Modification of the Raman Spectra in Graphene-Based Nanofluids and Its Correlation with Thermal Properties
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussions
3.1. Thermal Conductivity and Viscosity Measurements
3.2. Raman Measurements
3.2.1. Raman of DMAc- and DMF-Based Nanofluids
Graphene-DMAc Nanofluids
Graphene-DMF Nanofluids
3.2.2. Raman in NMP-Based Nanofluids
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Shi, L.; Dames, C.; Lukes, J.R.; Reddy, P.; Duda, J.; Cahill, D.G.; Lee, J.; Marconnet, A.; Goodson, K.E.; Bahk, J.-H.; et al. Evaluating Broader Impacts of Nanoscale Thermal Transport Research. Nanoscale Microscale Thermophys. Eng. 2015, 19, 127–165. [Google Scholar] [CrossRef]
- International Roadmap for Devices and Systems (IRDS): Beyond CMOS. Available online: https://irds.ieee.org/images/files/pdf/2017/2017IRDS_BC.pdf (accessed on 26 May 2019).
- Ekpu, M.; Bhatti, R.; Ekere, N.; Mallik, S. Advanced thermal management materials for heat sinks used in microelectronics. In Proceedings of the 18th European Microelectronics & Packaging Conference, Brighton, UK, 12–15 September 2011; pp. 1–8. [Google Scholar]
- Ye, H.; Zhang, G. A review of passive thermal management of LED module. J. Semicond. 2011, 32, 14008. [Google Scholar] [CrossRef]
- Sohel Murshed, S.M.; Nieto de Castro, C.A. A critical review of traditional and emerging techniques and fluids for electronics cooling. Renew. Sustain. Energy Rev. 2017, 78, 821–833. [Google Scholar] [CrossRef]
- Bar-Cohen, A.; Arik, M.; Ohadi, M. Direct Liquid Cooling of High Flux Micro and Nano Electronic Components. Proc. IEEE 2006, 94, 1549–1570. [Google Scholar] [CrossRef]
- Siddique, A.R.M.; Muresan, H.; Majid, S.H.; Mahmud, S. An adjustable closed-loop liquid-based thermoelectric electronic cooling system for variable load thermal management. Therm. Sci. Eng. Prog. 2019, 10, 245–252. [Google Scholar] [CrossRef]
- Tasaka, M.; Shinohara, K.; Hayashi, C.; Kashima, S.; Koyama, K. Cooling performance of heat sinks with corrugated-fins. In Proceedings of the ITherm’98. Sixth Intersociety Conference on Thermal and Thermomechanical Phenomena in Electronic Systems (Cat. No.98CH36208), Seattle, WA, USA, 27–30 May 1998; pp. 104–111. [Google Scholar]
- Budelman, G.A. US-Patent: Heatsink with integrated blower for improved heat transfer. No. 6244331, 2001. [Google Scholar]
- Mahajan, R.; Chia-pin Chiu; Chrysler, G. Cooling a Microprocessor Chip. Proc. IEEE 2006, 94, 1476–1486. [Google Scholar] [CrossRef]
- Leng, C.; Wang, X.-D.; Wang, T.-H. An improved design of double-layered microchannel heat sink with truncated top channels. Appl. Therm. Eng. 2015, 79, 54–62. [Google Scholar] [CrossRef]
- Molina, J.; Prieto, R.; Narciso, J.; Louis, E. The effect of porosity on the thermal conductivity of Al–12wt.% Si/SiC composites. Scr. Mater. 2009, 60, 582–585. [Google Scholar] [CrossRef]
- Caccia, M.; Rodríguez, A.; Narciso, J. Diamond Surface Modification to Enhance Interfacial Thermal Conductivity in Al/Diamond Composites. JOM 2014, 66, 920–925. [Google Scholar] [CrossRef]
- Molina, J.-M.; Rodríguez-Guerrero, A.; Louis, E.; Rodríguez-Reinoso, F.; Narciso, J. Porosity Effect on Thermal Properties of Al-12 wt % Si/Graphite Composites. Materials (Basel) 2017, 10, 177. [Google Scholar] [CrossRef]
- Kheirabadi, A.C.; Groulx, D. Cooling of server electronics: A design review of existing technology. Appl. Therm. Eng. 2016, 105, 622–638. [Google Scholar] [CrossRef]
- Lv, L.C.; Li, J. Micro Flat Heat Pipes for Microelectronics Cooling: Review. Recent Patents Mech. Eng. 2013, 6, 169–184. [Google Scholar] [CrossRef]
- Garimella, S.V.; Persoons, T.; Weibel, J.A.; Gektin, V. Electronics Thermal Management in Information and Communications Technologies: Challenges and Future Directions. IEEE Trans. Compon. Packag. Manuf. Technol. 2017, 7, 1191–1205. [Google Scholar] [CrossRef]
- Patil, M.; Kim, S.; Seo, J.; Lee, M. Review of the Thermo-Physical Properties and Performance Characteristics of a Refrigeration System Using Refrigerant-Based Nanofluids. Energies 2015, 9, 22. [Google Scholar] [CrossRef]
- Mahian, O.; Kianifar, A.; Kalogirou, S.a.; Pop, I.; Wongwises, S. A review of the applications of nanofluids in solar energy. Int. J. Heat Mass Transf. 2013, 57, 582–594. [Google Scholar] [CrossRef]
- Safaei, M.; Ahmadi, G.; Goodarzi, M.; Safdari Shadloo, M.; Goshayeshi, H.; Dahari, M. Heat Transfer and Pressure Drop in Fully Developed Turbulent Flows of Graphene Nanoplatelets–Silver/Water Nanofluids. Fluids 2016, 1, 20. [Google Scholar] [CrossRef]
- Choi, S.U.S.; Eastman, J.A. Enhancing thermal conductivity of fluids with nanoparticles. ASME Int. Mech. Eng. Congr. Expo. 1995, 66, 99–105. [Google Scholar]
- Branson, B.T.; Beauchamp, P.S.; Beam, J.C.; Lukehart, C.M.; Davidson, J.L. Nanodiamond Nanofluids for Enhanced Thermal Conductivity. ACS Nano 2013, 7, 3183–3189. [Google Scholar] [CrossRef]
- Patil, M.; Seo, J.-H.; Kang, S.-J.; Lee, M.-Y. Review on Synthesis, Thermo-Physical Property, and Heat Transfer Mechanism of Nanofluids. Energies 2016, 9, 840. [Google Scholar] [CrossRef]
- Azmi, W.H.; Sharma, K.V.; Mamat, R.; Najafi, G.; Mohamad, M.S. The enhancement of effective thermal conductivity and effective dynamic viscosity of nanofluids – A review. Renew. Sustain. Energy Rev. 2016, 53, 1046–1058. [Google Scholar] [CrossRef]
- Bhanushali, S.; Jason, N.N.; Ghosh, P.; Ganesh, A.; Simon, G.P.; Cheng, W. Enhanced Thermal Conductivity of Copper Nanofluids: The Effect of Filler Geometry. ACS Appl. Mater. Interfaces 2017, 9, 18925–18935. [Google Scholar] [CrossRef] [PubMed]
- Sekrani, G.; Poncet, S. Ethylene- and Propylene-Glycol Based Nanofluids: A Litterature Review on Their Thermophysical Properties and Thermal Performances. Appl. Sci. 2018, 8, 2311. [Google Scholar] [CrossRef]
- Rodríguez-Laguna, M.R.; Castro-Alvarez, A.; Sledzinska, M.; Maire, J.; Costanzo, F.; Ensing, B.; Pruneda, M.; Ordejón, P.; Sotomayor Torres, C.M.; Gómez-Romero, P.; et al. Mechanisms behind the enhancement of thermal properties of graphene nanofluids. Nanoscale 2018, 10, 15402–15409. [Google Scholar] [CrossRef]
- Wei, C.; Nan, Z.; Wang, X.; Tan, Z. Investigation on Thermodynamic Properties of a Water-Based Hematite Nanofluid. J. Chem. Eng. Data 2010, 55, 2524–2528. [Google Scholar] [CrossRef]
- Shin, D.; Banerjee, D. Enhanced Specific Heat of Silica Nanofluid. J. Heat Transf. 2011, 133, 24501. [Google Scholar] [CrossRef]
- Tiznobaik, H.; Shin, D. Enhanced specific heat capacity of high-temperature molten salt-based nanofluids. Int. J. Heat Mass Transf. 2013, 57, 542–548. [Google Scholar] [CrossRef]
- Ho, M.X.; Pan, C. Optimal concentration of alumina nanoparticles in molten Hitec salt to maximize its specific heat capacity. Int. J. Heat Mass Transf. 2014, 70, 174–184. [Google Scholar] [CrossRef]
- Shahrul, I.M.; Mahbubul, I.M.; Khaleduzzaman, S.S.; Saidur, R.; Sabri, M.F.M. A comparative review on the specific heat of nanofluids for energy perspective. Renew. Sustain. Energy Rev. 2014, 38, 88–98. [Google Scholar] [CrossRef]
- Riazi, H.; Murphy, T.; Webber, G.B.; Atkin, R.; Tehrani, S.S.M.; Taylor, R.A. Specific heat control of nanofluids: A critical review. Int. J. Therm. Sci. 2016, 107, 25–38. [Google Scholar] [CrossRef]
- Hu, Y.; He, Y.; Zhang, Z.; Wen, D. Enhanced heat capacity of binary nitrate eutectic salt-silica nanofluid for solar energy storage. Sol. Energy Mater. Sol. Cells 2019, 192, 94–102. [Google Scholar] [CrossRef]
- Roberts, N.A.; Walker, D.G. Convective Performance of Nanofluids in Commercial Electronics Cooling Systems. Appl. Therm. Eng. 2010, 30, 2499–2504. [Google Scholar] [CrossRef]
- Escher, W.; Brunschwiler, T.; Shalkevich, N.; Shalkevich, A.; Burgi, T.; Michel, B.; Poulikakos, D. On the Cooling of Electronics With Nanofluids. J. Heat Transf. 2011, 133, 51401. [Google Scholar] [CrossRef]
- Rafati, M.; Hamidi, A.A.; Shariati Niaser, M. Application of nanofluids in computer cooling systems (heat transfer performance of nanofluids). Appl. Therm. Eng. 2012, 45–46, 9–14. [Google Scholar] [CrossRef]
- Ijam, A.; Saidur, R. Nanofluid as a coolant for electronic devices (cooling of electronic devices). Appl. Therm. Eng. 2012, 32, 76–82. [Google Scholar] [CrossRef]
- Nazari, M.; Karami, M.; Ashouri, M. Comparing the thermal performance of water, Ethylene Glycol, Alumina and CNT nanofluids in CPU cooling: Experimental study. Exp. Therm. Fluid Sci. 2014, 57, 371–377. [Google Scholar] [CrossRef]
- Liu, Z.-H.; Li, Y.-Y. A new frontier of nanofluid research – Application of nanofluids in heat pipes. Int. J. Heat Mass Transf. 2012, 55, 6786–6797. [Google Scholar] [CrossRef]
- Sureshkumar, R.; Mohideen, S.T.; Nethaji, N. Heat transfer characteristics of nanofluids in heat pipes: A review. Renew. Sustain. Energy Rev. 2013, 20, 397–410. [Google Scholar] [CrossRef]
- Alawi, O.A.; Sidik, N.A.C.; Mohammed, H.A.; Syahrullail, S. Fluid flow and heat transfer characteristics of nanofluids in heat pipes: A review. Int. Commun. Heat Mass Transf. 2014, 56, 50–62. [Google Scholar] [CrossRef]
- Das, S.; Giri, A.; Samanta, S.; Kanagaraj, S. Role of graphene nanofluids on heat transfer enhancement in thermosyphon. J. Sci. Adv. Mater. Devices 2019, 4, 163–169. [Google Scholar] [CrossRef]
- Poplaski, L.M.; Benn, S.P.; Faghri, A. Thermal performance of heat pipes using nanofluids. Int. J. Heat Mass Transf. 2017, 107, 358–371. [Google Scholar] [CrossRef]
- Sarkar, S.; Selvam, R.P. Molecular dynamics simulation of effective thermal conductivity and study of enhanced thermal transport mechanism in nanofluids. J. Appl. Phys. 2007, 102, 074302. [Google Scholar] [CrossRef]
- Prasher, R.; Bhattacharya, P.; Phelan, P.E. Brownian-Motion-Based Convective-Conductive Model for the Effective Thermal Conductivity of Nanofluids. J. Heat Transf. 2006, 128, 588. [Google Scholar] [CrossRef]
- Prasher, R.; Phelan, P.E.; Bhattacharya, P. Effect of Aggregation Kinetics on the Thermal Conductivity of Nanoscale Colloidal Solutions (Nanofluid). Nano Lett. 2006, 6, 1529–1534. [Google Scholar] [CrossRef]
- Keblinski, P.; Thomin, J. Hydrodynamic field around a Brownian particle. Phys. Rev. E 2006, 73, 10502. [Google Scholar] [CrossRef]
- Keblinski, P.; Phillpot, S.R.; Choi, S.U.S.; Eastman, J.A. Mechanisms of heat flow in suspensions of nano-sized particles (nanofluids). Int. J. Heat Mass Transf. 2002, 45, 855–863. [Google Scholar] [CrossRef]
- Trisaksri, V.; Wongwises, S. Critical review of heat transfer characteristics of nanofluids. Renew. Sustain. Energy Rev. 2007, 11, 512–523. [Google Scholar] [CrossRef]
- Cahill, D.G. Thermal conductivity measurement from 30 to 750 K: the 3ω method. Rev. Sci. Instrum. 1990, 61, 802. [Google Scholar] [CrossRef]
- Cahill, D.G. Erratum: “Thermal conductivity measurement from 30 to 750 K: The 3ω method” [Rev. Sci. Instrum. 61, 802 (1990)]. Rev. Sci. Instrum. 2002, 73, 3701. [Google Scholar] [CrossRef]
- Oh, D.W.; Jain, A.; Eaton, J.K.; Goodson, K.E.; Lee, J.S. Thermal conductivity measurement and sedimentation detection of aluminum oxide nanofluids by using the 3ω method. Int. J. Heat Fluid Flow 2008, 29, 1456–1461. [Google Scholar] [CrossRef]
- Lubner, S.D.; Choi, J.; Wehmeyer, G.; Waag, B.; Mishra, V.; Natesan, H.; Bischof, J.C.; Dames, C. Reusable bi-directional 3 ω sensor to measure thermal conductivity of 100-μ m thick biological tissues. Rev. Sci. Instrum. 2015, 86, 014905. [Google Scholar] [CrossRef]
- Chavez-Angel, E.; Reuter, N.; Komar, P.; Heinz, S.; Kolb, U.; Kleebe, H.-J.; Jakob, G. Subamorphous Thermal Conductivity of Crystalline Half-Heusler Superlattices. Nanoscale Microscale Thermophys. Eng. 2019, 23, 1–9. [Google Scholar] [CrossRef]
- Kole, M.; Dey, T.K. Investigation of thermal conductivity, viscosity, and electrical conductivity of graphene based nanofluids. J. Appl. Phys. 2013, 113, 84307. [Google Scholar] [CrossRef]
- Mehrali, M.; Sadeghinezhad, E.; Tahan Latibari, S.; Mehrali, M.; Togun, H.; Zubir, M.N.M.; Kazi, S.N.; Metselaar, H.S.C. Preparation, characterization, viscosity, and thermal conductivity of nitrogen-doped graphene aqueous nanofluids. J. Mater. Sci. 2014, 49, 7156–7171. [Google Scholar] [CrossRef]
- Vallejo, J.P.; Żyła, G.; Fernández-Seara, J.; Lugo, L. Rheological behaviour of functionalized graphene nanoplatelet nanofluids based on water and propylene glycol:water mixtures. Int. Commun. Heat Mass Transf. 2018, 99, 43–53. [Google Scholar] [CrossRef]
- Vallejo, J.; Żyła, G.; Fernández-Seara, J.; Lugo, L. Influence of Six Carbon-Based Nanomaterials on the Rheological Properties of Nanofluids. Nanomaterials 2019, 9, 146. [Google Scholar] [CrossRef] [PubMed]
- Chalapathi, V.V.; Ramiah, K.V. Normal vibrations of N-Dimethylformamide and N, N-Dimethylacetamide. Proc. Indian Acad. Sci. 1968, 91, 109–122. [Google Scholar] [CrossRef]
- Peek, P.S.; McDermott, D.P. Vibrational modes and frequencies of 2-pyrrolidinones and their deutero-isotopomers. Spectrochim. Acta Part A Mol. Spectrosc. 1988, 44, 371–377. [Google Scholar] [CrossRef]
- Xu, W.; Wang, H.; Tao, Y.; Zheng, X. The structural organization of N-methyl-2-pyrrolidinone in binary mixtures probed by Raman spectroscopy: Experimental and quantum chemical results. J. Raman Spectrosc. 2018, 49, 362–371. [Google Scholar] [CrossRef]
- Rozpłoch, F.; Patyk, J.; Stankowski, J. Graphenes Bonding Forces in Graphite. Acta Phys. Pol. A 2007, 112, 557–562. [Google Scholar] [CrossRef]
- Kemnitz, C.R.; Loewen, M.J. “Amide Resonance” Correlates with a Breadth of C−N Rotation Barriers. J. Am. Chem. Soc. 2007, 129, 2521–2528. [Google Scholar] [CrossRef] [PubMed]
- Adams, W.A.; Kruus, P.; Patraboy, T.J. The system sulfur dioxide–N-methyl-2-pyrrolidinone. Can. J. Chem. 1983, 61, 37–44. [Google Scholar] [CrossRef]
- Basma, N.S.; Headen, T.F.; Shaffer, M.S.P.; Skipper, N.T.; Howard, C.A. Local Structure and Polar Order in Liquid N -Methyl-2-pyrrolidone (NMP). J. Phys. Chem. B 2018, 122, 8963–8971. [Google Scholar] [CrossRef] [PubMed]
- Yau, H.C.; Bayazit, M.K.; Steinke, J.H.G.; Shaffer, M.S.P. Sonochemical degradation of N-methylpyrrolidone and its influence on single walled carbon nanotube dispersion. Chem. Commun. 2015, 51, 16621–16624. [Google Scholar] [CrossRef] [PubMed]
- Ogilvie, S.P.; Large, M.J.; Fratta, G.; Meloni, M.; Canton-Vitoria, R.; Tagmatarchis, N.; Massuyeau, F.; Ewels, C.P.; King, A.A.K.; Dalton, A.B. Considerations for spectroscopy of liquid-exfoliated 2D materials: Emerging photoluminescence of N-methyl-2-pyrrolidone. Sci. Rep. 2017, 7, 16706. [Google Scholar] [CrossRef] [PubMed]
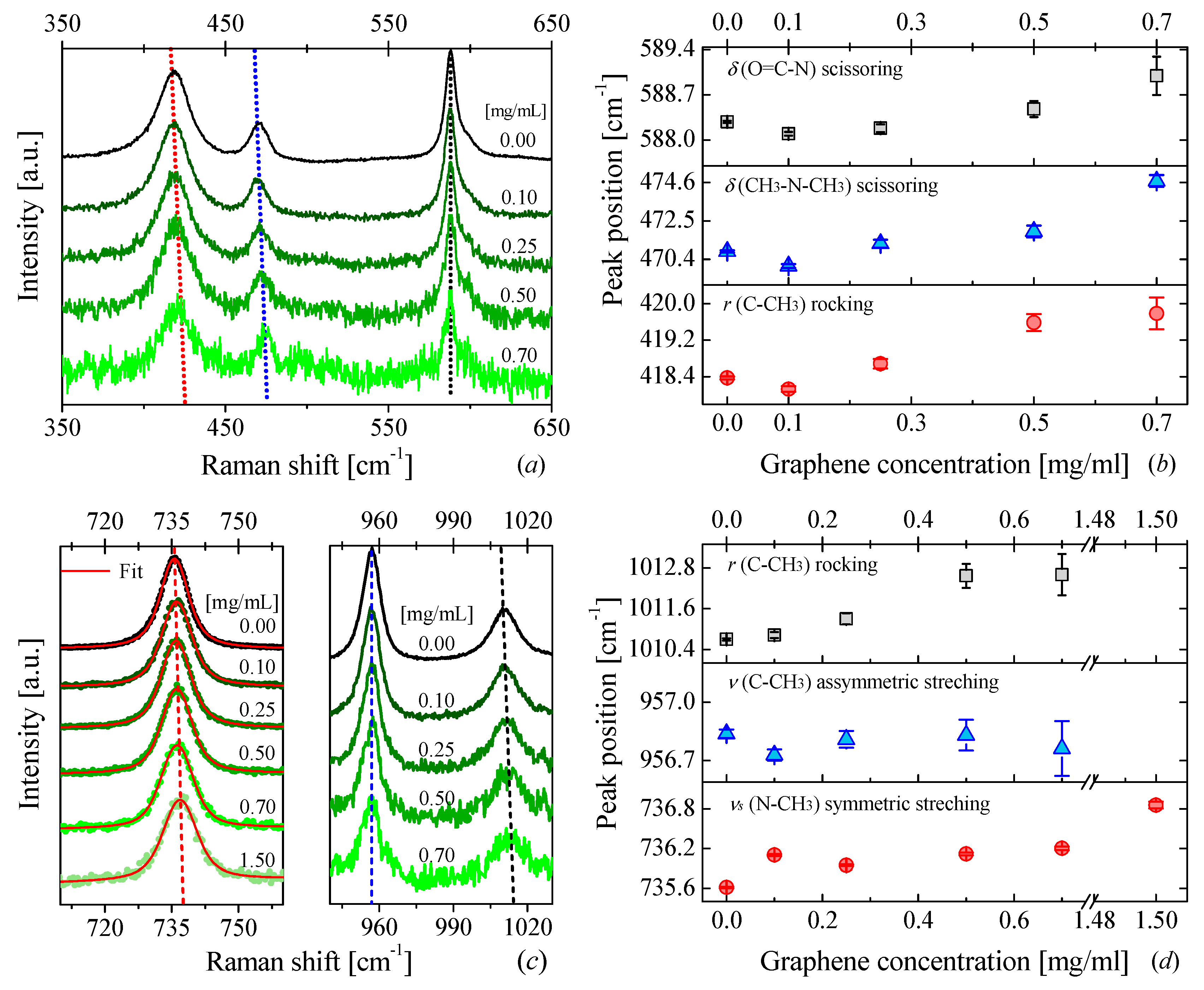
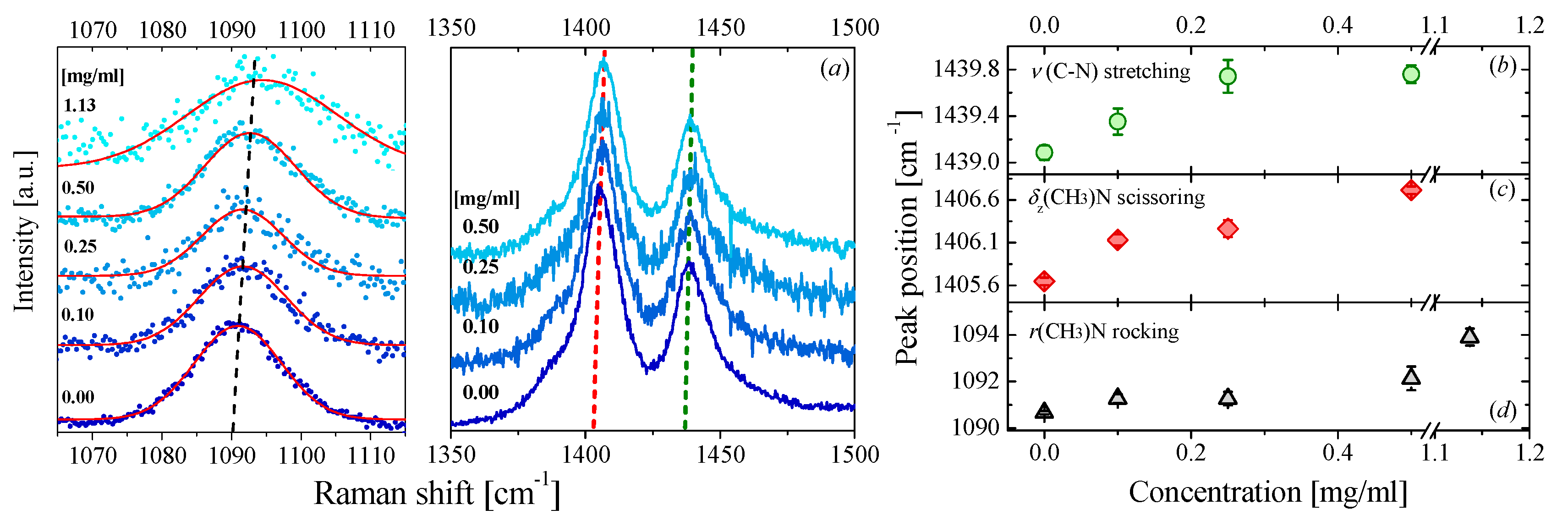
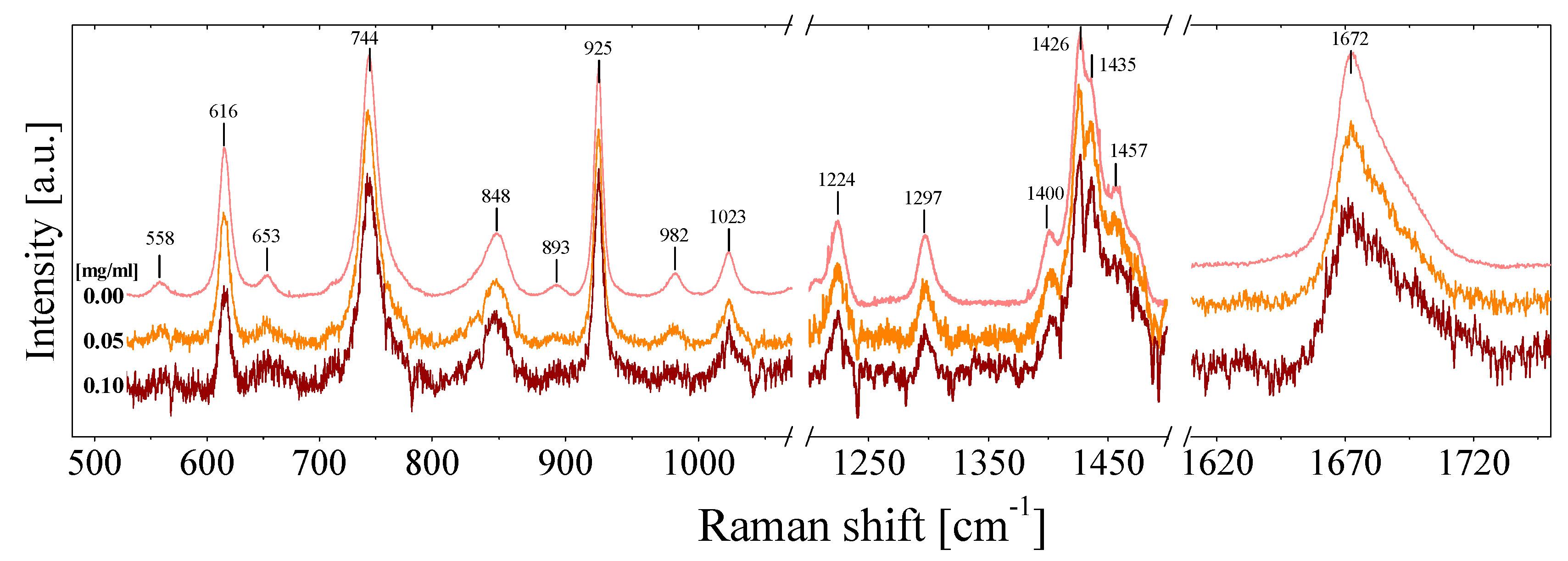


| Concentration | Samples | ||||||
|---|---|---|---|---|---|---|---|
| DMAc * | DMF * | NMP (This Work) | |||||
| mg/mL | wt% | k (W m−1 K−1) | Viscosity (mPa·s) | K (W m−1 K−1) | Viscosity (mPa·s) | k (W m−1 K−1) | Viscosity (mPa·s) |
| 0.00 | 0 | 0.175 | 1.19 | 0.183 | 0.94 | 0.235 | 2.07 |
| 0.05 | 0.005 | - | - | - | - | 0.234 | 2.19 |
| 0.10 | 0.01 | 0.180 | 1.17 | 0.194 | 0.99 | 0.236 | 2.21 |
| 0.25 | 0.03 | 0.196 | 1.18 | 0.203 | 1.01 | - | - |
| 0.50 | 0.05 | 0.206 | 1.26 | 0.228 | 1.08 | 0.213 | 2.92 |
| 1.13 | 0.12 | - | - | - | 1.26 | - | - |
| 1.50 | 0.18 | 0.259 | 1.68 | - | - | - | - |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rodríguez-Laguna, M.d.R.; Gómez-Romero, P.; Sotomayor Torres, C.M.; Chavez-Angel, E. Modification of the Raman Spectra in Graphene-Based Nanofluids and Its Correlation with Thermal Properties. Nanomaterials 2019, 9, 804. https://doi.org/10.3390/nano9050804
Rodríguez-Laguna MdR, Gómez-Romero P, Sotomayor Torres CM, Chavez-Angel E. Modification of the Raman Spectra in Graphene-Based Nanofluids and Its Correlation with Thermal Properties. Nanomaterials. 2019; 9(5):804. https://doi.org/10.3390/nano9050804
Chicago/Turabian StyleRodríguez-Laguna, María del Rocío, Pedro Gómez-Romero, Clivia M. Sotomayor Torres, and Emigdio Chavez-Angel. 2019. "Modification of the Raman Spectra in Graphene-Based Nanofluids and Its Correlation with Thermal Properties" Nanomaterials 9, no. 5: 804. https://doi.org/10.3390/nano9050804
APA StyleRodríguez-Laguna, M. d. R., Gómez-Romero, P., Sotomayor Torres, C. M., & Chavez-Angel, E. (2019). Modification of the Raman Spectra in Graphene-Based Nanofluids and Its Correlation with Thermal Properties. Nanomaterials, 9(5), 804. https://doi.org/10.3390/nano9050804








