TiO2, SiO2 and ZrO2 Nanoparticles Synergistically Provoke Cellular Oxidative Damage in Freshwater Microalgae
Abstract
:1. Introduction
2. Methods
2.1. Test Material and Test Medium
2.2. Physicochemical Analysis
2.3. Algal Growth Assays
2.4. Chlorophyll Analysis
2.5. Mitochondrial Membrane Potential
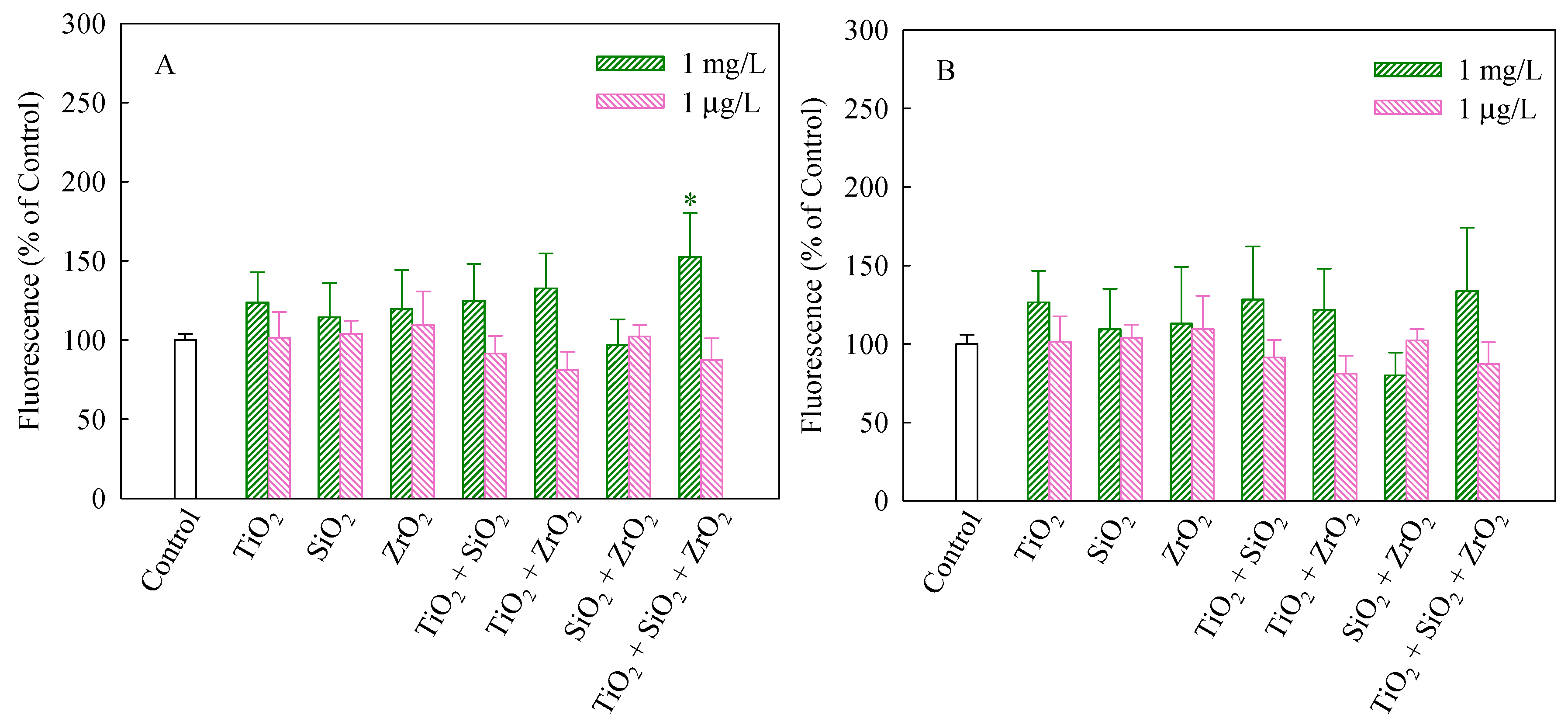
2.6. Permeability of Cell Membrane
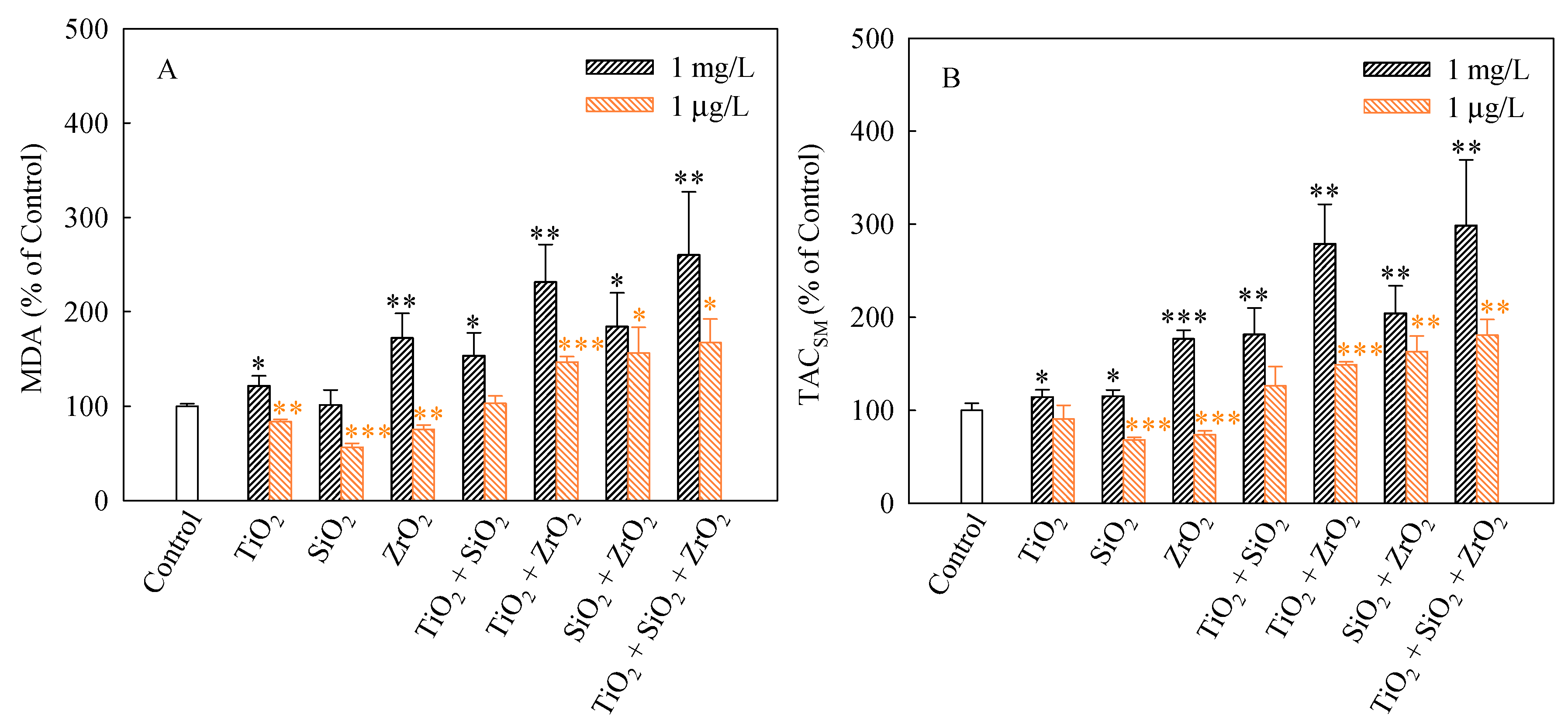
2.7. Oxidative Stress Biomarker Assays
2.8. Statistical Analysis
3. Results and Discussion
3.1. Physicochemical Characterizations
3.2. Effects of Single and Mixtures of NPs on Chlorophyll Contents
3.3. Effects of Single and Mixtures of NPs on Cellular Responses in Algal Cells
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Guinée, J.B.; Heijungs, R.; Vijver, M.G.; Peijnenburg, W.J.G.M. Setting the stage for debating the roles of risk assessment and life-cycle assessment of engineered nanomaterials. Nat. Nanotechnol. 2017, 12, 727–733. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Mahendra, S.; Alvarez, P.J.J. Nanomaterials in the construction industry: A review of their applications and environmental health and safety considerations. ACS Nano 2010, 4, 3580–3590. [Google Scholar] [CrossRef] [PubMed]
- Hoseinzadeh, E.; Makhdoumi, P.; Taha, P.; Hossini, H.; Stelling, J.; Kamal, M.A.; Ashraf, G.M. A review on nano-antimicrobials: Metal nanoparticles, methods and mechanisms. Curr. Drug Metab. 2017, 18, 120–128. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.H.; Balasubramanian, R. Effects of cerium oxide and ferrocene nanoparticles addition as fuel-borne catalysts on diesel engine particulate emissions: Environmental and health implications. Environ. Sci. Technol. 2017, 51, 4248–4258. [Google Scholar] [CrossRef] [PubMed]
- Walser, T.; Demou, E.; Lang, D.J.; Hellweg, S. Prospective environmental life cycle assessment of nanosilver T-shirts. Environ. Sci. Technol. 2011, 45, 4570–4578. [Google Scholar] [CrossRef] [PubMed]
- De la Calle, I.; Menta, M.; Klein, M.; Séby, F. Screening of TiO2 and Au nanoparticles in cosmetics and determination of elemental impurities by multiple techniques (DLS, SP-ICP-MS, ICP-MS and ICP-OES). Talanta 2017, 171, 291–306. [Google Scholar] [CrossRef] [PubMed]
- Tourinho, P.S.; van Gestel, C.A.; Lofts, S.; Svendsen, C.; Soares, A.M.V.M.; Loureiro, S. Metal-based nanoparticles in soil: Fate, behavior, and effects on soil invertebrates. Environ. Toxicol. Chem. 2012, 31, 1679–1692. [Google Scholar] [CrossRef] [PubMed]
- Lowry, G.V.; Gregory, K.B.; Apte, S.C.; Lead, J.R. Transformations of nanomaterials in the environment. Environ. Sci. Technol. 2012, 46, 6893–6899. [Google Scholar] [CrossRef] [PubMed]
- Coll, C.; Notter, D.; Gottschalk, F.; Sun, T.; Som, C.; Nowack, B. Probabilistic environmental risk assessment of five nanomaterials (nano-TiO2, nano-Ag, nano-ZnO, CNT, and fullerenes). Nanotoxicology 2016, 10, 436–444. [Google Scholar] [CrossRef] [PubMed]
- Thwala, M.; Klaine, S.J.; Musee, N. Interactions of metal-based engineered nanoparticles with aquatic higher plants: A review of the state of current knowledge. Environ. Toxicol. Chem. 2016, 35, 1677–1694. [Google Scholar] [CrossRef] [PubMed]
- Ashauer, R.; O’Connor, I.; Escher, B.I. Toxic mixtures in time-the sequence makes the poison. Environ. Sci. Technol. 2017, 51, 3084–3092. [Google Scholar] [CrossRef] [PubMed]
- Yu, R.; Wu, J.; Liu, M.; Zhu, G.; Chen, L.; Chang, Y.; Lu, H. Toxicity of binary mixtures of metal oxide nanoparticles to Nitrosomonas europaea. Chemosphere 2016, 153, 187–197. [Google Scholar] [CrossRef] [PubMed]
- Ye, N.; Wang, Z.; Fang, H.; Wang, S.; Zhang, F. Combined ecotoxicity of binary zinc oxide and copper oxide nanoparticles to Scenedesmus obliquus. J. Environ. Sci. Health. A Tox. Hazard. Subst. Environ. Eng. 2017, 52, 555–560. [Google Scholar] [CrossRef] [PubMed]
- Tong, T.; Wilke, C.M.; Wu, J.; Binh, C.T.; Kelly, J.J.; Gaillard, J.F.; Gray, K.A. Combined toxicity of nano-ZnO and nano-TiO2: From single- to multinanomaterial systems. Environ. Sci. Technol. 2015, 49, 8113–8123. [Google Scholar] [CrossRef] [PubMed]
- Tsugita, M.; Morimoto, N.; Nakayama, M. SiO2 and TiO2 nanoparticles synergistically trigger macrophage inflammatory responses. Part. Fiber Toxicol. 2017, 14, 11. [Google Scholar] [CrossRef] [PubMed]
- Hua, J.; Peijnenburg, W.J.G.M.; Vijver, M.G. TiO2 nanoparticles reduce the effects of ZnO nanoparticles and Zn ions on zebrafish embryos (Danio rerio). NanoImpact 2016, 2, 45–53. [Google Scholar] [CrossRef]
- Toussaint, M.W.; Shedd, T.R.; Van der Schalie, W.H.; Leather, G.R. A comparison of standard acute toxicity tests with rapid-screening toxicity tests. Environ. Toxicol. Chem. 1995, 14, 907–915. [Google Scholar] [CrossRef]
- Domingos, R.F.; Simon, D.F.; Hauser, C.; Wilkinson, K.J. Bioaccumulation and effects of CdTe/CdS quantum dots on Chlamydomonas reinhardtii—Nanoparticles or the free ions? Environ. Sci. Technol. 2011, 45, 7664–7669. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Li, J.; Zhao, J.; Xing, B. Toxicity and internalization of CuO nanoparticles to prokaryotic alga Microcystis aeruginosa as affected by dissolved organic matter. Environ. Sci. Technol. 2011, 45, 6032–6040. [Google Scholar] [CrossRef] [PubMed]
- Schwab, F.; Bucheli, T.D.; Lukhele, L.P.; Magrez, A.; Nowack, B.; Sigg, L.; Knauer, K. Are carbon nanotube effects on green algae caused by shading and agglomeration? Environ. Sci. Technol. 2011, 45, 6136–6144. [Google Scholar] [CrossRef] [PubMed]
- Tang, D.; Han, W.; Li, P.; Miao, X.; Zhong, J. CO2 biofixation and fatty acid composition of Scenedesmus obliquus and Chlorella pyrenoidosa in response to different CO2 levels. Bioresour. Technol. 2011, 102, 3071–3076. [Google Scholar] [CrossRef] [PubMed]
- OECD. Freshwater Alga and Cyanobacteria, Growth Inhibition Test, Nr. 201, OECD Guidelines for the Testing of Chemicals; Organization for Economic Cooperation and Development (OECD): Paris, France, 2011; Available online: http://www.oecd.org (accessed on 10 January 2018).
- Wang, Z.; Chen, J.W.; Li, X.H.; Shao, J.P.; Peijnenburg, W.J.G.M. Aquatic toxicity of nanosilver colloids to different trophic organisms: Contributions of particles and free silver ion. Environ. Toxicol. Chem. 2012, 31, 2408–2413. [Google Scholar] [CrossRef] [PubMed]
- Gottschalk, F.; Sun, T.; Nowack, B. Environmental concentrations of engineered nanomaterials: Review of modeling and analytical studies. Environ. Pollut. 2013, 181, 287–300. [Google Scholar] [CrossRef] [PubMed]
- Wellburn, A.R.; Lichtenthaler, H. Formulae and program to determine total carotenoids and chlorophylls A and B of leaf extracts in different solvents. In Advances in Photosynthesis Research, Advances in Agricultural Biotechnology; Sybesma, C., Ed.; Springer: Dordrecht, The Netherlands, 1984; p. 2. [Google Scholar]
- Liu, W.; Chen, S.; Quan, X.; Jin, Y.H. Toxic effect of serial perfluorosulfonic and perfluorocarboxylic acids on the membrane system of a freshwater alga measured by flow cytometry. Environ. Toxicol. Chem. 2008, 27, 1597–1604. [Google Scholar] [CrossRef] [PubMed]
- Vigneault, B.; Percot, A.; Lafleur, M.; Campbell, P.G. Permeability changes in model and phytoplankton membranes in the presence of aquatic humic substances. Environ. Sci. Technol. 2000, 34, 3907–3913. [Google Scholar] [CrossRef]
- Hu, X.; Ouyang, S.; Mu, L.; An, J.; Zhou, Q. Effects of graphene oxide and oxidized carbon nanotubes on the cellular division, microstructure, uptake, oxidative stress, and metabolic profiles. Environ. Sci. Technol. 2015, 49, 10825–10833. [Google Scholar] [CrossRef] [PubMed]
- Grimm, B.; Porra, R.J.; Rüdiger, W.; Scheer, H. (Eds.) Chlorophylls and Bacteriochlorophylls: Biochemistry, Biophysics, Functions and Applications; Springer: Dordrecht, The Netherlands, 2006; pp. 1–26. [Google Scholar]
- Stensberg, M.C.; Madangopal, R.; Yale, G.; Wei, Q.; Ochoa-Acuna, H.; Wei, A.; McLamore, E.S.; Rickus, J.; Porterfield, D.M.; Sepulveda, M.S. Silver nanoparticle-specific mitotoxicity in Daphnia magna. Nanotoxicology 2014, 8, 833–842. [Google Scholar] [CrossRef] [PubMed]
- Karlsson, H.L.; Cronholm, P.; Gustafsson, J.; Möller, L. Copper oxide nanoparticles are highly toxic: A comparison between metal oxide nanoparticles and carbon nanotubes. Chem. Res. Toxicol. 2008, 21, 1726–1732. [Google Scholar]
- Weckx, J.E.J.; Clijsters, H.M.M. Oxidative damage and defense mechanisms in primary leaves of Phaseolus vulgaris as a result of root assimilation of toxic amounts of copper. Physiol. Plant. 1996, 96, 506–512. [Google Scholar]
- Foyer, C.H.; Shigeoka, S. Understanding oxidative stress and antioxidant functions to enhance photosynthesis. Plant Physiol. 2011, 155, 93–100. [Google Scholar] [CrossRef] [PubMed]
- Hu, C.; Wang, Q.; Zhao, H.; Wang, L.; Guo, S.; Li, X. Ecotoxicological effects of graphene oxide on the protozoan Euglena gracilis. Chemosphere 2015, 128, 184–190. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Hu, C.; Zhu, Q.; Chen, L.; Kong, Z.; Liu, Z. Copper and zinc induction of lipid peroxidation and effects on antioxidant enzyme activities in the microalga Pavlova viridis (Prymnesiophyceae). Chemosphere 2006, 62, 565–572. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Miao, Y.; Huang, L.; Li, J.; Sun, H.; Zhao, Y.; Yang, J.; Zhou, W. Antioxidant activities of saponins extracted from Radix Trichosanthis: An in vivo and in vitro evaluation. BMC Complement. Altern. Med. 2014, 14, 86. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Zhang, F.; Wang, S.; Peijnenburg, W.J.G.M. Assessment and prediction of joint algal toxicity of binary mixtures of graphene and ionic liquids. Chemosphere 2017, 185, 681–689. [Google Scholar] [CrossRef] [PubMed]

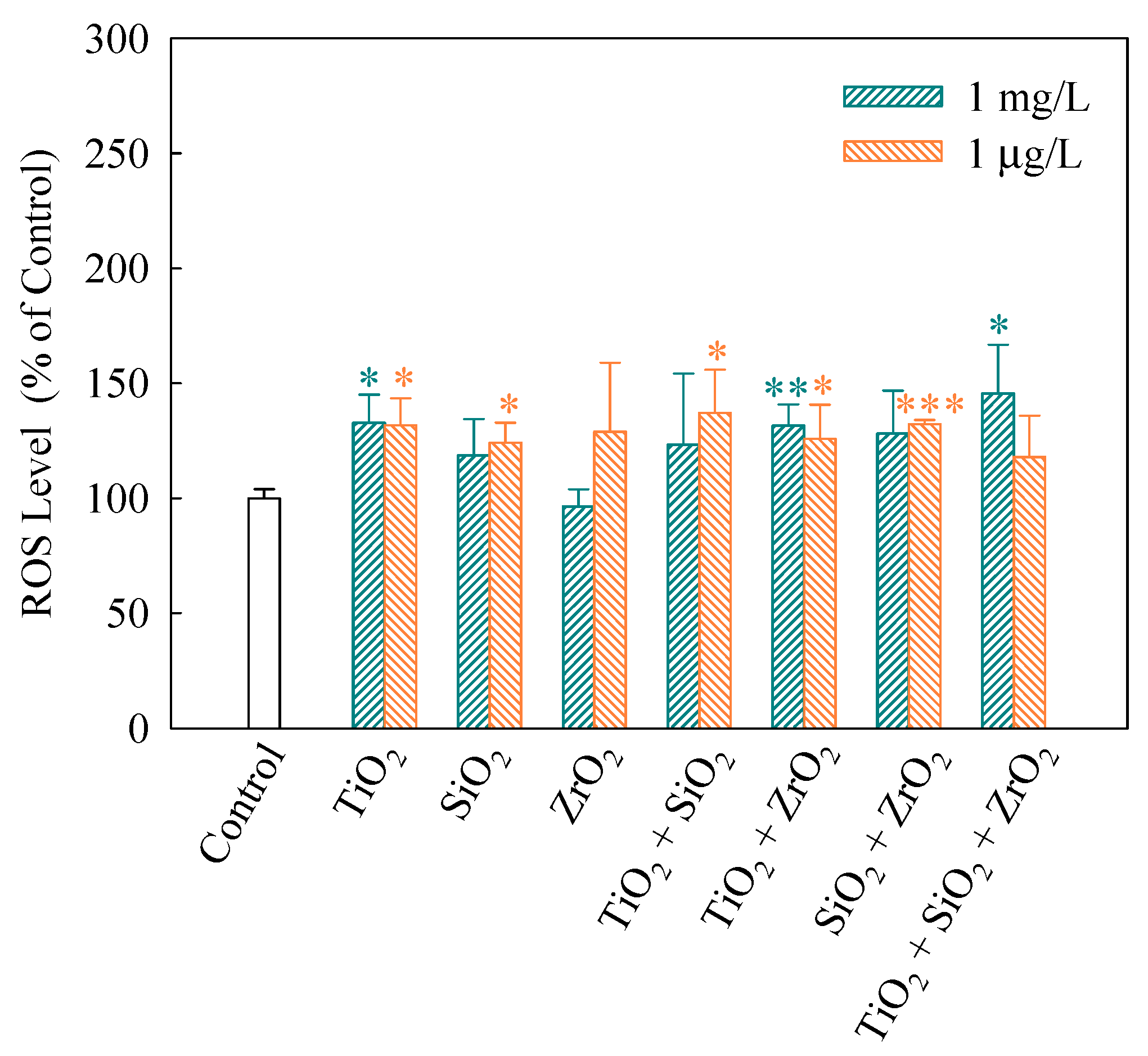
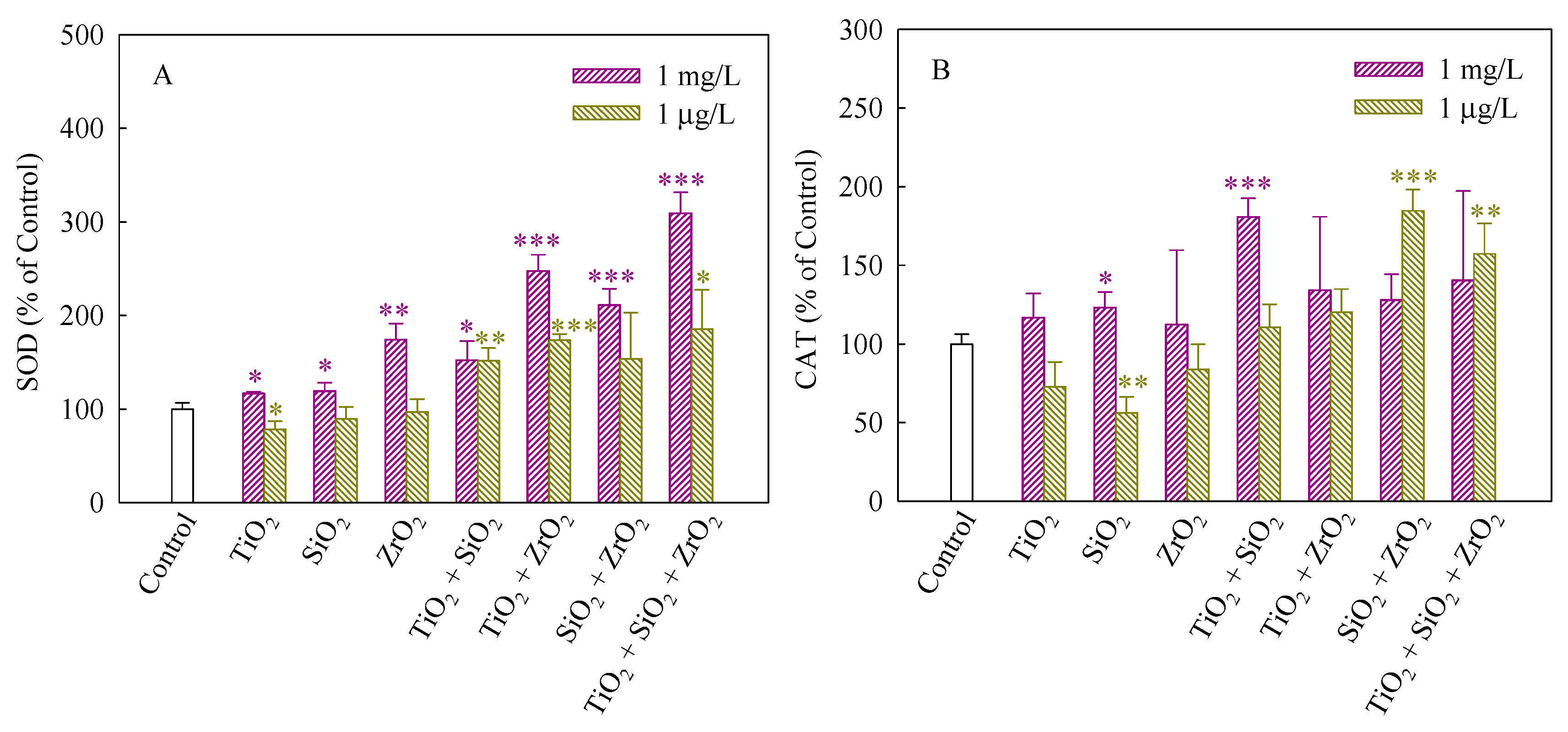

| Test NPs | 0 h | 96 h | ||
|---|---|---|---|---|
| ZP | DH | ZP | DH | |
| mV | nm | mV | nm | |
| TiO2 | −20.9 ± 1.2 | 1111 ± 410 | −15.3 ± 0.9 | 1278 ± 924 |
| SiO2 | −22.3 ± 1.6 | 525 ± 373 | −17.9 ± 1.1 | 147 ± 38 |
| ZrO2 | −19.0 ± 0.5 | 333 ± 12 | −16.3 ± 0.2 | 336 ± 41 |
| TiO2 + SiO2 | −16.7 ± 0.5 | 1095 ± 213 | −13.8 ± 1.1 | 511 ± 319 |
| TiO2 + ZrO2 | −18.8 ± 1.0 | 596 ± 87 | −16.3 ± 1.0 | 352 ± 22 |
| SiO2 + ZrO2 | −20.0 ± 0.5 | 327 ± 46 | −19.8 ± 0.6 | 285 ± 30 |
| TiO2 + SiO2 + ZrO2 | −18.1 ± 0.2 | 523 ± 39 | −18.5 ± 0.5 | 333 ± 10 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, Y.; Wang, S.; Wang, Z.; Ye, N.; Fang, H.; Wang, D. TiO2, SiO2 and ZrO2 Nanoparticles Synergistically Provoke Cellular Oxidative Damage in Freshwater Microalgae. Nanomaterials 2018, 8, 95. https://doi.org/10.3390/nano8020095
Liu Y, Wang S, Wang Z, Ye N, Fang H, Wang D. TiO2, SiO2 and ZrO2 Nanoparticles Synergistically Provoke Cellular Oxidative Damage in Freshwater Microalgae. Nanomaterials. 2018; 8(2):95. https://doi.org/10.3390/nano8020095
Chicago/Turabian StyleLiu, Yinghan, Se Wang, Zhuang Wang, Nan Ye, Hao Fang, and Degao Wang. 2018. "TiO2, SiO2 and ZrO2 Nanoparticles Synergistically Provoke Cellular Oxidative Damage in Freshwater Microalgae" Nanomaterials 8, no. 2: 95. https://doi.org/10.3390/nano8020095
APA StyleLiu, Y., Wang, S., Wang, Z., Ye, N., Fang, H., & Wang, D. (2018). TiO2, SiO2 and ZrO2 Nanoparticles Synergistically Provoke Cellular Oxidative Damage in Freshwater Microalgae. Nanomaterials, 8(2), 95. https://doi.org/10.3390/nano8020095