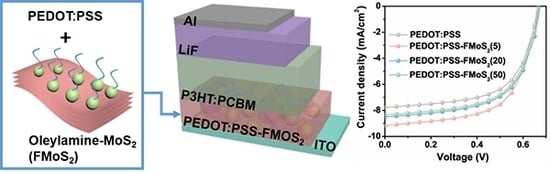
Solution-Processed PEDOT:PSS/MoS2 Nanocomposites as Efficient Hole-Transporting Layers for Organic Solar Cells
Abstract
1. Introduction
2. Experimental
2.1. Materials and Methods
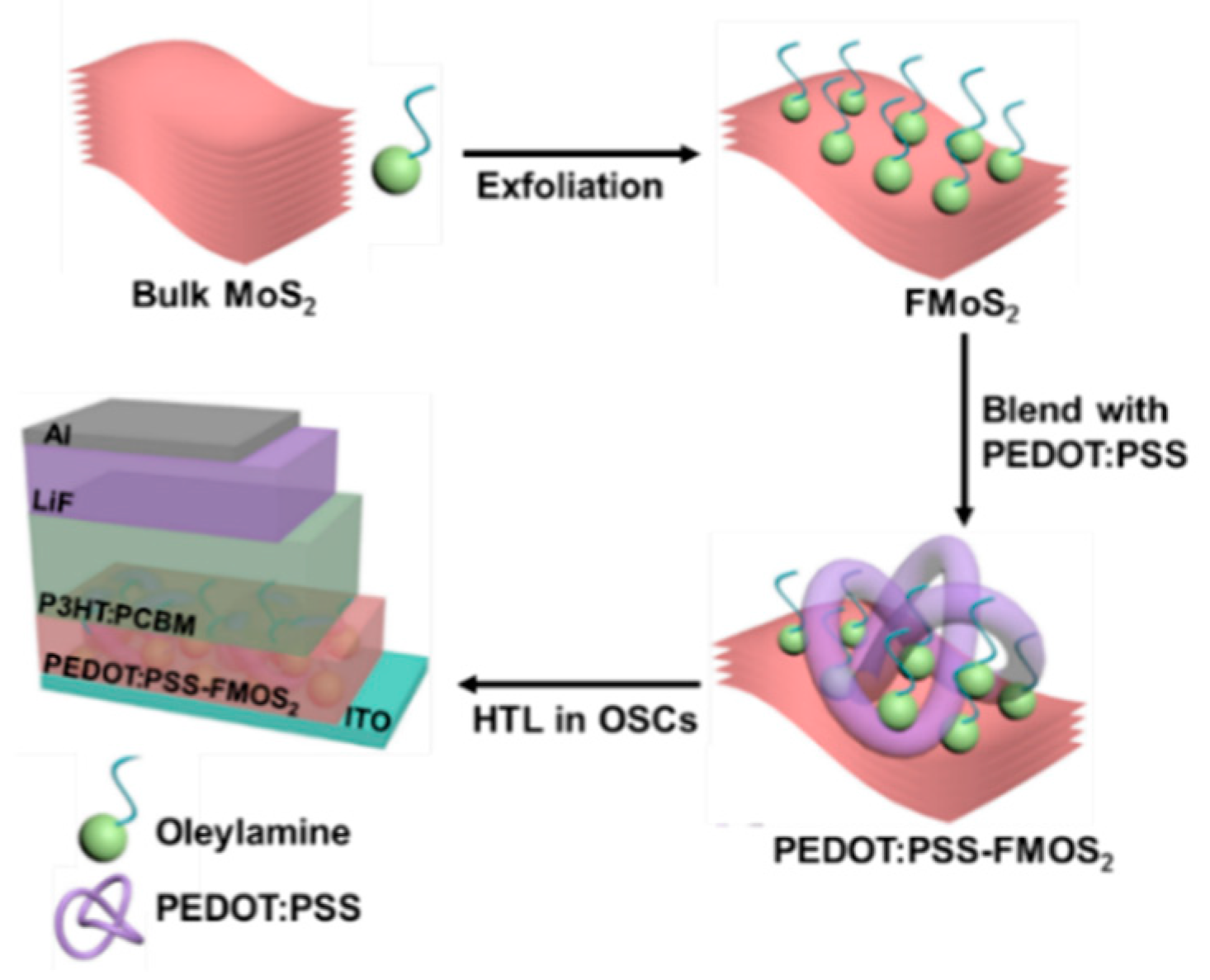
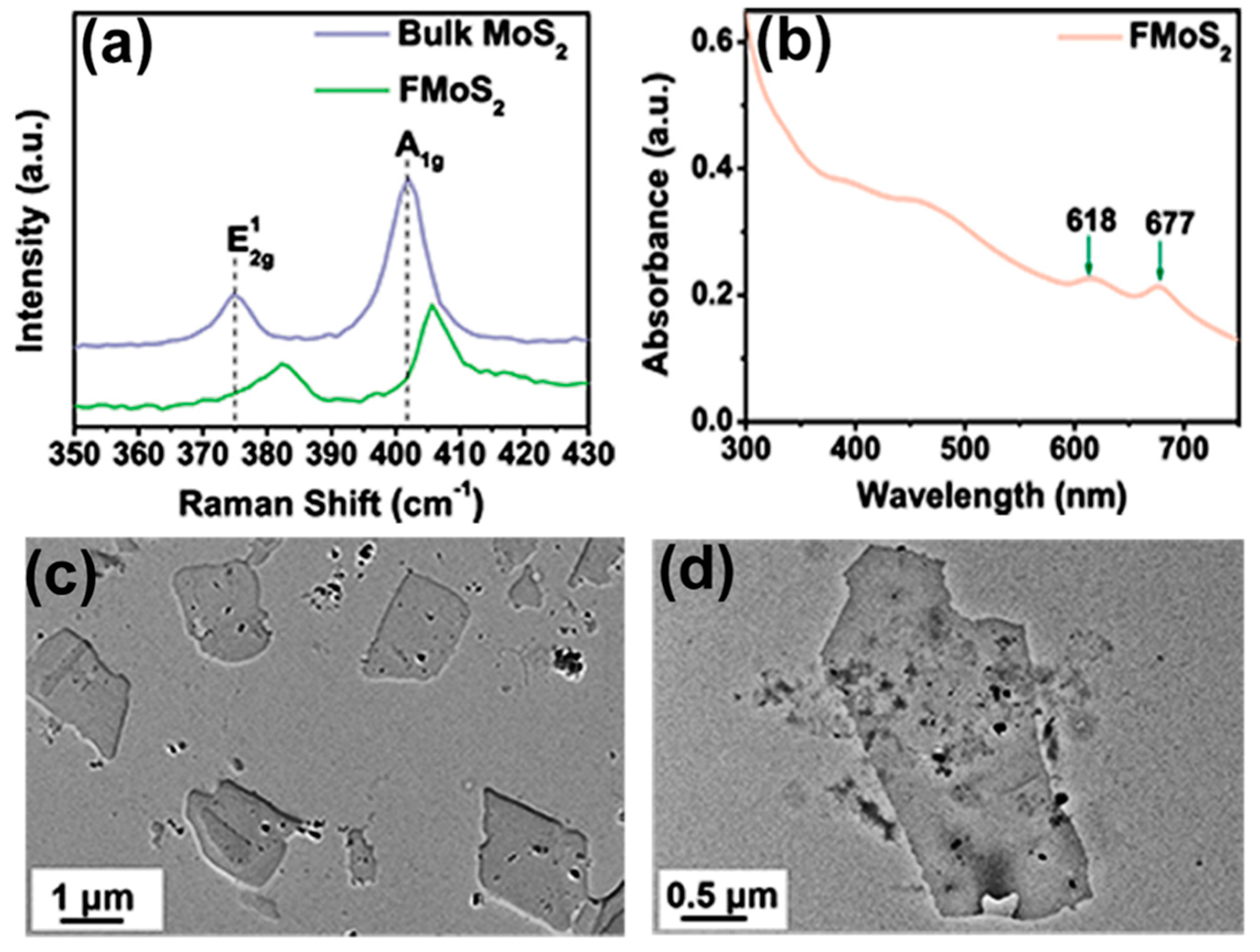
2.2. Synthesis of FMoS2 Nanosheets and PEDOT:PSS/FMoS2 Hybrids
2.3. Fabrication of OSCs
2.4. Characterization
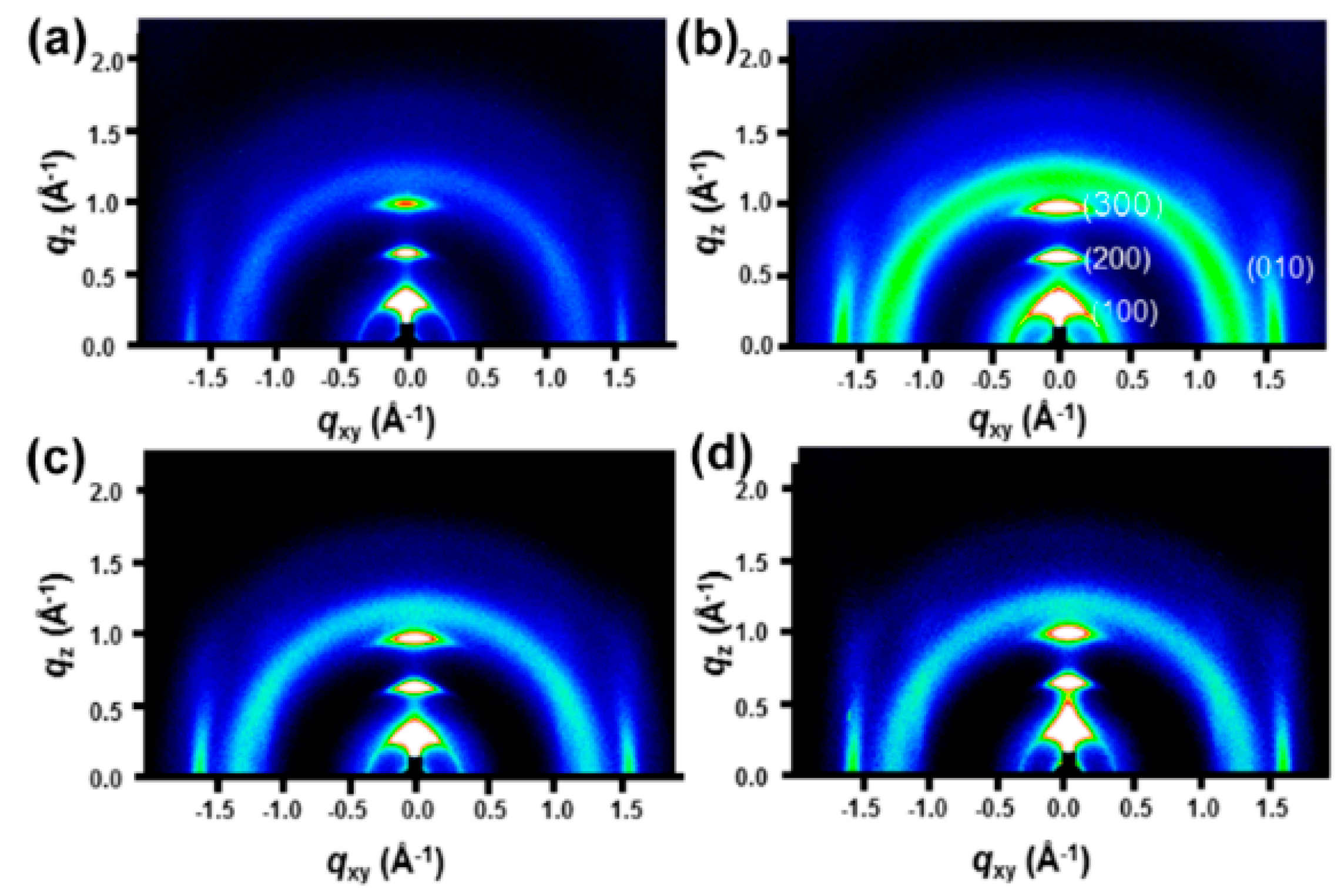
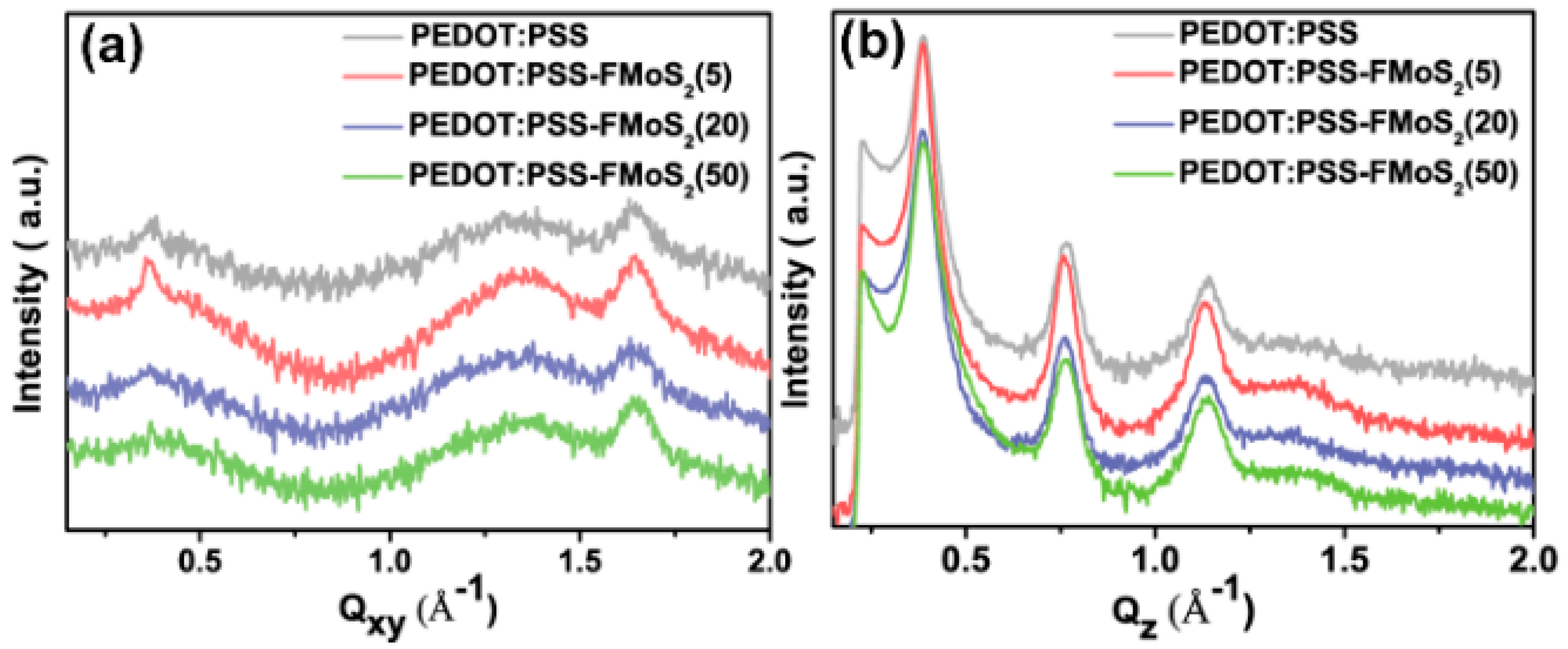
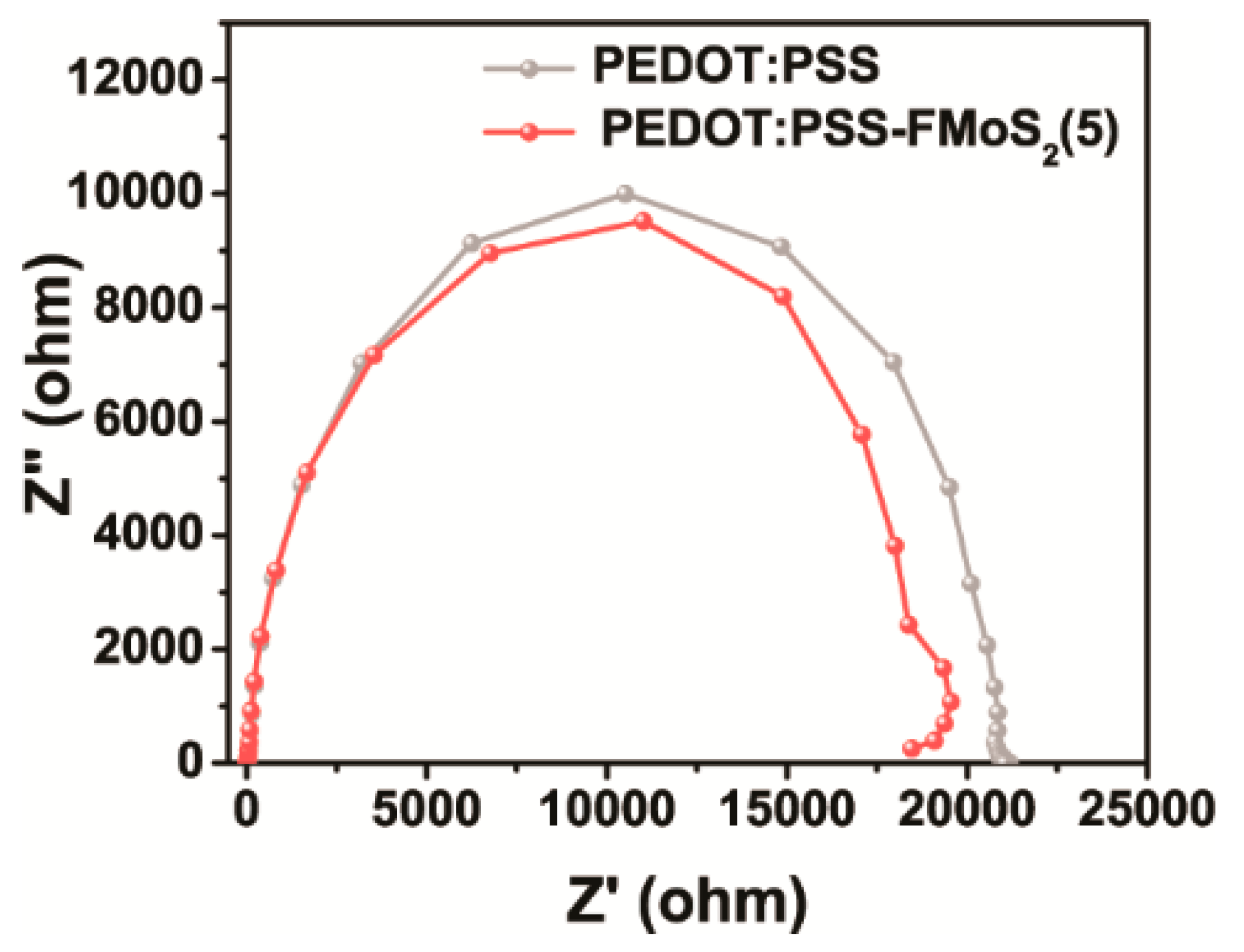
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Jang, Y.H.; Jang, Y.J.; Kim, S.; Quan, L.N.; Chung, K.; Kim, D.H. Plasmonic solar cells: From rational design to mechanism overview. Chem. Rev. 2016, 116, 14982–15034. [Google Scholar] [CrossRef] [PubMed]
- Oh, Y.; Lim, J.W.; Kim, J.G.; Wang, H.; Kang, B.H.; Park, Y.W.; Kim, H.; Jang, Y.J.; Kim, J.; Kim, D.H.; et al. Plasmonic periodic nanodot arrays via laser interference lithography for organic photovoltaic cells with ˃10% efficiency. ACS Nano 2016, 10, 10143–10151. [Google Scholar] [CrossRef] [PubMed]
- Xie, L.; Lee, J.S.; Jang, Y.; Ahn, H.; Kim, Y.H.; Kim, K. Organic photovoltaics utilizing a polymer nanofiber/fullerene interdigitated bilayer prepared by sequential solution deposition. J. Phys. Chem. C 2016, 120, 12933–12940. [Google Scholar] [CrossRef]
- Zhu, M.; Kim, H.; Jang, Y.J.; Park, S.; Ryu, D.Y.; Kim, K.; Tang, P.; Qiu, F.; Kim, D.H.; Peng, J. Toward high efficiency organic photovoltaic devices with enhanced thermal stability utilizing P3HT-b-P3PHT block copolymer additives. J. Mater. Chem. A 2016, 4, 18432–18443. [Google Scholar] [CrossRef]
- Kim, Y.; Son, J.; Shafian, S.; Kim, K.; Hyun, J.K. Semitransparent blue, green, and red organic solar cells Using Color Filtering Electrodes. Adv. Opt. Mater. 2018, 6, 1800051. [Google Scholar] [CrossRef]
- Xie, L.; Yoon, S.; Cho, Y.J.; Kim, K. Effective protection of sequential solution-processed polymer/fullerene bilayer solar cell against charge recombination and degradation. Org. Electron. 2015, 25, 212–218. [Google Scholar] [CrossRef]
- Zhao, W.; Li, S.; Yao, H.; Zhang, S.; Zhang, Y.; Yang, B.; Hou, J. Molecular optimization enables over 13% efficiency in organic solar cells. J. Am. Chem. Soc. 2017, 139, 7148–7151. [Google Scholar] [CrossRef] [PubMed]
- Ahn, S.; Jeong, S.H.; Han, T.H.; Lee, T.W. Conducting polymers as anode buffer materials in organic and perovskite optoelectronics. Adv. Opt. Mater. 2017, 5, 1600512. [Google Scholar] [CrossRef]
- Zhang, S.; Yu, Z.; Li, P.; Li, B.; Isikgor, F.H.; Du, D.; Sun, K.; Xia, Y.; Ouyang, J. Poly(3,4-ethylenedioxythiophene): Polystyrene sulfonate films with low conductivity and low Acidity through a treatment of their solutions with probe ultrasonication and their application as hole transport layer in polymer solar cells and perovskite solar cells. Org. Electron. 2016, 32, 149–156. [Google Scholar]
- Lee, J.J.; Lee, S.H.; Kim, F.S.; Choi, H.H.; Kim, J.H. Simultaneous enhancement of the efficiency and stability of organic solar cells using PEDOT: PSS grafted with a PEGME buffer layer. Org. Electron. 2015, 26, 191–199. [Google Scholar] [CrossRef]
- Zhou, H.; Zhang, Y.; Mai, C.K.; Seifter, J.; Nguyen, T.Q.; Bazan, G.C.; Heeger, A.J. Solution-processed pH-neutral conjugated polyelectrolyte improves interfacial contact in organic solar cells. ACS Nano 2015, 9, 371–377. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Zhang, Y.; Mai, C.K.; Collins, S.D.; Nguyen, T.Q.; Bazan, G.C.; Heeger, A.J. Conductive conjugated polyelectrolyte as hole-transporting layer for organic bulk heterojunction solar cells. Adv. Mater. 2014, 26, 780–785. [Google Scholar] [CrossRef] [PubMed]
- Jasieniak, J.J.; Seifter, J.; Jo, J.; Mates, T.; Heeger, A.J. A solution-processed MoOx anode interlayer for use within organic photovoltaic devices. Adv. Funct. Mater. 2012, 22, 2594–2605. [Google Scholar] [CrossRef]
- Xie, F.; Choy, W.C.H.; Wang, C.; Li, X.; Zhang, S.; Hou, J. Low-temperature solution-processed hydrogen molybdenum and vanadium bronzes for an efficient hole-transport layer in organic electronics. Adv. Mater. 2013, 25, 2051–2055. [Google Scholar] [CrossRef] [PubMed]
- Bao, X.; Zhu, Q.; Wang, T.; Guo, J.; Yang, C.; Yu, D.; Wang, N.; Chen, W.; Yang, R. Simple O2 plasma-processed V2O5 as an anode buffer layer for high performance polymer solar cells. ACS Appl. Mater. Interfaces 2015, 7, 7613–7618. [Google Scholar] [CrossRef] [PubMed]
- Bai, S.; Jin, Y.; Dai, X.; Liang, X.; Ye, Z.; Li, M.; Cheng, J.; Xiao, X.; Wu, Z.; Xia, Z.; et al. Low-temperature combustion-synthesized nickel oxide thin films as hole-transport interlayers for solution processed optoelectronic devices. Adv. Energy Mater. 2014, 4, 1301460. [Google Scholar] [CrossRef]
- Yin, Z.; Wei, J.; Zheng, Q. Interfacial materials for organic solar cells: Recent advances and perspectives. Adv. Sci. 2016, 3, 1500362. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Xue, Y.; Dai, L. Sulfated graphene oxide as a hole-extraction layer in high-performance polymer solar cells. J. Phys. Chem. Lett. 2012, 3, 1928–1933. [Google Scholar] [CrossRef]
- Li, S.S.; Tu, K.H.; Lin, C.C.; Chen, C.W.; Chhowalla, M. Solution-processable graphene oxide as an efficient hole transport layer in polymer solar cells. ACS Nano 2010, 4, 3169–3174. [Google Scholar] [CrossRef]
- Yun, J.M.; Yeo, J.S.; Kim, J.; Jeong, H.G.; Kim, D.Y.; Noh, Y.J.; Kim, S.S.; Ku, B.C.; Na, S.I. Solution-processable reduced graphene oxide as a novel alternative to PEDOT:PSS hole transport layers for highly efficient and stable polymer solar cells. Adv. Mater. 2011, 23, 4923–4928. [Google Scholar] [CrossRef]
- Niu, J.; Yang, D.; Ren, X.; Yang, Z.; Liu, Y.; Zhu, X.; Zhao, W.; Liu, S. Graphene-oxide doped PEDOT: PSS as a superior hole transport material for high-efficiency perovskite solar cell. Org. Electron. 2017, 48, 165–171. [Google Scholar] [CrossRef]
- Drakonakis, V.M.; Savva, A.; Kokonou, M.; Choulis, S.A. Investigating electrodes degradation in organic photovoltaics through reverse engineering under accelerated humidity lifetime conditions. Sol. Energy Mater. Sol. Cells 2014, 130, 544–550. [Google Scholar] [CrossRef]
- Kawano, K.; Pacios, R.; Poplavskyy, D.; Nelson, J.; Bradley, D.D.C.; Durrant, J.R. Degradation of organic solar cells due to air exposure. Sol. Energy Mater. Sol. Cells 2006, 90, 3520–3530. [Google Scholar] [CrossRef]
- Lim, F.J.; Ananthanarayanan, K.; Luther, J.; Ho, G.W. Influence of a novel fluorosurfactant modified PEDOT: PSS hole transport layer on the performance of inverted organic solar cells. J. Mater. Chem. 2012, 22, 25057–25064. [Google Scholar] [CrossRef]
- Arulkashmir, A.; Krishnamoorthy, K. Disassembly of micelles to impart donor and acceptor gradation to enhance organic solar cell efficiency. Chem. Commun. 2016, 52, 3486–3489. [Google Scholar] [CrossRef]
- Lim, D.C.; Kim, K.D.; Park, S.Y.; Hong, E.M.; Seo, H.O.; Lim, J.H.; Lee, K.W.; Jeong, Y.J.; Song, C.; Lee, E.; et al. Towards fabrication of high-performing organic photovoltaics: New donor polymer, atomic layer deposited thin buffer layer and plasmonic effects. Energy Environ. Sci. 2012, 5, 9803–9807. [Google Scholar] [CrossRef]
- Wang, K.; Yi, C.; Hu, X.; Liu, C.; Sun, Y.; Hou, J.; Li, Y.; Zheng, J.; Chuang, S.; Karim, A.; et al. Enhanced performance of polymer solar cells using PEDOT:PSS doped with Fe3O4 magnetic nanoparticles aligned by an external magnetostatic field as an anode buffer layer. ACS Appl. Mater. Interfaces 2014, 6, 13201–13208. [Google Scholar] [CrossRef]
- Yao, K.; Salvador, M.; Chueh, C.C.; Xin, X.K.; Xu, Y.X.; de Quilettes, D.W.; Hu, T.; Chen, Y.; Ginger, D.S.; Jen, A.K.Y. A general route to enhance polymer solar cell performance using plasmonic nanoprisms. Adv. Energy Mater. 2014, 4, 1400206. [Google Scholar] [CrossRef]
- Zhao, Z.; Wu, Q.; Xia, F.; Chen, X.; Liu, Y.; Zhang, W.; Zhu, J.; Dai, S.; Yang, S. Improving the conductivity of PEDOT: PSS hole transport layer in polymer solar cells via copper(II) bromide salt doping. ACS Appl. Mater. Interfaces 2015, 7, 1439–1448. [Google Scholar] [CrossRef]
- Kadem, B.; Cranton, W.; Hassan, A. Metal salt modified PEDOT: PSS as anode buffer layer and its effect on power conversion efficiency of organic solar cells. Org. Electron. 2015, 24, 73–79. [Google Scholar] [CrossRef]
- Kang, D.J.; Cho, H.H.; Lee, I.; Kim, K.H.; Kim, H.J.; Liao, K.; Kim, T.S.; Kim, B.J. Enhancing mechanical properties of highly efficient polymer solar cells using size-tuned polymer nanoparticles. ACS Appl. Mater. Interfaces 2015, 7, 2668–2676. [Google Scholar] [CrossRef]
- Hossain, J.; Liu, Q.; Miura, T.; Kasahara, K.; Harada, D.; Ishikawa, R.; Ueno, K.; Shirai, H. Nafion-modified PEDOT: PSS as a transparent hole-transporting layer for high-performance crystalline-Si/organic heterojunction solar cells with improved light soaking stability. ACS Appl. Mater. Interfaces 2016, 8, 31926–31934. [Google Scholar] [CrossRef]
- Hilal, M.; Han, J.I. Significant improvement in the photovoltaic stability of bulk heterojunction organic solar cells by the molecular level interaction of graphene oxide with a PEDOT: PSS composite hole transport layer. Sol. Energy 2018, 167, 24–34. [Google Scholar] [CrossRef]
- HDehsari, S.; Shalamzari, E.K.; Gavgani, J.N.; Taeomi, F.A.; Ghanbary, S. Efficient preparation of ultralarge graphene oxide using a PEDOT: PSS/GO composite layer as hole transport layer in polymer-based optoelectronic devices. RSC Adv. 2014, 4, 55067–55076. [Google Scholar] [CrossRef]
- Walker, B.; Choi, H.; Kim, J.Y. Interfacial engineering for highly efficient organic solar cells. Curr. Appl. Phys. 2017, 17, 370–391. [Google Scholar] [CrossRef]
- Pham, V.P.; Yeom, G.Y. Recent advances in doping of molybdenum disulfide: Industrial applications and future prospects. Adv. Mater. 2016, 28, 9024–9059. [Google Scholar] [CrossRef]
- Chhowalla, M.; Shin, H.S.; Eda, G.; Li, L.J.; Loh, K.P.; Zhang, H. The chemistry of two-dimensional layered transition metal dichalcogenide nanosheets. Nat. Chem. 2013, 5, 263–275. [Google Scholar] [CrossRef]
- Li, H.; Wu, J.; Yin, Z.; Zhang, H. Preparation and Applications of Mechanically Exfoliated Single-Layer and Multilayer MoS2 and WSe2 Nanosheets. Acc. Chem. Res. 2014, 47, 1067–1075. [Google Scholar] [CrossRef]
- Lee, Y.H.; Zhang, X.Q.; Zhang, W.; Chang, M.T.; Lin, C.T.; Chang, K.D.; Yu, Y.C.; Wang, J.T.W.; Chang, C.S.; Li, L.J.; et al. Synthesis of large-area MoS2 atomic layers with chemical vapor deposition. Adv. Mater. 2012, 24, 2320–2325. [Google Scholar] [CrossRef]
- Zhang, X.; Lai, Z.; Tan, C.; Zhang, H. Solution-processed two-Dimensional MoS2 nanosheets: Preparation, hybridization, and applications. Angew. Chem. Int. Ed. 2016, 55, 8816–8838. [Google Scholar] [CrossRef]
- Le, Q.V.; Nguyen, T.P.; Jang, H.W.; Kim, S.Y. The use of UV/ozone-treated MoS2 nanosheets for extended air stability in organic photovoltaic cells. Phys. Chem. Chem. Phys. 2014, 16, 13123–13128. [Google Scholar] [CrossRef]
- Yun, J.M.; Noh, Y.J.; Yeo, J.S.; Go, Y.J.; Na, S.I.; Jeong, H.G.; Kim, J.; Lee, S.; Kim, S.S.; Koo, H.Y.; et al. Efficient work-function engineering of solution-processed MoS2 thin-films for novel hole and electron transport layers leading to high-performance solar cells. J. Mater. Chem. C 2013, 1, 3777–3783. [Google Scholar] [CrossRef]
- Yang, X.; Fu, W.; Liu, W.; Hong, J.; Cai, Y.; Jin, C.; Xu, M.; Wang, H.; Yang, D.; Chen, H. Engineering crystalline structures of two-dimensional MoS2 sheets for high-performance organic solar cells. J. Mater. Chem. A 2014, 2, 7727–7733. [Google Scholar] [CrossRef]
- Xing, W.; Chen, Y.; Wu, X.; Xu, X.; Ye, P.; Zhu, T.; Guo, Q.; Yang, L.; Li, W.; Huang, H. PEDOT: PSS-assisted exfoliation and functionalization of 2D nanosheets for high-performance organic solar cells. Adv. Funct. Mater. 2017, 27, 1701622. [Google Scholar] [CrossRef]
- Ahmad, R.; Srivastava, R.; Yadav, S.; Chand, S.; Sapra, S. Functionalized 2D-MoS2-incorporated polymer ternary solar cells: Role of nanosheet-induced long-range ordering of polymer chains on charge transport. ACS Appl. Mater. Interfaces 2017, 9, 34111–34121. [Google Scholar] [CrossRef]
- Wu, J.B.; Lin, M.L.; Cong, X.; Liu, H.N.; Tan, P.H. Raman spectroscopy of graphene-based materials and its applications in related devices. Chem. Soc. Rev. 2018, 47, 1822–1873. [Google Scholar] [CrossRef]
- Cho, K.; Min, M.; Kim, T.Y.; Jeong, H.; Pak, J.; Kim, J.K.; Jang, J.; Yun, S.J.; Lee, Y.H.; Hong, W.K.; et al. Electrical and optical characterization of MoS2 with Sulfur vacancy passivation by treatment with alkanethiol molecules. ACS Nano 2015, 9, 8044–8053. [Google Scholar] [CrossRef]
- Yao, Y.; Tolentino, L.; Yang, Z.; Song, X.; Zhang, W.; Chen, Y.; Wong, C. High-concentration aqueous dispersions of MoS2. Adv. Funct. Mater. 2013, 23, 3577–3583. [Google Scholar] [CrossRef]
- Luo, S.; Qi, X.; Ren, L.; Hao, G.; Fan, Y.; Liu, Y.; Han, W.; Zang, C.; Li, J.; Zhong, J. Photoresponse properties of large-area MoS2 atomic layer synthesized by vapor phase deposition. J. Appl. Phys. 2014, 116, 164304. [Google Scholar] [CrossRef]
- Zhou, K.G.; Mao, N.N.; Wang, H.X.; Peng, Y.; Zhang, H.L. A Mixed-solvent strategy for efficient exfoliation of inorganic graphene analogues. Angew. Chem. Int. Ed. 2011, 50, 10839–10842. [Google Scholar] [CrossRef]
- Chen, X.; Berner, N.C.; Backes, C.; Duesberg, G.S.; McDonald, A.R. Functionalization of two-dimensional MoS2: On the reaction between MoS2 and organic thiols. Angew. Chem. Int. Ed. 2016, 55, 5803–5808. [Google Scholar] [CrossRef]
- Splendiani, A.; Sun, L.; Zhang, Y.; Li, T.; Kim, J.; Chim, C.Y.; Galli, G.; Wang, F. Emerging photoluminescence in monolayer MoS2. Nano Lett. 2010, 10, 1271–1275. [Google Scholar] [CrossRef]
- Eda, G.; Yamaguchi, H.; Voiry, D.; Fujita, T.; Chen, M.; Chhowalla, M. Photoluminescence from chemically exfoliated MoS2. Nano. Lett. 2011, 11, 5111–5116. [Google Scholar] [CrossRef]
- Qi, Y.; Wang, N.; Xu, Q.; Li, H.; Zhou, P.; Lu, X.; Zhao, G. A green route to fabricate MoS2 nanosheets in water-ethanol-CO2. Chem. Commun. 2015, 51, 6726–6729. [Google Scholar] [CrossRef]
- Kim, D.H.; Park, Y.D.; Jang, Y.; Yang, H.; Kim, Y.H.; Han, J.I.; Moon, D.G.; Park, S.; Chang, T.; Chang, C.; et al. Enhancement of field-effect mobility due to surface-mediated molecular ordering in regioregular polythiophene thin film transistors. Adv. Funct. Mater. 2005, 15, 77–82. [Google Scholar] [CrossRef]
- Kim, D.H.; Jang, Y.; Park, Y.D.; Cho, K. Surface-induced conformational changes in poly(3-hexylthiophene) monolayer films. Langmuir 2005, 21, 3203–3206. [Google Scholar] [CrossRef]
- Huang, J.S.; Goh, T.; Li, X.; Sfeir, M.Y.; Bielinski, E.A.; Tomasulo, S.; Lee, M.L.; Hazari, N.; Taylor, A.D. Polymer bulk heterojunction solar cells employing Forster resonance energy transfer. Nat. Photonics 2013, 7, 479–485. [Google Scholar] [CrossRef]
- Liu, Y.; Renna, L.A.; Page, Z.A.; Thompson, H.B.; Kim, P.Y.; Barnes, M.D.; Emrick, T.; Venkataramanan, D.; Russell, T.P. A polymer hole extraction layer for inverted perovskite solar cells from aqueous solutions. Adv. Energy Mater. 2016, 6, 1600664. [Google Scholar] [CrossRef]
- Choi, H.; Kim, H.B.; Ko, S.J.; Kim, J.Y.; Heeger, A.J. An organic surface modifier to produce a high work function transparent electrode for high performance polymer solar cells. Adv. Mater. 2015, 27, 892–896. [Google Scholar] [CrossRef]
- Noriega, R.; Rivnay, J.; Vandewal, K.; Koch, F.P.V.; Stingelin, N.; Smith, P.; Toney, M.F.; Salleo, A. A general relationship between disorder, aggregation and charge transport in conjugated polymers. Nat. Mater. 2013, 12, 1038–1044. [Google Scholar] [CrossRef]
- Lan, Y.K.; Huang, C. A theoretical study of the charge transfer behavior of the highly regioregular poly-3-hexylthiophene in the ordered state. J. Phys. Chem. B 2008, 112, 14857–14862. [Google Scholar] [CrossRef]
- Skrypnychuk, V.; Boulanger, N.; Yu, V.; Hilke, M.; Mannsfeld, S.C.B.; Toney, M.F.; Barbero, D.R. Enhanced vertical charge transport in a semiconducting P3HT thin Film on single layer grapheme. Adv. Funct. Mater. 2015, 25, 664–670. [Google Scholar] [CrossRef]
- Boulanger, N.; Yu, V.; Hilke, M.; Toney, M.F.; Barbero, D.R. In situ probing of the crystallization kinetics of rr-P3HT on single layer graphene as a function of temperature. Phys. Chem. Chem. Phys. 2017, 19, 8496–8503. [Google Scholar] [CrossRef]
- Lim, J.W.; Wang, H.; Choi, C.H.; Quan, L.N.; Chung, K.; Park, W.T.; Noh, Y.Y.; Kim, D.H. Polyethylenimine ethoxylated interlayer-mediated ZnO interfacial engineering for high-performance and low-temperature processed flexible perovskite solar cells: A simple and viable route for one-step processed CH3NH3PbI3. J. Power Sources 2019, 438, 226956. [Google Scholar] [CrossRef]
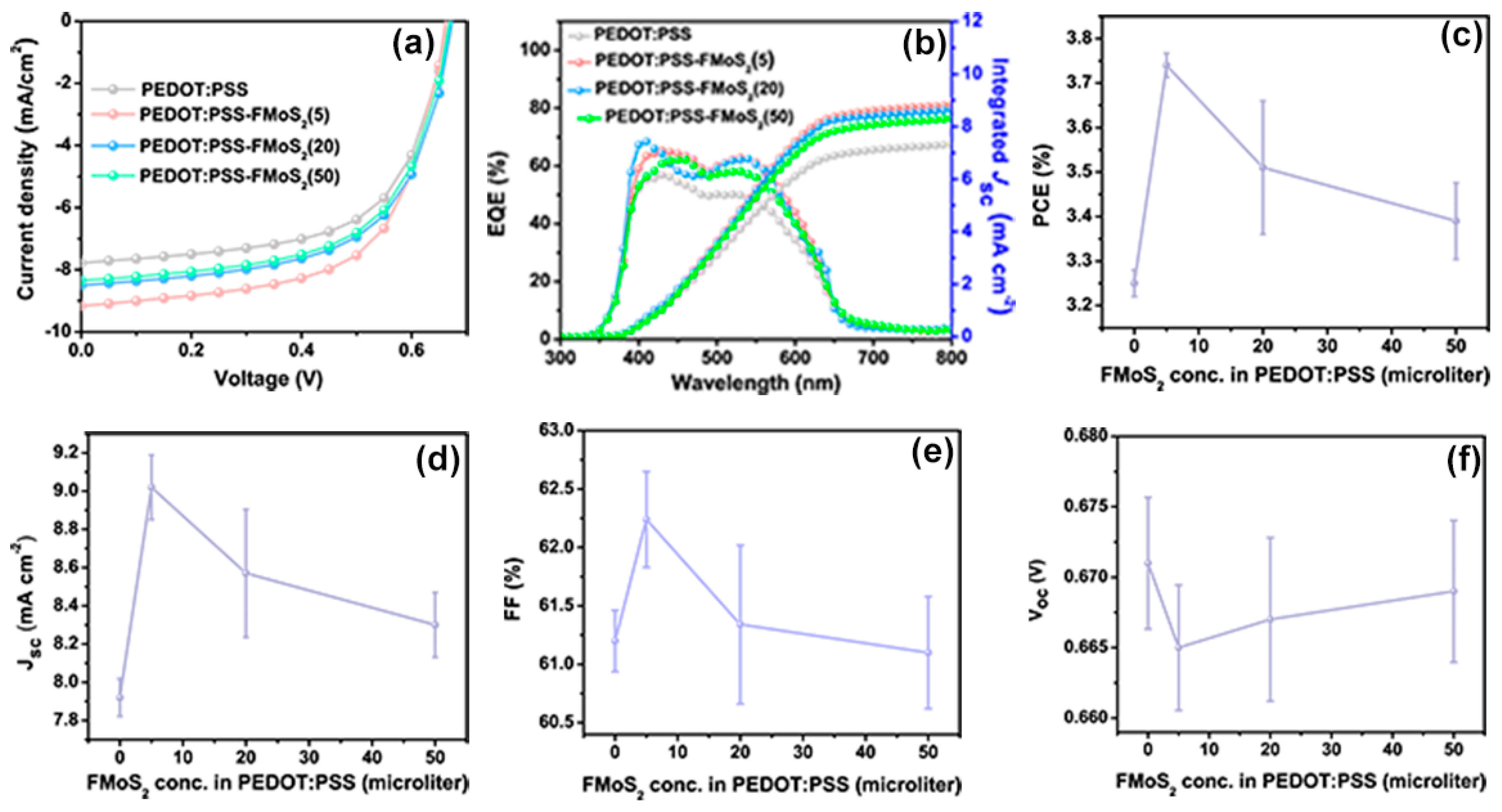
| FMoS2 Concentration (µL) in PEDOT:PSS | PCE (%) | Voc (V) | Jsc (mA cm−2) | FF (%) |
|---|---|---|---|---|
| 0 (Reference) | 3.25 ± 0.03 | 0.671 ± 0.004 | 7.92 ± 0.09 | 61.2 ± 0.26 |
| 5 | 3.74 ± 0.02 | 0.665 ± 0.004 | 9.02 ± 0.17 | 62.24 ± 0.41 |
| 20 | 3.51 ± 0.15 | 0.667 ± 0.006 | 8.57 ± 0.33 | 61.34 ± 0.68 |
| 50 | 3.39 ± 0.08 | 0.669 ± 0.005 | 8.30 ± 0.17 | 61.10 ± 0.48 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ramasamy, M.S.; Ryu, K.Y.; Lim, J.W.; Bibi, A.; Kwon, H.; Lee, J.-E.; Kim, D.H.; Kim, K. Solution-Processed PEDOT:PSS/MoS2 Nanocomposites as Efficient Hole-Transporting Layers for Organic Solar Cells. Nanomaterials 2019, 9, 1328. https://doi.org/10.3390/nano9091328
Ramasamy MS, Ryu KY, Lim JW, Bibi A, Kwon H, Lee J-E, Kim DH, Kim K. Solution-Processed PEDOT:PSS/MoS2 Nanocomposites as Efficient Hole-Transporting Layers for Organic Solar Cells. Nanomaterials. 2019; 9(9):1328. https://doi.org/10.3390/nano9091328
Chicago/Turabian StyleRamasamy, Madeshwaran Sekkarapatti, Ka Yeon Ryu, Ju Won Lim, Asia Bibi, Hannah Kwon, Ji-Eun Lee, Dong Ha Kim, and Kyungkon Kim. 2019. "Solution-Processed PEDOT:PSS/MoS2 Nanocomposites as Efficient Hole-Transporting Layers for Organic Solar Cells" Nanomaterials 9, no. 9: 1328. https://doi.org/10.3390/nano9091328
APA StyleRamasamy, M. S., Ryu, K. Y., Lim, J. W., Bibi, A., Kwon, H., Lee, J.-E., Kim, D. H., & Kim, K. (2019). Solution-Processed PEDOT:PSS/MoS2 Nanocomposites as Efficient Hole-Transporting Layers for Organic Solar Cells. Nanomaterials, 9(9), 1328. https://doi.org/10.3390/nano9091328