Nanomaterials-Based Ion-Imprinted Electrochemical Sensors for Heavy Metal Ions Detection: A Review
Abstract
:1. Introduction
2. Preparation of IIPs
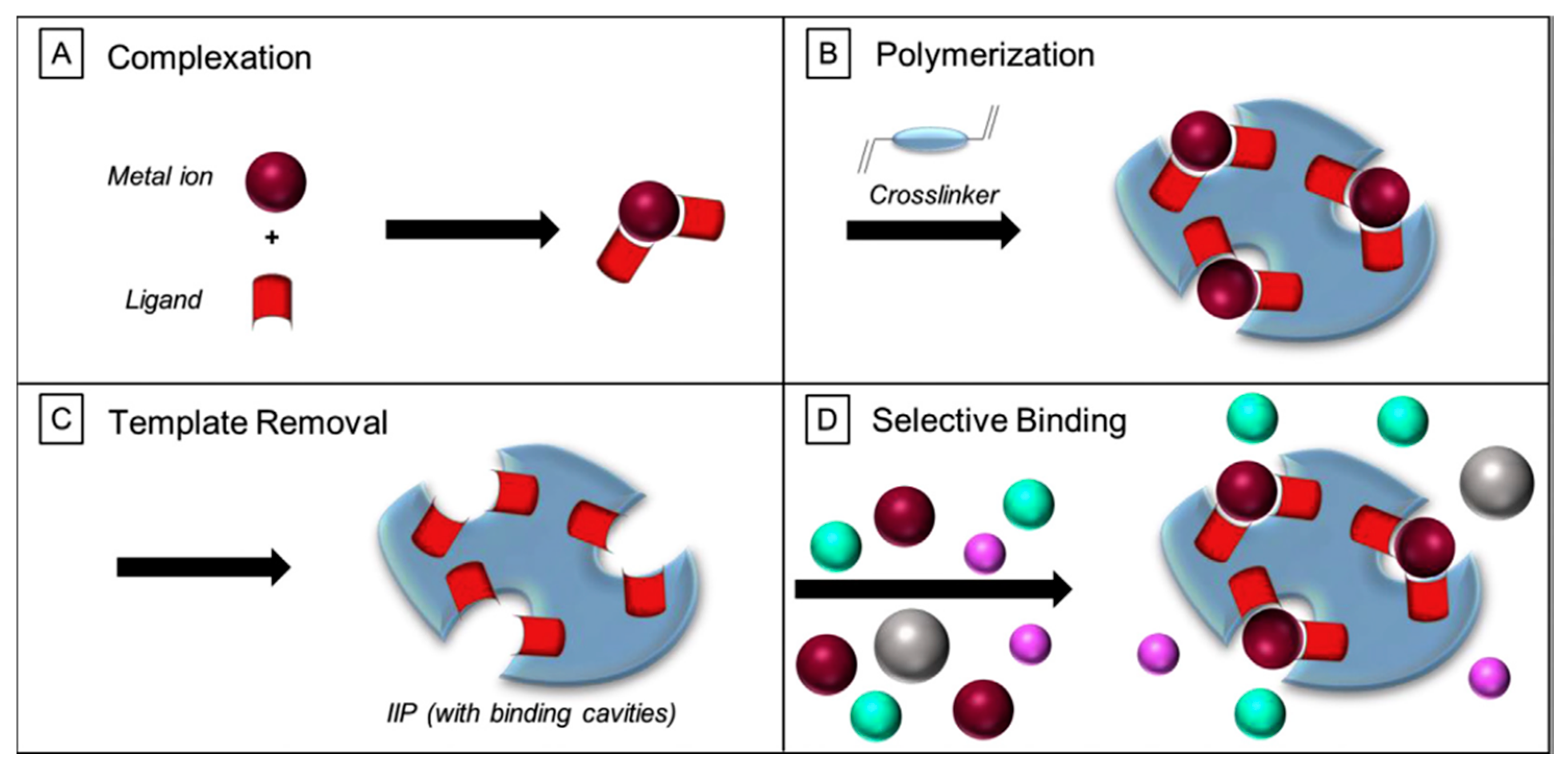
2.1. Preparation Principle of IIPs
2.2. Preparation Methods of IIPs
2.2.1. Bulk Polymerization
2.2.2. Precipitation Polymerization
2.2.3. Suspension Polymerization
2.2.4. Emulsion Polymerization
2.2.5. Surface Imprinting Method
2.3. Preparation of HMIIECSs
2.3.1. Preparation Principle of HMIIECSs
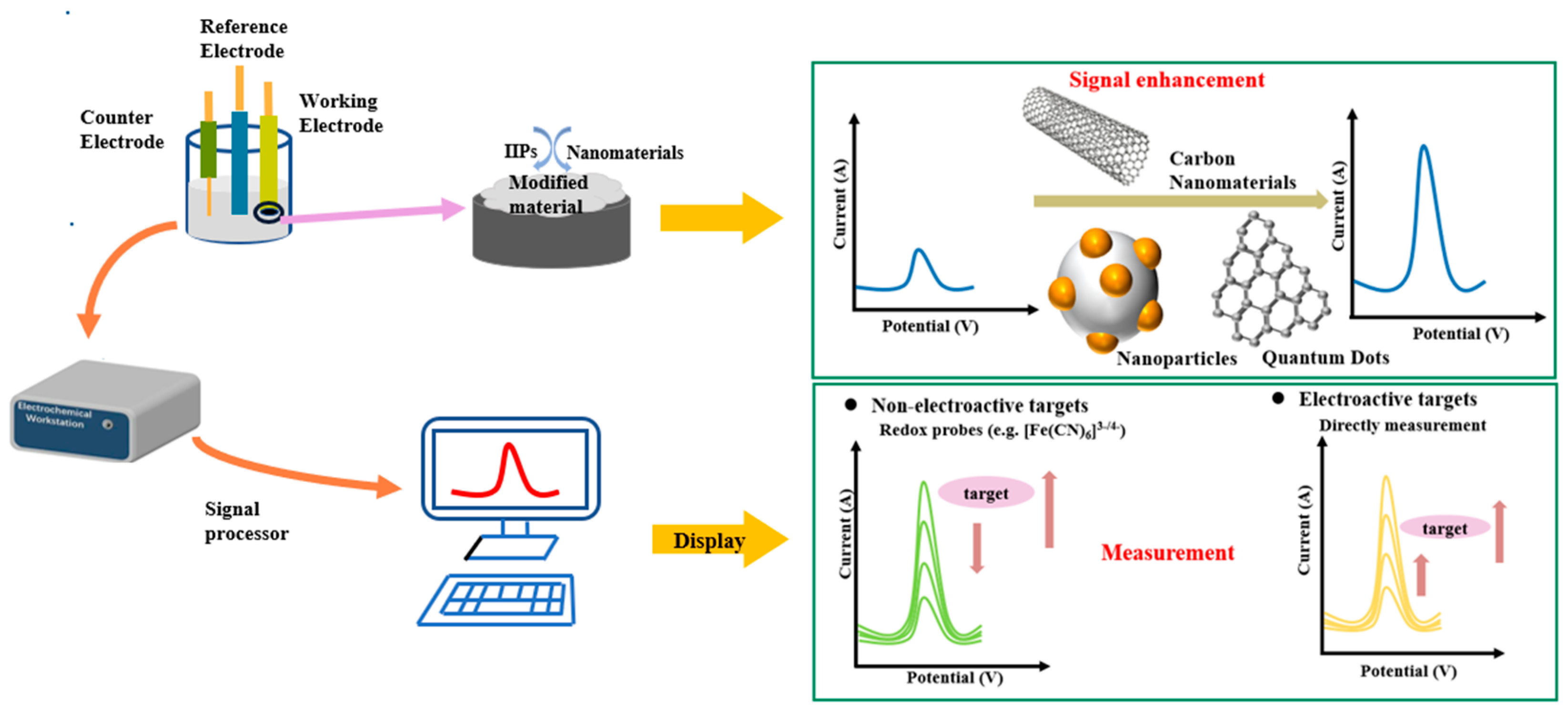
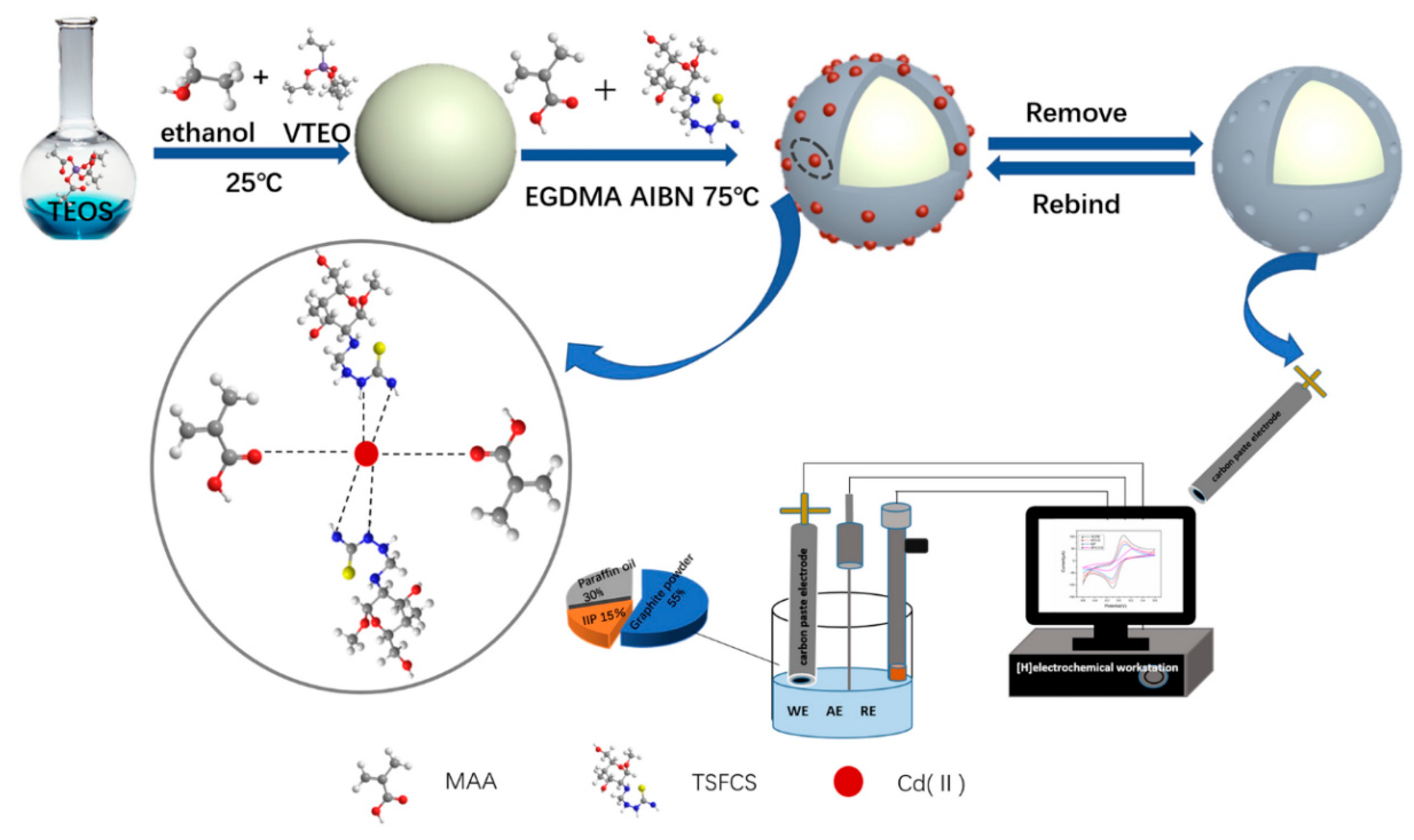
2.3.2. Preparation Methods of HMIIECS
The Direct Method
The Indirect Method
3. Application of Nanomaterials in HMIECSs
3.1. Carbon-Based Nanomaterials
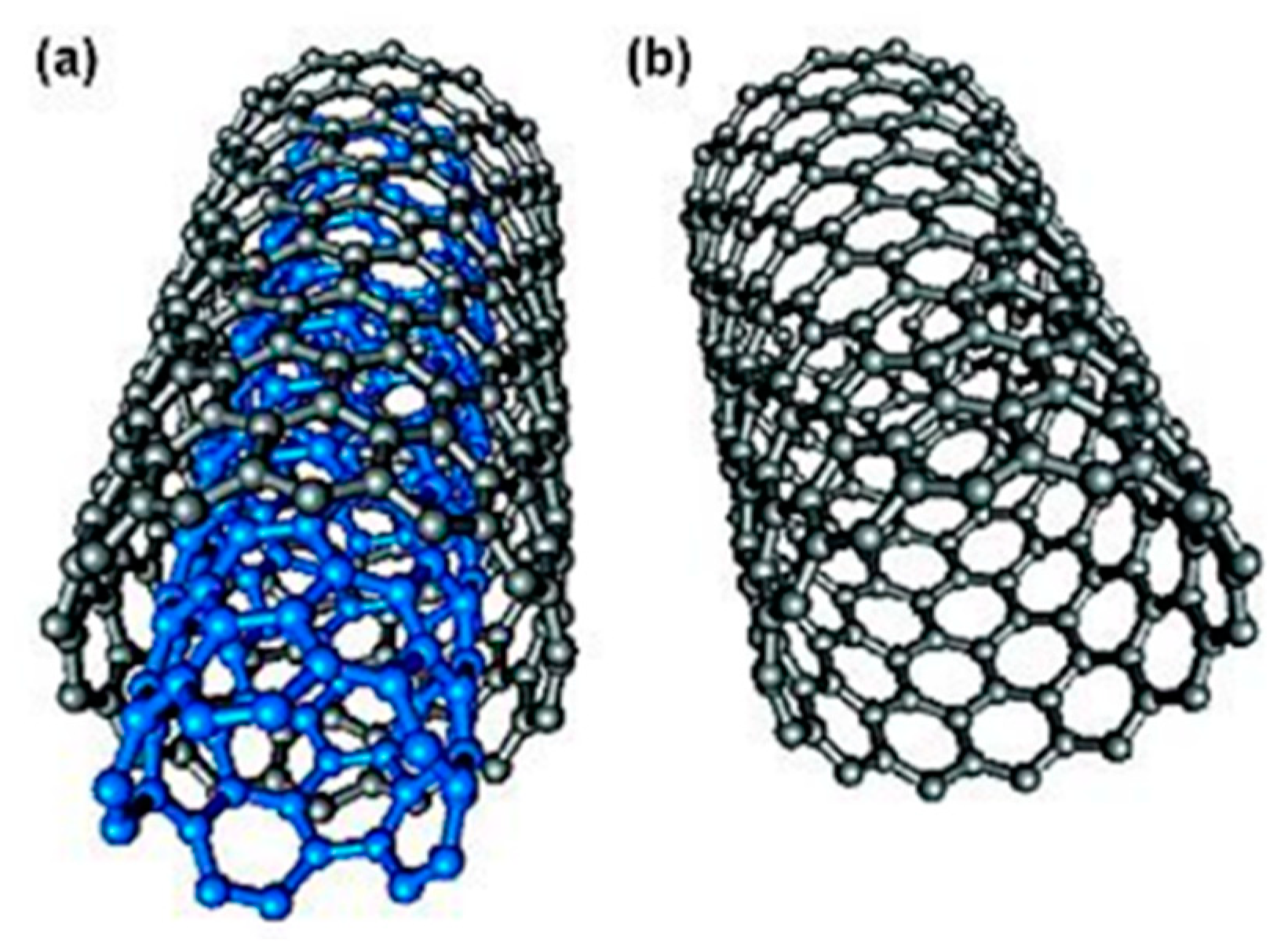
3.1.1. Carbon Nanotube Materials
3.1.2. Graphene Materials
3.1.3. Graphitic Carbonitride Nanomaterials
3.2. Metal Nanomaterials
Gold Nanomaterials
3.3. Metal Oxide Nanomaterials
3.4. Magnetic Ferric Oxide Nanomaterials
3.5. Silica Nanomaterials
4. Conclusions and Future Outlook
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| HMIs | Heavy metal ions | MAA | Methacrylic acid |
| IIECSs | Ion-imprinted electrochemical sensors | OPD | O-phenylenediamine |
| IIPs | Ion-imprinted polymers | AA | Acrylic acid |
| WHO | World Health Organization | AM | Acrylamide |
| IQ | Intelligence quotient | 4-VP | 4-Vinylpyridine |
| SERS | Surface-enhanced Raman spectroscopy | Py | Pyrrole |
| AAS | Atomic absorption spectrometry | ECSs | Electrochemical sensors |
| CE | Capillary electrophoresis | CS | Chitosan |
| ICP-AES | Inductively coupled plasma-atomic emission spectroscopy | NNMBA | N,N′-methylene double acrylamide |
| IC-UV-vis | Ion chromatography-ultraviolet vis spectrometry | TPPZ | 2,3,5,6-Tetra(2-pyridyl) pyrazine |
| ICP-MS | Inductively coupled plasma mass spectrometry | MMWCNTs | Magnetic multi-walled carbon nanotubes |
| MP | Microprobe | NPs | Nanoparticles |
| XFS | X-ray fluorescence spectroscopy | WE | Working electrodes |
| MIPs | Molecularly imprinted polymers | RE | Reference electrodes |
| MIECSs | Molecularly imprinted electrochemical sensors | CE | Counter electrodes |
| MIT | Molecular imprinting technique | ISEs | Ion selective electrodes |
| HMIIECSs | Heavy metal ions imprinted electrochemical sensors | LOD | Low limit of detection |
| CPE | Carbon paste electrode | IIMs | Ion-imprinted membranes |
| APA | Allyl phenoxyacetate | Th | Thiophene |
| EGDMA | Ethylene glycol dimethacrylate | SCE | Saturated calomel electrode |
| AIBN | Azobisisobutyronitrile | CV | Cyclic voltammetry |
| CNF | Carbon nanofibers-grafted | DPV | Differential pulse voltammetry |
| SEM | Scanning electron microscopy | PPD | P-phenylenediamine |
| AAM | Acryl amide | THPP | 5,10,15,20-Tetrakis(3-hydroxyphenyl)-porphyrin |
| BAAP | Bis-(2-acryloylamino-ethyl)-phosphinic acid | IL | Ionic liquid |
| SPCE | Screen-printed carbon electrode | GR | Graphene |
| PGE | Pencil graphite electrode | rGO | Reduced graphene oxide |
| PC | Poly catechol | GP | Graphite powder |
| SPE | Screen printed electrode | EAAP | Ethyl 6-(allyloxy)-2-amino-4-phenyl-4H-benzo[f]chromene-3-carboxylate |
| DPC | 1,5-Dipenylcarbazone | ASV | Anodic stripping voltammetry |
| CNTs | Carbon nanotubes | DDBHCT | 2,2’-(9E,10E)-1,4-Dihydroxy-anthrace-9,10-dimethylene)bis(hydrazi-1-carbonthiona-mide) |
| SWCNTs | Single-walled carbon nanotubes | APS | Ammonium persulfate |
| GCE | Glassy carbon electrode | TEM | Transmission electron microscope |
| DPASV | Differential pulse anodic stripping voltammetry | PPy | Polypyrrole |
| PAN | 1-(2-Pyridazo)-2-naphthol | ERGO | Electrochemical reduction of graphene oxide |
| LOQ | Limit of quantification | PoPD | Poly (o-phenylenediamine) |
| SWASV | Square wave anodic stripping voltammetry | EDTA | Ethylene diamine tetraacetic acid |
| RSD | Relative standard deviation | GQDs | Graphene quantum dots |
| EIS | Electrochemical impedance spectroscopy | GQDTU | Thiourea derivative-functionalized GQDs |
| LEDs | Light-emitting devices | g-C3N4 | Graphitic carbonitride |
| ITA | Itaconic acid | NPG | Nanoporous gold |
| AuNPs | Gold nanomaterials | GE | Gold electrode |
| MMIECS | Magnetic molecularly imprinted electrochemical sensors | MNMs | Magnetic nanomaterials |
| GF-AAS | Graphite furnace atomic absorption spectrometry | 2-CBT | Benzothiazole-2-carbaldehyde |
| APTES | 3-Aminopropyltriethoxysilane | MPS | Methacryloxypropyltrimethoxysilane |
| VTES | Vinyltriethoxysilane | SCMNPs | SiO2-coated magnetite nanomaterials |
| PVC | Polyvinylchloride |
References
- Liu, X.; Yao, Y.; Ying, Y.; Ping, J. Recent advances in nanomaterial-enabled screen-printed electrochemical sensors for heavy metal detection. TrAC Trends Anal. Chem. 2019, 115, 187–202. [Google Scholar] [CrossRef]
- Nourbakhsh, A.; Rahimnejad, M.; Asghary, M.; Younesi, H. Simultaneous electro-determination of trace copper, lead, and cadmium in tap water by using silver nanoparticles and graphene nanoplates as nanocomposite modified graphite electrode. Microchem. J. 2022, 175, 107137. [Google Scholar] [CrossRef]
- Guerrini, L.; Alvarez-Puebla, R.A. Surface-Enhanced Raman Scattering Sensing of Transition Metal Ions in Waters. ACS Omega 2021, 6, 1054–1063. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Numan, A.; Zhan, Y.; Singh, V.; Van Hung, T.; Nam, N.D. A novel highly efficient and ultrasensitive electrochemical detection of toxic mercury (II) ions in canned tuna fish and tap water based on a copper metal-organic framework. J. Hazard. Mater. 2020, 399, 123042. [Google Scholar] [CrossRef] [PubMed]
- Pathak, P.; Hwang, J.H.; Li, R.H.T.; Rodriguez, K.L.; Rex, M.M.; Lee, W.H.; Cho, H.J. Flexible copper-biopolymer nanocomposite sensors for trace level lead detection in water. Sens. Actuators B Chem. 2021, 344, 130263. [Google Scholar] [CrossRef]
- Waheed, A.; Mansha, M.; Ullah, N. Nanomaterials-based electrochemical detection of heavy metals in water: Current status, challenges and future direction. TrAC Trends Anal. Chem. 2018, 105, 37–51. [Google Scholar] [CrossRef]
- Guo, Z.M.; Chen, P.; Yosri, N.; Chen, Q.S.; Elseedi, H.R.; Zou, X.B.; Yang, H.S. Detection of heavy metals in food and agricultural products by surface-enhanced raman spectroscopy. Food Rev. Int. 2021, 1–22. [Google Scholar] [CrossRef]
- Devaraj, M.; Sasikumar, Y.; Rajendran, S.; Ponce, L.C. Review-metal organic framework based nanomaterials for electrochemical sensing of toxic heavy metal ions: Progress and their prospects. J. Electrochem. Soc. 2021, 168, 037513. [Google Scholar] [CrossRef]
- Liu, Y.; Deng, Y.; Dong, H.; Liu, K.; He, N. Progress on sensors based on nanomaterials for rapid detection of heavy metal ions. Sci. China Chem. 2016, 60, 329–337. [Google Scholar] [CrossRef]
- Li, Y.; Jiang, C. Trypsin electrochemical sensing using two-dimensional molecularly imprinted polymers on 96-well microplates. Biosens. Bioelectron. 2018, 119, 18–24. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Wang, R. Review on Fundamentals, Preparations and Applications of Imprinted Polymers. Curr. Org. Chem. 2018, 22, 1600–1618. [Google Scholar] [CrossRef]
- Dickey, F.H. Specific adsorption. J. Phys. Chem. 1955, 59, 695–707. [Google Scholar] [CrossRef]
- Beluomini, M.A.; da Silva, J.L.; de Sá, A.C.; Buffon, E.; Pereira, T.C.; Stradiotto, N.R. Electrochemical sensors based on molecularly imprinted polymer on nanostructured carbon materials: A review. J. Electroanal. Chem. 2019, 840, 343–366. [Google Scholar] [CrossRef]
- Zhao, X.; Guo, Z.; Hou, Y.; Gbologah, L.; Qiu, S.; Zeng, X.; Cao, R.; Zhang, J. AuNP-/rGO-/GCE-based molecular imprinted electrochemical sensor for rapid and sensitive detection of coumarin. Chem. Pap. 2022, 76, 3679–3690. [Google Scholar] [CrossRef]
- Zhong, C.; Yang, B.; Jiang, X.; Li, J. Current Progress of Nanomaterials in Molecularly Imprinted Electrochemical Sensing. Crit. Rev. Anal. Chem. 2017, 48, 15–32. [Google Scholar] [CrossRef]
- Cui, B.; Liu, P.; Liu, X.; Liu, S.; Zhang, Z. Molecularly imprinted polymers for electrochemical detection and analysis: Progress and perspectives. J. Mater. Res. Technol. 2020, 9, 12568–12584. [Google Scholar] [CrossRef]
- Wei, X.; Wu, T.; Yuan, Y.; Ma, X.; Li, J. Highly sensitive analysis of organometallic compounds based on molecularly imprinted electrochemical sensors. Anal. Methods 2017, 9, 1771–1778. [Google Scholar] [CrossRef]
- Pardeshi, S.; Dhodapkar, R. Advances in fabrication of molecularly imprinted electrochemical sensors for detection of contaminants and toxicants. Environ. Res. 2022, 212, 113359. [Google Scholar] [CrossRef]
- Zhou, X.; Wang, B.; Wang, R. Insights into ion-imprinted materials for the recovery of metal ions: Preparation, evaluation and application. Sep. Purif. Technol. 2022, 298, 121469. [Google Scholar] [CrossRef]
- Zhang, G.Y.; Ali, M.M.; Feng, X.; Zhou, J.T.; Hu, L.H. Mesoporous molecularly imprinted materials: From preparation to biorecognition and analysis. TrAC Trends Anal. Chem. 2021, 144, 116426. [Google Scholar] [CrossRef]
- Jakavula, S.; Biata, N.R.; Dimpe, K.M.; Pakade, V.E.; Nomngongo, P.N. A Critical Review on the Synthesis and Application of Ion-Imprinted Polymers for Selective Preconcentration, Speciation, Removal and Determination of Trace and Essential Metals from Different Matrices. Crit. Rev. Anal. Chem. 2022, 52, 314–326. [Google Scholar] [CrossRef]
- Lah, N.F.C.; Ahmad, A.L.; Low, S.C.; Shoparwe, N.F. The role of porogen-polymer complexation in atrazine imprinted polymer to work as an electrochemical sensor in water. J. Environ. Chem. Eng. 2019, 7, 103500. [Google Scholar] [CrossRef]
- Ye, L. Molecularly imprinted polymers with multi-functionality. Anal. Bioanal. Chem. 2016, 408, 1727–1733. [Google Scholar] [CrossRef]
- Sala, A.; Brisset, H.; Margaillan, A.; Mullot, J.-U.; Branger, C. Electrochemical sensors modified with ion-imprinted polymers for metal ion detection. TrAC Trends Anal. Chem. 2022, 148, 116536. [Google Scholar] [CrossRef]
- Wu, S.P.; Li, K.H.; Dai, X.Z.; Zhang, Z.H.; Ding, F.Y.; Li, S.J. An ultrasensitive electrochemical platform based on imprinted chitosan/gold nanoparticles/graphene nanocomposite for sensing cadmium (II) ions. Microchem. J. 2020, 155, 104710. [Google Scholar] [CrossRef]
- Shi, M.; Lu, T.; Li, X.; Yang, Y. Preparation and properties of GO-based lanthanum ion-imprinted polymer, La-IIP-MAA/Fe3O4-GO. J. Rare Earths 2022, 40, 135–142. [Google Scholar] [CrossRef]
- Topcu, C.; Lacin, G.; Yilmaz, V.; Coldur, F.; Caglar, B.; Cubuk, O.; Isildak, I. Electrochemical Determination of Copper(II) in Water Samples Using a Novel Ion-Selective Electrode Based on a Graphite Oxide-Imprinted Polymer Composite. Anal. Lett. 2018, 51, 1890–1910. [Google Scholar] [CrossRef]
- Baig, N.; Sajid, M.; Saleh, T.A. Recent trends in nanomaterial-modified electrodes for electroanalytical applications. TrAC Trends Anal. Chem. 2019, 111, 47–61. [Google Scholar] [CrossRef]
- Li, Y.-K.; Yang, T.; Chen, M.-L.; Wang, J.-H. Recent Advances in Nanomaterials for Analysis of Trace Heavy Metals. Crit. Rev. Anal. Chem. 2021, 51, 353–372. [Google Scholar] [CrossRef]
- Ann Maria, C.G.; Varghese, A.; Nidhin, M. Recent Advances in Nanomaterials Based Molecularly Imprinted Electrochemical Sensors. Crit. Rev. Anal. Chem. 2021, 1–10, online ahead of print. [Google Scholar] [CrossRef]
- Limthin, D.; Leepheng, P.; Klamchuen, A.; Phromyothin, D. Enhancement of Electrochemical Detection of Gluten with Surface Modification Based on Molecularly Imprinted Polymers Combined with Superparamagnetic Iron Oxide Nanoparticles. Polymers 2021, 14, 91. [Google Scholar] [CrossRef]
- Rebolledo-Perales, L.E.; Ibarra, I.; Guzmán, M.F.; Islas, G.; Romero, G.A. A novel ion-imprinted polymer based on pyrrole as functional monomer for the voltammetric determination of Hg(II) in water samples. Electrochim. Acta 2022, 434, 141258. [Google Scholar] [CrossRef]
- Chen, J.; Chen, Y.; Liang, Y. Application of chitosan-N-doped graphene oxide ion-imprinted sensor in Cd (II) ions detection. Diam. Relat. Mater. 2021, 119, 108591. [Google Scholar] [CrossRef]
- Qiu, M.; Mao, X.; Zhang, C. Lab-on-cloth integrated with a photoelectrochemical cell and ion imprinting for point-of-care testing of Hg(Ⅱ). Sens. Actuators B Chem. 2022, 361, 131689. [Google Scholar] [CrossRef]
- Zhu, F.; Li, L.; Xing, J. Selective adsorption behavior of Cd(II) ion imprinted polymers synthesized by microwave-assisted inverse emulsion polymerization: Adsorption performance and mechanism. J. Hazard. Mater. 2017, 321, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Mostafa, A.M.; Barton, S.J.; Wren, S.P.; Barker, J. Review on molecularly imprinted polymers with a focus on their application to the analysis of protein biomarkers. TrAC Trends Anal. Chem. 2021, 144, 116431. [Google Scholar] [CrossRef]
- Mafu, L.D.; Msagati, T.A.M.; Mamba, B.B. Ion-imprinted polymers for environmental monitoring of inorganic pollutants: Synthesis, characterization, and applications. Environ. Sci. Pollut. Res. 2013, 20, 790–802. [Google Scholar] [CrossRef] [PubMed]
- Fadillah, G.; Saputra, O.A.; Saleh, T.A. Trends in polymers functionalized nanostructures for analysis of environmental pollutants. Trends Environ. Anal. Chem. 2020, 26, e00084. [Google Scholar] [CrossRef]
- Shakerian, F.; Kim, K.-H.; Kwon, E.; Szulejko, J.E.; Kumar, P.; Dadfarnia, S.; Shabani, A.M.H. Advanced polymeric materials: Synthesis and analytical application of ion imprinted polymers as selective sorbents for solid phase extraction of metal ions. TrAC Trends Anal. Chem. 2016, 83, 55–69. [Google Scholar] [CrossRef]
- Rahman, S.; Bozal-Palabiyik, B.; Unal, D.N.; Erkmen, C.; Siddiq, M.; Shah, A.; Uslu, B. Molecularly imprinted polymers (MIPs) combined with nanomaterials as electrochemical sensing applications for environmental pollutants. Trends Environ. Anal. Chem. 2022, 36, e00176. [Google Scholar] [CrossRef]
- Chen, J.; Bai, H.; Xia, J.; Liu, X.; Liu, Y.; Cao, Q. Trace detection of Ce3+ by adsorption strip voltammetry at a carbon paste electrode modified with ion imprinted polymers. J. Rare Earths 2018, 36, 1121–1126. [Google Scholar] [CrossRef]
- Liu, X.; Wu, F.; Au, C.; Tao, Q.; Pi, M.; Zhang, W. Synthesis of molecularly imprinted polymer by suspension polymerization for selective extraction of p-hydroxybenzoic acid from water. J. Appl. Polym. Sci. 2019, 136, 46984. [Google Scholar] [CrossRef]
- Hande, P.; Samui, A.B.; Kulkarni, P.S. Highly selective monitoring of metals by using ion-imprinted polymers. Environ. Sci. Pollut. Res. 2015, 22, 7375–7404. [Google Scholar] [CrossRef] [PubMed]
- Chaipuang, A.; Phungpanya, C.; Thongpoon, C.; Watla-Iad, K.; Inkaew, P.; Machan, T.; Suwantong, O. Synthesis of copper(II) ion-imprinted polymers via suspension polymerization. Polym. Adv. Technol. 2018, 29, 3134–3141. [Google Scholar] [CrossRef]
- Mishra, S.; Verma, N. Surface ion imprinting-mediated carbon nanofiber-grafted highly porous polymeric beads: Synthesis and application towards selective removal of aqueous Pb(II). Chem. Eng. J. 2017, 313, 1142–1151. [Google Scholar] [CrossRef]
- He, J.; Shang, H.; Zhang, X.; Sun, X. Synthesis and application of ion imprinting polymer coated magnetic multi-walled carbon nanotubes for selective adsorption of nickel ion. Appl. Surf. Sci. 2018, 428, 110–117. [Google Scholar] [CrossRef]
- Wei, P.; Li, Z.; Zhao, X.; Song, R.; Zhu, Z. Fe3O4/SiO2/CS surface ion-imprinted polymer modified glassy carbon electrode for highly sensitivity and selectivity detection of toxic metal ions. J. Taiwan Inst. Chem. Eng. 2020, 113, 107–113. [Google Scholar] [CrossRef]
- Ghanei-Motlagh, M.; Taher, M. Novel imprinted polymeric nanoparticles prepared by sol-gel technique for electrochemical detection of toxic cadmium(II) ions. Chem. Eng. J. 2017, 327, 135–141. [Google Scholar] [CrossRef]
- Ismail, A.S.; Ishak, N.; Kamarudin, Q.; Ewe, V.S.H.; Mustapa, N.B.; Nasir, A.M. Synthesis of Graphite-Based Ion-Imprinted Polymer for the Selective Removal of Nitrate Ions. Chem. Eng. Technol. 2022, 45, 1803–1810. [Google Scholar] [CrossRef]
- Fayazi, M.; Taher, M.A.; Afzali, D.; Mostafavi, A.; Ghanei-Motlagh, M. Synthesis and application of novel ion-imprinted polymer coated magnetic multi-walled carbon nanotubes for selective solid phase extraction of lead(II) ions. Mater. Sci. Eng. C 2016, 60, 365–373. [Google Scholar] [CrossRef]
- Wei, J.; Liu, C.; Wu, T.; Zeng, W.; Hu, B.; Zhou, S.; Wu, L. A review of current status of ratiometric molecularly imprinted electrochemical sensors: From design to applications. Anal. Chim. Acta 2022, 1230, 340273. [Google Scholar] [CrossRef]
- Wang, X.; Qi, Y.; Shen, Y.; Yuan, Y.; Zhang, L.; Zhang, C.; Sun, Y. A ratiometric electrochemical sensor for simultaneous detection of multiple heavy metal ions based on ferrocene-functionalized metal-organic framework. Sens. Actuators B Chem. 2020, 310, 127756. [Google Scholar] [CrossRef]
- Lochab, A.; Sharma, R.; Kumar, S.; Saxena, R. Recent advances in carbon based nanomaterials as electrochemical sensor for toxic metal ions in environmental applications. Mater. Today Proc. 2021, 45, 3741–3753. [Google Scholar] [CrossRef]
- Ab Latif, F.E.; Numan, A.; Mubarak, N.M.; Khalid, M.; Abdullah, E.C.; Manaf, N.A.; Walvekar, R. Evolution of MXene and its 2D heterostructure in electrochemical sensor applications. Coord. Chem. Rev. 2022, 471, 214755. [Google Scholar] [CrossRef]
- Moradi, O. A review on nanomaterial-based electrochemical sensors for determination of vanillin in food samples. Food Chem. Toxicol. 2022, 168, 113391. [Google Scholar] [CrossRef]
- Ardalani, M.; Shamsipur, M.; Besharati-Seidani, A. A new generation of highly sensitive potentiometric sensors based on ion imprinted polymeric nanoparticles/multiwall carbon nanotubes/polyaniline/graphite electrode for sub-nanomolar detection of lead(II) ions. J. Electroanal. Chem. 2020, 879, 114788. [Google Scholar] [CrossRef]
- Rebelo, P.; Costa-Rama, E.; Seguro, I.; Pacheco, J.G.; Nouws, H.P.A.; Cordeiro, M.N.D.S.; Delerue-Matos, C. Molecularly imprinted polymer-based electrochemical sensors for environmental analysis. Biosens. Bioelectron. 2021, 172, 112719. [Google Scholar] [CrossRef] [PubMed]
- Mostafiz, B.; Bigdeli, S.A.; Banan, K.; Afsharara, H.; Hatamabadi, D.; Mousavi, P.; Hussain, C.M.; Kecili, R.; Ghorbani-Bidkorbeh, F. Molecularly imprinted polymer-carbon paste electrode (MIP-CPE)-based sensors for the sensitive detection of organic and inorganic environmental pollutants: A review. Trends. Environ. Anal. Chem. 2021, 32, e00144. [Google Scholar] [CrossRef]
- Di Masi, S.; Pennetta, A.; Guerreiro, A.; Canfarotta, F.; De Benedetto, G.E.; Malitesta, C. Sensor based on electrosynthesised imprinted polymeric film for rapid and trace detection of copper(II) ions. Sens. Actuators B Chem. 2019, 307, 127648. [Google Scholar] [CrossRef]
- Sharma, P.S.; Pietrzyk-Le, A.; D’Souza, F.; Kutner, W. Electrochemically synthesized polymers in molecular imprinting for chemical sensing. Anal. Bioanal. Chem. 2012, 402, 3177–3204. [Google Scholar] [CrossRef]
- Torkashvand, M.; Gholivand, M.; Azizi, R. Synthesis, characterization and application of a novel ion-imprinted polymer based voltammetric sensor for selective extraction and trace determination of cobalt (II) ions. Sens. Actuators B Chem. 2017, 243, 283–291. [Google Scholar] [CrossRef]
- Wu, S.; Dai, X.; Cheng, T.; Li, S. Highly sensitive and selective ion-imprinted polymers based on one-step electrodeposition of chitosan-graphene nanocomposites for the determination of Cr(VI). Carbohydr. Polym. 2018, 195, 199–206. [Google Scholar] [CrossRef] [PubMed]
- Prasad, B.B.; Jauhari, D. Double-ion imprinted polymer @magnetic nanoparticles modified screen printed carbon electrode for simultaneous analysis of cerium and gadolinium ions. Anal. Chim. Acta 2015, 875, 83–91. [Google Scholar] [CrossRef] [PubMed]
- Kumar, D.; Madhuri, R.; Tiwari, M.P.; Sinha, P.; Prasad, B.B. Molecularly imprinted polymer-modified electrochemical sensor for simultaneous determination of copper and zinc. Adv. Mater. Lett. 2011, 2, 294–297. [Google Scholar] [CrossRef]
- Wei, P.; Zhu, Z.; Song, R.; Li, Z.; Chen, C. An ion-imprinted sensor based on chitosan-graphene oxide composite polymer modified glassy carbon electrode for environmental sensing application. Electrochim. Acta 2019, 317, 93–101. [Google Scholar] [CrossRef]
- Wu, S.; Li, K.; Zhang, Z.; Chen, L. Synthesis of imprinted chitosan/AuNPs/graphene-coated MWCNTs/Nafion film for detection of lead ions. New J. Chem. 2020, 44, 14129–14135. [Google Scholar] [CrossRef]
- Hu, S. An Electrochemical Sensor Based on ion Imprinted PPy/rGO Composite for Cd(II) Determination in Water. Int. J. Electrochem. Sci. 2019, 14, 11714–11730. [Google Scholar] [CrossRef]
- Wang, J.; Hu, J.; Hu, S.; Gao, G.; Song, Y. A Novel Electrochemical Sensor Based on Electropolymerized Ion Imprinted PoPD/ERGO Composite for Trace Cd(II) Determination in Water. Sensors 2020, 20, 1004. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fu, X.-C.; Wu, J.; Nie, L.; Xie, C.-G.; Liu, J.-H.; Huang, X.-J. Electropolymerized surface ion imprinting films on a gold nanoparticles/single-wall carbon nanotube nanohybrids modified glassy carbon electrode for electrochemical detection of trace mercury(II) in water. Anal. Chim. Acta 2012, 720, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Ait-Touchente, Z.; Sakhraoui, H.E.E.; Fourati, N.; Zerrouki, C.; Maouche, N.; Yaakoubi, N.; Touzani, R.; Chehimi, M.M. High performance zinc oxide nanorod-doped ion imprinted polypyrrole for the selective electrosensing of mercury(II) ions. Appl. Sci. 2020, 10, 7010. [Google Scholar] [CrossRef]
- Ma, W.; Chang, Q.; Zhao, J.; Ye, B.-C. Novel electrochemical sensing platform based on ion imprinted polymer with nanoporous gold for ultrasensitive and selective determination of As3+. Mikrochim. Acta 2020, 187, 571. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Bai, H.; Xia, J.; Li, Z.; Liu, P.; Cao, Q. Electrochemical sensor for detection of europium based on poly-catechol and ion-imprinted sol-gel film modified screen-printed electrode. J. Electroanal. Chem. 2018, 824, 32–38. [Google Scholar] [CrossRef]
- Fadillah, G.; Inayatussholeha, E.N.; Mukarom, N.A.; Rattyananda, B.S.; Wicaksono, W.P.; Fatimah, I.; Saleh, T.A. Ion imprinted-carbon paste electrode as electrochemical sensor for ultra-trace recognizing speciation of mercury. Results Chem. 2022, 4, 100489. [Google Scholar] [CrossRef]
- Zhang, H.; Cui, J.; Zeng, Y.X.; Zhang, Y.; Pei, Y.S. Direct electrodeposition of carbon dots modifying bismuth film electrode for sensitive detection of Cd2+ and Pb2+. J. Electrochem. Soc. 2022, 169, 017501. [Google Scholar] [CrossRef]
- Power, A.C.; Gorey, B.; Chandra, S.; Chapman, J. Carbon nanomaterials and their application to electrochemical sensors: A review. Nanotechnol. Rev. 2018, 7, 19–41. [Google Scholar] [CrossRef]
- Alhans, R.; Singh, A.; Singhal, C.; Narang, J.; Wadhwa, S.; Mathur, A. Comparative analysis of single-walled and multi-walled carbon nanotubes for electrochemical sensing of glucose on gold printed circuit boards. Mater. Sci. Eng. C 2018, 90, 273–279. [Google Scholar] [CrossRef] [PubMed]
- Karimi-Maleh, H.; Beitollahi, H.; Kumar, P.S.; Tajik, S.; Jahani, P.M.; Karimi, F.; Karaman, C.; Vasseghian, Y.; Baghayeri, M.; Rouhi, J.; et al. Recent advances in carbon nanomaterials-based electrochemical sensors for food azo dyes detection. Food Chem. Toxicol. 2022, 164, 112961. [Google Scholar] [CrossRef] [PubMed]
- Deshmukh, M.A.; Shirsat, M.D.; Ramanaviciene, A.; Ramanavicius, A. Composites Based on Conducting Polymers and Carbon Nanomaterials for Heavy Metal Ion Sensing (Review). Crit. Rev. Anal. Chem. 2018, 48, 293–304. [Google Scholar] [CrossRef] [PubMed]
- Ahammad, A.J.S.; Lee, J.-J.; Rahman, A. Electrochemical Sensors Based on Carbon Nanotubes. Sensors 2009, 9, 2289–2319. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.L.; Stoddart, J.F. Noncovalent functionalization of single-walled carbon nanotubes. Acc. Chem. Res. 2009, 42, 1161–1171. [Google Scholar] [CrossRef]
- Rajabi, H.R.; Roushani, M.; Shamsipur, M. Development of a highly selective voltammetric sensor for nanomolar detection of mercury ions using glassy carbon electrode modified with a novel ion imprinted polymeric nanobeads and multi-wall carbon nanotubes. J. Electroanal. Chem. 2013, 693, 16–22. [Google Scholar] [CrossRef]
- Nasiri-Majd, M.; Taher, M.A.; Fazelirad, H. Synthesis and application of nano-sized ionic imprinted polymer for the selective voltammetric determination of thallium. Talanta 2015, 144, 204–209. [Google Scholar] [CrossRef] [PubMed]
- Alizadeh, T.; Ganjali, M.R.; Akhoundian, M.; Norouzi, P. Voltammetric determination of ultratrace levels of cerium(III) using a carbon paste electrode modified with nano-sized cerium-imprinted polymer and multiwalled carbon nanotubes. Mikrochim. Acta 2016, 183, 1123–1130. [Google Scholar] [CrossRef]
- Alizadeh, T.; Mirzaee, S.; Rafiei, F. All-solid-state Cr(III)-selective potentiometric sensor based on Cr(III)-imprinted polymer nanomaterial/MWCNTs/carbon nanocomposite electrode. Int. J. Environ. Anal. Chem. 2017, 97, 1283–1297. [Google Scholar] [CrossRef]
- Zhiani, R.; Ghanei-Motlag, M.; Razavipanah, I. Selective voltammetric sensor for nanomolar detection of silver ions using carbon paste electrode modified with novel nanosized Ag(I)-imprinted polymer. J. Mol. Liq. 2016, 219, 554–560. [Google Scholar] [CrossRef]
- Tarley, C.R.T.; Basaglia, A.M.; Segatelli, M.G.; Prete, M.C.; Suquila, F.A.C.; de Oliveira, L.L.G. Preparation and application of nanocomposite based on imprinted poly(methacrylic acid)-PAN/MWCNT as a new electrochemical selective sensing platform of Pb2+ in water samples. J. Electroanal. Chem. 2017, 801, 114–121. [Google Scholar] [CrossRef]
- Alizadeh, T.; Hamidi, N.; Ganjali, M.R.; Rafiei, F. An extraordinarily sensitive voltammetric sensor with picomolar detection limit for Pb2+ determination based on carbon paste electrode impregnated with nano-sized imprinted polymer and multi-walled carbon nanotubes. J. Environ. Chem. Eng. 2017, 5, 4327–4336. [Google Scholar] [CrossRef]
- Sebastian, M.; Mathew, B. Carbon nanotube-based ion imprinted polymer as electrochemical sensor and sorbent for Zn(II) ion from paint industry wastewater. Int. J. Polym. Anal. Charact. 2017, 23, 18–28. [Google Scholar] [CrossRef]
- Aravind, A.; Mathew, B. Electrochemical sensor based on nanostructured ion imprinted polymer for the sensing and extraction of Cr(III) ions from industrial wastewater. Polym. Int. 2018, 67, 1595–1604. [Google Scholar] [CrossRef]
- Sebastian, M.; Mathew, B. Multiwalled carbon nanotube based ion imprinted polymer as sensor and sorbent for environmental hazardous cobalt ion. J. Macromol. Sci. Part A 2018, 55, 455–465. [Google Scholar] [CrossRef]
- Aravind, A.; Mathew, B. An electrochemical sensor and sorbent based on mutiwalled carbon nanotube supported ion imprinting technique for Ni(II) ion from electroplating and steel industries. SN Appl. Sci. 2018, 1, 23. [Google Scholar] [CrossRef] [Green Version]
- Sebastian, M.; Mathew, B. Ion imprinting approach for the fabrication of an electrochemical sensor and sorbent for lead ions in real samples using modified multiwalled carbon nanotubes. J. Mater. Sci. 2017, 53, 3557–3572. [Google Scholar] [CrossRef]
- Aravind, A.; Mathew, B. Tailoring of nanostructured material as an electrochemical sensor and sorbent for toxic Cd(II) ions from various real samples. J. Anal. Sci. Technol. 2018, 9, 22. [Google Scholar] [CrossRef] [Green Version]
- Aravind, A.; Mathew, B. Nano layered ion imprinted polymer based electrochemical sensor and sorbent for Mn (II) ions from real samples. J. Macromol. Sci. Part A 2020, 57, 256–265. [Google Scholar] [CrossRef]
- Roushani, M.; Saedi, Z.; Hamdi, F.; Dizajdizi, B.Z. Preparation an electrochemical sensor for detection of manganese (II) ions using glassy carbon electrode modified with multi walled carbon nanotube-chitosan-ionic liquid nanocomposite decorated with ion imprinted polymer. J. Electroanal. Chem. 2017, 804, 1–6. [Google Scholar] [CrossRef]
- Reddy, Y.V.M.; Shin, J.H.; Palakollu, V.N.; Sravani, B.; Choi, C.-H.; Park, K.; Kim, S.-K.; Madhavi, G.; Park, J.P.; Shetti, N.P. Strategies, advances, and challenges associated with the use of graphene-based nanocomposites for electrochemical biosensors. Adv. Colloid Interface Sci. 2022, 304, 102664. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Ruan, T.; Chen, Y.; Jin, F.; Peng, L.; Zhou, Y.; Wang, D.; Dou, S. Graphene-based composites for electrochemical energy storage. Energy Storage Mater. 2020, 24, 22–51. [Google Scholar] [CrossRef]
- Hsine, Z.; Mlika, R.; Jaffrezic-Renault, N.; Korri-Youssoufi, H. Review—Recent Progress in Graphene Based Modified Electrodes for Electrochemical Detection of Dopamine. Chemosensors 2022, 10, 249. [Google Scholar] [CrossRef]
- Amiri, M.; Nekoueian, K.; Saberi, R.S. Graphene-family materials in electrochemical aptasensors. Anal. Bioanal. Chem. 2021, 413, 673–699. [Google Scholar] [CrossRef]
- Bai, H.; Wang, S.; Liu, P.; Xiong, C.; Zhang, K.; Cao, Q. Electrochemical sensor based on in situ polymerized ion-imprinted membranes at graphene modified electrode for palladium determination. J. Electroanal. Chem. 2016, 771, 29–36. [Google Scholar] [CrossRef]
- Zuo, Y.; Xu, J.; Zhu, X.; Duan, X.; Lu, L.; Yu, Y. Graphene-derived nanomaterials as recognition elements for electrochemical determination of heavy metal ions: A review. Microchim. Acta 2019, 186, 171. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhang, B.; Tang, Y.; Zhao, F.; Zeng, B. Fabrication and application of a rutin electrochemical sensor based on rose-like AuNPs-MoS2-GN composite and molecularly imprinted chitosan. Microchem. J. 2021, 168, 106505. [Google Scholar] [CrossRef]
- Shirzadmehr, A.; Rezaei, M.; Bagheri, H.; Khoshsafar, H. Novel potentiometric sensor for the trace-level determination of Zn2+ based on a new nanographene/ion imprinted polymer composite. Int. J. Environ. Anal. Chem. 2016, 96, 929–944. [Google Scholar] [CrossRef]
- Yavuz, S.; Erkal, A.; Kariper, A.; Solak, A.O.; Jeon, S.; Mülazımoğlu, I.E.; Üstündağ, Z. Carbonaceous Materials-12: A Novel Highly Sensitive Graphene Oxide-Based Carbon Electrode: Preparation, Characterization, and Heavy Metal Analysis in Food Samples. Food Anal. Methods 2015, 9, 322–331. [Google Scholar] [CrossRef]
- Yasinzai, M.; Mustafa, G.; Asghar, N.; Ullah, I.; Zahid, M.; Lieberzeit, P.A.; Han, D.; Latif, U. Ion-imprinted polymer-based receptors for sensitive and selective detection of mercury ions in aqueous environment. J. Sens. 2018, 2018, 8972549. [Google Scholar] [CrossRef]
- Abdallah, A.B.; El-Kholany, M.R.; Molouk, A.F.S.; Ali, T.A.; El-Shafei, A.A.; Khalifa, M.E. Selective and sensitive electrochemical sensors based on an ion imprinting polymer and graphene oxide for the detection of ultra-trace Cd(ii) in biological samples. RSC Adv. 2021, 11, 30771–30780. [Google Scholar] [CrossRef]
- Wu, S.; He, Q.; Tan, C.; Wang, Y.; Zhang, H. Graphene-Based Electrochemical Sensors. Small 2013, 9, 1160–1172. [Google Scholar] [CrossRef] [PubMed]
- Roy, E.; Patra, S.; Kumar, D.; Madhuri, R.; Sharma, P.K. RETRACTED: Multifunctional magnetic reduced graphene oxide dendrites: Synthesis, characterization and their applications. Biosens. Bioelectron. 2015, 68, 726–735. [Google Scholar] [CrossRef] [PubMed]
- Ghanei-Motlagh, M.; Taher, M.A.; Heydari, A.; Ghanei-Motlagh, R.; Gupta, V.K. A novel voltammetric sensor for sensitive detection of mercury(II) ions using glassy carbon electrode modified with graphene-based ion imprinted polymer. Mater. Sci. Eng. C 2016, 63, 367–375. [Google Scholar] [CrossRef] [PubMed]
- Shirzadmehr, A.; Afkhami, A.; Madrakian, T. A new nano-composite potentiometric sensor containing an Hg2+-ion imprinted polymer for the trace determination of mercury ions in different matrices. J. Mol. Liq. 2015, 204, 227–235. [Google Scholar] [CrossRef]
- Yan, Y.; Gong, J.; Chen, J.; Zeng, Z.; Huang, W.; Pu, K.; Liu, J.; Chen, P. Recent advances on graphene quantum dots-from chemistry and physics to applications. Adv. Mater. 2019, 31, 1808283. [Google Scholar] [CrossRef]
- Kumar, Y.R.; Deshmukh, K.; Sadasivuni, K.K.; Pasha, S.K.K. Graphene quantum dot based materials for sensing, bio-imaging and energy storage applications: A review. RSC Adv. 2020, 10, 23861–23898. [Google Scholar] [CrossRef] [PubMed]
- Campuzano, S.; Yáñez-Sedeño, P.; Pingarrón, J.M. Carbon Dots and Graphene Quantum Dots in Electrochemical Biosensing. Nanomaterials 2019, 9, 634. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Soman, S.; Aswathy, P.V.; Kala, R. Covalently modified graphene quantum dot using a thiourea based imprinted polymer for the selective electrochemical sensing of Hg(II) ions. J. Polym. Res. 2021, 28, 359. [Google Scholar] [CrossRef]
- Wu, L.; Chen, Y.; Li, Y.; Meng, Q.; Duan, T. Functionally integrated g-C3N4@wood-derived carbon with an orderly interconnected porous structure. Appl. Surf. Sci. 2021, 540, 148440. [Google Scholar] [CrossRef]
- Wang, P.; Liu, Y.; Jiang, N.; Jing, R.; Li, S.; Zhang, Q.; Liu, H.; Xiu, J.; Li, Z.; Liu, Y. Double S-scheme AgBr heterojunction co-modified with g-C3N4 and black phosphorus nanosheets greatly improves the photocatalytic activity and stability. J. Mol. Liq. 2021, 329, 115540. [Google Scholar] [CrossRef]
- Xie, K.; Wang, Y.; Zhang, K.; Zhao, R.; Chai, Z.; Du, J.; Li, J. Controllable band structure of ZnO/g-C3N4 aggregation to enhance gas sensing for the dimethylamine detection. Sens. Actuators Rep. 2022, 4, 100084. [Google Scholar] [CrossRef]
- Ahmad, R.; Tripathy, N.; Khosla, A.; Khan, M.; Mishra, P.; Ansari, W.A.; Syed, M.A.; Hahn, Y.B. Review-recent advances in nanostructured graphitic carbon nitride as a sensing material for heavy metal ions. J. Electrochem. Soc. 2020, 167, 037519. [Google Scholar] [CrossRef]
- Ganjali, M.R. A Highly Sensitive and Selective Electrochemical Mercury(II) Sensor Based on Nanoparticles of Hg(II)-imprinted Polymer and Graphitic Carbon Nitride (g-C3N4). Int. J. Electrochem. Sci. 2019, 14, 6420–6430. [Google Scholar] [CrossRef]
- Peng, L.; Li, B.L.; Zhou, C.; Li, N.B.; Setyawati, M.I.; Zou, H.L. “Naked-eye” recognition: Emerging gold nano-family for visual sensing. Appl. Mater. Today 2018, 11, 166–188. [Google Scholar] [CrossRef]
- Zhu, Y.; Pan, D.; Hu, X.; Han, H.; Lin, M.; Wang, C. An electrochemical sensor based on reduced graphene oxide/gold nanoparticles modified electrode for determination of iron in coastal waters. Sens. Actuators B Chem. 2017, 243, 1–7. [Google Scholar] [CrossRef]
- Xiao, T.; Huang, J.; Wang, D.; Meng, T.; Yang, X. Au and Au-Based nanomaterials: Synthesis and recent progress in electrochemical sensor applications. Talanta 2020, 206, 120210. [Google Scholar] [CrossRef]
- Huang, W.; Liu, Y.; Wang, N.; Song, G.; Yin, X.; Zhang, L.; Ni, X.; Xu, W. A Sensitive Electrochemical Sensor Based on Ion Imprinted Polymers with Gold Nanoparticles for High Selective Detecting Cd (II) Ions in Real Samples. J. Inorg. Organomet. Polym. Mater. 2021, 31, 2043–2053. [Google Scholar] [CrossRef]
- Yang, Z.; Li, J.; Liu, P.; Zhang, A.; Wang, J.; Huang, Y.; Wang, J.; Wang, Z. Highly sensitive non-enzymatic hydrogen peroxide monitoring platform based on nanoporous gold via a modified solid-phase reaction method. RSC Adv. 2021, 11, 36753–36759. [Google Scholar] [CrossRef]
- Cheng, W.; Zhang, Q.; Wu, D.; Yang, Y.; Zhang, Y.; Tang, X. A facile electrochemical method for rapid determination of 3-chloropropane-1,2-diol in soy sauce based on nanoporous gold capped with molecularly imprinted polymer. Food Control 2022, 134, 108750. [Google Scholar] [CrossRef]
- Ahmed, M.M.; Zhao, R.H.; Du, J.P.; Li, J.P. Review-nanostructural ZnO-based electrochemical sensor for environmental application. J. Electrochem. Soc. 2022, 169, 020573. [Google Scholar] [CrossRef]
- Nie, Y.; Wang, Z.; Wang, J.; Bao, F.; Zhang, J.; Ma, Y.; Sham, T.-K.; Sun, X. Synthesis and Structure-Dependent Optical Properties of ZnO Nanocomb and ZnO Nanoflag. J. Phys. Chem. C 2017, 121, 26076–26085. [Google Scholar] [CrossRef]
- Ahmadi, M.; Ghoorchian, A.; Dashtian, K.; Kamalabadi, M.; Madrakian, T.; Afkhami, A. Application of magnetic nanomaterials in electroanalytical methods: A review. Talanta 2021, 225, 121974. [Google Scholar] [CrossRef]
- Yang, Y.; Yan, W.; Guo, C.; Zhang, J.; Yu, L.; Zhang, G.; Wang, X.; Fang, G.; Sun, D. Magnetic molecularly imprinted electrochemical sensors: A review. Anal. Chim. Acta 2020, 1106, 1–21. [Google Scholar] [CrossRef]
- Zhou, S.-F.; Han, X.-J.; Liu, Y.-Q. SWASV performance toward heavy metal ions based on a high-activity and simple magnetic chitosan sensing nanomaterials. J. Alloys Compd. 2016, 684, 1–7. [Google Scholar] [CrossRef]
- Ghanei-Motlagh, M.; Taher, M.A. Magnetic silver(I) ion-imprinted polymeric nanoparticles on a carbon paste electrode for voltammetric determination of silver(I). Mikrochim. Acta 2017, 184, 1691–1699. [Google Scholar] [CrossRef]
- Ghanei-Motlagh, M.; Taher, M.A. An electrochemical sensor based on novel ion imprinted polymeric nanoparticles for selective detection of lead ions. Anal. Bioanal. Chem. 2017, 4, 295–306. [Google Scholar]
- Dahaghin, Z.; Kilmartin, P.A.; Mousavi, H.Z. Simultaneous determination of lead(II) and cadmium(II) at a glassy carbon electrode modified with GO@Fe 3 O 4 @benzothiazole-2-carboxaldehyde using square wave anodic stripping voltammetry. J. Mol. Liq. 2018, 249, 1125–1132. [Google Scholar] [CrossRef]
- Dahaghin, Z.; Kilmartin, P.A.; Mousavi, H.Z. Novel ion imprinted polymer electrochemical sensor for the selective detection of lead(II). Food Chem. 2020, 303, 125374. [Google Scholar] [CrossRef]
- An, Z.; Liu, W.; Liang, Q.; Yan, G.; Qin, L.; Chen, L.; Wang, M.; Yang, Y.; Liu, X. Ion-Imprinted Polymers Modified Sensor for Electrochemical Detection of Cu2+. Nano 2019, 13, 1850140. [Google Scholar] [CrossRef]
- Fresco-Cala, B.; Batista, A.D.; Cárdenas, S. Molecularly Imprinted Polymer Micro- and Nano-Particles: A Review. Molecules 2020, 25, 4740. [Google Scholar] [CrossRef]
- Khairnar, N.A.; Jirimali, H.D.; Patil, K.P.; Gite, V.V. Zinc ion-imprinted polymer based on silica particles modified carbon paste electrodes for highly selective electrochemical determination of zinc ions. Polym. Technol. Mater. 2020, 59, 1698–1714. [Google Scholar] [CrossRef]
- Dahaghin, Z.; Kilmartin, P.A.; Mousavi, H.Z. Determination of cadmium(II) using a glassy carbon electrode modified with a Cd-ion imprinted polymer. J. Electroanal. Chem. 2018, 810, 185–190. [Google Scholar] [CrossRef]
- Afkhami, A.; Madrakian, T.; Soltani-Shahrivar, M.; Ahmadi, M.; Ghaedi, H. Selective and Sensitive Electrochemical Determination of Trace Amounts of Mercury Ion in Some Real Samples Using an Ion Imprinted Polymer Nano-Modifier. J. Electrochem. Soc. 2015, 163, B68–B75. [Google Scholar] [CrossRef]
- Wang, L.; Wang, Z.; Zhou, C.; Song, W.; Sun, C. Potentiometric microsensor based on ion-imprinted polymer for the trace determination of cesium(I) ions. J. Dispers. Sci. Technol. 2020, 41, 1095–1103. [Google Scholar] [CrossRef]
- Afkhami, A.; Moradi, M.; Bahiraei, A.; Madrakian, T. Fabrication of an electrochemical sensor based on a new nano-ion imprinted polymer for highly selective and sensitive determination of molybdate. Anal. Bioanal. Chem. 2018, 5, 41–53. [Google Scholar]
- Rebolledo-Perales, L.E.; Romero, G.A.; Ibarra-Ortega, I.S.; Galán-Vidal, C.A.; Pérez-Silva, I. Review—Electrochemical Determination of Heavy Metals in Food and Drinking Water Using Electrodes Modified with Ion-Imprinted Polymers. J. Electrochem. Soc. 2021, 168, 067516. [Google Scholar] [CrossRef]
- Martín-Yerga, D.; González-García, M.B.; Costa-García, A. Electrochemical determination of mercury: A review. Talanta 2013, 116, 1091–1104. [Google Scholar] [CrossRef] [PubMed]
- Paez-Hernandez, M.E.; Aguilar-Arteaga, K.; Galan-Vidal, C.A.; Palomar-Pardave, M.; Romero-Romo, M.; Ramirez-Silva, M.T. Mercury ions removal from aqueous solution using an activated composite membrane. Environ. Sci. Technol. 2005, 39, 7667–7670. [Google Scholar] [CrossRef] [PubMed]











| Target Ion | Electrode | Nanomaterial | Monomer | Electrochemical Method | Linear Range (μM) | LOD (μM) | Ref. |
|---|---|---|---|---|---|---|---|
| Co2+ | GCE | Fe3O4 | AAM | DPCSV | 5.0 × 10−4–0.02 and 0.02–0.5 | 1.0 × 10−4 | [61] |
| Cr6+ | GE | GR | CS | DPV | 1.0 × 10−3–10 | 6.4 × 10−4 | [62] |
| Ce4+ | SPCE a | Fe3O4 | But-2-enedioic acid bis-[(2-amino-ethyl)-amide] | DPASV | 1.78 × 10−3–4.45 × 10−2 | 5.0 × 10−4 | [63] |
| Gd3+ | SPCE | Fe3O4 | But-2-enedioic acid bis-[(2-amino-ethyl)-amide] | DPASV | 4.71 × 10−3–6.02 × 10−2 | 1.2 × 10−4 | [63] |
| Zn2+ | PGE b | MWCNTs | BAAP c | DPASV | 4.21 × 10−4 | 1.5 × 10−3–0.364 | [64] |
| Cu2+ | PGE | MWCNTs | BAAP | DPASV | 2.5 × 10−4 | 1.54 × 10−3–0.375 | [64] |
| GCE | GO | CS | DPASV | 0.5–100 | 0.15 | [65] |
| Target Ion | Electrode | Nanomaterial | Monomer | Electrochemical Method | Linear Range (μM) | LOD (μM) | Ref. |
|---|---|---|---|---|---|---|---|
| Pb2+ | GCE | AuNPs/GR/MWCNTs | CS | DPV | 1.00 × 10−3–50 | 2.83 × 10−4 | [66] |
| Cd2+ | GCE | AuNPs/GR | CS | DPV | 0.10–0.90 | 1.62 × 10−4 | [25] |
| GCE | RGO | PPy | SWASV | 8.90 × 10−3–0.89 | 2.30 × 10−3 | [67] | |
| GCE | ERGO | PoPD | SWASV | 8.90 × 10−3–0.44 | 1.20 × 10−3 | [68] | |
| GCE | N-rGO | CS | DPV | 0.01–0.10 | 3.51 × 10−3 | [33] | |
| Hg2+ | GCE | AuNPs/SWCNTs | Mercaptobenzotriazole | DPASV | 4.00 × 10−4–9.60 × 10−2 | 8.00 × 10−5 | [69] |
| GE | Au/ZnO | Py | SWV | —— | ~1 × 10−6 | [70] | |
| As3+ | GE | NPG | P-phenylened-iamine | CV | 2.00 × 10−5–9.00 × 10−3 | 7.10 × 10−6 | [71] |
| EC Sensing Platform | Technique | Metal Selectivity | Detection Limit | Linear Range | Ref. |
|---|---|---|---|---|---|
| GCE–IIP–MWCNTs | DPV | Hg2+ | 5.0 nM | 1 × 10−8–7.0 × 10−4M | [81] |
| Tl-IP-MWCNT-CPE | DPASV | Tl+ | 0.76 ng/mL | 3.0–240 ng/mL | [82] |
| CPE-IIP-MWCNTs | —— | Cr3+ | 5.9 × 10−7 M | 1.0 μM–1.0 M | [84] |
| Ce-IIP-MWCNTs | SWV | Ce3+ | 10.0 pM | 1.0 μM–25 pM | [83] |
| Ag-IIP- MWCNTs | DPV | Ag+ | 1.2 × 10−10 M | 0.5 × 10−9–2.8 × 10−7 M | [85] |
| GCE–IIP–MWCNTs | DPV | Pb2+ | 0.16 μg/L | 5.0–10.0 μg/L | [86] |
| IIP–MWCNTs-CPE | SWASV | Pb2+ | 3.8 pM | 1.0 × 10−11–8.0 × 10−8 M | [87] |
| MWCNT-CH=CH2-IIP | DPV | Zn2+ | 1.32 × 10−4 µM | 1–5 ppm | [88] |
| MWCNT-CH=CH2-IIP | DPV | Cr3+ | 0.051 μM | 1–5 ppm | [89] |
| MWCNT-CH=CH2-IIP/PE | DPV | Co2+ | 1.01 × 10−5 μM | 1–5 ppm | [90] |
| MWCNT-CH=CH2-IIP/Pt | DPV | Ni2+ | 0.028 µM | 1–5 ppm | [91] |
| MWCNT-CH=CH2-IIP | DPV | Pb2+ | 2 × 10−2 μM | 1–5 ppm | [92] |
| MWCNT-CH=CH2-IIP/Pt | DPV | Cd2+ | 0.03 μM | 1–5 ppm | [93] |
| MWCNT-CH=CH2-IIP/Pt | DPV | Mn2+ | 0.0138 μM | 1–5 ppm | [94] |
| IIP/MWCNT/CS/IL/GCE | SWASVs | Mn2+ | 0.15 μM | 2.0–9.0 μM | [95] |
| GR-IIM-GCE | i-t | Pd2+ | 6.4 × 10−9 M | 2.0 × 10−8–2.0 × 10−4 M | [100] |
| CS-GR-IIP | DPV | Cr6+ | 6.4 × 10−10 M | 1.0 × 10−9–1.0 × 10−5 M | [62] |
| CS/AuNPs/GR/GCE | DPV | Cd2+ | 1.62 × 10−4 μM | 0.1–0.9 μM | [25] |
| Ag-GR-IIP-CPE | EMF | Zn2+ | 1.93 × 10−1 μg/L | 2.62 × 10−1–6.54 × 10−5 μg/L | [103] |
| GO-IIP-IDE | —— | Hg2+ | 1 ppm | —— | [105] |
| CS/GO-IIP | DPASV | Cu2+ | 0.15 μM | 0.5–100 μM | [65] |
| GO–IIP-GCE | ASV | Cd2+ | 7 × 10−14 M | 4.2 × 10−12–5.6 × 10−3 M | [106] |
| GO/MWCNT/IIP | —— | Cu2+ | 4.0 × 10−7 M | 1.0 × 10−6–1.0 × 10−1 M | [27] |
| RGO–IIP | SWASV | Hg2+ | 0.02 μg/L | 0.07–80 μg/L | [109] |
| PPy/rGO/IIP | SWASV | Cd2+ | 0.26 μg/L | 1–100 μg/L | [67] |
| PoPD/ERGO/IIP/GCE | SWASV | Cd2+ | 0.13 ng/mL | 1–50 ng/mL | [68] |
| Al2O3/rGO/IIP | —— | Hg2+ | 1.95 × 10−9 M | 4.00 × 10−9–1.30 × 10−3 M | [110] |
| N-rGO-CS-IIP | DPV | Cd2+ | 3.51 nM | 0.01–0.1 μM | [33] |
| GQDTU-IIP | DPV | Hg2+ | 23.5 nM | 5 × 10−8M–2.3 × 10−5 M | [114] |
| GQDTU-IIP | CV | Hg2+ | 30.2 nM | 6 × 10−8M–8.5 × 10−7 M and 1.4 × 10−6 M–7 × 10−6 M | [114] |
| g-C3N4/IIP | SWASV | Hg2+ | 18 pM | 0.06–25.0 nM | [119] |
| Target Ion | Electrochemical Sensor | Monomer | Crosslinker | Initiator | Imprinting Method | Detection Technique | Linear Range | LOD | Sample | Maximum Permissibe Limit | Ref. |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Pb2+ | MWCNTs/GCE | MAA | EGDMA | AIBN | Precipitation polymerization | DPV | 5.0–10.0 μg/L | 0.16 μg/L | Drinking water, physiologi-cal serum (NaCl 0.9% m/v),and synthetic urine | 10.0 μg/L (Drinki-ng water), 0.03 mg/Kg (Fruit juice) | [86] |
| MWCNTs/CPE | ITA | EGDMA | AIBN | Precipitation polymerization | SWASV | 1.0 × 10−11–8.0 × 10−8 M | 3.8 pM | Sea and river | [87] | ||
| MWCNT-CH=CH2/Pt | AA | NNMBA | AIBN | Surface imprinting | DPV | 1–5 ppm | 2 × 10−2 μM | Waste water, lake, food sample and cosmetics | [92] | ||
| MWCNTs/Fe3O4/C-PE | 2-VP a | EGDMA | AIBN | Surface imprinting | DPSV | 3–55 μg/L | 0.5 μg/L | River, waste water | [132] | ||
| Fe3O4/GCE | 4-VP | EGDMA | AIBN | Precipitation polymerization | DPV | 0.1–80 ng/mL | 0.05 ng/mL | Fruit juice, drinking water | [134,142] | ||
| Cd2+ | MWCNT-CH=CH2/Pt | MAA | NNMBA | K2S2O8 | Precipitation polymerization | DPV | 1–5 ppm | 0.03 μM | Lake water, pigments, cosmetics and fertilizers | 3 ng/mL (Drin-king water), 3.0–5.0 μg/L (Human body) | [68,93] |
| GO/GCE | Benzo[f]chrom-ene scaffold | EGDMA | AIBN | Thermal polymerization | ASV | 4.2 × 10−12–5.6 × 10−3 | 7 × 10−14 M | Blood serum and human hair samples | [106] | ||
| AuNPs/CPE | Thiosemicarba-mide functionalized CS; MAA | EGDMA | AIBN | Surface imprinting | DPV | 10−3–100 μM | 1.43 × 10−10 M | Rice, drinking water | [123] | ||
| Fe3O4/SiO2/GCE | 2-Aminobenzimi-dazole | EGDMA | AIBN | Precipitation polymerization | DPV | 0.008–0.05 μM | 1 × 10−4 μM | Waste and drinking water | [138] | ||
| Cu2+ | GO/MWCNT/CPE | 5-methyl-2-thiozylmethacryl-amide | EGDMA | AIBN | Precipitation polymerization | —— | 1.0 × 10−6–1.0 × 10−1 M | 0.4 μM | Spiked river, dam, and tap water | 31.5 μM (Dri-nking water) | [27] |
| Fe3O4/C/GCE | NIPAM b | MBA c | APS | Surface imprinting | DPV | 1.0 × 10−5–1.0 × 10−3 M | 5.99 μM | River, tap, and mineral water | [135] | ||
| Fe3O4/SiO2/CS/Naf-ion/GCE | CS | GA d | —— | Surface imprinting | DPASV | 0.01–20 μM | 5 nM | Tap and river water | [47] | ||
| Hg2+ | MWCNTs/GCE | MAA | EGDMA | AIBN | Precipitation polymerization | DPV | 0.01–700 μM | 5.0 nM | Waste and ground water | 2 μg/L (Drinking water), 0.249 μM (In-dustrial waste water), 0.05 mg/kg (Veg-etables) | [81] |
| GO/IDE | Styrene | EGDMA | AIBN | Thermal polymerization | —— | —— | 1 ppm | —— | [105] | ||
| RGO/GCE | MAA | EGDMA | APS | Surface imprinting | SWASV | 0.07–80 μg/L | 0.02 μg/L | Waste and drinking water | [109,143] | ||
| Al2O3/rGO/CPE | MAA | EGDMA | AIBN | Thermal polymerization | —— | 0.004–1300 μM | 0.00195 μM | Drinking water, industrial waste water, food and human hair | [110,144] | ||
| GQDTU/GCE | GQDTU | EGDMA | AIBN | Suspension polymerisation | DPV | 0.05–23 μM | 23.5 nM | River and tap water | [114] | ||
| GQDTU | EGDMA | AIBN | Suspension polymerisation | CV | 0.06–0.85 and 1.4–7 μM | 30.2 nM | River and tap water | [114] | |||
| g-C3N4/CPE | ITA | EGDMA | AIBN | Precipitation polymerization | SWASV | 0.06–25.0 nM | 18 pM | Tap and sea water | [119] | ||
| SCMNPs/CPE | Functional mercaptoethylam-ino monomer | EGDMA | APS | Self-assembly homo polymerization | SWV | 0.20–1600.0 ng/mL | 0.04 ng/mL | River, waste water, and vegetables samples | [139,142] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yu, L.; Sun, L.; Zhang, Q.; Zhou, Y.; Zhang, J.; Yang, B.; Xu, B.; Xu, Q. Nanomaterials-Based Ion-Imprinted Electrochemical Sensors for Heavy Metal Ions Detection: A Review. Biosensors 2022, 12, 1096. https://doi.org/10.3390/bios12121096
Yu L, Sun L, Zhang Q, Zhou Y, Zhang J, Yang B, Xu B, Xu Q. Nanomaterials-Based Ion-Imprinted Electrochemical Sensors for Heavy Metal Ions Detection: A Review. Biosensors. 2022; 12(12):1096. https://doi.org/10.3390/bios12121096
Chicago/Turabian StyleYu, Liangyun, Liangju Sun, Qi Zhang, Yawen Zhou, Jingjing Zhang, Bairen Yang, Baocai Xu, and Qin Xu. 2022. "Nanomaterials-Based Ion-Imprinted Electrochemical Sensors for Heavy Metal Ions Detection: A Review" Biosensors 12, no. 12: 1096. https://doi.org/10.3390/bios12121096
APA StyleYu, L., Sun, L., Zhang, Q., Zhou, Y., Zhang, J., Yang, B., Xu, B., & Xu, Q. (2022). Nanomaterials-Based Ion-Imprinted Electrochemical Sensors for Heavy Metal Ions Detection: A Review. Biosensors, 12(12), 1096. https://doi.org/10.3390/bios12121096





