Enhancing the Microparticle Deposition Stability and Homogeneity on Planer for Synthesis of Self-Assembly Monolayer
Abstract
:1. Introduction
2. Experimental and Simulation Setup
2.1. Aerosol Generation and Deposition System
2.2. Measurement and Analysis of Microparticle Deposition
2.3. Simulation Method
3. Results and Discussion
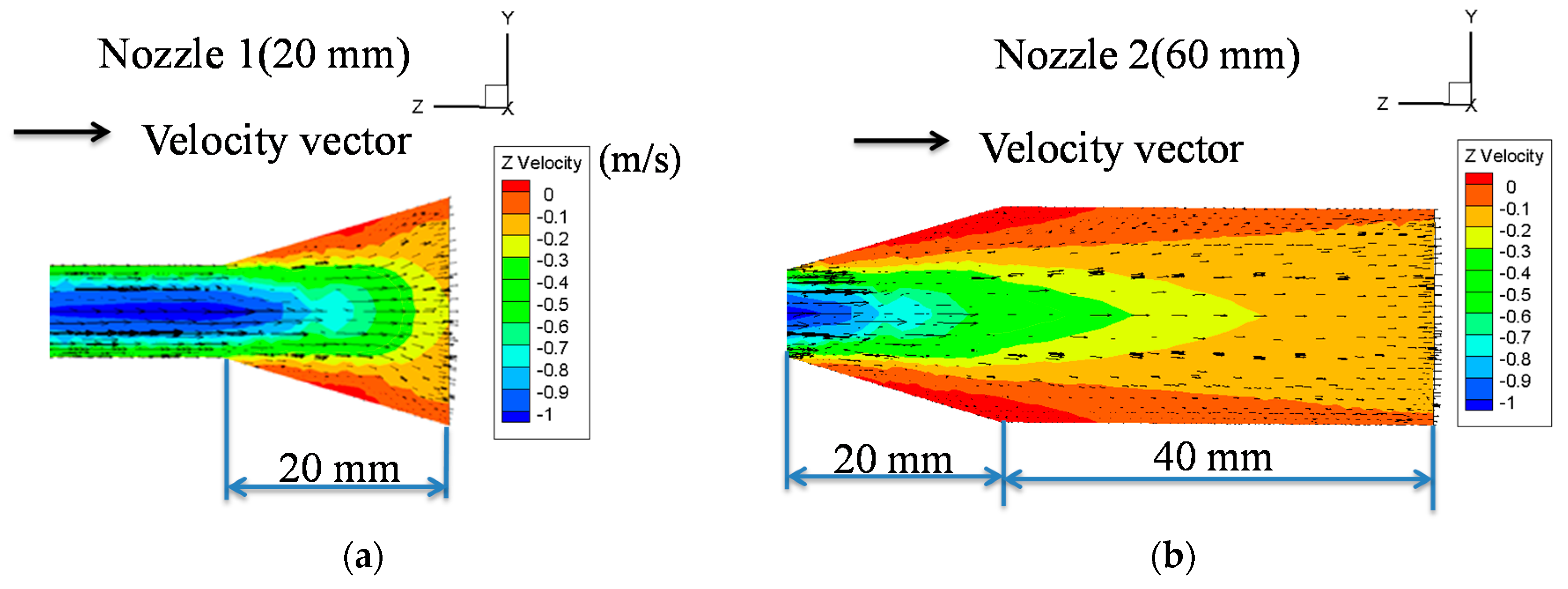
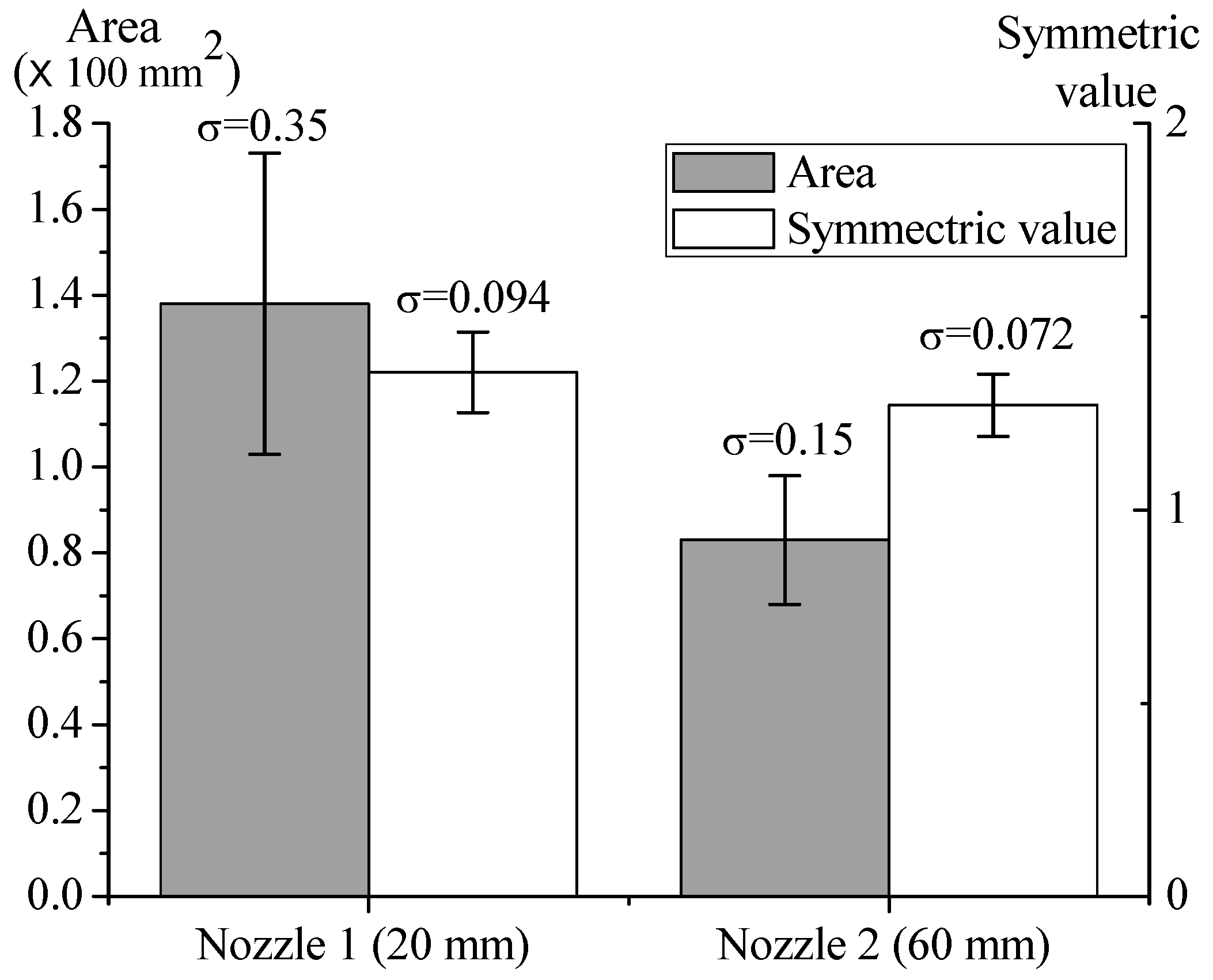
3.1. Nozzle Length and Flowrate Effects on the Stability of Deposited Microparticle Layer
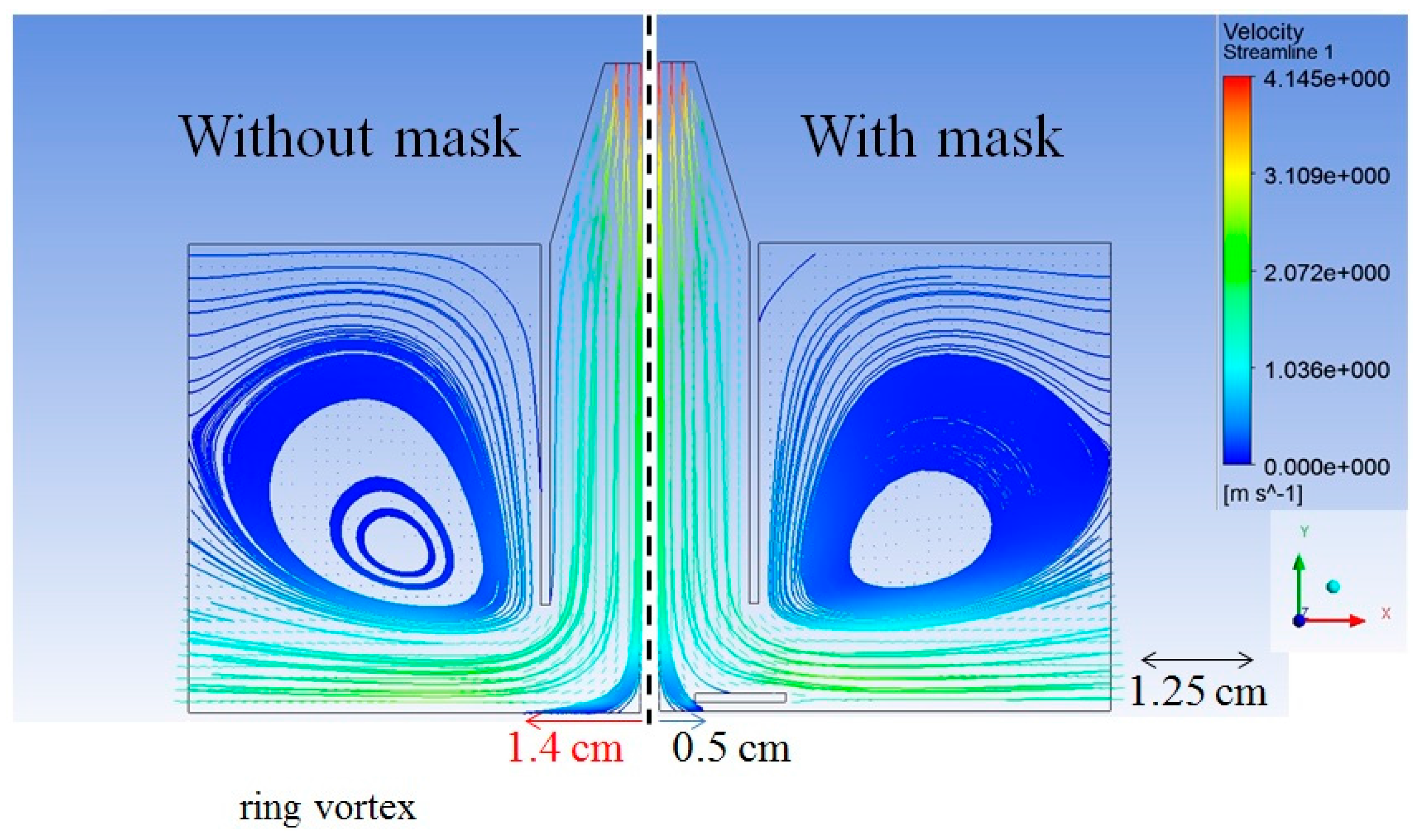
3.2. Mask for Removing the Ring Vortex and Enhancing Deposition Stability
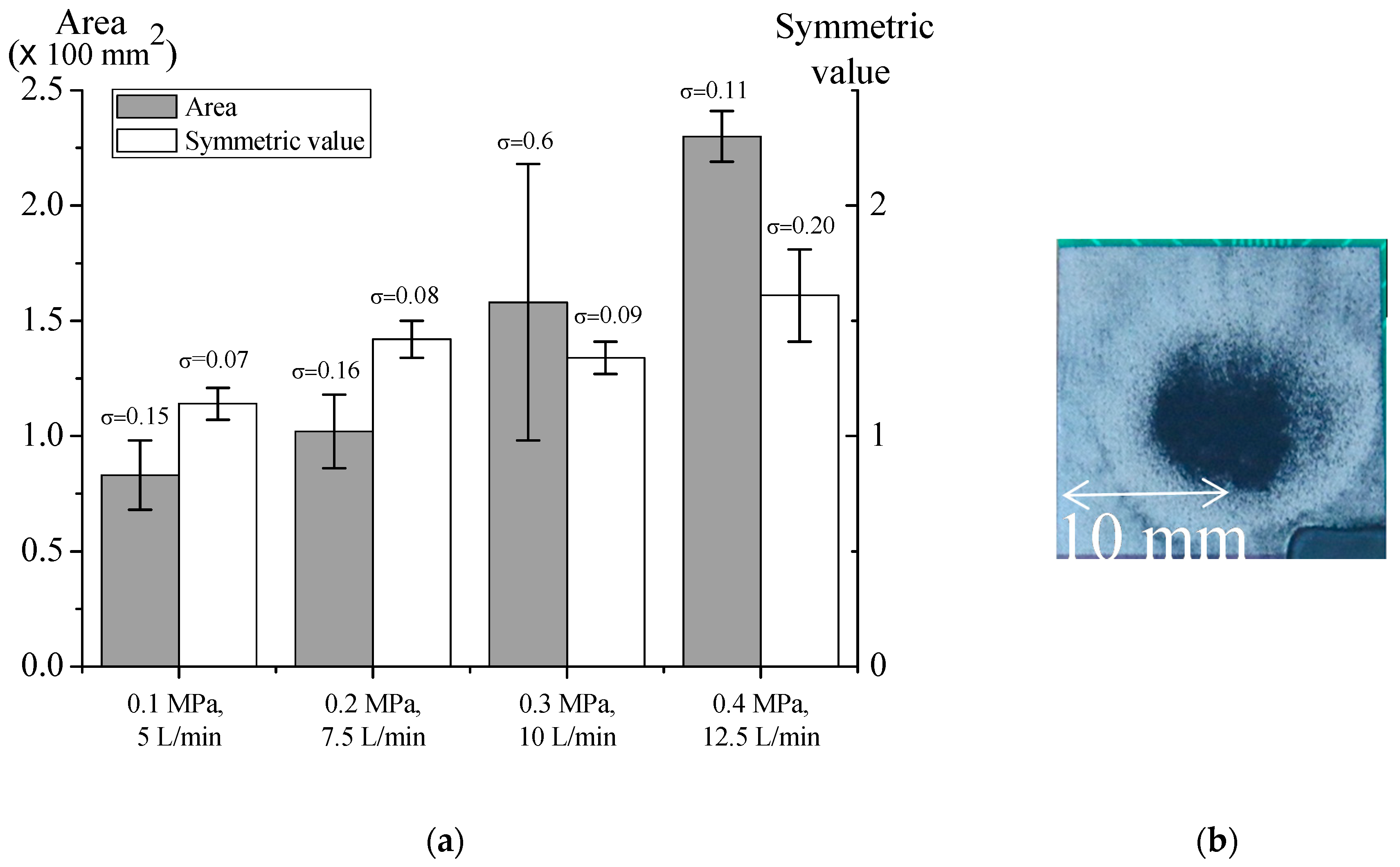
3.3. Effects of Mask Size, Pressure, and Flow Rate on Microparticle Deposition Stability with Mask
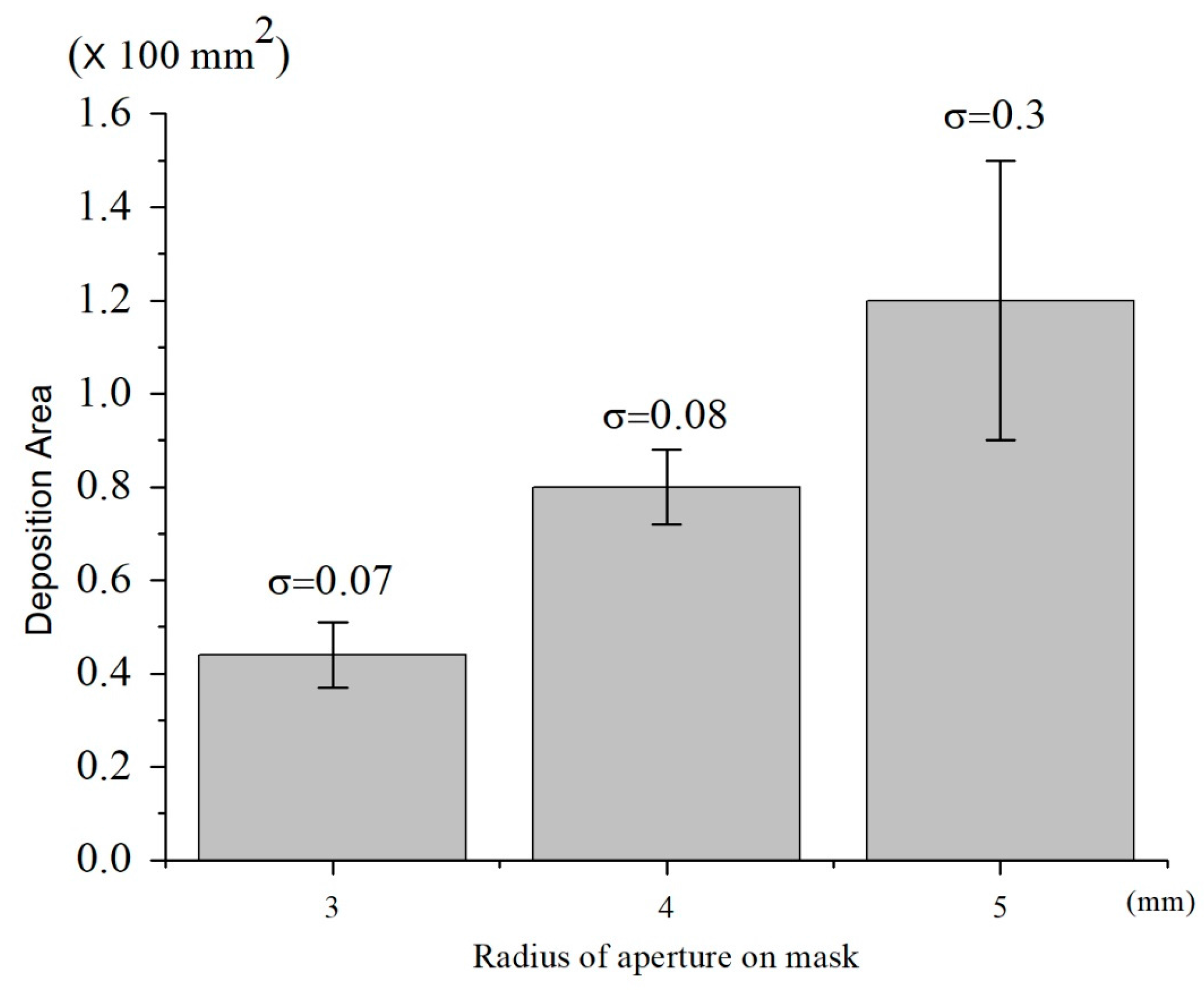
3.3.1. Effects of Mask Size
3.3.2. Effects of Pressure and Flow Rate
3.4. Particle Deposition in Initial Condition and after Optimization
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Blandenet, G.; Court, M.; Lagarde, Y. Thin layers deposited by the pyrosol process. Thin Solid Films 1981, 77, 81–90. [Google Scholar] [CrossRef]
- Mooney, J.B.; Radding, S.B. Spray pyrolysis processing. Annu. Rev. Mater. Sci. 1982, 12, 81–101. [Google Scholar] [CrossRef]
- Su, B.; Choy, K. Microstructure and properties of the CdS thin films prepared by electrostatic spray assisted vapour deposition (ESAVD) method. Thin Solid Films 2000, 359, 160–164. [Google Scholar] [CrossRef]
- Kashu, S.; Fuchita, E.; Manabe, T.; Hayashi, C. Deposition of ultra fine particles using a gas jet. Jpn. J. Appl. Phys. 1984, 23, L910. [Google Scholar] [CrossRef]
- Adamczyk, Z.; Van De Ven, T.G. Deposition of particles under external forces in laminar flow through parallel-plate and cylindrical channels. J. Colloid Interface Sci. 1981, 80, 340–356. [Google Scholar] [CrossRef]
- Park, J.; Jeong, J.; Kim, C.; Hwang, J. Deposition of charged aerosol particles on a substrate by collimating through an electric field assisted coaxial flow nozzle. Aerosol Sci. Technol. 2013, 47, 512–519. [Google Scholar] [CrossRef]
- Kim, H.; Kim, J.; Yang, H.; Suh, J.; Kim, T.; Han, B.; Kim, S.; Kim, D.S.; Pikhitsa, P.V.; Choi, M. Parallel patterning of nanoparticles via electrodynamic focusing of charged aerosols. Nat. Nanotechnol. 2006, 1, 117–121. [Google Scholar] [CrossRef] [PubMed]
- Qi, L.; McMurry, P.H.; Norris, D.J.; Girshick, S.L. Micropattern deposition of colloidal semiconductor nanocrystals by aerodynamic focusing. Aerosol Sci. Technol. 2010, 44, 55–60. [Google Scholar] [CrossRef]
- Bowen, B.D.; Epstein, N. Fine particle deposition in smooth parallel-plate channels. J. Colloid Interface Sci. 1979, 72, 81–97. [Google Scholar] [CrossRef]
- Lee, H.; You, S.; Woo, C.G.; Lim, K.; Jun, K.; Choi, M. Focused patterning of nanoparticles by controlling electric field induced particle motion. Appl. Phys. Lett. 2009, 94, 53104. [Google Scholar] [CrossRef]
- Stadler, V.; Felgenhauer, T.; Beyer, M.; Fernandez, S.; Leibe, K.; Güttler, S.; Gröning, M.; König, K.; Torralba, G.; Hausmann, M.; et al. Combinatorial synthesis of peptide arrays with a laser printer. Angew. Chem. Int. Ed. 2008, 47, 7132–7135. [Google Scholar] [CrossRef] [PubMed]
- Pai, D.M.; Springett, B.E. Physics of electrophotography. Rev. Mod. Phys. 1993, 65, 163. [Google Scholar] [CrossRef]
- Li, W.; Liang, C.; Zhou, W.; Qiu, J.; Li, H.; Sun, G.; Xin, Q. Homogeneous and controllable Pt particles deposited on multi-wall carbon nanotubes as cathode catalyst for direct methanol fuel cells. Carbon 2004, 42, 436–439. [Google Scholar] [CrossRef]
- Imanaka, Y.; Amada, H.; Kumasaka, F. Dielectric and insulating properties of embedded capacitor for flexible electronics prepared by aerosol-type nanoparticle deposition. Jpn. J. Appl. Phys. 2013, 52, 5DA2. [Google Scholar] [CrossRef]
- Huang, C.; Becker, M.F.; Keto, J.W.; Kovar, D. Annealing of nanostructured silver films produced by supersonic deposition of nanoparticles. J. Appl. Phys. 2007, 102, 54308. [Google Scholar] [CrossRef]
- Lebedev, M.; Akedo, J.; Akiyama, Y. Actuation properties of lead zirconate titanate thick films structured on Si membrane by the aerosol deposition method. Jpn. J. Appl. Phys. 2000, 39, 5600. [Google Scholar] [CrossRef]
- Tsukamoto, M.; Fujihara, T.; Abe, N.; Miyake, S.; Katto, M.; Nakayama, T.; Akedo, J. Hydroxyapatite coating on titanium plate with an ultrafine particle beam. Jpn. J. Appl. Phys. 2003, 42, L120. [Google Scholar] [CrossRef]
- Murakami, H.; Nishino, J.; Yaegashi, S.; Shiohara, Y.; Tanaka, S. Preparation of YBa2CuZ3O7−x and YBa2Cu4O8 Thick Films by Gas Deposition Using Fine Powders. Jpn. J. Appl. Phys. 1991, 30, L185. [Google Scholar] [CrossRef]
- Beyer, M.; Nesterov, A.; Block, I.; König, K.; Felgenhauer, T.; Fernandez, S.; Leibe, K.; Torralba, G.; Hausmann, M.; Trunk, U. Combinatorial synthesis of peptide arrays onto a microchip. Science 2007, 318, 1888. [Google Scholar] [CrossRef] [PubMed]
- Breitling, F.; Felgenhauer, T.; Nesterov, A.; Lindenstruth, V.; Stadler, V.; Bischoff, F.R. Particle-based synthesis of peptide arrays. ChemBioChem 2009, 10, 803–808. [Google Scholar] [CrossRef] [PubMed]
- Maerkle, F.; Loeffler, F.F.; Schillo, S.; Foertsch, T.; Muenster, B.; Striffler, J.; Schirwitz, C.; Bischoff, F.R.; Breitling, F.; Nesterov-Mueller, A. High-density peptide arrays with combinatorial laser fusing. Adv. Mater. 2014, 26, 3730–3734. [Google Scholar] [CrossRef] [PubMed]
- Frank, R. Spot-synthesis: An easy technique for the positionally addressable, parallel chemical synthesis on a membrane support. Tetrahedron 1992, 48, 9217–9232. [Google Scholar] [CrossRef]
- Fodor, S.P.; Read, J.L.; Pirrung, M.C.; Stryer, L.; Lu, A.T.; Solas, D. Light-directed, spatially addressable parallel chemical synthesis. Science 1991, 251, 767–773. [Google Scholar] [CrossRef] [PubMed]
- Breitling, F.; Nesterov, A.; Stadler, V.; Felgenhauer, T.; Bischoff, F.R. High-density peptide arrays. Mol. Biosyst. 2009, 5, 224–234. [Google Scholar] [CrossRef] [PubMed]
- Löffler, F.; Wagner, J.; König, K.; Märkle, F.; Fernandez, S.; Schirwitz, C.; Torralba, G.; Hausmann, M.; Lindenstruth, V.; Bischoff, F. High-precision combinatorial deposition of micro particle patterns on a microelectronic chip. Aerosol Sci. Technol. 2011, 45, 65–74. [Google Scholar] [CrossRef]
- Huh, S.H.; Riu, D.H.; Naono, Y.; Taguchi, Y.; Kawabata, S.; Nakajima, A. Generation of high-quality lines and arrays using nanoparticle controlling processes. Appl. Phys. Lett. 2007, 91, 93118. [Google Scholar] [CrossRef]
- Loeffler, F.; Schirwitz, C.; Wagner, J.; Koenig, K.; Maerkle, F.; Torralba, G.; Hausmann, M.; Bischoff, F.R.; Nesterov-Mueller, A.; Breitling, F. Biomolecule arrays using functional combinatorial particle patterning on microchips. Adv. Funct. Mater. 2012, 22, 2503–2508. [Google Scholar] [CrossRef]
- Tang, P.; Fletcher, D.; Chan, H.-K.; Raper, J.A. Simple and cost-effective powder disperser for aerosol particle size measurement. Powder Technol. 2008, 187, 27–36. [Google Scholar] [CrossRef]
- Sahu, K.C.; Govindarajan, R. Stability of flow through a slowly diverging pipe. J. Fluid Mech. 2005, 531, 325–334. [Google Scholar] [CrossRef]
- Peixinho, J.; Besnard, H. Transition to turbulence in slowly divergent pipe flow. Phys. Fluids 2013, 25, 111702. [Google Scholar] [CrossRef]
- Peixinho, J. Flow in a Slowly Divergent Pipe Section. In Seventh IUTAM Symposium on Laminar-Turbulent Transition; Springer: Dordrecht, the Netherlands, 2010; Available online: https://www.amazon.com/Seventh-IUTAM-Symposium-Laminar-Turbulent-Transition/dp/9048137225 (accessed on 1 March 2018).
- Cheng, Y.C. CMOS-Chip Based Printing System for Combinatorial Synthesis. Ph.D. Thesis, Technischen Universität Darmstadt, Darmstadt, Germany, 2012. [Google Scholar]
- Loeffler, F.; Cheng, Y.; Muenster, B.; Striffler, J.; Liu, F.; Bischoff, R.F.; Doersam, E.; Breitling, F.; Nesterov-Mueller, A. Printing peptide arrays with a complementary metal oxide semiconductor chip. Adv. Biochem. Eng. Biotechnol. 2013, 137, 1–23. [Google Scholar] [PubMed]
- Loeffler, F.; Foertsch, T.; Popov, R.; Mattes, D.; Schlageter, M.; Sedlmayr, M.; Ridder, B.; Dang, F.; Bojničić-Kninski, F.; Weber, L.; et al. High-flexibility combinatorial peptide synthesis with laser-based transfer of monomers in solid matrix material. Nat. Commun. 2016, 7, 11844. [Google Scholar] [CrossRef] [PubMed]
- Ridder, B.; Foertsch, T.; Wellec, A.; Mattes, D.; Bojnicic-Kninski, C.; Loeffler, F.; Nesterov-Mueller, A.; Meier, M.; Breitling, F. Development of a poly(dimethylacrylamide) based matrix material for solid phase high density peptide array synthesis employing a laser based material transfer. Appl. Surf. Sci. 2016, 389, 942–951. [Google Scholar] [CrossRef]
- Bojnicic-Kninski, C.; Bykovskaya, V.; Maerkle, F.; Popov, R.; Palermoa, A.; Mattes, D.; Weber, L.; Ridder, B.; Foertsch, T.; Loeffler, F.; et al. Microcavity functionalization: Selective functionalization of microstructured surfaces by laser-assisted particle transfer. Adv. Funct. Mater. 2016, 26, 7067–7073. [Google Scholar] [CrossRef]
- Bojnicic-Kninski, C.; Popov, R.; Dörsam, E.; Loeffler, F.; Breitling, F.; Nesterov-Mueller, A. Combinatorial particle patterning. Adv. Funct. Mater. 2017, 27, 1703511. [Google Scholar] [CrossRef]
- Webera, L.; Palermoa, A.; Kügler, J.; Nesterov-Mueller, A.; Breitling, F.; Loeffler, F. Single amino acid fingerprinting of the human antibody repertoire with high density peptide arrays. J. Immunol. Methods 2017, 443, 45–54. [Google Scholar] [CrossRef] [PubMed]
- Ridder, B.; Mattes, D.; Nesterov-Mueller, A.; Breitling, F.; Meier, M. Peptide array functionalization via the Ugi four-component reaction. Chem. Commun. 2017, 53, 5553–5556. [Google Scholar] [CrossRef] [PubMed]



© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shih, A.-C.; Han, C.-J.; Kuo, T.-C.; Cheng, Y.-C. Enhancing the Microparticle Deposition Stability and Homogeneity on Planer for Synthesis of Self-Assembly Monolayer. Nanomaterials 2018, 8, 164. https://doi.org/10.3390/nano8030164
Shih A-C, Han C-J, Kuo T-C, Cheng Y-C. Enhancing the Microparticle Deposition Stability and Homogeneity on Planer for Synthesis of Self-Assembly Monolayer. Nanomaterials. 2018; 8(3):164. https://doi.org/10.3390/nano8030164
Chicago/Turabian StyleShih, An-Ci, Chi-Jui Han, Tsung-Cheng Kuo, and Yun-Chien Cheng. 2018. "Enhancing the Microparticle Deposition Stability and Homogeneity on Planer for Synthesis of Self-Assembly Monolayer" Nanomaterials 8, no. 3: 164. https://doi.org/10.3390/nano8030164
APA StyleShih, A.-C., Han, C.-J., Kuo, T.-C., & Cheng, Y.-C. (2018). Enhancing the Microparticle Deposition Stability and Homogeneity on Planer for Synthesis of Self-Assembly Monolayer. Nanomaterials, 8(3), 164. https://doi.org/10.3390/nano8030164



