2016, 95, e2502. [Google Scholar] [CrossRef] [PubMed]
- Tong, Q.; Wang, X.L.; Li, S.B.; Yang, G.L.; Jin, S.; Gao, Z.Y.; Liu, X.B. Combined detection of IL-6 and IL-8 is beneficial to the diagnosis of early stage esophageal squamous cell cancer: A preliminary study based on the screening of serum markers using protein chips. Onco. Targets. Ther. 2018, 11, 5777–5787. [Google Scholar] [CrossRef] [Green Version]
- Trovato, M.; Sciacchitano, S.; Facciolà, A.; Valenti, A.; Visalli, G.; di Pietro, A. Interleukin-6 signalling as a valuable cornerstone for molecular medicine (review). Int. J. Molec. Med. 2021, 47, e107. [Google Scholar] [CrossRef]
- Kampan, N.C.; Madondo, M.T.; Reynolds, J.; Hallo, J.; McNally, O.M.; Jobling, T.W.; Stephens, A.N.; Quinn, M.A.; Plebanski, M. Pre-operative sera interleukin-6 in the diagnosis of high-grade serous ovarian cancer. Sci. Rep. 2020, 10, e221. [Google Scholar] [CrossRef]
- Montero, A.; Pascual, C.B.; Anaut, M.B.; López-Andrés, N.; Antona, G.; Martín-Calvo, N. Diagnostic performance of serum interleukin-6 in pediatric acute appendicitis: A systematic review. World J. Pediatr. 2022, 18, 91–99. [Google Scholar] [CrossRef]
- Sanchez-Zauco, N.; Torres, J.; Gómez, A.; Camorlinga-Ponce, M.; Muñoz-Pérez, L.; Herrera-Goepfert, R.; Medrano-Guzmán, R.; Giono-Cerezo, S.; Maldonado-Bernal, C. Circulating blood levels of IL-6, IFN-g, and IL-10 as potential diagnostic biomarkers in gastric cancer: A controlled study. BMC Cancer 2017, 17, e384. [Google Scholar] [CrossRef] [Green Version]
- Chou, T.H.; Chuang, C.-Y.; Wu, C.-M. Quantification of Interleukin-6 in cell culture medium using surface plasmon resonance biosensors. Cytokine 2010, 51, 107–111. [Google Scholar] [CrossRef] [PubMed]
- Gorodkiewicz, E.; Sankiewicz, A.; Laudanski, P. Surface plasmon resonance imaging biosensors for aromatase based on a potent inhibitor and a specific antibody: Sensor development and application for biological material. Cent. Eur. J. Chem. 2014, 12, 557–567. [Google Scholar] [CrossRef]
| Interferent | IL-6 Spike (pg mL−1) | Found IL-6 (pg mL−1) | Recovery (%) |
|---|---|---|---|
| CEA | 20.0 | 19.4 ± 2.68 | 97.0 |
| CA 125 | 20.0 | 19.7 ± 1.31 | 98.5 |
| Leptin | 20.0 | 20.6 ± 3.05 | 103 |
| MMP 2 | 20.0 | 20.5 ± 3.94 | 102 |
| Albumin | 20.0 | 19.0 ± 5.88 | 97.9 |
| Interferent | IL-6 Spike (pg mL−1) | Found IL-6 (pg mL−1) | Recovery (%) |
|---|---|---|---|
| CEA | 20.0 | 20.8 ± 1.34 | 104 |
| CA 125 | 20.0 | 19.7 ± 9.2 | 98.6 |
| Leptin | 20.0 | 19.9 ± 1.07 | 99.4 |
| MMP 2 | 20.0 | 20.5 ± 8.75 | 102 |
| albumin | 20.0 | 20.5 ± 1.64 | 102 |
| Biosensor with | IL-6 Spike (pg mL−1) | Found (pg mL−1) | SD (pg mL−1) | RSD (%) | Recovery (%) |
|---|---|---|---|---|---|
| antibody | 3.1 | 3.02 | 0.30 | 9.7 | 97.4 |
| 10 | 10.7 | 0.98 | 9.8 | 107 | |
| 20 | 19.6 | 1.6 | 8.0 | 97.9 | |
| inhibitor | 3.1 | 3.24 | 0.11 | 3.5 | 104 |
| 10 | 10.5 | 0.93 | 9.3 | 105 | |
| 20 | 20.2 | 1.8 | 8.9 | 101 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Szymanska, B.; Lukaszewski, Z.; Oldak, L.; Zelazowska-Rutkowska, B.; Hermanowicz-Szamatowicz, K.; Gorodkiewicz, E. Two Biosensors for the Determination of Interleukin-6 in Blood Plasma by Array SPRi. Biosensors 2022, 12, 412. https://doi.org/10.3390/bios12060412
Szymanska B, Lukaszewski Z, Oldak L, Zelazowska-Rutkowska B, Hermanowicz-Szamatowicz K, Gorodkiewicz E. Two Biosensors for the Determination of Interleukin-6 in Blood Plasma by Array SPRi. Biosensors. 2022; 12(6):412. https://doi.org/10.3390/bios12060412
Chicago/Turabian StyleSzymanska, Beata, Zenon Lukaszewski, Lukasz Oldak, Beata Zelazowska-Rutkowska, Kinga Hermanowicz-Szamatowicz, and Ewa Gorodkiewicz. 2022. "Two Biosensors for the Determination of Interleukin-6 in Blood Plasma by Array SPRi" Biosensors 12, no. 6: 412. https://doi.org/10.3390/bios12060412
APA StyleSzymanska, B., Lukaszewski, Z., Oldak, L., Zelazowska-Rutkowska, B., Hermanowicz-Szamatowicz, K., & Gorodkiewicz, E. (2022). Two Biosensors for the Determination of Interleukin-6 in Blood Plasma by Array SPRi. Biosensors, 12(6), 412. https://doi.org/10.3390/bios12060412






