Electrically and Thermally Conductive Low Density Polyethylene-Based Nanocomposites Reinforced by MWCNT or Hybrid MWCNT/Graphene Nanoplatelets with Improved Thermo-Oxidative Stability
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of LDPE-Based Nanocomposites
2.3. Preparation of Testing Samples
2.4. Characterization Methods
- (a)
- for resistivities lower than 104 Ω cm according to the standard PN-EN ISO 3915;
- (b)
- for resistivities higher than 104 Ω cm according to the standard PN-88/E-04405.
3. Results and Discussion
3.1. Morphology of LDPE-Based Nanocomposites
3.2. Electrical and Thermal Conductivity
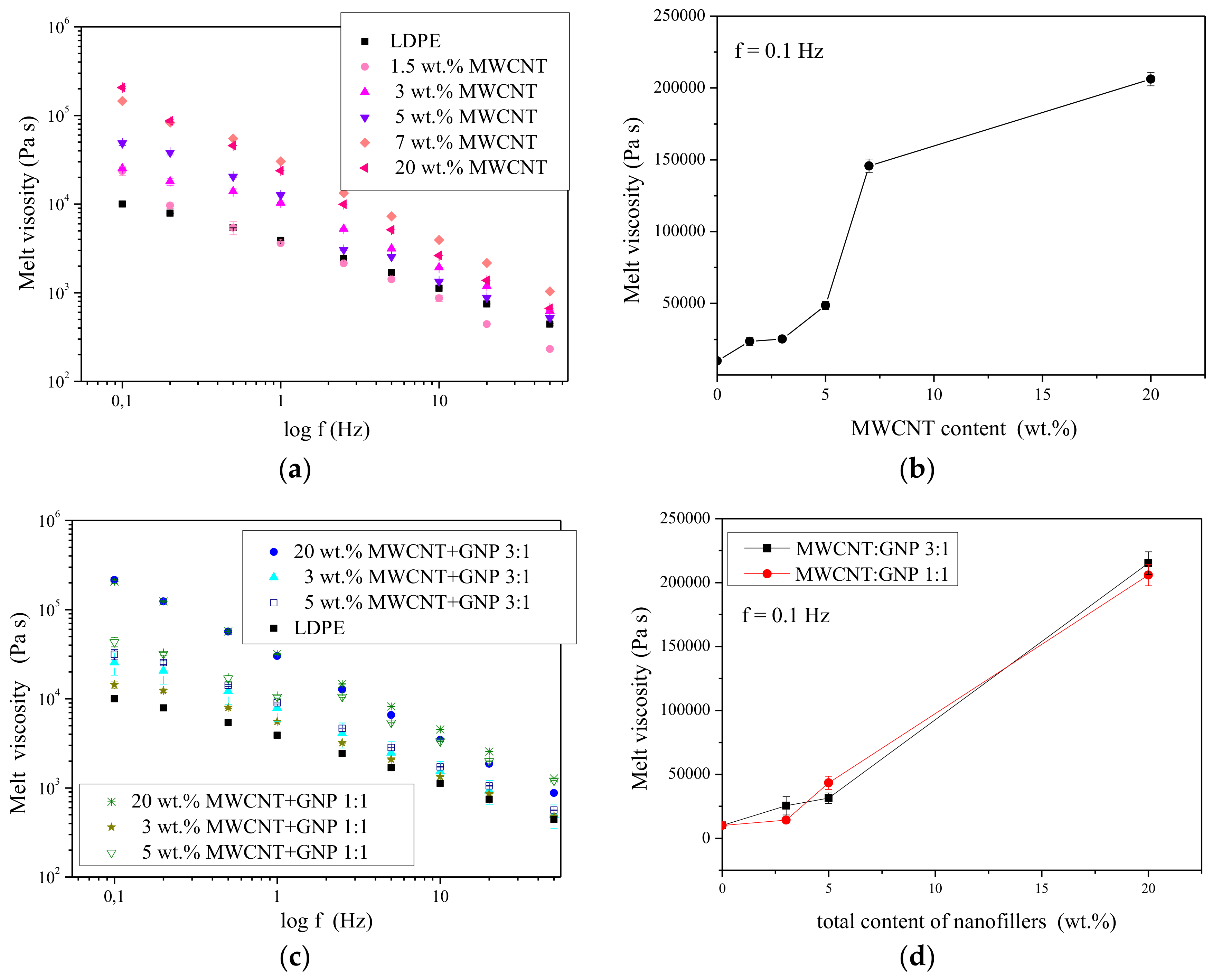
3.3. Structural and Rheological Characteristics
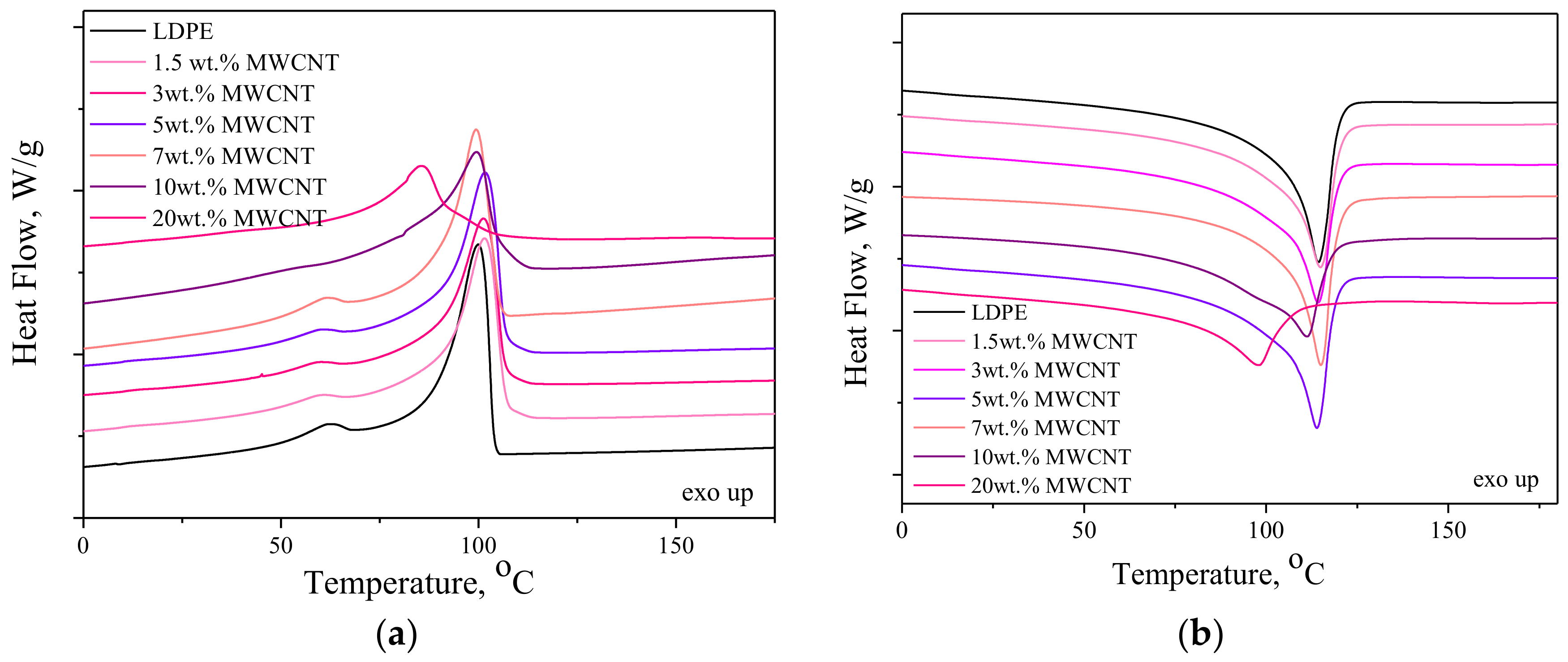
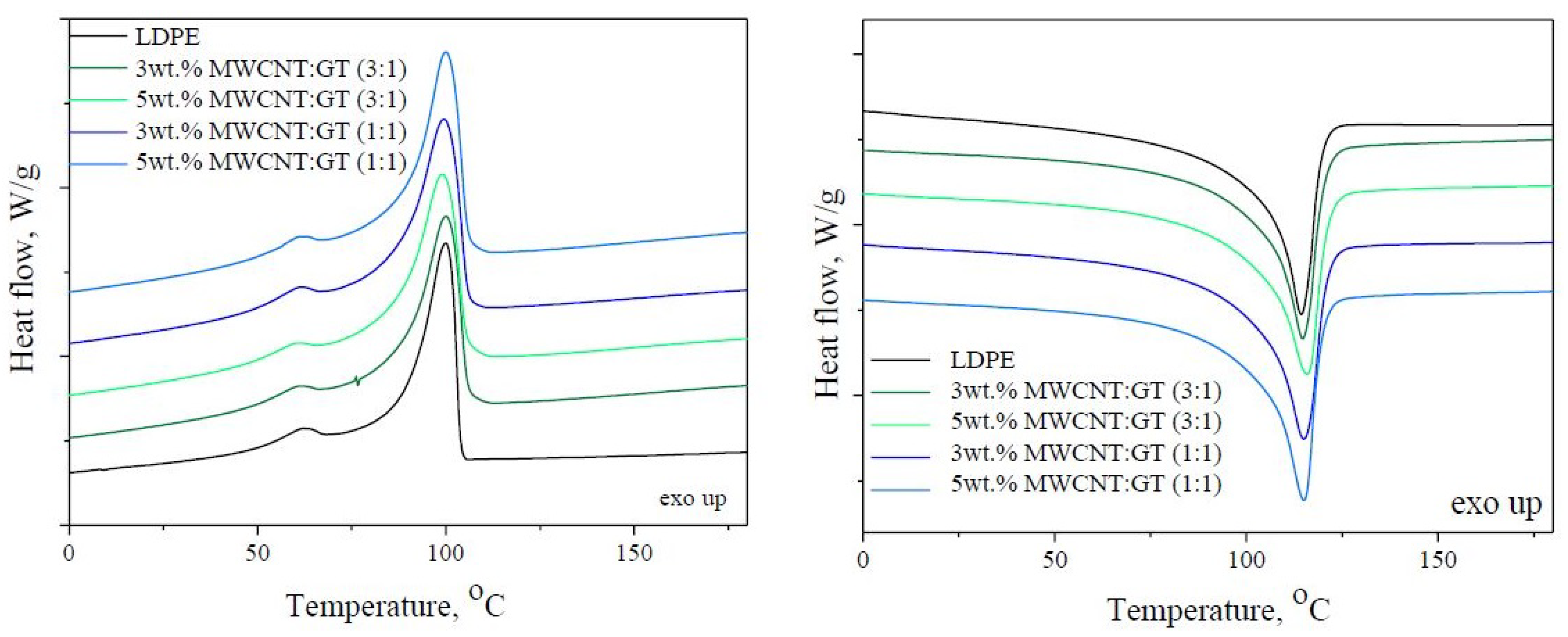
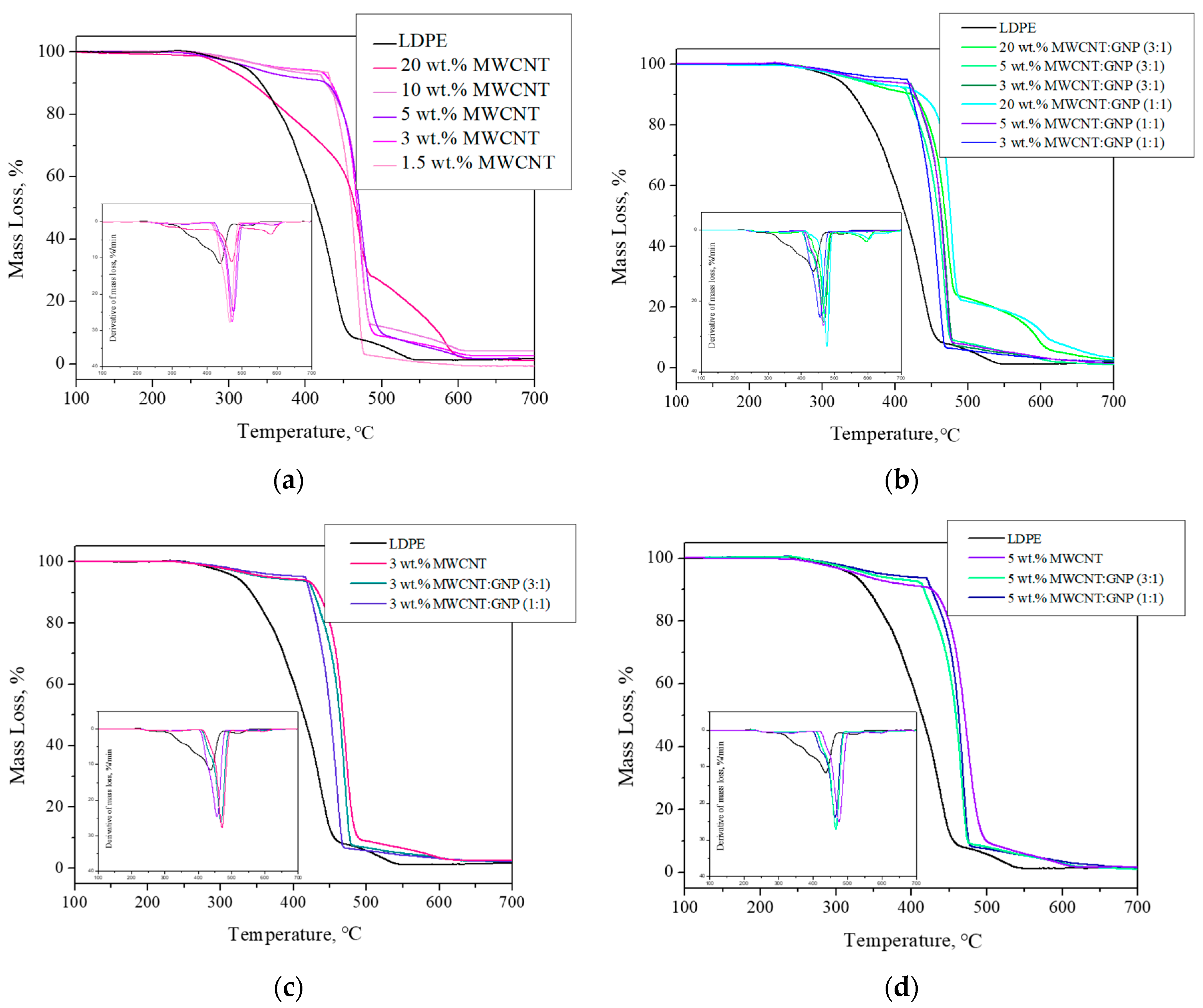
3.4. Thermo-Oxidative Stability
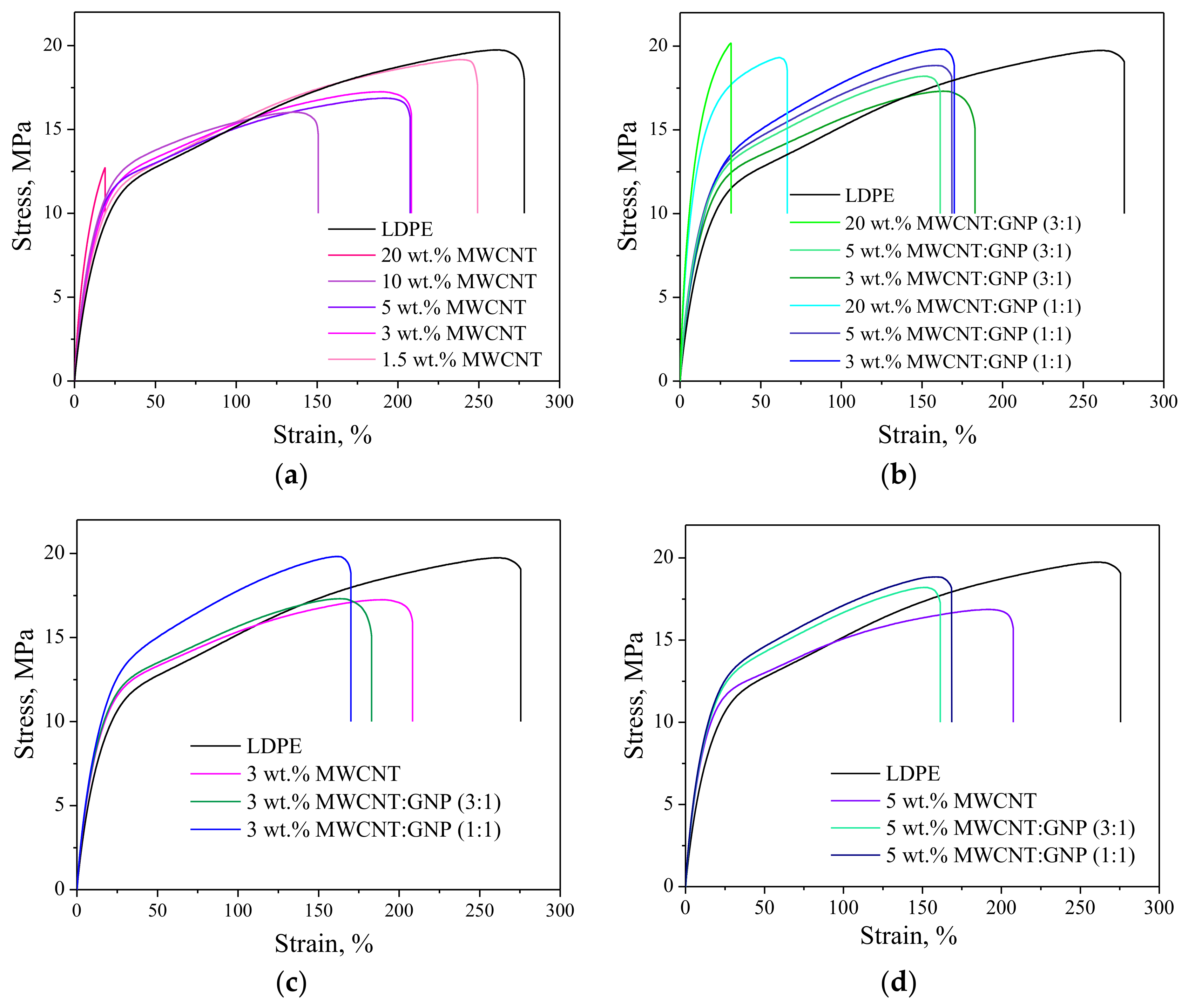
3.5. Tensile Properties
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kulshreshtha, A.K.; Talapatra, S. Competitive New Technologies in Polyolefin Synthesis and Materials. In Handbook of Polyolefins, 2nd ed.; Vasile, C., Ed.; CRR Press: Boca Raton, FL, USA, 2000. [Google Scholar]
- Malpass, D.B. Introduction to Industrial Polyethylene: Properties, Catalysts, and Processes; John Wiley & Sons, Inc.: Hoboken, NJ, USA; Scrivener Publishing LCC: Salem, MA, USA, 2010; ISBN 978-0-470-62598-9. [Google Scholar]
- Soares, J.B.P.; McKenna, T.F.L. Polyolefin Reaction Engineering, 1st ed.; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2012. [Google Scholar]
- Cousins, K. Polymers for Wire and Cable Changes within an Industry; A Rapra Industry Analysis Report; Smithers Rapra Technology: Shawbury, UK, 2000; ISBN 13 978-1847351906. [Google Scholar]
- Thue, W. Electrical Power Cable Engineering, 2nd ed.; Marcel Dekker Inc.: New York, NY, USA, 2003; ISBN 0-8247-4303-2. [Google Scholar]
- Belli, S.; Bareggi, A.; Dell’Anna, G.; Scelza, C.; Donazzi, F. Continuous process for manufacturing electrical cables. PAT. 205143, 31 March 2003. [Google Scholar]
- Zhu, H.; Xu, Z.; Xie, D.; Fang, Y. Graphene: Fabrication, Characterizations, Properties and Applications, 1st ed.; Academic Press: Cambridge, UK, 2017; ISBN 978-0-128-12651-6. [Google Scholar]
- Kim, H.; Abdala, A.A.; Macosko, C.W. Graphene/polymer nanocomposites. Macromolecule 2010, 43, 6515–6530. [Google Scholar] [CrossRef]
- Slonczewski, J.C.; Weiss, P.R. Band structure of graphite. Phys. Rev. Lett. 1958, 109, 272–279. [Google Scholar] [CrossRef]
- Soldano, C.; Mahmood, A.; Dujardin, E. Production, properties and potential of graphene. Carbon 2010, 48, 2127–2150. [Google Scholar] [CrossRef]
- Novoselov, K.S.; Geim, A.K.; Morozov, S.V.; Jiang, D.; Zhang, Y.; Dubonos, S.V.; Grigorieva, I.V.; Firsov, A.A. Electric field effect in atomically thin carbon films. Science 2004, 306, 666–669. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.; Wei, X.; Kysar, J.W.; Hone, J. Measurement of the elastic properties and intrinsic strength of monolayer grapheme. Science 2008, 321, 385–388. [Google Scholar] [CrossRef] [PubMed]
- Balandin, A.A.; Ghosh, S.; Bao, W.; Calizo, I.; Teweldebrhan, D.; Miao, F.; Lau, C.N. Superior thermal conductivity of single-layer grapheme. Nano Lett. 2008, 8, 902–907. [Google Scholar] [CrossRef] [PubMed]
- Razali, M.; Kim, J.F.; Attfield, M.; Budd, P.M.; Drioli, E.; Lee, Y.M.; Szekely, G. Sustainable wastewater treatment and recycling in membrane manufacturing. Green Chem. 2015, 17, 5196–5205. [Google Scholar] [CrossRef]
- Vilela, D.; Parmar, J.; Zeng, Y.; Zhao, Y.; Sanchez, S. Graphene-based microbots for toxic heavy metal removal and recovery from water. Nano Lett. 2016, 16, 2860–2866. [Google Scholar] [CrossRef] [PubMed]
- Bunch, J.S.; Verbridge, S.S.; Alden, J.S.; van der Zande, A.M.; Parpia, J.M.; Craighead, H.G.; McEuen, P.L. Impermeable atomic membranes from graphene sheets. Nano Lett. 2008, 8, 2458–2462. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Miura, Y.; Macosko, C.W. Graphene/polyurethane nanocomposites for improved gas barrier and electrical conductivity. Chem. Mater. 2010, 22, 3441–3450. [Google Scholar] [CrossRef]
- El Achaby, M.; Arrakhiz, F.-A.; Vaudreuil, S.; El Kacem Qaiss, A.; Bousmina, M.; Fassi-Fehri, O. Mechanical, thermal, and rheological properties of graphene-based polypropylene nanocomposites prepared by melt mixing. Polym. Compos. 2012, 33, 733–744. [Google Scholar] [CrossRef]
- Gao, F. Advances in Polymer Nanocomposites, 1st ed.; Woodhead Publishing Lmt.: Sawston, UK, 2012; ISBN 978-1-84569-940-6. [Google Scholar]
- Du, J.; Zhao, L.; Zeng, Y.; Zhang, L.; Li, F.; Liu, P.; Liu, C. Comparison of electrical properties between multi-walled carbon nanotube and graphene nanosheet/high density polyethylene composites with a segregated network structure. Carbon 2011, 49, 1094–1100. [Google Scholar] [CrossRef]
- Pang, H.; Piao, Y.-Y.; Cui, C.-H.; Bao, Y.; Lei, J.; Yuan, G.-P.; Zhang, C.-L. Preparation and performance of segregated polymer composites with hybrid fillers of octadecylamine functionalized graphene and carbon nanotubes. J. Polym. Res. 2013, 20, 304. [Google Scholar] [CrossRef]
- Liu, F.; Hu, N.; Ning, H.; Atobe, S.; Yan, C.; Liu, Y.; Wu, L.; Liu, X.; Fu, S.; Xu, C.; et al. Investigation on the interfacial mechanical properties of hybrid graphene-carbon nanotube/polymer nanocomposites. Carbon 2017, 115, 694–700. [Google Scholar] [CrossRef]
- Che, J.; Wu, K.; Lin, Y.; Wang, K.; Fu, Q. Largely improved thermal conductivity of HDPE/expanded graphite/carbon nanotubes ternary composites via filler network-network synergy. Compos. Part A Appl. Sci. Manuf. 2017, 99, 32–40. [Google Scholar] [CrossRef]
- Al-Saleh, M.H. Electrical and mechanical properties of graphene/carbon nanotube hybrid nanocomposites. Synth. Met. 2015, 209, 41–45. [Google Scholar] [CrossRef]
- Paszkiewicz, S.; Kwiatkowska, M.; Szymczyk, A.; Jotko, M.; Lisiecki, S.; Roslaniec, Z. The influence of different shaped nanofillers (1D, 2D) on barrier and mechanical properties of polymer hybrid nanocomposites based on PET prepared by in situ polymerization. Polym. Compos. 2016, 37, 1949–1959. [Google Scholar] [CrossRef]
- Paszkiewicz, S.; Pawelec, I.; Szymczyk, A.; Rosłaniec, Z. Mechanical and thermal properties of PTT based hybrid nanocomposites containing SWCNT+EG prepared by in situ polymerization. Polimery 2016, 61, 172–180. [Google Scholar] [CrossRef]
- Paszkiewicz, S.; Szymczyk, A.; Sui, X.M.; Wagner, H.D.; Linares, A.; Cirera, A.; Varea, A.; Ezquerra, T.A.; Rosłaniec, Z. Electrical conductivity and transparency of polymer hybrid nanocomposites based on poly(trimethylene terephthalate) containing single walled carbon nanotubes and expanded graphite. J. Appl. Polym. Sci. 2017, 134, 32–40. [Google Scholar] [CrossRef]
- Li, Y.; Yang, T.; Yu, T.; Zheng, L.; Liao, K. Synergistic effect of hybrid carbon nanotube–graphene oxide as a nanofiller in enhancing the mechanical properties of PVA composites. J. Mater. Chem. 2011, 21, 10844–10851. [Google Scholar] [CrossRef]
- Araby, S.; Saber, N.; Ma, X.; Kawashima, N.; Kang, H.; Shen, H.; Zhang, L.; Xu, J.; Majewski, P.; Ma, J. Implication of multi-walled carbon nanotubes on polymer/graphene composites. Mater. Des. 2015, 65, 690–699. [Google Scholar] [CrossRef]
- Szeluga, U.; Kumanek, B.; Trzebicka, B. Synergy in hybrid polymer/nanocarbon composites. A review. Compos. Part A Appl. Sci. Manuf. 2015, 73, 204–231. [Google Scholar] [CrossRef]
- Paszkiewicz, S.; Szymczyk, A.; Sui, X.M.; Wagner, H.D.; Linares, A.; Ezquerra, T.A.; Rosłaniec, Z. Synergetic effect of single-walled carbon nanotubes (SWCNT) and graphene nanoplatelets (GNP) in electrically conductive PTT-block-PTMO hybrid nanocomposites prepared by in situ polymerization. Compos. Sci. Technol. 2015, 118, 72–77. [Google Scholar] [CrossRef]
- Paszkiewicz, S.; Szymczyk, A.; Livanov, K.; Wagner, H.D.; Rosłaniec, Z. Enhanced thermal and mechanical properties of PTT-block-PTMO based hybrid nanocomposites prepared by in situ polymerization via synergy effect between swcnts and graphene nanoplatelets. Exp. Polym. Lett. 2015, 9, 509–524. [Google Scholar] [CrossRef]
- Paszkiewicz, S.; Szymczyk, A.; Pilawka, R.; Przybyszewski, B.; Czulak, A.; Rosłaniec, Z. Improved thermal conductivity of poly(trimethylene terephthalate-block-poly(tetramethylene oxide) based nanocomposites containing hybrid single-walled carbon nanotubes/graphene nanoplatelets fillers. Adv. Polym. Technol. 2017, 36, 236–242. [Google Scholar] [CrossRef]
- Celzard, A.; McRae, E.; Deleuze, C.; Dufort, M.; Furdin, G.; Marêché, J.F. Critical concentration in percolating systems containing a high-aspect-ratio filler. Phys. Rev. B 1996, 53, 6209–6214. [Google Scholar] [CrossRef]
- Pawlikowska, D.; Szymczyk, A.; Paszkiewicz, S.; Zenker, M.; Stanik, R.; Subocz, J.; Masztak, R. Electrically and thermally conductive polymer nanocomposites based on low-density polyethylene containing graphene nanoplatelets. Przem. Chem. 2017, 96, 1973–1978. [Google Scholar] [CrossRef]
- Wunderlich, B.; Doyle, M. Specific heat of synthetic high polymers. VIII. Low pressure polyethylene. J. Polym. Sci. 1957, 24, 201–213. [Google Scholar] [CrossRef]
- Verdejo, R.; Bernal, M.M.; Romasanta, L.J.; Lopez-Manchado, M.A. Graphene filled polymer nanocomposites. J. Mater. Chem. 2011, 21, 3301–3310. [Google Scholar] [CrossRef]
- Ma, P.-C.; Siddiqui, N.A.; Marom, G.; Kim, J.-K. Dispersion and functionalization of carbon nanotubes for polymer-based nanocomposites: A review. Compos. Part A Appl. Sci. Manuf. 2010, 41, 1345–1367. [Google Scholar] [CrossRef]
- Potts, J.R.; Dreyer, D.R.; Bielawski, C.W.; Ruoff, R.S. Graphene-based polymer nanocomposites. Polymer 2011, 52, 5–25. [Google Scholar] [CrossRef]
- Dresselhaus, M.S.; Dresselhaus, G.; Avouris, P.H. Carbon Nanotubes: Synthesis, Structure, Properties, and Applications; Springer: Berlin, Germany, 2001. [Google Scholar]
- Ebbesen, T. Carbon Nanotubes: Preparation and Properties; CRC: New York, NY, USA, 1997. [Google Scholar]
- Safdari, M.; Al-Haik, M.S. Synergistic electrical and thermal transport properties of hybrid polymeric nanocomposites based on carbon nanotubes and graphite nanoplatelets. Carbon 2013, 64, 111–121. [Google Scholar] [CrossRef]
- Thostenson, E.K.; Li, C.; Chou, T.W. Nanocomposites in context. Compos. Sci. Technol. 2005, 65, 491–516. [Google Scholar] [CrossRef]
- He, X.J.; Du, J.H.; Ying, Z.; Cheng, H.M. Positive temperature coefficient effect in multiwalled carbon nanotube/high-density polyethylene composites. Appl. Phys. Lett. 2005, 86, 062112. [Google Scholar] [CrossRef]
- Zhang, Q.H.; Rastogi, S.; Chen, D.J.; Lippits, D.; Lemstra, P.J. Low percolation threshold in single-walled carbon nanotube/high density polyethylene composites prepared by melt processing technique. Carbon 2006, 44, 778–785. [Google Scholar] [CrossRef]
- Ciselli, P.; Zhang, R.; Wang, Z.; Reynolds, C.T.; Baxendale, M.; Peijs, T. Oriented UHMW–PE/CNT composite tapes by a solution casting-drawing process using mixed-solvents. Carbon 2009, 45, 2741–2748. [Google Scholar] [CrossRef]
- Xie, S.H.; Liu, Y.Y.; Li, J.Y. Comparison of the effective conductivity between composites reinforced by graphene nanosheets and carbon nanotubes. Appl. Phys. Lett. 2008, 92, 243121. [Google Scholar] [CrossRef]
- Han, Z.; Fina, A. Thermal conductivity of carbon nanotubes and their polymer nanocomposites: A review. Prog. Polym. Sci. 2011, 36, 914–944. [Google Scholar] [CrossRef]
- Pop, E.; Varshney, V.; Roy, A.K. Thermal properties of graphene: Fundamentals and applications. MRS Bull. 2012, 37, 1273–1281. [Google Scholar] [CrossRef]
- Chen, H.; Ginzburg, V.V.; Yang, J.; Yunfeng Yang, Y.; Liu, W.; Huang, Y.; Du, L.; Chen, B. Thermal conductivity of polymer-based composites. Fundamentals and applications. Prog. Polym. Sci. 2016, 59, 41–85. [Google Scholar] [CrossRef]
- Yu, A.; Ramesh, P.; Sun, X.; Bekyarova, E.; Itkis, M.E.; Haddon, R.C. Enhanced thermal conductivity in a hybrid graphite nanoplatelet–carbon nanotube filler for epoxy composites. Adv. Mater. 2008, 20, 4740–4744. [Google Scholar] [CrossRef]
- King, J.A.; Johnson, B.A.; Via, M.D.; Ciarkowski, C.J. Effects of carbon fillers in thermally conductive polypropylene based resins. Polym. Compos. 2010, 31, 497–506. [Google Scholar] [CrossRef]
- King, J.A.; Gaxiola, D.L.; Johnson, B.A.; Keith, J.M. Thermal conductivity of carbon-filled polypropylene-based resins. J. Compos. Mater. 2010, 44, 839–855. [Google Scholar] [CrossRef]
- Im, H.; Kim, J. Thermal conductivity of a graphene oxide-carbon nanotube hybrid composite. Carbon 2012, 50, 5429–5440. [Google Scholar] [CrossRef]
- Shenogin, S.; Ozisik, R.; Keblinski, P.; Cahill, D.G. Role of thermal boundary resistance on the heat flow in carbon-nanotube composites. J. Appl. Phys. 2004, 95, 8136. [Google Scholar] [CrossRef]
- Shonogina, N.; Shenogin, S.; Xue, L.P.; Keblinski, P. On the lack of thermal percolation in carbon nanotube composites. Appl. Phys. Lett. 2005, 87, 133106. [Google Scholar] [CrossRef]
- Chang, T.E.; Jensen, L.R.; Kisliuk, A.; Pipes, R.B.; Pyrz, R.; Sokolov, A.P. Microscopic mechanism of reinforcement in single-wall carbon nanotube/polypropylene nanocomposite. Polymer 2005, 46, 439–444. [Google Scholar] [CrossRef]
- Kodjie, S.L.; Li, L.; Li, B.; Cai, W.; Li, C.Y.; Keating, M. Morphology and Crystallization Behavior of HDPE/CNT Nanocomposite. J. Macromol. Sci. Part B Phys. 2006, 45, 231–245. [Google Scholar] [CrossRef]
- Pavlidou, S.; Papaspyrides, C.D. A review on polymer–layered silicate nanocomposites. Prog. Polym. Sci. 2008, 33, 1119–1198. [Google Scholar] [CrossRef]
- Wegrzyn, M.; Galindo, B.; Benedito, A.; Gimenez, E. Morphology, thermal, and electrical properties of polypropylene hybrid composites co-filled with multi-walled carbon nanotubes and graphene nanoplatelets. J. Appl. Polym. Sci. 2015, 132, 42793. [Google Scholar] [CrossRef]
- Tarani, E.; Papageorgiou, D.G.; Valles, C.; Wurm, A.; Terzopoulou, Z.; Bikiaris, D.N.; Schick, C.; Chrissafis, K.; Vourlias, G. Insights into crystallization and melting of high density polyethylene/graphene nanocomposites studied by fast scanning calorimetry. Polym. Test. 2018, in press. [Google Scholar] [CrossRef]
- Tarani, E.; Terzopoulou, Z.; Bikiaris, D.N.; Kyratsi, T.; Chrissafis, K.; Vourlias, G. Thermal conductivity and degradation behavior of HDPE/graphene nanocomposites: Pyrolysis, kinetics and mechanism. J. Therm. Anal. Calorim. 2017, 129, 1715–1726. [Google Scholar] [CrossRef]
- Papageorgiou, D.G.; Kinloch, I.A.; Young, R.J. Hybrid multifunctional graphene/glass-fibre polypropylene composites. Compos. Sci. Technol. 2015, 137, 44–51. [Google Scholar] [CrossRef]
- Durmus, A.; Kasgoz, A.; Macosko, C.W. Linear low density polyethylene (LLDPE)/clay nanocomposites. Part I: Structural characterization and quantifying clay dispersion by melt rheology. Polymer 2007, 48, 4492–4502. [Google Scholar] [CrossRef]
- Jang, B.Z.; Zhamu, A. Processing of nanographene platelets (NGPs) and NGP nanocomposites: A review. J. Mater. Sci. 2008, 43, 5092–5101. [Google Scholar] [CrossRef]
- Poetschke, P.; Abdel-Goad, M.; Alig, I.; Dudkin, S.; Lellinger, D. Rheological and dielectrical characterization of melt mixed polycarbonate-multiwalled carbon nanotube composites. Polymer 2004, 45, 8863–8870. [Google Scholar] [CrossRef]
- Paszkiewicz, S.; Szymczyk, A.; Kasprowiak, I.; Zenker, M.; Pilawka, R.; Linares, A.; Ezquerra, T.A.; Rosłaniec, Z. Electrical and rheological characterization of poly(trimethylene terephthalate) hybrid nanocomposites filled with COOH functionalized MWCNT and graphene nanosheets. Polym. Compos. 2017. (Early view version). [Google Scholar] [CrossRef]
- McNally, T.; Poetschke, P.; Halley, P.; Murphy, M.; Martin, D.; Bell, S.E.J.; Brennan, G.P.; Bein, D.; Lemoine, P.; Quinn, J.P. Polyethylene multiwalled carbon nanotube composites. Polymer 2005, 46, 8222–8232. [Google Scholar] [CrossRef]
- She, Y.; Chen, G.; Wu, D. Fabrication of polyethylene/graphite nanocomposite from modified expanded graphite. Polym. Int. 2007, 56, 679–685. [Google Scholar] [CrossRef]


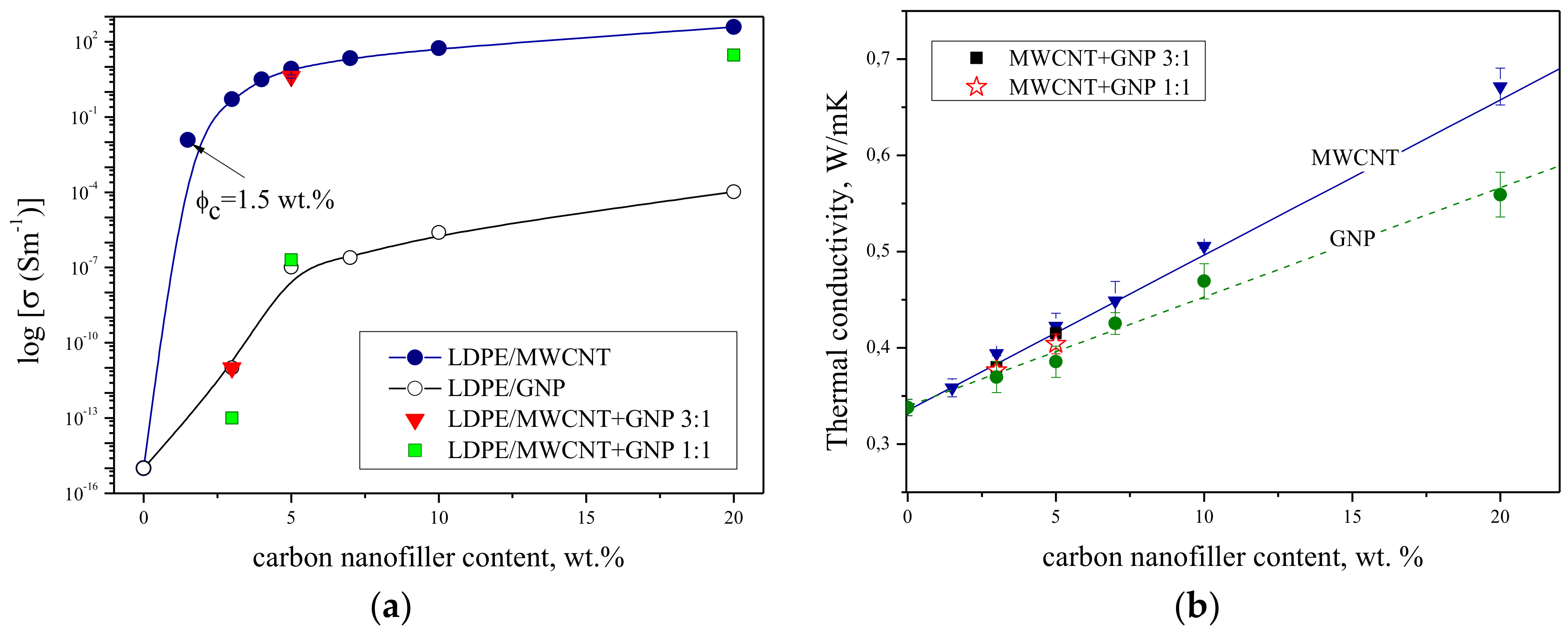
| Content of MWCNT wt. % | Resistivity */Ω·m | ||
|---|---|---|---|
| at 150 rpm | at 60 rpm | at 40 rpm | |
| 1.5 | (8.1 ± 2.1) × 103 | 8.4 × 104 | 1 × 106 |
| 3 | 12.0 ± 7.5 | 27.0 ± 17 | 1.94 ± 12 |
| 4 | - | 0.36 ± 0.04 | 0.32 ± 0.04 |
| 5 | 1.22 ± 0.76 | 0.12 ± 0.07 | 0.12 ± 0.04 |
| 20 | (0.3 ± 0.01) × 10−2 | - | (0.26 ± 0.01) × 10−2 |
| Sample | Tm [°C] | ΔHm [J/g] | Tc [°C] | ΔHc [J/g] | Xc [%] | d [g/cm3] |
|---|---|---|---|---|---|---|
| LDPE | 114 | 128.9 | 100 | 129.1 | 44.0 | 0.934 ± 0.002 |
| LDPE/1.5 wt. % MWCNT | 115 | 120.2 | 101 | 120.3 | 41.7 | 0.942 ± 0.001 |
| LDPE/3 wt. % MWCNT | 114 | 112.6 | 101 | 112.2 | 39.6 | 0.951 ± 0.001 |
| LDPE/5 wt. % MWCNT | 114 | 119.1 | 102 | 119.4 | 42.7 | 0.962 ± 0.002 |
| LDPE/7 wt. % MWCNT | 114 | 104.8 | 100 | 104.8 | 38.4 | 0.968 ± 0.001 |
| LDPE/10 wt. % MWCNT | 110 | 104.1 | 100 | 101.3 | 39.4 | 0.985 ± 0.003 |
| LDPE/20 wt. % MWCNT | 97 | 102.1 | 85 | 102.7 | 38.7 | 0.934 ± 0.002 |
| LDPE/3 wt. % MWCNT:GNP (3:1) | 115 | 126.4 | 100 | 122.8 | 44.5 | 0.953 ± 0.001 |
| LDPE/5 wt. % MWCNT:GNP (3:1) | 116 | 121.8 | 99 | 121.6 | 43.8 | 0.962 ± 0.001 |
| LDPE/3 wt. % MWCNT:GNP (1:1) | 115 | 127.7 | 100 | 123.5 | 44.9 | 0.951 ± 0.003 |
| LDPE/5 wt. % MWCNT:GNP (1:1) | 115 | 124.0 | 100 | 122.5 | 44.5 | 0.961 ± 0.006 |
| Sample | E [MPa] | σm [MPa] | εb [%] |
|---|---|---|---|
| LDPE | 137.5 ± 3.30 | 19.77 ± 0.51 | 280.87 ± 8.16 |
| LDPE/1.5 wt. % MWCNT | 144.30 ± 8.07 | 19.03 ± 0.13 | 240.61 ± 26.39 |
| LDPE/3 wt. % MWCNT | 144.25 ± 6.65 | 17.94 ± 0.42 | 166.20 ± 10.66 |
| LDPE/5 wt. % MWCNT | 150.09 ± 8.06 | 16.76 ± 0.24 | 223.81 ± 11.25 |
| LDPE/10 wt. % MWCNT | 170.53 ± 7.19 | 15.90 ± 0.17 | 142.40 ± 6.76 |
| LDPE/20 wt. % MWCNT | 201.26 ± 8.80 | 12.57 ± 0.63 | 19.01± 2.78 |
| LDPE/3 wt. % MWCNT:GNP (3:1) | 158.78 ± 5.40 | 17.07 ± 0.21 | 187.05 ± 5.40 |
| LDPE/5 wt. % MWCNT:GNP (3:1) | 167.91 ± 3.29 | 18.06 ± 0.27 | 171.2 ± 13.59 |
| LDPE/3 wt. % MWCNT:GNP (1:1) | 164.32 ± 3.48 | 19.46 ± 0.31 | 178.4 ± 18.84 |
| LDPE/5 wt. % MWCNT:GNP (1:1) | 177.78 ± 5.09 | 18.79 ± 0.33 | 163.3 ± 10.77 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Paszkiewicz, S.; Szymczyk, A.; Pawlikowska, D.; Subocz, J.; Zenker, M.; Masztak, R. Electrically and Thermally Conductive Low Density Polyethylene-Based Nanocomposites Reinforced by MWCNT or Hybrid MWCNT/Graphene Nanoplatelets with Improved Thermo-Oxidative Stability. Nanomaterials 2018, 8, 264. https://doi.org/10.3390/nano8040264
Paszkiewicz S, Szymczyk A, Pawlikowska D, Subocz J, Zenker M, Masztak R. Electrically and Thermally Conductive Low Density Polyethylene-Based Nanocomposites Reinforced by MWCNT or Hybrid MWCNT/Graphene Nanoplatelets with Improved Thermo-Oxidative Stability. Nanomaterials. 2018; 8(4):264. https://doi.org/10.3390/nano8040264
Chicago/Turabian StylePaszkiewicz, Sandra, Anna Szymczyk, Daria Pawlikowska, Jan Subocz, Marek Zenker, and Roman Masztak. 2018. "Electrically and Thermally Conductive Low Density Polyethylene-Based Nanocomposites Reinforced by MWCNT or Hybrid MWCNT/Graphene Nanoplatelets with Improved Thermo-Oxidative Stability" Nanomaterials 8, no. 4: 264. https://doi.org/10.3390/nano8040264
APA StylePaszkiewicz, S., Szymczyk, A., Pawlikowska, D., Subocz, J., Zenker, M., & Masztak, R. (2018). Electrically and Thermally Conductive Low Density Polyethylene-Based Nanocomposites Reinforced by MWCNT or Hybrid MWCNT/Graphene Nanoplatelets with Improved Thermo-Oxidative Stability. Nanomaterials, 8(4), 264. https://doi.org/10.3390/nano8040264









