Recent Development of Fluorescent Nanodiamonds for Optical Biosensing and Disease Diagnosis
Abstract
1. Introduction
2. Intracellular Heat Generation and Thermal Sensing
2.1. Optical Nanothermometry
2.2. Intracellular Thermometry
2.3. Quantifying the Intracellular Metabolic Heat
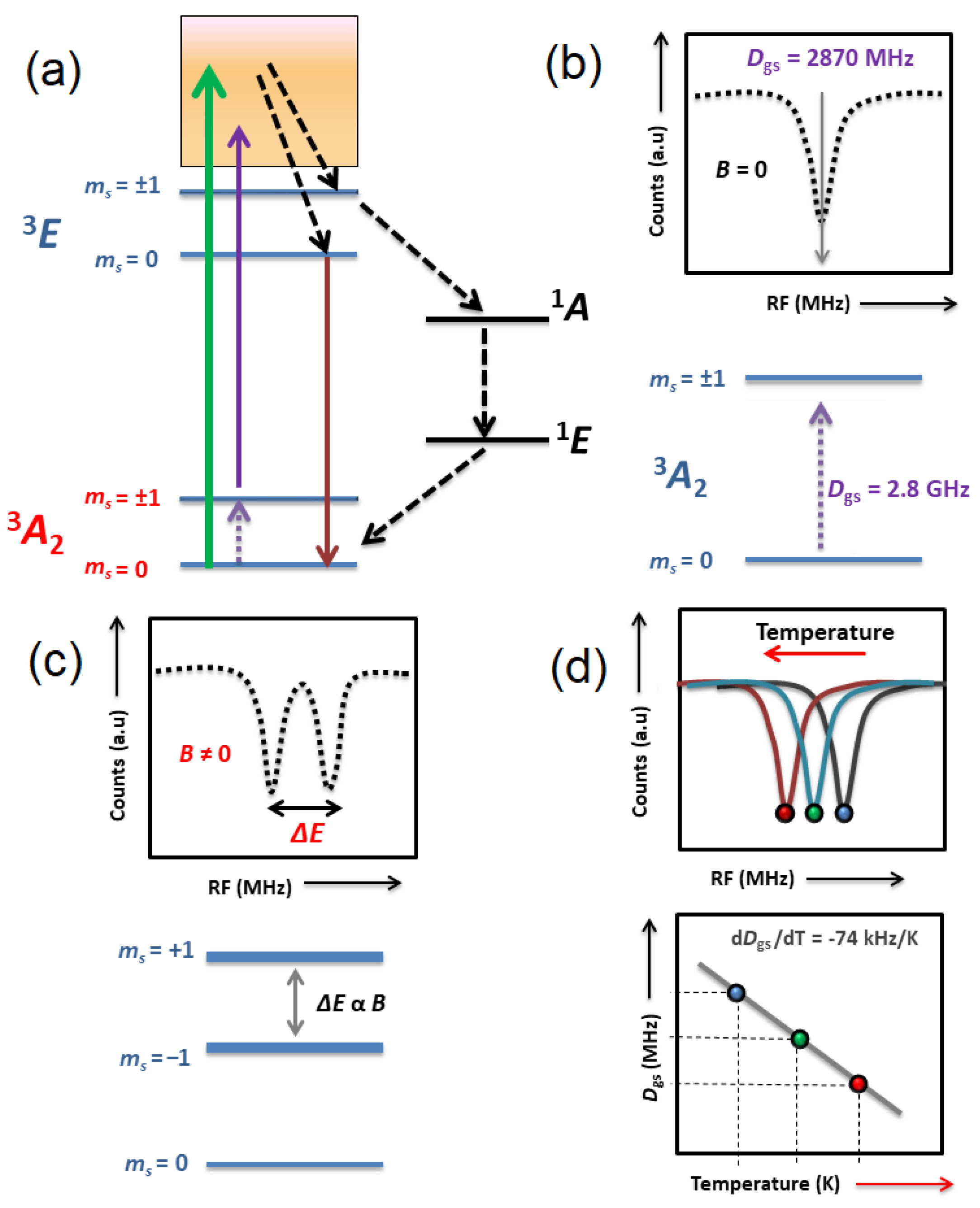
3. FNDs in Magnetic Sensing and Microscopy
3.1. Optical Magnetometry
3.2. Magnetometry in Biological Species
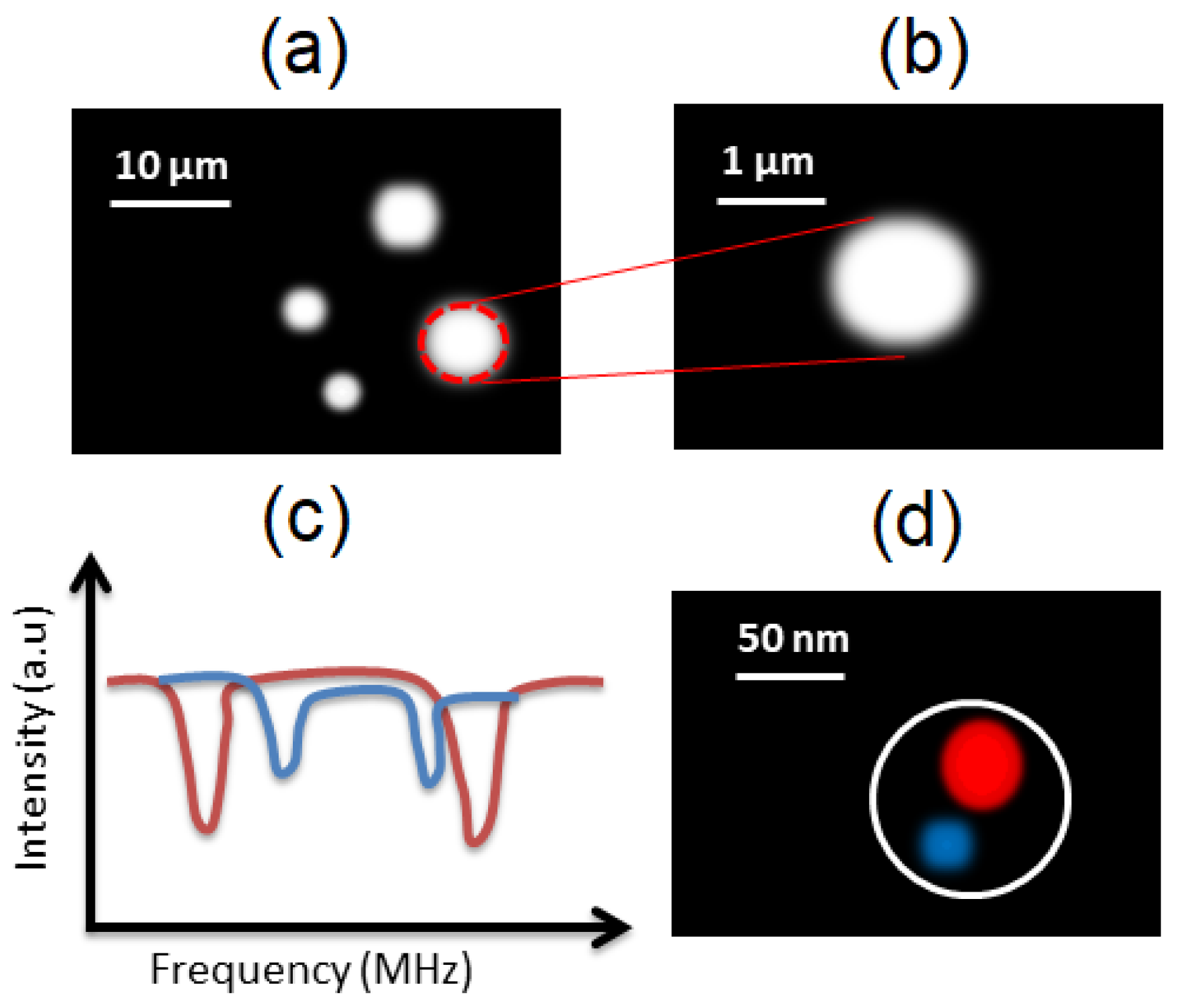
3.3. Intracellular Imaging and Tracking
3.4. Superresolution Microscopy
4. Intracellular Quantum-Sensing Applications of Surface-Enhanced FNDs
5. FNDs for the Detection of Infectious Viruses and Malicious Diseases
5.1. Biosensing of Infectious Viruses
5.2. FND as Nanomedicine for Cancer Therapy and Disease Diagnosis
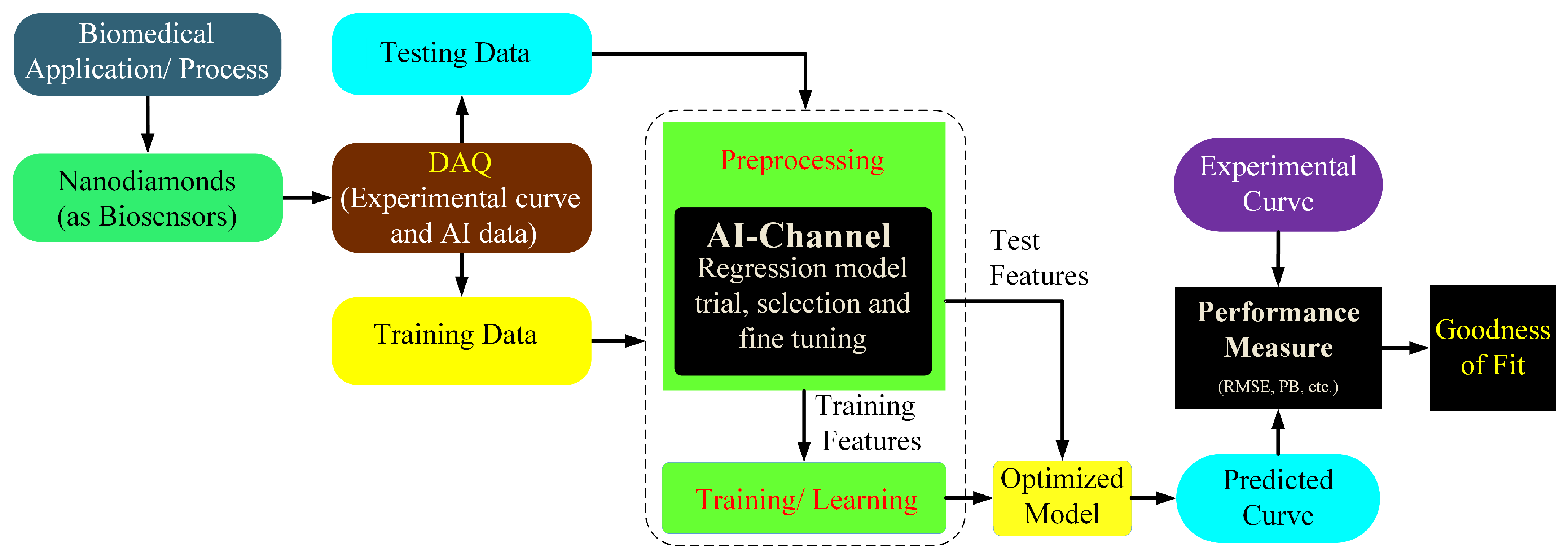
6. Application of FND Bioimaging with Artificial Intelligence
7. Future Challenges and Recommendations
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Schirhagl, R.; Chang, K.; Loretz, M.; Degen, C.L. Nitrogen-vacancy centers in diamond: Nanoscale sensors for physics and biology. Annu. Rev. Phys. Chem. 2014, 65, 83–105. [Google Scholar] [CrossRef] [PubMed]
- Jariwala, D.H.; Patel, D.; Wairkar, S. Surface functionalization of nanodiamonds for biomedical applications. Mater. Sci. Eng. C 2020, 113, 110996. [Google Scholar] [CrossRef]
- Ozawa, H.; Hatano, Y.; Iwasaki, T.; Harada, Y.; Hatano, M. Formation of perfectly aligned high-density nv centers in (111) cvd-grown diamonds for magnetic field imaging of magnetic particles. Jpn. J. Appl. Phys. 2019, 58, SIIB26. [Google Scholar] [CrossRef]
- Balasubramanian, G.; Neumann, P.; Twitchen, D.; Markham, M.; Kolesov, R.; Mizuochi, N.; Isoya, J.; Achard, J.; Beck, J.; Tissler, J. Ultralong spin coherence time in isotopically engineered diamond. Nat. Mater. 2009, 8, 383–387. [Google Scholar] [CrossRef] [PubMed]
- Stehlik, S.; Varga, M.; Ledinsky, M.; Jirasek, V.; Artemenko, A.; Kozak, H.; Ondic, L.; Skakalova, V.; Argentero, G.; Pennycook, T. Size and purity control of hpht nanodiamonds down to 1 nm. J. Phys. Chem. C 2015, 119, 27708–27720. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Miao, X.; Ma, H.; Guo, L.; Wang, Z.; Yang, Z.; Fang, C.; Jia, X. Synthesis and characterization of diamonds with different nitrogen concentrations under high pressure and high temperature conditions. CrystEngComm 2018, 20, 7164–7169. [Google Scholar] [CrossRef]
- Smeltzer, B.; Childress, L.; Gali, A. 13c hyperfine interactions in the nitrogen-vacancy centre in diamond. New J. Phys. 2011, 13, 025021. [Google Scholar] [CrossRef]
- Felton, S.; Edmonds, A.; Newton, M.; Martineau, P.; Fisher, D.; Twitchen, D.; Baker, J. Hyperfine interaction in the ground state of the negatively charged nitrogen vacancy center in diamond. Phys. Rev. B 2009, 79, 075203. [Google Scholar] [CrossRef]
- Antonov, D.; Häußermann, T.; Aird, A.; Roth, J.; Trebin, H.-R.; Müller, C.; McGuinness, L.; Jelezko, F.; Yamamoto, T.; Isoya, J. Statistical investigations on nitrogen-vacancy center creation. Appl. Phys. Lett. 2014, 104, 012105. [Google Scholar] [CrossRef]
- Orwa, J.; Santori, C.; Fu, K.; Gibson, B.; Simpson, D.; Aharonovich, I.; Stacey, A.; Cimmino, A.; Balog, P.; Markham, M. Engineering of nitrogen-vacancy color centers in high purity diamond by ion implantation and annealing. J. Appl. Phys. 2011, 109, 083530. [Google Scholar] [CrossRef]
- Pezzagna, S.; Naydenov, B.; Jelezko, F.; Wrachtrup, J.; Meijer, J. Creation efficiency of nitrogen-vacancy centres in diamond. New J. Phys. 2010, 12, 065017. [Google Scholar] [CrossRef]
- Neumann, P.; Jakobi, I.; Dolde, F.; Burk, C.; Reuter, R.; Waldherr, G.; Honert, J.; Wolf, T.; Brunner, A.; Shim, J.H.J.N.l. High-precision nanoscale temperature sensing using single defects in diamond. Nano Lett. 2013, 13, 2738–2742. [Google Scholar] [CrossRef] [PubMed]
- Hsiao, W.W.-W.; Hui, Y.Y.; Tsai, P.-C.; Chang, H.-C. Fluorescent nanodiamond: A versatile tool for long-term cell tracking, super-resolution imaging, and nanoscale temperature sensing. Acc. Chem. Res. 2016, 49, 400–407. [Google Scholar] [CrossRef] [PubMed]
- Kucsko, G.; Maurer, P.C.; Yao, N.Y.; Kubo, M.; Noh, H.J.; Lo, P.K.; Park, H.; Lukin, M.D. Nanometre-scale thermometry in a living cell. Nature 2013, 500, 54–58. [Google Scholar] [CrossRef]
- Barry, J.F.; Turner, M.J.; Schloss, J.M.; Glenn, D.R.; Song, Y.; Lukin, M.D.; Park, H.; Walsworth, R.L. Optical magnetic detection of single-neuron action potentials using quantum defects in diamond. Proc. Natl. Acad. Sci. USA 2016, 113, 14133–14138. [Google Scholar] [CrossRef]
- Le Sage, D.; Arai, K.; Glenn, D.R.; DeVience, S.J.; Pham, L.M.; Rahn-Lee, L.; Lukin, M.D.; Yacoby, A.; Komeili, A.; Walsworth, R.L. Optical magnetic imaging of living cells. Nature 2013, 496, 486–489. [Google Scholar] [CrossRef]
- Hall, L.; Beart, G.; Thomas, E.; Simpson, D.; McGuinness, L.; Cole, J.; Manton, J.; Scholten, R.; Jelezko, F.; Wrachtrup, J. High spatial and temporal resolution wide-field imaging of neuron activity using quantum nv-diamond. Sci. Rep. 2012, 2, 1–9. [Google Scholar] [CrossRef]
- Karadas, M.; Wojciechowski, A.M.; Huck, A.; Dalby, N.O.; Andersen, U.L.; Thielscher, A. Feasibility and resolution limits of opto-magnetic imaging of neural network activity in brain slices using color centers in diamond. Sci. Rep. 2018, 8, 1–14. [Google Scholar] [CrossRef]
- Dolde, F.; Fedder, H.; Doherty, M.W.; Nöbauer, T.; Rempp, F.; Balasubramanian, G.; Wolf, T.; Reinhard, F.; Hollenberg, L.C.; Jelezko, F. Electric-field sensing using single diamond spins. Nat. Phys. 2011, 7, 459–463. [Google Scholar] [CrossRef]
- Michl, J.; Steiner, J.; Denisenko, A.; Bülau, A.; Zimmermann, A.; Nakamura, K.; Sumiya, H.; Onoda, S.; Neumann, P.; Isoya, J. Robust and accurate electric field sensing with solid state spin ensembles. Nano Lett. 2019, 19, 4904–4910. [Google Scholar] [CrossRef]
- Ivády, V.; Simon, T.; Maze, J.R.; Abrikosov, I.; Gali, A. Pressure and temperature dependence of the zero-field splitting in the ground state of nv centers in diamond: A first-principles study. Phys. Rev. B 2014, 90, 235205. [Google Scholar] [CrossRef]
- Hsieh, S.; Bhattacharyya, P.; Zu, C.; Mittiga, T.; Smart, T.; Machado, F.; Kobrin, B.; Höhn, T.; Rui, N.; Kamrani, M. Imaging stress and magnetism at high pressures using a nanoscale quantum sensor. Science 2019, 366, 1349–1354. [Google Scholar] [CrossRef] [PubMed]
- Dolde, F.; Doherty, M.W.; Michl, J.; Jakobi, I.; Naydenov, B.; Pezzagna, S.; Meijer, J.; Neumann, P.; Jelezko, F.; Manson, N.B. Nanoscale detection of a single fundamental charge in ambient conditions using the nv− center in diamond. Phys. Rev. Lett. 2014, 112, 097603. [Google Scholar] [CrossRef]
- Degen, C.L.; Reinhard, F.; Cappellaro, P. Quantum sensing. Rev. Mod. Phys. 2017, 89, 035002. [Google Scholar] [CrossRef]
- Radtke, M.; Bernardi, E.; Slablab, A.; Nelz, R.; Neu, E. Nanoscale sensing based on nitrogen vacancy centers in single crystal diamond and nanodiamonds: Achievements and challenges. Nano Futur. 2019, 3, 042004. [Google Scholar] [CrossRef]
- Beams, R.; Smith, D.; Johnson, T.W.; Oh, S.-H.; Novotny, L.; Vamivakas, A.N. Nanoscale fluorescence lifetime imaging of an optical antenna with a single diamond nv center. Nano Lett. 2013, 13, 3807–3811. [Google Scholar] [CrossRef]
- Rondin, L.; Tetienne, J.-P.; Hingant, T.; Roch, J.-F.; Maletinsky, P.; Jacques, V. Magnetometry with nitrogen-vacancy defects in diamond. Rep. Prog. Phys. 2014, 77, 056503. [Google Scholar] [CrossRef]
- Acosta, V.M.; Bauch, E.; Ledbetter, M.P.; Waxman, A.; Bouchard, L.-S.; Budker, D. Temperature dependence of the nitrogen-vacancy magnetic resonance in diamond. Phys. Rev. Lett. 2010, 104, 070801. [Google Scholar] [CrossRef]
- Plakhotnik, T.; Doherty, M.W.; Cole, J.H.; Chapman, R.; Manson, N.B. All-optical thermometry and thermal properties of the optically detected spin resonances of the nv–center in nanodiamond. Nano Lett. 2014, 14, 4989–4996. [Google Scholar] [CrossRef]
- Sotoma, S.; Epperla, C.P.; Chang, H.C. Diamond nanothermometry. ChemNanoMat 2018, 4, 15–27. [Google Scholar] [CrossRef]
- Plakhotnik, T.; Aman, H.; Chang, H.-C. All-optical single-nanoparticle ratiometric thermometry with a noise floor of 0.3 k hz− 1/2. Nanotechnology 2015, 26, 245501. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.-D.; Dong, C.-H.; Sun, F.-W.; Zou, C.-L.; Cui, J.-M.; Han, Z.-F.; Guo, G.-C.J.A.P.L. Temperature dependent energy level shifts of nitrogen-vacancy centers in diamond. Appl. Phys. Lett. 2011, 99, 161903. [Google Scholar] [CrossRef]
- Toyli, D.; Christle, D.; Alkauskas, A.; Buckley, B.; Van de Walle, C.; Awschalom, D. Measurement and control of single nitrogen-vacancy center spins above 600 k. Phys. Rev. X 2012, 2, 031001. [Google Scholar] [CrossRef]
- Choi, S.; Agafonov, V.N.; Davydov, V.A.; Plakhotnik, T. Ultrasensitive all-optical thermometry using nanodiamonds with a high concentration of silicon-vacancy centers and multiparametric data analysis. ACS Photonics 2019, 6, 1387–1392. [Google Scholar] [CrossRef]
- Liu, C.-F.; Leong, W.-H.; Xia, K.; Feng, X.; Finkler, A.; Denisenko, A.; Wrachtrup, J.; Li, Q.; Liu, R.-B. Ultra-sensitive hybrid diamond nanothermometer. Natl. Sci. Rev. 2021, 8, nwaa194. [Google Scholar] [CrossRef]
- Wang, N.; Liu, G.-Q.; Leong, W.-H.; Zeng, H.; Feng, X.; Li, S.-H.; Dolde, F.; Fedder, H.; Wrachtrup, J.; Cui, X.-D. Magnetic criticality enhanced hybrid nanodiamond thermometer under ambient conditions. Phys. Rev. X 2018, 8, 011042. [Google Scholar] [CrossRef]
- Bommidi, D.K.; Pickel, A.D.J.A.P.L. Temperature-dependent excited state lifetimes of nitrogen vacancy centers in individual nanodiamonds. Appl. Phys. Lett. 2021, 119, 254103. [Google Scholar] [CrossRef]
- Tsai, P.C.; Epperla, C.P.; Huang, J.S.; Chen, O.Y.; Wu, C.C.; Chang, H.C. Measuring nanoscale thermostability of cell membranes with single gold–diamond nanohybrids. Angew. Chem. Int. Ed. 2017, 56, 3025–3030. [Google Scholar] [CrossRef]
- Fujiwara, M.; Sun, S.; Dohms, A.; Nishimura, Y.; Suto, K.; Takezawa, Y.; Oshimi, K.; Zhao, L.; Sadzak, N.; Umehara, Y. Real-time nanodiamond thermometry probing in vivo thermogenic responses. Sci. Adv. 2020, 6, eaba9636. [Google Scholar] [CrossRef]
- Sotoma, S.; Zhong, C.; Kah, J.C.Y.; Yamashita, H.; Plakhotnik, T.; Harada, Y.; Suzuki, M. In situ measurements of intracellular thermal conductivity using heater-thermometer hybrid diamond nanosensors. Sci. Adv. 2021, 7, eabd7888. [Google Scholar] [CrossRef]
- Suzuki, M.; Plakhotnik, T. Opportunities for hybrid diamond nanosensors targeting photothermal applications in biological systems. Appl. Phys. Lett. 2021, 119, 190502. [Google Scholar] [CrossRef]
- Fedotov, I.V.; Solotenkov, M.A.; Pochechuev, M.S.; Ivashkina, O.I.; Kilin, S.Y.; Anokhin, K.V.; Zheltikov, A.M. All-optical brain thermometry in freely moving animals. ACS Photonics 2020, 7, 3353–3360. [Google Scholar] [CrossRef]
- Chretien, D.; Bénit, P.; Ha, H.-H.; Keipert, S.; El-Khoury, R.; Chang, Y.-T.; Jastroch, M.; Jacobs, H.T.; Rustin, P.; Rak, M. Mitochondria are physiologically maintained at close to 50 c. PLoS Biol. 2018, 16, e2003992. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Xu, R.; Tian, W.; Jiang, X.; Cui, Z.; Wang, M.; Sun, H.; Fang, K.; Gu, N. Determining intracellular temperature at single-cell level by a novel thermocouple method. Cell Res. 2011, 21, 1517–1519. [Google Scholar] [CrossRef] [PubMed]
- Acosta, V.; Jarmola, A.; Bauch, E.; Budker, D. Optical properties of the nitrogen-vacancy singlet levels in diamond. Phys. Rev. B 2010, 82, 201202. [Google Scholar] [CrossRef]
- Acosta, V.; Bauch, E.; Jarmola, A.; Zipp, L.; Ledbetter, M.; Budker, D. Broadband magnetometry by infrared-absorption detection of nitrogen-vacancy ensembles in diamond. Appl. Phys. Lett. 2010, 97, 174104. [Google Scholar] [CrossRef]
- Yu, H.; Xie, Y.; Zhu, Y.; Rong, X.; Du, J. Enhanced sensitivity of the nitrogen-vacancy ensemble magnetometer via surface coating. Appl. Phys. Lett. 2020, 117, 204002. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, Z.; Feng, Y.; Guo, H.; Wen, H.; Tang, J.; Liu, J. High-sensitivity dc magnetic field detection with ensemble nv centers by pulsed quantum filtering technology. Opt. Express 2020, 28, 16191–16201. [Google Scholar] [CrossRef]
- Wolf, T.; Neumann, P.; Nakamura, K.; Sumiya, H.; Ohshima, T.; Isoya, J.; Wrachtrup, J. Subpicotesla diamond magnetometry. Phys. Rev. X 2015, 5, 041001. [Google Scholar] [CrossRef]
- Webb, J.L.; Troise, L.; Hansen, N.W.; Achard, J.; Brinza, O.; Staacke, R.; Kieschnick, M.; Meijer, J.; Perrier, J.-F.; Berg-Sørensen, K. Optimization of a diamond nitrogen vacancy centre magnetometer for sensing of biological signals. Front. Phys. 2020, 8, 430. [Google Scholar] [CrossRef]
- Hegyi, A.; Yablonovitch, E. Molecular imaging by optically detected electron spin resonance of nitrogen-vacancies in nanodiamonds. Nano Lett. 2013, 13, 1173–1178. [Google Scholar] [CrossRef] [PubMed]
- Price, J.C.; Levett, S.J.; Radu, V.; Simpson, D.A.; Barcons, A.M.; Adams, C.F.; Mather, M.L. Quantum sensing in a physiological-like cell niche using fluorescent nanodiamonds embedded in electrospun polymer nanofibers. Small 2019, 15, 1900455. [Google Scholar] [CrossRef] [PubMed]
- Barbiero, M.; Castelletto, S.; Zhang, Q.; Chen, Y.; Charnley, M.; Russell, S.; Gu, M. Nanoscale magnetic imaging enabled by nitrogen vacancy centres in nanodiamonds labelled by iron–oxide nanoparticles. Nanoscale 2020, 12, 8847–8857. [Google Scholar] [CrossRef] [PubMed]
- Gulka, M.; Salehi, H.; Varga, B.; Middendorp, E.; Pall, O.; Raabova, H.; Cloitre, T.; Cuisinier, F.J.; Cigler, P.; Nesladek, M. Simultaneous label-free live imaging of cell nucleus and luminescent nanodiamonds. Sci. Rep. 2020, 10, 1–9. [Google Scholar] [CrossRef]
- Lin, Y.C.; Perevedentseva, E.; Tsai, L.W.; Wu, K.T.; Cheng, C.L. Nanodiamond for intracellular imaging in the microorganisms in vivo. J. Biophotonics 2012, 5, 838–847. [Google Scholar] [CrossRef]
- Chen, E.H.; Gaathon, O.; Trusheim, M.E.; Englund, D. Wide-field multispectral super-resolution imaging using spin-dependent fluorescence in nanodiamonds. Nano Lett. 2013, 13, 2073–2077. [Google Scholar] [CrossRef]
- Hemelaar, S.R.; Nagl, A.; Bigot, F.; Rodríguez-García, M.M.; de Vries, M.P.; Chipaux, M.; Schirhagl, R. The interaction of fluorescent nanodiamond probes with cellular media. Microchim. Acta 2017, 184, 1001–1009. [Google Scholar] [CrossRef]
- Claveau, S.; Bertrand, J.-R.; Treussart, F. Fluorescent nanodiamond applications for cellular process sensing and cell tracking. Micromachines 2018, 9, 247. [Google Scholar] [CrossRef]
- Price, J.C.; Mesquita-Ribeiro, R.; Dajas-Bailador, F.; Mather, M.L. Widefield, spatiotemporal mapping of spontaneous activity of mouse cultured neuronal networks using quantum diamond sensors. Front. Phys. 2020, 8, 255. [Google Scholar] [CrossRef]
- Simpson, D.A.; Morrisroe, E.; McCoey, J.M.; Lombard, A.H.; Mendis, D.C.; Treussart, F.; Hall, L.T.; Petrou, S.; Hollenberg, L.C. Non-neurotoxic nanodiamond probes for intraneuronal temperature mapping. ACS Nano 2017, 11, 12077–12086. [Google Scholar] [CrossRef]
- Kaufmann, S.; Simpson, D.A.; Hall, L.T.; Perunicic, V.; Senn, P.; Steinert, S.; McGuinness, L.P.; Johnson, B.C.; Ohshima, T.; Caruso, F. Detection of atomic spin labels in a lipid bilayer using a single-spin nanodiamond probe. Proc. Natl. Acad. Sci. USA 2013, 110, 10894–10898. [Google Scholar] [CrossRef] [PubMed]
- Hall, L.T.; Hill, C.D.; Cole, J.H.; Städler, B.; Caruso, F.; Mulvaney, P.; Wrachtrup, J.; Hollenberg, L.C. Monitoring ion-channel function in real time through quantum decoherence. Proc. Natl. Acad. Sci. USA 2010, 107, 18777–18782. [Google Scholar] [CrossRef] [PubMed]
- Nie, L.; Nusantara, A.; Damle, V.; Sharmin, R.; Evans, E.; Hemelaar, S.; Van der Laan, K.; Li, R.; Perona Martinez, F.; Vedelaar, T. Quantum monitoring of cellular metabolic activities in single mitochondria. Sci. Adv. 2021, 7, eabf0573. [Google Scholar] [CrossRef] [PubMed]
- Holzgrafe, J.; Gu, Q.; Beitner, J.; Kara, D.M.; Knowles, H.S.; Atatüre, M. Nanoscale nmr spectroscopy using nanodiamond quantum sensors. Phys. Rev. Appl. 2020, 13, 044004. [Google Scholar] [CrossRef]
- Aslam, N.; Pfender, M.; Neumann, P.; Reuter, R.; Zappe, A.; Fávaro de Oliveira, F.; Denisenko, A.; Sumiya, H.; Onoda, S.; Isoya, J. Nanoscale nuclear magnetic resonance with chemical resolution. Science 2017, 357, 67–71. [Google Scholar] [CrossRef]
- Liang, L.; Zheng, P.; Jia, S.; Ray, K.; Chen, Y.; Barman, I. DNA self-assembled plasmonic nanodiamonds for biological sensing. bioRxiv 2021. [Google Scholar] [CrossRef]
- Wu, Y.; Alam, M.N.A.; Balasubramanian, P.; Ermakova, A.; Fischer, S.; Barth, H.; Wagner, M.; Raabe, M.; Jelezko, F.; Weil, T. Nanodiamond theranostic for light-controlled intracellular heating and nanoscale temperature sensing. Nano Lett. 2021, 21, 3780–3788. [Google Scholar] [CrossRef]
- Qureshi, S.A.; ul Rehman, A. Optical techniques, computed tomography and deep learning role in the diagnosis of COVID-19 pandemic towards increasing the survival rate of vulnerable populations. Photodiagnosis Photodyn. Ther. 2020, 31, 101880. [Google Scholar] [CrossRef]
- Zhang, T.; Pramanik, G.; Zhang, K.; Gulka, M.; Wang, L.; Jing, J.; Xu, F.; Li, Z.; Wei, Q.; Cigler, P. Toward quantitative bio-sensing with nitrogen–vacancy center in diamond. ACS Sens. 2021, 6, 2077–2107. [Google Scholar] [CrossRef]
- Shi, F.; Zhang, Q.; Wang, P.; Sun, H.; Wang, J.; Rong, X.; Chen, M.; Ju, C.; Reinhard, F.; Chen, H. Single-protein spin resonance spectroscopy under ambient conditions. Science 2015, 347, 1135–1138. [Google Scholar] [CrossRef]
- Sushkov, A.; Chisholm, N.; Lovchinsky, I.; Kubo, M.; Lo, P.; Bennett, S.; Hunger, D.; Akimov, A.; Walsworth, R.L.; Park, H. All-optical sensing of a single-molecule electron spin. Nano Lett. 2014, 14, 6443–6448. [Google Scholar] [CrossRef] [PubMed]
- Hui, Y.Y.; Chen, O.J.; Lin, H.-H.; Su, Y.-K.; Chen, K.Y.; Wang, C.-Y.; Hsiao, W.W.W.; Chang, H.-C. Magnetically modulated fluorescence of nitrogen-vacancy centers in nanodiamonds for ultrasensitive biomedical analysis. Anal. Chem. 2021, 93, 7140–7147. [Google Scholar] [CrossRef] [PubMed]
- Hsiao, W.W.-W.; Sharma, N.; Le, T.-N.; Cheng, Y.-Y.; Lee, C.-C.; Vo, D.-T.; Hui, Y.Y.; Chang, H.-C.; Chiang, W.-H. Fluorescent nanodiamond-based spin-enhanced lateral flow immunoassay for detection of SARS-CoV-2 nucleocapsid protein and spike protein from different variants. Anal. Chim. Acta 2022, 1230, 340389. [Google Scholar] [CrossRef] [PubMed]
- Shi, F.; Kong, F.; Zhao, P.; Zhang, X.; Chen, M.; Chen, S.; Zhang, Q.; Wang, M.; Ye, X.; Wang, Z. Single-DNA electron spin resonance spectroscopy in aqueous solutions. Nat. Methods 2018, 15, 697–699. [Google Scholar] [CrossRef]
- Li, C.; Soleyman, R.; Kohandel, M.; Cappellaro, P. SARS-CoV-2 quantum sensor based on nitrogen-vacancy centers in diamond. Nano Lett. 2021, 22, 43–49. [Google Scholar] [CrossRef]
- Gui, R.; Jin, H.; Bu, X.; Fu, Y.; Wang, Z.; Liu, Q. Recent advances in dual-emission ratiometric fluorescence probes for chemo/biosensing and bioimaging of biomarkers. Coord. Chem. Rev. 2019, 383, 82–103. [Google Scholar] [CrossRef]
- Roy, U.; Drozd, V.; Durygin, A.; Rodriguez, J.; Barber, P.; Atluri, V.; Liu, X.; Voss, T.G.; Saxena, S.; Nair, M. Characterization of nanodiamond-based anti-hiv drug delivery to the brain. Sci. Rep. 2018, 8, 1–12. [Google Scholar] [CrossRef]
- Miller, B.S.; Bezinge, L.; Gliddon, H.D.; Huang, D.; Dold, G.; Gray, E.R.; Heaney, J.; Dobson, P.J.; Nastouli, E.; Morton, J.J. Spin-enhanced nanodiamond biosensing for ultrasensitive diagnostics. Nature 2020, 587, 588–593. [Google Scholar] [CrossRef]
- Wu, K.; Vedelaar, T.A.; Damle, V.G.; Morita, A.; Mougnaud, J.; San Martin, C.R.; Zhang, Y.; van der Pol, D.P.; Ende-Metselaar, H.; Rodenhuis-Zybert, I. Applying nv center-based quantum sensing to study intracellular free radical response upon viral infections. Redox Biol. 2022, 52, 102279. [Google Scholar] [CrossRef]
- Wu, Y.; Cao, S.; Alam, M.N.A.; Raabe, M.; Michel-Souzy, S.; Wang, Z.; Wagner, M.; Ermakova, A.; Cornelissen, J.J.; Weil, T. Fluorescent nanodiamonds encapsulated by cowpea chlorotic mottle virus (ccmv) proteins for intracellular 3d-trajectory analysis. J. Mater. Chem. B 2021, 9, 5621–5627. [Google Scholar] [CrossRef]
- Feuerstein, G.Z.; Mansfield, M.A.; Lelkes, P.I.; Alesci, S.; Marcinkiewicz, C.; Butlin, N.; Sternberg, M. The use of near-infrared light-emitting fluorescent nanodiamond particles to detect ebola virus glycoprotein: Technology development and proof of principle. Int. J. Nanomed. 2020, 15, 7583. [Google Scholar] [CrossRef] [PubMed]
- Ivanova, V.; Ivanova, M.; Spitsyn, B.; Garina, K.; Trushakova, S.; Manykin, A.; Korzhenevsky, A.; Burseva, E. Interaction of Nanodiamonds Materials with Influenza Viruses; Journal of Physics: Conference Series; IOP Publishing: Bristol, UK, 2012; p. 012019. [Google Scholar]
- Leung, H.M.; Lau, C.H.; Ho, J.W.-T.; Chan, M.S.; Chang, T.J.H.; Law, L.H.; Wang, F.; Tam, D.Y.; Liu, L.S.; Chan, K.W.Y. Targeted brain tumor imaging by using discrete biopolymer-coated nanodiamonds across the blood–brain barrier. Nanoscale 2021, 13, 3184–3193. [Google Scholar] [CrossRef] [PubMed]
- Tang, G.; Zhang, M.; Liu, Q.; Tian, X.; Mai, R. Applications of nanodiamonds in the diagnosis and treatment of neurological diseases. J. Nanoparticle Res. 2022, 24, 1–23. [Google Scholar] [CrossRef]
- Panich, A.M.; Salti, M.; Prager, O.; Swissa, E.; Kulvelis, Y.V.; Yudina, E.B.; Aleksenskii, A.E.; Goren, S.D.; Vul’, A.Y.; Shames, A.I. Pvp-coated gd-grafted nanodiamonds as a novel and potentially safer contrast agent for in vivo mri. Magn. Reson. Med. 2021, 86, 935–942. [Google Scholar] [CrossRef]
- Zou, Y.; Komatsu, N. Quantitative investigation of the interaction between proteins and charged functional groups on the polyglycerol-grafted nanodiamond surface. Carbon 2020, 163, 395–401. [Google Scholar] [CrossRef]
- Mochalin, V.; Shenderova, O.; Ho, D.; Gogotsi, Y. The properties and applications of nanodiamonds. Nano-Enabled Med. Appl. 2020, 313–350. [Google Scholar]
- Xi, G.; Robinson, E.; Mania-Farnell, B.; Vanin, E.F.; Shim, K.-W.; Takao, T.; Allender, E.V.; Mayanil, C.S.; Soares, M.B.; Ho, D. Convection-enhanced delivery of nanodiamond drug delivery platforms for intracranial tumor treatment. Nanomed. Nanotechnol. Biol. Med. 2014, 10, 381–391. [Google Scholar] [CrossRef]
- Liu, K.-K.; Cheng, C.-L.; Chang, C.-C.; Chao, J.-I. Biocompatible and detectable carboxylated nanodiamond on human cell. Nanotechnology 2007, 18, 325102. [Google Scholar] [CrossRef]
- Locharoenrat, K. Efficacy of nanodiamond–doxorubicin complexes on human breast adenocarcinoma cell lines. Artif. Cells Nanomed. Biotechnol. 2019, 47, 4053–4058. [Google Scholar] [CrossRef]
- Lam, P.; Au-Yeung, K.M.; Cheng, P.W.; Wei, W.I.; Yuen, A.P.-W.; Trendell-Smith, N.; Li, J.H.; Li, R. Correlating mri and histologic tumor thickness in the assessment of oral tongue cancer. AJR Am. J. Roentgenol. 2004, 182, 803–808. [Google Scholar] [CrossRef] [PubMed]
- Feng, Q.; Yan, Y.Q.; Zhu, J.; Xu, J.R. T staging of rectal cancer: Accuracy of diffusion-weighted imaging compared with t2-weighted imaging on 3.0 tesla mri. J. Dig. Dis. 2014, 15, 188–194. [Google Scholar] [CrossRef] [PubMed]
- Curvo-Semedo, L.; Lambregts, D.M.; Maas, M.; Beets, G.L.; Caseiro-Alves, F.; Beets-Tan, R.G. Diffusion-weighted mri in rectal cancer: Apparent diffusion coefficient as a potential noninvasive marker of tumor aggressiveness. J. Magn. Reson. Imaging 2012, 35, 1365–1371. [Google Scholar] [CrossRef] [PubMed]
- Murphy, G.; Haider, M.; Ghai, S.; Sreeharsha, B. The expanding role of mri in prostate cancer. AJR Am. J. Roentgenol. 2013, 201, 1229–1238. [Google Scholar] [CrossRef]
- Heacock, L.; Melsaether, A.N.; Heller, S.L.; Gao, Y.; Pysarenko, K.M.; Babb, J.S.; Kim, S.G.; Moy, L. Evaluation of a known breast cancer using an abbreviated breast mri protocol: Correlation of imaging characteristics and pathology with lesion detection and conspicuity. Eur. J. Radiol. 2016, 85, 815–823. [Google Scholar] [CrossRef]
- Trimboli, R.M.; Verardi, N.; Cartia, F.; Carbonaro, L.A.; Sardanelli, F. Breast cancer detection using double reading of unenhanced mri including t1-weighted, t2-weighted stir, and diffusion-weighted imaging: A proof of concept study. AJR Am. J. Roentgenol. 2014, 203, 674–681. [Google Scholar] [CrossRef]
- Manus, L.M.; Mastarone, D.J.; Waters, E.A.; Zhang, X.-Q.; Schultz-Sikma, E.A.; MacRenaris, K.W.; Ho, D.; Meade, T.J. Gd (iii)-nanodiamond conjugates for mri contrast enhancement. Nano Lett. 2010, 10, 484–489. [Google Scholar] [CrossRef]
- Waddington, D.E.; Sarracanie, M.; Zhang, H.; Salameh, N.; Glenn, D.R.; Rej, E.; Gaebel, T.; Boele, T.; Walsworth, R.L.; Reilly, D.J. Nanodiamond-enhanced mri via in situ hyperpolarization. Nat. Commun. 2017, 8, 1–8. [Google Scholar] [CrossRef]
- Liu, Y.-Y.; Chang, B.-M.; Chang, H.-C. Nanodiamond-enabled biomedical imaging. Nanomedicine 2020, 15, 1599–1616. [Google Scholar] [CrossRef]
- Hou, W.; Toh, T.B.; Abdullah, L.N.; Yvonne, T.W.Z.; Lee, K.J.; Guenther, I.; Chow, E.K.-H. Nanodiamond–manganese dual mode mri contrast agents for enhanced liver tumor detection. Nanomed. Nanotechnol. Biol. Med. 2017, 13, 783–793. [Google Scholar] [CrossRef]
- Rammohan, N.; MacRenaris, K.W.; Moore, L.K.; Parigi, G.; Mastarone, D.J.; Manus, L.M.; Lilley, L.M.; Preslar, A.T.; Waters, E.A.; Filicko, A. Nanodiamond–gadolinium (iii) aggregates for tracking cancer growth in vivo at high field. Nano Lett. 2016, 16, 7551–7564. [Google Scholar] [CrossRef] [PubMed]
- McClung Pasqualino, H. Functional mri: Basic principles and emerging clinical applications for anesthesiology and the neurologic sciences. Anesthesiology 2021, 134, 136–137. [Google Scholar] [CrossRef] [PubMed]
- Gitto, S.; Cuocolo, R.; Albano, D.; Morelli, F.; Pescatori, L.C.; Messina, C.; Imbriaco, M.; Sconfienza, L.M. Ct and mri radiomics of bone and soft-tissue sarcomas: A systematic review of reproducibility and validation strategies. Insights Into Imaging 2021, 12, 1–14. [Google Scholar] [CrossRef]
- Buchbender, C.; Heusner, T.A.; Lauenstein, T.C.; Bockisch, A.; Antoch, G. Oncologic pet/mri, part 1: Tumors of the brain, head and neck, chest, abdomen, and pelvis. J. Nucl. Med. 2012, 53, 928–938. [Google Scholar] [CrossRef] [PubMed]
- Browning, L.M.; Mugridge, O.; Chatfield, M.D.; Dixon, A.K.; Aitken, S.W.; Joubert, I.; Prentice, A.M.; Jebb, S.A. Validity of a new abdominal bioelectrical impedance device to measure abdominal and visceral fat: Comparison with mri. Obesity 2010, 18, 2385–2391. [Google Scholar] [CrossRef]
- O’Connor, S.C.; Rooks, V.J.; Smith, A.B. Magnetic Resonance Imaging of the Fetal Central Nervous System, Head, Neck, and Chest; Seminars in Ultrasound, CT and MRI; Elsevier: Amsterdam, The Netherlands, 2012; pp. 86–101. [Google Scholar]
- Browning, L.M.; Mugridge, O.; Dixon, A.K.; Aitken, S.W.; Prentice, A.M.; Jebb, S.A. Measuring abdominal adipose tissue: Comparison of simpler methods with mri. Obes. Facts 2011, 4, 9–15. [Google Scholar] [CrossRef]
- Hong, S.; Grinolds, M.S.; Pham, L.M.; Le Sage, D.; Luan, L.; Walsworth, R.L.; Yacoby, A. Nanoscale magnetometry with nv centers in diamond. MRS Bull. 2013, 38, 155–161. [Google Scholar] [CrossRef]
- Chen, S.; Li, W.; Zheng, X.; Yu, P.; Wang, P.; Sun, Z.; Xu, Y.; Jiao, D.; Ye, X.; Cai, M. Immunomagnetic microscopy of tumor tissues using quantum sensors in diamond. Proc. Natl. Acad. Sci. USA 2022, 119, e2118876119. [Google Scholar] [CrossRef]
- Mistur, R.; Mosconi, L.; De Santi, S.; Guzman, M.; Li, Y.; Tsui, W.; de Leon, M.J. Current challenges for the early detection of alzheimer’s disease: Brain imaging and csf studies. J. Clin. Neurol. 2009, 5, 153–166. [Google Scholar] [CrossRef]
- Morales-Zavala, F.; Casanova-Morales, N.; Gonzalez, R.B.; Chandía-Cristi, A.; Estrada, L.D.; Alvizú, I.; Waselowski, V.; Guzman, F.; Guerrero, S.; Oyarzún-Olave, M. Functionalization of stable fluorescent nanodiamonds towards reliable detection of biomarkers for alzheimer’s disease. J. Nanobiotechnol. 2018, 16, 1–14. [Google Scholar] [CrossRef]
- Cui, F.; Yue, Y.; Zhang, Y.; Zhang, Z.; Zhou, H.S. Advancing biosensors with machine learning. ACS Sens. 2020, 5, 3346–3364. [Google Scholar] [CrossRef] [PubMed]
- Dolenko, T.A.; Burikov, S.A.; Vervald, A.M.; Vlasov, I.I.; Dolenko, S.A.; Laptinskiy, K.A.; Rosenholm, J.M.; Shenderova, O.A. Optical imaging of fluorescent carbon biomarkers using artificial neural networks. J. Biomed. Opt. 2014, 19, 117007. [Google Scholar] [CrossRef] [PubMed]
- Burikov, S.A.; Vervald, A.M.; Vlasov, I.I.; Dolenko, S.A.; Laptinskiy, K.; Dolenko, T.A. Use of neural network algorithms for elaboration of fluorescent biosensors on the base of nanoparticles. Opt. Mem. Neural Netw. 2013, 22, 156–165. [Google Scholar] [CrossRef]
- Loh, K.P.; Ho, D.; Chiu, G.N.C.; Leong, D.T.; Pastorin, G.; Chow, E.K.H. Clinical applications of carbon nanomaterials in diagnostics and therapy. Adv. Mater. 2018, 30, 1802368. [Google Scholar] [CrossRef]
- Blasiak, A.; Khong, J.; Kee, T. Curate. Ai: Optimizing personalized medicine with artificial intelligence. SLAS Technol. 2020, 25, 95–105. [Google Scholar] [CrossRef]
- Wang, H.; Lee, D.-K.; Chen, K.-Y.; Chen, J.-Y.; Zhang, K.; Silva, A.; Ho, C.-M.; Ho, D. Mechanism-independent optimization of combinatorial nanodiamond and unmodified drug delivery using a phenotypically driven platform technology. ACS Nano 2015, 9, 3332–3344. [Google Scholar] [CrossRef]
- Qureshi, S.A.; Raza, S.E.A.; Hussain, L.; Malibari, A.A.; Nour, M.K.; Rehman, A.u.; Al-Wesabi, F.N.; Hilal, A.M. Intelligent ultra-light deep learning model for multi-class brain tumor detection. Appl. Sci. 2022, 12, 3715. [Google Scholar] [CrossRef]
- Tao, H.; Wu, T.; Aldeghi, M.; Wu, T.C.; Aspuru-Guzik, A.; Kumacheva, E. Nanoparticle synthesis assisted by machine learning. Nat. Rev. Mater. 2021, 6, 701–716. [Google Scholar] [CrossRef]




| Sensing Type | Scheme | Sensitivity Achieved | References |
|---|---|---|---|
| Magnetic field | DC magnetometry Using ensembles of NV centers | 164 pTHz−1/2 | [47] |
| Magnetic field | CW-ODMR using ensembles of NV centers | 1 nTHz−1/2 | [48] |
| AC magnetic field at 20 KHz | Ensembles of NV centers | 0.9 pTHz−1/2 | [49] |
| AC magnetic field | Biological media (mouse brain) | 100 pTHz−1/2 | [50] |
| Property | Organic-Dye | Quantum-Dot | NV Center |
|---|---|---|---|
| Size (nm) | 1 | 10 | 5 |
| Emission band | UV-IR | UV-IR | 600–800 nm |
| Absorption band (nm) | 400–700 | 350–750 | 450–650 |
| Observed quantum yield | 1 | 0.8 | 0.8 |
| Photostability level | Low | High | Exceptionally High |
| Biocompatibility level | High | Low | High |
| Thermal sensitivity at room temperature (K−1) | 0.02–0.3 | 10−4–10−3 | 0.01 |
| Operating range (K) | 287–465 | 100–400 | 290–400 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qureshi, S.A.; Hsiao, W.W.-W.; Hussain, L.; Aman, H.; Le, T.-N.; Rafique, M. Recent Development of Fluorescent Nanodiamonds for Optical Biosensing and Disease Diagnosis. Biosensors 2022, 12, 1181. https://doi.org/10.3390/bios12121181
Qureshi SA, Hsiao WW-W, Hussain L, Aman H, Le T-N, Rafique M. Recent Development of Fluorescent Nanodiamonds for Optical Biosensing and Disease Diagnosis. Biosensors. 2022; 12(12):1181. https://doi.org/10.3390/bios12121181
Chicago/Turabian StyleQureshi, Shahzad Ahmad, Wesley Wei-Wen Hsiao, Lal Hussain, Haroon Aman, Trong-Nghia Le, and Muhammad Rafique. 2022. "Recent Development of Fluorescent Nanodiamonds for Optical Biosensing and Disease Diagnosis" Biosensors 12, no. 12: 1181. https://doi.org/10.3390/bios12121181
APA StyleQureshi, S. A., Hsiao, W. W.-W., Hussain, L., Aman, H., Le, T.-N., & Rafique, M. (2022). Recent Development of Fluorescent Nanodiamonds for Optical Biosensing and Disease Diagnosis. Biosensors, 12(12), 1181. https://doi.org/10.3390/bios12121181







