Impact of Tetrazolium Ionic Liquid Thermal Decomposition in Solvothermal Reaction on the Remarkable Photocatalytic Properties of TiO2 Particles
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of the ILs–TiO2 Particles
2.2. Characterization of ILs–TiO2 Particles
2.3. Photocatalytic Test
3. Results and Discussion
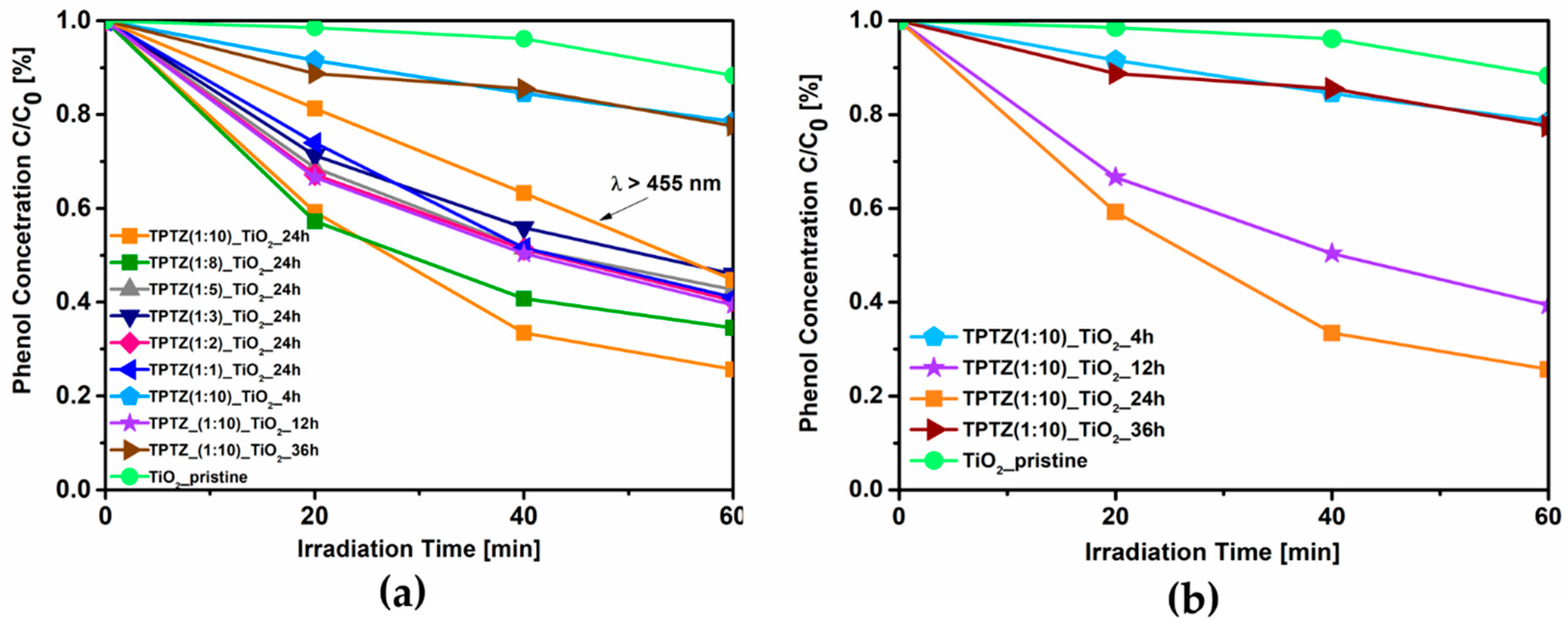
3.1. Photocatalytic Activity
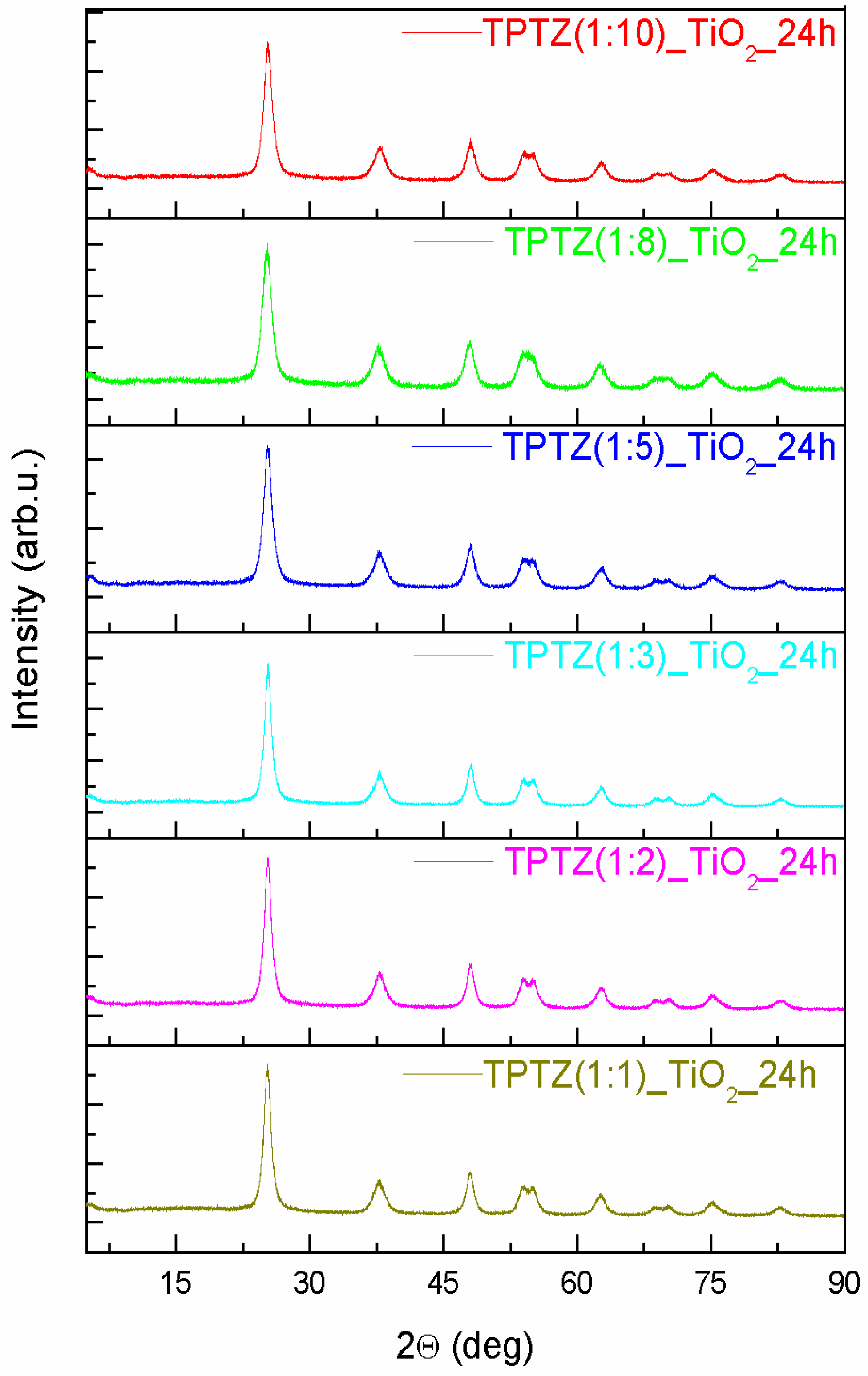
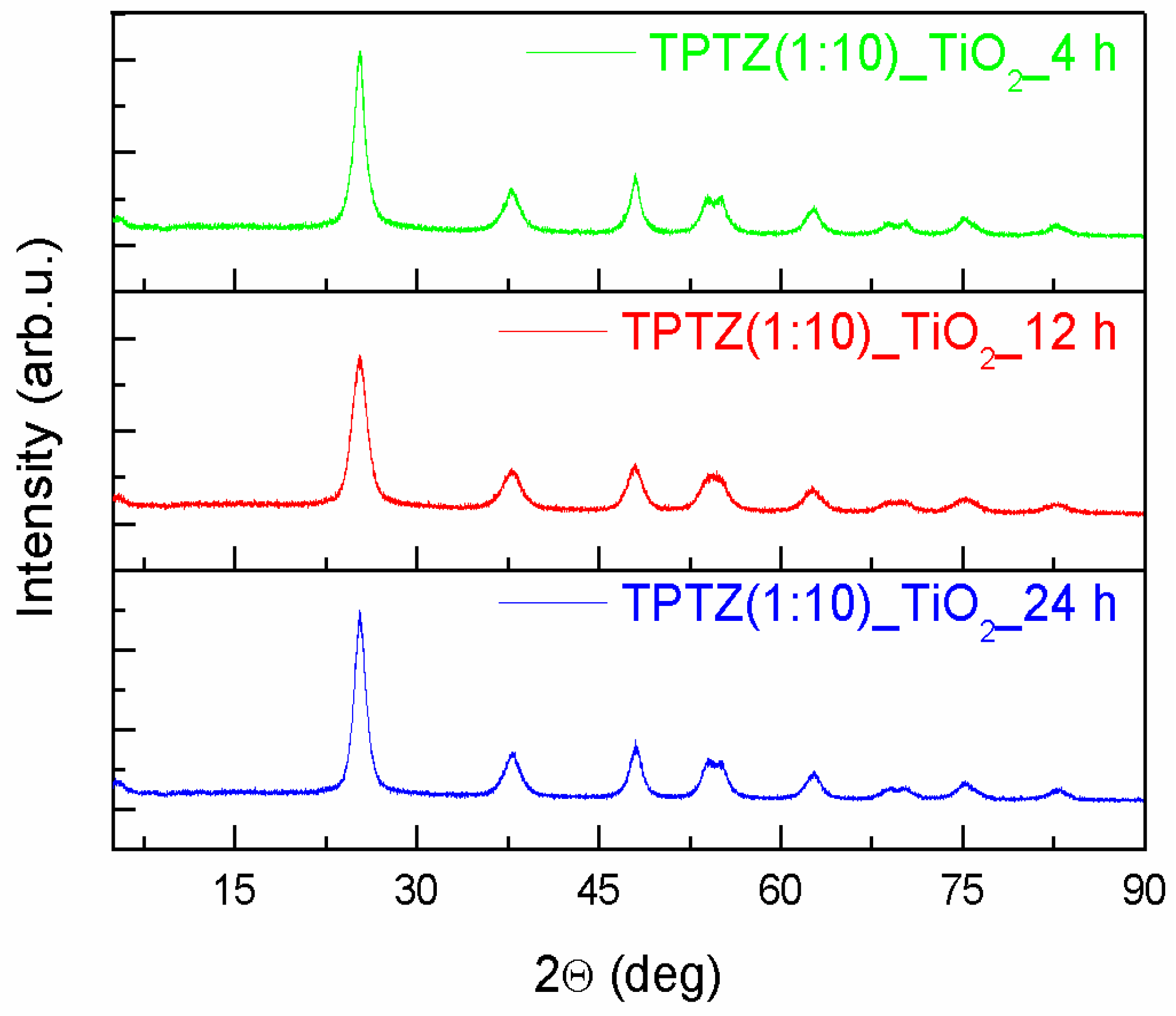
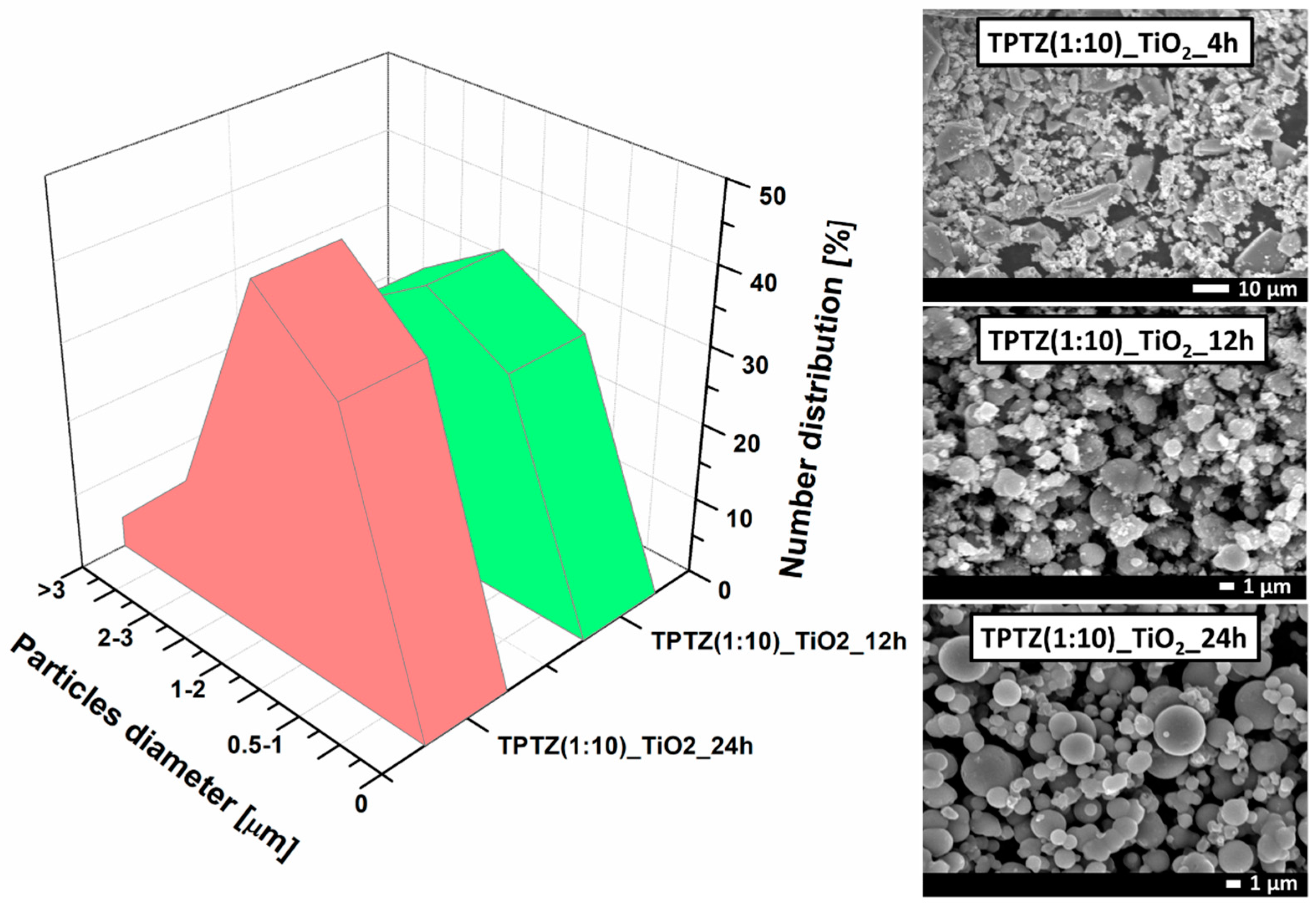
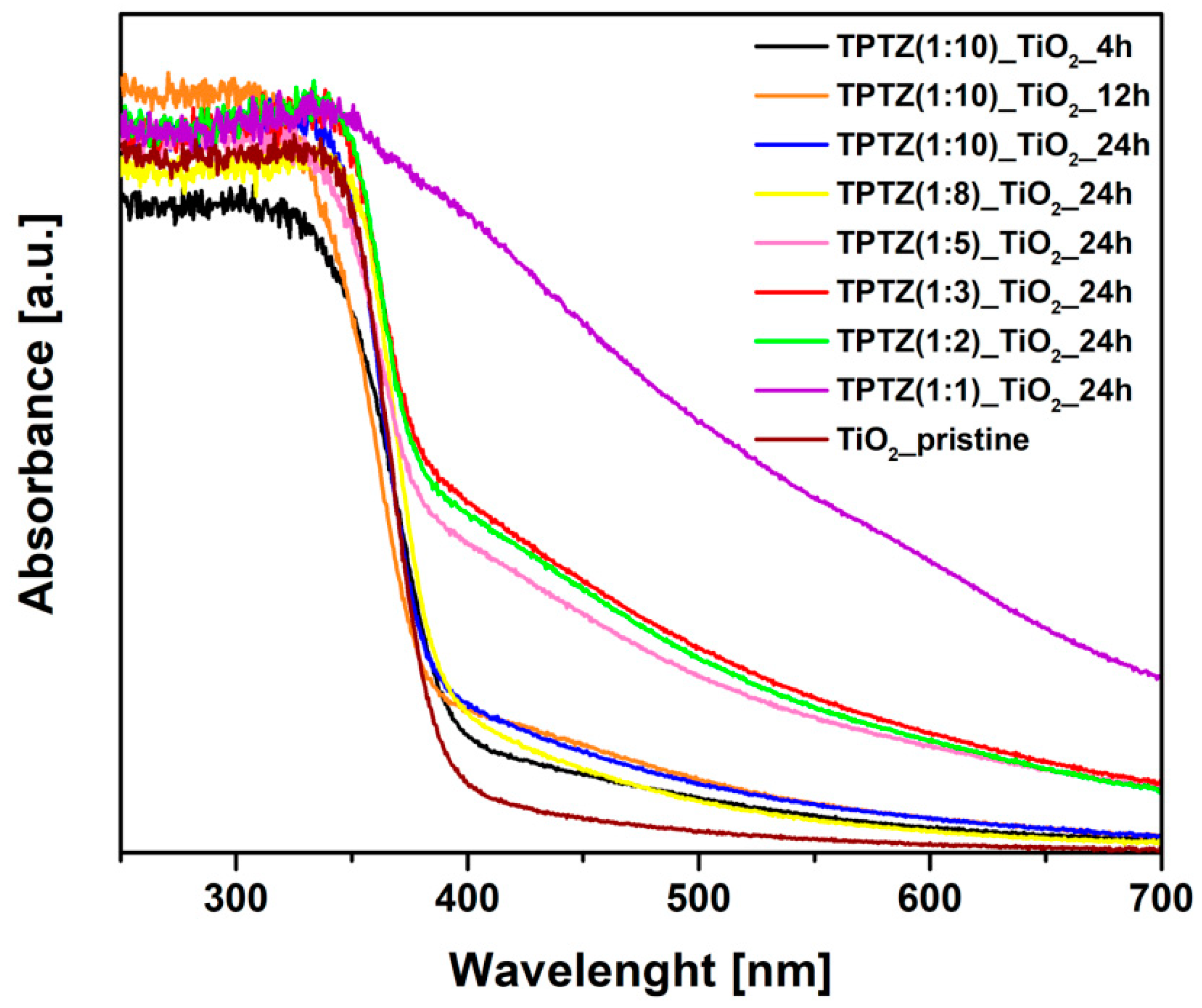
3.2. Structure, Morphology, and Absorption Properties
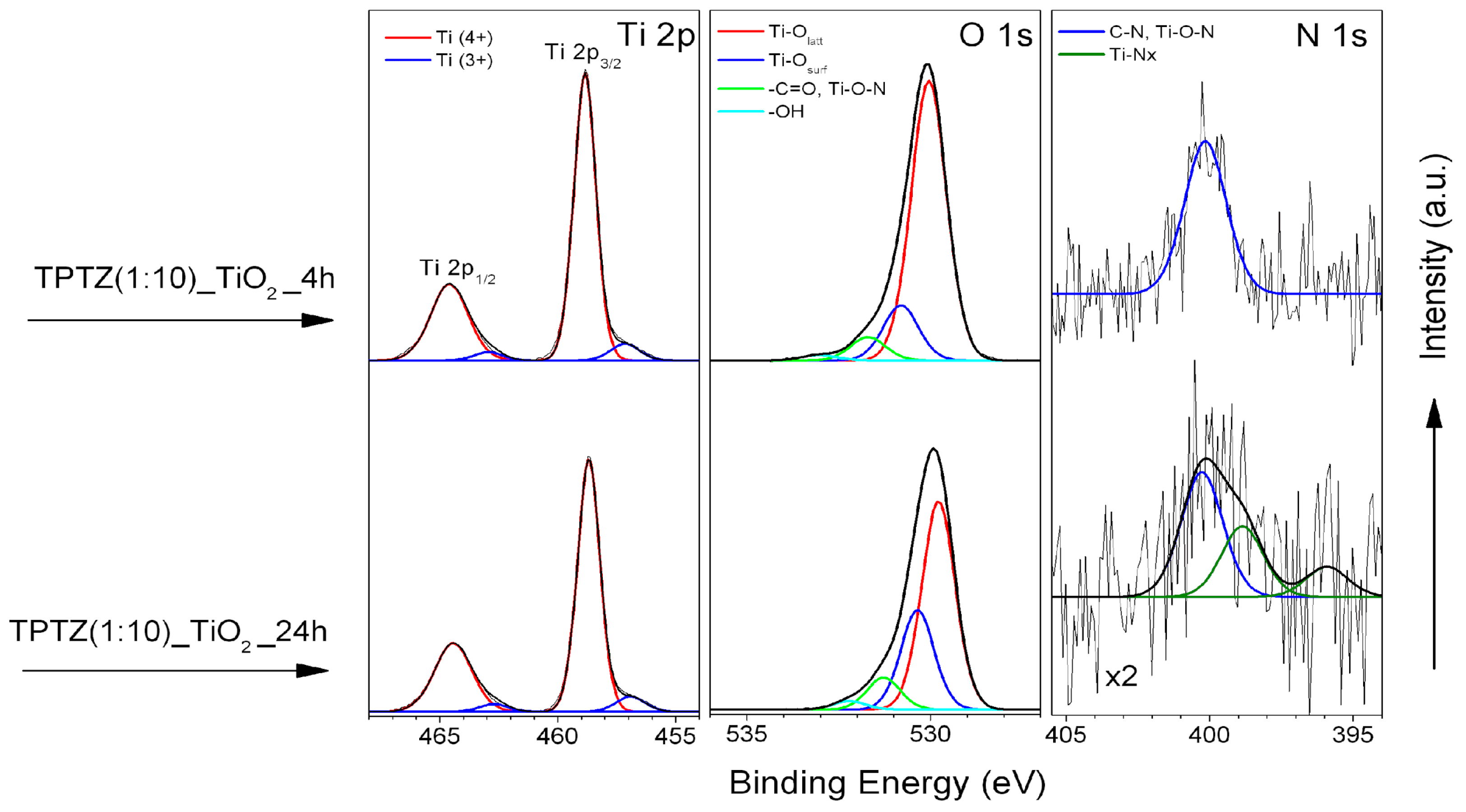
3.3. Surface Composition
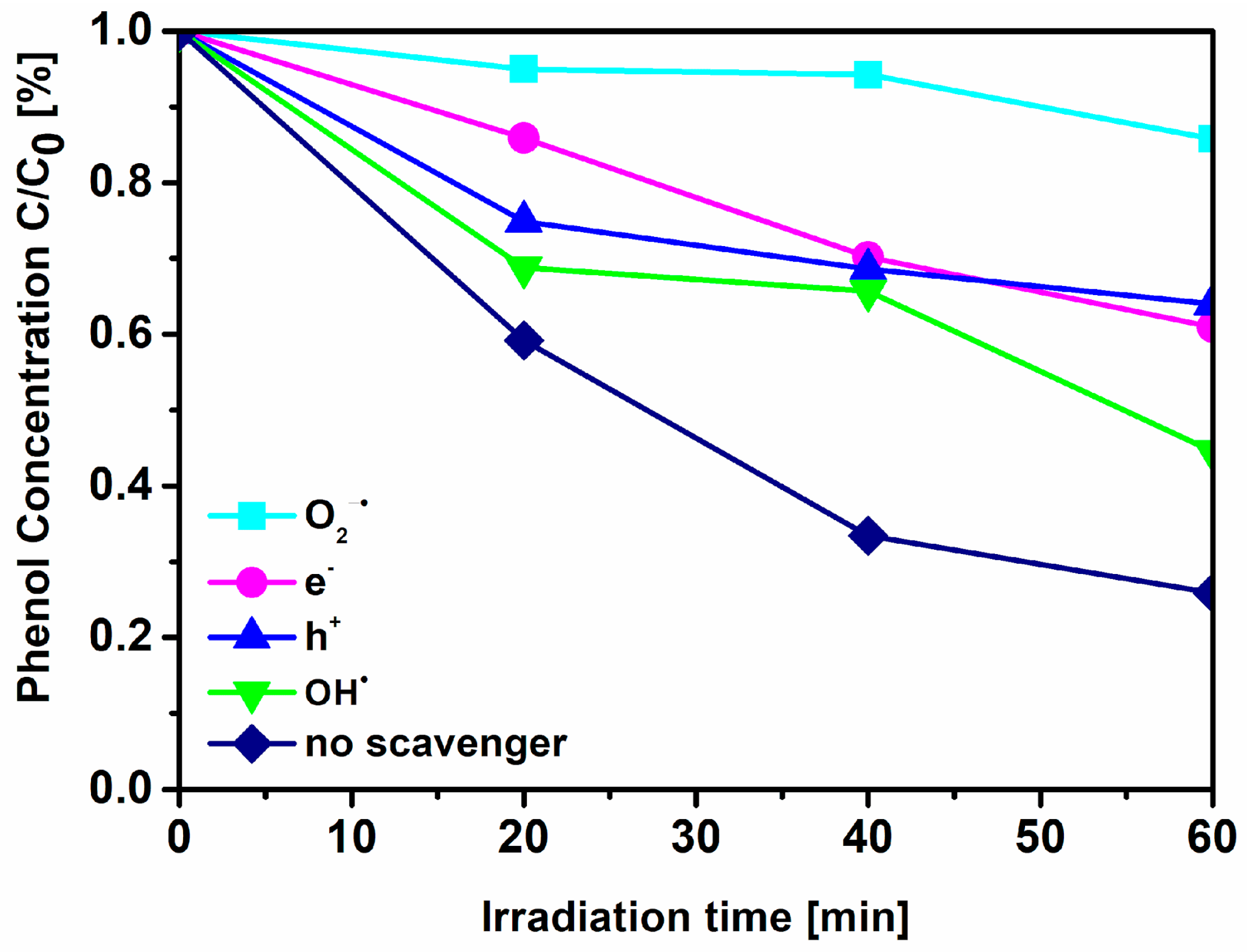
3.4. Mechanism of the Photocatalytic Activity of TiO2_[TPTZ][Cl] Microparticles
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Izgorodina, E.I.; Seeger, Z.L.; Scarborough, D.L.A.; Tan, S.Y.S. Quantum Chemical Methods for the Prediction of Energetic, Physical, and Spectroscopic Properties of Ionic Liquids. Chem. Rev. 2017, 117, 6696–6754. [Google Scholar] [CrossRef]
- Liu, H.; Yu, H. Ionic liquids for electrochemical energy storage devices applications. J. Mater. Sci. Technol. 2019, 35, 674–686. [Google Scholar] [CrossRef]
- Egorova, K.S.; Ananikov, V.P. Fundamental importance of ionic interactions in the liquid phase: A review of recent studies of ionic liquids in biomedical and pharmaceutical applications. J. Mol. Liq. 2018, 272, 271–300. [Google Scholar] [CrossRef]
- Mahamat Nor, S.B.; Woi, P.M.; Ng, S.H. Characterisation of ionic liquids nanoemulsion loaded with piroxicam for drug delivery system. J. Mol. Liq. 2017, 234, 30–39. [Google Scholar] [CrossRef]
- Cognigni, A.; Kampichler, S.; Bica, K. Surface-active ionic liquids in catalysis: Impact of structure and concentration on the aerobic oxidation of octanol in water. J. Colloid Interf. Sci. 2017, 492, 136–145. [Google Scholar] [CrossRef]
- Łuczak, J.; Paszkiewicz, M.; Krukowska, A.; Malankowska, A.; Zaleska-Medynska, A. Ionic liquids for nano- and microstructures preparation. Part 2: Application in synthesis. Adv. Colloid Interface Sci. 2016, 227, 1–52. [Google Scholar] [CrossRef] [PubMed]
- Łuczak, J.; Paszkiewicz-Gawron, M.; Długokęcka, M.; Lisowski, W.; Grabowska, E.; Makurat, S.; Rak, J.; Zaleska-Medynska, A. Visible light photocatalytic activity of ionic liquid-TiO2 spheres: Effect of the ionic liquid’s anion structure. ChemCatChem 2017, 9, 4377–4388. [Google Scholar] [CrossRef]
- Paszkiewicz, M.; Łuczak, J.; Lisowski, W.; Patyk, P.; Zaleska-Medynska, A. The ILs-assisted solvothermal synthesis of TiO2 spheres: The effect of ionic liquids on morphology and photoactivity of TiO2. Appl. Catal. B-Environ. 2016, 184, 223–237. [Google Scholar] [CrossRef]
- Nakata, K.; Fujishima, A. TiO2 photocatalysis: Design and applications. J. Photochem. Photobiol. C 2012, 13, 169–189. [Google Scholar] [CrossRef]
- Schneider, J.; Matsuoka, M.; Takeuchi, M.; Zhang, J.; Horiuchi, Y.; Anpo, M.; Bahnemann, D.W. Understanding TiO2 photocatalysis: Mechanisms and materials. Chem. Rev. 2014, 114, 9919–9986. [Google Scholar] [CrossRef]
- Linsebigler, A.L.; Lu, G.; Yates, J.T., Jr. Photocatalysis on TiO2 surfaces: Principles, mechanisms, and selected results. Chem. Rev. 1995, 95, 735–758. [Google Scholar] [CrossRef]
- Yun, E.-T.; Yoo, H.-Y.; Kim, W.; Kim, H.-E.; Kang, G.; Lee, H.; Lee, S.; Park, T.; Lee, C.; Kim, J.-H.; et al. Visible-light-induced activation of periodate that mimics dye-sensitization of TiO2: Simultaneous decolorization of dyes and production of oxidizing radicals. Appl. Catal. B-Environ. 2017, 203, 475–484. [Google Scholar] [CrossRef]
- Huang, F.; Yan, A.; Zhao, H. Influences of Doping on Photocatalytic Properties of TiO2 Photocatalyst. In Semiconductor Photocatalysis—Materials, Mechanisms and Applications; Cao, W., Ed.; InTech: Rijeka, Croatia, 2016; Chapter 02. [Google Scholar]
- Gołąbiewska, A.; Zielińska-Jurek, A.; Zaleska, A. Characterization of TiO2 modified with bimetallic Ag/Au nanoparticles obtained in microemulsion system. J. Adv. Oxid. Technol. 2012, 15, 71–77. [Google Scholar]
- Wang, Y.; Tao, J.; Wang, X.; Wang, Z.; Zhang, M.; He, G.; Sun, Z. A unique Cu2O/TiO2 nanocomposite with enhanced photocatalytic performance under visible light irradiation. Ceram. Int. 2017, 43, 4866–4872. [Google Scholar] [CrossRef]
- Chen, Y.; Li, W.; Wang, J.; Gan, Y.; Liu, L.; Ju, M. Microwave-assisted ionic liquid synthesis of Ti3+ self-doped TiO2 hollow nanocrystals with enhanced visible-light photoactivity. Appl. Catal. B-Environ. 2016, 191, 94–105. [Google Scholar] [CrossRef]
- Ramanathan, R.; Bansal, V. Ionic liquid mediated synthesis of nitrogen, carbon and fluorine-codoped rutile TiO 2 nanorods for improved UV and visible light photocatalysis. RSC Adv. 2015, 5, 1424–1429. [Google Scholar] [CrossRef]
- Yu, J.; Li, Q.; Liu, S.; Jaroniec, M. Ionic-Liquid-Assisted Synthesis of Uniform Fluorinated B/C-Codoped TiO2 Nanocrystals and Their Enhanced Visible-Light Photocatalytic Activity. Chem.-Eur. J. 2013, 19, 2433–2441. [Google Scholar] [CrossRef]
- Mazierski, P.; Łuczak, J.; Lisowski, W.; Winiarski, M.J.; Klimczuk, T.; Zaleska-Medynska, A. The ILs-assisted electrochemical synthesis of TiO2 nanotubes: The effect of ionic liquids on morphology and photoactivity. Appl. Catal. B-Environ. 2017, 214, 100–113. [Google Scholar] [CrossRef]
- Li, F.-T.; Wang, X.-J.; Zhao, Y.; Liu, J.-X.; Hao, Y.-J.; Liu, R.-H.; Zhao, D.-S. Ionic-liquid-assisted synthesis of high-visible-light-activated N–B–F-tri-doped mesoporous TiO2 via a microwave route. Appl. Catal. B-Environ. 2014, 144, 442–453. [Google Scholar] [CrossRef]
- Hu, S.; Wang, A.; Li, X.; Wang, Y.; Löwe, H. Hydrothermal Synthesis of Ionic Liquid [Bmim] OH-Modified TiO2 Nanoparticles with Enhanced Photocatalytic Activity under Visible Light. Chem.-Asian J. 2010, 5, 1171–1177. [Google Scholar] [CrossRef]
- Paszkiewicz-Gawron, M.; Długokȩcka, M.; Lisowski, W.; Cristina Paganini, M.; Giamello, E.; Klimczuk, T.; Paszkiewicz, M.; Grabowska, E.; Zaleska-Medynska, A.; Łuczak, J. Dependence between Ionic Liquid Structure and Mechanism of Visible-Light-Induced Activity of TiO2 Obtained by Ionic-Liquid- Assisted Solvothermal Synthesis. ACS Sustain. Chem. Eng. 2018, 6, 3927–3937. [Google Scholar] [CrossRef]
- Gołąbiewska, A.; Paszkiewicz-Gawron, M.; Sadzińska, A.; Lisowski, W.; Grabowska, E.; Zaleska-Medynska, A.; Łuczak, J. Fabrication and photoactivity of ionic liquid–TiO2 structures for efficient visible-light-induced photocatalyticdecomposition of organic pollutants in aqueous phase. Beilstein J. Nanotechnol. 2018, 9, 580–590. [Google Scholar] [CrossRef]
- Deng, F.; Luo, X.; Li, K.; Tu, X.; Luo, S.; Yang, L.; Zhou, N.; Shu, H. The effect of vinyl-containing ionic liquid on the photocatalytic activity of iron-doped TiO2. J. Mol. Catal. A-Chem. 2013, 366, 222–227. [Google Scholar] [CrossRef]
- Liu, H.; Wang, M.; Wang, Y.; Liang, Y.; Cao, W.; Su, Y. Ionic liquid-templated synthesis of mesoporous CeO2–TiO2 nanoparticles and their enhanced photocatalytic activities under UV or visible light. J. Photochem. Photobiol. A-Chem. 2011, 223, 157–164. [Google Scholar] [CrossRef]
- Liu, S.-H.; Syu, H.-R. High visible-light photocatalytic hydrogen evolution of C,N-codoped mesoporous TiO2 nanoparticles prepared via an ionic-liquid-template approach. Int. J. Hydrog. Energy 2013, 38, 13856–13865. [Google Scholar] [CrossRef]
- Gołąbiewska, A.; Checa-Suárez, M.; Paszkiewicz-Gawron, M.; Lisowski, W.; Raczuk, E.; Klimczuk, T.; Polkowska, Ż.; Grabowska, E.; Zaleska-Medynska, A.; Łuczak, J. Highly Active TiO2 Microspheres Formation in the Presence of Ethylammonium Nitrate Ionic Liquid. Catalysts 2018, 8, 279. [Google Scholar] [CrossRef]
- Qi, L.; Yu, J.; Jaroniec, M. Enhanced and suppressed effects of ionic liquid on the photocatalytic activity of TiO2. Adsorption 2013, 19, 557–561. [Google Scholar] [CrossRef]
- Łuczak, J.; Paszkiewicz, M.; Krukowska, A.; Malankowska, A.; Zaleska-Medynska, A. Ionic liquids for nano- and microstructures preparation. Part 1: Properties and multifunctional role. Adv. Colloid Interfac. 2016, 230, 13–28. [Google Scholar] [CrossRef]
- Naumkin, A.V.; Kraut-Vass, A.; Gaarenstroom, S.W.; Powell, C.J. NIST X-ray Photoelectron Spectroscopy Database 20, Version 4.1; National Institute of Standards and Technology: Gaithersburg, MD, USA, 2012.
- Cong, Y.; Zhang, J.; Chen, F.; Anpo, M. Synthesis and Characterization of Nitrogen-Doped TiO2 Nanophotocatalyst with High Visible Light Activity. J. Phys. Chem. C 2007, 111, 6976–6982. [Google Scholar] [CrossRef]
- Asahi, R.; Morikawa, T.; Ohwaki, T.; Aoki, K.; Taga, Y. Visible-Light Photocatalysis in Nitrogen-Doped Titanium Oxides. Science 2001, 293, 269–271. [Google Scholar] [CrossRef]
- Antony, R.P.; Mathews, T.; Panda, K.; Sundaravel, B.; Dash, S.; Tyagi, A. Enhanced field emission properties of electrochemically synthesized self-aligned nitrogen-doped TiO2 nanotube array thin films. J. Phys. Chem. C 2012, 116, 16740–16746. [Google Scholar] [CrossRef]
- Dunnill, C.W.; Parkin, I.P. Nitrogen-doped TiO2 thin films: Photocatalytic applications for healthcare environments. Dalton Trans. 2011, 40, 1635–1640. [Google Scholar] [CrossRef]
- Viswanathan, B.; Krishanmurthy, K.R. Nitrogen Incorporation in TiO2: Does It Make a Visible Light Photo-Active Material? Int. J. Photoenergy 2012, 269654. [Google Scholar] [CrossRef]
- Jiang, Z.; Kong, L.; Alenazey, F.S.; Qian, Y.; France, L.; Xiao, T.; Edwards, P.P. Enhanced visible-light-driven photocatalytic activity of mesoporous TiO2−xNxderived from the ethylenediamine-based complex. Nanoscale 2013, 5, 5396–5402. [Google Scholar] [CrossRef][Green Version]
- Hu, S.; Wang, A.; Li, X.; Löwe, H. Hydrothermal synthesis of well-dispersed ultrafine N-doped TiO2nanoparticles with enhanced photocatalytic activity under visible light. J. Phys. Chem. Solids 2010, 71, 156–162. [Google Scholar] [CrossRef]
- Ansari, S.A.; Khan, M.M.; Ansari, M.O.; Cho, M.H. Nitrogen-doped titanium dioxide (N-doped TiO2) for visible light photocatalysis. New J. Chem. 2016, 146, 199–204. [Google Scholar] [CrossRef]
- Di Valentin, C.; Pacchioni, G.; Selloni, A.; Livraghi, S.; Giamello, E. Characterization of Paramagnetic Species in N-Doped TiO2 Powders by EPR Spectroscopy and DFT Calculations. J. Phys. Chem. B 2005, 109, 11414–11419. [Google Scholar] [CrossRef]
- Wang, Y.; Zhu, L.; Ba, N.; Xie, H. Effects of NH4F quantity on N-doping level, photodegradation and photocatalytic H2 production activities of N-doped TiO2 nanotube array films. Mater. Res. Bull. 2017, 86, 268–276. [Google Scholar] [CrossRef]
- Wang, Y.; Feng, C.; Zhang, M.; Yang, J.; Zhang, Z. Enhanced visible light photocatalytic activity of N-doped TiO2 in relation to single-electron-trapped oxygen vacancy and doped-nitrogen. Appl. Catal. B-Environ. 2010, 100, 84–90. [Google Scholar] [CrossRef]
- Wood, P.M. The potential diagram for oxygen at pH 7. Biochem. J. 1988, 253, 287–289. [Google Scholar] [CrossRef]
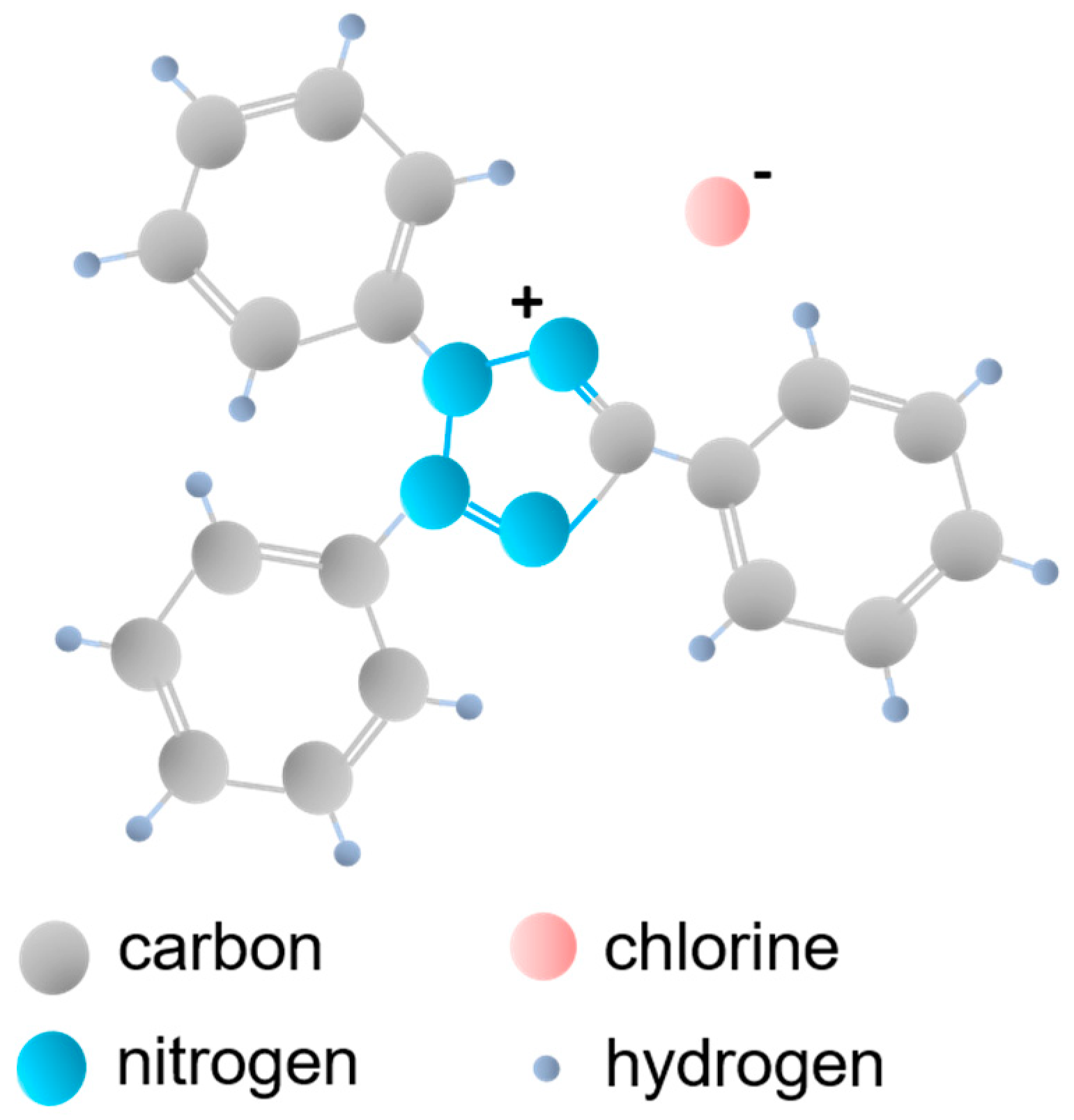

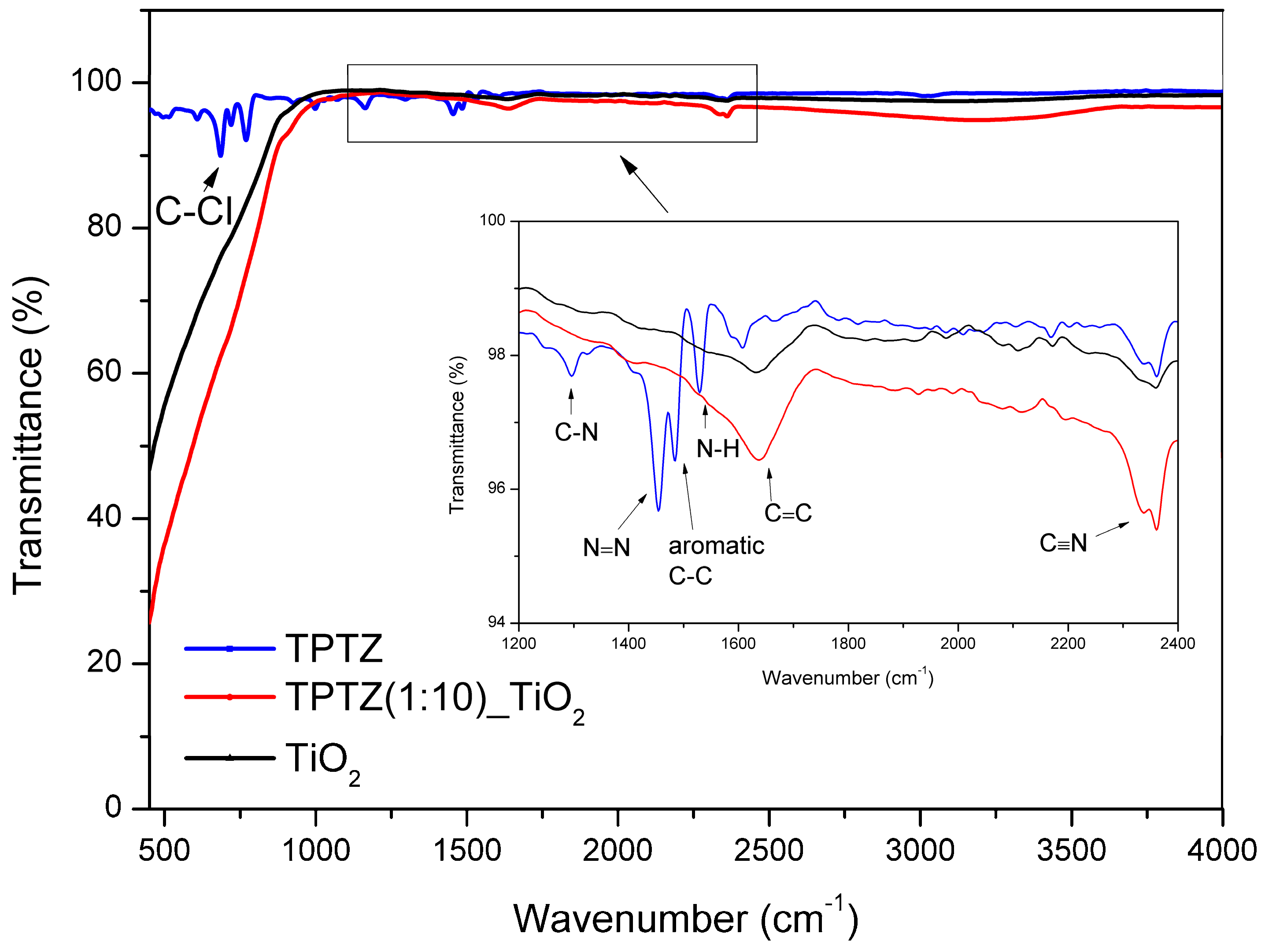
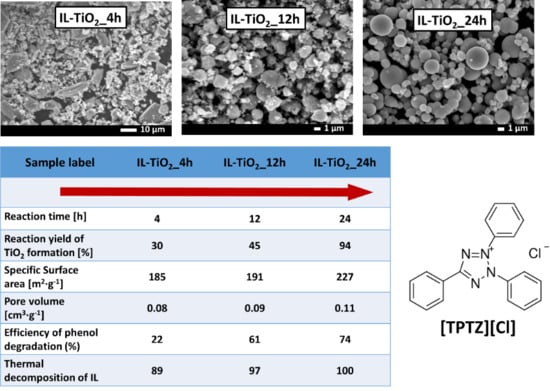
| Sample Label | IL:TBOT Molar Ratio | Crystalline Phase | Specific Surface Area (m2·g−1) | Pore Volume (cm3·g−1) | Efficiency of Phenol Degradation under Visible Irradiation (λ > 420 nm) (%) |
|---|---|---|---|---|---|
| TiO2_pristine | - | anatase | 184 | 0.07 | 7 |
| TPTZ(1:10)_TiO2_24 h | 1:10 | 227 | 0.11 | 74 (55% λ > 455 nm) * | |
| TPTZ(1:8)_TiO2_24 h | 1:8 | 212 | 0.10 | 66 | |
| TPTZ(1:5)_TiO2_24 h | 1:5 | 187 | 0.09 | 57 | |
| TPTZ(1:3)_TiO2_24 h | 1:3 | 201 | 0.10 | 58 | |
| TPTZ(1:2)_TiO2_24 h | 1:2 | 198 | 0.10 | 59 | |
| TPTZ(1:1)_TiO2_24 h | 1:1 | 219 | 0.09 | 59 | |
| TPTZ(1:10)_TiO2_1 h | 1:10 | No precipitate (product) on the bottom of the Teflon-lined autoclave was obtained. | |||
| TPTZ(1:10)_TiO2_4 h | 1:10 | anatase | 185 | 0.08 | 22 |
| TPTZ(1:10)_TiO2_12 h | 1:10 | 191 | 0.09 | 61 | |
| TPTZ(1:10)_TiO2_36 h | 1:10 | 165 | 0.08 | 23 | |
| Sample | a = b (Å) | c (Å) | V (nm3) | Crystallite Size (Å) |
|---|---|---|---|---|
| TiO2_pristine | ||||
| TPTZ(1:10)_TiO2_4 h | 3.7768(3) | 9.5470(6) | 136.18 | 74 |
| TPTZ(1:10)_TiO2_12 h | 3.7963(4) | 9.5095(5) | 137.05 | 71 |
| TPTZ(1:10)_TiO2_24 h | 3.7913(3) | 9.4922(0) | 136.44 | 57 |
| TPTZ(1:8)_TiO2_24 h | 3.7860(2) | 9.5474(2) | 136.85 | 66 |
| TPTZ(1:5)_TiO2_24 h | 3.7823(5) | 9.5261(2) | 136.28 | 59 |
| TPTZ(1:3)_TiO2_24 h | 3.7814(7) | 9.4890(6) | 135.68 | 81 |
| TPTZ(1:2)_TiO2_24 h | 3.7844(0) | 9.5254(4) | 136.42 | 60 |
| TPTZ(1:1)_TiO2_24 h | 3.7890(1) | 9.5190(0) | 136.66 | 88 |
| Sample | Elemental Composition (at. %) | |||||||
|---|---|---|---|---|---|---|---|---|
| Ti | O | C | N | Cl | C/N | Cl/N | N/Ti | |
| TiO2 | 29.44 | 66.27 | 4.14 | - | - | - | - | - |
| TPTZ(1:1)_TiO2_24 h | 19.15 | 45.59 | 32.50 | 2.63 | 0.15 | 12.3 | 0.057 | 0.1373 |
| TPTZ(1:2)_TiO2_24 h | 21.01 | 54.78 | 23.01 | 1.86 | 0.19 | 12.4 | 0.102 | 0.0885 |
| TPTZ(1:3)_TiO2_24 h | 23.59 | 61.30 | 14.43 | 0.64 | 0.04 | 22.5 | 0.063 | 0.0271 |
| TPTZ(1:5)_TiO2_24 h | 23.54 | 56.53 | 19.36 | 0.56 | 0.11 | 34.6 | 0.196 | 0.0238 |
| TPTZ(1:8)_TiO2_24 h | 25.18 | 64.64 | 9.64 | 0.38 | 0.15 | 25.4 | 0.395 | 0.0151 |
| TPTZ(1:10)_TiO2_24 h | 24.36 | 62.12 | 13.20 | 0.17 | 0.14 | 77.6 | 0.824 | 0.0070 |
| TPTZ(1:10)_TiO2_4 h | 24.84 | 60.56 | 12.38 | 0.24 | 1.97 | 51.6 | 8.208 | 0.0097 |
| Sample | Ti 2p3/2 Fractions (%) | O1s Fractions (%) | C1s Fractions (%) | N1s Fractions | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ti(4+) 458.8 ± 0.2 eV | Ti(3+) 457.3 ± 0.1 eV | Ti-Olatt 530.0 ± 0.1 eV | Ti-Osurf 530.7 ± 0.1 eV | –C=O, Ti-O-N, 531.7 ± 0.1 eV | –OH 532.5 ± 0.1 eV | “A” C–C 284.8 eV | “B” C–O, C–Cl C–N 286.1 ± 0.1 eV | “C” –C=O, N–C=N 289.0 ± 0.1 eV | N+ 401.5 ± 0.1 eV | C–N Ti–O–N 400 ± 0.1 eV | Ti-Nx 396.1–398.9 eV | |
| TPTZ(1:1)_TiO2_24 h | 96.24 | 3.76 | 84.03 | 8.67 | 5.68 | 1.63 | 81.94 | 15.92 | 2.14 | 15.89 | 84.11 | 0 |
| TPTZ(1:2)_TiO2_24 h | 96.39 | 3.61 | 77.51 | 10.60 | 9.43 | 2.46 | 89.44 | 7.34 | 3.22 | 9.50 | 90.50 | 0 |
| TPTZ(1:3)_TiO2_24 h | 93.71 | 6.29 | 65.45 | 24.65 | 7.93 | 1.97 | 73.12 | 25.09 | 1.79 | 0 | 100 | 0 |
| TPTZ(1:5)_TiO2_24 h | 93.58 | 6.42 | 65.83 | 23.53 | 8.03 | 2.61 | 74.63 | 21.24 | 4.13 | 0 | 100 | 0 |
| TPTZ(1:8)_TiO2_24 h | 96.46 | 3.54 | 74.47 | 16.44 | 5.92 | 3.17 | 70.56 | 13.72 | 15.72 | 0 | 86.41 | 13.59 |
| TPTZ(1:10)_TiO2_24 h | 92.47 | 7.53 | 59.74 | 28.51 | 9.26 | 2.49 | 66.34 | 29.84 | 3.82 | 0 | 55.25 | 44.75 |
| TPTZ(1:10)_TiO2_4 h | 93.74 | 6.26 | 71.14 | 20.35 | 6.84 | 1.67 | 72.17 | 24.73 | 3.10 | 0 | 100 | 0 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Paszkiewicz-Gawron, M.; Gołąbiewska, A.; Pancielejko, A.; Lisowski, W.; Zwara, J.; Paszkiewicz, M.; Zaleska-Medynska, A.; Łuczak, J. Impact of Tetrazolium Ionic Liquid Thermal Decomposition in Solvothermal Reaction on the Remarkable Photocatalytic Properties of TiO2 Particles. Nanomaterials 2019, 9, 744. https://doi.org/10.3390/nano9050744
Paszkiewicz-Gawron M, Gołąbiewska A, Pancielejko A, Lisowski W, Zwara J, Paszkiewicz M, Zaleska-Medynska A, Łuczak J. Impact of Tetrazolium Ionic Liquid Thermal Decomposition in Solvothermal Reaction on the Remarkable Photocatalytic Properties of TiO2 Particles. Nanomaterials. 2019; 9(5):744. https://doi.org/10.3390/nano9050744
Chicago/Turabian StylePaszkiewicz-Gawron, Marta, Anna Gołąbiewska, Anna Pancielejko, Wojciech Lisowski, Julia Zwara, Monika Paszkiewicz, Adriana Zaleska-Medynska, and Justyna Łuczak. 2019. "Impact of Tetrazolium Ionic Liquid Thermal Decomposition in Solvothermal Reaction on the Remarkable Photocatalytic Properties of TiO2 Particles" Nanomaterials 9, no. 5: 744. https://doi.org/10.3390/nano9050744
APA StylePaszkiewicz-Gawron, M., Gołąbiewska, A., Pancielejko, A., Lisowski, W., Zwara, J., Paszkiewicz, M., Zaleska-Medynska, A., & Łuczak, J. (2019). Impact of Tetrazolium Ionic Liquid Thermal Decomposition in Solvothermal Reaction on the Remarkable Photocatalytic Properties of TiO2 Particles. Nanomaterials, 9(5), 744. https://doi.org/10.3390/nano9050744