1. Introduction
With the emergence of big data, cloud computing, the internet of things and artificial intelligence technology, the amount of information that needs to be stored and analyzed is exploding. However, as the mainstream in current semiconductor memory market, flash memory has encountered serious challenges to further scaling down [
1]. Hence both industry and academia have exerted great efforts to explore appropriate candidates for the next-generation non-volatile memory (NVM) [
2,
3]. Due to the simple device structure, low operation voltages, fast operation speed, excellent scaling potential, multilevel data storage and uncomplicated fabrication process which is compatible with complementary metal-oxide-semiconductor (CMOS) technology, resistive random access memory (RRAM) with a simple metal-insulator-metal (MIM) structure is considered to be one of the most promising candidates for the next generation NVM [
4,
5]. RRAM can store information in the form of resistance states which rely on the history of applied voltage and can be reversibly switched. The resistive switching (RS) modes in RRAM devices can either be unipolar and bipolar. During bipolar RS mode, the devices transform from high-resistance state (HRS) to low-resistance state (LRS) at one polarity (defined as SET) and regain the original resistive state at reverse polarity (defined as RESET), whereas for the unipolar RS mode a change in polarity is not required [
6]. A filamentary switching mechanism has been proposed to explain the RS phenomenon by the formation/rupture of nanoscale conductive filament (CF) [
7]. As research continues, several well-accepted filamentary switching mechanisms have been raised to understand the underlying principle of the RS behaviors. They are respectively electrochemical metallization mechanism (ECM), thermochemical mechanism (TCM), valence change mechanism (VCM) and threshold switching (TS) for selector devices [
1]. Moreover, the interfacial coupling mechanism (ICM) was also introduced to explain the RS between HRS and LRS [
8].
Binary metal oxides such as TiO
2, WO
3, VO
2, ZnO, Ta
2O
5, HfO
2 and Fe
2O
3 have been extensively studied as RS layers due to their controllable components and simple structures [
9,
10,
11,
12,
13,
14,
15]. Among various binary metal oxides, hafnium oxide (HfO
x) shows distinguished advantages of large dielectric constant (~25), wide band-gap (~6 eV) and good thermal stability, leading to a potentially ultrathin storage layer with both enhanced function density and optical transparency [
16]. Hence, HfO
x-based memristive devices are promising candidates for future applications such as non-volatile memories, logic operations as well as artificial synapses in neuronal systems [
16,
17,
18]. However, an uncontrolled diameter and number of CFs will cause poor uniformity and reliability of memristive devices. To address this problem, various approaches have been proposed to optimize the RS performance by modulating the growth and rupture of CFs, including device structure design [
19,
20], operating schemes optimization [
21], and materials modulation [
22,
23].
In previous reports, defects and impurities in RS layers are the main factors that induce RS in RRAM devices. Therefore, the distribution of defects and impurities in RS layers can be controlled intentionally to improve the switching properties. Several methods have been investigated to modulate the RS layers’ materials, among which plasma treatment is proven to be a simple and effective way as well as impurity doping and crystallization modulating [
24,
25].
T. Kawashima et al. found that the RS performance in the SiO
2/Cu stack could be modified by Ar plasma treatment in terms of operating voltages and light response, which were ascribed to oxygen vacancies produced in the surface region of the SiO
2 by Ar plasma treatment [
26]. Boncheol Ku et al. achieved the properties of forming-free process, faster switching speed, tighter low-resistance state and high-resistance state current distribution, smaller variations of SET/RESET voltages, and enhanced retention/endurance characteristics under HRS in HfO
2-based RRAM through appropriate Ar plasma treatment [
27]. For oxides-based memristive devices, the RS is presumably caused by the migration of oxygen ions, the concentration and distribution of oxygen ions/vacancies will affect the RS performance directly. Therefore oxygen plasma treatment (OPT) is considered to be a more effective method to modulate RS properties. For instance, Umesh Chand et al. suppressed the endurance degradation of HfO
2-based RS memory by utilizing OPT which could increase extra available oxygen ions [
28]. Xiaorong Chen et al. achieved improved RRAM stability due to the large quantity Ta
2O
5 near the top electrode after OPT, which played an important role in resistive switching of the devices [
29]. Nevertheless, the lack of experimental evidence observed directly from the microscopic level of materials and appropriate understandings on how OPT modulates switching properties hinders its practical application.
In this paper, we revealed the OPT-enhanced RS performance in HfOx-based memristive devices by advanced conductive atomic force microscopy (C-AFM) combined with X-ray photoelectron spectroscopic (XPS) surface analysis. It was found that the structural deformations of the HfOx nanofilm induced by migration of oxygen ions and interfacial electrochemical reactions can be eliminated by OPT effectively. Moreover, this type of deformations in HfOx nanofilm no longer occur after OPT, which directly illustrates the enhanced quantity of HfOx nanofilm and the nature of enhanced RS properties after OPT. Finally, we revealed the underlying mechanisms of the enhanced RS performance after OPT according to material analysis and electrical measurements.
3. Results and Discussion
As the development of emerging nanoionics, ions transport process and related electrochemical reactions at nanoscale have been studied extensively [
30,
31]. According to previous studies, ions can easily diffuse by vacancies and transport along grain boundaries [
32,
33]. As a result, the formation, rupture and reaggregation of CFs in polycrystalline HfO
x nanofilm-based memristive devices may be stochastic, which will cause inhomogenous programming parameters. Therefore, efforts can be devoted to modulating the electronic structure of polycrystalline HfO
x nanofilm by carefully adjusting the concentration and distribution of oxygen ions/vacancies.
In order to investigate the influence of OPT on prepared HfO
x nanofilm, a series of electrical measurements and material analysis were carried out. The HfO
x nanofilm with the thickness of 5 nm was firstly deposited onto a commercial Pt/Ti/SiO
2 substrate by the RF magnetron sputtering technique, as visualized by the cross-sectional TEM image in
Figure 1a. Then the as-prepared HfO
x nanofilm was observed by HRTEM as shown in
Figure 1b, we can clearly see that the HfO
x nanofilm exhibits a polycrystalline structure with typical grain boundaries (
Figure S1 shows the topographic AFM image of grains in the as-deposited HfO
x nanofilm). The electrical transport characteristics of the RS layer can be affected by its element compositions significantly. Therefore, the high-angle annular dark-field scanning transmission electron microscopy (HAADF STEM) image and energy-dispersive spectroscopy (EDS) mappings of the HfO
x nanofilm are presented in
Figure 1c,d, respectively. One can see that the atomic ratio of hafnium and oxygen in HfO
x nanofilm is nearly 1:1.25, indicating that the as-grown HfO
x nanofilm possesses vast oxygen vacancies which will cause unstable lattice structure and uncontrollable CFs during the RS processes.
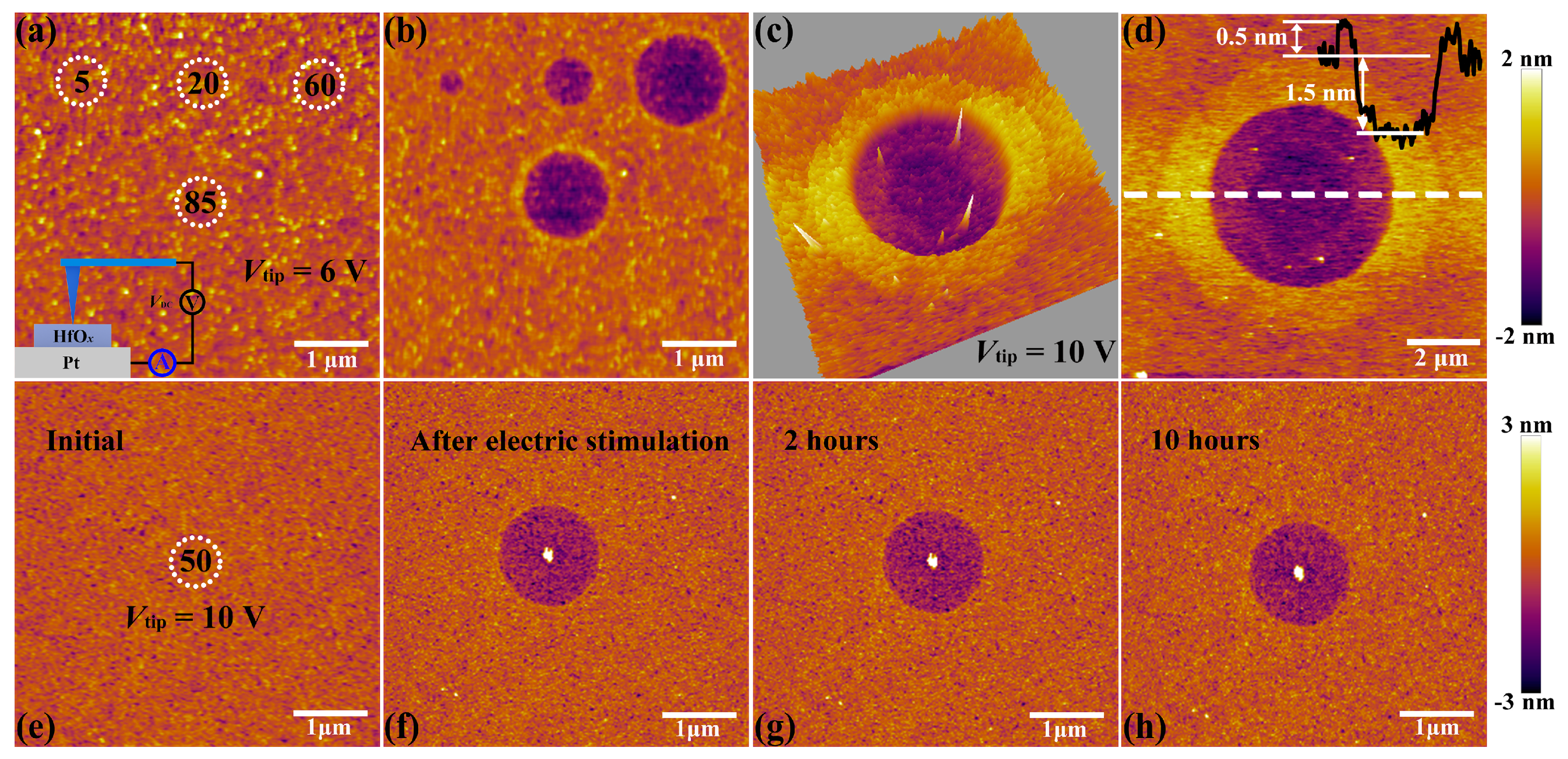
The C-AFM measurements were performed by utilizing a Ti/Ir coated conductive tip in contact mode as a top microelectrode, as shown in the insert part of
Figure 2a. Then DC voltage sweepings from 0 to 6 V with a compliance current (
Icc) of 20 nA were subjected onto the tip at four positions for 5, 20, 60 and 85 times, respectively (see
Figure 2a). It is obvious that structural deformations took place at the positions of applied electric stimulation (see
Figure 2b). To be more specific, the diameter and depth of circular pits increase from 0.3 to 1.3 μm and 0.7 to 1.5 nm respectively, corresponding to the sweeping cycles increase from 5 to 60 times. It is worth noting that the shape and dimension of circular pits cannot enlarge continuously when it reaches a certain limit (comparing the sweeping cycles between 60 and 85 times), which is attributed to the constraint of local electric field, indicating that this process is driven by the applied electric field. Such structural deformations have been studied to be a result of oxygen ions migration and subsequent oxygen gas eruption induced by electrochemical reactions at the top interface between conductive tip and HfO
x nanofilm, which also result in the accumulation of vast interstitial oxygen atoms and oxygen vacancies near these circular pits [
34,
35]. To further explore the localization of oxygen ions’ motion and related electrochemical reactions, we applied a DC voltage bias of 10 V onto the conductive tip to scan a region of 1.0 × 1.0 μm
2 for 5 times with the scan rate of 1 Hz in contact mode. Soon afterwards, an extended area of 10 × 10 μm
2 was scanned with no bias applied to the tip. As shown in
Figure 2c,d, more drastic oxygen ions’ migration, accumulation and related electrochemical reactions at the top interface were detected, which result in the severer collapse of lattice structure with the diameter of circular pit up to about 5 μm. This phenomenon illustrates that the oxygen ions’ migration and related electrochemical reactions are indeed induced by the external electric field rather than the particularity of selected regions. The prominent ring around the middle pit may be attributed to the lattice distortion that resulted from interstitial oxygen atoms and vacancies which can induce expansion of lattice structure around the central region under the external electric field.
Next, the HfO
x nanofilm with a circular pit which is created by the migration of oxygen ions accompanied with occurrence of electrochemical reactions and generation of oxygen gas in the external electric field was treated by oxygen plasma with exposure time of 30 s. It is found that structural deformations of the HfO
x nanofilm induced by electrical stimuli were repaired by OPT (see
Figure 3b), compared with the morphology before OPT in
Figure 3a (the positioning method before and after OPT was presented in
Figure S2). To illustrate the disappearance of circular pit is the effect of OPT instead of selfreparing without any external field, we investigated the retentivity of the circular pit. As depicted in
Figure 2e, C-AFM measurements were performed on the sample with DC voltage sweepings to 10 V at the central location for 50 times.
Figure 2f–h show topographic measurements 0, 2 and 10 h after removing the electrical stimulation, respectively. One can see that the structural deformations occurred on the sample still exist even after 10 h. It is necessary to declare that the total time of the OPT process is less than 2 h, including placing the sample, pumping vacuum, setting up parameters, etc. Hence we can confirm that the repairation of structural deformations is attributed to OPT. The reparation of a circular pit can be understood as a chemical reaction, which is the recombination of oxygen ions and oxygen vacancies or the formation of Hf–O bonds. To further illuminate the physical nature of the repairation phenomenon in HfO
x nanofilm after OPT, the same C-AFM measurement procedures were applied to the oxygen plasma-treated HfO
x nanofilm. DC voltage sweepings from 0 to 10 V with a
Icc of 20 nA were applied to the tip at four positions for 10, 40, 70 and 150 times, respectively (see
Figure 3c). Remarkably, we can see that there is no structural deformations took place at the positions where electric stimulation was applied (see
Figure 3d). On the basis of previous discussions, oxygen vacancies in polycrystal HfO
x nanofilm could be partly filled with oxygen ions after OPT, which resulted in the more stable lattice structure. More specifically, oxygen vacancies near the top surface of the HfO
x nanofilm are easier to fill than the deeper ones due to the energy loss of oxygen ions in oxygen plasma with the increasing implantation depth, which ensures the more robust lattice structure at the top surface than the deeper regions. Therefore, the physical deformations that occurred at the mechanically weakest part of the thin films cannot arise in the HfO
x nanofilm after OPT due to the more stable lattice structure especially near the top interface [
35].
To further illustrate the influence of OPT on the switching characteristics of HfO
x nanofilm, the C-AFM measurements were performed with a DC voltage was applied to the Ti/Ir-coated Si tip and the bottom Pt electrode was grounded. Through in situ morphology and electrical measurements, we primarily researched the influence of structural deformations mentioned above on the electrical properties of the as-prepared HfO
x nanofilm. With the pretreatment of applying 10 V voltage sweepings for 10, 40 times respectively, we subsequently applied the voltages (0 to 6 V, 6 to 0 V, 0 to −6 V, −6 to 0 V) onto the tip to measure the
I-V curves under the two pretreatment conditions, respectively. Based on the discussions above, the pretreatment of 40 times produced larger structural deformations than 10 times, suggesting more oxygen vacancies generated by the pretreatment, which enhanced the conductance of HfO
x nanofilm. Therefore, the lower switching voltages were obtained for the pretreatment of 40 times than 10 times (see
Figure 4a). However, we observed no remarkable excursion in
I-V curves when the same operations were applied on the HfO
x nanofilm after OPT (see
Figure 4b), which can be accounted for by the more stable lattice structure. As plotted in
Figure 4c, the SET curves of the as-grown HfO
x nanofilm show a significant fluctuation, which mainly caused by the continuous generation of oxygen vacancies at the area of circular pit induced by electrochemical reactions, which changes the conductance of CFs in HfO
x nanofilm after each SET process. Nevertheless, after OPT, as shown in
Figure 4d, the HfO
x nanofilm exhibits a more stable SET process. The lower
VSET can be explained by the migration of non-lattice oxygen ions supplied by OPT, which facilitates the arrival of
Icc.
Figure 4e presents the
VSET cumulative probability of the HfO
x nanofilm before and after OPT. We can observe clearly that the distribution of statistical
VSET after OPT is more concentrated than that before OPT. In addition,
Figure 4f gives the linear fitting of extracted
VSET before and after OPT from SET sweepings for 150 times, the decrease of |slope| of
VSET from 0.0033 to 0.00131 after OPT implies not only the generation of oxygen vacancies near the circular pit before OPT but also the positive effect of an undamaged structure during the electric stimulation after OPT. The significant improvement of uniformity in SET processes after OPT can be attributed to the more stable lattice structure, which avoided physical disruption during the SET processes [
35].
The enhanced RS characteristics were also found in the prepared HfO
x-based memristive devices after OPT. As plotted in
Figure 5a, the Pt/HfO
x/Pt device before OPT shows typical bipolar RS behaviors in
I-
V curves, displaying a dramatic fluctuation (insert part shows the schematic of the memory device). Oxygen gas bubbles which were caused by oxygen ions’ migration and related electrochemical reactions were observed in the top Pt electrode of Pt/as-deposited HfO
x/Pt device during RS processes (see
Figure S3) [
35]. After OPT, we found that the RS curves show a much better stability and repeatability (see
Figure 5b). The histograms and statistical charts in
Figure 5c,d show the switching voltage distributions of the Pt/HfO
x/Pt devices without/with OPT obtained from the RS curves, respectively. The switching voltages of the devices without OPT are distributed in widespread ranges of 0.2 to 2 V (
VSET) and −0.2 to −1 V (
VRESET), while the switching voltages of the devices with OPT are distributed in scales of 0.3 to 1.1 V (
VSET) and −0.3 to −0.5 V (
VRESET). Obviously, the switching voltages of the Pt/HfO
x/Pt devices with OPT are much more stable than those without OPT. Also, the decrease of switching voltages can be attributed to the participation of non-lattice oxygen ions supplied by OPT during the RS processes which are conductive to the forming of the fine conductive filaments.
As shown in
Figure 6, XPS measurements were performed to further explain the physical nature of the enhanced RS performance observed in the HfO
x-based memristive devices after OPT.
Figure 6a,b show the XPS spectra of O 1s in the as-grown HfO
x nanofilm before and after OPT, respectively. The O 1s signal with the binding energy of 529.5 eV are mainly arising from the lattice oxygen (Hf-O bonding, HfO
2 components). While the higher binding energy of O 1s at 530.14 eV are associated with the non-lattice oxygen anions. One can see that lattice oxygen species increase from 52.2% to 54.31% and non-lattice oxygen also increases from 20.75% to 27.78%. In addition, Hf 4f XPS spectra also provide essential information of the composition change in HfO
x nanofilm after OPT. As shown in
Figure 6c,d, the presence of the Hf 4f
7/2 and Hf 4f
5/2 species with the binding energies of 16.29 eV and 17.9 eV corresponds to the predominant HfO
x components where
x is less than 2, while the minor high binding energy Hf 4f
7/2 and Hf 4f
5/2 species at 16.6 eV and 18.1 eV are associated with the HfO
2 components. In addition, the binding energy Hf species at 16 eV correspond to metallic Hf. After OPT, the HfO
2 species increase from 55.04% to 67.73%, the HfO
x species decrease from 38.39% to 32.27% where
x is less than 2 and the metallic Hf disappeared. The above XPS results indicate that the OPT can increase the number of non-lattice oxygen and the concentration of HfO
2 which improved the stability of lattice structure.
Considering all the above experimental results, the mechanisms of the RS properties’ improvement in the oxygen plasma-treated Pt/HfO
x/Pt devices were proposed based on the theory of CFs.
Figure 7 schematically show the processes of RS behaviors in a single device. The as-grown polycrystalline HfO
x possesses a large amount of intrinsic vacancies in crystalline regions (violet dotted circle in
Figure 7a), which is consistent with aforementioned EDS and XPS analysis. According to previous reports, when a positive voltage is applied to the inert Pt top electrode while the Pt bottom electrode is grounded, oxygen ions will be attracted to the top interface, subsequently the CFs consisting of oxygen vacancies will be formed mainly along the grain boundaries in polycrystalline HfO
x nanofilms [
32]. Nevertheless, a certain amount of oxygen vacancies apart from grain boundaries can also provide transport routes for oxygen ions in polycrystalline HfO
x nanofilm. Hence we can believe that multiple and stochastic CFs will be formed not only along grain boundaries but also the location beyond grain boundaries but rich in oxygen vacancies after electric stimuli. During the forming process, oxygen ions will not only transport along the grain boundaries but also by means of the oxygen vacancies (see
Figure 7b). As a result, the stochastic forming of the multiple CFs will lead to severe fluctuation of the programming voltages and the device resistances (see
Figure 7c). In the oxygen plasma-treated Pt/HfO
x/Pt device, oxygen vacancies in crystalline regions drastically reduced by the active oxygen ions’ implantation (see
Figure 7d), as proved by the XPS analysis and AFM experiments above. Therefore, the oxygen ions’ transport depends considerably on the grain boundaries in polycrystalline HfO
x nanofilm [
36]. During the forming process, oxygen ions transport through the assistance of intrinsic oxygen vacancies in crystalline regions will be restrained effectively (see
Figure 7e). As a result, the CFs will be formed mainly along the grain boundaries (see
Figure 7f), and better stability and uniformity will be achieved in RS processes. During the RESET process, the increased non-lattice oxygen can migrate back to recombine with oxygen vacancies which increase the probability of the recombination, promoting the stability of the RESET process [
28]. It is worth noting that the transport of non-lattice oxygen during the RS processes also relies considerably on the grain boundaries, hence, this type of transport can not cause additional instability.
Last but not least, the significant fluctuation in the Pt/as-deposited HfO
x/Pt device also partly due to the continuous generation of oxygen vacancies at the area of the circular pit which is the mechanically weakest part of the HfO
x nanofilm forming CFs during RS cycles. The continuous generation of oxygen vacancies increase the conductance of CFs as the increasing sweeping cycles (see
Figure 7g–i), which is consistent with the former C-AFM topographic measurements (see
Figure 2b). Through OPT (see
Figure 7j), the increase of HfO
2 components especially near the top interface ensures a more stable lattice structure and eliminates the physical structural deformations effectively, which were proved by direct experimental evidence from microscopic level of materials (see
Figure 3b,d). Therefore, the conductance of CFs can remain stable during the RS processes (see
Figure 7k–m), which further ensures the uniformity and repeatability in RS processes. Overall, after OPT, the more stable lattice structure and increased available non-lattice oxygen improve the RS properties of the HfO
x-based memristive devices.