A Novel Immunoreagent for the Specific and Sensitive Detection of the Explosive Triacetone Triperoxide (TATP)
Abstract
:1. Introduction
2. Experimental Section
2.1. Reagents
2.2. Safety Note
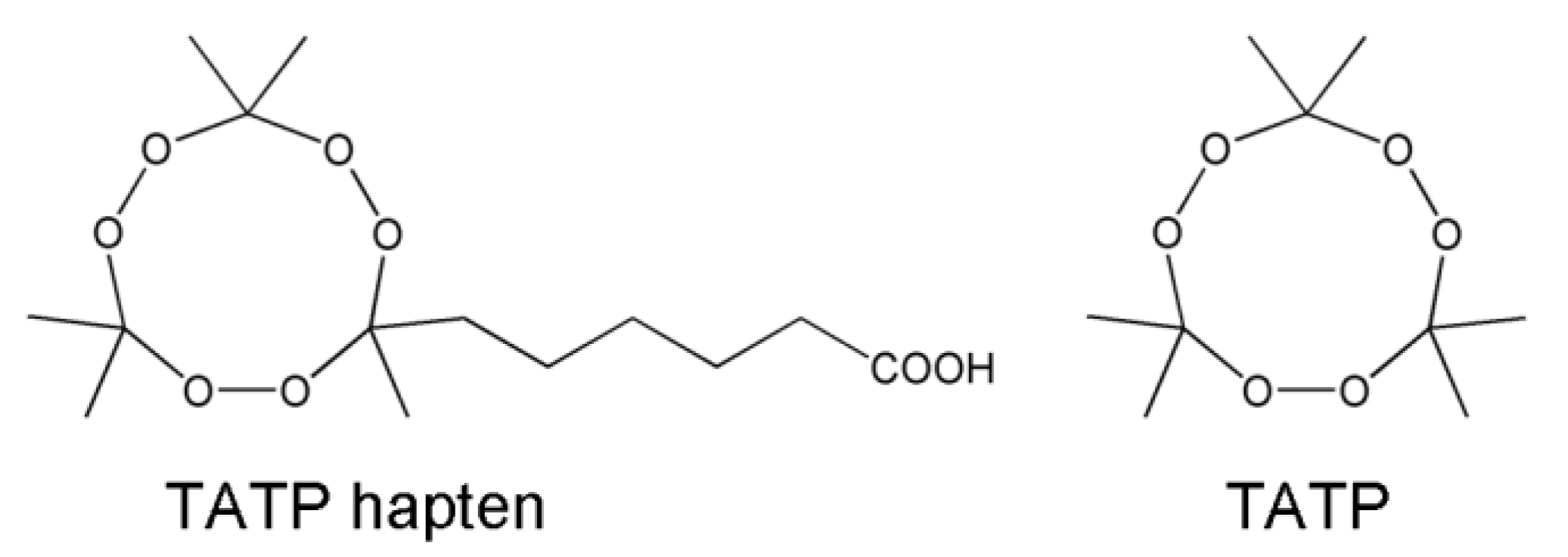
2.3. Synthesis of TATP, TATP Hapten, Immunogen and Enzyme Tracer
2.4. Immunization
2.5. Enzyme-Linked Immunosorbent Assay (ELISA)
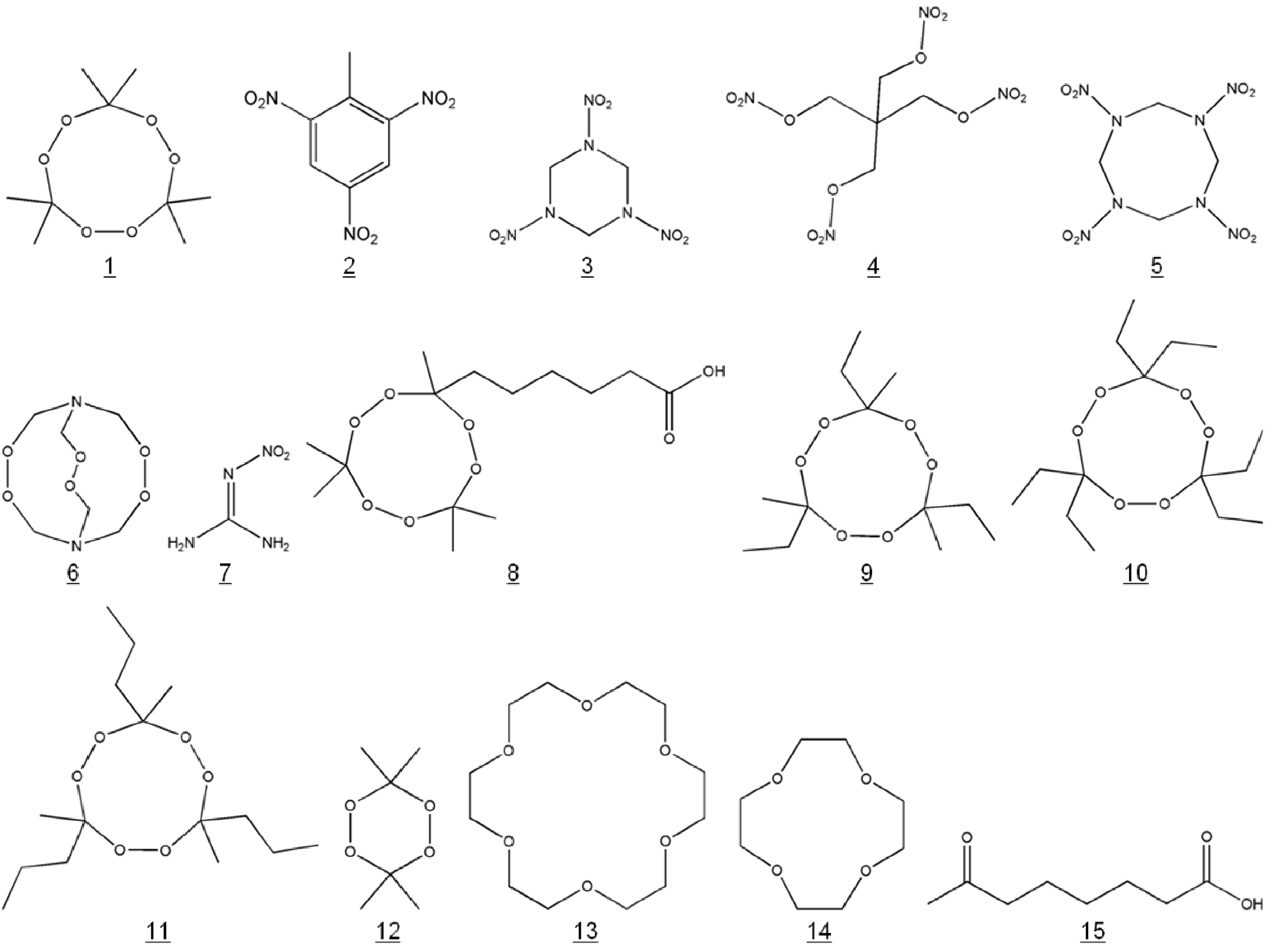
2.6. Syntheses of Cross-Reactants and Determination of Cross-Reactivities

3. Results and Discussion
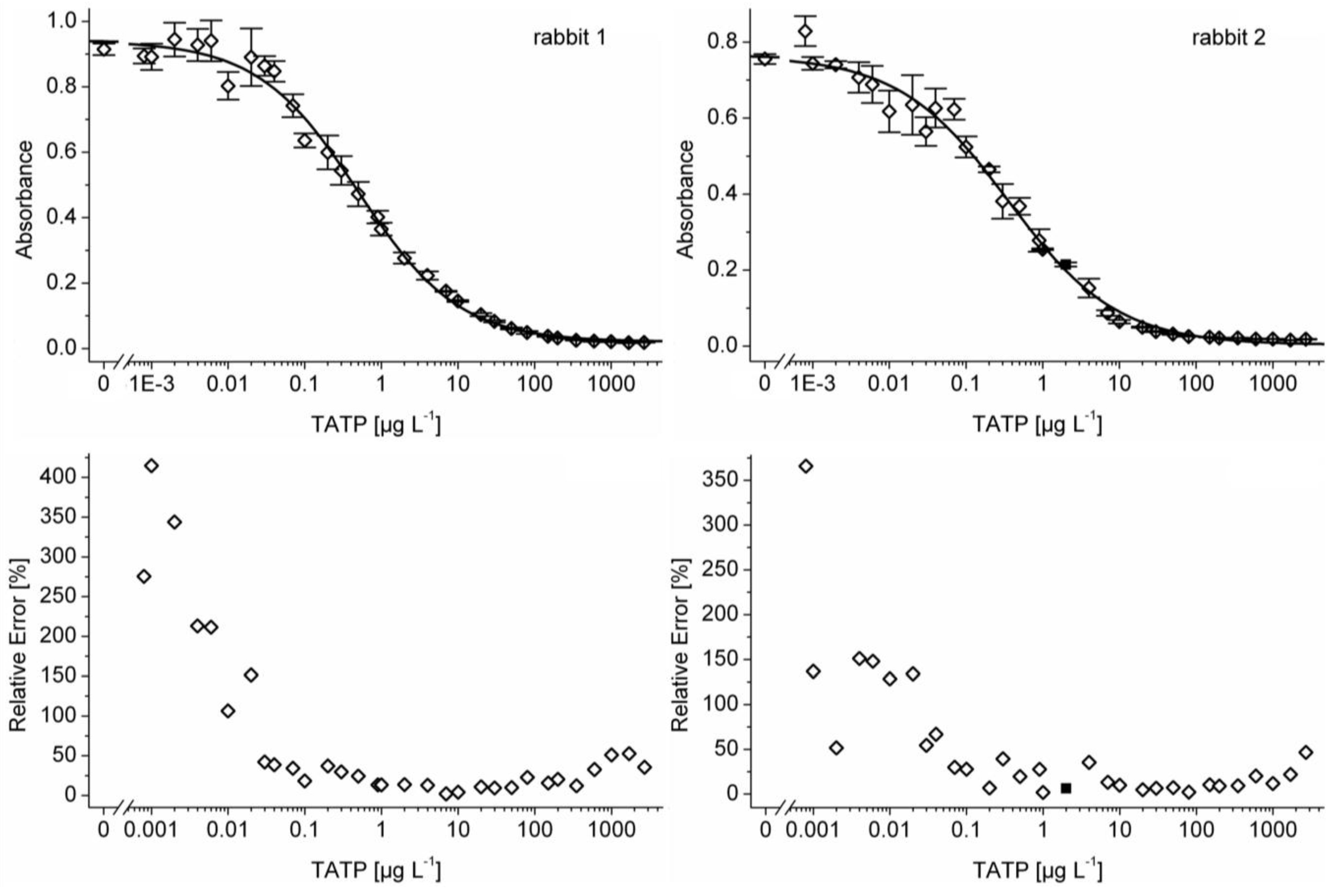
3.1. Antibody Development
3.2. Limit of Detection and Quantification Range
3.3. Cross-Reactivities
| Rabbit 1 | Rabbit 2 | Rabbit 1 | Rabbit 2 | |||
|---|---|---|---|---|---|---|
| Name | MW [g mol−1] | CR in % | molar CR in % | |||
| 1 | TATP | 222.24 | 100 | 100 | 100 | 100 |
| 2 | TNT | 227.13 | <0.01 | <0.01 | <0.01 | <0.01 |
| 3 | RDX | 222.12 | <0.01 | <0.01 | <0.01 | <0.01 |
| 4 | PETN | 316.14 | <0.01 | <0.01 | <0.01 | <0.01 |
| 5 | HMX | 296.16 | <0.01 | <0.01 | <0.01 | <0.01 |
| 6 | HMTD | 208.17 | <0.01 | <0.01 | <0.01 | <0.01 |
| 7 | Nitroguanidine | 104.07 | <0.01 | <0.01 | <0.01 | <0.01 |
| Ammonium nitrate (NH4NO3) | 80.04 | <0.01 | <0.01 | <0.01 | <0.01 | |
| Rabbit 1 | Rabbit 2 | Rabbit 1 | Rabbit 2 | |||
|---|---|---|---|---|---|---|
| Name | MW [g mol−1] | CR in % | molar CR in % | |||
| 8 | TATP hapten | 322.35 | 340 ± 70 | 330 ± 10 | 490 ± 100 | 470 ± 10 |
| 9 | Tri-butanone triperoxide * | 264.32 | 4 | 2 | 4 | 3 |
| 10 | Tri-3-pentanone triperoxide | 306.40 | 0.01 | 0.01 | 0.02 | 0.01 |
| 11 | Tri-2-pentanone triperoxide * | 306.40 | <0.01 | 0.01 | <0.01 | 0.01 |
| 12 | Diacetone diperoxide (DADP) | 148.16 | <0.01 | <0.01 | <0.01 | <0.01 |
| 13 | 18-Crown-6 | 264.32 | <0.01 | <0.01 | <0.01 | <0.01 |
| 14 | 12-Crown-4 | 176.21 | <0.01 | <0.01 | <0.01 | <0.01 |
| 15 | 7-Oxooctanoic acid | 158.19 | <0.01 | <0.01 | <0.01 | <0.01 |
| Acetone (CH3COCH3) | 58.08 | <0.01 | <0.01 | <0.01 | <0.01 | |
| Hydrogen peroxide (H2O2) | 34.01 | <0.01 | <0.01 | <0.01 | <0.01 | |
4. Conclusions
Supplementary Files
Acknowledgments
Supplementary Material
Material Availability
References
- Wolffenstein, R. Ueber die Einwirkung von Wasserstoffsuperoxyd auf Aceton und Mesityloxyd. Ber. Dtsch. Chem. Ges. 1895, 28, 2265–2269. [Google Scholar] [CrossRef]
- Muller, D.; Levy, A.; Shelef, R.; Abramovich-Bar, S.; Sonenfeld, D.; Tamiri, T. Improved method for the detection of TATP after explosion. J. Forensic Sci. 2004, 49, 935–938. [Google Scholar]
- Räsänen, R.M.; Nousiainen, M.; Perakorpi, K.; Sillanpaa, M.; Polari, L.; Anttalainen, O.; Utriainen, M. Determination of gas phase triacetone triperoxide with aspiration ion mobility spectrometry and gas chromatography-mass spectrometry. Anal. Chim. Acta 2008, 623, 59–65. [Google Scholar]
- Buttigieg, G.A.; Knight, A.K.; Denson, S.; Pommier, C.; Denton, M.B. Characterization of the explosive triacetone triperoxide and detection by ion mobility spectrometry. Forensic Sci. Intern. 2003, 135, 53–59. [Google Scholar] [CrossRef]
- Oxley, J.C.; Smith, J.L.; Kirschenbaum, L.J.; Marimganti, S.; Vadlamannati, S. Detection of explosives in hair using ion mobility spectrometry. J. Forensic Sci. 2008, 53, 690–693. [Google Scholar] [CrossRef]
- Germain, M.E.; Knapp, M.J. Turn-on fluorescence detection of H2O2 and TATP. Inorg. Chem. 2008, 47, 9748–9750. [Google Scholar] [CrossRef]
- Lin, H.W.; Suslick, K.S. A colorimetric sensor array for detection of triacetone triperoxide vapor. J. Am. Chem. Soc. 2010, 132, 15519–15521. [Google Scholar] [CrossRef]
- Schulte-Ladbeck, R.; Karst, U. Liquid chromatography—Post-column photochemical conversion and electrochemical detection for determination of peroxide-based explosives. Chromatographia 2003, 57, S61–S65. [Google Scholar] [CrossRef]
- Schulte-Ladbeck, R.; Karst, U. Determination of triacetonetriperoxide in ambient air. Anal. Chim. Acta 2003, 482, 183–188. [Google Scholar] [CrossRef]
- Girotti, S.; Ferri, E.; Maiolini, E.; Bolelli, L.; D’Elia, M.; Coppe, D.; Romolo, F.S. A quantitative chemiluminescent assay for analysis of peroxide-based explosives. Anal. Bioanal. Chem. 2011, 400, 313–320. [Google Scholar] [CrossRef]
- Malashikhin, S.; Finney, N.S. Fluorescent signaling based on sulfoxide profluorophores: Application to the visual detection of the explosive TATP. J. Am. Chem. Soc. 2008, 130, 12846–12447. [Google Scholar] [CrossRef]
- Schulte-Ladbeck, R.; Kolla, P.; Karst, U. A field test for the detection of peroxide-based explosives. Analyst 2002, 127, 1152–1154. [Google Scholar] [CrossRef]
- Widmer, L.; Watson, S.; Schlatter, K.; Crowson, A. Development of an LC/MS method for the trace analysis of triacetone triperoxide (TATP). Analyst 2002, 127, 1627–1632. [Google Scholar] [CrossRef]
- Sigman, M.E.; Clark, C.D.; Caiano, T.; Mullen, R. Analysis of triacetone triperoxide (TATP) and TATP synthetic intermediates by electrospray ionization mass spectrometry. Rapid Comm. Mass Spectr. 2008, 22, 84–90. [Google Scholar] [CrossRef]
- Cotte-Rodriguez, I.; Chen, H.; Cooks, R.G. Rapid trace detection of triacetone triperoxide (TATP) by complexation reactions during desorption electrospray ionization. Chem. Commun. 2006, 9, 953–955. [Google Scholar] [CrossRef]
- Cotte-Rodriguez, I.; Hernandez-Soto, H.; Chen, H.; Cooks, R.G. In situ trace detection of peroxide explosives by desorption electrospray ionization and desorption atmospheric pressure chemical ionization. Anal. Chem. 2008, 80, 1512–1519. [Google Scholar] [CrossRef]
- Benson, S.J.; Lennard, C.J.; Maynard, P.; Hill, D.M.; Andrew, A.S.; Roux, C. Forensic analysis of explosives using isotope ratio mass spectrometry (IRMS)—Preliminary study on TATP and PETN. Sci. Justice 2009, 49, 81–86. [Google Scholar] [CrossRef]
- Schulte-Ladbeck, R.; Edelmann, A.; Quintas, G.; Lendl, B.; Karst, U. Determination of peroxide-based explosives using liquid chromatography with on-line infrared detection. Anal. Chem. 2006, 78, 8150–8155. [Google Scholar] [CrossRef]
- Jensen, L.; Mortensen, P.M.; Trane, R.; Harris, P.; Berg, R.W. Reaction kinetics of acetone peroxide formation and structure investigations using raman spectroscopy and x-ray diffraction. Appl. Spectr. 2009, 63, 92–97. [Google Scholar]
- Willer, U.; Schade, W. Photonic sensor devices for explosive detection. Anal. Bioanal. Chem. 2009, 395, 275–282. [Google Scholar] [CrossRef]
- Burks, R.M.; Hage, D.S. Current trends in the detection of peroxide-based explosives. Anal. Bioanal. Chem. 2009, 395, 301–313. [Google Scholar] [CrossRef]
- Bromage, E.S.; Lackie, T.; Unger, M.A.; Ye, J.; Kaattari, S.L. The development of a real-time biosensor for the detection of trace levels of trinitrotoluene (TNT) in aquatic environments. Biosens. Bioelectron. 2007, 22, 2532–2538. [Google Scholar] [CrossRef]
- Conroy, P.J.; Hearty, S.; Leonard, P.; O’Kennedy, R.J. Antibody production, design and use for biosensor-based applications. Semin. Cell Develop. Biol. 2009, 20, 10–26. [Google Scholar] [CrossRef]
- Holt, D.B.; Gauger, P.R.; Kusterbeck, A.W.; Ligler, F.S. Fabrication of a capillary immunosensor in polymethyl methacrylate. Biosens. Bioelectron. 2002, 17, 95–103. [Google Scholar] [CrossRef]
- Whelan, J.P.; Kusterbeck, A.W.; Wemhoff, G.A.; Bredehorst, R.; Ligler, F.S. Continuous-flow immunosensor for detection of explosives. Anal. Chem. 1993, 65, 3561–3565. [Google Scholar] [CrossRef]
- Goodrow, M.H.; Hammock, B.D. Hapten design for compound-selective antibodies: ELISAS for environmentally deleterious small molecules. Anal. Chim. Acta 1998, 376, 83–91. [Google Scholar] [CrossRef]
- Marco, M.P.; Gee, S.; Hammock, B.D. Immunochemical techniques for environmental-analysis II. Antibody-production and immunoassay development. Trends Anal. Chem. 1995, 14, 415–425. [Google Scholar]
- Szurdoki, F.; Bekheit, H.K.M.; Marco, M.P.; Goodrow, M.H.; Hammock, B.D. Important Factors in Hapten Design and Enzyme-Linked Immunosorbent Assay Development. In New Frontiers in Agrochemical Immunoassay; Kurtz, D.A., Skerritt, J.H., Stanker, L., Eds.; AOAC International: Arlington, VA, USA, 1995; pp. 39–62. [Google Scholar]
- Winklmair, M.; Weller, M.G.; Mangler, J.; Schlosshauer, B.; Niessner, R. Development of a highly sensitive enzyme-immunoassay for the determination of triazine herbicides. Fresenius J. Anal. Chem. 1997, 358, 614–622. [Google Scholar] [CrossRef]
- Abdulhalim, I.; Zourob, M.; Lakhtakia, A. Surface plasmon resonance for biosensing: A mini-review. Electromagnetics 2008, 28, 214–242. [Google Scholar] [CrossRef]
- Anderson, G.P.; Goldman, E.R. TNT detection using llama antibodies and a two-step competitive fluid array immunoassay. J. Immunol. Meth. 2008, 339, 47–54. [Google Scholar] [CrossRef]
- Kawaguchi, T.; Shankaran, D.R.; Kim, S.J.; Gobi, K.V.; Matsumoto, K.; Toko, K.; Miura, N. Fabrication of a novel immunosensor using functionalized self-assembled monolayer for trace level detection of TNT by surface plasmon resonance. Talanta 2007, 72, 554–560. [Google Scholar] [CrossRef]
- Kawaguchi, T.; Shankaran, D.R.; Kim, S.J.; Matsumoto, K.; Toko, K.; Miura, N. Surface plasmon resonance immunosensor using Au nanoparticle for detection of TNT. Sens. Actuat. B 2008, 133, 467–472. [Google Scholar] [CrossRef]
- Shankaran, D.R.; Matsumoto, K.; Toko, K.; Miura, N. Development and comparison of two immunoassays for the detection of 2,4,6-trinitrotoluene (TNT) based on surface plasmon resonance. Sens. Actuators B 2006, 114, 71–79. [Google Scholar] [CrossRef]
- Charles, P.T.; Adams, A.A.; Howell, P.B.; Trammell, S.A.; Deschamps, J.R.; Kusterbeck, A.W. Fluorescence-based sensing of 2,4,6-Trinitrotoluene (TNT) using a multi-channeled poly(methyl methacrylate) (PMMA) microimmunosensor. Sensors 2010, 10, 876–889. [Google Scholar] [CrossRef]
- Girotti, S.; Eremin, S.; Montoya, A.; Moreno, M.J.; Caputo, P.; D’Elia, M.; Ripani, L.; Romolo, F.S.; Maiolini, E. Development of a chemiluminescent ELISA and a colloidal gold-based LFIA for TNT detection. Anal. Bioanal. Chem. 2010, 396, 687–695. [Google Scholar] [CrossRef]
- Onodera, T.; Mizuta, Y.; Horikawa, K.; Singh, P.; Matsumoto, K.; Miura, N.; Toko, K. Displacement immunosensor based on surface plasmon resonance for rapid and highly sensitive detection of 2,4,6-trinitrotoluene. Sens. Mater. 2011, 23, 39–52. [Google Scholar]
- Smith, R.G.; D’Souza, N.; Nicklin, S. A review of biosensors and biologically-inspired systems for explosives detection. Analyst 2008, 133, 571–584. [Google Scholar] [CrossRef]
- Yinon, J. Field detection and monitoring of explosives. Trends Anal. Chem. 2002, 21, 292–301. [Google Scholar] [CrossRef]
- Singh, S. Sensors—An effective approach for the detection of explosives. J. Hazard. Mater. 2007, 144, 15–28. [Google Scholar] [CrossRef]
- Mitchell, J. Small molecule immunosensing using surface plasmon resonance. Sensors 2010, 10, 7323–7346. [Google Scholar] [CrossRef]
- Bowen, J.; Noe, L.J.; Sullivan, B.P.; Morris, K.; Martin, V.; Donnelly, G. Gas-phase detection of trinitrotoluene utilizing a solid-phase antibody immobilized on a gold film by means of surface plasmon resonance spectroscopy. Appl. Spectr. 2003, 57, 906–914. [Google Scholar] [CrossRef]
- Bowen, J.M.; Noe, L.J.; Sullivan, P. Immunochemical Detection of an Explosive Substance in the Gas Phase through Surface Plasmon Resonance Spectroscopy. U.S. Patent 6,573,107, 3 June 2003. [Google Scholar]
- Hunt, W.D.; Lee, S.H.; Stubbs, D.D.; Edmonson, P.J. Clues from digital radio regarding biomolecular recognition. IEEE Trans. Biomed. Circ. Syst. 2007, 1, 50–55. [Google Scholar] [CrossRef]
- Weller, M.G.; Schuetz, A.J.; Winklmair, M.; Niessner, R. Highly parallel affinity sensor for the detection of environmental contaminants in water. Anal. Chim. Acta 1999, 393, 29–41. [Google Scholar] [CrossRef]
- Long, F.; He, M.A.; Zhu, A.N.; Song, B.D.; Sheng, J.W.; Shi, H.C. Compact quantitative optic fiber-based immunoarray biosensor for rapid detection of small analytes. Biosens. Bioelectron. 2010, 26, 16–22. [Google Scholar] [CrossRef]
- Bunte, G.; Hurttlen, J.; Pontius, H.; Hartlieb, K.; Krause, H. Gas phase detection of explosives such as 2,4,6-trinitrotoluene by molecularly imprinted polymers. Anal. Chim. Acta 2007, 591, 49–56. [Google Scholar] [CrossRef]
- Walker, N.R.; Linman, M.J.; Timmers, M.M.; Dean, S.L.; Burkett, C.M.; Lloyd, J.A.; Keelor, J.D.; Baughman, B.M.; Edmiston, P.L. Selective detection of gas-phase TNT by integrated optical waveguide spectrometry using molecularly imprinted sol-gel sensing films. Anal. Chim. Acta 2007, 593, 82–91. [Google Scholar] [CrossRef]
- Ehrentreich-Förster, E.; Orgel, D.; Krause-Griep, A.; Cech, B.; Erdmann, V.A.; Bier, F.; Scheller, F.W.; Rimmele, M. Biosensor-based on-site explosives detection using aptamers as recognition elements. Anal. Bioanal. Chem. 2008, 391, 1793–1800. [Google Scholar] [CrossRef]
- Fodey, T.; Leonard, P.; O’Mahony, J.; O’Kennedy, R.; Danaher, M. Developments in the production of biological and synthetic binders for immunoassay and sensor-based detection of small molecules. Trends Anal. Chem. 2011, 30, 254–269. [Google Scholar] [CrossRef]
- Röck, F.; Barsan, N.; Weimar, U. Electronic nose: Current status and future trends. Chem. Rev. 2008, 108, 705–725. [Google Scholar] [CrossRef]
- Hirst, E.R.; Yuan, Y.J.; Xu, W.L.; Bronlund, J.E. Bond-rupture immunosensors—A review. Biosens. Bioelectron. 2008, 23, 1759–1768. [Google Scholar] [CrossRef]
- Heiss, C.; Weller, M.G.; Niessner, R. Dip-and-read test strips for the determination of trinitrotoluene (TNT) in drinking water. Anal. Chim. Acta 1999, 396, 309–316. [Google Scholar] [CrossRef]
- Gonzalez-Martinez, M.A.; Puchades, R.; Maquieira, A. Optical immunosensors for environmental monitoring: How far have we come? Anal. Bioanal. Chem. 2007, 387, 205–218. [Google Scholar] [CrossRef]
- Van Dorst, B.; Mehta, J.; Bekaert, K.; Rouah-Martin, E.; De Coen, W.; Dubruel, P.; Blust, R.; Robbens, J. Recent advances in recognition elements of food and environmental biosensors: A review. Biosens. Bioelectron. 2010, 26, 1178–1194. [Google Scholar] [CrossRef]
- Walter, M.A.; Pfeifer, D.; Kraus, W.; Emmerling, F.; Schneider, R.J.; Panne, U.; Weller, M.G. Triacetone triperoxide (TATP): Hapten design and development of antibodies. Langmuir 2010, 26, 15418–15423. [Google Scholar] [CrossRef]
- Tatake, J.G.; Knapp, M.M.; Ressler, C. Synthesis and characterization of protein and polylysine conjugates of sulfamethoxazole and sulfanilic acid for investigation of sulfonamide drug allergy. Bioconjug. Chem. 1991, 2, 124–132. [Google Scholar] [CrossRef]
- Weller, M.G.; Weil, L.; Niessner, R. Determination of triazine herbicides by ELISA—optimization of enzyme tracer synthesis. Fresenius J. Anal. Chem. 1992, 343, 51–52. [Google Scholar] [CrossRef]
- Bahlmann, A.; Weller, M.G.; Panne, U.; Schneider, R.J. Monitoring carbamazepine in surface and wastewaters by an immunoassay based on a monoclonal antibody. Anal. Bioanal. Chem. 2009, 395, 1809–1820. [Google Scholar] [CrossRef]
- Dudley, R.A.; Edwards, P.; Ekins, R.P.; Finney, D.J.; Mckenzie, I.G.M.; Raab, G.M.; Rodbard, D.; Rodgers, R.P.C. Guidelines for immunoassay data-processing. Clin. Chem. 1985, 31, 1264–1271. [Google Scholar]
- Carvalho, J.J.; Weller, M.G.; Panne, U.; Schneider, R.J. A highly sensitive caffeine immunoassay based on a monoclonal antibody. Anal. Bioanal. Chem. 2010, 396, 2617–2628. [Google Scholar] [CrossRef]
- Abraham, G.E. Solid-phase radioimmunoassay of estradiol-17beta. J. Clin. Endocrin. Metabol. 1969, 29, 866–870. [Google Scholar] [CrossRef]
- Ekins, R. The “Precision Profile”: Its use in RIA assessment and design. Ligand Q 1981, 4, 33–44. [Google Scholar]
- Reid, V.; Doherty, J.; McIntosh, G.; Cowell, S.; Lee, M.; Rees, M. The first quantitative comparison of immunohistochemical rabbit and mouse monoclonal antibody affinities using Biacore analysis. J. Histotechnol. 2007, 30, 177–182. [Google Scholar] [CrossRef]
- Rocha, R.; Nunes, C.; Rocha, G.; Oliveira, F.; Sanches, F.; Gobbi, H. Rabbit monoclonal antibodies show higher sensitivity than mouse monoclonals for estrogen and progesterone receptor evaluation in breast cancer by immunohistochemistry. Pathol. Res. Pract. 2008, 204, 655–662. [Google Scholar] [CrossRef]
- Rossi, S.; Laurino, L.; Furlanetto, A.; Chinellato, S.; Orvieto, E.; Canal, F.; Facchetti, F.; Tos, A.P.D. A comparative study between a novel category of immunoreagents and the corresponding mouse monoclonal antibodies. Am. J. Clin. Pathol. 2005, 124, 295–302. [Google Scholar] [CrossRef]
- Saito, M.; Sakurai, S.; Motegi, A.; Saito, K.; Sano, T.; Nakajima, T. Comparative study using rabbit-derived polyclonal, mouse-derived monoclonal, and rabbit-derived monoclonal antibodies for KIT immunostaining in GIST and other tumors. Pathol. Intern. 2007, 57, 200–204. [Google Scholar] [CrossRef]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Walter, M.A.; Panne, U.; Weller, M.G. A Novel Immunoreagent for the Specific and Sensitive Detection of the Explosive Triacetone Triperoxide (TATP). Biosensors 2011, 1, 93-106. https://doi.org/10.3390/bios1030093
Walter MA, Panne U, Weller MG. A Novel Immunoreagent for the Specific and Sensitive Detection of the Explosive Triacetone Triperoxide (TATP). Biosensors. 2011; 1(3):93-106. https://doi.org/10.3390/bios1030093
Chicago/Turabian StyleWalter, Maria Astrid, Ulrich Panne, and Michael G. Weller. 2011. "A Novel Immunoreagent for the Specific and Sensitive Detection of the Explosive Triacetone Triperoxide (TATP)" Biosensors 1, no. 3: 93-106. https://doi.org/10.3390/bios1030093
APA StyleWalter, M. A., Panne, U., & Weller, M. G. (2011). A Novel Immunoreagent for the Specific and Sensitive Detection of the Explosive Triacetone Triperoxide (TATP). Biosensors, 1(3), 93-106. https://doi.org/10.3390/bios1030093