Enhanced Peroxidase-Like Activity of MoS2 Quantum Dots Functionalized g-C3N4 Nanosheets towards Colorimetric Detection of H2O2
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials and Reagents
2.2. Preparation of the Catalysts
2.3. Characterization
2.4. Peroxidase-Like Activities and Steady-State Kinetic Assay
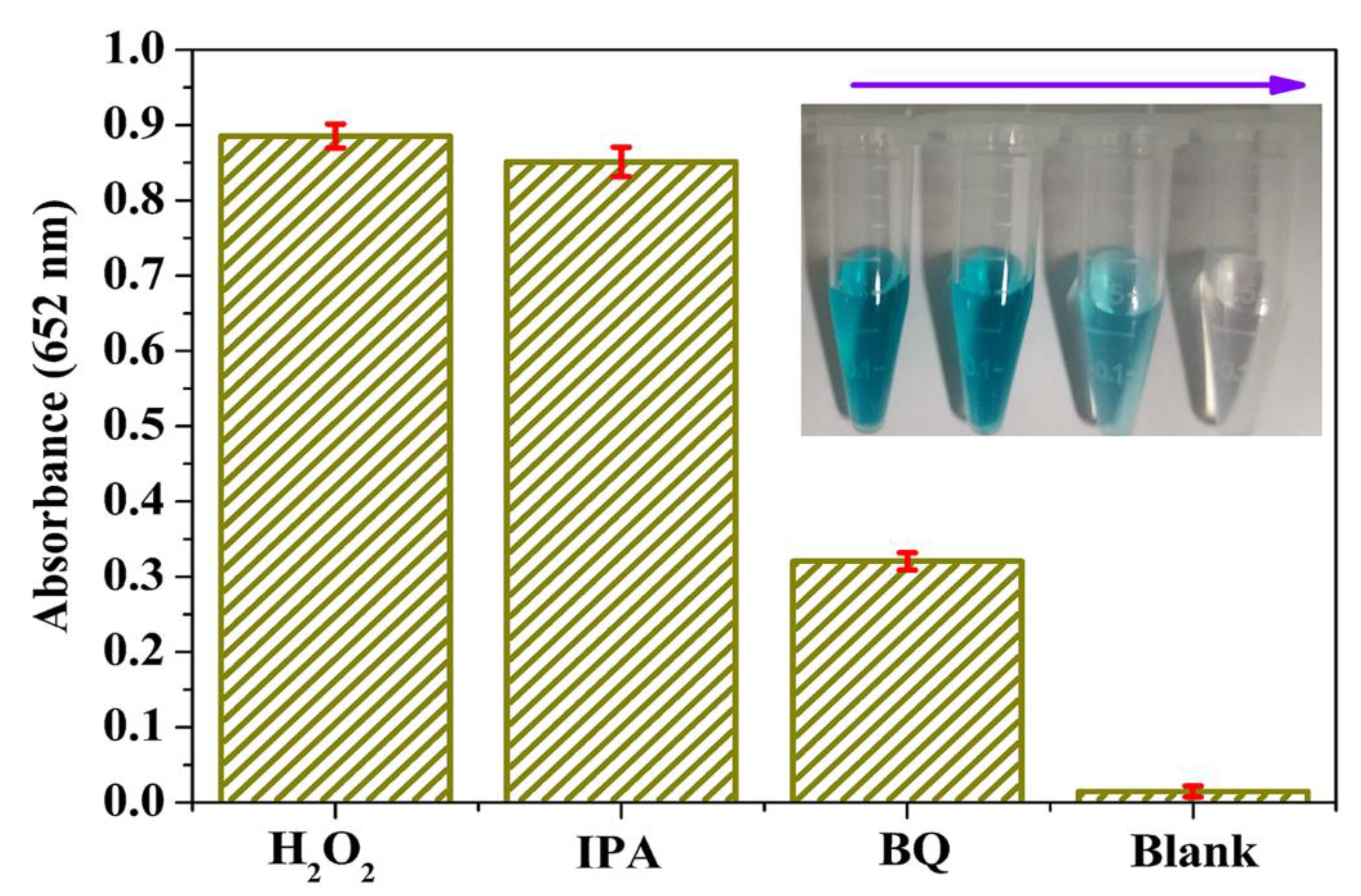
2.5. Analysis of Active Species
2.6. H2O2 Detection
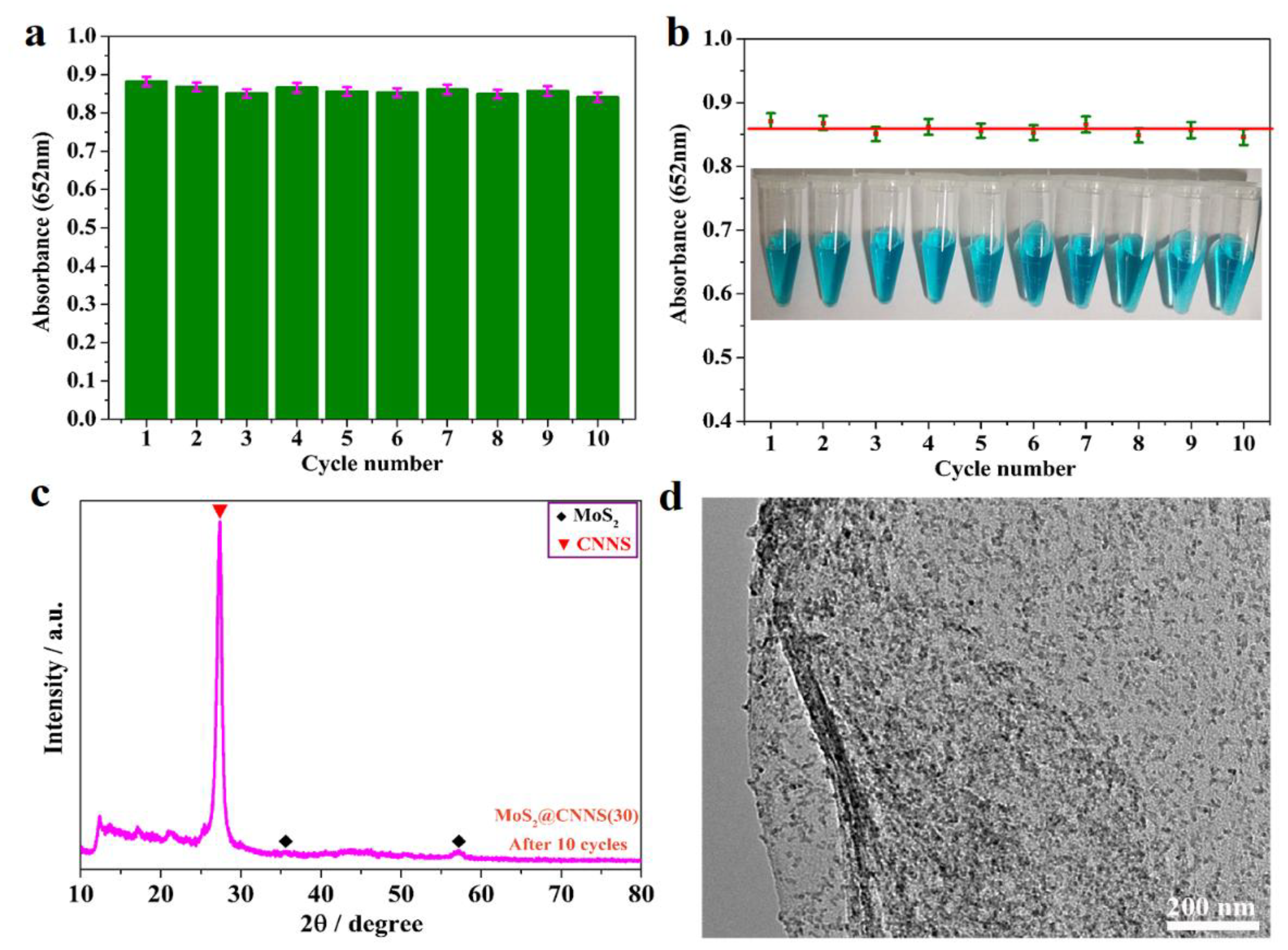
2.7. Stability and Reusability of the Catalysts
3. Results
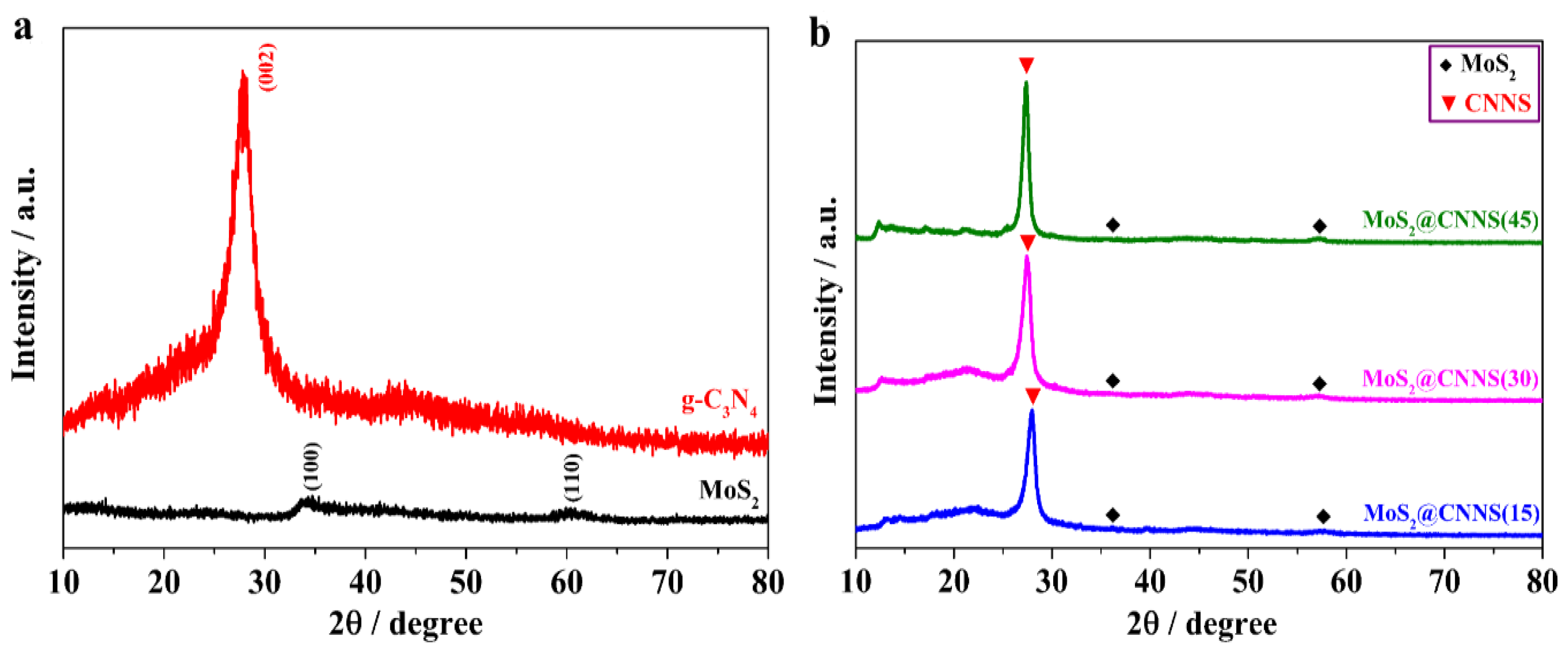
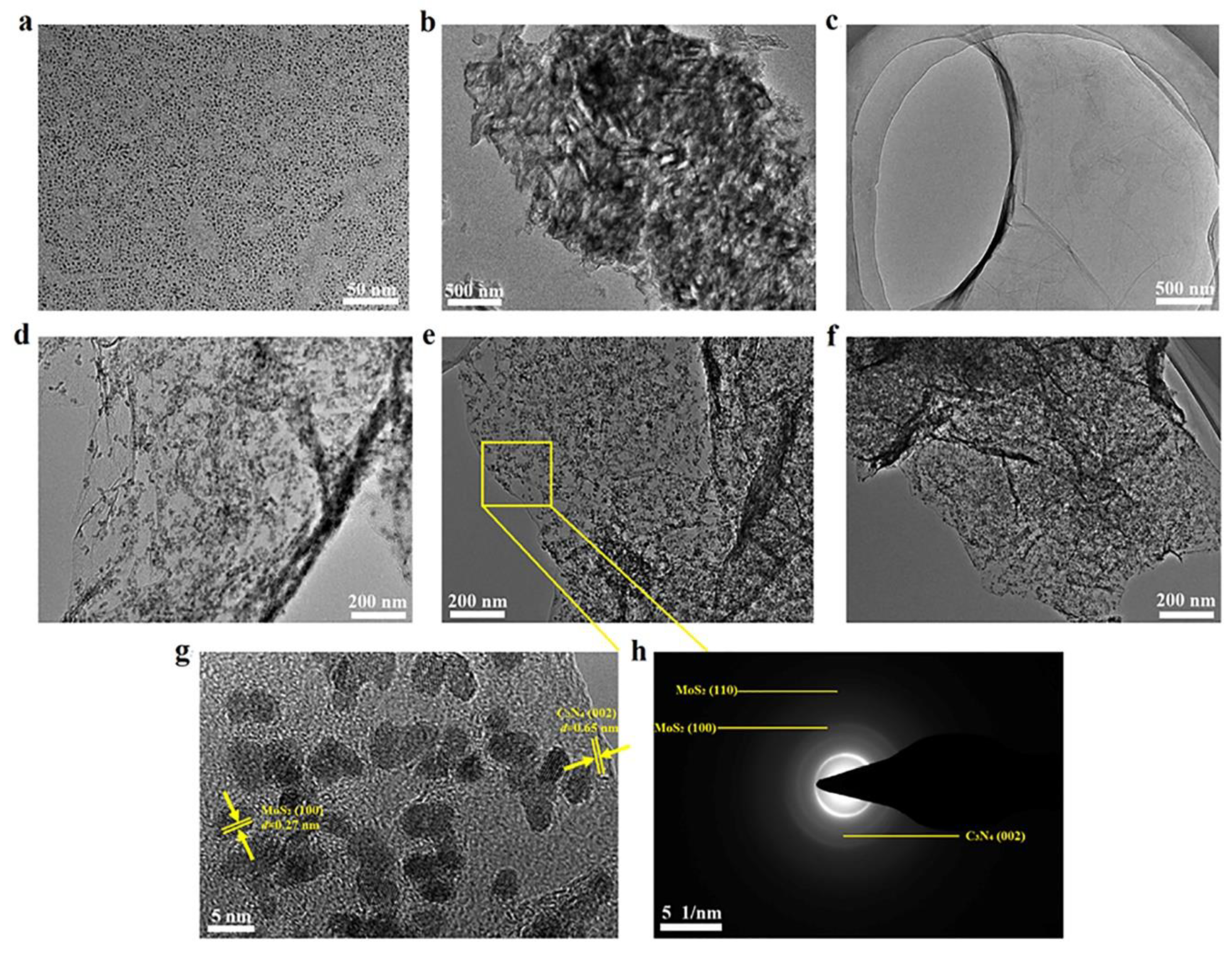
3.1. Characterization of the Catalysts
3.2. Peroxidase-Like Activities of MoS2@CNNS(30) Nanocomposites
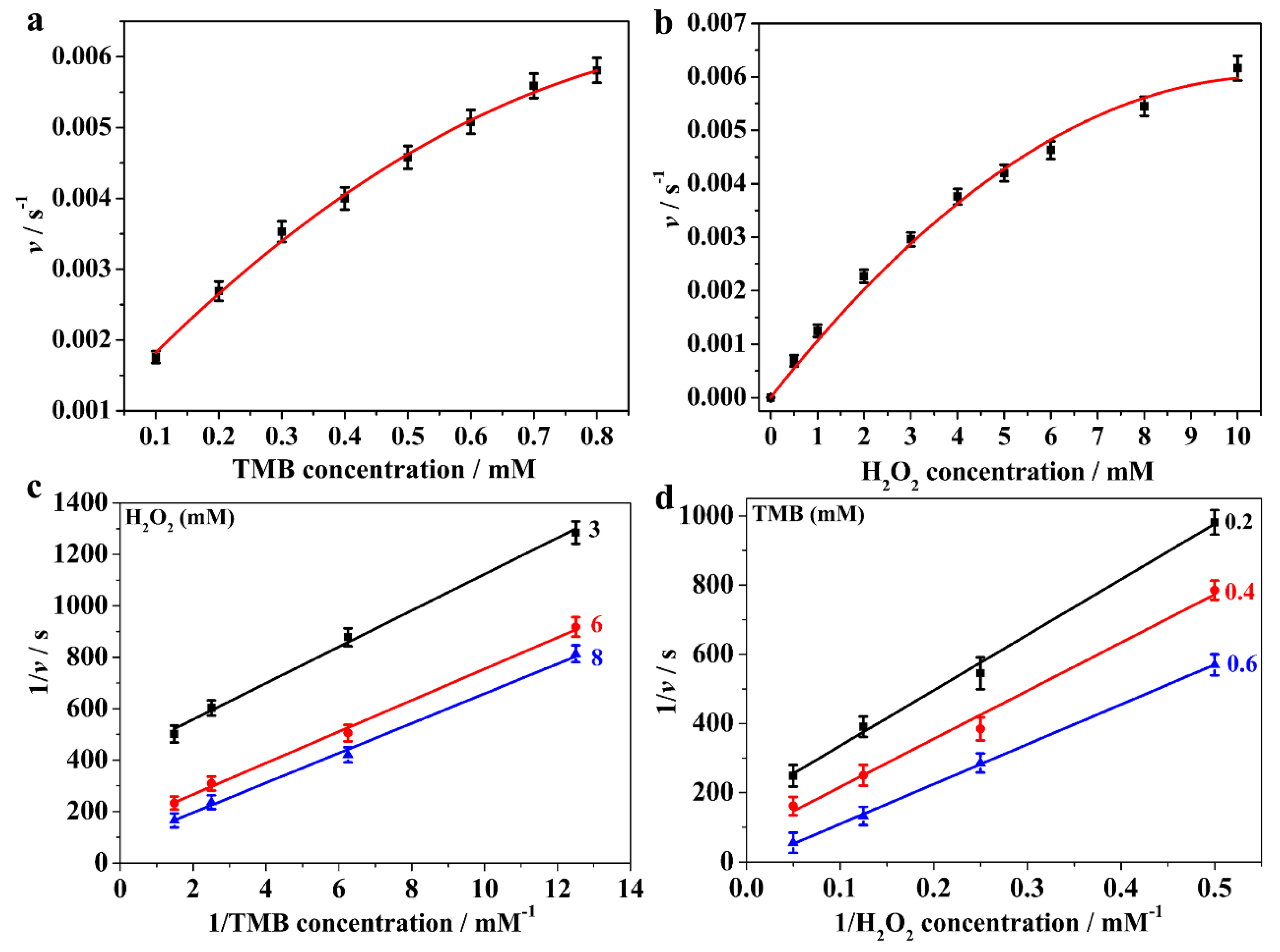
3.3. Steady-State Kinetics Assay
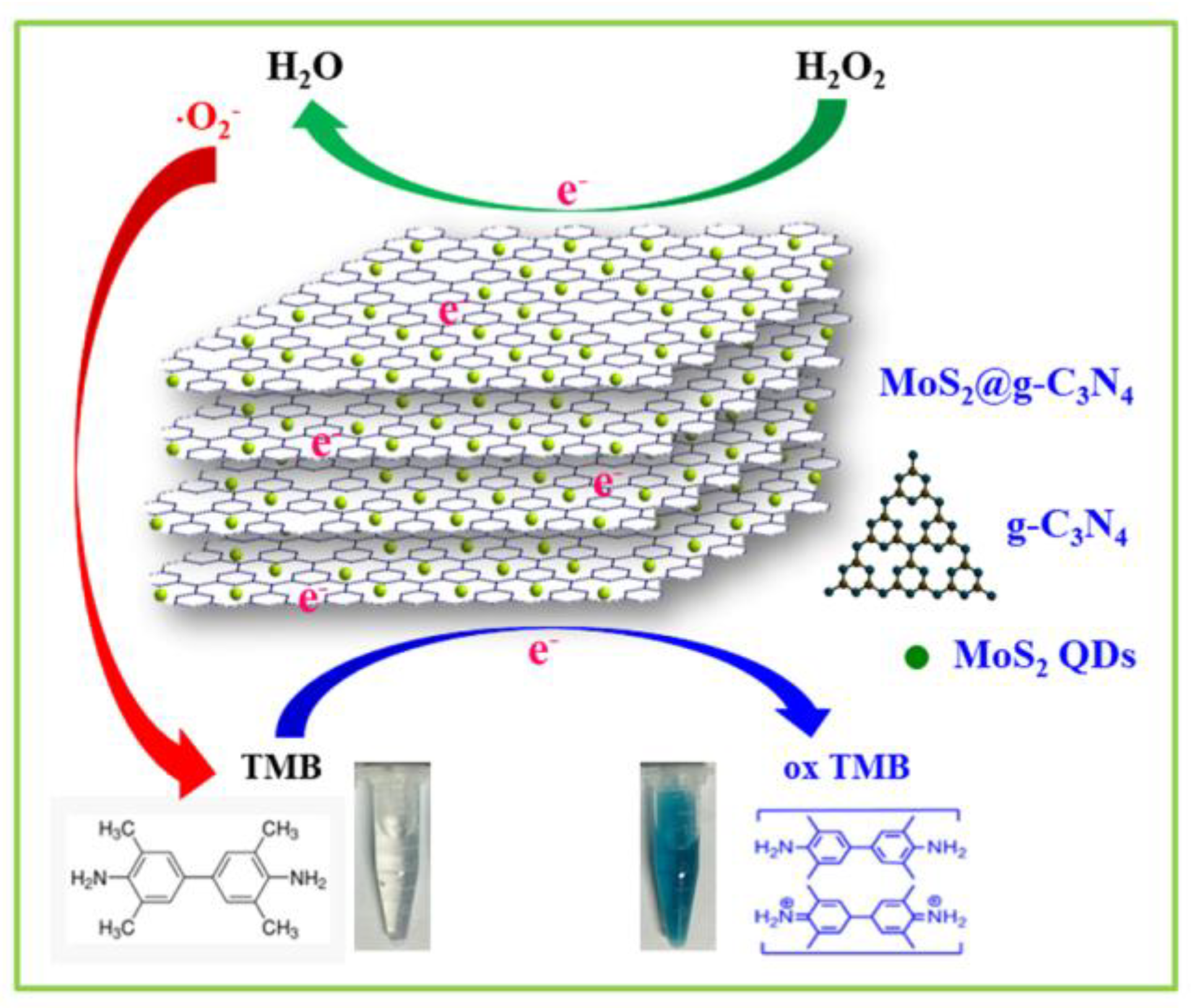
3.4. Active Species Analysis and Peroxidase-Like Catalytic Mechanism Study
3.5. Detection of H2O2 by MoS2@CNNS(30)-Based Assay System
3.6. Selectivity and Applicability of MoS2@CNNS(30)-Based Assay System
3.7. Stability and Reusability of MoS2@CNNS(30)-Based Assay System
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Song, Y.J.; Wei, W.L.; Qu, X.G. Colorimetric biosensing using smart materials. Adv. Mater. 2011, 23, 4215–4236. [Google Scholar] [CrossRef] [PubMed]
- Wei, H.; Wang, E.K. Nanomaterials with enzyme-like characteristics (nanozymes): Next-generation artificial enzymes. Chem. Soc. Rev. 2013, 42, 6060–6093. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.Q.; Wei, H.; Zhang, Z.Q.; Wang, E.K.; Dong, S.J. Nanozyme: An emerging alternative to natural enzyme for biosensing and immunoassay. TrAC-Trend. Anal. Chem. 2018, 105, 218–224. [Google Scholar] [CrossRef]
- Gao, L.; Zhuang, J.; Nie, L.; Zhang, J.; Zhang, Y.; Gu, N.; Wang, T.; Feng, J.; Yang, D.; Perrett, S.; Yan, X. Intrinsic peroxidase-like activity of ferromagnetic nanoparticles. Nat. Nanotechnol. 2007, 2, 577–583. [Google Scholar] [CrossRef] [PubMed]
- Tian, R.; Sun, J.H.; Qi, Y.F.; Zhang, B.Y.; Guo, S.L.; Zhao, M.M. Influence of VO2 nanoparticle morphology on the colorimetric assay of H2O2 and glucose. Nanomaterials 2017, 7, 347. [Google Scholar] [CrossRef] [PubMed]
- Zheng, H.Q.; Liu, C.Y.; Zeng, X.Y.; Chen, J.; Lv, J.; Lin, R.G.; Cao, R.; Lin, Z.J.; Su, J.W. MOF-808: A metal-organic framework with intrinsic peroxidase-like catalytic activity at neutral pH for colorimetric bciosensing. Inorg. Chem. 2018, 57, 9096–9104. [Google Scholar] [CrossRef] [PubMed]
- Zhou, R.; Guzman, M.I. Photocatalytic reduction of fumarate to succinate on ZnS mineral surfaces. J. Phys. Chem. C 2016, 120, 7349–7357. [Google Scholar] [CrossRef]
- Vázquez-Fernández, M.Á.; Bermejo, M.R.; Fernández-García, M.I.; González-Riopedre, G.; Rodríguez-Doutón, M.J.; Maneiro, M. Influence of the geometry around the manganese ion on the peroxidase and catalase activities of Mn(III)=Schiff base complexes. J. Inorg. Biochem. 2011, 105, 1538–1547. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Ji, H. 2-Hydroxypropyl-β-cyclodextrin polymer as a mimetic enzyme for mediated synthesis of benzaldehyde in water. ACS Sustain. Chem. Eng. 2013, 1, 1172–1179. [Google Scholar] [CrossRef]
- Wang, Q.G.; Yang, Z.M.; Zhang, X.Q.; Xiao, X.D.; Chang, C.K.; Xu, B. A supramolecular-hydrogel-encapsulated hemin as an artificial enzyme to mimic peroxidase. Angew. Chem. Int. Ed. 2007, 46, 4285–4289. [Google Scholar] [CrossRef] [PubMed]
- Tan, B.; Zhao, H.M.; Wu, W.H.; Liu, X.; Zhang, Y.B.; Quan, X. Fe3O4-AuNPs anchored 2D metal-organic framework nanosheets with DNA regulated switchable peroxidase-like activity. Nanoscale 2017, 9, 18699–18710. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.H.; Wan, G.P.; Hu, N.; He, Z.Y.; Shi, S.H.; Suo, Y.R.; Wang, K.; Xu, X.F.; Tang, Y.L.; Wang, G.Z. Synthesis of porous CoFe2O4 and its application as a peroxidase mimetic for colorimetric detection of H2O2 and organic pollutant degradation. Nanomaterials 2017, 7, 451. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.Z.; Ju, P.; Zhang, D.; Han, X.X.; Yin, X.F.; Zheng, L.; Sun, C.J. Peroxidase-like activity of FeVO4 nanobelts and its analytical application for optical detection of hydrogen peroxide. Sensor. Actuator. B Chem. 2016, 233, 162–172. [Google Scholar] [CrossRef]
- Wang, H.; Li, P.H.; Yu, D.Q.; Zhang, Y.; Wang, Z.Z.; Liu, C.Q.; Qiu, H.; Liu, Z.; Ren, J.S.; Qu, X.G. Unraveling the enzymatic activity of oxygenated carbon nanotubes and their application in the treatment of bacterial infections. Nano Lett. 2018, 6, 3344–3351. [Google Scholar] [CrossRef] [PubMed]
- Singh, V.K.; Yadav, P.K.; Chandra, S.; Bano, D.; Talat, M.; Hasan, S.H. Peroxidase mimetic activity of fluorescent NS-carbon quantum dots and their application in colorimetric detection of H2O2 and glutathione in human blood serum. J. Mater. Chem. B 2018, 6, 5256–5268. [Google Scholar] [CrossRef]
- Song, Y.; Qu, K.; Zhao, C.; Ren, J.; Qu, X. Graphene oxide: intrinsic peroxidase catalytic activity and its application to glucose detection. Adv. Mater. 2010, 22, 2206–2210. [Google Scholar] [CrossRef] [PubMed]
- Lin, T.R.; Zhong, L.S.; Wang, J.; Guo, L.Q.; Wu, H.Y.; Guo, Q.Q.; Fu, F.F.; Chen, G.N. Graphite-like carbon nitrides as peroxidase mimetics and their applications to glucose detection. Biosens. Bioelectron. 2014, 59, 89–93. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.J.; Wang, E.K. Noble metal nanomaterials: Controllable synthesis and application in fuel cells and analytical sensors. Nano Today 2011, 6, 240–264. [Google Scholar] [CrossRef]
- Xiang, Z.B.; Wang, Y.; Ju, P.; Zhang, D. Optical determination of hydrogen peroxide by exploiting the peroxidase-like activity of AgVO3 nanobelts. Microchim. Acta 2016, 183, 457–463. [Google Scholar] [CrossRef]
- Qiao, F.M.; Wang, Z.; Xu, K.; Ai, S.Y. Double enzymatic cascade reactions within FeSe-Pt@SiO2 nanospheres: synthesis and application toward colorimetric biosensing of H2O2 and glucose. Analyst 2015, 140, 6684–6691. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.S.; Kweon, S.H.; Cho, S.Y.; An, S.S.A.; Kim, M.I.; Doh, J.S.; Lee, J.W. Pt-decorated magnetic nanozymes for facile and sensitive point-of-care bioassay. ACS Appl. Mater. Interfaces 2017, 9, 35133–35140. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.Y.; Shin, H.Y.; Kim, M.I. Nanohybrids consisting of magnetic nanoparticles and gold nanoclusters as effective peroxidase mimics and their application for colorimetric detection of glucose. Biointerphases 2017, 12, 01A401. [Google Scholar] [CrossRef] [PubMed]
- Ju, P.; Xiang, Y.H.; Xiang, Z.B.; Wang, M.; Zhao, Y.; Zhang, D.; Yu, J.Q.; Han, X.X. BiOI hierarchical nanoflowers as novel robust peroxidase mimetics for colorimetric detection of H2O2. RSC Adv. 2016, 6, 17483–17493. [Google Scholar] [CrossRef]
- Ju, P.; Yu, Y.Z.; Wang, M.; Zhao, Y.; Zhang, D.; Sun, C.J.; Han, X.X. Synthesis of EDTA-assisted CeVO4 nanorods as robust peroxidase mimics towards colorimetric detection of H2O2. J. Mater. Chem. B 2016, 4, 6316–6325. [Google Scholar] [CrossRef]
- Yu, J.; Ma, D.Q.; Mei, L.Q.; Gao, Q.; Yin, W.Y.; Zhang, X.; Yan, L.; Gu, Z.J.; Ma, X.Y.; Zhao, Y.L. Peroxidase-like activity of MoS2 nanoflakes with different modifications and their application for H2O2 and glucose detection. J. Mater. Chem. B 2018, 6, 487–498. [Google Scholar] [CrossRef]
- Fan, H.; Wang, N.; Tian, Y.J.; Ai, S.Y.; Zhan, J.H. Acetic acid induced synthesis of laminated activated carbon nitride nanostructures. Carbon 2016, 107, 747–753. [Google Scholar] [CrossRef]
- Li, Y.; Jin, R.; Xing, Y.; Li, J.; Song, S.; Liu, X.; Li, M.; Jin, R. Macroscopic foam-like holey ultrathin g-C3N4 nanosheets for drastic improvement of visible-light photocatalytic activity. Adv. Energy Mater. 2016, 24, 1601273. [Google Scholar] [CrossRef]
- Wang, N.; Han, Z.W.; Fan, H.; Ai, S.Y. Copper nanoparticles modified graphitic carbon nitride nanosheets as a peroxidase mimetic for glucose detection. RSC Adv. 2015, 5, 91302–91307. [Google Scholar] [CrossRef]
- Qiao, F.M.; Qi, Q.Q.; Wang, Z.Z.; Xu, K.; Ai, S.Y. MnSe-loaded g-C3N4 nanocomposite with synergistic peroxidase-like catalysis: Synthesis and application toward colorimetric biosensing of H2O2 and glucose. Sensor. Actuator. B Chem. 2016, 229, 379–386. [Google Scholar] [CrossRef]
- Tian, J.Q.; Liu, Q.; Asiri, A.M.; Qusti, A.H.; Al-Youbi, A.O.; Sun, X.P. Ultrathin graphitic carbon nitride nanosheets: a novel peroxidase mimetic, Fe doping-mediated catalytic performance enhancement and application to rapid, highly sensitive optical detection of glucose. Nanoscale 2013, 5, 11604–11609. [Google Scholar] [CrossRef] [PubMed]
- Mu, J.S.; Li, J.; Zhao, X.; Yang, E.C.; Zhao, X.J. Cobalt-doped graphitic carbon nitride with enhanced peroxidase-like activity for wastewater treatment. RSC Adv. 2016, 6, 35568–35576. [Google Scholar] [CrossRef]
- Ouyang, H.; Tu, X.M.; Fu, Z.F.; Wang, W.W.; Fu, S.F.; Zhu, C.Z.; Du, D.; Lin, Y.H. Colorimetric and chemiluminescent dual-readout immunochromatographic assay for detection of pesticide residues utilizing g-C3N4/BiFeO3 nanocomposites. Biosens. Bioelectron. 2018, 106, 43–49. [Google Scholar] [CrossRef] [PubMed]
- Chhowalla, M.; Shin, H.S.; Eda, G.; Li, L.J.; Loh, K.P.; Zhang, H. The chemistry of two-dimensional layered transition metal dichalcogenide nanosheets. Nat. Chem. 2013, 5, 263–275. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.X.; Guo, J.H.; Kang, Y.J.; Ai, Y.; Li, C.M. Two dimensional atomically thin MoS2 nanosheets and their sensing applications. Nanoscale 2015, 7, 19358–19376. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.J.; Wu, Q.; Jiang, K.L.; Wang, C.X.; Zhang, C. One-step synthesis of water-soluble and highly fluorescent MoS2 quantum dots for detection of hydrogen peroxide and glucose. Sensor. Actuator. B Chem. 2017, 252, 183–190. [Google Scholar] [CrossRef]
- Wang, M.; Ju, P.; Zhao, Y.; Li, J.J.; Han, X.X.; Hao, Z.M. In situ ion exchange synthesis of MoS2/g-C3N4 heterojunction for highly efficient hydrogen production. New J. Chem. 2018, 42, 910–917. [Google Scholar] [CrossRef]
- Zhao, K.; Gu, W.; Zheng, S.S.; Zhang, C.L.; Xian, Y.Z. SDS-MoS2 nanoparticles as highly-efficient peroxidase mimetics for colorimetric detection of H2O2 and glucose. Talanta 2015, 141, 47–52. [Google Scholar] [CrossRef] [PubMed]
- Peng, J.; Weng, J. Enhanced peroxidase-like activity of MoS2/graphene oxide hybrid with light irradiation for glucose detection. Biosens. Bioelectron. 2017, 89, 652–658. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhou, Z.F.; Wen, F.F.; Tan, J.; Peng, T.; Luo, B.Q.; Wang, H.G.; Yin, S.X. A flower-like MoS2-decorated MgFe2O4 nanocomposite: Mimicking peroxidase and colorimetric detection of H2O2 and glucose. Sensor. Actuator. B Chem. 2018, 275, 155–162. [Google Scholar] [CrossRef]
- Vinita; Nirala, N.R.; Prakash, R. One step synthesis of AuNPs@MoS2-QDs composite as a robust peroxidase-mimetic for instant unaided eye detection of glucose in serum, saliva and tear. Sensor. Actuator. B Chem. 2018, 263, 109–119. [Google Scholar] [CrossRef]
- Gopalakrishnan, D.; Damien, D.; Shaijumon, M.M. MoS2 quantum dot-interspersed exfoliated MoS2 nanosheets. ACS Nano 2014, 8, 5297–5303. [Google Scholar] [CrossRef] [PubMed]
- Wei, H.; Wang, E.K. Fe3O4 magnetic nanoparticles as peroxidase mimetics and their applications in H2O2 and glucose detection. Anal. Chem. 2008, 80, 2250–2254. [Google Scholar] [CrossRef] [PubMed]
- Ye, L.; Chen, J.; Tian, L.; Liu, J.; Peng, T.; Deng, K.; Zan, L. BiOI thin film via chemical vapor transport: photocatalytic activity, durability, selectivity and mechanism. Appl. Catal. B Environ. 2013, 130–131, 1–7. [Google Scholar] [CrossRef]
- Zhang, Y.; Thomas, A.; Antonietti, M.; Wang, X. Activation of carbon nitride solids by protonation: Morphology changes, enhanced ionic conductivity, and photoconduction experiments. J. Am. Chem. Soc. 2009, 131, 50–51. [Google Scholar] [CrossRef] [PubMed]
- Ju, P.; Wang, Y.; Sun, Y.; Zhang, D. Controllable one-pot synthesis of a nest-like Bi2WO6/BiVO4 composite with enhanced photocatalytic antifouling performance under visible light irradiation. Dalton Trans. 2016, 45, 4588–4602. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.J.; Sun, B.; Wang, X.; Qiao, F.M.; Ai, S.Y. 2D ultrathin nanosheets of Co-Al layered double hydroxides prepared in L-asparagine solution enhanced peroxidase-like activity and colorimetric detection of glucose. J. Mater. Chem. B 2013, 1, 2268–2274. [Google Scholar] [CrossRef]
- Qiao, F.M.; Chen, L.J.; Li, X.; Li, L.; Ai, S.Y. Peroxidase-like activity of manganese selenide nanoparticles and its analytical application for visual detection of hydrogen peroxide and glucose. Sensor. Actuator. B Chem. 2014, 193, 255–262. [Google Scholar] [CrossRef]
- Porter, D.J.; Bright, H.J. The mechanism of oxidation of nitroalkanes by horseradish peroxidase. J. Biol. Chem. 1983, 258, 9913–9924. [Google Scholar] [PubMed]
- Li, L.; Ai, L.; Zhang, C.; Jiang, J. Hierarchical {001}-faceted BiOBr microspheres as a novel biomimetic catalyst dark catalysis towards colorimetric biosensing and pollutant degradation. Nanoscale 2014, 6, 4627–4634. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Ju, P.; Zhang, D.; Han, X.X.; Zheng, L.; Yin, X.F.; Sun, C.J. Colorimetric detection of H2O2 using flower-like Fe2(MoO4)3 microparticles as a peroxidase mimic. Microchim. Acta 2016, 183, 3025–3033. [Google Scholar] [CrossRef]
- Chen, Q.; Chen, J.; Gao, C.J.; Zhang, M.L.; Chen, J.Y.; Qiu, H.D. Hemin-functionalized WS2 nanosheets as highly active peroxidase mimetics for label-free colorimetric detection of H2O2 and glucose. Analyst 2015, 140, 2857–2863. [Google Scholar] [CrossRef] [PubMed]



© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ju, P.; He, Y.; Wang, M.; Han, X.; Jiang, F.; Sun, C.; Wu, C. Enhanced Peroxidase-Like Activity of MoS2 Quantum Dots Functionalized g-C3N4 Nanosheets towards Colorimetric Detection of H2O2. Nanomaterials 2018, 8, 976. https://doi.org/10.3390/nano8120976
Ju P, He Y, Wang M, Han X, Jiang F, Sun C, Wu C. Enhanced Peroxidase-Like Activity of MoS2 Quantum Dots Functionalized g-C3N4 Nanosheets towards Colorimetric Detection of H2O2. Nanomaterials. 2018; 8(12):976. https://doi.org/10.3390/nano8120976
Chicago/Turabian StyleJu, Peng, Yunhong He, Min Wang, Xiuxun Han, Fenghua Jiang, Chengjun Sun, and Chi Wu. 2018. "Enhanced Peroxidase-Like Activity of MoS2 Quantum Dots Functionalized g-C3N4 Nanosheets towards Colorimetric Detection of H2O2" Nanomaterials 8, no. 12: 976. https://doi.org/10.3390/nano8120976
APA StyleJu, P., He, Y., Wang, M., Han, X., Jiang, F., Sun, C., & Wu, C. (2018). Enhanced Peroxidase-Like Activity of MoS2 Quantum Dots Functionalized g-C3N4 Nanosheets towards Colorimetric Detection of H2O2. Nanomaterials, 8(12), 976. https://doi.org/10.3390/nano8120976






