Inhibition of TRPV1 Channel Activity in Human CD4+ T Cells by Nanodiamond and Nanoplatinum Liquid, DPV576
Abstract
1. Introduction
2. Methods
2.1. Blood Donors
2.2. DPV576 Liquid
2.3. CD4+ T Cell Purification
2.4. Effect of DPV576 on T Cells Activation
2.5. Stimulation with Anti-CD3 and Anti-CD28 Beads in the Presence and Absence of TRPV1 Inhibitor SB366791 and DPV576
2.6. Stimulation with Anti-CD3 and Anti-CD28 Beads in the Presence and Absence of Capsaicin and DPV576
2.7. Statistical Analysis
3. Results
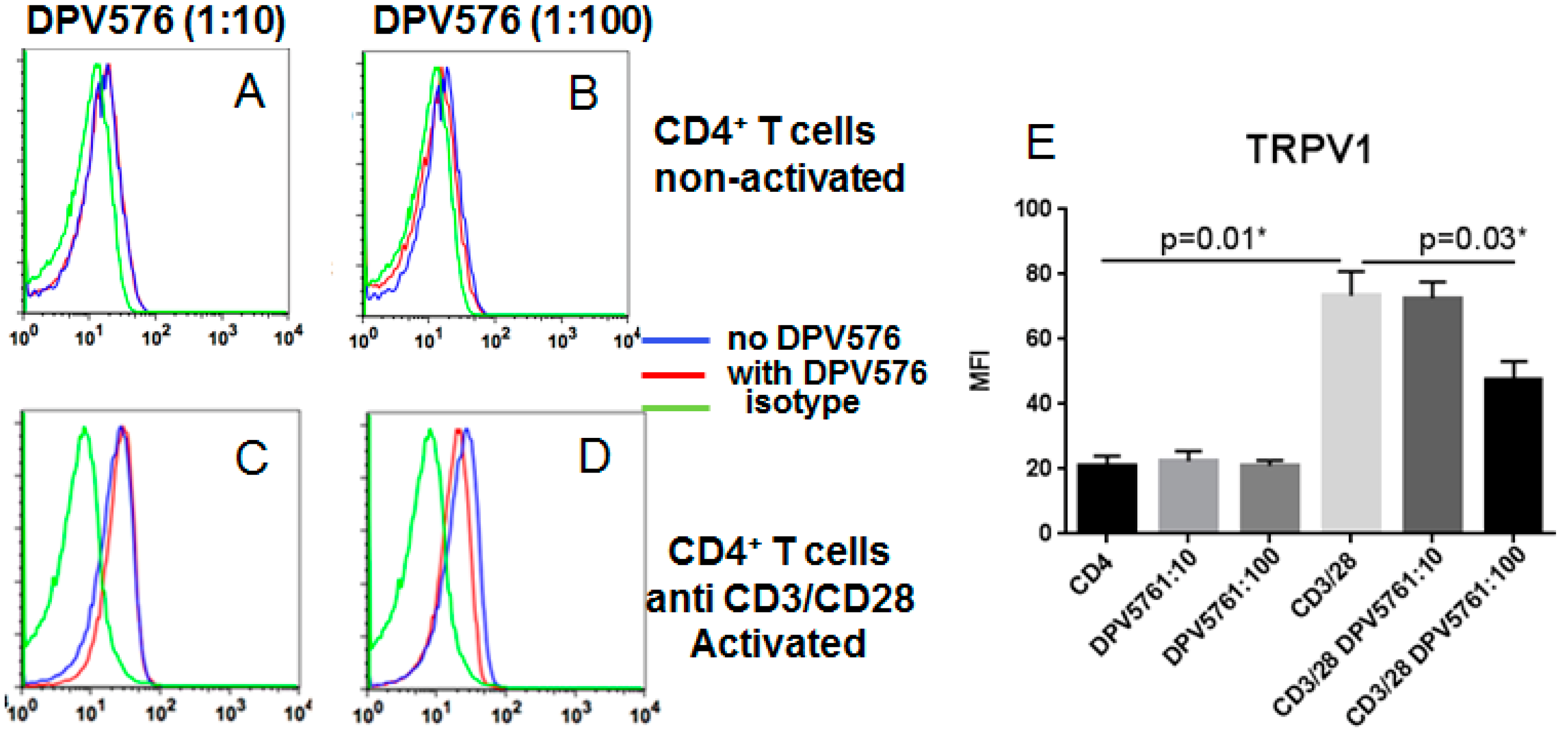
3.1. DPV576 Down Modulates the Expression of TRPV1 on Activated CD4+ T Cells
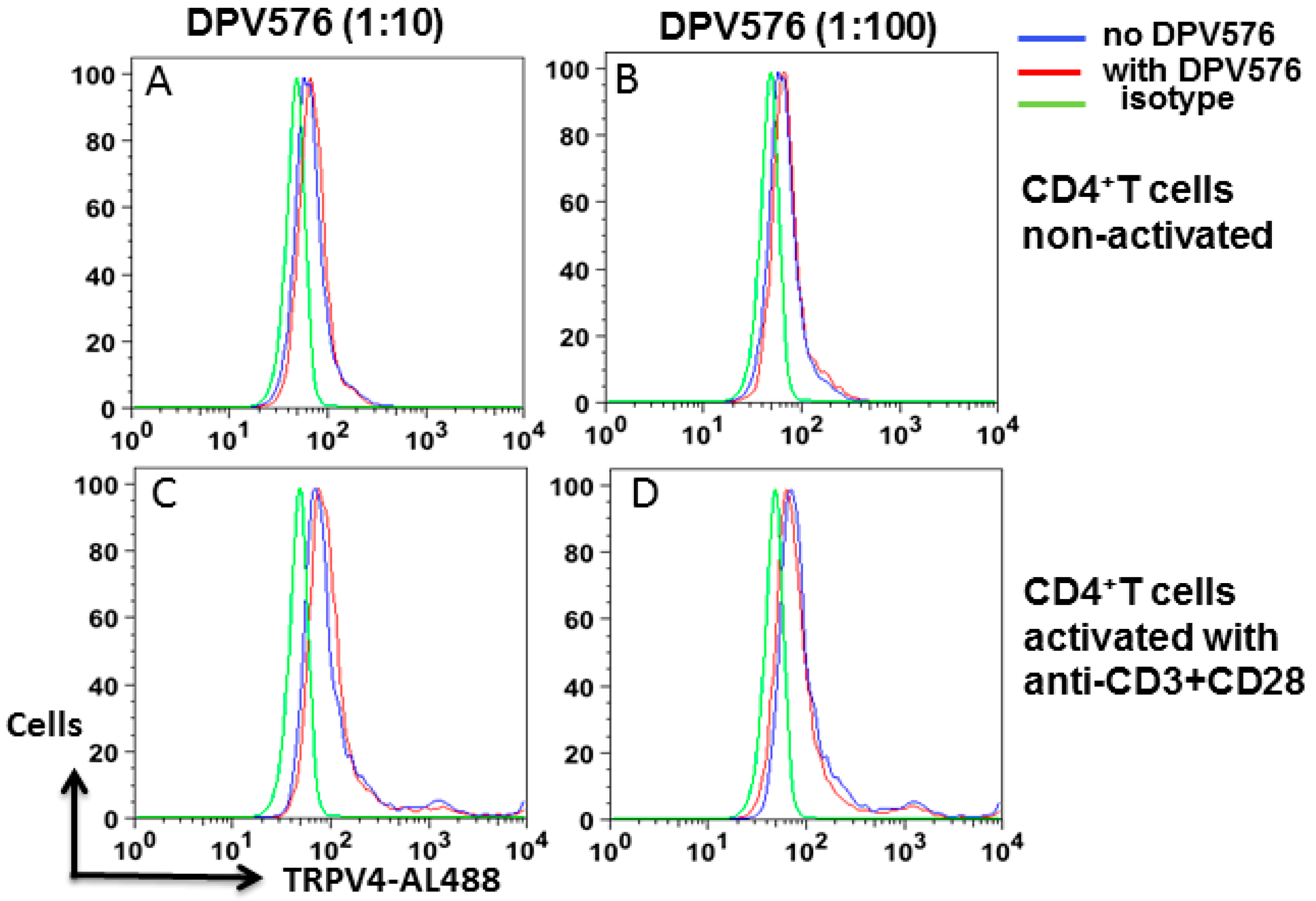
3.2. DPV576 Does Not Modulate the Expression of TRPV4 on CD4+ T Cells
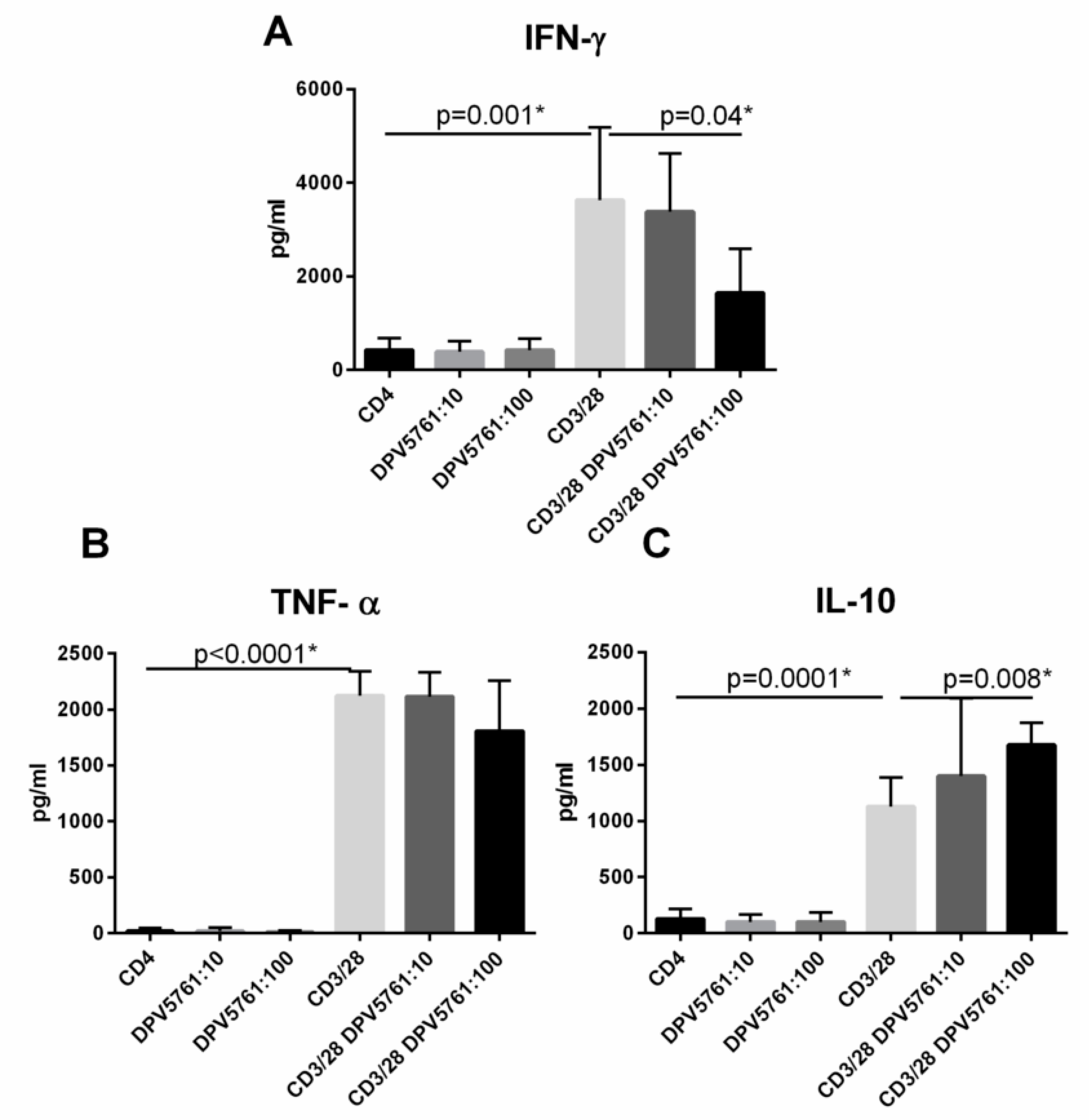
3.3. DPV576 Modulates Cytokine Secretion from Activated CD4+ T Cells
3.4. TRPV1 Antagonist, SB366791, Inhibits the Effect of DPV576 on Cytokine Secretion by CD4+ T Cells
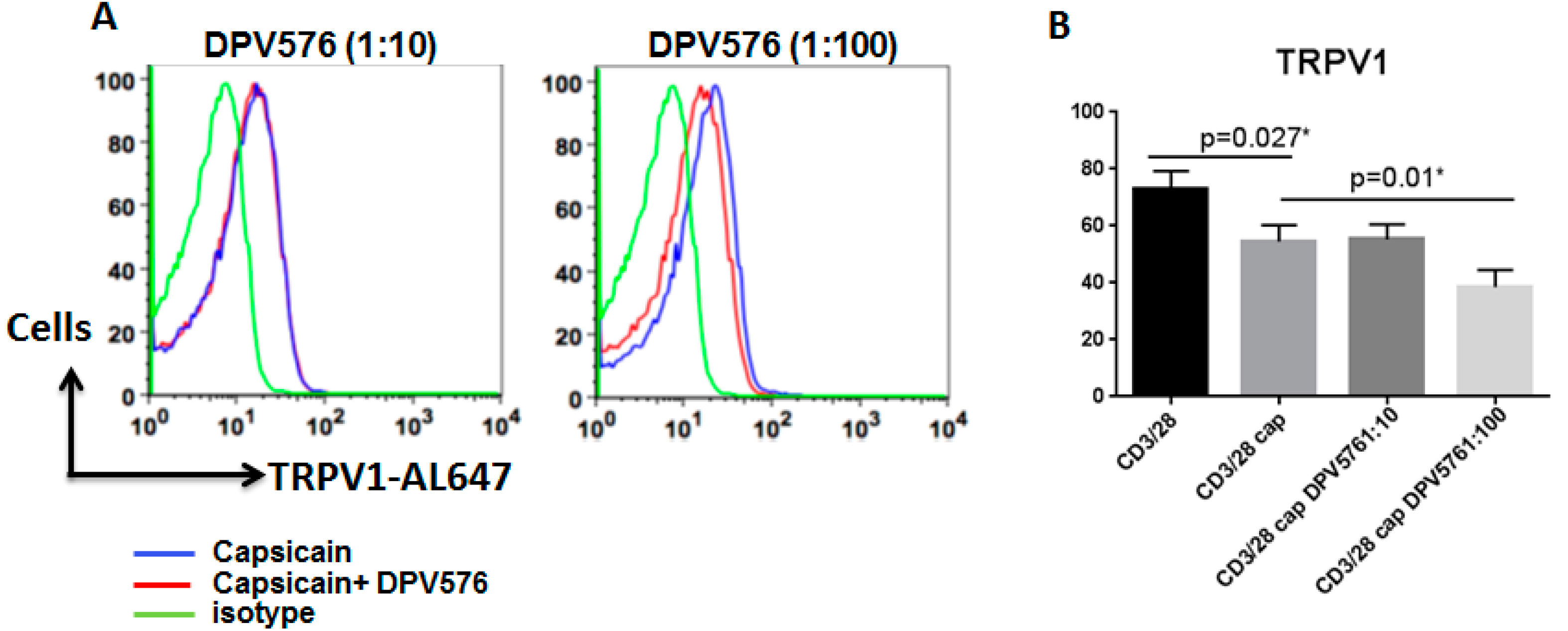
3.5. DPV576 Synergizes with Capsaicin to Downregulate the Expression of TRPV1 on CD4+ T Cells
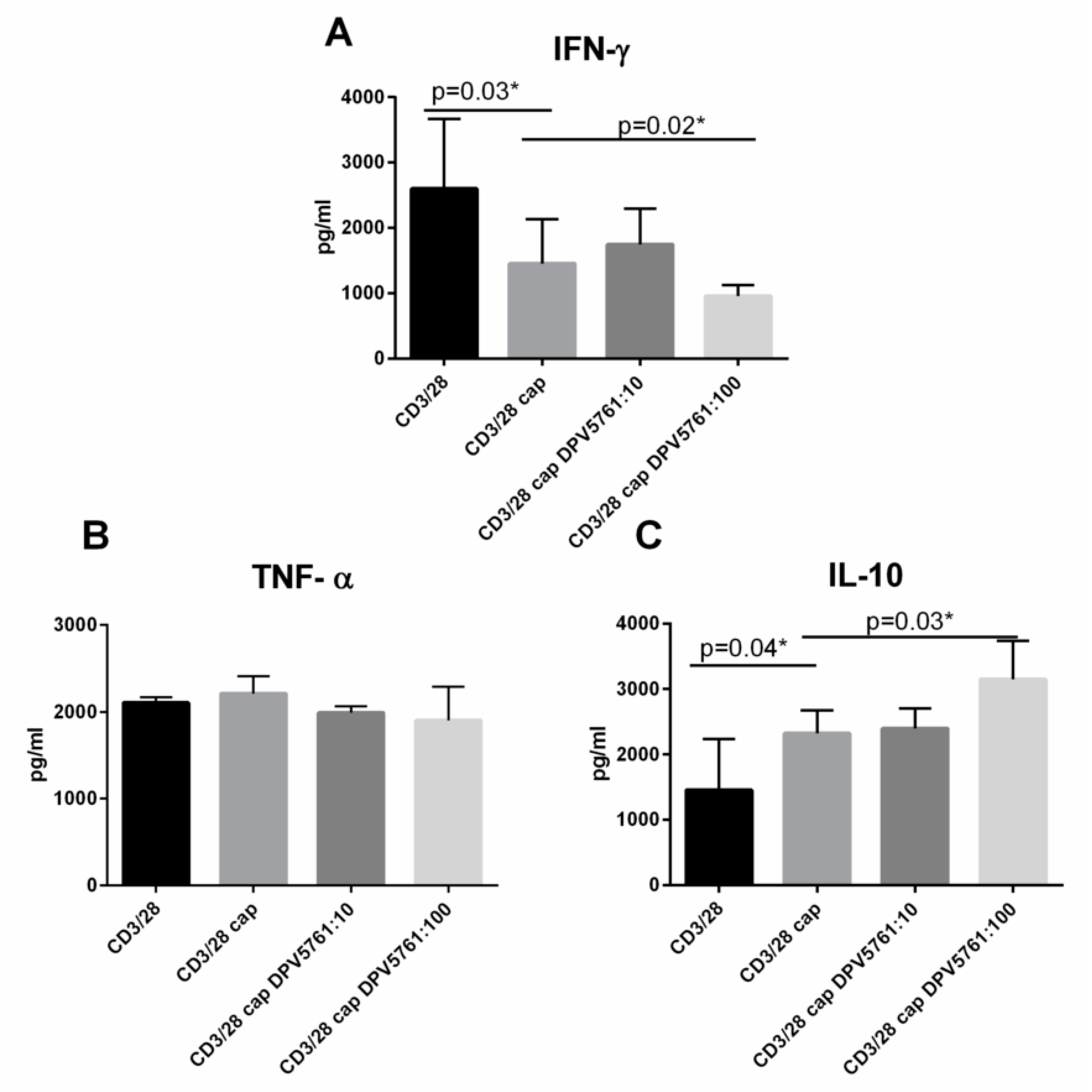
3.6. DPV576 Modulates Cytokine Secretion of TRPV1 Agonist, Capsaicin, in CD4+ T Cells
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Vezzani, A.; Viviani, B. Neuromodulatory properties of inflammatory cytokines and their impact on neuronal excitability. Neuropharmacol. 2015, 96, 70–82. [Google Scholar] [CrossRef] [PubMed]
- Venkatachalam, K.; Montell, C. TRP channels. Annu. Rev. Biochem. 2007, 76, 387–417. [Google Scholar] [CrossRef] [PubMed]
- Julius, D. TRP channels and pain. Annu. Rev. Cell Dev. Biol. 2013, 29, 355–384. [Google Scholar] [CrossRef] [PubMed]
- Gees, M.; Colsoul, B.; Nilius, B. The role of transient receptor potential cation channels in Ca2+ signaling. Cold Spring Harb. Perspect. Biol. 2010, 2, a003962. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, E.S.; Fernandes, M.A.; Keeble, J.E. The functions of TRPA1 and TRPV1: Moving away from sensory nerves. Br. J. Pharmacol. 2012, 166, 510–521. [Google Scholar] [CrossRef] [PubMed]
- Nilius, B.; Szallasi, A. Transient receptor potential channels as drug targets: From the science of basic research to the art of medicine. Pharmacol. Rev. 2014, 66, 676–814. [Google Scholar] [CrossRef] [PubMed]
- Bertin, S.; Aoki-Nonaka, Y.; de Jong, P.R.; Nohara, L.L.; Xu, H.; Stanwood, S.R.; Srikanth, S.; Lee, J.; To, K.; Abramson, L.; et al. The ion channel TRPV1 regulates the activation and proinflammatory properties of CD4+ T cells. Nat. Immunol. 2014, 15, 1055–1063. [Google Scholar] [CrossRef] [PubMed]
- Owsianik, G.; Talavera, K.; Voets, T.; Nilius, B. Permeation and selectivity of TRP channels. Annu. Rev. Physiol. 2006, 68, 685–717. [Google Scholar] [CrossRef] [PubMed]
- Van Goor, M.K.C.; Hoenderop, J.G.J.; van der Wijst, J. TRP channels in calcium homeostasis: From hormonal control to structure-function relationship of TRPV5 and TRPV6. Biochim. Biophys. Acta 2017, 1864, 883–893. [Google Scholar] [CrossRef] [PubMed]
- Vassilieva, I.O.; Tomilin, V.N.; Marakhova, I.I.; Shatrova, A.N.; Negulyaev, Y.A.; Semenova, S.B. Expression of transient receptor potential vanilloid channels TRPV5 and TRPV6 in human blood lymphocytes and Jurkat leukemia T cells. J. Membr. Biol. 2013, 246, 131–140. [Google Scholar] [CrossRef] [PubMed]
- Vasil’eva, I.O.; Neguliaev, I.; Marakhova, I.I.; Semenova, S.B. TRPV5 and TRPV6 calcium channels in human T cells. Tsitologiia 2008, 50, 953–957. [Google Scholar] [CrossRef] [PubMed]
- Bertin, S.; Raz, E. A novel role for the TRPV1 ion channel in CD4+ T cell activation. Med. Sci. 2015, 31, 135–136. [Google Scholar]
- Szallasi, A.; Cortright, D.N.; Blum, C.A.; Eid, S.R. The vanilloid receptor TRPV1: 10 years from channel cloning to antagonist proof-of-concept. Nat. Rev. Drug Discov. 2007, 6, 357–372. [Google Scholar] [CrossRef] [PubMed]
- Ghoneum, M.H.; Katano, H.; Agrawal, S.; Ganguly, S.; Agrawal, A. Effect of nanodiamond and nanoplatinum liquid, DPV576, on human primary keratinocytes. J. Biomed. Nanotechnol. 2017, 13, 110–116. [Google Scholar] [CrossRef] [PubMed]
- Ghoneum, M.; Ghoneum, A.; Tolentino, L.; Gimzewski, J. Modulation of aged murine T lymphocytes in vivo by DPV576-C, a nanodiamond- and nanoplatinum-coated material. In Vivo 2010, 24, 141–146. [Google Scholar] [PubMed]
- Samivel, R.; Kim, D.W.; Son, H.R.; Rhee, Y.H.; Kim, E.H.; Kim, J.H.; Bae, J.S.; Chung, Y.J.; Chung, P.S.; Raz, E.; et al. The role of TRPV1 in the CD4+ T cell-mediated inflammatory response of allergic rhinitis. Oncotarget 2016, 7, 148–160. [Google Scholar] [CrossRef] [PubMed]
- Wenning, A.S.; Neblung, K.; Strauss, B.; Wolfs, M.J.; Sappok, A.; Hoth, M.; Schwarz, E.C. TRP expression pattern and the functional importance of TRPC3 in primary human T-cells. Biochim. Biophys. Acta 2011, 1813, 412–423. [Google Scholar] [CrossRef] [PubMed]
- Saunders, C.I.M.; Kunde, D.A.; Crawford, A.; Geraghty, D.P. Expression of transient receptor potential vanilloid 1 (TRPV1) and 2 (TRPV2) in human peripheral blood. Mol. Immunol. 2007, 44, 1429–1435. [Google Scholar] [CrossRef] [PubMed]
- Spinsanti, G.; Zannolli, R.; Panti, C.; Ceccarelli, I.; Marsili, L.; Bachiocco, V.; Frati, F.; Aloisi, A.M. Quantitative Real-Time PCR detection of TRPV1-4 gene expression in human leukocytes from healthy and hyposensitive subjects. Mol. Pain 2008, 4, 51. [Google Scholar] [CrossRef] [PubMed]
- Bachiocco, V.; Bergamaschi, R.; Spinsanti, G.; Lima, M.; Romagnoli, R.; Sorda, G.; Aloisi, A.M. Lymphocyte TRPV 1–4 gene expression and MIF blood levels in a young girl clinically diagnosed with HSAN IV. Clin. J. Pain 2011, 27, 631–634. [Google Scholar] [CrossRef] [PubMed]
- Amantini, C.; Mosca, M.; Lucciarini, R.; Perfumi, M.; Morrone, S.; Piccoli, M.; Santoni, G. Distinct thymocyte subsets express the vanilloid receptor VR1 that mediates capsaicin-induced apoptotic cell death. Cell Death Differ. 2004, 11, 1342–1356. [Google Scholar] [CrossRef] [PubMed]
- Farfariello, V.; Amantini, C.; Santoni, G. Transient receptor potential vanilloid 1 activation induces autophagy in thymocytes through ROS-regulated AMPK and Atg4C pathways. J. Leukoc. Biol. 2012, 92, 421–423. [Google Scholar] [CrossRef] [PubMed]
- Nilius, B.; Owsianik, G.; Voets, T.; Peters, J.A. Transient receptor potential cation channels in disease. Physiol. Rev. 2007, 87, 165–217. [Google Scholar] [CrossRef] [PubMed]
- Majhi, R.K.; Sahoo, S.S.; Yadav, M.; Pratheek, B.M.; Chattopadhyay, S.; Goswami, C. Functional expression of TRPV channels in T cells and their implications in immune regulation. FEBS J. 2015, 282, 2661–2668. [Google Scholar] [CrossRef] [PubMed]
- Bertin, S.; Raz, E. Transient receptor potential (TRP) channels in T cells. Semin. Immunopathol. 2016, 38, 309–319. [Google Scholar] [CrossRef] [PubMed]
- Frias, B.; Merighi, A. Capsaicin, Nociception and Pain. Molecules 2016, 21, 797. [Google Scholar] [CrossRef] [PubMed]
- Moran, M.M.; Szallasi, A. Targeting nociceptive transient receptor potential channels to treat chronic pain: Current state of the field. Br. J. Pharmacol. 2017, 175, 2185–2203. [Google Scholar] [CrossRef] [PubMed]
- Austin, P.J.; Moalem-Taylor, G. The neuro-immune balance in neuropathic pain: Involvement of inflammatory immune cells, immune-like glial cells and cytokines. J. Neuroimmunol. 2010, 229, 26–50. [Google Scholar] [CrossRef] [PubMed]
- Tian, L.; Ma, L.; Kaarela, T.; Li, Z. Neuroimmune crosstalk in the central nervous system and its significance for neurological diseases. J. Neuroinflamm. 2012, 9, 155. [Google Scholar] [CrossRef] [PubMed]
- Perkins, N.M.; Tracey, D.J. Hyperalgesia due to nerve injury: Role of neutrophils. Neuroscience 2000, 101, 745–757. [Google Scholar] [CrossRef]
- Liu, H.H.; Xia, X.Y.; Wu, Y.M.; Pan, L.J.; Jin, B.F.; Shang, X.J.; Huang, Y.F. Detection of peripheral blood Th1/Th2 cell ratio in patients with chronic abacterial prostatitis/chronic pelvic pain syndrome. Zhonghua Nan Ke Xue 2006, 12, 330–332. [Google Scholar] [PubMed]
- Schinkel, C.; Gaertner, A.; Zaspel, J.; Zedler, S.; Faist, E.; Schuermann, M. Inflammatory mediators are altered in the acute phase of posttraumatic complex regional pain syndrome. Clin. J. Pain 2006, 22, 235–239. [Google Scholar] [CrossRef] [PubMed]
- Kleinschnitz, C.; Hofstetter, H.H.; Meuth, S.G.; Braeuninger, S.; Sommer, C.; Stoll, G. T cell infiltration after chronic constriction injury of mouse sciatic nerve is associated with interleukin-17 expression. Exp. Neurol. 2006, 200, 480–485. [Google Scholar] [CrossRef] [PubMed]
- Austin, P.J.; Kim, C.F.; Perera, C.J.; Moalem-Taylor, G. Regulatory T cells attenuate neuropathic pain following peripheral nerve injury and experimental autoimmune neuritis. Pain 2012, 153, 1916–1931. [Google Scholar] [CrossRef] [PubMed]
- Caterina, M.J.; Schumacher, M.A.; Tominaga, M.; Rosen, T.A.; Levine, J.D.; Julius, D. The capsaicin receptor: A heat-activated ion channel in the pain pathway. Nature 1997, 389, 816–824. [Google Scholar] [PubMed]
- Phelps, C.B.; Wang, R.R.; Choo, S.S.; Gaudet, R. Differential Regulation of TRPV1, TRPV3 and TRPV4 Sensitivity through a Conserved Binding Site on the Ankyrin Repeat Domain. J. Biol. Chem. 2010, 285, 731–740. [Google Scholar] [CrossRef] [PubMed]
- Ota, H.; Katanosaka, K.; Murase, S.; Kashio, M.; Tominaga, M.; Mizumura, K. TRPV1 and TRPV4 Play Pivotal Roles in Delayed Onset Muscle Soreness. PLoS ONE 2013, 8, e65751. [Google Scholar] [CrossRef] [PubMed]
- Tóth, A.; Czikora, Á.; Pásztor, E.T.; Dienes, B.; Bai, P.; Csernoch, L.; Rutki, I.; Csato, V.; Manyine, I.S.; Porszasz, R.; et al. Vanilloid Receptor-1 (TRPV1) Expression and Function in the Vasculature of the Rat. J. Histochem. Cytochem. 2014, 62, 129–144. [Google Scholar] [CrossRef] [PubMed]
- Raison, C.L.; Capuron, L.; Miller, A.H. Cytokines sing the blues: Inflammation and the pathogenesis of depression. Trends Immunol. 2006, 27, 24–31. [Google Scholar] [CrossRef] [PubMed]
- Dantzer, R.; O’Connor, J.C.; Freund, G.G.; Johnson, R.W.; Kelley, K.W. From inflammation to sickness and depression: When the immune system subjugates the brain. Nat. Rev. Neurosci. 2008, 9, 46–57. [Google Scholar] [CrossRef] [PubMed]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ghoneum, M.H.; Gimzewski, J.K.; Ghoneum, A.; Katano, H.; Paw U, C.N.; Agrawal, A. Inhibition of TRPV1 Channel Activity in Human CD4+ T Cells by Nanodiamond and Nanoplatinum Liquid, DPV576. Nanomaterials 2018, 8, 770. https://doi.org/10.3390/nano8100770
Ghoneum MH, Gimzewski JK, Ghoneum A, Katano H, Paw U CN, Agrawal A. Inhibition of TRPV1 Channel Activity in Human CD4+ T Cells by Nanodiamond and Nanoplatinum Liquid, DPV576. Nanomaterials. 2018; 8(10):770. https://doi.org/10.3390/nano8100770
Chicago/Turabian StyleGhoneum, Mamdooh H., James K. Gimzewski, Aya Ghoneum, Hideki Katano, Clarissa Nila Paw U, and Anshu Agrawal. 2018. "Inhibition of TRPV1 Channel Activity in Human CD4+ T Cells by Nanodiamond and Nanoplatinum Liquid, DPV576" Nanomaterials 8, no. 10: 770. https://doi.org/10.3390/nano8100770
APA StyleGhoneum, M. H., Gimzewski, J. K., Ghoneum, A., Katano, H., Paw U, C. N., & Agrawal, A. (2018). Inhibition of TRPV1 Channel Activity in Human CD4+ T Cells by Nanodiamond and Nanoplatinum Liquid, DPV576. Nanomaterials, 8(10), 770. https://doi.org/10.3390/nano8100770





