Lipid-Based Nanoparticles: Application and Recent Advances in Cancer Treatment
Abstract
1. Introduction
2. Lipid-Based Nanoparticles
2.1. Liposomes
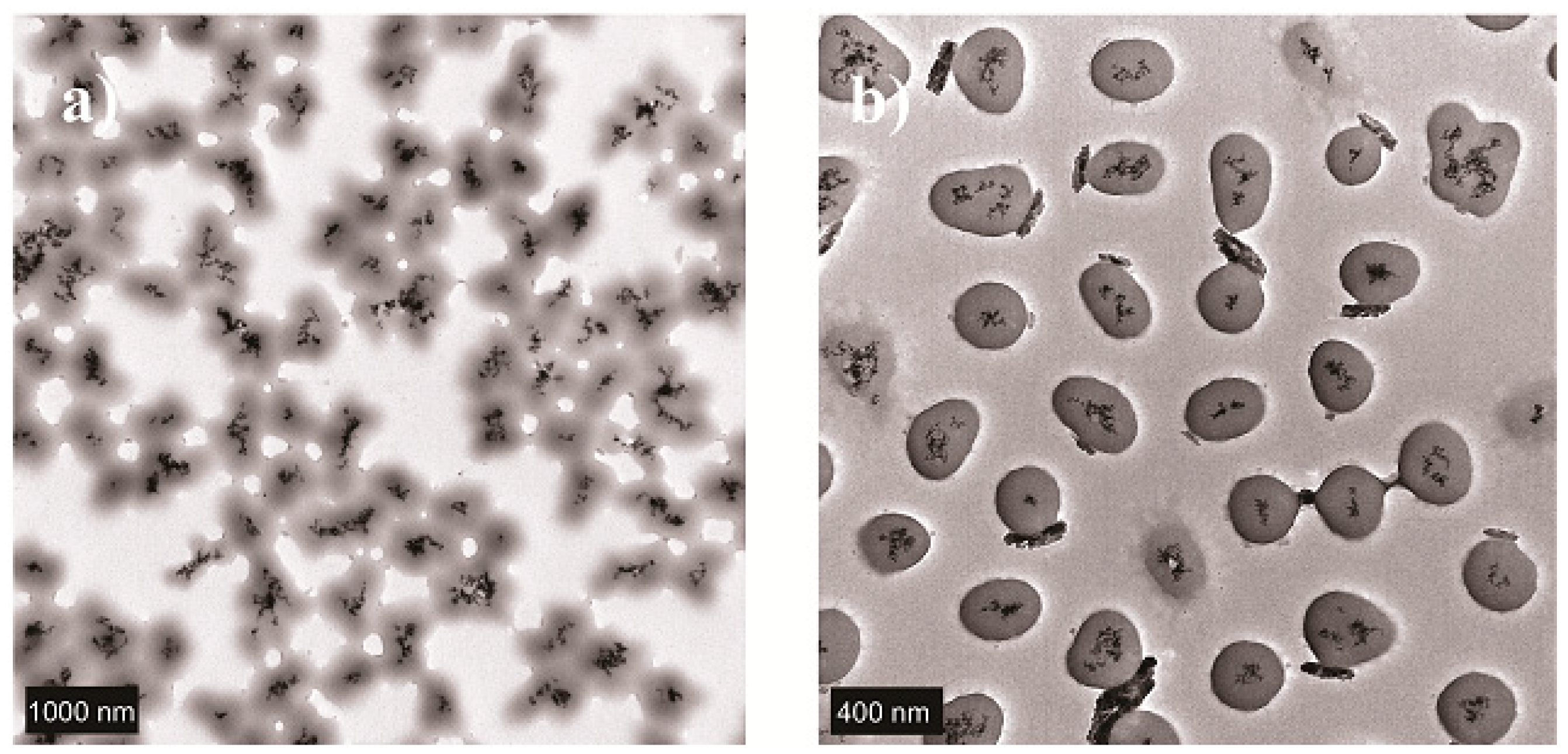
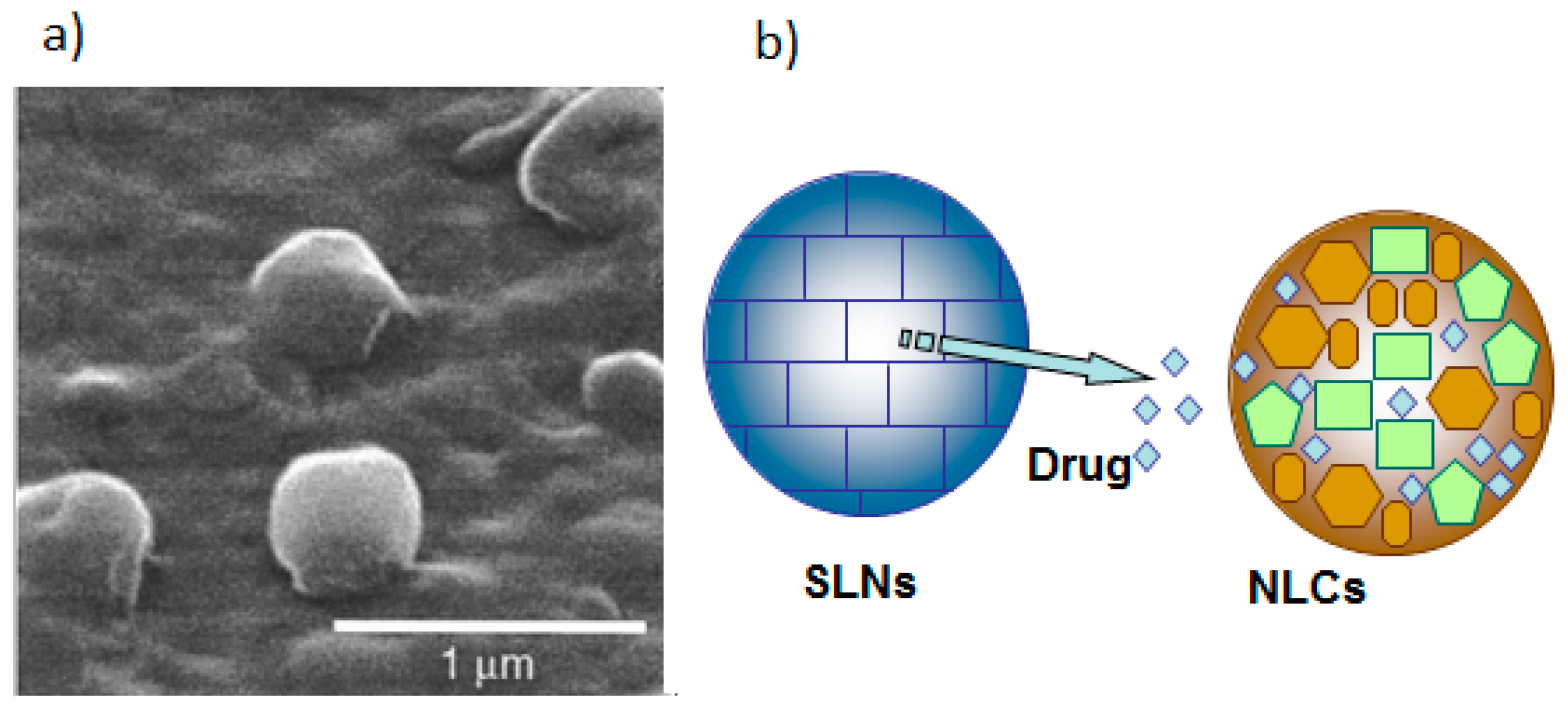
2.2. Solid Lipid Nanoparticles (SLN)
2.3. Nanostructured Lipid Carriers (NLC)
3. Nanoparticles and Cancer
4. Application of Lipid-Based Nanoparticles in Cancer Therapy
4.1. Gastrointestinal Cancer
4.1.1. Gastric and Esophageal Cancer
4.1.2. Colorectal Cancer
4.1.3. Pancreatic Cancer
4.1.4. Liver Cancer
4.2. Nervous System Cancer: Glioblastoma Multiforme
4.3. Lung Cancer
4.4. Breast Cancer
4.5. Prostate Cancer
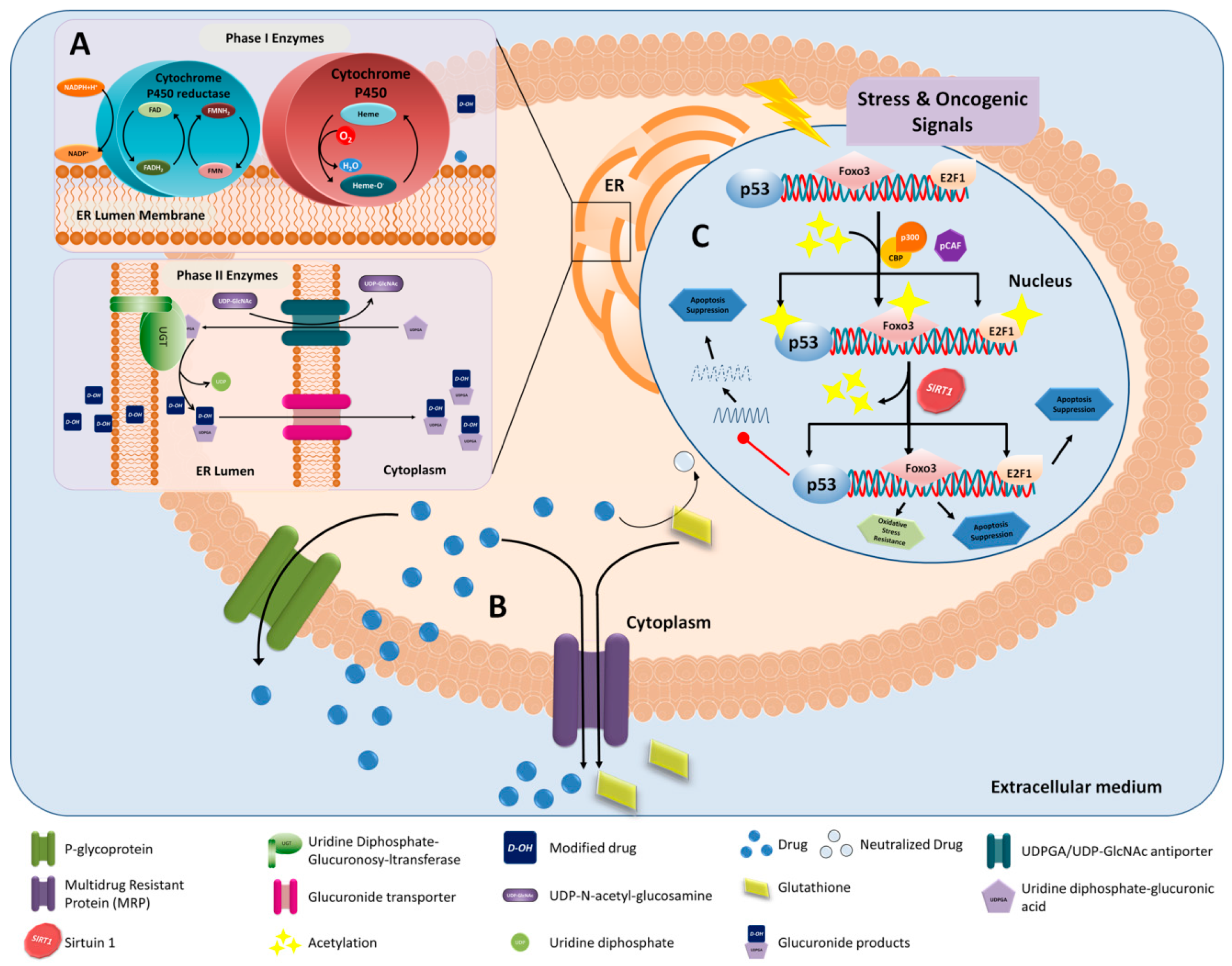
5. Lipid-Based Nanoparticles, Drug Resistance and Epithelial-Mesenchymal Transition
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Bor, G.; Mat Azmi, I.D.; Yaghmur, A. Nanomedicines for cancer therapy: Current status, challenges and future prospects. Ther. Deliv. 2019, 10, 113–132. [Google Scholar] [CrossRef]
- Miele, E.; Spinelli, G.P.; Miele, E.; Di Fabrizio, E.; Ferretti, E.; Tomao, S.; Gulino, A. Nanoparticle-based delivery of small interfering RNA: Challenges for cancer therapy. Int. J. Nanomed. 2012, 7, 3637–3657. [Google Scholar]
- Cormode, D.P.; Naha, P.C.; Fayad, Z.A. Nanoparticle contrast agents for computed tomography: A focus on micelles. Contrast Media Mol. Imaging 2014, 9, 37–52. [Google Scholar] [CrossRef]
- Ozpolat, B.; Sood, A.K.; Lopez-Berestein, G. Liposomal siRNA nanocarriers for cancer therapy. Adv. Drug Deliv. Rev. 2014, 66, 110–116. [Google Scholar] [CrossRef]
- Rama, A.R.; Jimenez-Lopez, J.; Cabeza, L.; Jimenez-Luna, C.; Leiva, M.C.; Perazzoli, G.; Hernandez, R.; Zafra, I.; Ortiz, R.; Melguizo, C.; et al. Last Advances in Nanocarriers-Based Drug Delivery Systems for Colorectal Cancer. Curr. Drug Deliv. 2016, 13, 830–838. [Google Scholar] [CrossRef]
- Alavi, M.; Hamidi, M. Passive and active targeting in cancer therapy by liposomes and lipid nanoparticles. Drug Metab. Pers. Ther. 2019, 34. [Google Scholar] [CrossRef]
- Yingchoncharoen, P.; Kalinowski, D.S.; Richardson, D.R. Lipid-Based Drug Delivery Systems in Cancer Therapy: What Is Available and What Is Yet to Come. Pharmacol. Rev. 2016, 68, 701–787. [Google Scholar] [CrossRef]
- Gogoi, M.; Kumar, N.; Patra, S. 27—Multifunctional Magnetic Liposomes for Cancer Imaging and Therapeutic Applications. In Nanoarchitectonics for Smart Delivery and Drug Targeting; Holban, A.M., Grumezescu, A.M., Eds.; William Andrew Publishing: Norwich, NY, USA, 2016; pp. 743–782. ISBN 978-0-323-47347-7. [Google Scholar]
- Kunjachan, S.; Ehling, J.; Storm, G.; Kiessling, F.; Lammers, T. Noninvasive Imaging of Nanomedicines and Nanotheranostics: Principles, Progress, and Prospects. Chem. Rev. 2015, 115, 10907–10937. [Google Scholar] [CrossRef]
- Hardiansyah, A.; Huang, L.-Y.; Yang, M.-C.; Liu, T.-Y.; Tsai, S.-C.; Yang, C.-Y.; Kuo, C.-Y.; Chan, T.-Y.; Zou, H.-M.; Lian, W.-N.; et al. Magnetic liposomes for colorectal cancer cells therapy by high-frequency magnetic field treatment. Nanoscale Res. Lett. 2014, 9, 497. [Google Scholar] [CrossRef]
- Park, J.-H.; Cho, H.-J.; Yoon, H.Y.; Yoon, I.-S.; Ko, S.-H.; Shim, J.-S.; Cho, J.-H.; Park, J.H.; Kim, K.; Kwon, I.C.; et al. Hyaluronic acid derivative-coated nanohybrid liposomes for cancer imaging and drug delivery. J. Control. Release 2014, 174, 98–108. [Google Scholar] [CrossRef]
- Ninomiya, K.; Kawabata, S.; Tashita, H.; Shimizu, N. Ultrasound-mediated drug delivery using liposomes modified with a thermosensitive polymer. Ultrason. Sonochem. 2014, 21, 310–316. [Google Scholar] [CrossRef]
- Legut, M.; Lipka, D.; Filipczak, N.; Piwoni, A.; Kozubek, A.; Gubernator, J. Anacardic acid enhances the anticancer activity of liposomal mitoxantrone towards melanoma cell lines-in vitro studies. Int. J. Nanomed. 2014, 9, 653–668. [Google Scholar]
- Yan, L.; Crayton, S.H.; Thawani, J.P.; Amirshaghaghi, A.; Tsourkas, A.; Cheng, Z. A pH-Responsive Drug-Delivery Platform Based on Glycol Chitosan-Coated Liposomes. Small 2015, 11, 4870–4874. [Google Scholar] [CrossRef]
- Ramadass, S.K.; Anantharaman, N.V.; Subramanian, S.; Sivasubramanian, S.; Madhan, B. Paclitaxel/epigallocatechin gallate coloaded liposome: A synergistic delivery to control the invasiveness of MDA-MB-231 breast cancer cells. Colloids Surf. B Biointerfaces 2015, 125, 65–72. [Google Scholar] [CrossRef]
- Ali, S.; Sheikh, S.; Ahmad, A.; Ahmad, M.; Chen, P.; Paithankar, M.; Choundhury, K.; Makadia, R.; Kumar, A.; Velavan, K.; et al. Bioequivalence Study of Pegylated Doxorubicin Hydrochloride Liposome (PEGADRIA) and DOXIL® in Ovarian Cancer Patients: Physicochemical Characterization and Pre-clinical Studies. J. Nanomed. Nanotechnol. 2016, 7, 2. [Google Scholar]
- Meng, J.; Guo, F.; Xu, H.; Liang, W.; Wang, C.; Yang, X.-D. Combination Therapy using Co-encapsulated Resveratrol and Paclitaxel in Liposomes for Drug Resistance Reversal in Breast Cancer Cells in vivo. Sci. Rep. 2016, 6, 22390. [Google Scholar] [CrossRef]
- Xiao, Y.; Liu, Y.; Yang, S.; Zhang, B.; Wang, T.; Jiang, D.; Zhang, J.; Yu, D.; Zhang, N. Sorafenib and gadolinium co-loaded liposomes for drug delivery and MRI-guided HCC treatment. Colloids Surf. B Biointerfaces 2016, 141, 83–92. [Google Scholar] [CrossRef]
- Lakkadwala, S.; Singh, J. Dual Functionalized 5-Fluorouracil Liposomes as Highly Efficient Nanomedicine for Glioblastoma Treatment as Assessed in an In Vitro Brain Tumor Model. J. Pharm. Sci. 2018, 107, 2902–2913. [Google Scholar] [CrossRef]
- Deshpande, P.; Jhaveri, A.; Pattni, B.; Biswas, S.; Torchilin, V. Transferrin and octaarginine modified dual-functional liposomes with improved cancer cell targeting and enhanced intracellular delivery for the treatment of ovarian cancer. Drug Deliv. 2018, 25, 517–532. [Google Scholar] [CrossRef]
- Sesarman, A.; Tefas, L.; Sylvester, B.; Licarete, E.; Rauca, V.; Luput, L.; Patras, L.; Porav, S.; Banciu, M.; Porfire, A. Co-delivery of curcumin and doxorubicin in PEGylated liposomes favored the antineoplastic C26 murine colon carcinoma microenvironment. Drug Deliv. Transl. Res. 2019, 9, 260–272. [Google Scholar] [CrossRef]
- Tian, Y.; Zhang, H.; Qin, Y.; Li, D.; Liu, Y.; Wang, H.; Gan, L. Overcoming drug-resistant lung cancer by paclitaxel-loaded hyaluronic acid-coated liposomes targeted to mitochondria. Drug Dev. Ind. Pharm. 2018, 44, 2071–2082. [Google Scholar] [CrossRef] [PubMed]
- Mydin, R.B.S.M.N.; Moshawih, S. Nanoparticles in Nanomedicine Application: Lipid-Based Nanoparticles and Their Safety Concerns. In Nanotechnology: Applications in Energy, Drug and Food; Springer: Cham, Switzerland, 2007; pp. 227–232. [Google Scholar]
- Rajabi, M.; Mousa, S.A. Lipid Nanoparticles and their Application in Nanomedicine. Curr. Pharm. Biotechnol. 2016, 17, 662–672. [Google Scholar] [CrossRef] [PubMed]
- Pindiprolu, S.K.S.S.; Chintamaneni, P.K.; Krishnamurthy, P.T.; Ratna Sree Ganapathineedi, K. Formulation-optimization of solid lipid nanocarrier system of STAT3 inhibitor to improve its activity in triple negative breast cancer cells. Drug Dev. Ind. Pharm. 2018, 45, 304–313. [Google Scholar] [CrossRef]
- Eskiler, G.G. Synthetically Lethal BMN 673 (Talazoparib) Loaded Solid Lipid Nanoparticles for BRCA1 Mutant Triple Negative Breast Cancer. Pharm. Res. 2018, 35, 218. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, R.R.; Carrião, M.S.; Pacheco, M.T.; Branquinho, L.C.; de Souza, A.L.R.; Bakuzis, A.F.; Lima, E.M. Triggered release of paclitaxel from magnetic solid lipid nanoparticles by magnetic hyperthermia. Mater. Sci. Eng. C 2018, 92, 547–553. [Google Scholar] [CrossRef] [PubMed]
- Zielińska, A.; Martins-Gomes, C.; Ferreira, N.R.; Silva, A.M.; Nowak, I.; Souto, E.B. Anti-inflammatory and anti-cancer activity of citral: Optimization of citral-loaded solid lipid nanoparticles (SLN) using experimental factorial design and LUMiSizer®. Int. J. Pharm. 2018, 553, 428–440. [Google Scholar] [CrossRef]
- Wang, W.; Zhang, L.; Chen, T.; Guo, W.; Bao, X.; Wang, D.; Ren, B.; Wang, H.; Li, Y.; Wang, Y.; et al. Anticancer effects of resveratrol-loaded solid lipid nanoparticles on human breast cancer cells. Molecules 2017, 22, 1814. [Google Scholar] [CrossRef]
- Chirio, D.; Peira, E.; Battaglia, L.; Ferrara, B.; Barge, A.; Sapino, S.; Giordano, S.; Dianzani, C.; Gallarate, M. Lipophilic Prodrug of Floxuridine Loaded into Solid Lipid Nanoparticles: In Vitro Cytotoxicity Studies on Different Human Cancer Cell Lines. J. Nanosci. Nanotechnol. 2018, 18, 556–563. [Google Scholar] [CrossRef]
- Rahiminejad, A.; Dinarvand, R.; Johari, B.; Nodooshan, S.J.; Rashti, A.; Rismani, E.; Mahdaviani, P.; Saltanatpour, Z.; Rahiminejad, S.; Raigani, M.; et al. Preparation and investigation of Indirubin-loaded SLN nanoparticles and their anti-cancer effects on human glioblastoma U87MG cells. Cell Boil. Int. 2018, 43, 2–11. [Google Scholar] [CrossRef]
- Obeid, M.A.; Tate, R.J.; Mullen, A.B.; Ferro, V.A. Lipid-Based Nanoparticles for Cancer Treatment; Elsevier Inc.: Amsterdam, The Netherlands, 2018; ISBN 9780128136874. [Google Scholar]
- Fahmy, U.A. Augmentation of Fluvastatin Cytotoxicity Against Prostate Carcinoma PC3 Cell Line Utilizing Alpha Lipoic–Ellagic Acid Nanostructured Lipid Carrier Formula. AAPS PharmSciTech 2018, 19, 3454–3461. [Google Scholar] [CrossRef]
- Haron, A.S.; Syed Alwi, S.S.; Saiful Yazan, L.; Abd Razak, R.; Ong, Y.S.; Zakarial Ansar, F.H.; Roshini Alexander, H. Cytotoxic Effect of Thymoquinone-Loaded Nanostructured Lipid Carrier (TQ-NLC) on Liver Cancer Cell Integrated with Hepatitis B Genome, Hep3B. Evid.-Based Complement. Altern. Med. 2018, 2018, 1549805. [Google Scholar] [CrossRef]
- Li, J.; Jin, S.; Dong, X.-R.; Han, X.-F.; Wang, M.-Y. Construction of artesunate nanoparticles modified by hyaluronic acid and cell-penetrating peptides and its inhibitory effect on cancer cells in vitro. China J. Chin. Mater. Med. 2018, 43, 3668–3675. [Google Scholar]
- Nahak, P.; Gajbhiye, R.L.; Karmakar, G.; Guha, P.; Roy, B.; Besra, S.E.; Bikov, A.G.; Akentiev, A.V.; Noskov, B.A.; Nag, K.; et al. Orcinol Glucoside Loaded Polymer-Lipid Hybrid Nanostructured Lipid Carriers: Potential Cytotoxic Agents against Gastric, Colon and Hepatoma Carcinoma Cell Lines. Pharm. Res. 2018, 35, 198. [Google Scholar] [CrossRef] [PubMed]
- Wei, Q.; Yang, Q.; Wang, Q.; Sun, C.; Zhu, Y.; Niu, Y.; Yu, J.; Xu, X. Formulation, Characterization, and Pharmacokinetic Studies of 6-Gingerol-Loaded Nanostructured Lipid Carriers. AAPS PharmSciTech 2018, 19, 3661–3669. [Google Scholar] [CrossRef] [PubMed]
- Silva, C.; Pinho, J.; Lopes, J.; Almeida, A.; Gaspar, M.; Reis, C. Current Trends in Cancer Nanotheranostics: Metallic, Polymeric, and Lipid-Based Systems. Pharmaceutics 2019, 11, 22. [Google Scholar] [CrossRef] [PubMed]
- Sharma, G.; Sharma, A.R.; Lee, S.-S.; Bhattacharya, M.; Nam, J.-S.; Chakraborty, C. Advances in nanocarriers enabled brain targeted drug delivery across blood brain barrier. Int. J. Pharm. 2019, 559, 360–372. [Google Scholar] [CrossRef]
- Tapeinos, C.; Battaglini, M.; Ciofani, G. Advances in the design of solid lipid nanoparticles and nanostructured lipid carriers for targeting brain diseases. J. Control. Release 2017, 264, 306–332. [Google Scholar] [CrossRef] [PubMed]
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed]
- Ferlay, J.; Colombet, M.; Soerjomataram, I.; Mathers, C.; Parkin, D.M.; Piñeros, M.; Znaor, A.; Bray, F. Estimating the global cancer incidence and mortality in 2018: GLOBOCAN sources and methods. Int. J. Cancer 2019, 144, 1941–1953. [Google Scholar] [CrossRef]
- Palesh, O.; Scheiber, C.; Kesler, S.; Mustian, K.; Koopman, C.; Schapira, L. Management of side effects during and post-treatment in breast cancer survivors. Breast J. 2018, 24, 167–175. [Google Scholar] [CrossRef]
- Numico, G.; Longo, V.; Courthod, G.; Silvestris, N. Cancer survivorship: Long-term side-effects of anticancer treatments of gastrointestinal cancer. Curr. Opin. Oncol. 2015, 27, 351–357. [Google Scholar] [CrossRef] [PubMed]
- Tang, W.-L.; Tang, W.-H.; Li, S.-D. Cancer theranostic applications of lipid-based nanoparticles. Drug Discov. Today 2018, 23, 1159–1166. [Google Scholar] [CrossRef] [PubMed]
- Raucher, D.; Dragojevic, S.; Ryu, J. Macromolecular Drug Carriers for Targeted Glioblastoma Therapy: Preclinical Studies, Challenges, and Future Perspectives. Front. Oncol. 2018, 8, 624. [Google Scholar] [CrossRef]
- Angelova, A.; Garamus, V.M.; Angelov, B.; Tian, Z.; Li, Y.; Zou, A. Advances in structural design of lipid-based nanoparticle carriers for delivery of macromolecular drugs, phytochemicals and anti-tumor agents. Adv. Colloid Interface Sci. 2017, 249, 331–345. [Google Scholar] [CrossRef]
- Yaari, Z.; da Silva, D.; Zinger, A.; Goldman, E.; Kajal, A.; Tshuva, R.; Barak, E.; Dahan, N.; Hershkovitz, D.; Goldfeder, M.; et al. Theranostic barcoded nanoparticles for personalized cancer medicine. Nat. Commun. 2016, 7, 13325. [Google Scholar] [CrossRef]
- Ding, J.; Feng, M.; Wang, F.; Wang, H.; Guan, W. Targeting effect of PEGylated liposomes modified with the Arg-Gly-Asp sequence on gastric cancer. Oncol. Rep. 2015, 34, 1825–1834. [Google Scholar] [CrossRef]
- Yang, F.; Zheng, Z.; Zheng, L.; Qin, J.; Li, H.; Xue, X.; Gao, J.; Fang, G. SATB1 siRNA-encapsulated immunoliposomes conjugated with CD44 antibodies target and eliminate gastric cancer-initiating cells. Onco Targets Ther. 2018, 11, 6811–6825. [Google Scholar] [CrossRef] [PubMed]
- Wonder, E.; Simón-Gracia, L.; Scodeller, P.; Majzoub, R.N.; Kotamraju, V.R.; Ewert, K.K.; Teesalu, T.; Safinya, C.R. Competition of charge-mediated and specific binding by peptide-tagged cationic liposome–DNA nanoparticles in vitro and in vivo. Biomaterials 2018, 166, 52–63. [Google Scholar] [CrossRef]
- Wang, J.; Zhu, R.; Sun, X.; Zhu, Y.; Liu, H.; Wang, S.-L. Intracellular uptake of etoposide-loaded solid lipid nanoparticles induces an enhancing inhibitory effect on gastric cancer through mitochondria-mediated apoptosis pathway. Int. J. Nanomed. 2014, 9, 3987–3998. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Zhang, Y.; Meng, Y.-P.; Bo, L.-S.; Ke, W.-B. miR-542-3p Appended Sorafenib/All-trans Retinoic Acid (ATRA)-Loaded Lipid Nanoparticles to Enhance the Anticancer Efficacy in Gastric Cancers. Pharm. Res. 2017, 34, 2710–2719. [Google Scholar] [CrossRef]
- Ma, L.; Yang, D.; Li, Z.; Zhang, X.; Pu, L. Co-delivery of paclitaxel and tanespimycin in lipid nanoparticles enhanced anti-gastric-tumor effect in vitro and in vivo. Artif. Cells Nanomed. Biotechnol. 2018, 46, 904–911. [Google Scholar] [CrossRef]
- Jiang, H.; Pei, L.; Liu, N.; Li, J.; Li, Z.; Zhang, S. Etoposide-loaded nanostructured lipid carriers for gastric cancer therapy. Drug Deliv. 2015, 23, 1379–1382. [Google Scholar] [CrossRef]
- Jiang, H.; Geng, D.; Liu, H.; Li, Z.; Cao, J. Co-delivery of etoposide and curcumin by lipid nanoparticulate drug delivery system for the treatment of gastric tumors. Drug Deliv. 2016, 23, 3665–3673. [Google Scholar] [CrossRef]
- Qu, C.-Y.; Zhou, M.; Chen, Y.; Chen, M.; Feng Shen, F.; Xu, L.-M. Engineering of lipid prodrug-based, hyaluronic acid-decorated nanostructured lipid carriers platform for 5-fluorouracil and cisplatin combination gastric cancer therapy. Int. J. Nanomed. 2015, 10, 3911. [Google Scholar]
- Chang, C.-H.; Liu, S.-Y.; Chi, C.-W.; Yu, H.-L.; Chang, T.-J.; Tsai, T.-H.; Lee, T.-W.; Chen, Y.-J. External beam radiotherapy synergizes 188Re-liposome against human esophageal cancer xenograft and modulates 188Re-liposome pharmacokinetics. Int. J. Nanomed. 2015, 10, 3641–3649. [Google Scholar]
- Feng, Y.; Gao, Y.; Wang, D.; Xu, Z.; Sun, W.; Ren, P. Autophagy Inhibitor (LY294002) and 5-fluorouracil (5-FU) Combination-Based Nanoliposome for Enhanced Efficacy Against Esophageal Squamous Cell Carcinoma. Nanoscale Res. Lett. 2018, 13, 325. [Google Scholar] [CrossRef]
- Wang, L.-L.; Huang, S.; Guo, H.-H.; Han, Y.-X.; Zheng, W.-S.; Jiang, J.-D. In situ delivery of thermosensitive gel-mediated 5-fluorouracil microemulsion for the treatment of colorectal cancer. Drug Des. Dev. Ther. 2016, 10, 2855–2867. [Google Scholar] [CrossRef]
- Low, L.E.; Tan, L.T.-H.; Goh, B.-H.; Tey, B.T.; Ong, B.H.; Tang, S.Y. Magnetic cellulose nanocrystal stabilized Pickering emulsions for enhanced bioactive release and human colon cancer therapy. Int. J. Biol. Macromol. 2019, 127, 76–84. [Google Scholar] [CrossRef]
- Ektate, K.; Munteanu, M.C.; Ashar, H.; Malayer, J.; Ranjan, A. Chemo-immunotherapy of colon cancer with focused ultrasound and Salmonella-laden temperature sensitive liposomes (thermobots). Sci. Rep. 2018, 8, 13062. [Google Scholar] [CrossRef]
- Moghimipour, E.; Rezaei, M.; Ramezani, Z.; Kouchak, M.; Amini, M.; Angali, K.A.; Dorkoosh, F.A.; Handali, S. Folic acid-modified liposomal drug delivery strategy for tumor targeting of 5-fluorouracil. Eur. J. Pharm. Sci. 2018, 114, 166–174. [Google Scholar] [CrossRef]
- Kassem, M.A.; El-Sawy, H.S.; Abd-Allah, F.I.; Abdelghany, T.M.; El-Say, K.M. Maximizing the Therapeutic Efficacy of Imatinib Mesylate–Loaded Niosomes on Human Colon Adenocarcinoma Using Box-Behnken Design. J. Pharm. Sci. 2017, 106, 111–122. [Google Scholar] [CrossRef]
- Serini, S.; Cassano, R.; Corsetto, P.A.; Rizzo, A.M.; Calviello, G.; Trombino, S. Omega-3 PUFA Loaded in Resveratrol-Based Solid Lipid Nanoparticles: Physicochemical Properties and Antineoplastic Activities in Human Colorectal Cancer Cells In Vitro. Int. J. Mol. Sci. 2018, 19, 586. [Google Scholar] [CrossRef] [PubMed]
- Shen, M.-Y.; Liu, T.-I.; Yu, T.-W.; Kv, R.; Chiang, W.-H.; Tsai, Y.-C.; Chen, H.-H.; Lin, S.-C.; Chiu, H.-C. Hierarchically targetable polysaccharide-coated solid lipid nanoparticles as an oral chemo/thermotherapy delivery system for local treatment of colon cancer. Biomaterials 2019, 197, 86–100. [Google Scholar] [CrossRef]
- Negi, L.M.; Talegaonkar, S.; Jaggi, M.; Verma, A.K.; Verma, R.; Dobhal, S.; Kumar, V. Surface engineered nanostructured lipid carriers for targeting MDR tumor: Part I. Synthesis, characterization and in vitro investigation. Colloids Surf. B Biointerfaces 2014, 123, 600–609. [Google Scholar] [CrossRef]
- Chirio, D.; Peira, E.; Sapino, S.; Dianzani, C.; Barge, A.; Muntoni, E.; Morel, S.; Gallarate, M. Stearoyl-Chitosan Coated Nanoparticles Obtained by Microemulsion Cold Dilution Technique. Int. J. Mol. Sci. 2018, 19, 3833. [Google Scholar] [CrossRef] [PubMed]
- Abu-Fayyad, A.; Nazzal, S. Gemcitabine-vitamin E conjugates: Synthesis, characterization, entrapment into nanoemulsions, and in-vitro deamination and antitumor activity. Int. J. Pharm. 2017, 528, 463–470. [Google Scholar] [CrossRef] [PubMed]
- Wood, K.; Szewczuk, M.R.; Rousseau, D.; Neufeld, R.J. Oseltamivir phosphate released from injectable Pickering emulsions over an extended term disables human pancreatic cancer cell survival. Oncotarget 2018, 9, 12754. [Google Scholar] [CrossRef] [PubMed]
- Bisht, S.; Schlesinger, M.; Rupp, A.; Schubert, R.; Nolting, J.; Wenzel, J.; Holdenrieder, S.; Brossart, P.; Bendas, G.; Feldmann, G. A liposomal formulation of the synthetic curcumin analog EF24 (Lipo-EF24) inhibits pancreatic cancer progression: Towards future combination therapies. J. Nanobiotechnol. 2016, 14, 57. [Google Scholar] [CrossRef]
- Wei, Y.; Wang, Y.; Xia, D.; Guo, S.; Wang, F.; Zhang, X.; Gan, Y. Thermosensitive Liposomal Codelivery of HSA–Paclitaxel and HSA–Ellagic Acid Complexes for Enhanced Drug Perfusion and Efficacy Against Pancreatic Cancer. ACS Appl. Mater. Interfaces 2017, 9, 25138–25151. [Google Scholar] [CrossRef]
- Glassman, D.C.; Palmaira, R.L.; Covington, C.M.; Desai, A.M.; Ku, G.Y.; Li, J.; Harding, J.J.; Varghese, A.M.; O’Reilly, E.M.; Yu, K.H. Nanoliposomal irinotecan with fluorouracil for the treatment of advanced pancreatic cancer, a single institution experience. BMC Cancer 2018, 18, 693. [Google Scholar] [CrossRef]
- Wang-Gillam, A.; Li, C.-P.; Bodoky, G.; Dean, A.; Shan, Y.-S.; Jameson, G.; Macarulla, T.; Lee, K.-H.; Cunningham, D.; Blanc, J.F.; et al. Nanoliposomal irinotecan with fluorouracil and folinic acid in metastatic pancreatic cancer after previous gemcitabine-based therapy (NAPOLI-1): A global, randomised, open-label, phase 3 trial. Lancet 2016, 387, 545–557. [Google Scholar] [CrossRef]
- Barkat, M.A.; Rizwanullah, M.; Beg, S.; Pottoo, F.H.; Siddiqui, S.; Ahmad, F.J. Paclitaxel-loaded Nanolipidic Carriers with Improved Oral Bioavailability and Anticancer Activity against Human Liver Carcinoma. AAPS PharmSciTech 2019, 20, 87. [Google Scholar]
- Nasr, M.; Ghorab, M.K.; Abdelazem, A. In vitro and in vivo evaluation of cubosomes containing 5-fluorouracil for liver targeting. Acta Pharm. Sin. B 2015, 5, 79–88. [Google Scholar] [CrossRef] [PubMed]
- Grillone, A.; Riva, E.R.; Mondini, A.; Forte, C.; Calucci, L.; Innocenti, C.; de Julian Fernandez, C.; Cappello, V.; Gemmi, M.; Moscato, S.; et al. Active Targeting of Sorafenib: Preparation, Characterization, and In Vitro Testing of Drug-Loaded Magnetic Solid Lipid Nanoparticles. Adv. Healthc. Mater. 2015, 4, 1681–1690. [Google Scholar] [CrossRef]
- Lin, C.-C.; Lin, H.-Y.; Chi, M.-H.; Shen, C.-M.; Chen, H.-W.; Yang, W.-J.; Lee, M.-H. Preparation of curcumin microemulsions with food-grade soybean oil/lecithin and their cytotoxicity on the HepG2 cell line. Food Chem. 2014, 154, 282–290. [Google Scholar] [CrossRef] [PubMed]
- Qu, D.; Liu, M.; Huang, M.; Wang, L.; Chen, Y.; Liu, C.; Liu, Y. Octanoyl galactose ester-modified microemulsion system self-assembled by coix seed components to enhance tumor targeting and hepatoma therapy. Int. J. Nanomed. 2017, 12, 2045–2059. [Google Scholar] [CrossRef]
- Hu, H.; Xiao, C.; Wu, H.; Li, Y.; Zhou, Q.; Tang, Y.; Yu, C.; Yang, X.; Li, Z. Nanocolloidosomes with Selective Drug Release for Active Tumor-Targeted Imaging-Guided Photothermal/Chemo Combination Therapy. Appl. Mater. Interfaces 2017, 9, 42225–42238. [Google Scholar] [CrossRef]
- Zhang, X.; Lin, C.-C.; Chan, W.-K.-N.; Liu, K.-L.; Yang, Z.-J.; Zhang, H.-Q. Augmented Anticancer Effects of Cantharidin with Liposomal Encapsulation: In Vitro and In Vivo Evaluation. Molecules 2017, 22, 1052. [Google Scholar] [CrossRef]
- Chang, M.; Wu, M.; Li, H. Antitumor activities of novel glycyrrhetinic acid-modified curcumin-loaded cationic liposomes in vitro and in H22 tumor-bearing mice. Drug Deliv. 2018, 25, 1984–1995. [Google Scholar] [CrossRef]
- Beg, M.S.; Brenner, A.J.; Sachdev, J.; Borad, M.; Kang, Y.-K.; Stoudemire, J.; Smith, S.; Bader, A.G.; Kim, S.; Hong, D.S. Phase I study of MRX34, a liposomal miR-34a mimic, administered twice weekly in patients with advanced solid tumors. Investig. New Drugs 2017, 35, 180–188. [Google Scholar] [CrossRef]
- Sasmita, A.O.; Wong, Y.P.; Ling, A.P.K. Biomarkers and therapeutic advances in glioblastoma multiforme. Asia Pac. J. Clin. Oncol. 2018, 14, 40–51. [Google Scholar] [CrossRef]
- Kumar, A.; Ahuja, A.; Ali, J.; Baboota, S. Curcumin-loaded lipid nanocarrier for improving bioavailability, stability and cytotoxicity against malignant glioma cells. Drug Deliv. 2016, 23, 214–229. [Google Scholar] [CrossRef]
- Shinde, R.L.; Devarajan, P.V. Docosahexaenoic acid-mediated, targeted and sustained brain delivery of curcumin microemulsion. Drug Deliv. 2017, 24, 152–161. [Google Scholar] [CrossRef]
- Lakkadwala, S.; Singh, J. Co-delivery of doxorubicin and erlotinib through liposomal nanoparticles for glioblastoma tumor regression using an in vitro brain tumor model. Colloids Surf. B Biointerfaces 2019, 173, 27–35. [Google Scholar] [CrossRef]
- Kadari, A.; Pooja, D.; Gora, R.H.; Gudem, S.; Kolapalli, V.R.M.; Kulhari, H.; Sistla, R. Design of multifunctional peptide collaborated and docetaxel loaded lipid nanoparticles for antiglioma therapy. Eur. J. Pharm. Biopharm. 2018, 132, 168–179. [Google Scholar] [CrossRef]
- Carbone, C.; Campisi, A.; Musumeci, T.; Raciti, G.; Bonfanti, R.; Puglisi, G. FA-loaded lipid drug delivery systems: Preparation, characterization and biological studies. Eur. J. Pharm. Sci. 2014, 52, 12–20. [Google Scholar] [CrossRef]
- Zhang, J.; Xiao, X.; Zhu, J.; Gao, Z.; Lai, X.; Zhu, X.; Mao, G. Lactoferrin- and RGD-comodified, temozolomide and vincristine-coloaded nanostructured lipid carriers for gliomatosis cerebri combination therapy. Int. J. Nanomed. 2018, 13, 3039–3051. [Google Scholar] [CrossRef]
- Papachristodoulou, A.; Signorell, R.D.; Werner, B.; Brambilla, D.; Luciani, P.; Cavusoglu, M.; Grandjean, J.; Silginer, M.; Rudin, M.; Martin, E.; et al. Chemotherapy sensitization of glioblastoma by focused ultrasound-mediated delivery of therapeutic liposomes. J. Control. Release 2019, 295, 130–139. [Google Scholar] [CrossRef]
- Chastagner, P.; Devictor, B.; Geoerger, B.; Aerts, I.; Leblond, P.; Frappaz, D.; Gentet, J.-C.; Bracard, S.; André, N. Phase I study of non-pegylated liposomal doxorubicin in children with recurrent/refractory high-grade glioma. Cancer Chemother. Pharmacol. 2015, 76, 425–432. [Google Scholar] [CrossRef] [PubMed]
- Clarke, J.L.; Molinaro, A.M.; Cabrera, J.R.; DeSilva, A.A.; Rabbitt, J.E.; Prey, J.; Drummond, D.C.; Kim, J.; Noble, C.; Fitzgerald, J.B.; et al. A phase 1 trial of intravenous liposomal irinotecan in patients with recurrent high-grade glioma. Cancer Chemother. Pharmacol. 2017, 79, 603–610. [Google Scholar] [CrossRef]
- Jyoti, K.; Kaur, K.; Pandey, R.S.; Jain, U.K.; Chandra, R.; Madan, J. Inhalable nanostructured lipid particles of 9-bromo-noscapine, a tubulin-binding cytotoxic agent: In vitro and in vivo studies. J. Colloid Interface Sci. 2015, 445, 219–230. [Google Scholar] [CrossRef]
- Sun, L.; Wan, K.; Hu, X.; Zhang, Y.; Yan, Z.; Feng, J.; Zhang, J. Functional nanoemulsion-hybrid lipid nanocarriers enhance the bioavailability and anti-cancer activity of lipophilic diferuloylmethane. Nanotechnology 2016, 27, 085102. [Google Scholar] [CrossRef]
- Wan, K.; Sun, L.; Hu, X.; Yan, Z.; Zhang, Y.; Zhang, X.; Zhang, J. Novel nanoemulsion based lipid nanosystems for favorable in vitro and in vivo characteristics of curcumin. Int. J. Pharm. 2016, 504, 80–88. [Google Scholar] [CrossRef]
- Asmawi, A.A.; Salim, N.; Ngan, C.L.; Ahmad, H.; Abdulmalek, E.; Masarudin, M.J.; Abdul Rahman, M.B. Excipient selection and aerodynamic characterization of nebulized lipid-based nanoemulsion loaded with docetaxel for lung cancer treatment. Drug Deliv. Transl. Res. 2018, 9, 543–554. [Google Scholar] [CrossRef]
- Chen, T.; Gong, T.; Zhao, T.; Fu, Y.; Zhang, Z.; Gong, T. A comparison study between lycobetaine-loaded nanoemulsion and liposome using nRGD as therapeutic adjuvant for lung cancer therapy. Eur. J. Pharm. Sci. 2018, 111, 293–302. [Google Scholar] [CrossRef]
- Shi, S.; Han, L.; Deng, L.; Zhang, Y.; Shen, H.; Gong, T.; Zhang, Z.; Sun, X. Dual drugs (microRNA-34a and paclitaxel)-loaded functional solid lipid nanoparticles for synergistic cancer cell suppression. J. Control. Release 2014, 194, 228–237. [Google Scholar] [CrossRef]
- Li, S.; Wang, L.; Li, N.; Liu, Y.; Su, H. Combination lung cancer chemotherapy: Design of a pH-sensitive transferrin-PEG-Hz-lipid conjugate for the co-delivery of docetaxel and baicalin. Biomed. Pharmacother. 2017, 95, 548–555. [Google Scholar] [CrossRef]
- Liang, Y.; Tian, B.; Zhang, J.; Li, K.; Wang, L.; Han, J.; Wu, Z. Tumor-targeted polymeric nanostructured lipid carriers with precise ratiometric control over dual-drug loading for combination therapy in non-small-cell lung cancer. Int. J. Nanomed. 2017, 12, 1699–1715. [Google Scholar] [CrossRef]
- Lu, B.; Sun, L.; Yan, X.; Ai, Z.; Xu, J. Intratumoral chemotherapy with paclitaxel liposome combined with systemic chemotherapy: A new method of neoadjuvant chemotherapy for stage III unresectable non-small cell lung cancer. Med. Oncol. 2015, 32, 345. [Google Scholar] [CrossRef]
- Cao, X.; Luo, J.; Gong, T.; Zhang, Z.-R.; Sun, X.; Fu, Y. Coencapsulated doxorubicin and bromotetrandrine lipid nanoemulsions in reversing multidrug resistance in breast cancer in vitro and in vivo. Mol. Pharm. 2015, 12, 274–286. [Google Scholar] [CrossRef]
- Rocca, A.; Cecconetto, L.; Passardi, A.; Melegari, E.; Andreis, D.; Monti, M.; Maltoni, R.; Sarti, S.; Pietri, E.; Schirone, A.; et al. Phase Ib dose-finding trial of lapatinib plus pegylated liposomal doxorubicin in advanced HER2-positive breast cancer. Cancer Chemother. Pharmacol. 2017, 79, 863–871. [Google Scholar] [CrossRef] [PubMed]
- Lorusso, V.; Giotta, F.; Bordonaro, R.; Maiello, E.; Del Prete, S.; Gebbia, V.; Filipelli, G.; Pisconti, S.; Cinieri, S.; Romito, S.; et al. Non-pegylated liposome-encapsulated doxorubicin citrate plus cyclophosphamide or vinorelbine in metastatic breast cancer not previously treated with chemotherapy: A multicenter phase III study. Int. J. Oncol. 2014, 45, 2137–2142. [Google Scholar] [CrossRef] [PubMed]
- Behroozah, A.; Tabrizi, M.M.; Kazemi, S.M.; Choupani, E.; Kabiri, N.; Ilbeigi, D.; Nasab, A.H.; Khiyavi, A.A.; Kurdi, A.A.S. Evaluation the Anti-Cancer Effect of PEGylated Nano-Niosomal Gingerol, on Breast Cancer Cell lines (T47D), In-Vitro. Asian Pac. J. Cancer Prev. 2018, 19, 645. [Google Scholar]
- Salem, H.F.; Kharshoum, R.M.; Abo El-Ela, F.I.; Abdellatif, K.R.A. Evaluation and optimization of pH-responsive niosomes as a carrier for efficient treatment of breast cancer. Drug Deliv. Transl. Res. 2018, 8, 633–644. [Google Scholar] [CrossRef] [PubMed]
- Shaker, D.S.; Shaker, M.A.; Hanafy, M.S. Cellular uptake, cytotoxicity and in-vivo evaluation of Tamoxifen citrate loaded niosomes. Int. J. Pharm. 2015, 493, 285–294. [Google Scholar] [CrossRef] [PubMed]
- Davarpanah, F.; Khalili Yazdi, A.; Barani, M.; Mirzaei, M.; Torkzadeh-Mahani, M. Magnetic delivery of antitumor carboplatin by using PEGylated-Niosomes. DARU J. Pharm. Sci. 2018, 26, 57–64. [Google Scholar] [CrossRef]
- Amiri, B.; Ebrahimi-Far, M.; Saffari, Z.; Akbarzadeh, A.; Soleimani, E.; Chiani, M. Preparation, Characterization and Cytotoxicity of Silibinin-Containing Nanoniosomes in T47D Human Breast Carcinoma Cells. Asian Pac. J. Cancer Prev. 2016, 17, 3835–3838. [Google Scholar]
- Yazdi Rouholamini, S.E.; Moghassemi, S.; Maharat, Z.; Hakamivala, A.; Kashanian, S.; Omidfar, K. Effect of silibinin-loaded nano-niosomal coated with trimethyl chitosan on miRNAs expression in 2D and 3D models of T47D breast cancer cell line. Artif. Cells Nanomed. Biotechnol. 2018, 46, 524–535. [Google Scholar] [CrossRef]
- Barani, M.; Mirzaei, M.; Torkzadeh-Mahani, M.; Nematollahi, M.H. Lawsone-loaded Niosome and its antitumor activity in MCF-7 breast Cancer cell line: A Nano-herbal treatment for Cancer. DARU J. Pharm. Sci. 2018, 26, 11–17. [Google Scholar] [CrossRef]
- Tila, D.; Yazdani-Arazi, S.N.; Ghanbarzadeh, S.; Arami, S.; Pourmoazzen, Z. pH-sensitive, polymer modified, plasma stable niosomes: Promising carriers for anti-cancer drugs. EXCLI J. 2015, 14, 21–32. [Google Scholar]
- Rajput, S.; Puvvada, N.; Kumar, B.N.P.; Sarkar, S.; Konar, S.; Bharti, R.; Dey, G.; Mazumdar, A.; Pathak, A.; Fisher, P.B.; et al. Overcoming Akt Induced Therapeutic Resistance in Breast Cancer through siRNA and Thymoquinone Encapsulated Multilamellar Gold Niosomes. Mol. Pharm. 2015, 12, 4214–4225. [Google Scholar] [CrossRef]
- Alavi, S.E.; Mansouri, H.; Esfahani, M.K.M.; Movahedi, F.; Akbarzadeh, A.; Chiani, M. Archaeosome: As new drug carrier for delivery of Paclitaxel to breast cancer. Indian J. Clin. Biochem. 2014, 29, 150–153. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.; Li, J.-C.; Zhu, J.-X.; Zhu, N.; Zhang, H.-M.; Liang, L.; Sun, L. Folic Acid-Targeted Etoposide Cubosomes for Theranostic Application of Cancer Cell Imaging and Therapy. Med. Sci. Monit. 2017, 23, 2426–2435. [Google Scholar] [CrossRef] [PubMed]
- Yu, D.; Li, W.; Zhang, Y.; Zhang, B. Anti-tumor efficiency of paclitaxel and DNA when co-delivered by pH responsive ligand modified nanocarriers for breast cancer treatment. Biomed. Pharmacother. 2016, 83, 1428–1435. [Google Scholar] [CrossRef] [PubMed]
- Garg, N.K.; Singh, B.; Jain, A.; Nirbhavane, P.; Sharma, R.; Tyagi, R.K.; Kushwah, V.; Jain, S.; Katare, O.P. Fucose decorated solid-lipid nanocarriers mediate efficient delivery of methotrexate in breast cancer therapeutics. Colloids Surf. B Biointerfaces 2016, 146, 114–126. [Google Scholar] [CrossRef]
- Liang, T.; Yao, Z.; Ding, J.; Min, Q.; Jiang, L.; Zhu, J.-J. Cascaded Aptamers-Governed Multistage Drug-Delivery System Based on Biodegradable Envelope-Type Nanovehicle for Targeted Therapy of HER2-Overexpressing Breast Cancer. ACS Appl. Mater. Interfaces 2018, 10, 34050–34059. [Google Scholar] [CrossRef]
- Li, X.; Jia, X.; Niu, H. Nanostructured lipid carriers co-delivering lapachone and doxorubicin for overcoming multidrug resistance in breast cancer therapy. Int. J. Nanomed. 2018, 13, 4107–4119. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, G.; El Sadda, R.; Botchkina, G.; Ojima, I.; Egan, J.; Amiji, M. Nanoemulsion Formulation of a Novel Taxoid DHA-SBT-1214 Inhibits Prostate Cancer Stem Cell-Induced Tumor Growth. Cancer Lett. 2017, 406, 71–80. [Google Scholar] [CrossRef]
- Chen, B.-H.; Tsai, Y.J. Preparation of catechin extracts and nanoemulsions from green tea leaf waste and their inhibition effect on prostate cancer cell PC-3. Int. J. Nanomed. 2016, 11, 1907. [Google Scholar] [CrossRef]
- Nassir, A.M.; Ibrahim, I.A.A.; Md, S.; Waris, M.; Ain, M.R.; Ahmad, I.; Shahzad, N. Surface functionalized folate targeted oleuropein nano-liposomes for prostate tumor targeting: Invitro and invivo activity. Life Sci. 2019, 220, 136–146. [Google Scholar] [CrossRef]
- Hua, H.; Zhang, N.; Liu, D.; Song, L.; Liu, T.; Li, S.; Zhao, Y. Multifunctional gold nanorods and docetaxel-encapsulated liposomes for combined thermo- and chemotherapy. Int. J. Nanomed. 2017, 12, 7869–7884. [Google Scholar] [CrossRef]
- Saber, M.M.; Al-Mahallawi, A.M.; Nassar, N.N.; Stork, B.; Shouman, S.A. Targeting colorectal cancer cell metabolism through development of cisplatin and metformin nano-cubosomes. BMC Cancer 2018, 18, 822. [Google Scholar] [CrossRef] [PubMed]
- Norouzi-Barough, L.; Sarookhani, M.R.; Sharifi, M.; Moghbelinejad, S.; Jangjoo, S.; Salehi, R. Molecular mechanisms of drug resistance in ovarian cancer. J. Cell. Physiol. 2018, 233, 4546–4562. [Google Scholar] [CrossRef] [PubMed]
- Pan, S.-T.; Li, Z.-L.; He, Z.-X.; Qiu, J.-X.; Zhou, S.-F. Molecular mechanisms for tumour resistance to chemotherapy. Clin. Exp. Pharmacol. Physiol. 2016, 43, 723–737. [Google Scholar] [CrossRef]
- Baek, J.-S.; Cho, C.-W. A multifunctional lipid nanoparticle for co-delivery of paclitaxel and curcumin for targeted delivery and enhanced cytotoxicity in multidrug resistant breast cancer cells. Oncotarget 2017, 8, 30369. [Google Scholar] [CrossRef]
- Vicente-Dueñas, C.; Romero-Camarero, I.; Cobaleda, C.; Sánchez-García, I. Function of oncogenes in cancer development: A changing paradigm. EMBO J. 2013, 32, 1502–1513. [Google Scholar] [CrossRef]
- Zhao, Y.; Wang, W.; Guo, S.; Wang, Y.; Miao, L.; Xiong, Y.; Huang, L. PolyMetformin combines carrier and anticancer activities for in vivo siRNA delivery. Nat. Commun. 2016, 7, 11822. [Google Scholar] [CrossRef]
- Prieto-Vila, M.; Takahashi, R.; Usuba, W.; Kohama, I.; Ochiya, T. Drug Resistance Driven by Cancer Stem Cells and Their Niche. Int. J. Mol. Sci. 2017, 18, 2574. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Xiao, Y.; Lin, H.-P.; Reichel, D.; Bae, Y.; Lee, E.Y.; Jiang, Y.; Huang, X.; Yang, C.; Wang, Z. In vivo β-catenin attenuation by the integrin α5-targeting nano-delivery strategy suppresses triple negative breast cancer stemness and metastasis. Biomaterials 2019, 188, 160–172. [Google Scholar] [CrossRef] [PubMed]

| Cancer Type | NP Type | Target | Drug or Cargo | SS | Detection Method | Combined Therapy | Status | Reference |
|---|---|---|---|---|---|---|---|---|
| Gastric | Liposome | Integrin α5β1 | RGD Peptide & ICG | PEG | CoM, FC & IVV imaging | IVT + IVV | [49] | |
| Liposome | SATB1 | SATB1 siRNA | CD44 antibody | FC | IVT | [50] | ||
| Liposome | Affinity ligand | Plasmid DNA & Tumor-homing peptides | PEG | FC& IVV imaging | IVT + IVV | [51] | ||
| SLN | TopII | Etoposide (VP16) | IVT | [52] | ||||
| SLN | AEG-1, Ras/Raf/Mek/Erk cascade pathway & ReA receptors | miR-542-3p & Sorafenib | PEG | All-trans ReA | IVT | [53] | ||
| SLN | Tubulin & Hsp90 | PTX | 17-AAG | IVT + IVV | [54] | |||
| NLC | TopII | Etoposide (VP16) | IVT + IVV | [55] | ||||
| NLC | TopII | Etoposide (VP16) | CUR | IVT + IVV | [56] | |||
| NLC | Thymidylate synthase | 5-FU-stearic acid prodrug | Hyaluronic acid | Cisplatin | IVT + IVV | [57] | ||
| Esophageal | Liposome | Rhenium 188 | NanoSPECT/CT scanner system | IVT + IVV | [58] | |||
| Liposome | Thymidylate synthase | LY294002 | PEG | 5-FU | IVT + IVV | [59] | ||
| Colorectal | ME | Thymidylate synthase | 5-FU | IVT + IVV | [60] | |||
| PE | CUR | IVT | [61] | |||||
| Liposome | TopII | DOX | attenuated Salmonella | HIFU | IVT + IVV | [62] | ||
| Liposome | Thymidylate synthase | 5-FU | FoA | IVT + IVV | [63] | |||
| Niosome | Protein-tyrosine kinase | IM | IVT | [64] | ||||
| Cubosome | Cisplatin | Metformin | IVT | [125] | ||||
| SLN | Omega-3 PUFA—DHA & Linoleic acid (LNA) | IVT | [65] | |||||
| SLN | TopII | DOX | FoA & Dextran | SPIONs/high-frequency magnetic field (HFMF) | IVT + IVV | [66] | ||
| NLC | TopI | IRI | Hyaluronic acid | IVT | [67] | |||
| Pancreatic | ME | Cur | Stearoyl chitosan | Optical microscopy SEM | IVT + IVV | [68] | ||
| NE | Gemcitabine | γ-tocotrienol isomer of vitamin E | IVT | [69] | ||||
| PE | Neu1 | OsP | IVT | [70] | ||||
| Liposome | NF-kappaB | EF24 | PEG | TEM | Gemcitabine | IVT + IVV | [71] | |
| Liposome | HSA-PTX & HSA-Ellagic acid | Inverted FM | IVT + IVV | [72] | ||||
| Liposome | nal-IRI & 5-FU/leucovorin | CT Phase III | [74] | |||||
| Liver | ME | Cur | FM | IVT | [78] | |||
| ME | Coix seed components | Octanoyl galactose ester | In vivo near-infrared imaging system | IVT + IVV | [79] | |||
| PE | TopII | DOX & ICG | Galactose | NIR fluorescence imaging | NIR laser irradiation | IVT + IVV | [80] | |
| Liposome | Cantharidin | PEG | IVT + IVV | [81] | ||||
| Liposome | Cur | Glycyrrhetinic acid | IVT + IVV | [82] | ||||
| Liposome | miR-34a mimic | CT—Phase 1 | [83] | |||||
| Cubosome | Thymidylate synthase | 5-FU | IVT + IVV | [76] | ||||
| SLN | Sorafenib | SPIONs | IVT | [77] | ||||
| NLC | Tubulin | PTX | IVT + IVV | [75] | ||||
| GBM | ME | Cur | DHA-rich oil | IVT + IVV | [86] | |||
| NE | Cur | IVT + IVV | [85] | |||||
| Liposome | Tf receptors (TfR) | DOX | Tf & PFV | Erlotinib | IVT | [87] | ||
| Liposome | MGMT gene | O6BTG derivative | LIFU | [91] | ||||
| Liposome | Myocet® (DOX) | CT—Phase I | [92] | |||||
| Liposome | TopI | nal-IRI | CT—Phase I | [93] | ||||
| SLN | LRP-1 | Docetaxel | Angiopep-2 | Real time fluorescence imaging | IVT + IVV | [88] | ||
| NLC | Ferulic acid | IVT | [89] | |||||
| NLC | TMZ | Lactoferrin & RGD peptide | Vincristine | IVT + IVV | [90] | |||
| Lung | NE | 9-bromo-noscapine | Spray dried lactose | IVT + IVV | [94] | |||
| NE | Lipophilic diferuloylmethane | IVT + IVV | [95] | |||||
| NE | Cur | Tween80 & LipodS75 | IVT + IVV | [96] | ||||
| NE | Tubulin | Docetaxel | IVT | [97] | ||||
| NE | Lycobetaine & oleic acid (OA) | PEG-lecithin & nRGD peptide | IVT + IVV | [98] | ||||
| Liposome | Tubulin | PTX | Carboplatin & Gemcitabine | CT Phase III | [102] | |||
| SLN | Tubulin | PTX | miR-34a | FC& CoM | IVT + IVV | [99] | ||
| SLN | Tubulin & Tf receptors (TfR) | Docetaxel & Baicalin | PEG, Tf & Hydrazone | IVT + IVV | [100] | |||
| NLC | Tubulin & Glucose receptor | Gemcitabine & PTX | Glucose receptor-targeting ligand | FC& CoM | IVT | [101] | ||
| Breast | NE | TopII& P-gp | DOX & W198 | Whole body fluorescence imaging | IVT + IVV | [103] | ||
| Liposome | DOX | PEG | Lapatinib | CT Phase Ib | [104] | |||
| Liposome | Myocet© | Cyclophosphamide (MC) or vinorelbine (MV) | CT Phase III | [105] | ||||
| Niosome | Tamoxifen citrate (TXC) | IVT + IVV | [108] | |||||
| Niosome | TQ | Fluorescence Imaging & NIR | Akt-siRNA | IVT + IVV | [114] | |||
| Archaeosomes | PTX | IVT | [115] | |||||
| Cubosomes | TopII | Etoposide (VP16) | FoA-P407 | IVT + IVV | [116] | |||
| SLN | PTX & DNA | Hyaluronic acid | IVT + IVV | [117] | ||||
| SLN | Methotrexate | Fucose | IVT + IVV | [118] | ||||
| NLC | HER2+ | ATP aptamer-EGCG-protamine sulfate | HER2 aptamer | IVT + IVV | [119] | |||
| NLC | TopII & NQO-1 | Lapachone & DOX | Confocal laser scanning microscopy | IVT + IVV | [120] | |||
| Prostate | NE | Taxoid prodrug | Omega-3 fatty acid | CoM | IVT + IVV | [121] | ||
| NE | Catechin extract | TEM | IVT | [122] | ||||
| Liposome | Oleuropein | PEG | CoM | IVT + IVV | [123] | |||
| Liposome | LRP-1 | Docetaxel | PEG | FM | IVT + IVV | [124] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
García-Pinel, B.; Porras-Alcalá, C.; Ortega-Rodríguez, A.; Sarabia, F.; Prados, J.; Melguizo, C.; López-Romero, J.M. Lipid-Based Nanoparticles: Application and Recent Advances in Cancer Treatment. Nanomaterials 2019, 9, 638. https://doi.org/10.3390/nano9040638
García-Pinel B, Porras-Alcalá C, Ortega-Rodríguez A, Sarabia F, Prados J, Melguizo C, López-Romero JM. Lipid-Based Nanoparticles: Application and Recent Advances in Cancer Treatment. Nanomaterials. 2019; 9(4):638. https://doi.org/10.3390/nano9040638
Chicago/Turabian StyleGarcía-Pinel, Beatriz, Cristina Porras-Alcalá, Alicia Ortega-Rodríguez, Francisco Sarabia, Jose Prados, Consolación Melguizo, and Juan M. López-Romero. 2019. "Lipid-Based Nanoparticles: Application and Recent Advances in Cancer Treatment" Nanomaterials 9, no. 4: 638. https://doi.org/10.3390/nano9040638
APA StyleGarcía-Pinel, B., Porras-Alcalá, C., Ortega-Rodríguez, A., Sarabia, F., Prados, J., Melguizo, C., & López-Romero, J. M. (2019). Lipid-Based Nanoparticles: Application and Recent Advances in Cancer Treatment. Nanomaterials, 9(4), 638. https://doi.org/10.3390/nano9040638






