A Proof-of-Concept Electrochemical Skin Sensor for Simultaneous Measurement of Glial Fibrillary Acidic Protein (GFAP) and Interleukin-6 (IL-6) for Management of Traumatic Brain Injuries
Abstract
1. Introduction
2. Materials and Methods
2.1. Sensor Platform
2.2. Reagents and Instrumentation
2.3. Immunoassay Functionalization
2.4. Evaluation of Sweat Sensor Performance
2.5. Statistical Analyses
3. Results and Discussion
3.1. Sensor Design
3.2. Electrochemical Sensor Response for GFAP and IL-6
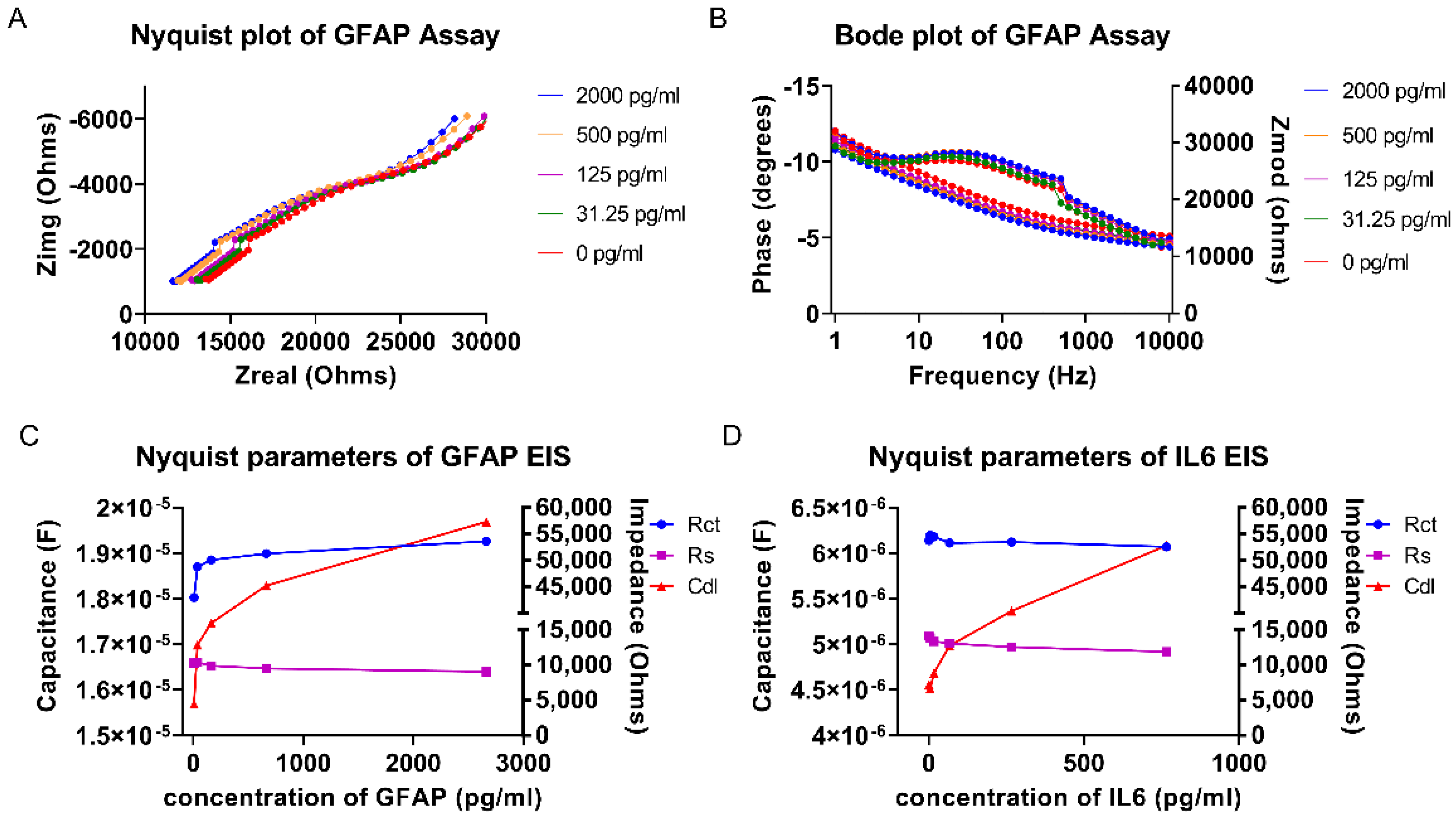
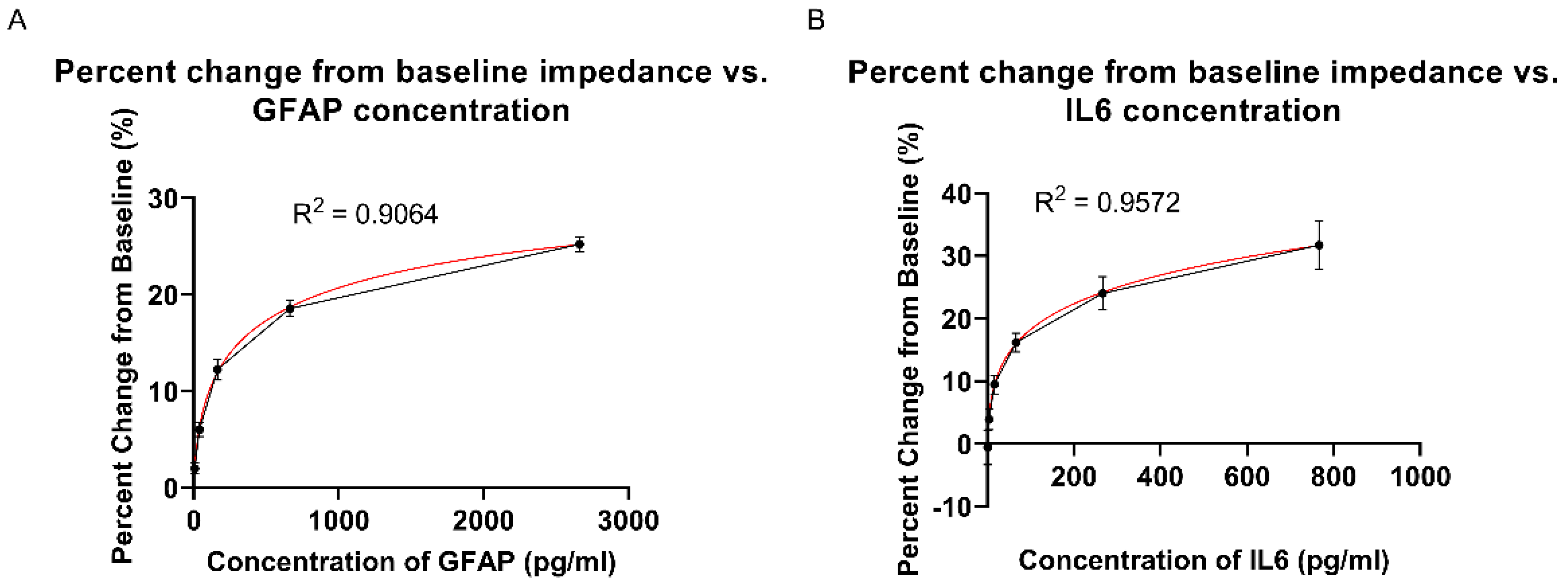
3.3. Calibrated Dose Response (CDR) Study Using EIS
3.4. Calibrated Dose Response (CDR) Study Using Chronoamperometry
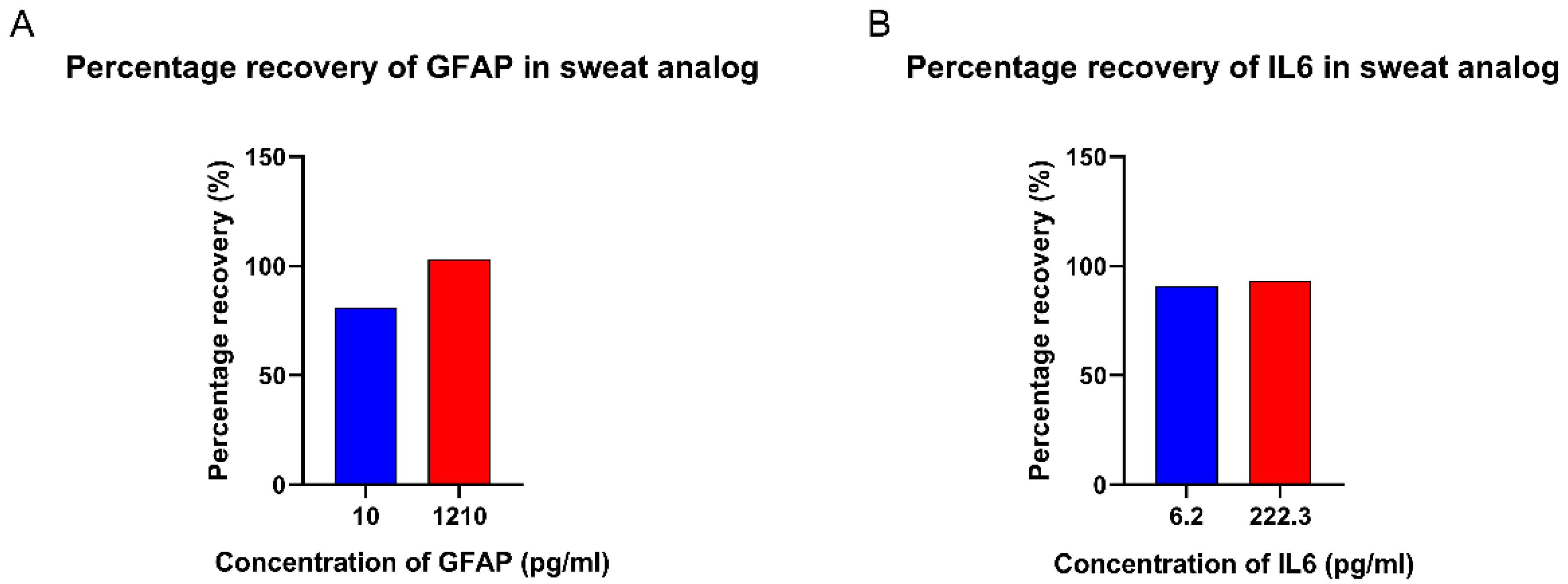
3.5. Spike and Recovery Study in Sweat Analogs
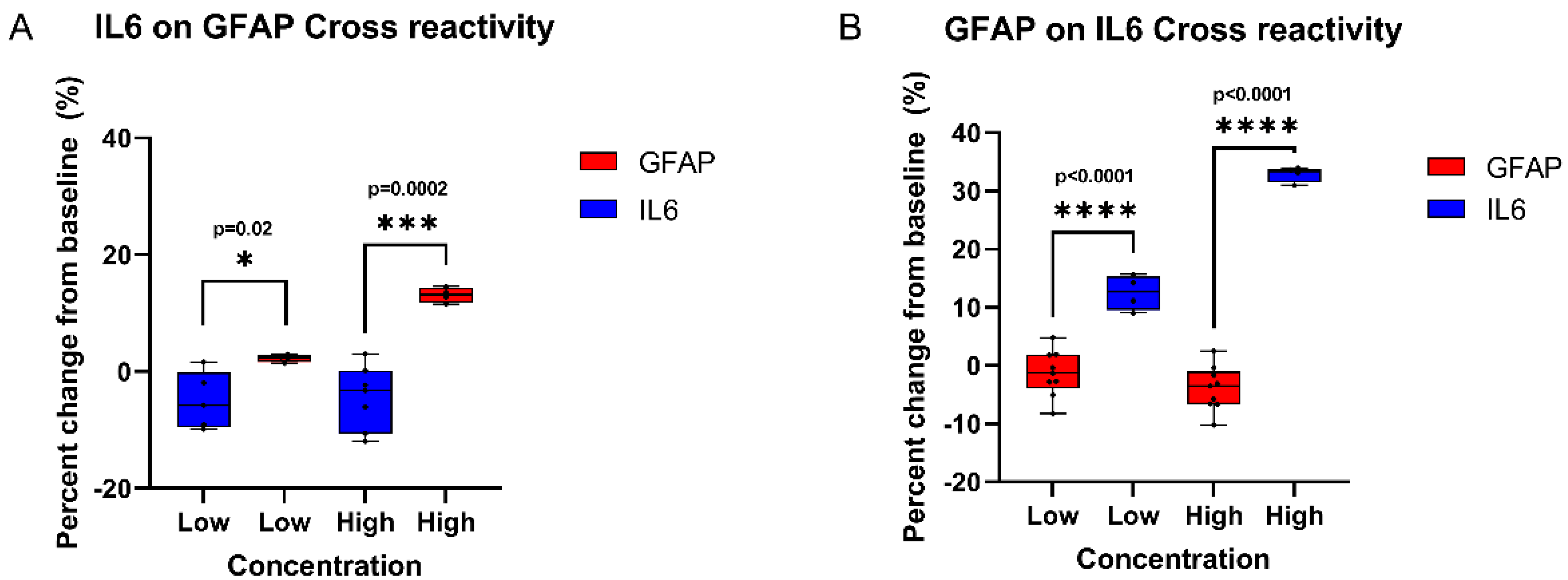
3.6. Cross-Reactivity Study
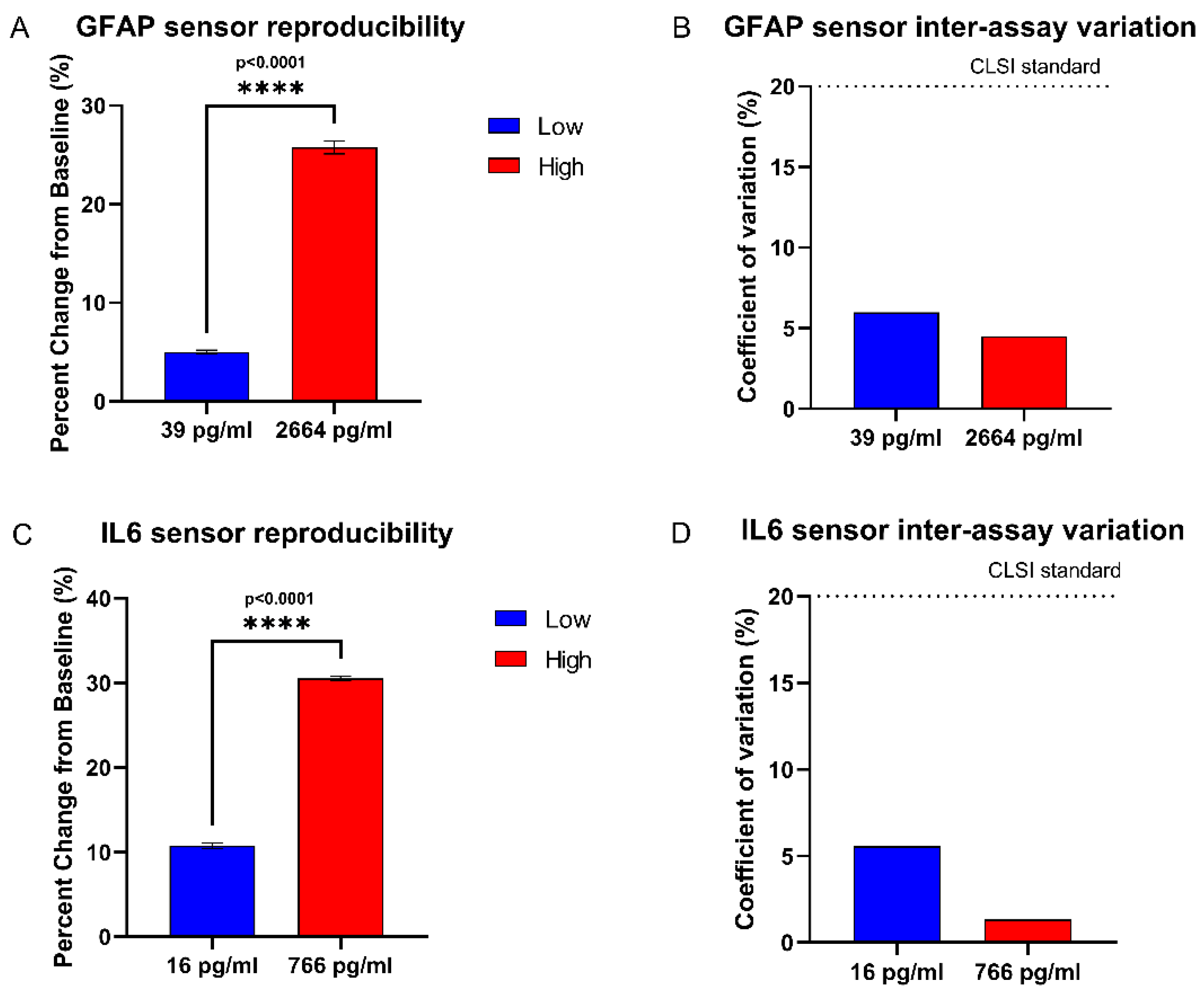
3.7. Reproducibility Study
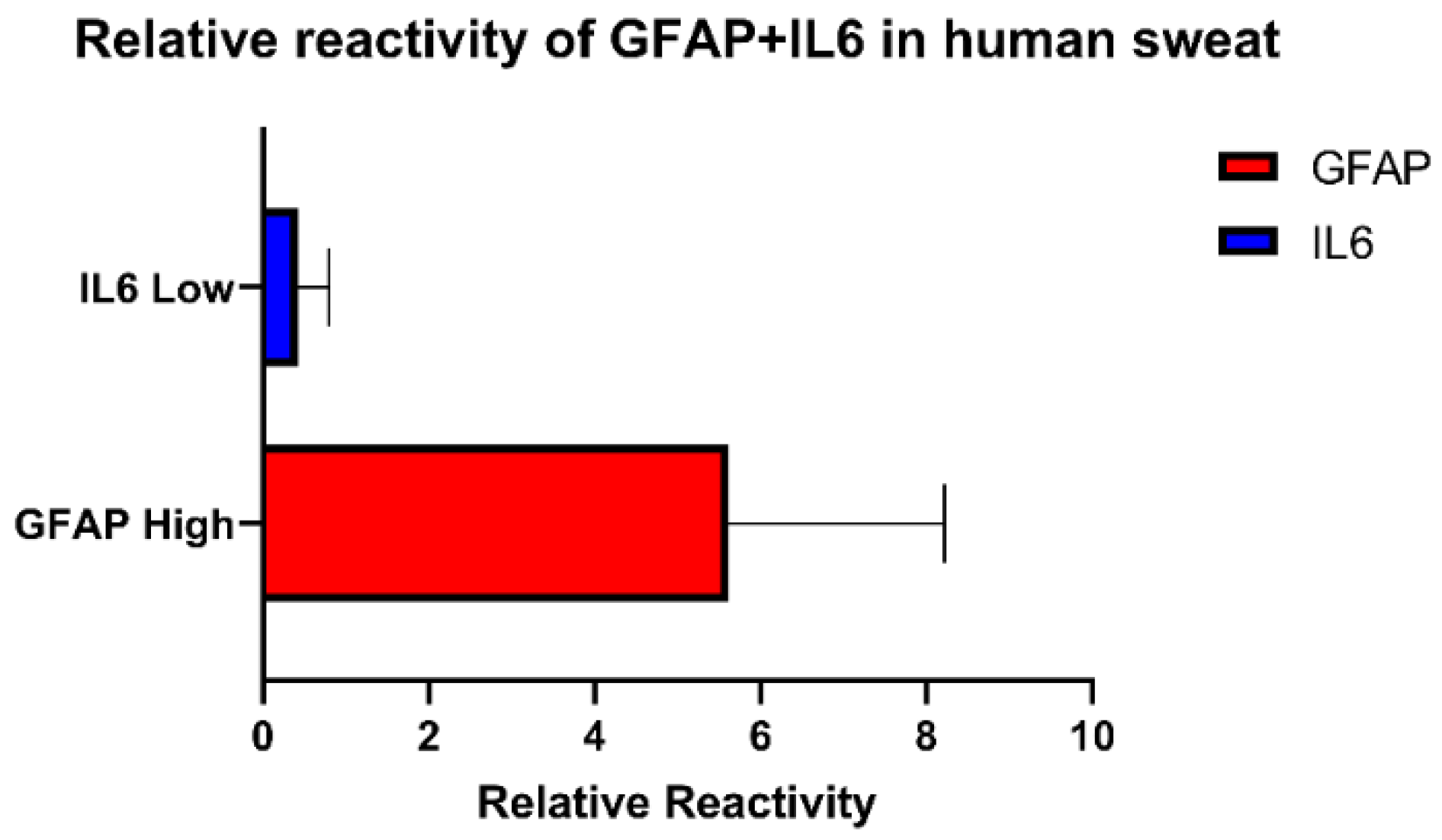
3.8. Proof of Feasibility Study in Interpreting Spiked TBI Conditions in Human Sweat
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kehoe, A.; Rennie, S.; Smith, J.E. Glasgow Coma Scale Is Unreliable for the Prediction of Severe Head Injury in Elderly Trauma Patients. Emerg. Med. J. 2014, 32, 613–615. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.K.; Yang, Z.; Zhu, T.; Shi, Y.; Rubenstein, R.; Tyndall, J.A.; Manley, G.T. An Update on Diagnostic and Prognostic Biomarkers for Traumatic Brain Injury. Expert Rev. Mol. Diagn. 2018, 18, 165–180. [Google Scholar] [CrossRef] [PubMed]
- Mena, J.H.; Sanchez, A.I.; Rubiano, A.M.; Peitzman, A.B.; Sperry, J.L.; Gutierrez, M.I.; Puyana, J.C. Effect of the Modified Glasgow Coma Scale Score Criteria for Mild Traumatic Brain Injury on Mortality Prediction: Comparing Classic and Modified Glasgow Coma Scale Score Model Scores of 13. J. Trauma: Inj. Infect. Crit. Care 2011, 71, 1185–1193. [Google Scholar] [CrossRef] [PubMed]
- Bodien, Y.; Barra, A.; Temkin, N.; Barber, J.; Foreman, B.; Robertson, C.; Vassar, M.; Manley, G.; Giacino, J.; Edlow, B. Diagnosing Level of Consciousness: The Limits of the Glasgow Coma Scale Total Score. Arch. Phys. Med. Rehabil. 2020, 101, e7. [Google Scholar] [CrossRef]
- Lannsjö, M.; Backheden, M.; Johansson, U.; af Geijerstam, J.L.; Borg, J. Does Head CT Scan Pathology Predict Outcome after Mild Traumatic Brain Injury? Eur. J. Neurol. 2012, 20, 124–129. [Google Scholar] [CrossRef] [PubMed]
- Eng, L.F.; Ghirnikar, R.S.; Lee, Y.L. Glial Fibrillary Acidic Protein: GFAP-Thirty-One Years (1969–2000). Neurochem. Res. 2000, 25, 1439–1451. [Google Scholar] [CrossRef] [PubMed]
- Shahim, P.; Politis, A.; van der Merwe, A.; Moore, B.; Ekanayake, V.; Lippa, S.M.; Chou, Y.-Y.; Pham, D.L.; Butman, J.A.; Diaz-Arrastia, R.; et al. Time Course and Diagnostic Utility of NFL, Tau, GFAP, and Uch-L1 in Subacute and Chronic TBI. Neurology 2020, 95, e623–e636. [Google Scholar] [CrossRef]
- Fraser, D.D.; Close, T.E.; Rose, K.L.; Ward, R.; Mehl, M.; Farrell, C.; Lacroix, J.; Creery, D.; Kesselman, M.; Stanimirovic, D.; et al. Severe Traumatic Brain Injury in Children Elevates Glial Fibrillary Acidic Protein in Cerebrospinal Fluid and Serum. Pediatr. Crit. Care Med. 2011, 12, 319–324. [Google Scholar] [CrossRef]
- Okonkwo, D.O.; Yue, J.K.; Puccio, A.M.; Panczykowski, D.M.; Inoue, T.; McMahon, P.J.; Sorani, M.D.; Yuh, E.L.; Lingsma, H.F.; Maas, A.I.R.; et al. GFAP-BDP as an Acute Diagnostic Marker in Traumatic Brain Injury: Results from the Prospective Transforming Research and Clinical Knowledge in Traumatic Brain Injury Study. J. Neurotrauma 2013, 30, 1490–1497. [Google Scholar] [CrossRef]
- Danielyan, L.; Tolstonog, G.; Traub, P.; Salvetter, J.; Gleiter, C.H.; Reisig, D.; Gebhardt, R.; Buniatian, G.H. Colocalization of Glial Fibrillary Acidic Protein, Metallothionein, and MHC II in Human, Rat, NOD/Scid, and Nude Mouse Skin Keratinocytes and Fibroblasts. J. Investig. Dermatol. 2007, 127, 555–563. [Google Scholar] [CrossRef][Green Version]
- Hirano, T.; Akira, S.; Taga, T.; Kishimoto, T. Biological and Clinical Aspects of Interleukin 6. Immunol. Today 1990, 11, 443–449. [Google Scholar] [CrossRef] [PubMed]
- Sjögren, F.; Anderson, C. Sterile Trauma to Normal Human Dermis Invariably Induces IL1beta, IL-6 and IL8 in an Innate Response to “Danger”. Acta Derm. Venereol. 2009, 89, 459–465. [Google Scholar] [CrossRef] [PubMed]
- Jawa, R.S.; Anillo, S.; Huntoon, K.; Baumann, H.; Kulaylat, M. Analytic Review: Interleukin-6 in Surgery, Trauma, and Critical Care: Part I: Basic Science. J. Intensive Care Med. 2011, 26, 3–12. [Google Scholar] [CrossRef] [PubMed]
- Ellison, V.J.; Mocatta, T.J.; Winterbourn, C.C.; Darlow, B.A.; Volpe, J.J.; Inder, T.E. The Relationship of CSF and Plasma Cytokine Levels to Cerebral White Matter Injury in the Premature Newborn. Pediatr. Res. 2005, 57, 282–286. [Google Scholar] [CrossRef] [PubMed]
- Jagannath, B.; Lin, K.C.; Pali, M.; Sankhala, D.; Muthukumar, S.; Prasad, S. Temporal Profiling of Cytokines in Passively Expressed Sweat for Detection of Infection Using Wearable Device. Bioeng. Transl. Med. 2021, 6, e10220. [Google Scholar] [CrossRef]
- Agostini, M.; Amato, F.; Vieri, M.L.; Greco, G.; Tonazzini, I.; Baroncelli, L.; Caleo, M.; Vannini, E.; Santi, M.; Signore, G.; et al. Glial-fibrillary-acidic-protein (GFAP) biomarker detection in Serum-matrix: Functionalization strategies and detection by an ultra-high-frequency surface-acoustic-wave (UHF-saw) lab-on-chip. Biosens. Bioelectron. 2021, 172, 112774. [Google Scholar] [CrossRef]
- Khetani, S.; Ozhukil Kollath, V.; Kundra, V.; Nguyen, M.D.; Debert, C.; Sen, A.; Karan, K.; Sanati-Nezhad, A. Polyethylenimine modified graphene-oxide electrochemical immunosensor for the detection of glial fibrillary acidic protein in central nervous system injury. ACS Sens. 2018, 3, 844–851. [Google Scholar] [CrossRef]
- Wang, T.; Fang, Y.; He, Z. Electrochemical quantitative detection of glial fibrillary acidic protein based on molecularly imprinted polymer sensor. Int. J. Electrochem. Sci. 2017, 12, 7341–7350. [Google Scholar] [CrossRef]
- Nylén, K.; Öst, M.; Csajbok, L.Z.; Nilsson, I.; Blennow, K.; Nellgård, B.; Rosengren, L. Increased serum-GFAP in patients with severe traumatic brain injury is related to outcome. J. Neurol. Sci. 2006, 240, 85–91. [Google Scholar] [CrossRef]
- Said, E.A.; Al-Reesi, I.; Al-Shizawi, N.; Jaju, S.; Al-Balushi, M.S.; Koh, C.Y.; Al-Jabri, A.A.; Jeyaseelan, L. Defining IL-6 levels in healthy individuals: A meta-analysis. J. Med. Virol. 2020, 93, 3915–3924. [Google Scholar] [CrossRef]
- Gebhard, F. Is Interleukin 6 an Early Marker of Injury Severity Following Major Trauma in Humans? Arch. Surg. 2000, 135, 291. [Google Scholar] [CrossRef] [PubMed]
- Hladek, M.D.; Szanton, S.L.; Cho, Y.-E.; Lai, C.; Sacko, C.; Roberts, L.; Gill, J. Using sweat to measure cytokines in older adults compared to younger adults: A pilot study. J. Immunol. Methods 2018, 454, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Yeung, K.K.; Huang, T.; Hua, Y.; Zhang, K.; Yuen, M.M.; Gao, Z. Recent Advances in Electrochemical Sensors for Wearable Sweat Monitoring: A Review. IEEE Sens. J. 2021, 21, 14522–14539. [Google Scholar] [CrossRef]
- Ates, H.C.; Mohsenin, H.; Wenzel, C.; Glatz, R.T.; Wagner, H.J.; Bruch, R.; Hoefflin, N.; Spassov, S.; Streicher, L.; Lozano-Zahonero, S.; et al. Biosensor-Enabled Multiplexed on-Site Therapeutic Drug Monitoring of Antibiotics. Adv. Mater. 2021, 34, 2104555. [Google Scholar] [CrossRef]
- Singh, A.; Sharma, A.; Ahmed, A.; Sundramoorthy, A.K.; Furukawa, H.; Arya, S.; Khosla, A. Recent Advances in Electrochemical Biosensors: Applications, Challenges, and Future Scope. Biosensors 2021, 11, 336. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Campbell, A.S.; de Ávila, B.E.-F.; Wang, J. Wearable Biosensors for Healthcare Monitoring. Nat. Biotechnol. 2019, 37, 389–406. [Google Scholar] [CrossRef]
- Chung, M.; Fortunato, G.; Radacsi, N. Wearable Flexible Sweat Sensors for Healthcare Monitoring: A Review. J. R. Soc. Interface 2019, 16, 20190217. [Google Scholar] [CrossRef]
- Jagannath, B.; Lin, K.-C.; Pali, M.; Sankhala, D.; Muthukumar, S.; Prasad, S. A Sweat-Based Wearable Enabling Technology for Real-Time Monitoring of Il-1β and CRP as Potential Markers for Inflammatory Bowel Disease. Inflamm. Bowel Dis. 2020, 26, 1533–1542. [Google Scholar] [CrossRef]
- Lin, K.-C.; Kinnamon, D.; Sankhala, D.; Muthukumar, S.; Prasad, S. Aware: A wearable awareness with real-time exposure, for monitoring alcohol consumption impact through ethyl glucuronide detection. Alcohol 2019, 81, 93–99. [Google Scholar] [CrossRef]
- Munje, R.D.; Muthukumar, S.; Prasad, S. Lancet-free and label-free diagnostics of glucose in sweat using zinc oxide based flexible bioelectronics. Sens. Actuators B Chem. 2017, 238, 482–490. [Google Scholar] [CrossRef]
- Jagannath, B.; Pali, M.; Lin, K.C.; Sankhala, D.; Naraghi, P.; Muthukumar, S.; Prasad, S. Novel Approach to Track the Lifecycle of Inflammation from Chemokine Expression to Inflammatory Proteins in Sweat Using Electrochemical Biosensor. Adv. Mater. Technol. 2022, 2101356. [Google Scholar] [CrossRef]
- Jagannath, B.; Muthukumar, S.; Prasad, S. Electrical Double Layer Modulation of Hybrid Room Temperature Ionic Liquid/Aqueous Buffer Interface for Enhanced Sweat Based Biosensing. Anal. Chim. Acta 2018, 1016, 29–39. [Google Scholar] [CrossRef] [PubMed]
- Tanak, A.S.; Jagannath, B.; Tamrakar, Y.; Muthukumar, S.; Prasad, S. Non-Faradaic Electrochemical Impedimetric Profiling of Procalcitonin and C-Reactive Protein as a Dual Marker Biosensor for Early Sepsis Detection. Anal. Chim. Acta X 2019, 3, 100029. [Google Scholar] [CrossRef] [PubMed]
- Kong, T.; Su, R.; Zhang, B.; Zhang, Q.; Cheng, G. CMOS-compatible, label-free silicon-nanowire biosensors to detect cardiac troponin I for acute myocardial infarction diagnosis. Biosens. Bioelectron. 2012, 34, 267–272. [Google Scholar] [CrossRef] [PubMed]
- Sadik, P.W.; Pearton, S.J.; Norton, D.P.; Lambers, E.; Ren, F. Functionalizing zn- and O-terminated zno with thiols. J. Appl. Phys. 2007, 101, 104514. [Google Scholar] [CrossRef]
- Munje, R.D.; Muthukumar, S.; Panneer Selvam, A.; Prasad, S. Flexible nanoporous tunable electrical double layer biosensors for Sweat Diagnostics. Sci. Rep. 2015, 5, 14586. [Google Scholar] [CrossRef]
- Randviir, E.P.; Banks, C.E. Electrochemical Impedance Spectroscopy: An Overview of Bioanalytical Applications. Anal. Methods 2013, 5, 1098. [Google Scholar] [CrossRef]
- Shrivastava, A.; Gupta, V.B. Methods for the determination of limit of detection and limit of quantitation of the analytical methods. Chron. Young Sci. 2011, 2, 21. [Google Scholar] [CrossRef]
- Yuan, J.-P.; Li, W.; Wang, C.; Ma, C.-Y.; Chen, L.-X.; Chen, D.-D. Nickel release rate of several nickel-containing stainless steels for jewelries. J. Iron Steel Res. Int. 2015, 22, 72–77. [Google Scholar] [CrossRef]
- Clinical and Laboratory Standards Institute. Evaluation of Precision of Quantitative Measurement Procedures: Approved Guideline; CLSI document EP05–A3; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2014. [Google Scholar]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shahub, S.; Lin, K.-C.; Muthukumar, S.; Prasad, S. A Proof-of-Concept Electrochemical Skin Sensor for Simultaneous Measurement of Glial Fibrillary Acidic Protein (GFAP) and Interleukin-6 (IL-6) for Management of Traumatic Brain Injuries. Biosensors 2022, 12, 1095. https://doi.org/10.3390/bios12121095
Shahub S, Lin K-C, Muthukumar S, Prasad S. A Proof-of-Concept Electrochemical Skin Sensor for Simultaneous Measurement of Glial Fibrillary Acidic Protein (GFAP) and Interleukin-6 (IL-6) for Management of Traumatic Brain Injuries. Biosensors. 2022; 12(12):1095. https://doi.org/10.3390/bios12121095
Chicago/Turabian StyleShahub, Sarah, Kai-Chun Lin, Sriram Muthukumar, and Shalini Prasad. 2022. "A Proof-of-Concept Electrochemical Skin Sensor for Simultaneous Measurement of Glial Fibrillary Acidic Protein (GFAP) and Interleukin-6 (IL-6) for Management of Traumatic Brain Injuries" Biosensors 12, no. 12: 1095. https://doi.org/10.3390/bios12121095
APA StyleShahub, S., Lin, K.-C., Muthukumar, S., & Prasad, S. (2022). A Proof-of-Concept Electrochemical Skin Sensor for Simultaneous Measurement of Glial Fibrillary Acidic Protein (GFAP) and Interleukin-6 (IL-6) for Management of Traumatic Brain Injuries. Biosensors, 12(12), 1095. https://doi.org/10.3390/bios12121095






