Polymer-Based Electrospun Nanofibers for Biomedical Applications
Abstract
1. Introduction
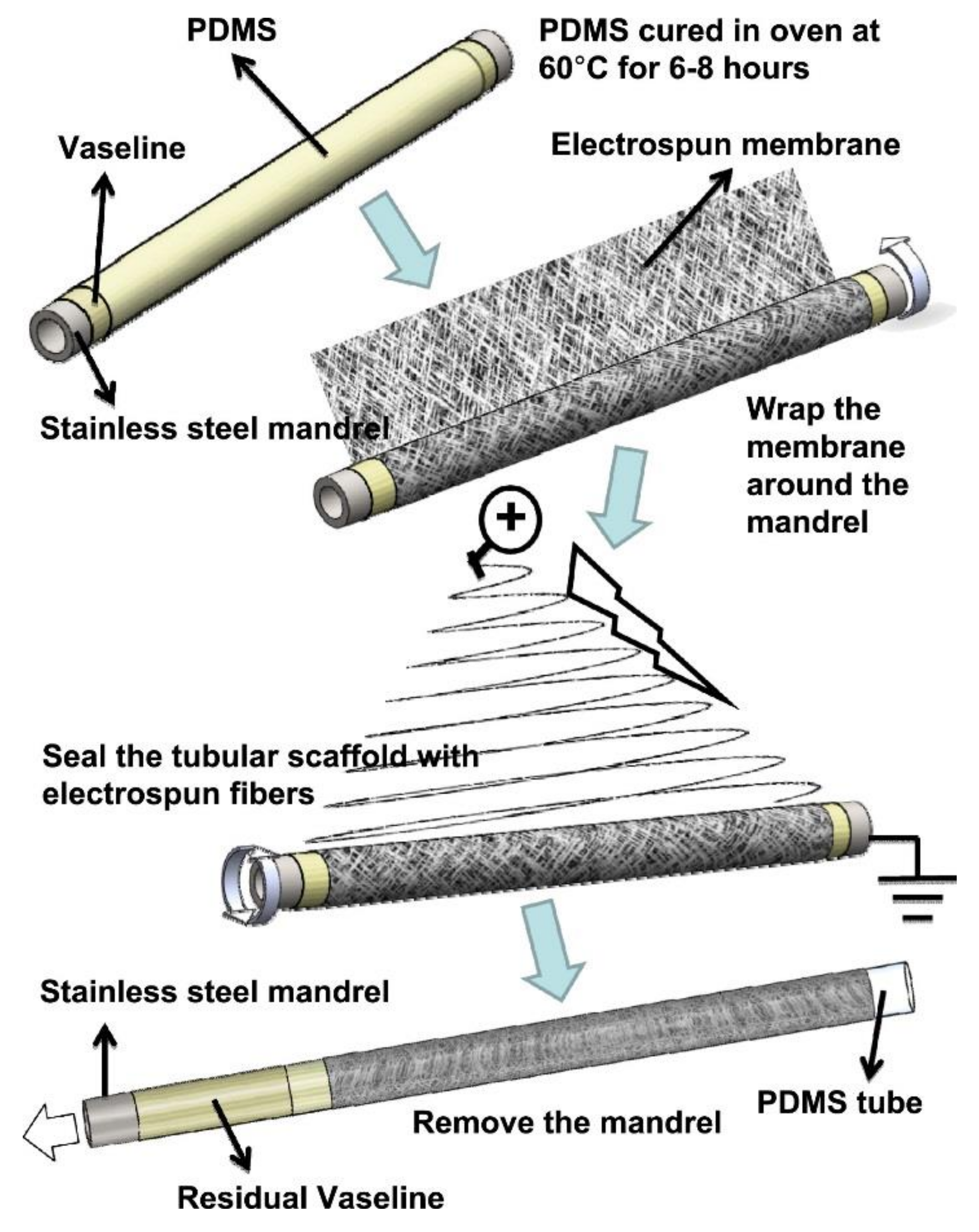
Electrospinning Process
2. Applications of Nanofibers
2.1. Biomedical Applications
2.1.1. Medical Prostheses
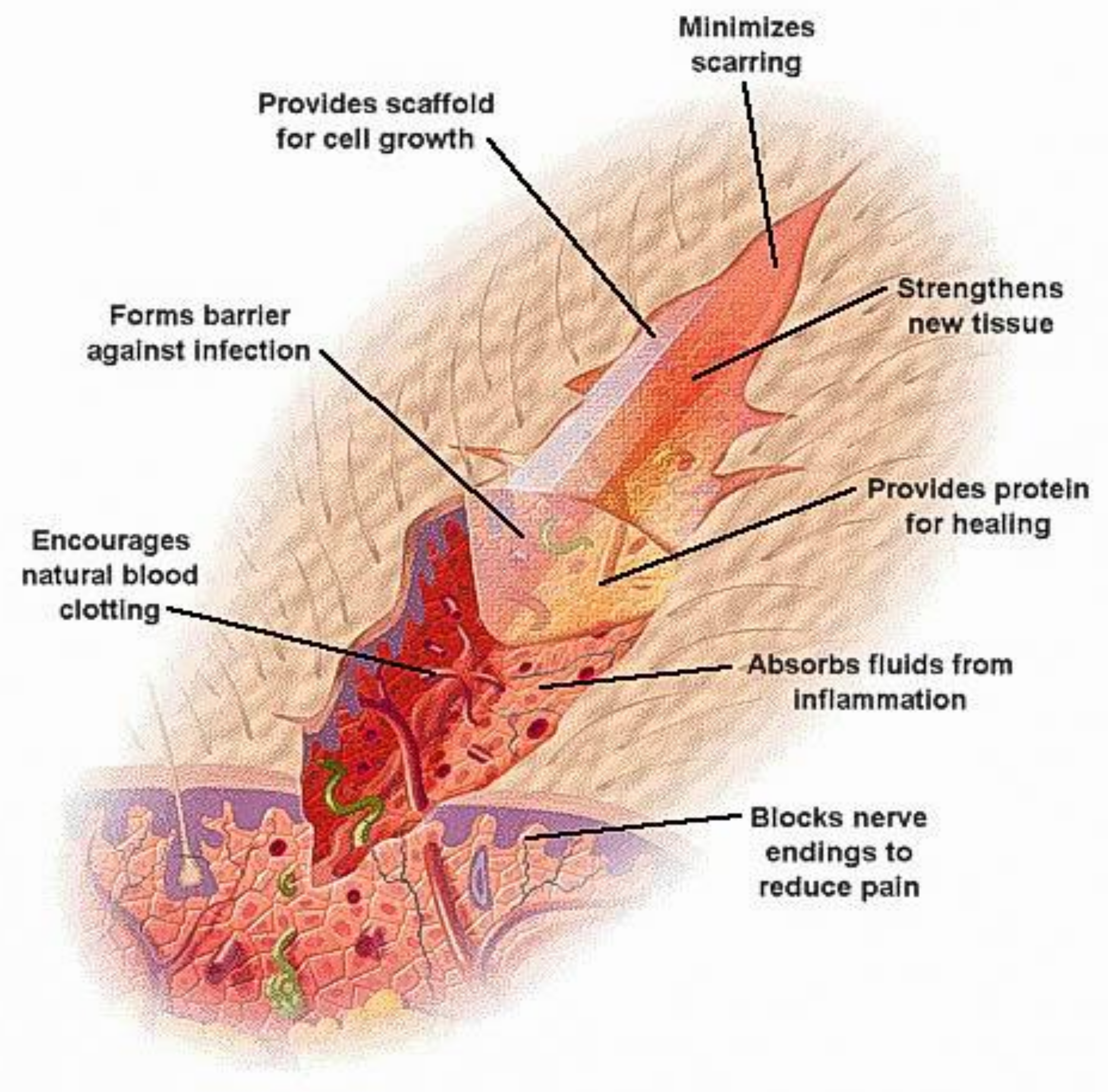
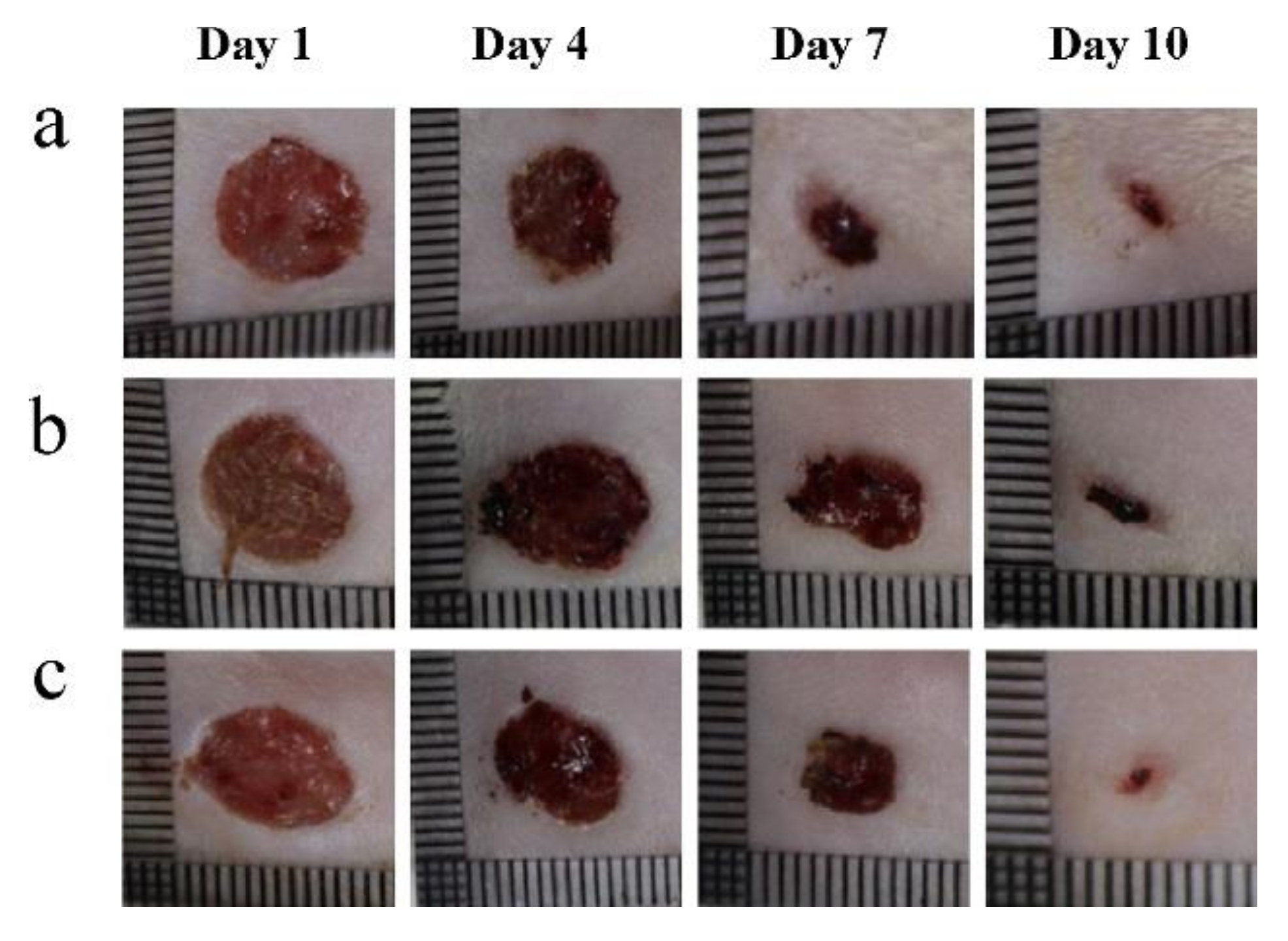
2.1.2. Wound Dressing
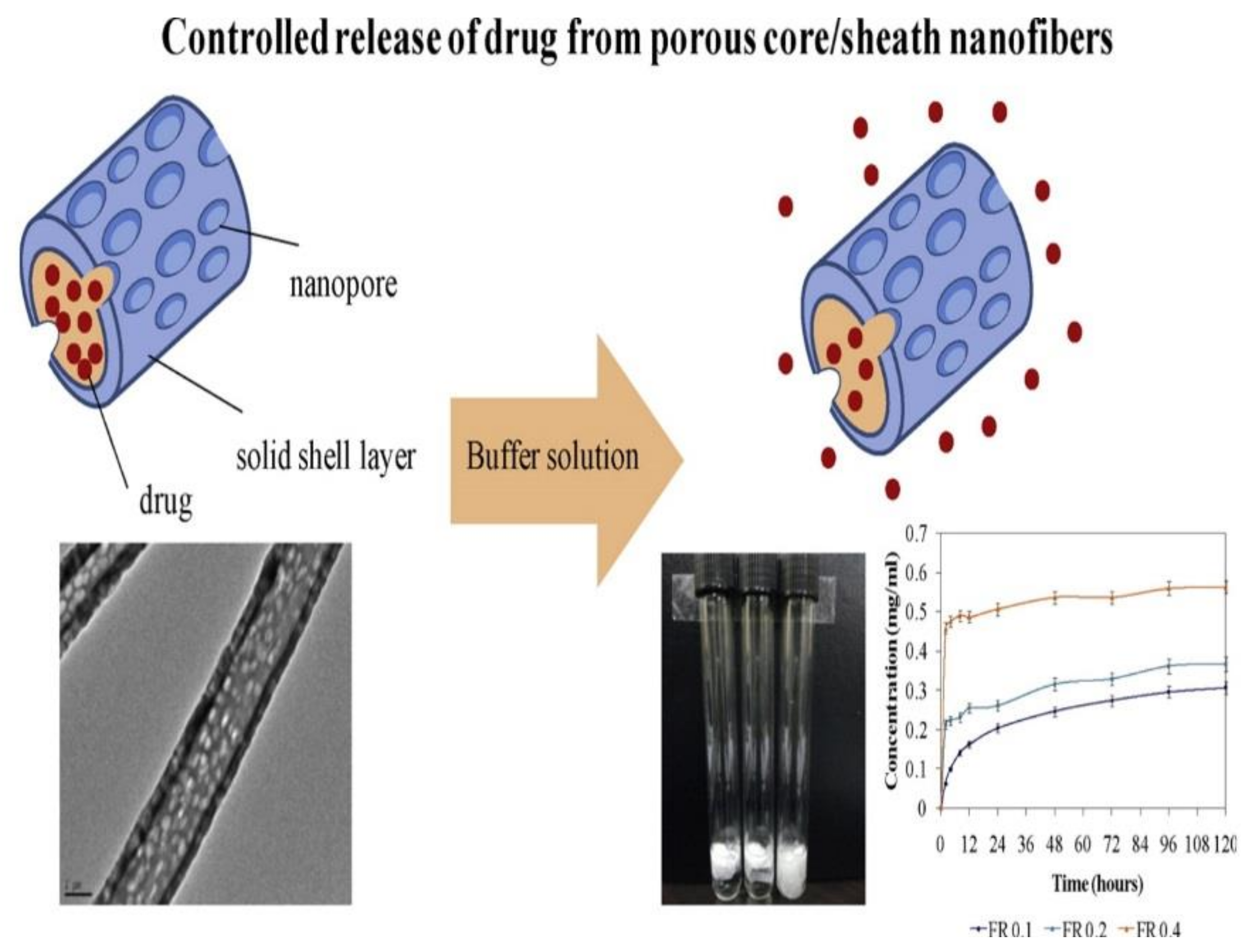
2.1.3. Drug Release
2.1.4. Tissue Engineering
Blood Vessels
Bone
3. Conclusions and Future Perspective
Author Contributions
Conflicts of Interest
References
- Huang, Z.-M.; Zhang, Y.-Z.; Kotaki, M.; Ramakrishna, S. A review on polymer nanofibers by electrospinning and their applications in nanocomposites. Compos. Sci. Technol. 2003, 63, 2223–2253. [Google Scholar] [CrossRef]
- Feng, L.; Li, S.; Li, H.; Zhai, J.; Song, Y.; Jiang, L.; Zhu, D. Super-Hydrophobic Surface of Aligned Polyacrylonitrile Nanofibers. Angew. Chem. Int. Ed. 2002, 41, 1221–1223. [Google Scholar] [CrossRef]
- Liu, G.; Ding, J.; Qiao, L.; Guo, A.; Dymov, B.; Gleeson, J.; Hashimoto, T.; Saijo, K. Polystyrene-block-poly(2-cinnamoylethyl methacrylate) Nanofibers-Preparation, Characterization, and Liquid Crystalline Properties. Chem. A Eur. J. 1999, 5, 2740–2749. [Google Scholar] [CrossRef]
- Whitesides, G.M.; Grzybowski, B. Self-Assembly at All Scales. Science 2002, 295, 2418–2421. [Google Scholar] [CrossRef] [PubMed]
- Ma, P.X.; Zhang, R. Synthetic nano-scale fibrous extracellular matrix. J. Biomed. Mater. Res. 1999, 46, 60–72. [Google Scholar] [CrossRef]
- Deitzel, J.M.; Kleinmeyer, J.D.; Hirvonen, J.K.; Beck Tan, N.C. Controlled deposition and collection of electro-spun poly(ethylene oxide) fibers. Polymer 2001, 42, 8163–8170. [Google Scholar] [CrossRef]
- Salem, D.R. Structure Formation in Polymeric Fibers; Hanser: Munich, Germany, 2001. [Google Scholar]
- Patil, J.V.; Mali, S.S.; Kamble, A.S.; Hong, C.K.; Kim, J.H.; Patil, P.S. Electrospinning: A versatile technique for making of 1D growth of nanostructured nanofibers and its applications: An experimental approach. Appl. Surf. Sci. 2017, 423, 641–674. [Google Scholar] [CrossRef]
- De Marco, I.; Reverchon, E. Nanostructured cellulose acetate filaments produced by supercritical antisolvent precipitation. J. Supercrit. Fluids 2011, 55, 1095–1103. [Google Scholar] [CrossRef]
- Sill, T.J.; von Recum, H.A. Electrospinning: Applications in drug delivery and tissue engineering. Biomaterials 2008, 29, 1989–2006. [Google Scholar] [CrossRef] [PubMed]
- Taylor, G. Electrically Driven Jets. Proc. R. Soc. A Math. Phys. Eng. Sci. 1969, 313, 453–475. [Google Scholar] [CrossRef]
- Shi, X.; Zhou, W.; Ma, D.; Ma, Q.; Bridges, D.; Ma, Y.; Hu, A. Electrospinning of Nanofibers and Their Applications for Energy Devices. J. Nanomater. 2015, 16, 122. [Google Scholar] [CrossRef]
- Ramakrishna, S.; Jose, R.; Archana, P.S.; Nair, A.S.; Balamurugan, R.; Venugopal, J.; Teo, W.E. Science and engineering of electrospun nanofibers for advances in clean energy, water filtration, and regenerative medicine. J. Mater. Sci. 2010, 45, 6283–6312. [Google Scholar] [CrossRef]
- Zagho, M.M.; Elzatahry, A. Recent Trends in Electrospinning of Polymer Nanofibers and their Applications as Templates for Metal Oxide Nanofibers Preparation. In Electrospinning—Material, Techniques, and Biomedical Applications; Haider, S., Ed.; InTech: London, UK, 2016; pp. 3–24. [Google Scholar]
- Khadka, D.B.; Haynie, D.T. Protein- and peptide-based electrospun nanofibers in medical biomaterials. Nanomed. Nanotechnol. Biol. Med. 2012, 8, 1242–1262. [Google Scholar] [CrossRef] [PubMed]
- Hynes, R.O. The Extracellular Matrix: Not Just Pretty Fibrils. Science 2009, 326, 1216–1219. [Google Scholar] [CrossRef] [PubMed]
- Hay, E. Cell Biology of Extracellular Matrix; Plenum: New York, NY, USA, 1991. [Google Scholar]
- Grafahrend, D.; Heffels, K.-H.; Möller, M.; Klee, D.; Groll, J. Electrospun, Biofunctionalized Fibers as Tailored in vitro Substrates for Keratinocyte Cell Culture. Macromol. Biosci. 2010, 10, 1022–1027. [Google Scholar] [CrossRef] [PubMed]
- Doshi, J.; Reneker, D.H. Electrospinning process and applications of electrospun fibers. J. Electrostat. 1995, 35, 151–160. [Google Scholar] [CrossRef]
- Reneker, D.H.; Yarin, A.L. Electrospinning jets and polymer nanofibers. Polymer 2008, 49, 2387–2425. [Google Scholar] [CrossRef]
- Bhardwaj, N.; Kundu, S.C. Electrospinning: A fascinating fiber fabrication technique. Biotechnol. Adv. 2010, 28, 325–347. [Google Scholar] [CrossRef] [PubMed]
- Pham, Q.P.; Sharma, U.; Mikos, A.G. Electrospun Poly(ε-caprolactone) Microfiber and Multilayer Nanofiber/Microfiber Scaffolds: Characterization of Scaffolds and Measurement of Cellular Infiltration. Biomacromolecules 2006, 7, 2796–2805. [Google Scholar] [CrossRef] [PubMed]
- Ahadian, S.; Obregón, R.; Salazar, G.; Ramalingam, M. 20. Clinical/Preclinical Aspects of Nanofiber Composites; Elsevier Ltd.: Amsterdam, The Netherlands, 2017; ISBN 9780081001738. [Google Scholar]
- Jannesari, M.; Varshosaz, J.; Morshed, M.; Zamani, M. Composite poly(vinyl alcohol)/poly(vinyl acetate) electrospun nanofibrous mats as a novel wound dressing matrix for controlled release of drugs. Int. J. Nanomed. 2011, 6, 993–1003. [Google Scholar] [CrossRef]
- Baumgarten, P.K. Electrostatic spinning of acrylic microfibers. J. Colloid Interface Sci. 1971, 36, 71–79. [Google Scholar] [CrossRef]
- Bornat, A. Production of Electrostatically Spun Products. U.S. Patent No. 4,689,186, 25 August 1987. [Google Scholar]
- Hohman, M.M.; Shin, M.; Rutledge, G.; Brenner, M.P. Electrospinning and electrically forced jets. II. Applications. Phys. Fluids 2001, 13, 2221–2236. [Google Scholar] [CrossRef]
- Martin, G.E.; Cockshott, I.D.; Fildes, F.J.T. Fibrillar Lining for Prosthetic Device. U.S. Patent No. 4,044,404, 30 August 1977. [Google Scholar]
- Martin, G.E.; Cockshott, I.D.; Fildes, F.J.T. Fibrillar Product. U.S. Patent No. 4,878,908, 7 November 1989. [Google Scholar]
- Bognitzki, M.; Czado, W.; Frese, T.; Schaper, A.; Hellwig, M.; Steinhart, M.; Greiner, A.; Wendorff, J.H. Nanostructured Fibers via Electrospinning. Adv. Mater. 2001, 13, 70–72. [Google Scholar] [CrossRef]
- The Johns Hopkins University Nanofiber Coating Prevents Infections of Prosthetic Joints. Available online: https://www.mdtmag.com/news/2016/10/nanofiber-coating-prevents-infections-prosthetic-joints (accessed on 25 October 2016).
- Popryadukhin, P.V.; Popov, G.I.; Dobrovolskaya, I.P.; Yudin, V.E.; Vavilov, V.N.; Yukina, G.Y.; Ivan’kova, E.M.; Lebedeva, I.O. Vascular Prostheses Based on Nanofibers from Aliphatic Copolyamide. Cardiovasc. Eng. Technol. 2016, 7, 78–86. [Google Scholar] [CrossRef] [PubMed]
- Semnani, D.; Naghashzargar, E.; Hadjianfar, M.; Manshadi, F.D.; Mohammadi, S.; Karbasi, S.; Effaty, F. Evaluation of PCL/chitosan electrospun nanofibers for liver tissue engineering. Int. J. Polym. Mater. Polym. Biomater. 2017, 66, 149–157. [Google Scholar] [CrossRef]
- Unnithan, A.R.; Barakat, N.A.M.; Pichiah, P.B.T.; Gnanasekaran, G.; Nirmala, R.; Cha, Y.-S.; Jung, C.-H.; El-Newehy, M.; Kim, H.Y. Wound-dressing materials with antibacterial activity from electrospun polyurethane–dextran nanofiber mats containing ciprofloxacin HCl. Carbohydr. Polym. 2012, 90, 1786–1793. [Google Scholar] [CrossRef] [PubMed]
- Wright, J.B.; Lam, K.; Buret, A.G.; Olson, M.E.; Burrell, R.E. Early healing events in a porcine model of contaminated wounds: Effects of nanocrystalline silver on matrix metalloproteinases, cell apoptosis, and healing. Wound Repair Regen. 2002, 10, 141–151. [Google Scholar] [CrossRef] [PubMed]
- Field, C.K.; Kerstein, M.D. Overview of wound healing in a moist environment. Am. J. Surg. 1994, 167, S2–S6. [Google Scholar] [CrossRef]
- Kenawy, E.-R.; Abdel-Fattah, Y.R. Antimicrobial properties of modified and electrospun poly(vinyl phenol). Macromol. Biosci. 2002, 2, 261–266. [Google Scholar] [CrossRef]
- Paul, W.; Sharma, C. Chitosan and alginate wound dressings: A short review. Trends Biomater. Artif. Organs 2004, 18, 18–23. [Google Scholar]
- Jayakumar, R.; Prabaharan, M.; Kumar, P.T.S.; Nair, S.V.; Tamura, H. Biomaterials based on chitin and chitosan in wound dressing applications. Biotechnol. Adv. 2011, 29, 322–337. [Google Scholar] [CrossRef] [PubMed]
- Jayakumar, R.; Prabaharan, M.; Nair, S.V.; Tamura, H. Novel chitin and chitosan nanofibers in biomedical applications. Biotechnol. Adv. 2010, 28, 142–150. [Google Scholar] [CrossRef] [PubMed]
- Jin, H.-J.; Fridrikh, S.V.; Rutledge, G.C.; Kaplan, D.L. Electrospinning Bombyx mori Silk with Poly(ethylene oxide). Biomacromolecules 2002, 3, 1233–1239. [Google Scholar] [CrossRef] [PubMed]
- Shibata, H.; Shioya, N.; Kuroyanagi, Y. Development of new wound dressing composed of spongy collagen sheet containing dibutyryl cyclic AMP. J. Biomater. Sci. Polym. Ed. 1997, 8, 601–621. [Google Scholar] [CrossRef] [PubMed]
- Draye, J.-P.; Delaey, B.; Van de Voorde, A.; Van Den Bulcke, A.; De Reu, B.; Schacht, E. In vitro and in vivo biocompatibility of dextran dialdehyde cross-linked gelatin hydrogel films. Biomaterials 1998, 19, 1677–1687. [Google Scholar] [CrossRef]
- Elsabee, M.Z.; Naguib, H.F.; Morsi, R.E. Chitosan based nanofibers, review. Mater. Sci. Eng. C 2012, 32, 1711–1726. [Google Scholar] [CrossRef]
- Jayakumar, R.; Menon, D.; Manzoor, K.; Nair, S.V.; Tamura, H. Biomedical applications of chitin and chitosan based nanomaterials—A short review. Carbohydr. Polym. 2010, 82, 227–232. [Google Scholar] [CrossRef]
- Chilarski, A.; Szosland, L.; Krucińska, I.; Kiekens, P.; Błasińska, A.; Schoukens, G.; Cisło, R.; Szumilewicz, J. Novel dressing materials accelerating wound healing made from dibutyrylchitin. Fibres Text. East. Eur. 2007, 15, 77–81. [Google Scholar]
- Blasinska, A.; Drobnik, J. Effects of nonwoven mats of di-O-butyrylchitin and related polymers on the process of wound healing. Biomacroloecules 2008, 9, 776–782. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Yang, D.; Chen, X.; Xu, Q.; Lu, F.; Nie, J. Electrospun Water-Soluble Carboxyethyl Chitosan/Poly(vinyl alcohol) Nanofibrous Membrane as Potential Wound Dressing for Skin Regeneration. Biomacromolecules 2008, 9, 349–354. [Google Scholar] [CrossRef] [PubMed]
- Ignatova, M.; Manolova, N.; Rashkov, I. Novel antibacterial fibers of quaternized chitosan and poly(vinyl pyrrolidone) prepared by electrospinning. Eur. Polym. J. 2007, 43, 1112–1122. [Google Scholar] [CrossRef]
- Ignatova, M.; Starbova, K.; Markova, N.; Manolova, N.; Rashkov, I. Electrospun nano-fibre mats with antibacterial properties from quaternised chitosan and poly(vinyl alcohol). Carbohydr. Res. 2006, 341, 2098–2107. [Google Scholar] [CrossRef] [PubMed]
- Watthanaphanit, A.; Supaphol, P.; Tamura, H.; Tokura, S.; Rujiravanit, R. Fabrication, structure, and properties of chitin whisker-reinforced alginate nanocomposite fibers. J. Appl. Polym. Sci. 2008, 110, 890–899. [Google Scholar] [CrossRef]
- Wang, C.-C.; Su, C.-H.; Chen, C.-C. Water absorbing and antibacterial properties of N-isopropyl acrylamide grafted and collagen/chitosan immobilized polypropylene nonwoven fabric and its application on wound healing enhancement. J. Biomed. Mater. Res. Part A 2008, 84A, 1006–1017. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.-P.; Chang, G.-Y.; Chen, J.-K. Electrospun collagen/chitosan nanofibrous membrane as wound dressing. Colloids Surf. A Physicochem. Eng. Asp. 2008, 313–314, 183–188. [Google Scholar] [CrossRef]
- Shalumon, K.T.; Anulekha, K.H.; Nair, S.V.; Nair, S.V.; Chennazhi, K.P.; Jayakumar, R. Sodium alginate/poly(vinyl alcohol)/nano ZnO composite nanofibers for antibacterial wound dressings. Int. J. Biol. Macromol. 2011, 49, 247–254. [Google Scholar] [CrossRef] [PubMed]
- Kossovich, L.Y.; Salkovskiy, Y.; Kirillova, I.V. Electrospun Chitosan Nanofiber Materials as Burn Dressing. In Proceedings of the 6th World Congress of Biomechanics (WCB 2010), Singapore, 1–6 August 2010; Lim, C.T., Goh, J.C.H., Eds.; Springer: Berlin/Heidelberg, Germany, 2010; pp. 1212–1214. [Google Scholar]
- Cai, Z.X.; Mo, X.M.; Zhang, K.H.; Fan, L.P.; Yin, A.L.; He, C.L.; Wang, H.S. Fabrication of chitosan/silk fibroin composite nanofibers for Wound-dressing Applications. Int. J. Mol. Sci. 2010, 11, 3529–3539. [Google Scholar] [CrossRef] [PubMed]
- Chong, E.J.; Phan, T.T.; Lim, I.J.; Zhang, Y.Z.; Bay, B.H.; Ramakrishna, S.; Lim, C.T. Evaluation of electrospun PCL/gelatin nanofibrous scaffold for wound healing and layered dermal reconstitution. Acta Biomater. 2007, 3, 321–330. [Google Scholar] [CrossRef] [PubMed]
- Costache, M.C.; Qu, H.; Ducheyne, P.; Devore, D.I. Polymer-xerogel composites for controlled release wound dressings. Biomaterials 2010, 31, 6336–6343. [Google Scholar] [CrossRef] [PubMed]
- Charernsriwilaiwat, N.; Opanasopit, P.; Rojanarata, T.; Ngawhirunpat, T. Lysozyme-loaded, electrospun chitosan-based nanofiber mats for wound healing. Int. J. Pharm. 2012, 427, 379–384. [Google Scholar] [CrossRef] [PubMed]
- Elzatahry, A.A.; Al-Enizi, A.M.; Elsayed, E.A.; Butorac, R.R.; Al-Deyab, S.S.; Wadaan, M.A.M.; Cowley, A.H. Nanofiber composites containing N-heterocyclic carbene complexes with antimicrobial activity. Int. J. Nanomed. 2012, 7, 2829–2832. [Google Scholar] [CrossRef]
- Li, Y.; Chen, F.; Nie, J.; Yang, D. Electrospun poly(lactic acid)/chitosan core–shell structure nanofibers from homogeneous solution. Carbohydr. Polym. 2012, 90, 1445–1451. [Google Scholar] [CrossRef] [PubMed]
- Rnjak-Kovacina, J.; Wise, S.G.; Li, Z.; Maitz, P.K.M.; Young, C.J.; Wang, Y.; Weiss, A.S. Electrospun synthetic human elastin:collagen composite scaffolds for dermal tissue engineering. Acta Biomater. 2012, 8, 3714–3722. [Google Scholar] [CrossRef] [PubMed]
- Pant, B.; Pant, H.R.; Pandeya, D.R.; Panthi, G.; Nam, K.T.; Hong, S.T.; Kim, C.S.; Kim, H.Y. Characterization and antibacterial properties of Ag NPs loaded nylon-6 nanocomposite prepared by one-step electrospinning process. Colloids Surf. A Physicochem. Eng. Asp. 2012, 395, 94–99. [Google Scholar] [CrossRef]
- Hassan, M.S.; Amna, T.; Sheikh, F.A.; Al-Deyab, S.S.; Choi, K.E.; Hwang, I.H.; Khil, M.-S. Bimetallic Zn/Ag doped polyurethane spider net composite nanofibers: A novel multipurpose electrospun mat. Ceram. Int. 2013, 39, 2503–2510. [Google Scholar] [CrossRef]
- Sofokleous, P.; Stride, E.; Bonfield, W.; Edirisinghe, M. Design, construction and performance of a portable handheld electrohydrodynamic multi-needle spray gun for biomedical applications. Mater. Sci. Eng. C 2013, 33, 213–223. [Google Scholar] [CrossRef] [PubMed]
- Rath, G.; Hussain, T.; Chauhan, G.; Garg, T.; Goyal, A.K. Collagen nanofiber containing silver nanoparticles for improved wound-healing applications. J. Drug Target. 2016, 24, 520–529. [Google Scholar] [CrossRef] [PubMed]
- Dubey, P.; Bhushan, B.; Sachdev, A.; Matai, I.; Uday Kumar, S.; Gopinath, P. Silver-nanoparticle-Incorporated composite nanofibers for potential wound-dressing applications. J. Appl. Polym. Sci. 2015, 132. [Google Scholar] [CrossRef]
- Sadri, M.; Arab-Sorkhi, S.; Vatani, H.; Bagheri-Pebdeni, A. New wound dressing polymeric nanofiber containing green tea extract prepared by electrospinning method. Fibers Polym. 2015, 16, 1742–1750. [Google Scholar] [CrossRef]
- Li, H.; Wang, M.; Williams, G.R.; Wu, J.; Sun, X.; Lv, Y.; Zhu, L.-M. Electrospun gelatin nanofibers loaded with vitamins A and E as antibacterial wound dressing materials. RSC Adv. 2016, 6, 50267–50277. [Google Scholar] [CrossRef]
- Sarhan, W.A.; Azzazy, H.M.E.; El-Sherbiny, I.M. Honey/Chitosan Nanofiber Wound Dressing Enriched with Allium sativum and Cleome droserifolia: Enhanced Antimicrobial and Wound Healing Activity. ACS Appl. Mater. Interfaces 2016, 8, 6379–6390. [Google Scholar] [CrossRef] [PubMed]
- Alavarse, A.C.; de Oliveira Silva, F.W.; Colque, J.T.; da Silva, V.M.; Prieto, T.; Venancio, E.C.; Bonvent, J.-J. Tetracycline hydrochloride-loaded electrospun nanofibers mats based on PVA and chitosan for wound dressing. Mater. Sci. Eng. C 2017, 77, 271–281. [Google Scholar] [CrossRef] [PubMed]
- Pásztor, N.; Rédai, E.; Szabó, Z.-I.; Sipos, E. Preparation and Characterization of Levofloxacin-Loaded Nanofibers as Potential Wound Dressings. Acta Med. Marisiensis 2017, 63, 66–69. [Google Scholar] [CrossRef]
- Jiang, S.; Ma, B.C.; Reinholz, J.; Li, Q.; Wang, J.; Zhang, K.A.I.; Landfester, K.; Crespy, D. Efficient Nanofibrous Membranes for Antibacterial Wound Dressing and UV Protection. ACS Appl. Mater. Interfaces 2016, 8, 29915–29922. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Oh, J.-H.; Park, C.H.; Baek, J.-H.; Ryoo, H.-M.; Woo, K.M. Effects of Dimethyloxalylglycine-Embedded Poly(ε-caprolactone) Fiber Meshes on Wound Healing in Diabetic Rats. ACS Appl. Mater. Interfaces 2017, 9, 7950–7963. [Google Scholar] [CrossRef] [PubMed]
- Kandhasamy, S.; Perumal, S.; Madhan, B.; Umamaheswari, N.; Banday, J.A.; Perumal, P.T.; Santhanakrishnan, V.P. Synthesis and Fabrication of Collagen-Coated Ostholamide Electrospun Nanofiber Scaffold for Wound Healing. ACS Appl. Mater. Interfaces 2017, 9, 8556–8568. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.-D.; Ma, Q.; Wang, K.; Chen, H.-W. Improving Antibacterial Activity and Biocompatibility of Bioinspired Electrospinning Silk Fibroin Nanofibers Modified by Graphene Oxide. ACS Omega 2018, 3, 406–413. [Google Scholar] [CrossRef]
- Kim, K.; Luu, Y.K.; Chang, C.; Fang, D.; Hsiao, B.S.; Chu, B.; Hadjiargyrou, M. Incorporation and controlled release of a hydrophilic antibiotic using poly(lactide-co-glycolide)-based electrospun nanofibrous scaffolds. J. Control. Release 2004, 98, 47–56. [Google Scholar] [CrossRef] [PubMed]
- Ramakrishna, S. An Introduction to Electrospinning and Nanofibers; World Scientific Publishing Company: Singapore, 2005. [Google Scholar]
- Yoo, H.S.; Kim, T.G.; Park, T.G. Surface-functionalized electrospun nanofibers for tissue engineering and drug delivery. Adv. Drug Deliv. Rev. 2009, 61, 1033–1042. [Google Scholar] [CrossRef] [PubMed]
- Zeng, J.; Xu, X.; Chen, X.; Liang, Q.; Bian, X.; Yang, L.; Jing, X. Biodegradable electrospun fibers for drug delivery. J. Control. Release 2003, 92, 227–231. [Google Scholar] [CrossRef]
- Zeng, J.; Yang, L.; Liang, Q.; Zhang, X.; Guan, H.; Xu, X.; Chen, X.; Jing, X. Influence of the drug compatibility with polymer solution on the release kinetics of electrospun fiber formulation. J. Control. Release 2005, 105, 43–51. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Wu, K.; Bosisio, R.G. Six-Port Networks. In Encyclopedia of RF and Microwave Engineering; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2005. [Google Scholar]
- Huang, Z.-Z.; Tang, Y. Unexpected Catalyst for Wittig-Type and Dehalogenation Reactions. J. Org. Chem. 2002, 67, 5320–5326. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.T.T.; Ghosh, C.; Hwang, S.-G.; Chanunpanich, N.; Park, J.S. Porous core/sheath composite nanofibers fabricated by coaxial electrospinning as a potential mat for drug release system. Int. J. Pharm. 2012, 439, 296–306. [Google Scholar] [CrossRef] [PubMed]
- Liang, D.; Hsiao, B.S.; Chu, B. Functional electrospun nanofibrous scaffolds for biomedical applications. Adv. Drug Deliv. Rev. 2007, 59, 1392–1412. [Google Scholar] [CrossRef] [PubMed]
- Kenawy, E.R.; Bowlin, G.L.; Mansfield, K.; Layman, J.; Simpson, D.G.; Sanders, E.H.; Wnek, G.E. Release of tetracycline hydrochloride from electrospun poly(ethylene-co-vinylacetate), poly(lactic acid), and a blend. J. Control. Release 2002, 81, 57–64. [Google Scholar] [CrossRef]
- Verreck, G.; Chun, I.; Peeters, J.; Rosenblatt, J.; Brewster, M.E. Preparation and characterization of nanofibers containing amorphous drug dispersions generated by electrostatic spinning. Pharm. Res. 2003, 20, 810–817. [Google Scholar] [CrossRef] [PubMed]
- Gunn, J.; Zhang, M. Polyblend nanofibers for biomedical applications: Perspectives and challenges. Trends Biotechnol. 2010, 28, 189–197. [Google Scholar] [CrossRef] [PubMed]
- Ma, G.; Liu, Y.; Peng, C.; Fang, D.; He, B.; Nie, J. Paclitaxel loaded electrospun porous nanofibers as mat potential application for chemotherapy against prostate cancer. Carbohydr. Polym. 2011, 86, 505–512. [Google Scholar] [CrossRef]
- Yu, D.-G.; Branford-White, C.; Li, L.; Wu, X.-M.; Zhu, L.-M. The compatibility of acyclovir with polyacrylonitrile in the electrospun drug-loaded nanofibers. J. Appl. Polym. Sci. 2010, 117, 1509–1515. [Google Scholar] [CrossRef]
- Huang, Z.-M.; He, C.-L.; Yang, A.; Zhang, Y.; Han, X.-J.; Yin, J.; Wu, Q. Encapsulating drugs in biodegradable ultrafine fibers through co-axial electrospinning. J. Biomed. Mater. Res. Part A 2006, 77A, 169–179. [Google Scholar] [CrossRef] [PubMed]
- He, C.-L.; Huang, Z.-M.; Han, X.-J. Fabrication of drug-loaded electrospun aligned fibrous threads for suture applications. J. Biomed. Mater. Res. Part A 2009, 89A, 80–95. [Google Scholar] [CrossRef] [PubMed]
- Xie, J.; Ng, W.J.; Lee, L.Y.; Wang, C.-H. Encapsulation of protein drugs in biodegradable microparticles by co-axial electrospray. J. Colloid Interface Sci. 2008, 317, 469–476. [Google Scholar] [CrossRef] [PubMed]
- Tiwari, S.K.; Tzezana, R.; Zussman, E.; Venkatraman, S.S. Optimizing partition-controlled drug release from electrospun core-shell fibers. Int. J. Pharm. 2010, 392, 209–217. [Google Scholar] [CrossRef] [PubMed]
- Vukomanović, M.; Škapin, S.D.; Poljanšek, I.; Žagar, E.; Kralj, B.; Ignjatović, N.; Uskoković, D. Poly(d,l-lactide-co-glycolide)/hydroxyapatite core-shell nanosphere. Part 2: Simultaneous release of a drug and a prodrug (clindamycin and clindamycin phosphate). Colloids Surf. B Biointerfaces 2011, 82, 414–421. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.-N.; Mo, H.-Y.; Yu, D.-G. Electrospun drug-loaded core–sheath PVP/zein nanofibers for biphasic drug release. Int. J. Pharm. 2012, 438, 232–239. [Google Scholar] [CrossRef] [PubMed]
- Song, B.; Wu, C.; Chang, J. Dual drug release from electrospun poly(lactic-co-glycolic acid)/mesoporous silica nanoparticles composite mats with distinct release profiles. Acta Biomater. 2012, 8, 1901–1907. [Google Scholar] [CrossRef] [PubMed]
- Loh, X.J.; Peh, P.; Liao, S.; Sng, C.; Li, J. Controlled drug release from biodegradable thermoresponsive physical hydrogel nanofibers. J. Control. Release 2010, 143, 175–182. [Google Scholar] [CrossRef] [PubMed]
- Goonoo, N.; Bhaw-Luximon, A.; Jhurry, D. Drug loading and release from electrospun biodegradable nanofibers. J. Biomed. Nanotechnol. 2014, 10, 2173–2199. [Google Scholar] [CrossRef] [PubMed]
- Demirci, S.; Celebioglu, A.; Aytac, Z.; Uyar, T. pH-responsive nanofibers with controlled drug release properties. Polym. Chem. 2014, 5, 2050–2056. [Google Scholar] [CrossRef]
- Slemming-Adamsen, P.; Song, J.; Dong, M.; Besenbacher, F.; Chen, M. In Situ Cross-Linked PNIPAM/Gelatin Nanofibers for Thermo-Responsive Drug Release. Macromol. Mater. Eng. 2015, 300, 1226–1231. [Google Scholar] [CrossRef]
- Zahedi, P.; Fallah-Darrehchi, M. Electrospun egg albumin-PVA nanofibers containing tetracycline hydrochloride: Morphological, drug release, antibacterial, thermal and mechanical properties. Fibers Polym. 2015, 16, 2184–2192. [Google Scholar] [CrossRef]
- Geiger, B.C.; Nelson, M.T.; Munj, H.R.; Tomasko, D.L.; Lannutti, J.J. Dual drug release from CO2-infused nanofibers via hydrophobic and hydrophilic interactions. J. Appl. Polym. Sci. 2015, 132. [Google Scholar] [CrossRef]
- Zupančič, Š.; Sinha-Ray, S.; Sinha-Ray, S.; Kristl, J.; Yarin, A.L. Controlled Release of Ciprofloxacin from Core–Shell Nanofibers with Monolithic or Blended Core. Mol. Pharm. 2016, 13, 1393–1404. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.-J.; Ebara, M.; Aoyagi, T. A Smart Hyperthermia Nanofiber with Switchable Drug Release for Inducing Cancer Apoptosis. Adv. Funct. Mater. 2013, 23, 5753–5761. [Google Scholar] [CrossRef]
- Fiorati, A.; Turco, G.; Travan, A.; Caneva, E.; Pastori, N.; Cametti, M.; Punta, C.; Melone, L. Mechanical and drug release properties of sponges from cross-linked cellulose nanofibers. Chempluschem 2017, 82, 848–858. [Google Scholar] [CrossRef]
- Mu, C.; Wu, Q. Electrospun Poly(ε-caprolactone) Composite Nanofibers with Controlled Release of Cis-Diamminediiodoplatinum for a Higher Anticancer Activity. Nanoscale Res. Lett. 2017, 12. [Google Scholar] [CrossRef] [PubMed]
- Laurencin, C.T.; Ambrosio, A.M.A.; Borden, M.D.; Cooper, J.A. Tissue Engineering: Orthopedic Applications. Annu. Rev. Biomed. Eng. 1999, 1, 19–46. [Google Scholar] [CrossRef] [PubMed]
- Buchko, C.J.; Chen, L.C.; Shen, Y.; Martin, D.C. Processing and microstructural characterization of porous biocompatible protein polymer thin films. Polymer 1999, 40, 7397–7407. [Google Scholar] [CrossRef]
- Fertala, A.; Han, W.B.; Ko, F.K. Mapping critical sites in collagen II for rational design of gene-engineered proteins for cell-supporting materials. J. Biomed. Mater. Res. 2001, 57, 48–58. [Google Scholar] [CrossRef]
- Huang, L.; McMillan, R.A.; Apkarian, R.P.; Pourdeyhimi, B.; Conticello, V.P.; Chaikof, E.L. Generation of Synthetic Elastin-Mimetic Small Diameter Fibers and Fiber Networks. Macromolecules 2000, 33, 2989–2997. [Google Scholar] [CrossRef]
- Li, C.; Vepari, C.; Jin, H.-J.; Kim, H.J.; Kaplan, D.L. Electrospun silk-BMP-2 scaffolds for bone tissue engineering. Biomaterials 2006, 27, 3115–3124. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Feng, Y.; Fang, Z.; Yuan, W.; Khan, M. Co-electrospun blends of PU and PEG as potential biocompatible scaffolds for small-diameter vascular tissue engineering. Mater. Sci. Eng. C 2012, 32, 2306–2315. [Google Scholar] [CrossRef]
- Coombes, A.G.A.; Verderio, E.; Shaw, B.; Li, X.; Griffin, M.; Downes, S. Biocomposites of non-crosslinked natural and synthetic polymers. Biomaterials 2002, 23, 2113–2118. [Google Scholar] [CrossRef]
- Xu, C.Y.; Inai, R.; Kotaki, M.; Ramakrishna, S. Aligned biodegradable nanofibrous structure: A potential scaffold for blood vessel engineering. Biomaterials 2004, 25, 877–886. [Google Scholar] [CrossRef]
- Drilling, S.; Gaumer, J.; Lannutti, J. Fabrication of burst pressure competent vascular grafts via electrospinning: Effects of microstructure. J. Biomed. Mater. Res. Part A 2009, 88A, 923–934. [Google Scholar] [CrossRef] [PubMed]
- Pektok, E.; Nottelet, B.; Tille, J.C.; Gurny, R.; Kalangos, A.; Moeller, M.; Walpoth, B.H. Degradation and healing characteristics of small-diameter poly(ε-caprolactone) vascular grafts in the rat systemic arterial circulation. Circulation 2008, 118, 2563–2570. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Nagapudi, K.; Apkarian, R.P.; Chaikof, E.L. Engineered collagen—PEO nanofibers and fabrics. J. Biomater. Sci. Polym. Ed. 2001, 12, 979–993. [Google Scholar] [CrossRef] [PubMed]
- Stitzel, J.; Liu, J.; Lee, S.J.; Komura, M.; Berry, J.; Soker, S.; Lim, G.; Van Dyke, M.; Czerw, R.; Yoo, J.J.; et al. Controlled fabrication of a biological vascular substitute. Biomaterials 2006, 27, 1088–1094. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.-J.; Chao, W.-C.; Lee, P.-Y.; Huang, C.-H. Construction and characterization of an electrospun tubular scaffold for small-diameter tissue-engineered vascular grafts: A scaffold membrane approach. J. Mech. Behav. Biomed. Mater. 2012, 13, 140–155. [Google Scholar] [CrossRef] [PubMed]
- Theron, A.; Zussman, E.; Yarin, A.L. Electrostatic field-assisted alignment of electrospun nanofibres. Nanotechnology 2001, 12, 384. [Google Scholar] [CrossRef]
- Li, D.; Wang, Y.; Xia, Y. Electrospinning of Polymeric and Ceramic Nanofibers as Uniaxially Aligned Arrays. Nano Lett. 2003, 3, 1167–1171. [Google Scholar] [CrossRef]
- Vatankhah, E.; Prabhakaran, M.P.; Semnani, D.; Razavi, S.; Morshed, M.; Ramakrishna, S. Electrospun tecophilic/gelatin nanofibers with potential for small diameter blood vessel tissue engineering. Biopolymers 2014, 101, 1165–1180. [Google Scholar] [CrossRef] [PubMed]
- Syazana, N.; Sukmana, I. Electrospun-based fibrous scaffold for cardiovascular engineering applications: A review. ARPN J. Eng. Appl. Sci. 2016, 11, 4778–4781. [Google Scholar]
- Yalcin Enis, I.; Gok Sadikoglu, T. Design parameters for electrospun biodegradable vascular grafts. J. Ind. Text. 2016. [Google Scholar] [CrossRef]
- Bertram, U.; Steiner, D.; Poppitz, B.; Dippold, D.; Köhn, K.; Beier, J.P.; Detsch, R.; Boccaccini, A.R.; Schubert, D.W.; Horch, R.E.; et al. Vascular Tissue Engineering: Effects of Integrating Collagen into a PCL Based Nanofiber Material. Biomed. Res. Int. 2017. [Google Scholar] [CrossRef] [PubMed]
- Abdal-hay, A.; Bartnikowski, M.; Hamlet, S.; Ivanovski, S. Electrospun biphasic tubular scaffold with enhanced mechanical properties for vascular tissue engineering. Mater. Sci. Eng. C 2018, 82, 10–18. [Google Scholar] [CrossRef] [PubMed]
- Castano, I.; Eltohamy, M.; Kim, H.W. Electrospinning technology in tissue regeneration. Methods Mol. Biol. 2010, 811, 127–140. [Google Scholar] [CrossRef]
- Shin, S.-H.; Purevdorj, O.; Castano, O.; Planell, J.A.; Kim, H.-W. A short review: Recent advances in electrospinning for bone tissue regeneration. J. Tissue Eng. 2012, 3. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.-C.; Chang, J.-J.; Lee, Y.-H.; Lin, Y.-C.; Wu, M.-H.; Yang, M.-C.; Chien, C.-T. Electrospun scaffolds composing of alginate, chitosan, collagen and hydroxyapatite for applying in bone tissue engineering. Mater. Lett. 2013, 93, 133–136. [Google Scholar] [CrossRef]
- Boakye, M.A.D.; Rijal, N.P.; Adhikari, U.; Bhattarai, N. Fabrication and Characterization of Electrospun PCL-MgO-Keratin-Based Composite Nanofibers for Biomedical Applications. Materials 2015, 8, 4080–4095. [Google Scholar] [CrossRef] [PubMed]
- Jo, Y.Y.; Kim, S.G.; Kwon, K.J.; Kweon, H.Y.; Chae, W.S.; Yang, W.G.; Lee, E.Y.; Seok, H. Silk fibroin-alginate-hydroxyapatite composite particles in bone tissue engineering applications in vivo. Int. J. Mol. Sci. 2017, 18, 858. [Google Scholar] [CrossRef] [PubMed]
- Farokhi, M.; Mottaghitalab, F.; Samani, S.; Shokrgozar, M.A.; Kundu, S.C.; Reis, R.L.; Fatahi, Y.; Kaplan, D.L. Silk fibroin/hydroxyapatite composites for bone tissue engineering. Biotechnol. Adv. 2018, 36, 68–91. [Google Scholar] [CrossRef] [PubMed]
- Seo, S.J.; Kim, H.W.; Lee, J.H. Electrospun Nanofibers Applications in Dentistry. J. Nanomater. 2016, 2016. [Google Scholar] [CrossRef]
- Gao, X.; Song, J.; Zhang, Y.; Xu, X.; Zhang, S.; Ji, P.; Wei, S. Bioinspired Design of Polycaprolactone Composite Nanofibers as Artificial Bone Extracellular Matrix for Bone Regeneration Application. ACS Appl. Mater. Interfaces 2016, 8, 27594–27610. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, K.; Prabakaran, M.; Ke, M.; Gang, X.; Chung, I.M.; Um, I.C.; Gopiraman, M.; Kim, I.S. Highly dispersed nanoscale hydroxyapatite on cellulose nanofibers for bone regeneration. Mater. Lett. 2016, 168, 56–61. [Google Scholar] [CrossRef]
- Guler, Z.; Silva, J.C.; Sezai Sarac, A. RGD functionalized poly(ε-caprolactone)/poly(m-anthranilic acid) electrospun nanofibers as high-performing scaffolds for bone tissue engineering RGD functionalized PCL/P3ANA nanofibers. Int. J. Polym. Mater. Polym. Biomater. 2017, 66, 139–148. [Google Scholar] [CrossRef]
- Nagarajan, S.; Belaid, H.; Pochat-Bohatier, C.; Teyssier, C.; Iatsunskyi, I.; Coy, E.; Balme, S.; Cornu, D.; Miele, P.; Kalkura, N.S.; et al. Design of Boron Nitride/Gelatin Electrospun Nanofibers for Bone Tissue Engineering. ACS Appl. Mater. Interfaces 2017, 9, 33695–33706. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Gleeson, S.E.; Yu, T.; Khan, N.; Yucha, R.W.; Marcolongo, M.; Li, C.Y. Hierarchically ordered polymer nanofiber shish kebabs as a bone scaffold material. J. Biomed. Mater. Res. Part A 2017, 105, 1786–1798. [Google Scholar] [CrossRef] [PubMed]
- Zhou, T.; Li, G.; Lin, S.; Tian, T.; Ma, Q.; Zhang, Q.; Shi, S.; Xue, C.; Ma, W.; Cai, X.; et al. Electrospun Poly(3-hydroxybutyrate-co-4-hydroxybutyrate)/Graphene Oxide Scaffold: Enhanced Properties and Promoted In Vivo Bone Repair in Rats. ACS Appl. Mater. Interfaces 2017, 9, 42589–42600. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.; Samanta, A.; Srivastava, R.K.; Hakkarainen, M. Starch-Derived Nanographene Oxide Paves the Way for Electrospinnable and Bioactive Starch Scaffolds for Bone Tissue Engineering. Biomacromolecules 2017, 18, 1582–1591. [Google Scholar] [CrossRef] [PubMed]
- Qian, Y.; Zhou, X.; Sun, H.; Yang, J.; Chen, Y.; Li, C.; Wang, H.; Xing, T.; Zhang, F.; Gu, N. Biomimetic Domain-Active Electrospun Scaffolds Facilitating Bone Regeneration Synergistically with Antibacterial Efficacy for Bone Defects. ACS Appl. Mater. Interfaces 2018, 10, 3248–3259. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Wang, J.; Jin, L.; Li, Y.; Wang, Z. Effects of Polyacrylonitrile/MoS2 Composite Nanofibers on the Growth Behavior of Bone Marrow Mesenchymal Stem Cells. ACS Appl. Nano Mater. 2018, 1, 337–343. [Google Scholar] [CrossRef]
- Qu, Y.; Wang, B.; Chu, B.; Liu, C.; Rong, X.; Chen, H.; Peng, J.; Qian, Z. Injectable and Thermosensitive Hydrogel and PDLLA Electrospun Nanofiber Membrane Composites for Guided Spinal Fusion. ACS Appl. Mater. Interfaces 2018, 10, 4462–4470. [Google Scholar] [CrossRef] [PubMed]


| Application | Fibrous Material | Reference | |
|---|---|---|---|
| Medical prostheses | Copolymer of ε–caprolactam and hexamethylendiaminadipate | Popryadukhin et al. [32]. | |
| Polycaprolactone (PCL)/chitosan (CS) | Semnani et al. [33] | ||
| Wound dressing | Polyurethane (PU)/dextran | Unnithan et al. [34] | |
| Alginate/chitin | Jayakumar et al. [45,51] | ||
| Dibutyrylchitin (DBC) | Chilarski et al. [46] | ||
| Carboxyethyl chitosan/poly(vinyl alcohol) (CECS/PVA) | Zhou et al. [48] | ||
| Quaternary chitosan (QCS) | Ignatova et al. [49] | ||
| CS/collagen | Wang et al. [52] | ||
| Polyethylene/chitosan/type 1 collagen | Chen et al. [53] | ||
| Sodium alginate (SA)/PVA | Shalumon et al. [54] | ||
| Silk fibroin/CS | Cai et al. [56] | ||
| PCL/gelatin | Chong et al. [57] | ||
| PVA/CS-ethylenediaminetetraacetic acid (CS-EDTA) | Charernsriwilaiwat et al. [59] | ||
| N-heterocyclic carbene complexes | Elzatahry et al. [60] | ||
| Poly(lactic acid)(PLA)/CS | Li et al. [61] | ||
| Collagen/tropoelastin | Rnjak-Kovacina et al. [62] | ||
| Nylon-6/Ag nanoparticles (Ag NPs) | Pant et al. [63] | ||
| ZnO/Ag/PU | Shamshi Hassan et al. [64] | ||
| Collagen/Ag NPs | Rath et al. [66] | ||
| Poly(ethylene oxide) (PEO)/PCL/Ag NPs | Dubey et al. [67] | ||
| CS/PEO/green tea extract | Sadri et al. [68] | ||
| Gelatin/vitamins A and E | Li et al. [69] | ||
| Honey/CS/Cleome droserifolia and Allium sativum | Sarhan et al. [70] | ||
| PVA/CS/tetracycline hydrochloride | Alavarse et al. [71] | ||
| PCL/Levofloxacin | Pásztor et al. [72] | ||
| Dimethyloxalylglycine-embedded PCL | Zhang et al. [74] | ||
| Collagen-coated ostholamide | Kandhasamy et al. [75] | ||
| Silk fibroin (SF)/graphene oxide (GO) | Wang et al. [76] | ||
| Drug release | PLA/ poly(ethylene-co-vinyl acetate) (PEVA)/tetracycline hydrochloride | Kenawy et al. [86] | |
| Hydroxylpropylmethyl cellulose (HPMC)/Itraconazole | Verreck et al. [87] | ||
| Poly(d,l-lactide-co-glycolide)/hydroxyapatite (PLGA/HAp)/clindamycin-base and clindamycin-2-phosphate | Vukomanovića et al. [95] | ||
| Zein/polyvinylpyrrolidone/ketoprofen (KET) | Jiang et al. [96] | ||
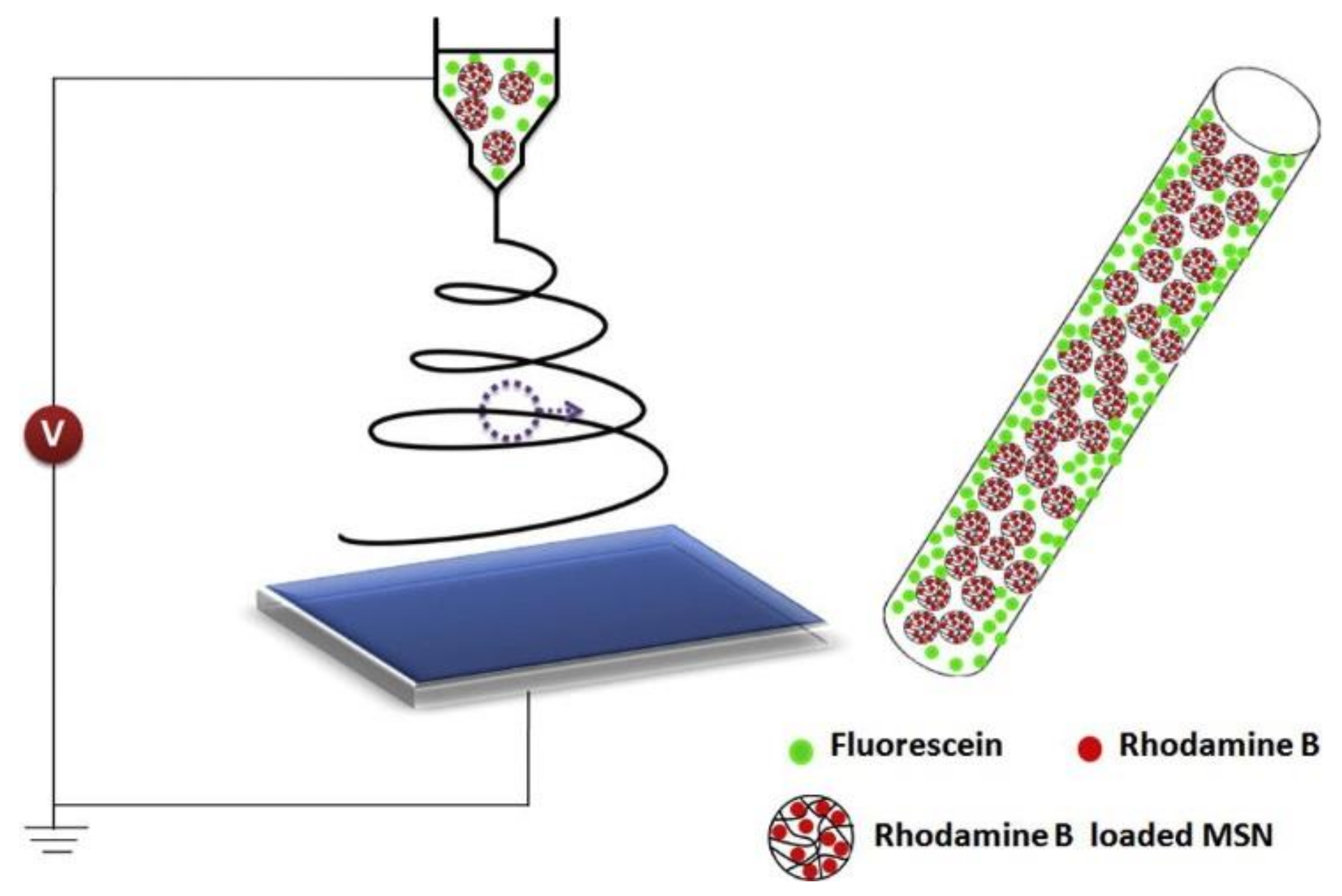
| Rhodamine B (RHB) and fluorescein (FLU)/poly(lactic-co-glycolic acid) (PLGA)/mesoporous silica NPs (MSNPs) | Song et al. [97] | ||
| Poly(4-vinylbenzoic acid-co-(ar-vinylbenzyl)trimethylammonium chloride) [poly(VBA-co-VBTAC)]/ciprofloxacin | Demirci et al. [100] | ||
| Poly(N-isopropylacrylamide) (PNIPAM)/gelatin | Slemming-Adamsen et al. [101] | ||
| Egg albumin/PVA | Zahedi et al. [102] | ||
| PCL/PCL–gelatin/BODIPY 493/503 and Rhodamine B fluorescent | Geiger et al. [103] | ||
| Poly(methyl methacrylate) (PMMA)/PVA/ciprofloxacin hydrochloride (CIP) | Zupančič et al. [104] | ||
| Oxidized cellulose/branched polyethyleneimine (bPEI)/amoxicillin (AM) and ibuprofen (IB) | Fiorati et al. [106] | ||
| cis-diamminediiodoplatinum (cis-DIDP)/PCL | Mu et al. [107] | ||
| Tissue engineering | Blood vessels | PEG/PU | Wang et al. [113] |
| PCL | Drilling et al. [116] and Hu et al. [120] | ||
| PCL and polytetrafluoroethylene (e-PTFE) | Pektok et al. [117] | ||
| Type I collagen-PEO | Huang et al. [118] | ||
| Polyglycolic acid/collagen/elastin | Stitzel et al. [119] | ||
| Gelatin/tecophilic | Vatankhah et al. [123] | ||
| Collagen/PCL | Bertram et al. [126] | ||
| PU/PCL | Abdal-hay et al. [127] | ||
| Bone | CS/alginate/AHp/collagen | Yu et al. [130] | |
| PCL/MgO/keratin | Boakye et al. [131] | ||
| PCL/HA | Gao et al. [135] | ||
| HA/cellulose | Yamaguchi et al. [136] | ||
| PCL/poly(m-anthranilic acid) | Guler et al. [137] | ||
| Boron nitride/gelatin | Nagarajan et al. [138] | ||
| PCL-b-poly(acrylic acid) copolymer/PCL | Chen at al. [139] | ||
| Poly(3-hydroxybutyrate-co-4-hydroxybutyrate)/graphene oxide | Zhou et al. [140] | ||
| Starch/GO | Wu et al. [141] | ||
| Chlorhexidine-doped-PLGA/PCL, PLGA/PCL and β–tricalcium phosphate-doped-PLGA/PCL | Qian et al. [142] | ||
| Polyacrylonitrile/MoS2 | Wu et al. [143] | ||
| Poly(d,l-lactide) (PDLLA) | Qu et al. [144] | ||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Al-Enizi, A.M.; Zagho, M.M.; Elzatahry, A.A. Polymer-Based Electrospun Nanofibers for Biomedical Applications. Nanomaterials 2018, 8, 259. https://doi.org/10.3390/nano8040259
Al-Enizi AM, Zagho MM, Elzatahry AA. Polymer-Based Electrospun Nanofibers for Biomedical Applications. Nanomaterials. 2018; 8(4):259. https://doi.org/10.3390/nano8040259
Chicago/Turabian StyleAl-Enizi, Abdullah M., Moustafa M. Zagho, and Ahmed A. Elzatahry. 2018. "Polymer-Based Electrospun Nanofibers for Biomedical Applications" Nanomaterials 8, no. 4: 259. https://doi.org/10.3390/nano8040259
APA StyleAl-Enizi, A. M., Zagho, M. M., & Elzatahry, A. A. (2018). Polymer-Based Electrospun Nanofibers for Biomedical Applications. Nanomaterials, 8(4), 259. https://doi.org/10.3390/nano8040259





