Enhanced UV-Visible Light Photocatalytic Activity by Constructing Appropriate Heterostructures between Mesopore TiO2 Nanospheres and Sn3O4 Nanoparticles
Abstract
:1. Introduction
2. Experimental Section
2.1. Chemicals
2.2. Synthesis of Samples
2.3. Characterization of Samples
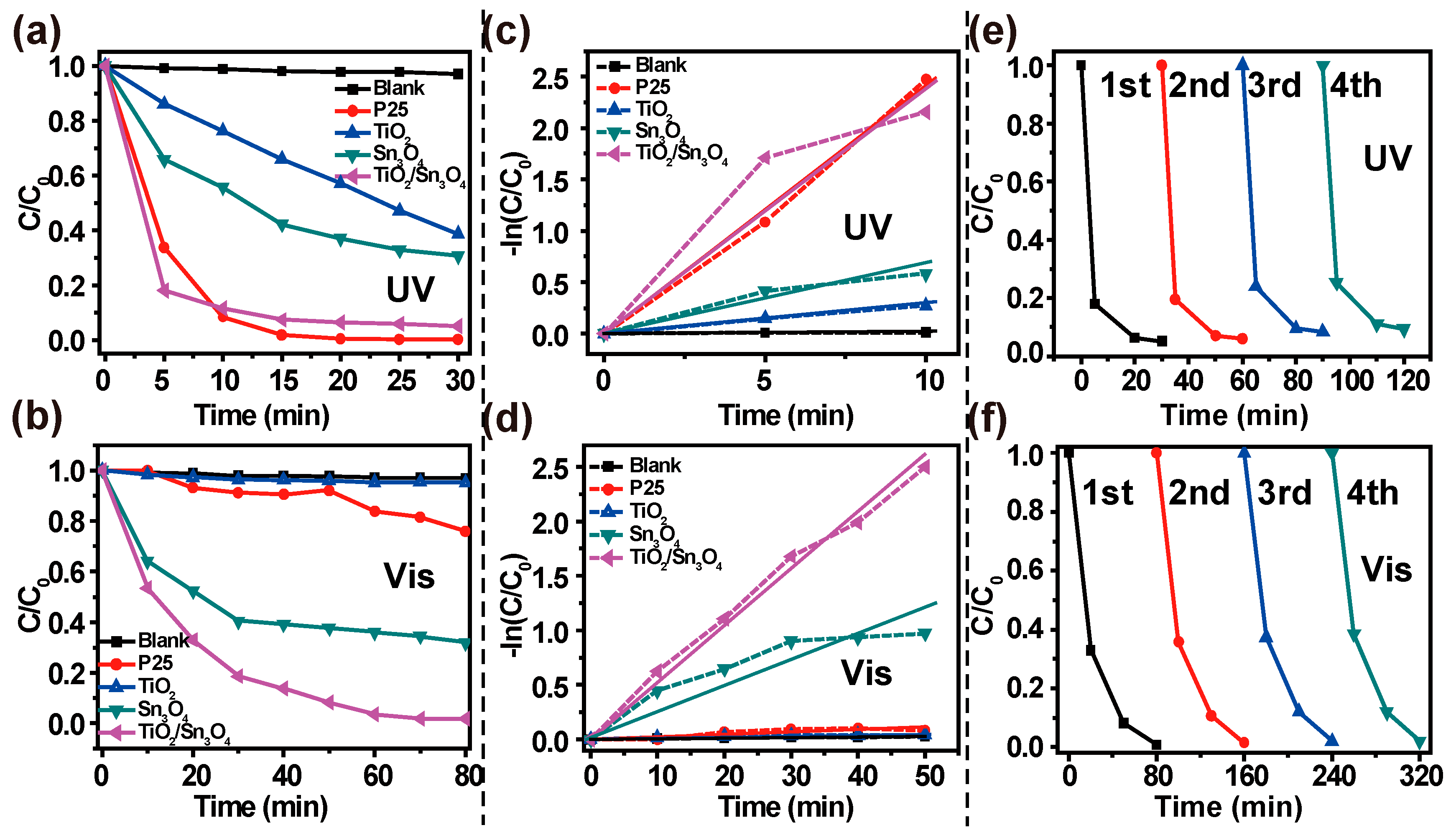
2.4. Photocatalytic Experiments
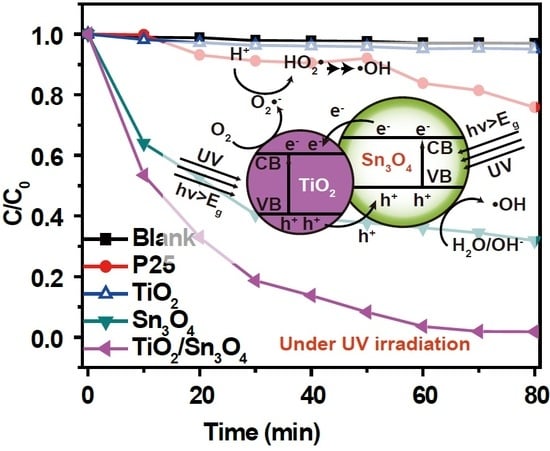
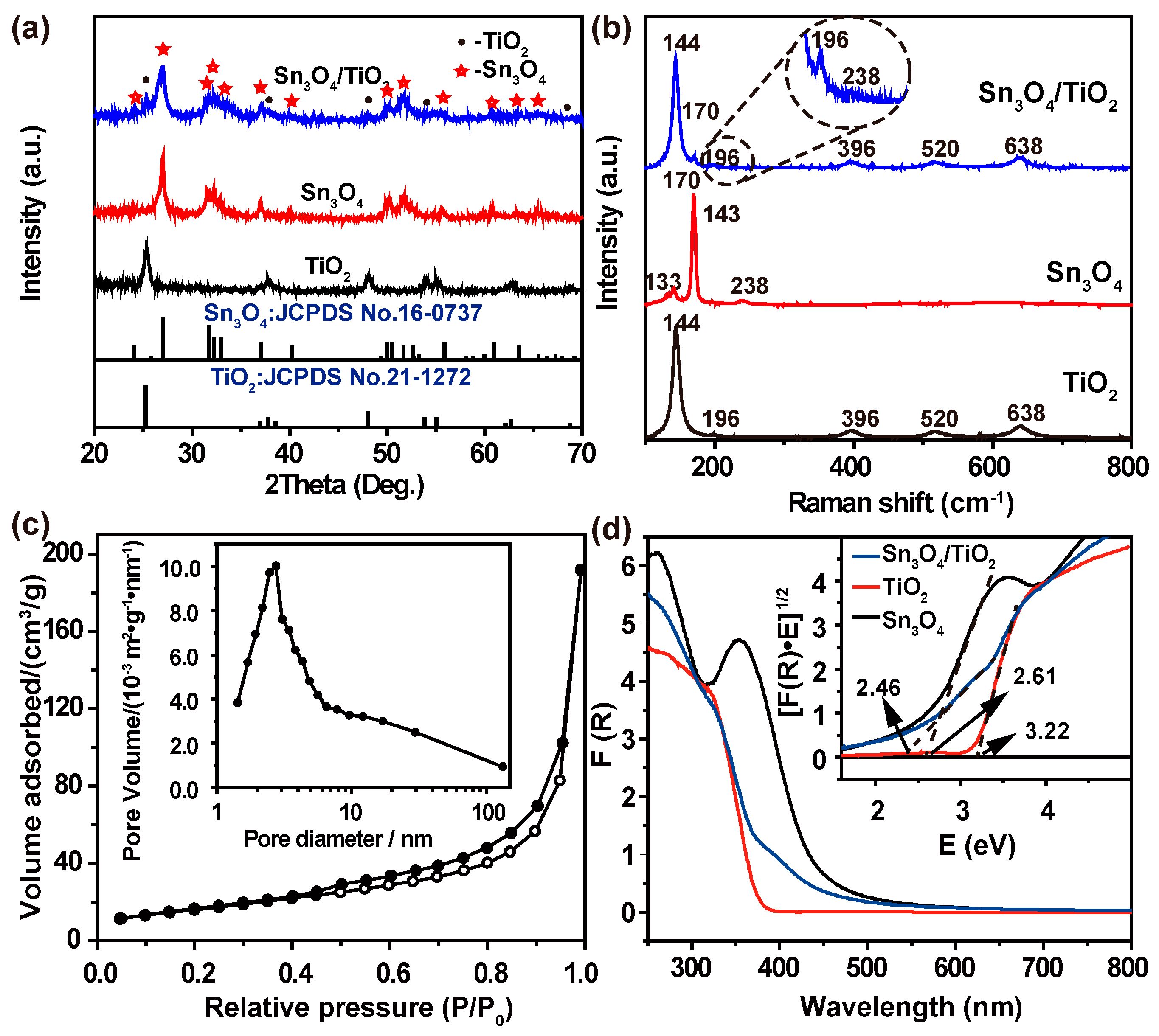
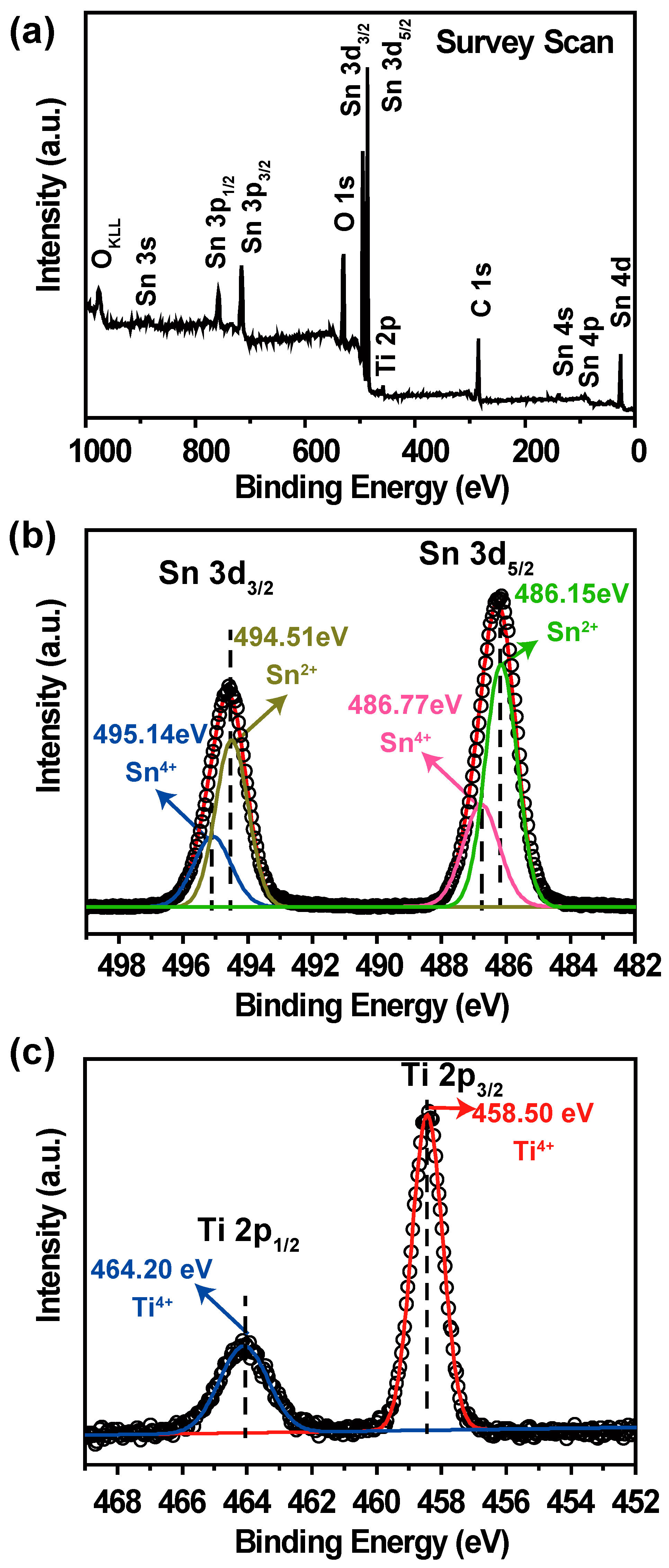
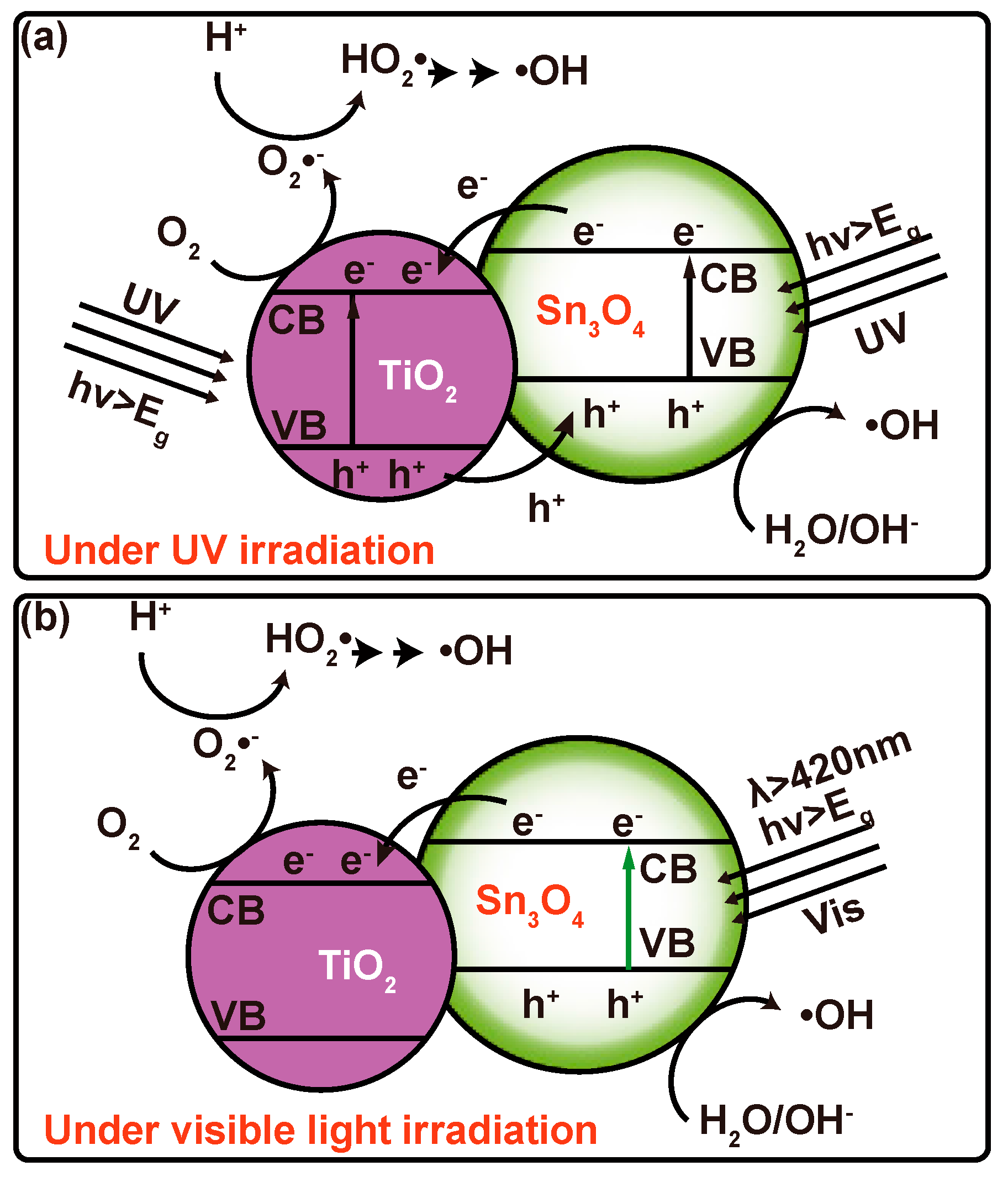
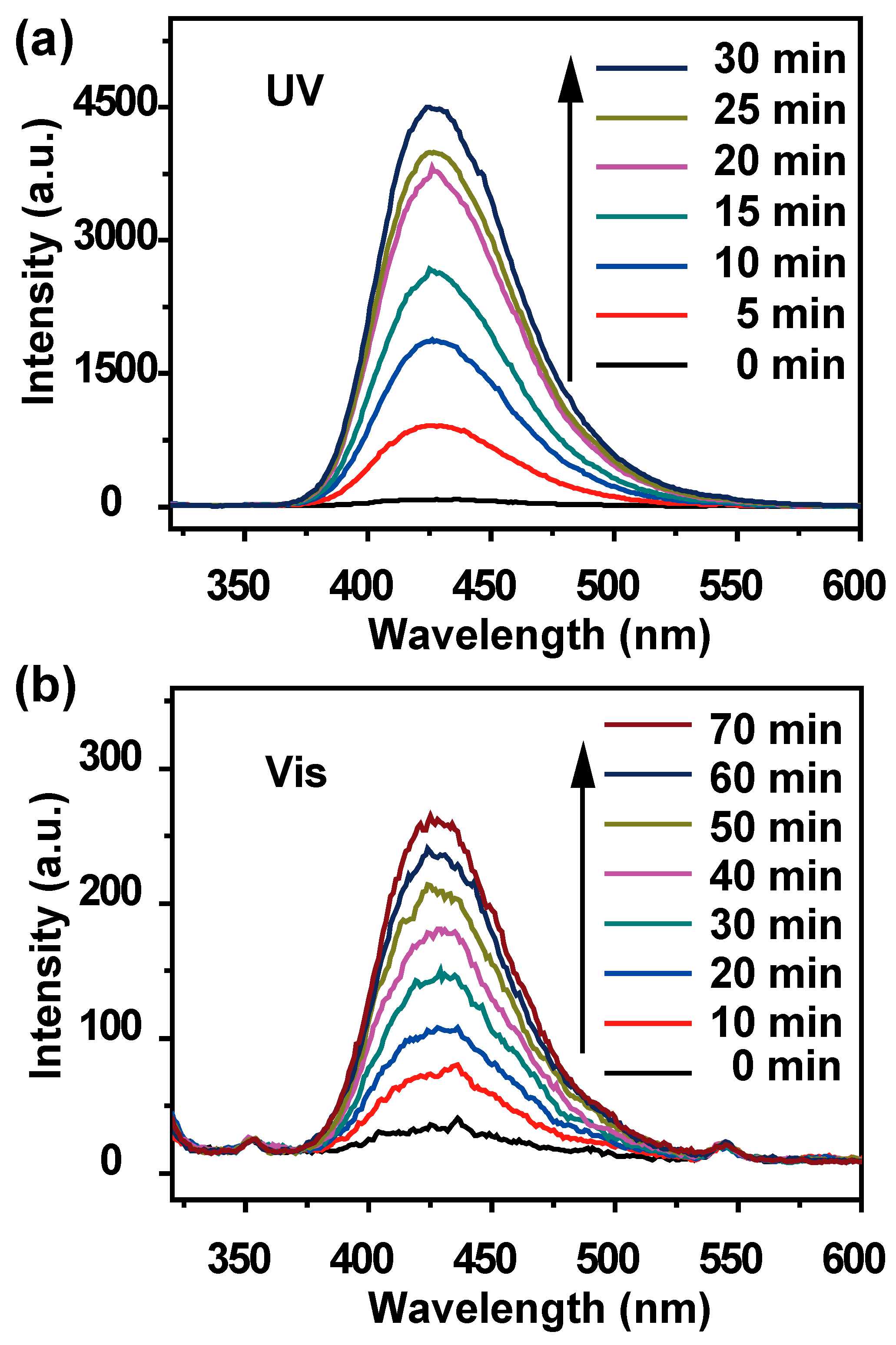
3. Results and Discussion
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Chen, X.; Liu, L.; Peter, Y.Y.; Mao, S.S. Increasing solar absorption for photocatalysis with black hydrogenated titanium dioxide nanocrystals. Science 2011, 331, 746–750. [Google Scholar] [CrossRef] [PubMed]
- Sun, Z.; Liao, T.; Sheng, L.; Kou, L.; Kim, J.H.; Dou, S.X. Deliberate design of TiO2 nanostructures towards superior photovoltaic cells. Chem.-A Eur. J. 2016, 22, 11357–11364. [Google Scholar] [CrossRef] [PubMed]
- Sun, Z.; Liao, T.; Kou, L. Strategies for designing metal oxide nanostructures. Sci. China Mater. 2017, 60, 1–24. [Google Scholar] [CrossRef]
- Asahi, R.; Morikawa, T.; Ohwaki, T.; Aoki, K.; Taga, Y. Visible-light photocatalysis in nitrogen-doped titanium oxides. Science 2001, 293, 269–271. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; Tian, J.; Sang, Y.; Cabot, A.; Liu, H. Structure, synthesis, and applications of TiO2 nanobelts. Adv. Mater. 2015, 27, 2557–2582. [Google Scholar] [CrossRef] [PubMed]
- Manikandan, M.; Tanabe, T.; Li, P.; Ueda, S.; Ramesh, G.V.; Kodiyath, R.; Wang, J.; Hara, T.; Dakshanamoorthy, A.; Ishihara, S. Photocatalytic water splitting under visible light by mixed-valence Sn3O4. ACS Appl. Mater. Interfaces 2014, 6, 3790–3793. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Li, D.; Chen, J.; Shao, Y.; Xian, J.; Zheng, X.; Wang, P. Sn3O4: A novel heterovalent-tin photocatalyst with hierarchical 3D nanostructures under visible light. RSC Adv. 2014, 4, 1266–1269. [Google Scholar] [CrossRef]
- Berengue, O.; Simon, R.; Chiquito, A.; Dalmaschio, C.; Leite, E.; Guerreiro, H.; Guimarães, F.E.G. Semiconducting Sn3O4 nanobelts: Growth and electronic structure. J. Appl. Phys. 2010, 107, 033717. [Google Scholar] [CrossRef]
- Chen, G.; Ji, S.; Sang, Y.; Chang, S.; Wang, Y.; Hao, P.; Claverie, J.; Liu, H.; Yu, G. Synthesis of scaly Sn3O4/TiO2 nanobelt heterostructures for enhanced UV-visible light photocatalytic activity. Nanoscale 2015, 7, 3117–3125. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wang, Q.; Zhan, X.; Wang, F.; Safdar, M.; He, J. Visible light driven type II heterostructures and their enhanced photocatalysis properties: A review. Nanoscale 2013, 5, 8326–8339. [Google Scholar] [CrossRef] [PubMed]
- Ye, W.; Shao, Y.; Hu, X.; Liu, C.; Sun, C. Highly Enhanced Photoreductive Degradation of Polybromodiphenyl Ethers with g-C3N4/TiO2 under Visible Light Irradiation. Nanomaterials 2017, 7, 76. [Google Scholar] [CrossRef] [PubMed]
- Low, J.; Yu, J.; Jaroniec, M.; Wageh, S.; Al-Ghamdi, A.A. Heterojunction photocatalysts. Adv. Mater. 2017, 43, 5234–5244. [Google Scholar] [CrossRef] [PubMed]
- Linsebigler, A.L.; Lu, G.; Yates, J.T., Jr. Photocatalysis on TiO2 surfaces: Principles, mechanisms, and selected results. Chem. Rev. 1995, 95, 735–758. [Google Scholar] [CrossRef]
- Liu, Q.; Sun, Z.; Dou, Y.; Kim, J.H.; Dou, S.X. Two-step self-assembly of hierarchically-ordered nanostructures. J. Mater. Chem. A 2015, 3, 11688–11699. [Google Scholar] [CrossRef]
- Sotelo-Vazquez, C.; Quesada-Cabrera, R.; Ling, M.; Scanlon, D.O.; Kafizas, A.; Thakur, P.K.; Lee, T.L.; Taylor, A.; Watson, G.W.; Palgrave, R.G. Evidence and Effect of Photogenerated Charge Transfer for Enhanced Photocatalysis in WO3/TiO2 Heterojunction Films: A Computational and Experimental Study. Adv. Funct. Mater. 2017, 27. [Google Scholar] [CrossRef]
- Nanakkal, A.; Alexander, L. Photocatalytic activity of graphene/ZnO nanocomposite fabricated by two-step electrochemical route. J. Chem. Sci. 2017, 129, 95–102. [Google Scholar] [CrossRef]
- Yu, X.; Wang, L.; Zhang, J.; Guo, W.; Zhao, Z.; Qin, Y.; Mou, X.; Li, A.; Liu, H. Hierarchical hybrid nanostructures of Sn3O4 on N doped TiO2 nanotubes with enhanced photocatalytic performance. J. Mater. Chem. A 2015, 3, 19129–19136. [Google Scholar] [CrossRef]
- Nanakkal, A.; Alexander, L. Graphene/BiVO4/TiO2 nanocomposite: Tuning band gap energies for superior photocatalytic activity under visible light. J. Mater. Sci. 2017, 52, 7997–8006. [Google Scholar] [CrossRef]
- Jiang, X.; Herricks, T.; Xia, Y. Monodispersed spherical colloids of titania: Synthesis, characterization, and crystallization. Adv. Mater. 2003, 15, 1205–1209. [Google Scholar] [CrossRef]
- Martínez, D.S.; Martínez-De La Cruz, A.; Cuéllar, E.L. Photocatalytic properties of WO3 nanoparticles obtained by precipitation in presence of urea as complexing agent. Appl. Catal. A Gen. 2011, 398, 179–186. [Google Scholar] [CrossRef]
- Cullity, B. Elements of X-Ray Diffractions; Addison-Wesley: Reading, MA, USA, 1978; p. 102. [Google Scholar]
- Sang, Y.; Yu, D.; Avdeev, M.; Qin, H.; Wang, J.; Liu, H.; Lv, Y. Yttrium aluminum garnet Nanoparticles with low antisite Defects studied with neutron and X-ray diffraction. J. Solid State Chem. 2012, 192, 366–370. [Google Scholar] [CrossRef]
- Khan, M.M.; Ansari, S.A.; Pradhan, D.; Ansari, M.O.; Lee, J.; Cho, M.H. Band gap engineered TiO2 nanoparticles for visible light induced photoelectrochemical and photocatalytic studies. J. Mater. Chem. A 2014, 2, 637–644. [Google Scholar] [CrossRef]
- Wang, F.; Zhou, X.; Zhou, J.; Sham, T.-K.; Ding, Z. Observation of single tin dioxide nanoribbons by confocal Raman microspectroscopy. J. Phys. Chem. C 2007, 111, 18839–18843. [Google Scholar] [CrossRef]
- Pawar, R.; Lee, C.S. Heterogeneous Nanocomposite-Photocatalysis for Water Purification; William Andrew: New York, NY, USA, 2015; pp. 68–76. [Google Scholar]
- Wang, J.; Lu, C.; Liu, X.; Wang, Y.; Zhu, Z.; Meng, D. Synthesis of tin oxide (SnO & SnO2) micro/nanostructures with novel distribution characteristic and superior photocatalytic performance. Mater. Des. 2017, 115, 103–111. [Google Scholar]
- Wagner, C.D. Handbook of X-Ray Photoelectron Spectroscopy; A Reference Book of Standard Data for Use in X-ray Photoelectron Spectroscopy; Physical Electronics Division, Perkin-Elmer Corporation: Eden Prairie, MN, USA, 1979; pp. 68–69. [Google Scholar]
- McDaniel, H.; Heil, P.E.; Tsai, C.-L.; Kim, K.; Shim, M. Integration of type II nanorod heterostructures into photovoltaics. ACS Nano 2011, 5, 7677–7683. [Google Scholar] [CrossRef] [PubMed]
- Dong, W.; Pan, F.; Xu, L.; Zheng, M.; Sow, C.H.; Wu, K.; Xu, G. Q.; Chen, W. Facile synthesis of CdS@ TiO2 core-shell nanorods with controllable shell thickness and enhanced photocatalytic activity under visible light irradiation. Appl. Surf. Sci. 2015, 349, 279–286. [Google Scholar] [CrossRef]
- Malik, V.; Pokhriyal, M.; Uma, S. Single step hydrothermal synthesis of beyerite, CaBi2O2(CO3)2 for the fabrication of UV-visible light photocatalyst BiOI/CaBi2O2(CO3)2. RSC Adv. 2016, 6, 38252–38262. [Google Scholar] [CrossRef]
- Cao, J.; Xu, B.; Luo, B.; Lin, H.; Chen, S. Novel BiOI/BiOBr heterojunction photocatalysts with enhanced visible light photocatalytic properties. Catal. Commun. 2011, 13, 63–68. [Google Scholar] [CrossRef]

| Photocatalyst | TiO2 | P25 | Sn3O4 | TiO2/Sn3O4 |
|---|---|---|---|---|
| κ (min−1) | κ (min−1) | κ (min−1) | κ (min−1) | |
| UV irradiation | 0.028 | 0.24 | 0.064 | 0.24 |
| Visible light | 0.0010 | 0.0023 | 0.024 | 0.052 |
| Surface Area (m2∙g−1) | 0.04 | 50 | 35.2 | 68.1 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hu, J.; Tu, J.; Li, X.; Wang, Z.; Li, Y.; Li, Q.; Wang, F. Enhanced UV-Visible Light Photocatalytic Activity by Constructing Appropriate Heterostructures between Mesopore TiO2 Nanospheres and Sn3O4 Nanoparticles. Nanomaterials 2017, 7, 336. https://doi.org/10.3390/nano7100336
Hu J, Tu J, Li X, Wang Z, Li Y, Li Q, Wang F. Enhanced UV-Visible Light Photocatalytic Activity by Constructing Appropriate Heterostructures between Mesopore TiO2 Nanospheres and Sn3O4 Nanoparticles. Nanomaterials. 2017; 7(10):336. https://doi.org/10.3390/nano7100336
Chicago/Turabian StyleHu, Jianling, Jianhai Tu, Xingyang Li, Ziya Wang, Yan Li, Quanshui Li, and Fengping Wang. 2017. "Enhanced UV-Visible Light Photocatalytic Activity by Constructing Appropriate Heterostructures between Mesopore TiO2 Nanospheres and Sn3O4 Nanoparticles" Nanomaterials 7, no. 10: 336. https://doi.org/10.3390/nano7100336
APA StyleHu, J., Tu, J., Li, X., Wang, Z., Li, Y., Li, Q., & Wang, F. (2017). Enhanced UV-Visible Light Photocatalytic Activity by Constructing Appropriate Heterostructures between Mesopore TiO2 Nanospheres and Sn3O4 Nanoparticles. Nanomaterials, 7(10), 336. https://doi.org/10.3390/nano7100336