Hierarchical Ta-Doped TiO2 Nanorod Arrays with Improved Charge Separation for Photoelectrochemical Water Oxidation under FTO Side Illumination
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Synthesis of Hierarchical Ta-Doped TiO2 Nanorod Arrays with Nanoparticles on the Top, on FTO Glass (Ta:TiO2)
3. Results and Discussion
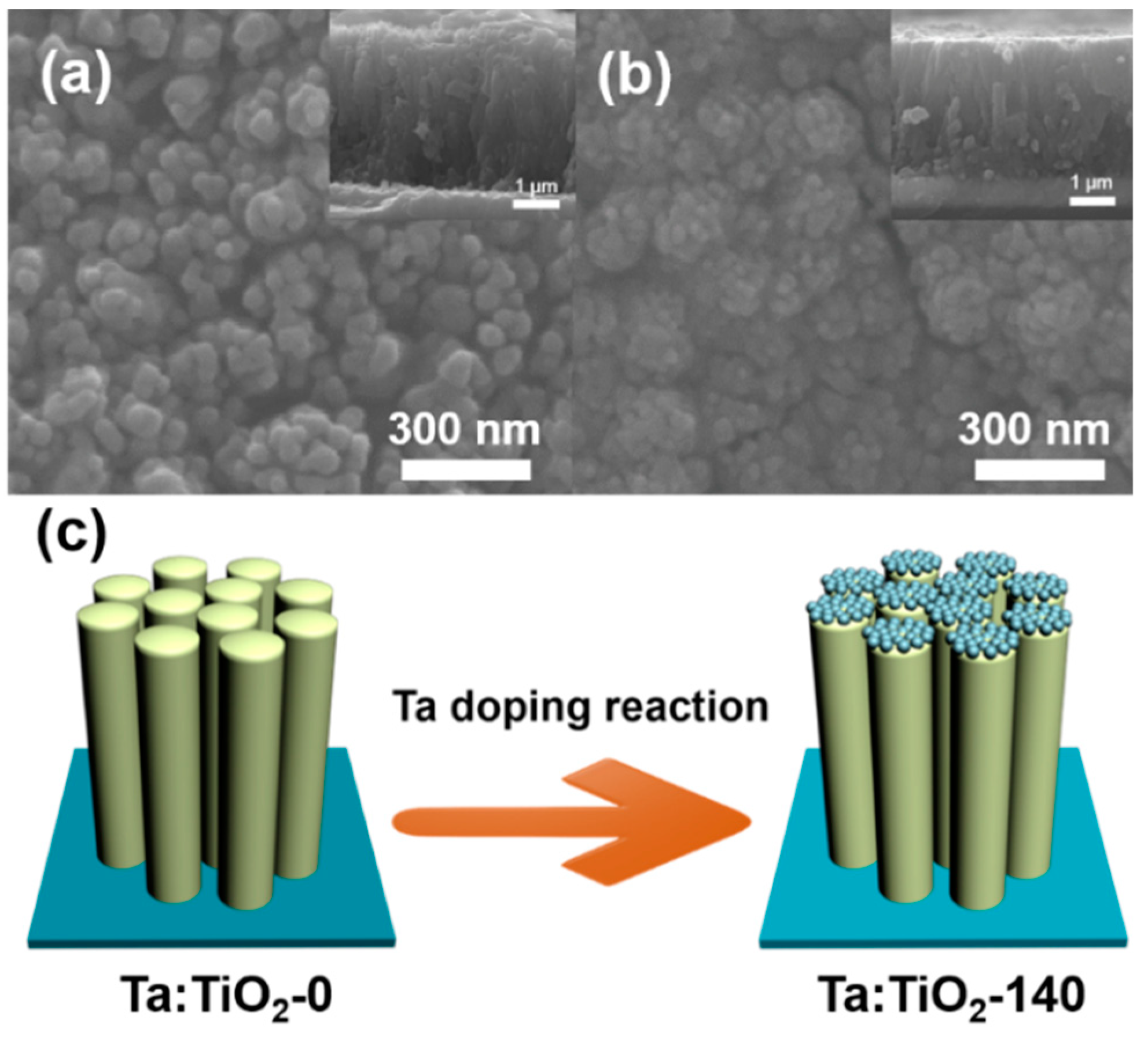
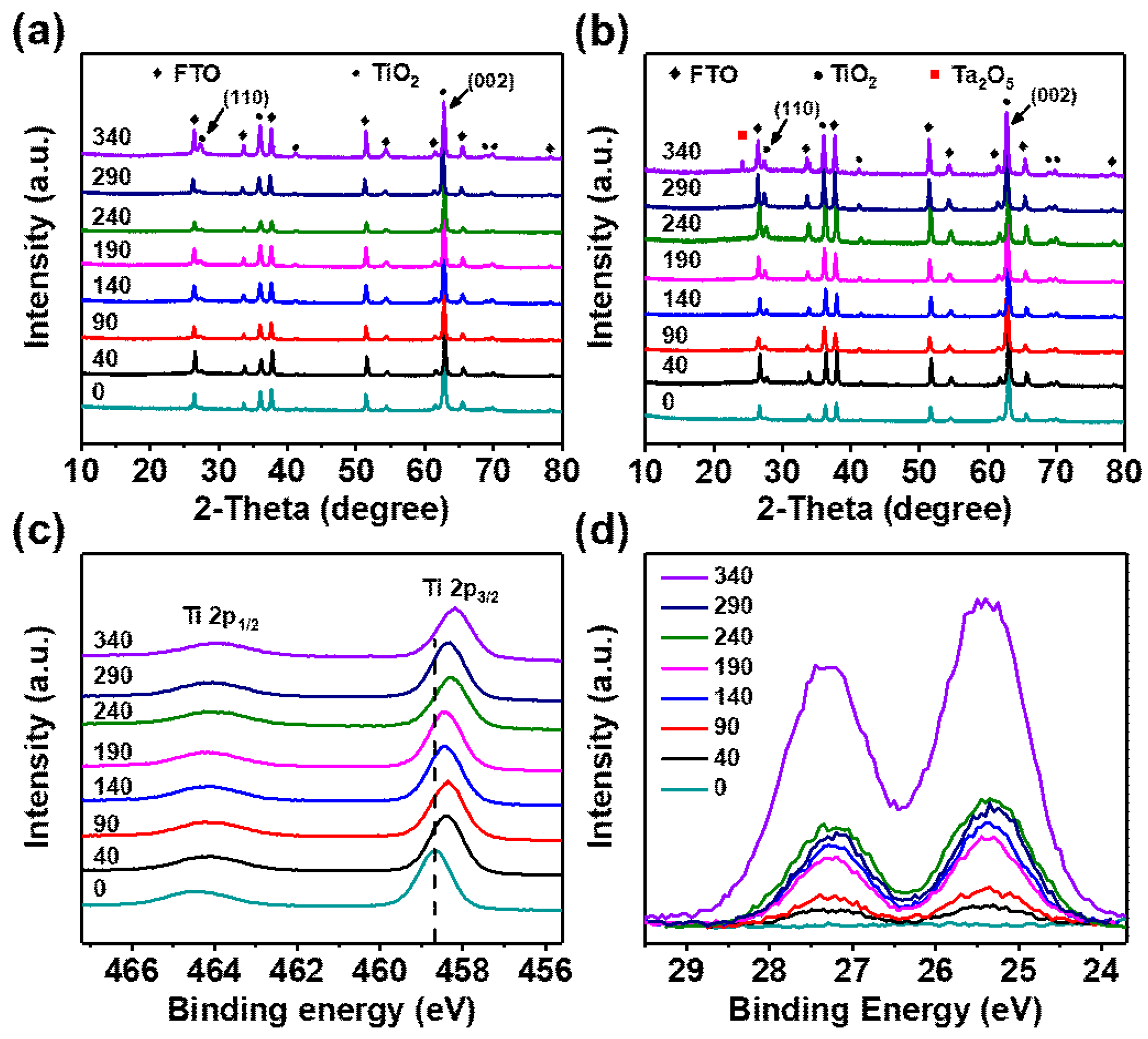
3.1. Morphology and Structural Characteristics
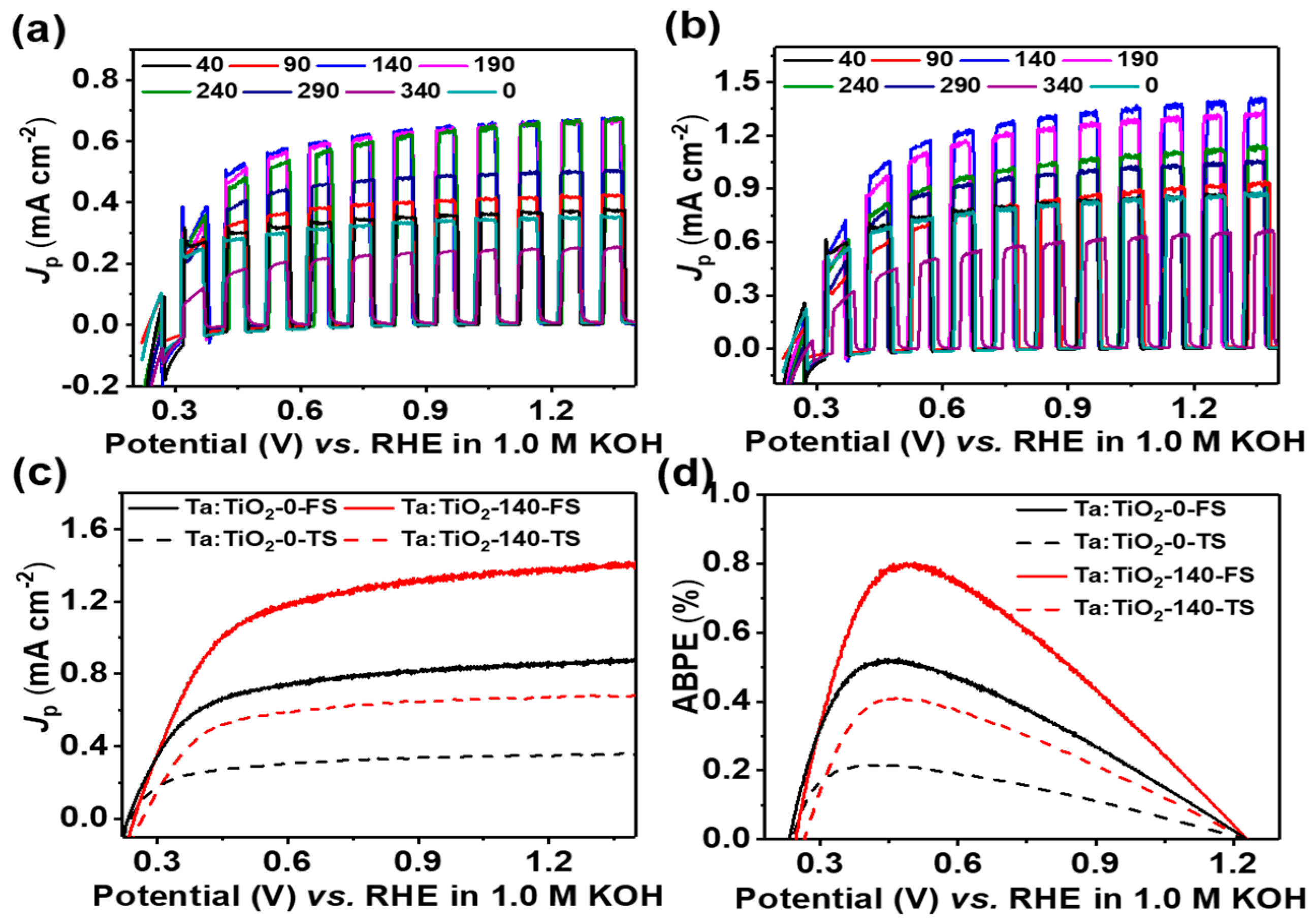
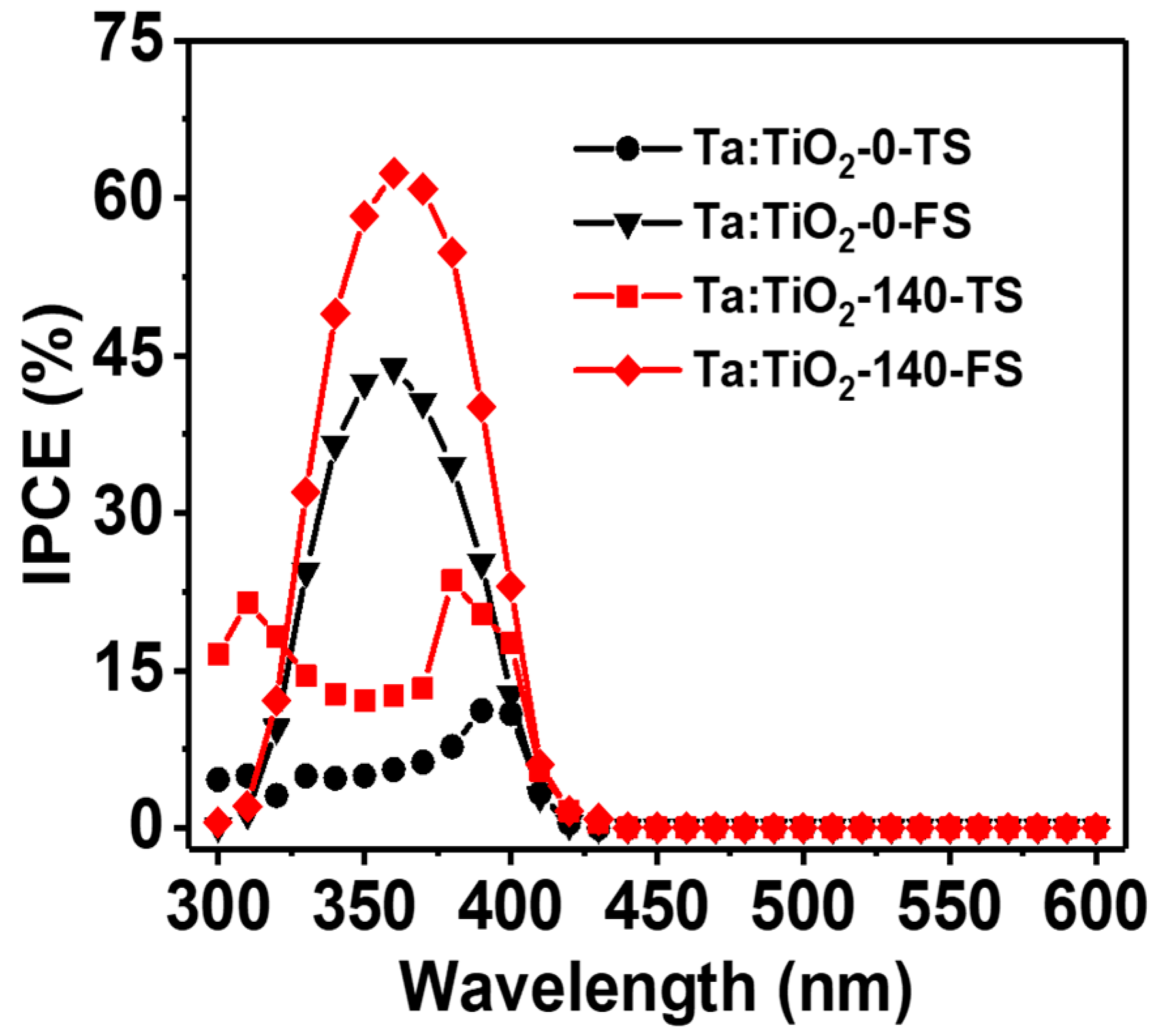
3.2. Photoelectrochemical Performance
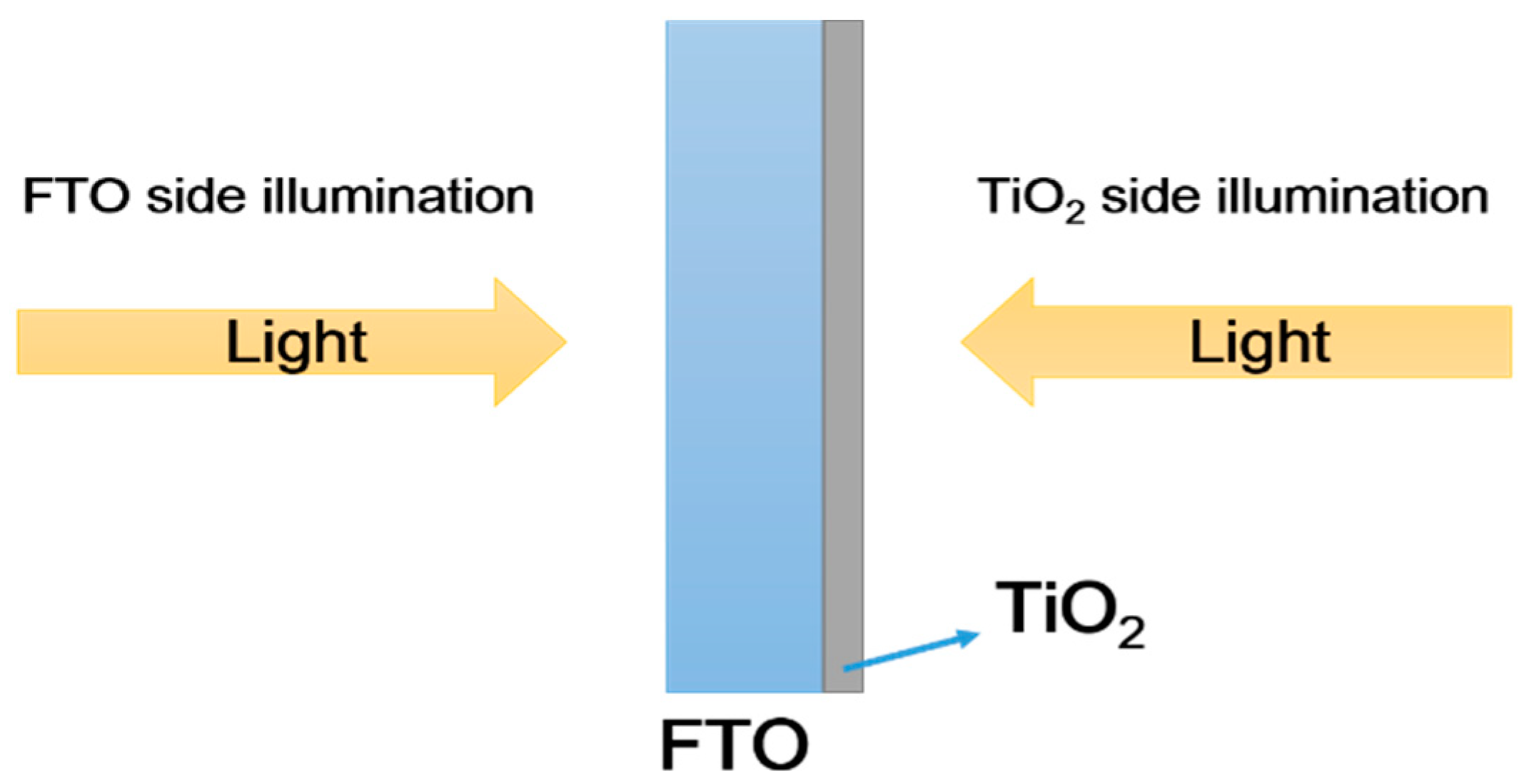
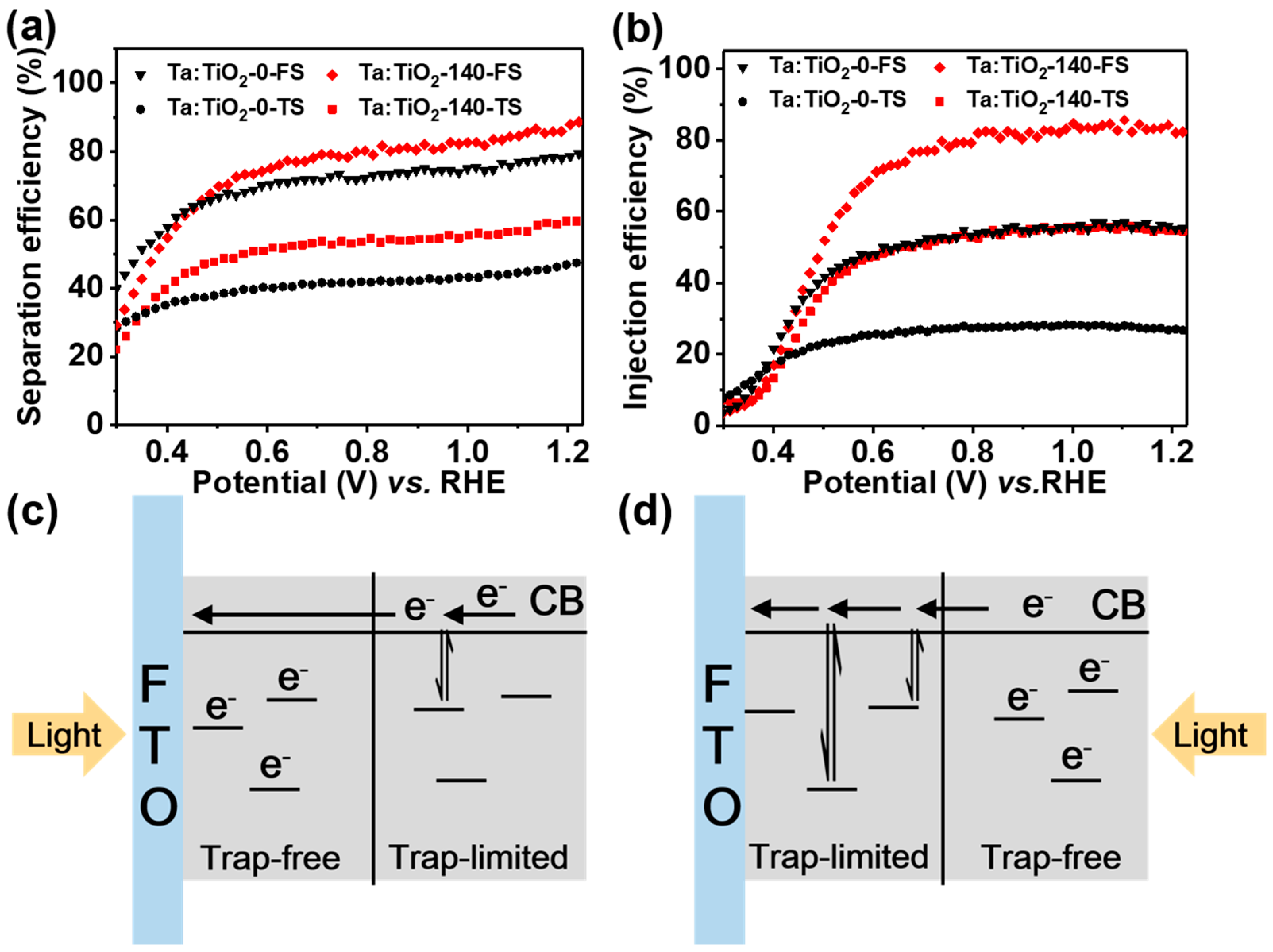
3.3. Charge Separation and Injection Mechanism
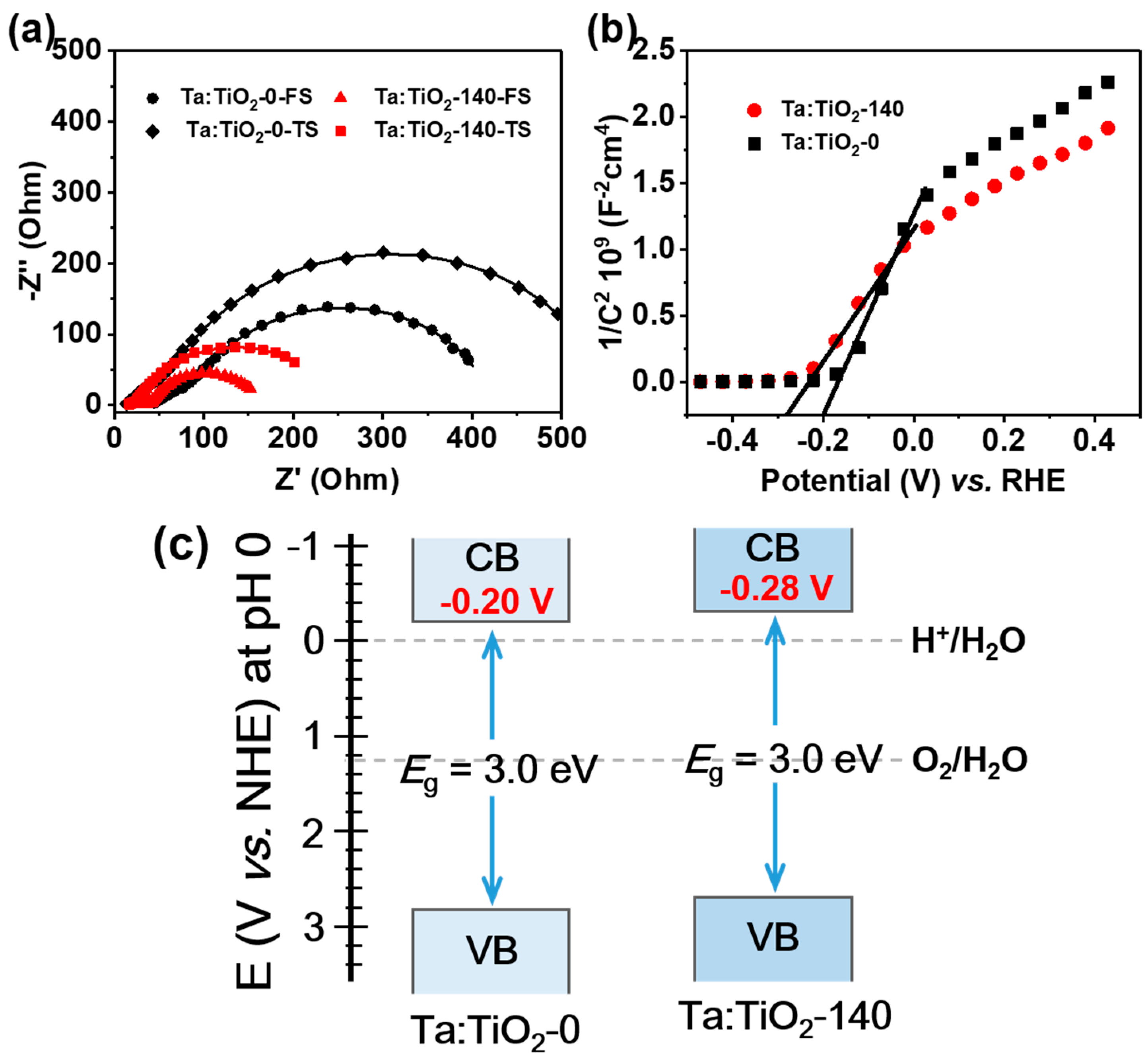
3.4. EIS and Band Structure Characteristics
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Chen, X.B.; Shen, S.H.; Guo, L.J.; Mao, S.S. Semiconductor-based photocatalytic hydrogen generation. Chem. Rev. 2010, 110, 6503–6570. [Google Scholar] [CrossRef] [PubMed]
- Grätzel, M. Photoelectrochemical cells. Nature 2001, 414, 338–344. [Google Scholar] [CrossRef] [PubMed]
- Fujishima, A.; Honda, K. Electrochemical photolysis of water at a semiconductor electrode. Nature 1972, 238, 37–38. [Google Scholar] [CrossRef] [PubMed]
- Hwang, Y.J.; Hahn, C.; Liu, B.; Yang, P.D. Photoelectrochemical properties of TiO2 nanowire arrays: A study of the dependence on length and atomic layer deposition coating. ACS Nano 2012, 6, 5060–5069. [Google Scholar] [CrossRef] [PubMed]
- Ge, M.; Li, Q.; Cao, C.; Huang, J.; Li, S.; Zhang, S.; Chen, Z.; Zhang, K.; Al-Deyab, S.S.; Lai, Y. One-dimensional TiO2 nanotube photocatalysts for solar water splitting. Adv. Sci. 2017, 4, 1600152. [Google Scholar] [CrossRef] [PubMed]
- Zhan, F.; Liu, W.; Li, H.; Yang, Y.; Wang, M. Ce-doped CdS quantum dot sensitized TiO2 nanorod films with enhanced visible-light photoelectrochemical properties. Appl. Surf. Sci. 2018, 455, 476–483. [Google Scholar] [CrossRef]
- Cheng, Q.; Fan, W.; He, Y.; Ma, P.; Vanka, S.; Fan, S.; Mi, Z.; Wang, D. Photorechargeable high voltage redox battery enabled by Ta3N5 and GaN/Si dual-photoelectrode. Adv. Mater. 2017, 29. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Bao, C.; Yu, T.; Hu, Y.; Luo, W.; Zhu, W.; Fu, G.; Li, Z.; Gao, H.; Li, F.; et al. Enhanced performance of photoelectrochemical water splitting with ITO@alpha-Fe2O3 core-shell nanowire array as photoanode. ACS Appl. Mater. Interfaces 2015, 7, 26482–26490. [Google Scholar] [CrossRef] [PubMed]
- Feng, X.J.; Shankar, K.; Paulose, M.; Grimes, C.A. Tantalum-doped titanium dioxide nanowire arrays for dye-sensitized solar cells with high open-circuit voltage. Angew. Chem. Int. Ed. 2009, 48, 8095–8098. [Google Scholar] [CrossRef] [PubMed]
- Gong, Y.Y.; Fu, C.X.; Ting, L.; Chen, J.R.; Zhao, Q.E.; Li, C. Exploring the effect of boron and tantalum codoping on the enhanced photocatalytic activity of TiO2. Appl. Surf. Sci. 2015, 351, 746–752. [Google Scholar] [CrossRef]
- Yan, Y.; Lee, J.; Cui, X. Enhanced photoelectrochemical properties of Ta-TiO2 nanotube arrays prepared by magnetron sputtering. Vacuum 2017, 138, 30–38. [Google Scholar] [CrossRef]
- Zhou, L.; Yang, Y.; Zhang, J.; Rao, P.M. Photoanode with enhanced performance achieved by coating BiVO4 onto ZnO-templated Sb-doped SnO2 nanotube scaffold. ACS Appl. Mater. Interfaces 2017, 9, 11356–11362. [Google Scholar] [CrossRef] [PubMed]
- Hoang, S.; Guo, S.W.; Mullins, C.B. Coincorporation of N and Ta into TiO2 nanowires for visible light driven photoelectrochemical water oxidation. J. Phys. Chem. C 2012, 116, 23283–23290. [Google Scholar] [CrossRef]
- Yang, X.F.; Chen, J.; Gong, L.; Wu, M.M.; Yu, J.C. Cross-medal arrays of Ta-doped rutile titania. J. Am. Chem. Soc. 2009, 131, 12048–12049. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Aydil, E.S. Growth of oriented single-crystalline rutile TiO2 nanorods on transparent conducting substrates for dye-sensitized solar cells. J. Am. Chem. Soc. 2009, 131, 3985–3990. [Google Scholar] [CrossRef] [PubMed]
- Bawaked, S.M.; Sathasivam, S.; Bhachu, D.S.; Chadwick, N.; Obaid, A.Y.; Al-Thabaiti, S.; Basahel, S.N.; Carmalt, C.J.; Parkin, I.P. Aerosol assisted chemical vapor deposition of conductive and photocatalytically active tantalum doped titanium dioxide films. J. Mater. Chem. A 2014, 2, 12849–12856. [Google Scholar] [CrossRef]
- Reitz, C.; Reinacher, J.; Hartmann, P.; Brezesinski, T. Polymer-templated ordered large-pore mesoporous anatase rutile TiO2:Ta nanocomposite films: Microstructure, electrical conductivity, and photocatalytic and photoelectrochemical properties. Catal. Today 2014, 225, 55–63. [Google Scholar] [CrossRef]
- Yang, X.F.; Jin, C.J.; Liang, C.L.; Chen, D.H.; Wu, M.M.; Yu, J.C. Nanoflower arrays of rutile TiO2. Chem. Commun. 2011, 47, 1184–1186. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.F.; Zhuang, J.L.; Li, X.Y.; Chen, D.H.; Ouyang, G.F.; Mao, Z.Q.; Han, Y.X.; He, Z.H.; Liang, C.L.; Wu, M.M.; et al. Hierarchically nanostructured rutile arrays. Acid vapor oxidation growth and tunable morphologies. ACS Nano 2009, 3, 1212–1218. [Google Scholar] [CrossRef] [PubMed]
- Altomare, M.; Lee, K.; Killian, M.S.; Selli, E.; Schmuki, P. Ta-Doped TiO2 nanotubes for enhanced solar-light photoelectrochemical water splitting. Chem.-Eur. J. 2013, 19, 5841–5844. [Google Scholar] [CrossRef] [PubMed]
- Li, A.; Wang, Z.; Yin, H.; Wang, S.; Yan, P.; Huang, B.; Wang, X.; Li, R.; Zong, X.; Han, H.; et al. Understanding the anatase-rutile phase junction in charge separation and transfer in a TiO2 electrode for photoelectrochemical water splitting. Chem. Sci. 2016, 7, 6076–6082. [Google Scholar] [CrossRef] [PubMed]
- Mao, C.Y.; Zuo, F.; Hou, Y.; Bu, X.H.; Feng, P.Y. In situ preparation of a Ti3+ self-doped TiO2 film with enhanced activity as photoanode by N2H4 reduction. Angew. Chem. Int. Ed. 2014, 53, 10485–10489. [Google Scholar] [CrossRef] [PubMed]
- Carroll, G.M.; Zhong, D.K.; Gamelin, D.R. Mechanistic insights into solar water oxidation by cobalt-phosphate-modified alpha-Fe2O3 photoanodes. Energy Environ. Sci. 2015, 8, 577–584. [Google Scholar] [CrossRef]
- Zhang, L.W.; Reisner, E.; Baumberg, J.J. Al-doped ZnO inverse opal networks as efficient electron collectors in BiVO4 photoanodes for solar water oxidation. Energy Environ. Sci. 2014, 7, 1402–1408. [Google Scholar] [CrossRef]
- Ai, G.J.; Mo, R.; Li, H.X.; Zhong, J.X. Cobalt phosphate modified TiO2 nanowire arrays as co-catalysts for solar water splitting. Nanoscale 2015, 7, 6722–6728. [Google Scholar] [CrossRef] [PubMed]
- Antony, R.P.; Bassi, P.S.; Abdi, F.F.; Chiam, S.Y.; Ren, Y.; Barber, J.; Loo, J.S.C.; Wong, L.H. Electrospun Mo-BiVO4 for efficient photoelectrochemical water oxidation: Direct evidence of improved hole diffusion length and charge separation. Electrochim. Acta 2016, 211, 173–182. [Google Scholar] [CrossRef]
- Hong, S.J.; Jun, H.; Borse, P.H.; Lee, J.S. Size effects of WO3 nanocrystals for photooxidation of water in particulate suspension and photoelectrochemical film systems. Int. J. Hydrogen Energy 2009, 34, 3234–3242. [Google Scholar] [CrossRef]
- Xiao, S.; Chen, H.; Yang, Z.; Long, X.; Wang, Z.; Zhu, Z.; Qu, Y.; Yang, S. Origin of the different photoelectrochemical performance of mesoporous BiVO4 photoanodes between the BiVO4 and the FTO side illumination. J. Phys. Chem. C 2015, 119, 23350–23357. [Google Scholar] [CrossRef]
- Ye, K.-H.; Chai, Z.; Gu, J.; Yu, X.; Zhao, C.; Zhang, Y.; Mai, W. BiOI-BiVO4 photoanodes with significantly improved solar water splitting capability: P-n junction to expand solar adsorption range and facilitate charge carrier dynamics. Nano Energy 2015, 18, 222–231. [Google Scholar] [CrossRef]
- Li, C.; Li, A.; Luo, Z.; Zhang, J.; Chang, X.; Huang, Z.; Wang, T.; Gong, J. Surviving high-temperature calcination: ZrO2-induced hematite nanotubes for photoelectrochemical water oxidation. Angew. Chem. Int. Ed. 2017, 129, 4214–4219. [Google Scholar] [CrossRef]
- Cho, I.S.; Lee, C.H.; Feng, Y.Z.; Logar, M.; Rao, P.M.; Cai, L.L.; Kim, D.R.; Sinclair, R.; Zheng, X.L. Codoping titanium dioxide nanowires with tungsten and carbon for enhanced photoelectrochemical performance. Nat. Commun. 2013, 4, 1723. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.W.; Choi, K.S. Nanoporous BiVO4 photoanodes with dual-layer oxygen evolution catalysts for solar water splitting. Science 2014, 343, 990–994. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Yu, Y.; Starr, M.B.; Yin, X.; Li, Z.; Kvit, A.; Wang, S.; Zhao, P.; Wang, X. Ferroelectric polarization-enhanced photoelectrochemical water splitting in TiO2-BaTiO3 core-shell nanowire photoanodes. Nano Lett. 2015, 15, 7574–7580. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.J.; Lee, S.; Jang, J.S.; Lee, J.S. Heterojunction BiVO4/WO3 electrodes for enhanced photoactivity of water oxidation. Energy Environ. Sci. 2011, 4, 1781–1787. [Google Scholar] [CrossRef]
- Yang, J.-S.; Wu, J.-J. Low-potential driven fully-depleted BiVO4/ZnO heterojunction nanodendrite array photoanodes for photoelectrochemical water splitting. Nano Energy 2017, 32, 232–240. [Google Scholar] [CrossRef]
- Yan, P.L.; Liu, G.J.; Ding, C.M.; Han, H.X.; Shi, J.Y.; Gan, Y.; Li, C. Photoelectrochemical water splitting promoted with a disordered surface layer created by electrochemical reduction. ACS Appl. Mater. Interfaces 2015, 7, 3791–3796. [Google Scholar] [CrossRef] [PubMed]
- Shao, M.F.; Ning, F.Y.; Wei, M.; Evans, D.G.; Duan, X. Hierarchical nanowire arrays based on ZnO core-layered double hydroxide shell for largely enhanced photoelectrochemical water splitting. Adv. Funct. Mater. 2014, 24, 580–586. [Google Scholar] [CrossRef]
- Li, J.; Guo, L.; Lei, N.; Song, Q.; Liang, Z. Metallic Bi nanocrystal-modified defective BiVO4 photoanodes with exposed (040) facets for photoelectrochemical water splitting. ChemElectroChem 2017, 4, 2852–2861. [Google Scholar] [CrossRef]
- Tong, X.L.; Yang, P.; Wang, Y.W.; Qin, Y.; Guo, X.Y. Enhanced photoelectrochemical water splitting performance of TiO2 nanotube arrays coated with an ultrathin nitrogen-doped carbon film by molecular layer deposition. Nanoscale 2014, 6, 6692–6700. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.H.; Wang, P. Optimization of photoelectrochemical water splitting performance on hierarchical TiO2 nanotube arrays. Energy Environ. Sci. 2012, 5, 6506–6512. [Google Scholar] [CrossRef]
- Hou, Y.; Zuo, F.; Dagg, A.P.; Liu, J.K.; Feng, P.Y. Branched WO3 nanosheet array with layered C3N4 heterojunctions and CoOx nanoparticles as a flexible photoanode for efficient photoelectrochemical water oxidation. Adv. Mater. 2014, 26, 5043–5049. [Google Scholar] [CrossRef] [PubMed]
- Dong, Z.; Ding, D.; Li, T.; Ning, C. Ni-doped TiO2 nanotubes photoanode for enhanced photoelectrochemical water splitting. Appl. Surf. Sci. 2018, 443, 321–328. [Google Scholar] [CrossRef]
- Chen, X.; Liu, L.; Yu, P.Y.; Mao, S.S. Increasing solar absorption for photocatalysis with black hydrogenated titanium dioxide nanocrystals. Science 2011, 331, 746–750. [Google Scholar] [CrossRef] [PubMed]
- He, S.; Meng, Y.; Wu, Q.; Yang, J.; Huang, S.; Li, X.; Tong, S.; Asefa, T.; Wu, M. Ta-doped porous TiO2 nanorod arrays by substrate-assisted synthesis: Efficient photoelectrocatalysts for water oxidation. Nanoscale 2018, 10, 19367–19374. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Zhang, W.D. Modification of TiO2 nanorod arrays by graphite-like C3N4 with high visible light photoelectrochemical activity. Electrochim. Acta 2012, 71, 10–16. [Google Scholar] [CrossRef]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
He, S.; Meng, Y.; Cao, Y.; Huang, S.; Yang, J.; Tong, S.; Wu, M. Hierarchical Ta-Doped TiO2 Nanorod Arrays with Improved Charge Separation for Photoelectrochemical Water Oxidation under FTO Side Illumination. Nanomaterials 2018, 8, 983. https://doi.org/10.3390/nano8120983
He S, Meng Y, Cao Y, Huang S, Yang J, Tong S, Wu M. Hierarchical Ta-Doped TiO2 Nanorod Arrays with Improved Charge Separation for Photoelectrochemical Water Oxidation under FTO Side Illumination. Nanomaterials. 2018; 8(12):983. https://doi.org/10.3390/nano8120983
Chicago/Turabian StyleHe, Shiman, Yuying Meng, Yangfei Cao, Senchuan Huang, Jingling Yang, Shengfu Tong, and Mingmei Wu. 2018. "Hierarchical Ta-Doped TiO2 Nanorod Arrays with Improved Charge Separation for Photoelectrochemical Water Oxidation under FTO Side Illumination" Nanomaterials 8, no. 12: 983. https://doi.org/10.3390/nano8120983
APA StyleHe, S., Meng, Y., Cao, Y., Huang, S., Yang, J., Tong, S., & Wu, M. (2018). Hierarchical Ta-Doped TiO2 Nanorod Arrays with Improved Charge Separation for Photoelectrochemical Water Oxidation under FTO Side Illumination. Nanomaterials, 8(12), 983. https://doi.org/10.3390/nano8120983





