High Refractive Organic–Inorganic Hybrid Films Prepared by Low Water Sol-Gel and UV-Irradiation Processes
Abstract
:1. Introduction
2. Results
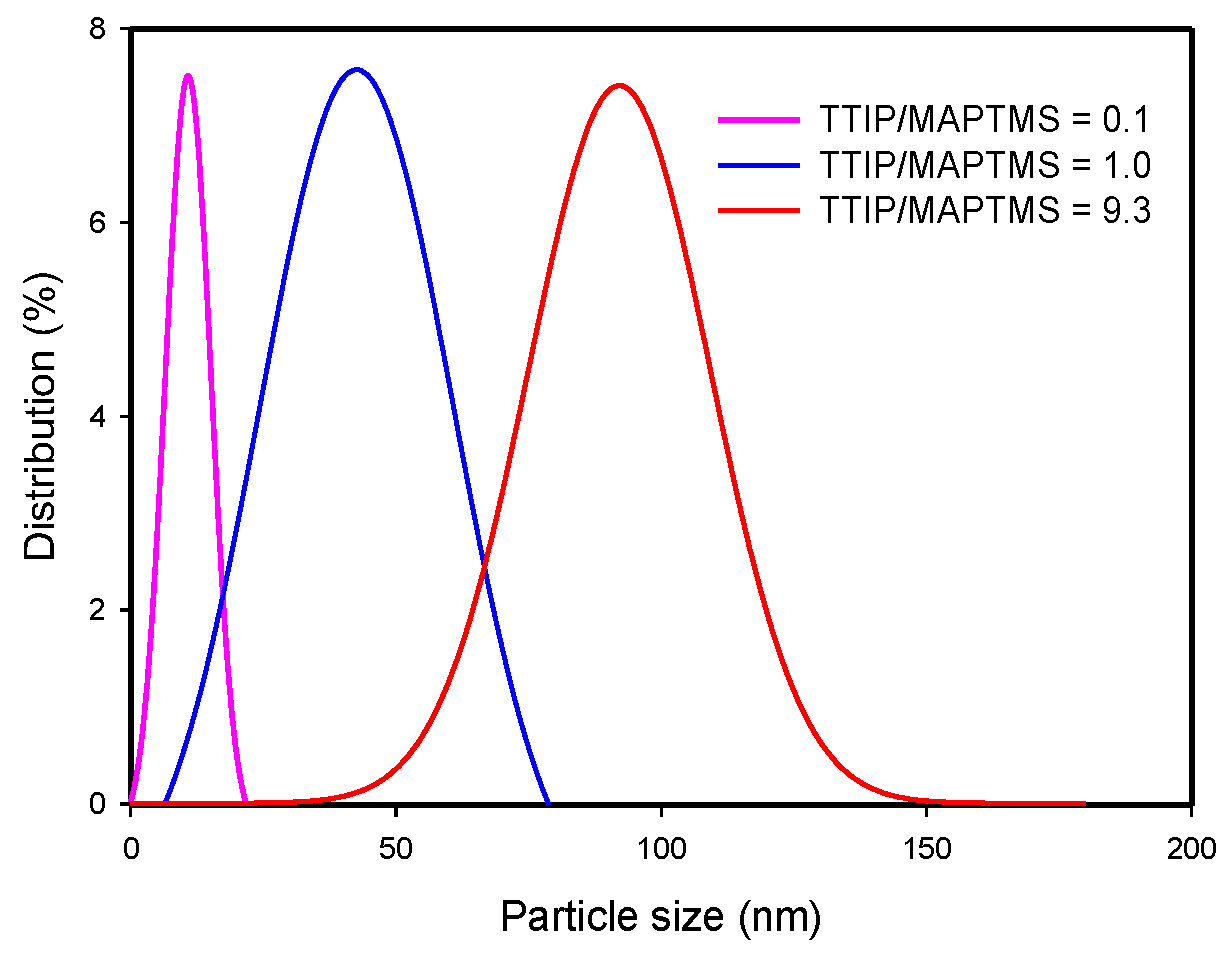
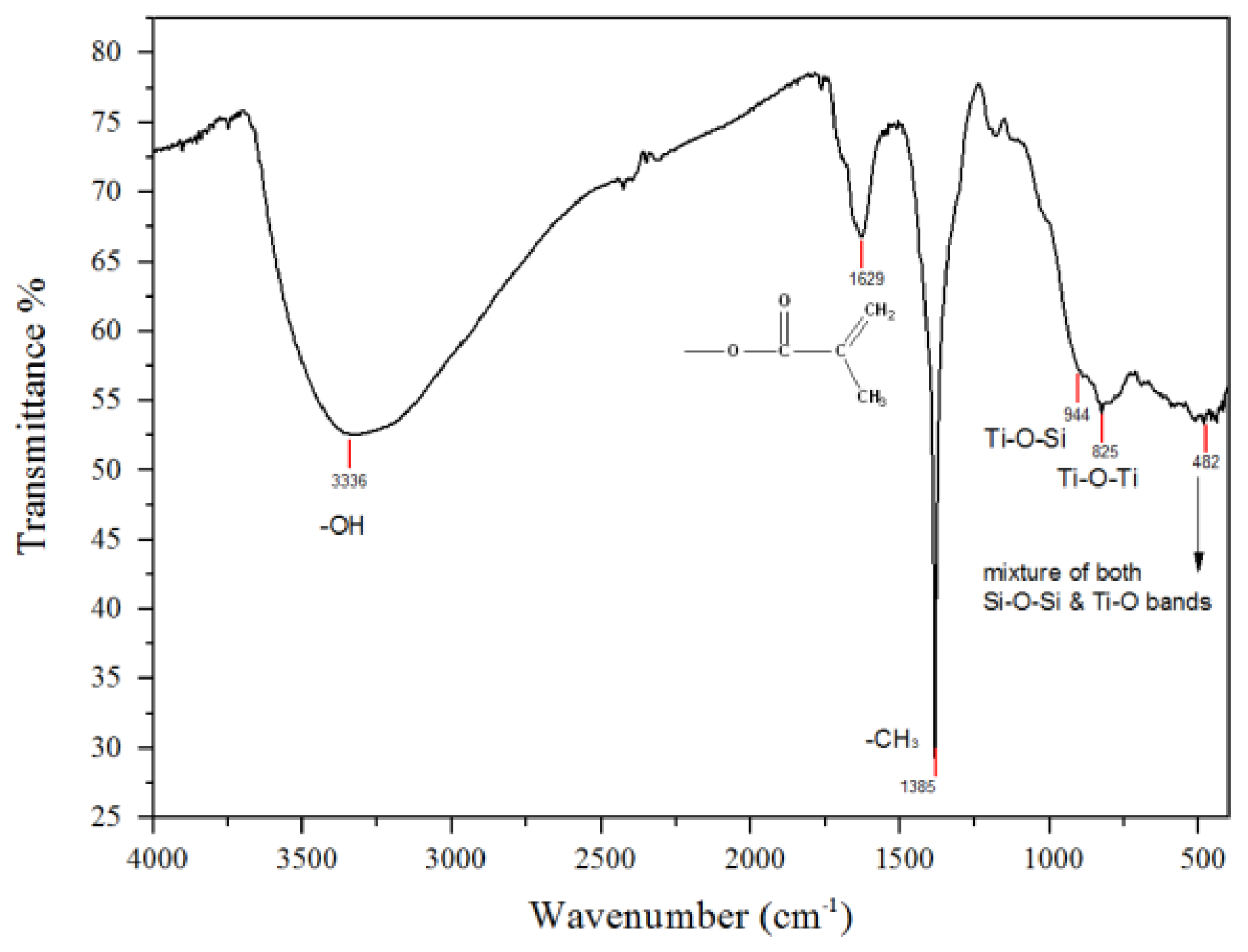
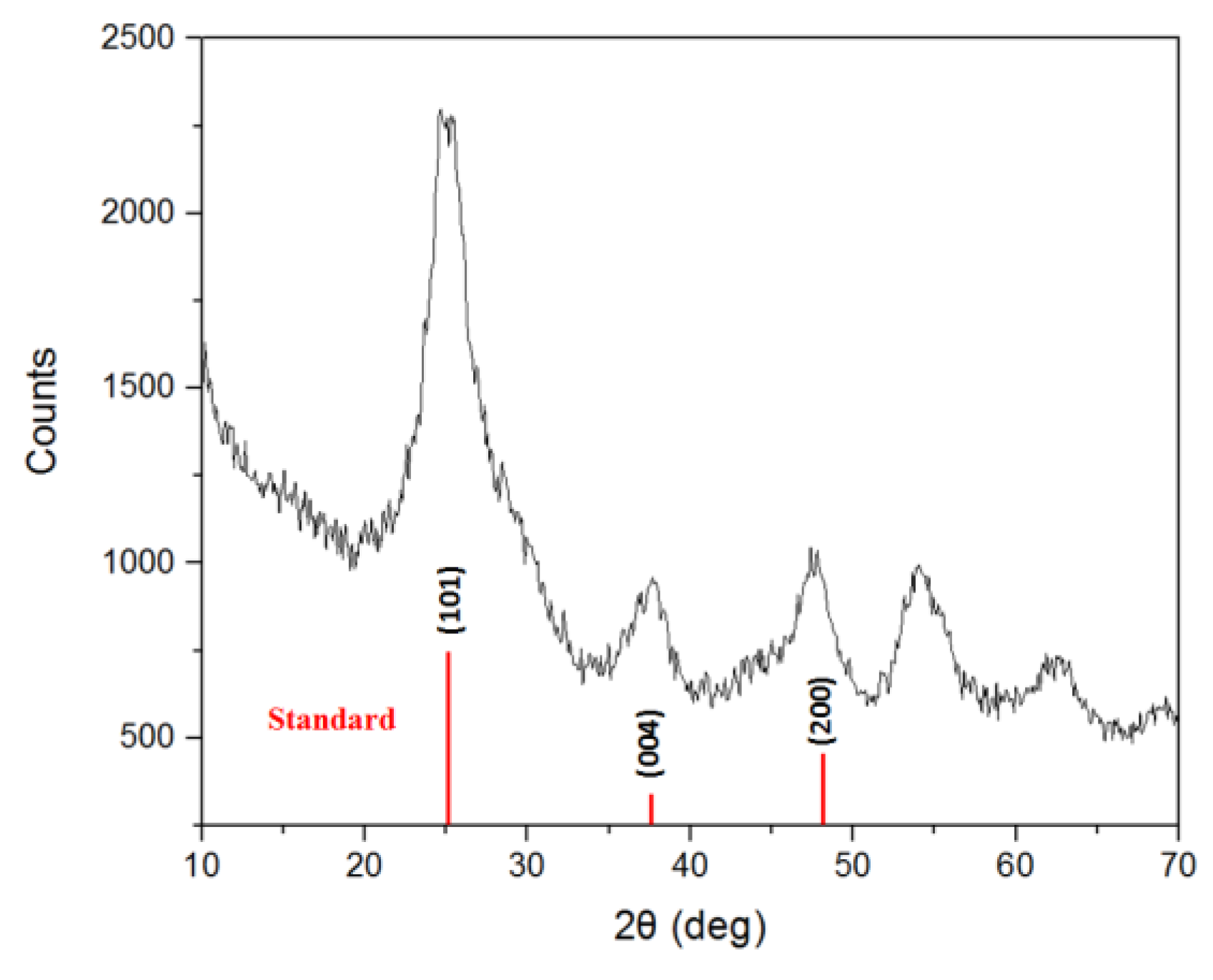
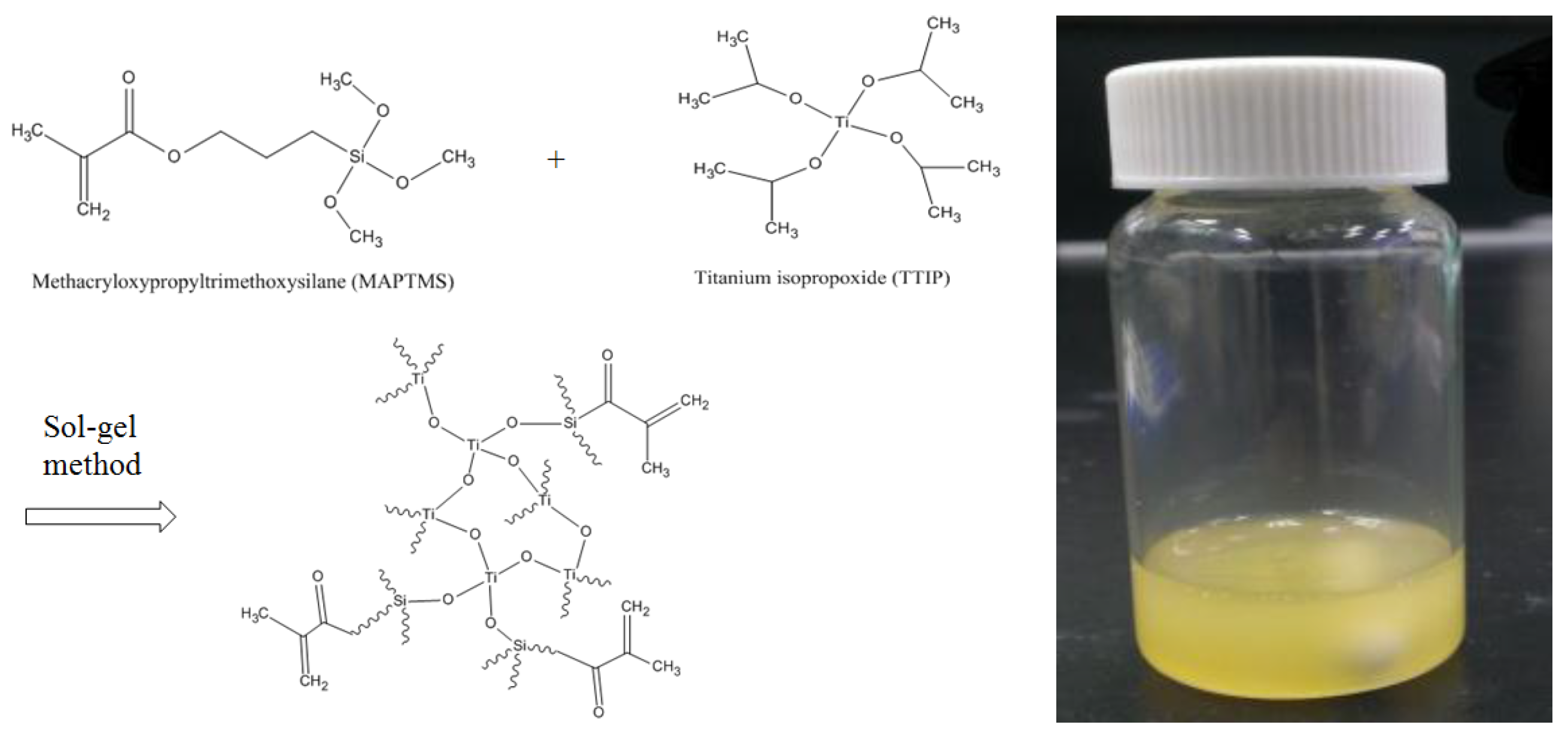
2.1. Sol-Gel Formation
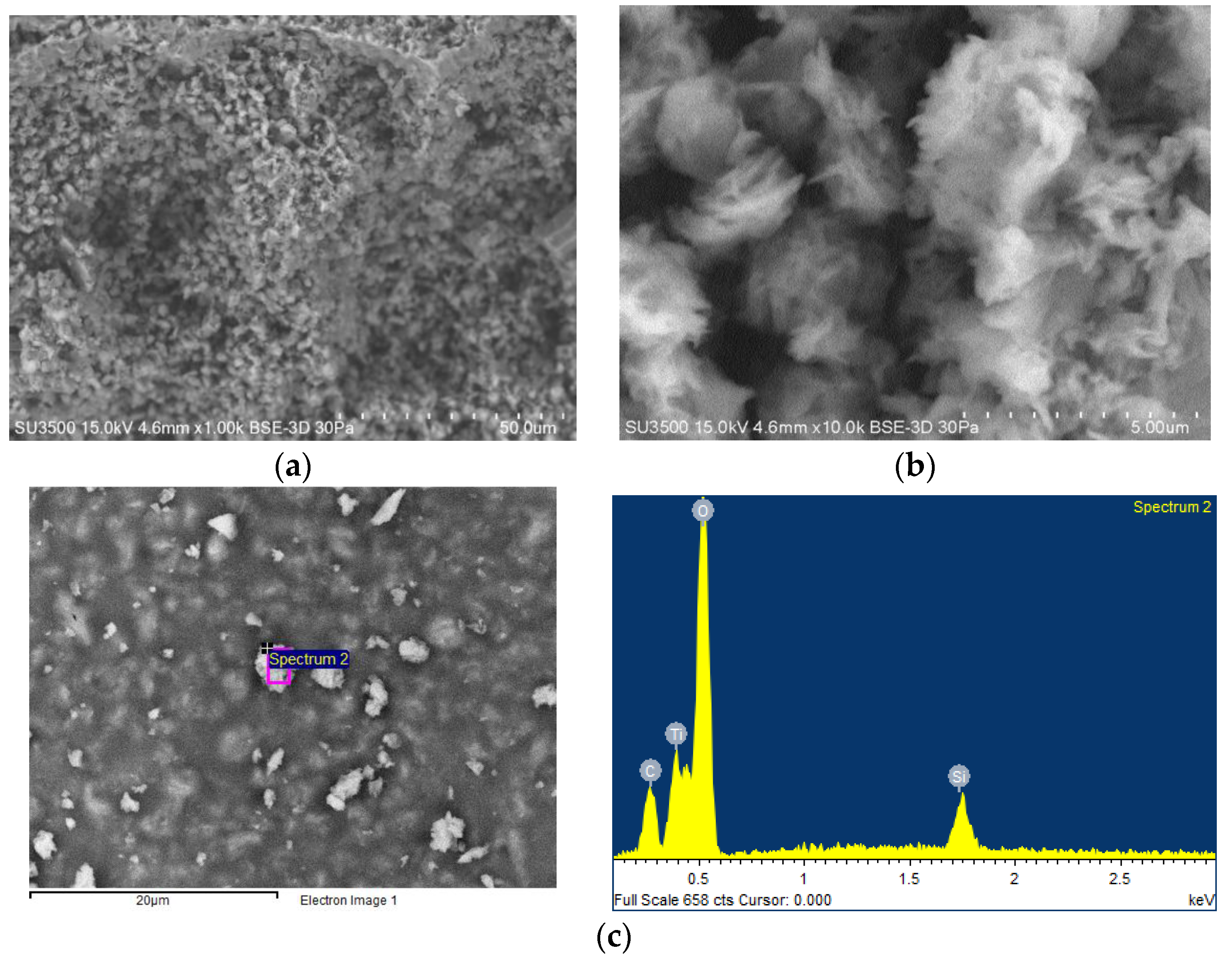
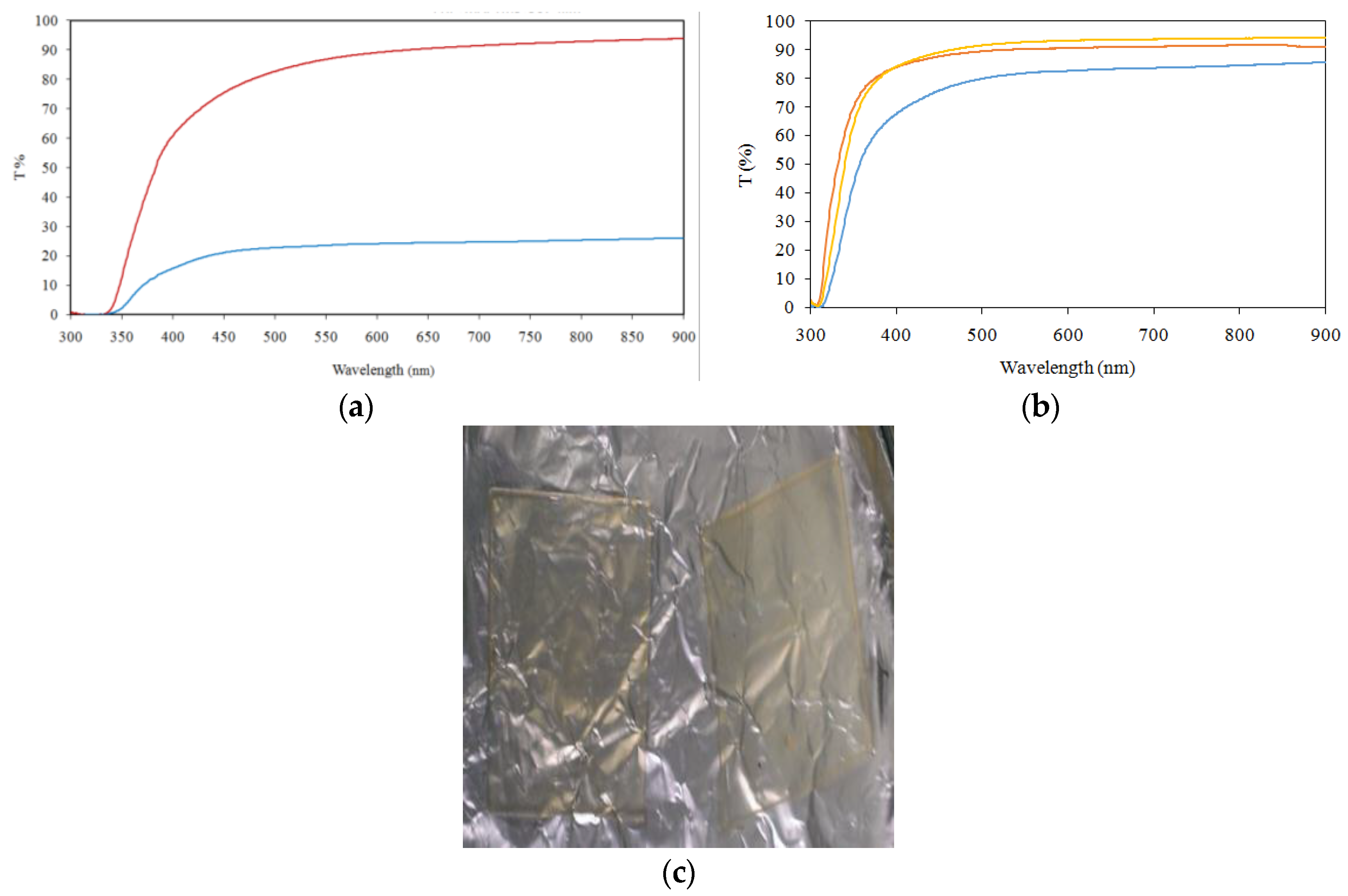
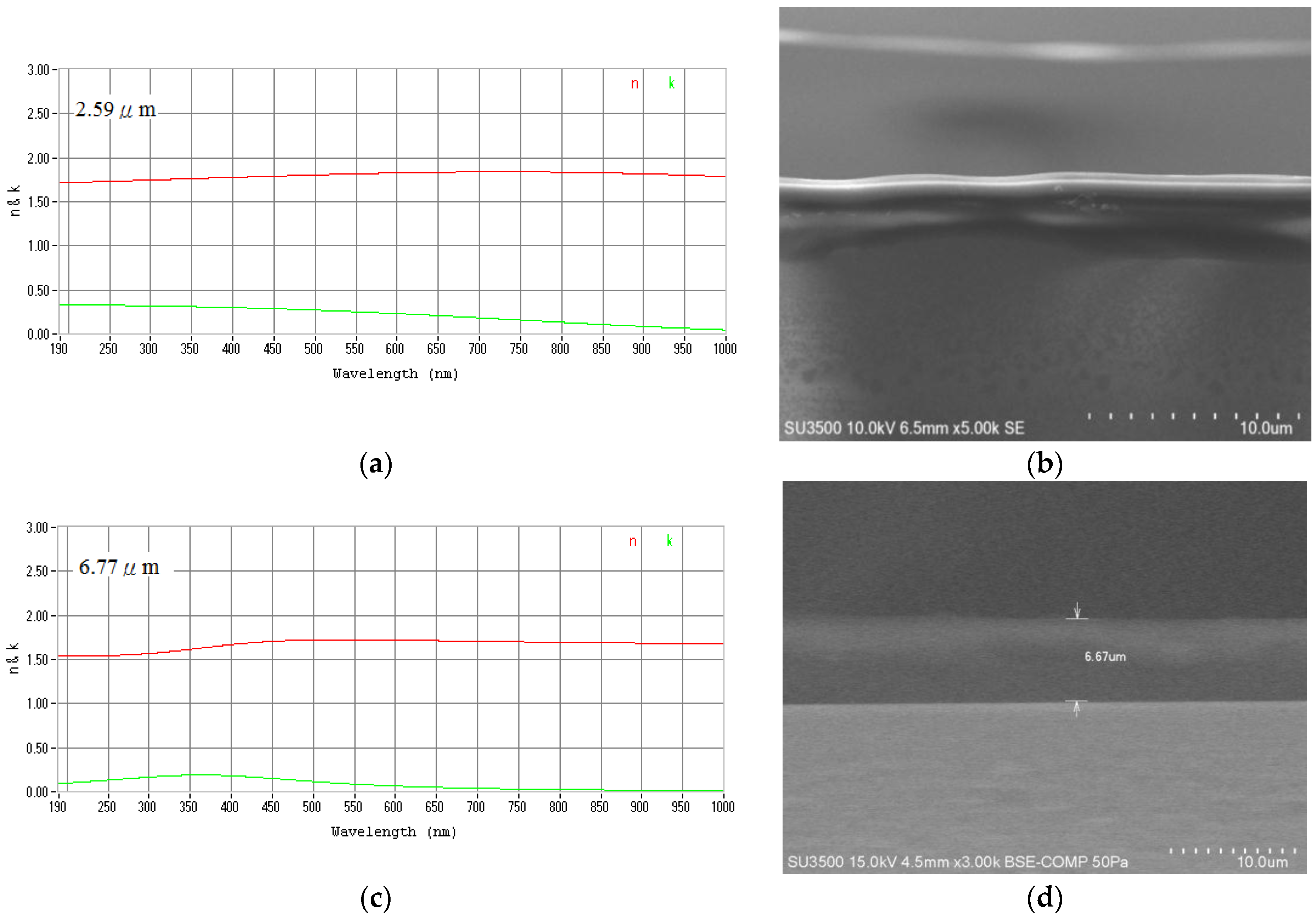
2.2. Organic-Inorganic Hybrid Polymer Films
3. Discussion
3.1. Sol-Gel Formation
3.2. Organic-Inorganic Hybrid Polymer Films
4. Materials and Methods
4.1. Chemicals
4.2. Synthesis of Organic-Inorganic Hybrid Sol-Gels
4.3. Preparation of the Organic-Inorganic Hybrid Coating
4.4. Measurements
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Paquet, C.; Cyr, P.W.; Kumacheva, E.; Manners, I. Rationalized approach to molecular tailoring of polymetallocenes with predictable optical properties. Chem. Mater. 2004, 16, 5205–5211. [Google Scholar] [CrossRef]
- Olshavsky, M.A.; Allcock, H.R. Polyphosphazenes with high refractive indices: Synthesis, characterization, and optical properties. Macromolecules 1995, 28, 6188–6197. [Google Scholar] [CrossRef]
- Olshavsky, M.A.; Allcock, H.R. Polyphosphazenes with high refractive indices: Optical dispersion and molar refractivity. Macromolecules 1997, 30, 4179–4183. [Google Scholar] [CrossRef]
- Sanchez, C.; Lebeau, B.; Chaput, F.; Boilot, J.P. Optical properties of functional hybrid organic-inorganic nanocomposites. Adv. Mater. 2003, 15, 1969–1993. [Google Scholar] [CrossRef]
- Balazs, A.C.; Emrick, T.; Russell, T.P. Nanoparticle Polymer Composites: Where Two Small Worlds Meet. Science 2006, 314, 1107–1110. [Google Scholar] [CrossRef] [PubMed]
- Arney, D.S.; Wood, T.E. Nanosize metal oxide particles for producing transparent metal oxide colloids and ceramers. U.S. Patent No. 6,432,526 B1, 13 August 2002. [Google Scholar]
- Chang, C.C.; Chen, W.C. Synthesis and optical properties of polyimide-silica hybrid thin films. Chem. Mater. 2002, 14, 4242–4248. [Google Scholar] [CrossRef]
- Lee, L.H.; Chen, W.C. High-refractive-index thin films prepared from trialkoxysilane-capped poly(methylmethacrylate)-titania materials. Chem. Mater. 2001, 13, 1137–1142. [Google Scholar] [CrossRef]
- Chau, J.L.H.; Lin, Y.M.; Li, A.K.; Su, W.F.; Chang, K.S.; Hsu, S.L.C.; Li, T.L. Transparent high refractive index nanocomposite thin films. Mater. Lett. 2007, 2908–2910. [Google Scholar] [CrossRef]
- Chang, C.C.; Cheng, L.P.; Huang, F.H.; Lin, C.Y.; Hsieh, C.F.; Wang, W.H. Preparation and characterization of TiO2 hybrid sol for UV-curable high-refractive-index organic-inorganic hybrid thin films. J. Sol-Gel Sci. Techn. 2010, 55, 199–206. [Google Scholar] [CrossRef]
- Liou, G.S.; Li, P.H.; Yen, H.J.; Yu, Y.Y.; Tsai, T.W.; Chen, W.C. Highly flexible and optical transparent 6F-PI/TiO2 optical hybrid films with tunable refractive index and excellent thermal stability. J. Mater. Chem. 2010, 20, 531–536. [Google Scholar] [CrossRef]
- Du, W.; Wang, H.; Zhong, W.; Shen, L.; Du, Q. High refractive index films prepared from titanium chloride and methyl methacrylate via a non-aqueous sol–gel route. J. Sol-Gel Sci. Technol. 2005, 34, 227–231. [Google Scholar] [CrossRef]
- Caseri, W. Nanocomposites of polymers and metals or semiconductors: Historical background and optical properties. Macromol. Rapid Commun. 2000, 21, 705–722. [Google Scholar] [CrossRef]
- Caseri, W.R. Nanocomposites of polymers and inorganic particles: Preparation, structure and properties. Mater. Sci. Technol. 2006, 22, 807–817. [Google Scholar] [CrossRef]
- Althues, H.; Henle, J.; Kaskel, S. Functional inorganic nanofillers for transparent polymers. Chem. Soc. Rev. 2007, 36, 1454–1465. [Google Scholar] [CrossRef] [PubMed]
- Antonello, A.; Guglielmi, M.; Bello, V.; Mattei, G.; Chiasera, A.; Ferrari, M.; Martucci, A. Titanate nanosheets as high refractive layer in vertical microcavity incorporating semiconductor quantum dots. J. Phys. Chem. C 2010, 114, 18423–18428. [Google Scholar] [CrossRef]
- Hodroj, A.; Chaix-Pluchery, O.; Audier, M.; Gottlieb, U.; Deschanvres, J.L. Thermal annealing Ti–Si–O thin films. J. Mater. Res. 2008, 23, 755–759. [Google Scholar] [CrossRef]
- Miskam, M.; Bakar, N.K.A.; Mohamad, S. Preparation and characterization of new sol-gel titanium(IV)butoxide-cyanopropyltriethoxysilane hybrid sorbent for extraction of polar aromatic amines. J. Sol-Gel Sci. Technol. 2013, 67, 121–129. [Google Scholar] [CrossRef]
- Zeitler, V.A.; Brown, C.A. The infrared spectra of some Ti–O–Si, Ti–O–Ti and Si–O–Si compounds. J. Phys. Chem. 1957, 61, 1174–1177. [Google Scholar] [CrossRef]
- Rodrigues, D.E.; Breman, A.B.; Betrabet, C.; Wang, B.; Wilkes, G.L. Structural features of sol-gel-derived hybrid inorganic-organic network creamer materials by small-angle X-ray scattering. Chem. Mater. 1992, 4, 1437–1446. [Google Scholar] [CrossRef]
- Romeo, H.E.; Fanovich, M.A.; Williams, R.J.J.; Matejka, L.; Plestil, J.; Brus, J. Self-assembly of a bridged silsesquioxane containing a pendant hydrophobic chain in the organic bridge. Macromolecules 2007, 40, 1435–1443. [Google Scholar] [CrossRef]
- Tsuzuki, T. Abnormal transmittance of refractive-index-modified ZnO/organic hybrid films. Macromol. Mater. Eng. 2008, 293, 109–113. [Google Scholar] [CrossRef]
- Sanchez, C.; de Soler-Illia, G.J.A.A.; Ribot, A.F.; Lalot, T.; Mayer, C.R.; Cabuil, V. Designed hybrid organic-inorganic nanocomposites from functional nanobuilding blocks. Chem. Mater. 2001, 13, 3061–3083. [Google Scholar] [CrossRef]
- Blanc, D.; Pelissier, S.; Saravanamuttu, K.; Najafi, S.I.; Andrews, M.P. Self-processing of surface-relief gratings in photosensitive hybrid sol-gel glasses. Adv. Mater. 1999, 11, 1508–1511. [Google Scholar] [CrossRef]
- Whang, C.M.; Yeo, C.S.; Kim, Y.H. Preparation and characterization of sol-gel derived SiO2-TiO2-PDMS composite films. Bull. Korean Chem. Soc. 2001, 22, 1366–1370. [Google Scholar]
- Varshneya, A.K. Fundamentals of Inorganic Glasses; Academic Press: San Diego, CA, USA, 1994; p. 459. [Google Scholar]
- Ku, C.C.; Liepins, R. Electrical Properties of Polymers: Chemical Principles; Hanser Publishers: New York, NY, USA, 1987; p. 41. [Google Scholar]

© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ma, H.-Y.; Wang, T.-L.; Chang, P.-Y.; Yang, C.-H. High Refractive Organic–Inorganic Hybrid Films Prepared by Low Water Sol-Gel and UV-Irradiation Processes. Nanomaterials 2016, 6, 44. https://doi.org/10.3390/nano6030044
Ma H-Y, Wang T-L, Chang P-Y, Yang C-H. High Refractive Organic–Inorganic Hybrid Films Prepared by Low Water Sol-Gel and UV-Irradiation Processes. Nanomaterials. 2016; 6(3):44. https://doi.org/10.3390/nano6030044
Chicago/Turabian StyleMa, Hsiao-Yuan, Tzong-Liu Wang, Pei-Yu Chang, and Chien-Hsin Yang. 2016. "High Refractive Organic–Inorganic Hybrid Films Prepared by Low Water Sol-Gel and UV-Irradiation Processes" Nanomaterials 6, no. 3: 44. https://doi.org/10.3390/nano6030044
APA StyleMa, H.-Y., Wang, T.-L., Chang, P.-Y., & Yang, C.-H. (2016). High Refractive Organic–Inorganic Hybrid Films Prepared by Low Water Sol-Gel and UV-Irradiation Processes. Nanomaterials, 6(3), 44. https://doi.org/10.3390/nano6030044





