A Brief Note on the Magnetowetting of Magnetic Nanofluids on AAO Surfaces
Abstract
:1. Introduction
2. Materials and Methods
2.1. Preparation of Magnetic Nanofluids
2.1.1. Co-Precipitation Method
2.1.2. Sol-Gel Method
2.2. Preparation of Anodic Aluminum Oxide Templates
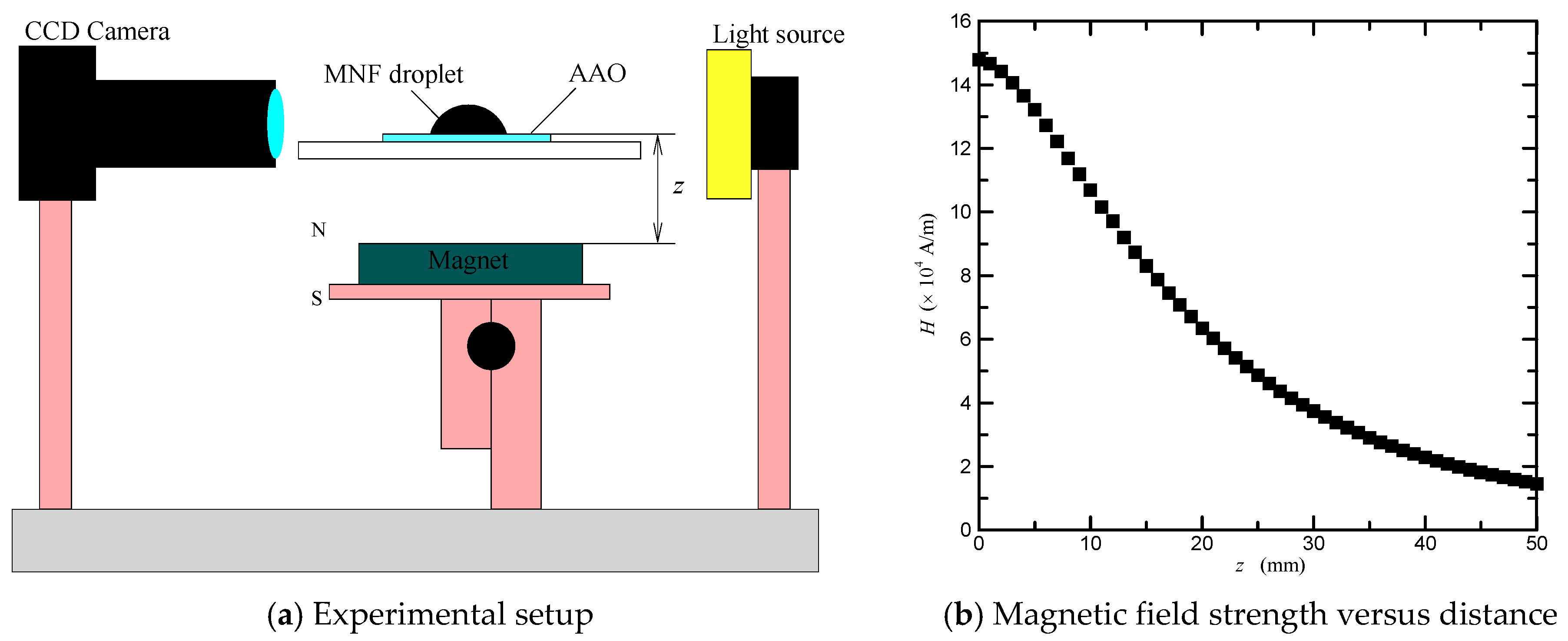
2.3. Magnetowetting Experimental Setup
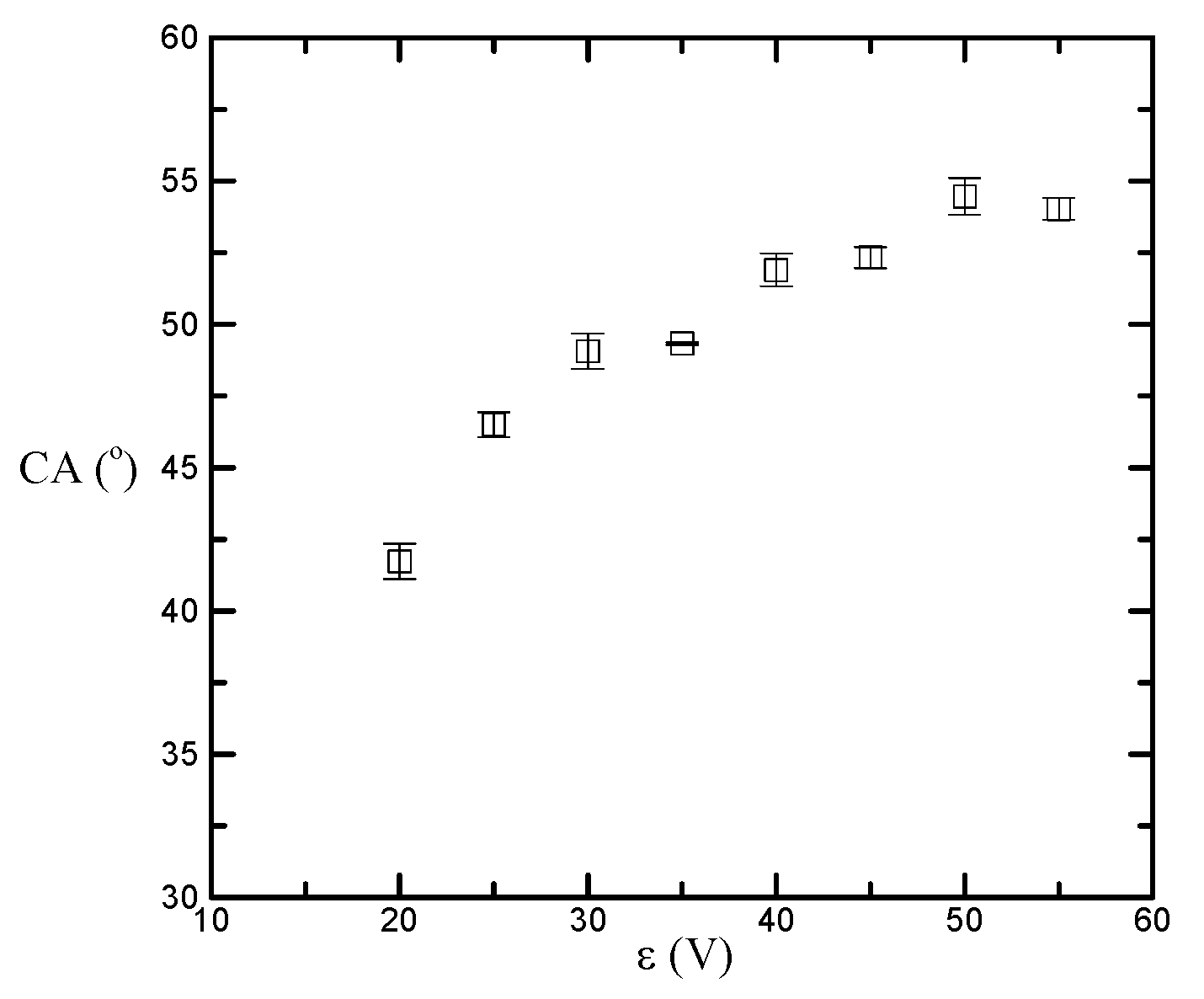
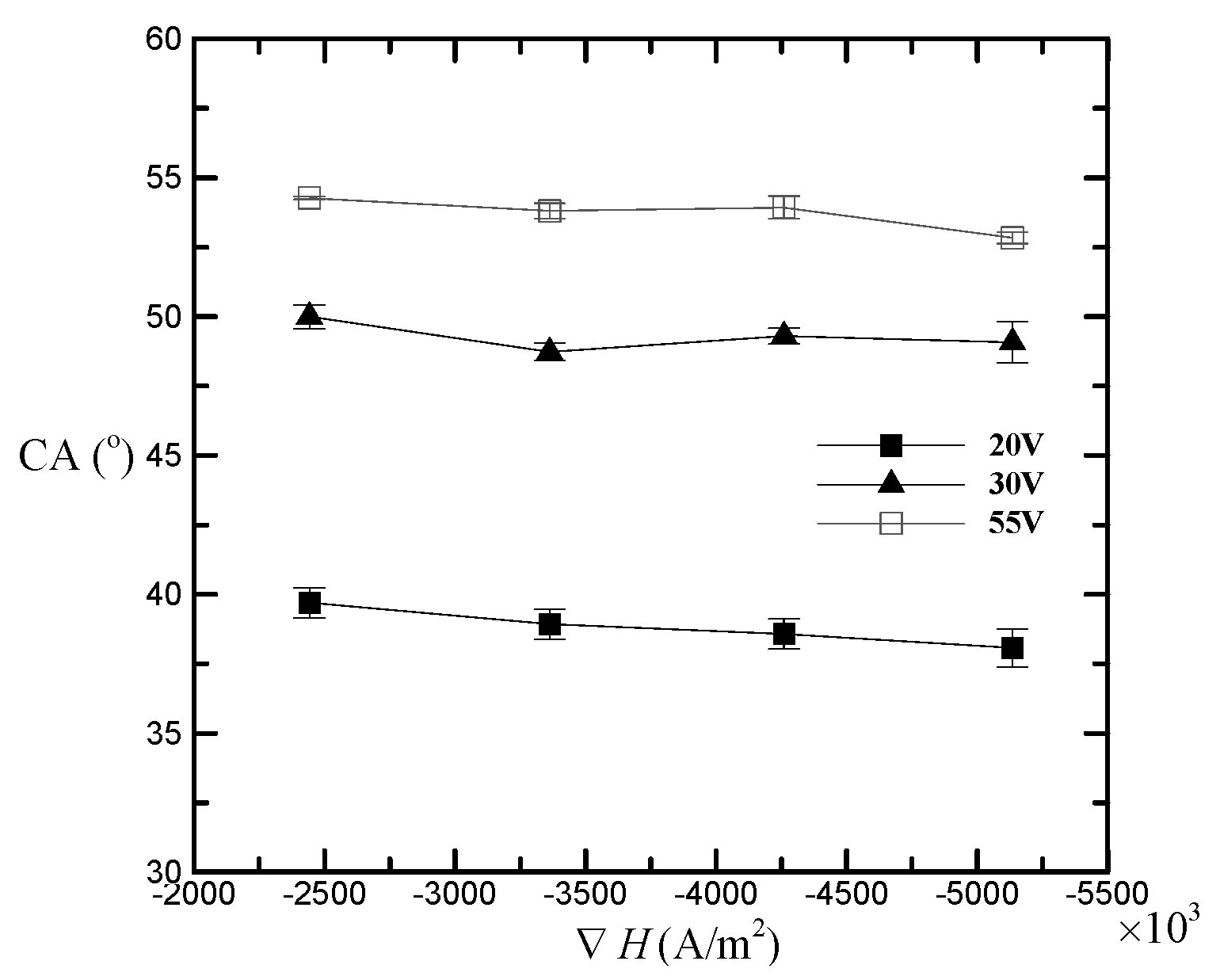
3. Results and Discussion
4. Conclusions
Author Contributions
Conflicts of Interest
References
- Li, H.; Yu, S.R. A robust superhydrophobic surface and origins of its self-cleaning properties. Appl. Surf. Sci. 2017, 420, 336–345. [Google Scholar] [CrossRef]
- Liu, Y.; Li, X.L.; Yan, Y.Y.; Han, Z.W.; Ren, L.Q. Anti-icing performance of superhydrophobic aluminum alloy surface and its rebounding mechanism of droplet under super-cold conditions. Surf. Coat. Technol. 2017, 331, 7–14. [Google Scholar] [CrossRef]
- Lay, K.K.; Cheong, B.M.Y.; Tong, W.L.; Tan, M.K.; Hung, Y.M. Effective micro-spray cooling for light-emitting diode with graphene nanoporous layers. Nanotechnology 2017, 28, 164003. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Hua, Y.Q.; Ye, Y.X.; Chen, R.F.; Li, Z.B. Transparent superhydrophobic solar glass prepared by fabricating groove-shaped arrays on the surface. Appl. Surf. Sci. 2017, 426, 957–964. [Google Scholar] [CrossRef]
- Holloway, P.J. Surface factors affecting the wetting of leaves. Pestic. Sci. 1970, 1, 156–163. [Google Scholar] [CrossRef]
- De Gennes, P.G. Wetting: statics and dynamics. Rev. Mod. Phys. 1985, 57, 827–863. [Google Scholar] [CrossRef]
- Verplanck, N.; Coffinier, Y.; Thomy, V.; Boukherroub, R. Wettability switching techniques on superhydrophobic surfaces. Nano. Res. Lett. 2007, 2, 577–596. [Google Scholar] [CrossRef]
- Quéré, D. Wetting and Roughness. Annu. Rev. Mater. Res. 2008, 38, 71–99. [Google Scholar] [CrossRef]
- Yuan, Y.; Lee, T.R. Contact angle and wetting properties. Surf. Sci. Tech. 2013, 51, 3–34. [Google Scholar] [CrossRef]
- Tadmor, R.; Das, R.; Gulec, S.; Liu, J.; N’guessan, H.E.; Shah, M.; Wasnik, P.S.; Yadav, S.B. Solid-Liquid Work of Adhesion. Langmuir 2017, 33, 3594–3600. [Google Scholar] [CrossRef] [PubMed]
- Timonen, J.V.I.; Latikka, M.; Ikkala, O.; Ras, R.H.A. Free-decay and resonant methods for investigating the fundamental limit of superhydrophobicity. Nat. Commun. 2013, 4, 2398. [Google Scholar] [CrossRef] [PubMed]
- Tadmor, R.; Bahadur, P.; Leh, A.; N’guessan, H.E.; Jaini, R.; Dang, L. Measurement of Lateral Adhesion Forces at the Interface between a Liquid Drop and a Substrate. Phys. Rev. Lett. 2009, 103, 266101. [Google Scholar] [CrossRef] [PubMed]
- Nishino, T.; Meguro, M.; Nakamae, K.; Matsushita, M.; Ueda, Y. The lowest surface free energy based on-CF3 alignment. Langmuir 1999, 15, 4321–4323. [Google Scholar] [CrossRef]
- Cao, M.; Song, X.; Zhai, J.; Wang, J.; Wang, Y. Fabrication of Highly Antireflective Silicon Surfaces with Superhydrophobicity. J. Phys. Chem. B 2006, 110, 13072. [Google Scholar] [CrossRef] [PubMed]
- Puukilainen, E.; Koponen, H.-K.; Xiao, Z.; Suvanto, S.; Pakkanen, T.A. Nanostructured and chemically modified hydrophobic polyolefin surfaces. Colloids Surf. Physicochem. Eng. Asp. 2006, 287, 175–181. [Google Scholar] [CrossRef]
- Hong, Y.C.; Uhm, H.S. Superhydrophobicity of a material made from multiwalled carbon nanotubes. Appl. Phys. Lett. 2006, 88, 244101. [Google Scholar] [CrossRef]
- Redón, R.; Vázquez-Olmos, A.; Mata-Zamora, M.E.; OrdóñezMedrano, A.; Rivera-Torres, F.; Saniger, J.M. Contact angle studies on anodic porous alumina. J. Colloid Interface Sci. 2005, 287, 664–670. [Google Scholar] [CrossRef] [PubMed]
- Petcu, C.; Purcar, V.; Spataru, C.-I.; Alexandrescu, E.; Somoghi, R.; Trica, B.; Nitu, S.G.; Panaitescu, D.M.; Donescu, D.; Jecu, M.-L. The Influence of New Hydrophobic Silica Nanoparticles on the Surface Properties of the Films Obtained from Bilayer Hybrids. Nanomaterials 2017, 7, 47. [Google Scholar] [CrossRef] [PubMed]
- Wenzel, R.N. Resistance of solid surfaces to wetting by water. Ind. Eng. Chem. 1936, 28, 988–994. [Google Scholar] [CrossRef]
- Cassie, A.B.D.; Baxter, S. Wettability of porous surface. Trans. Faraday Soc. 1944, 40, 546–551. [Google Scholar] [CrossRef]
- Shibuichi, S.; Onda, T.; Satoh, N.; Tsujii, K. Super Water-Repellent Surfaces Resulting from Fractal Structure. J. Phys. Chem. 1996, 100, 19512–19517. [Google Scholar] [CrossRef]
- Yoshimitsu, Z.; Nakajima, A.; Watanabe, T.; Hashimoto, K. Effects of Surface Structure on the Hydrophobicity and Sliding Behavior of Water Droplets. Langmuir 2002, 18, 5818–5822. [Google Scholar] [CrossRef]
- Thompson, G.E.; Wood, G.C. Porous anodic film formation on aluminium. Nature 1981, 290, 230–232. [Google Scholar] [CrossRef]
- Masuda, H.; Fukuda, K. Ordered metal nanohole arrays made by a two-step replication of honeycomb structures of anodic alumina. Science 1995, 268, 1466. [Google Scholar] [CrossRef] [PubMed]
- Jessensky, O.; Muller, F.; Gosele, U. Self-organized formation of hexagonal pore arrays in anodic alumina. Appl. Phys. Lett. 1998, 72, 1173–1175. [Google Scholar] [CrossRef]
- Li, A.P.; Muller, F.; Birner, A.; Nielsch, K.; Gosele, U. Hexagonal pore arrays with a 50–420 nm interpore distance formed by self-organization in anodic alumina. J. Appl. Phys. 1998, 84, 6023–6026. [Google Scholar] [CrossRef]
- Ran, C.; Ding, G.; Liu, W.; Deng, Y.; Hou, W. Wetting on nanoporous alumina surface: Transition between Wenzel and Cassie states controlled by surface structure. Langmuir 2008, 24, 9952–9955. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Polycarpou, A.A.; Liang, H. Electrical-potential induced surface wettability of porous metallic nanostructures. Appl. Surf. Sci. 2015, 351, 460–465. [Google Scholar] [CrossRef]
- Lippmann, G. Relations entre les phénomènes électriques et capillaires. Annales des Chimie et des Physique. 1875, 5, 494–549. [Google Scholar]
- Brochard, F. Motions of droplets on solid surface induced by chemical or thermal gradients. Langmuir 1989, 5, 432–438. [Google Scholar] [CrossRef]
- Wang, R.; Hashimoto, K.; Fujishima, A.; Chikuni, M.; Kojima, E.; Kitamura, A.; Shimohigoshi, M.; Watanabe, T. Light-induce amphiphilic surfaces. Nature 1997, 388, 431–432. [Google Scholar] [CrossRef]
- Welters, W.J.J.; Fokkink, L.G.J. Fast electrically switchable capillary effects. Langmuir 1998, 14, 1535–1538. [Google Scholar] [CrossRef]
- Egatz-Gómez, A.; Melle, S.; García, A.A.; Lindsay, S.A.; Márquez, M.; Domínguez-García, P.; Rubio, M.A.; Picraux, S.T.; Taraci, J.L.; Clement, T.; et al. Devens Gust. Discrete magnetic microfluidics. Appl. Phys. Lett. 2006, 89, 034106. [Google Scholar] [CrossRef]
- Berge, B. Electrocapillarity and wetting of insulator films by water. Comptes Rendus Acad. Sci. 1993, 317, 157–163. [Google Scholar] [CrossRef]
- Nguyen, N.-T.; Zhu, G.; Chua, Y.-C.; Phan, V.-N.; Tan, S.-H. Magnetowetting and sliding motion of a sessile ferrofluid droplet in the presence of a permanent magnet. Langmuir 2010, 26, 12553–12559. [Google Scholar] [CrossRef] [PubMed]
- Chien, Y.-C.; Weng, H.C. The effect of a magnetic field on the profile of sessile magnetic nanofluid droplets. Smart Sci. 2017, 5, 214–219. [Google Scholar] [CrossRef]
- Manukyan, S.; Schneider, M. Experimental Investigation of Wetting with Magnetic Fluids. Langmuir 2016, 32, 5135–5140. [Google Scholar] [CrossRef] [PubMed]
- Rigoni, C.; Pierno, M.; Mistura, G.; Talbot, D.; Massart, R.; Bacri, J.-C.; Abou-Hassan, A. Static Magnetowetting of Ferrofluid Drops. Langmuir 2016, 32, 7639–7646. [Google Scholar] [CrossRef] [PubMed]
- Shimoiizaka, J. Flocculation and dispersion of powders in liquids. J. Jpn. Soc. Powder Powder Metall. 1966, 13, 263–274. [Google Scholar]
- Shimoiizaka, J.; Nakatsuka, K.; Chubachi, R.; Sato, Y. On the Preparation of Magnetic Fluid and Its Behavior. J. Jpn. Soc. Powder Powder Metall. 1975, 22, 22–26. [Google Scholar] [CrossRef]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chien, Y.-C.; Weng, H.C. A Brief Note on the Magnetowetting of Magnetic Nanofluids on AAO Surfaces. Nanomaterials 2018, 8, 118. https://doi.org/10.3390/nano8020118
Chien Y-C, Weng HC. A Brief Note on the Magnetowetting of Magnetic Nanofluids on AAO Surfaces. Nanomaterials. 2018; 8(2):118. https://doi.org/10.3390/nano8020118
Chicago/Turabian StyleChien, Yu-Chin, and Huei Chu Weng. 2018. "A Brief Note on the Magnetowetting of Magnetic Nanofluids on AAO Surfaces" Nanomaterials 8, no. 2: 118. https://doi.org/10.3390/nano8020118
APA StyleChien, Y.-C., & Weng, H. C. (2018). A Brief Note on the Magnetowetting of Magnetic Nanofluids on AAO Surfaces. Nanomaterials, 8(2), 118. https://doi.org/10.3390/nano8020118




