Synthesis and Application of Amine Functionalized Iron Oxide Nanoparticles on Menaquinone-7 Fermentation: A Step towards Process Intensification
Abstract
:1. Introduction
2. Experimental Section
2.1. Materials
2.2. Synthesis and Characterization of l-Lys@IONs
2.3. Immobilization of Bacterial Cells with l-Lys@IONs and Fermentation
2.4. MK-7 Extraction and Measurement Procedure
3. Results and Discussion

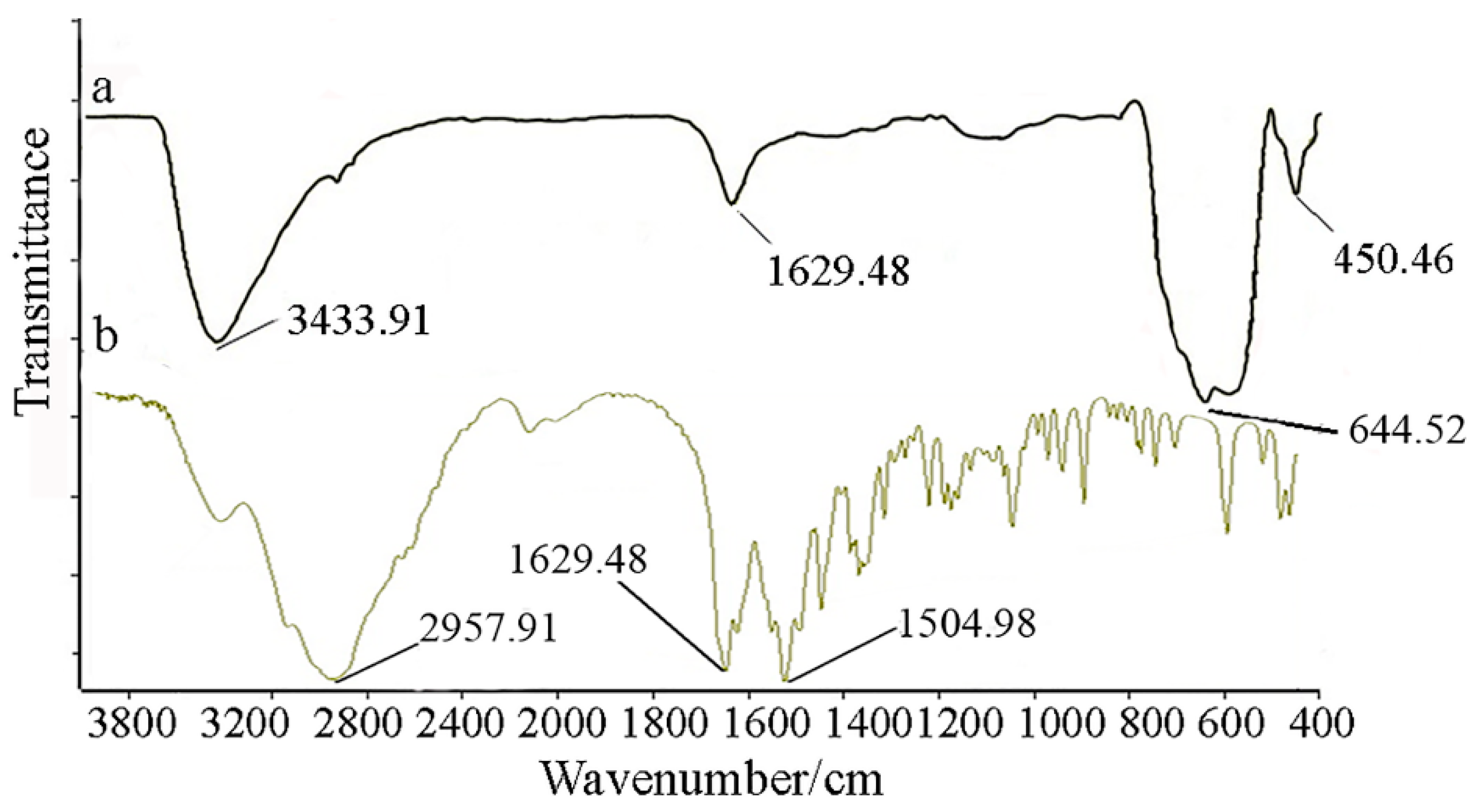
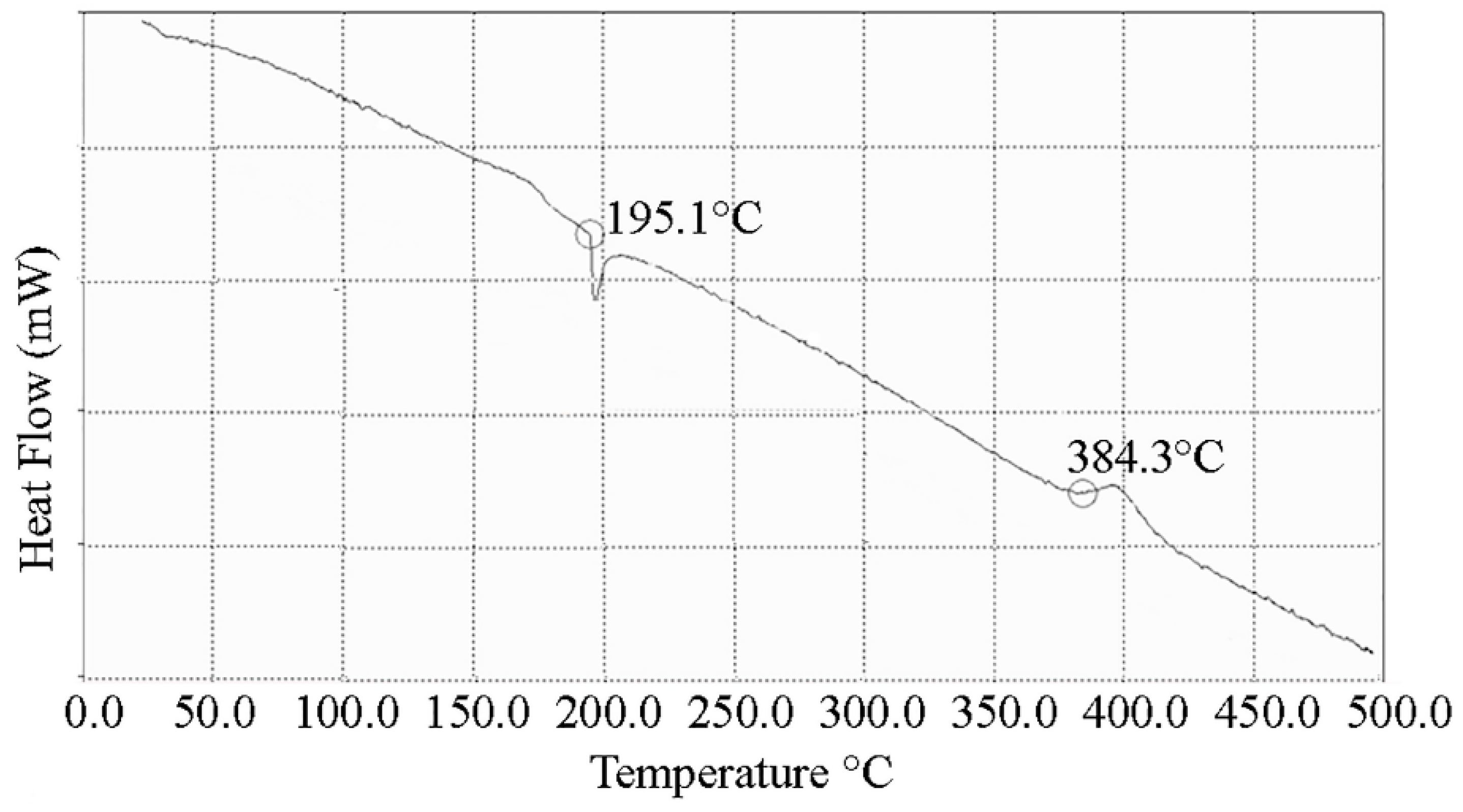
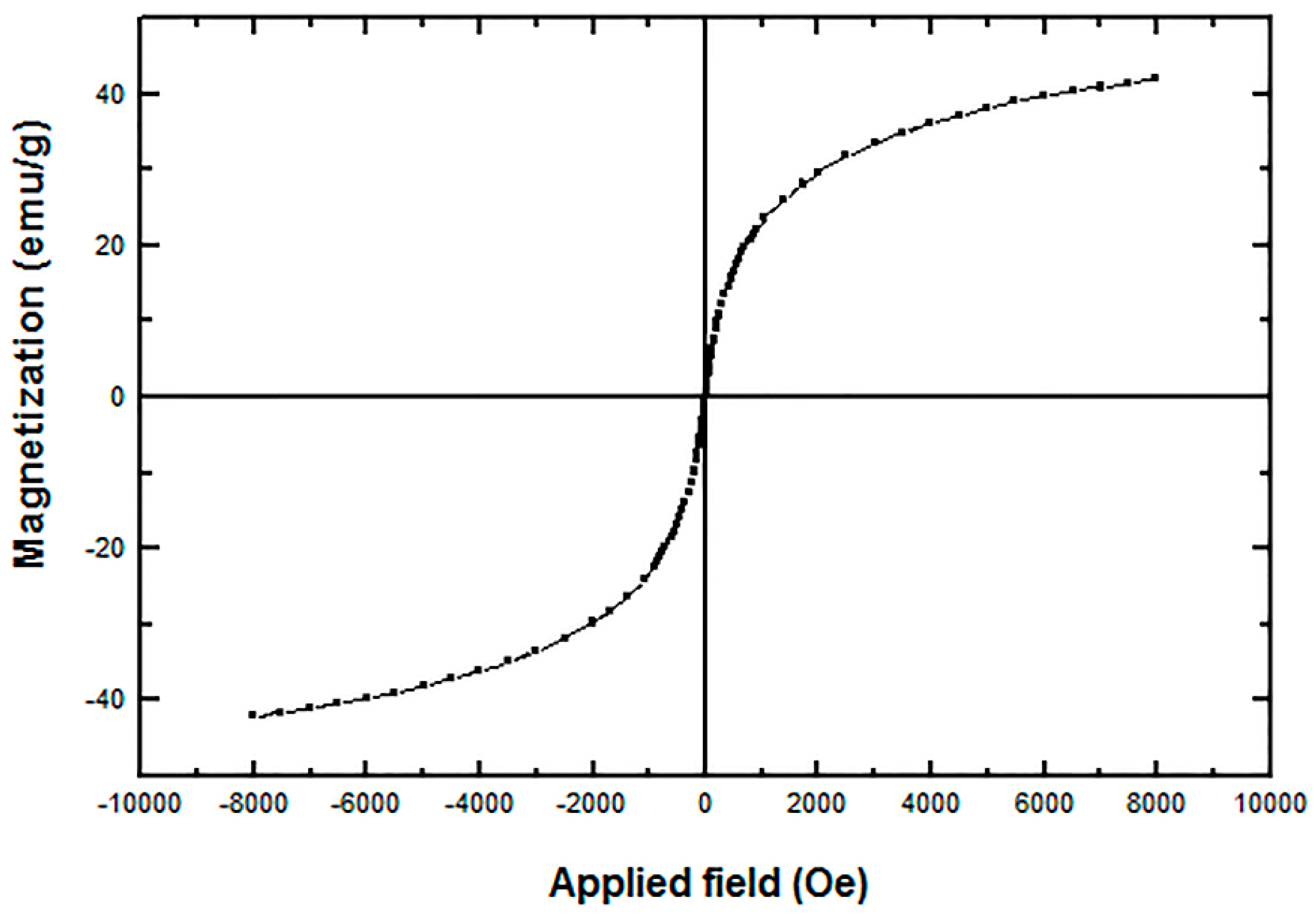
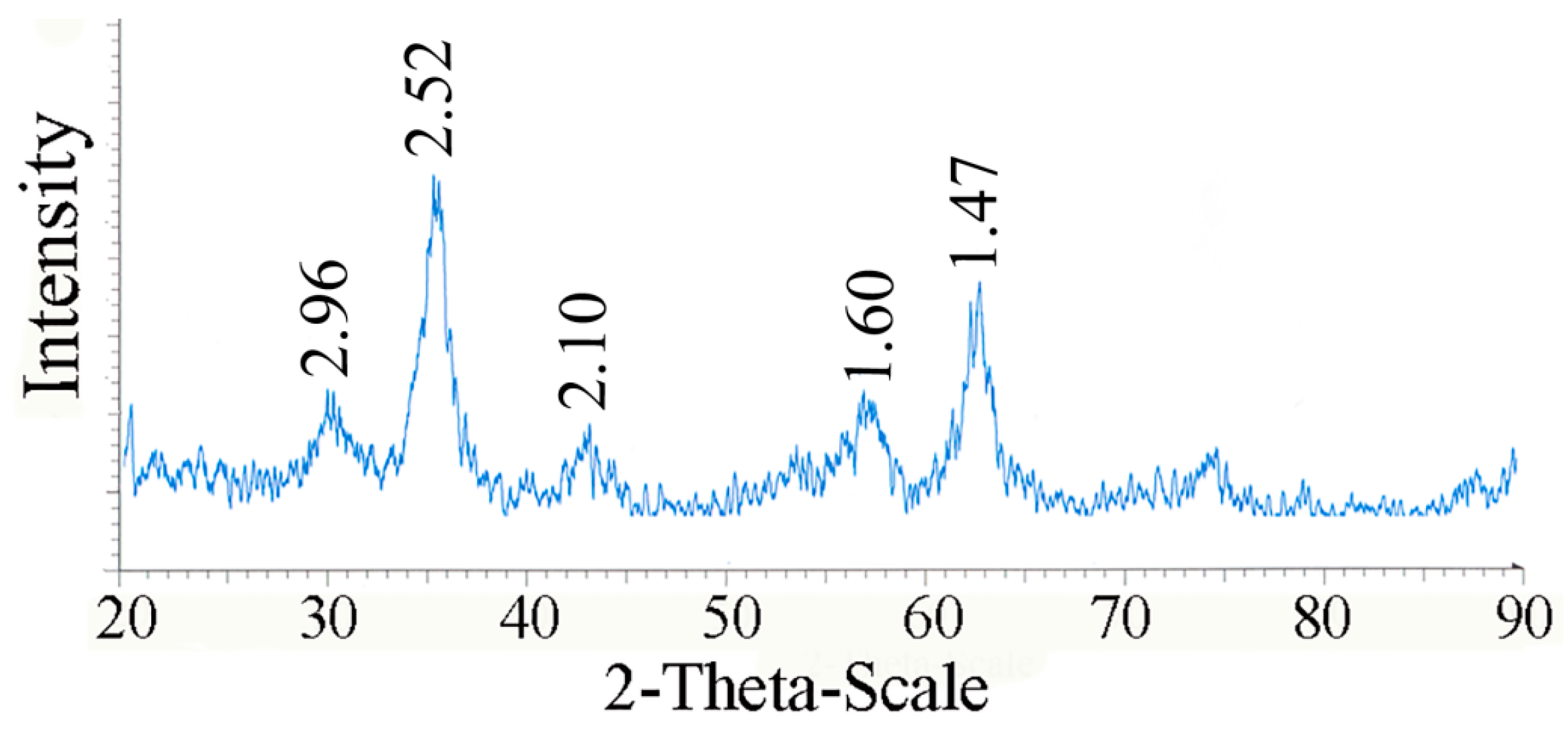
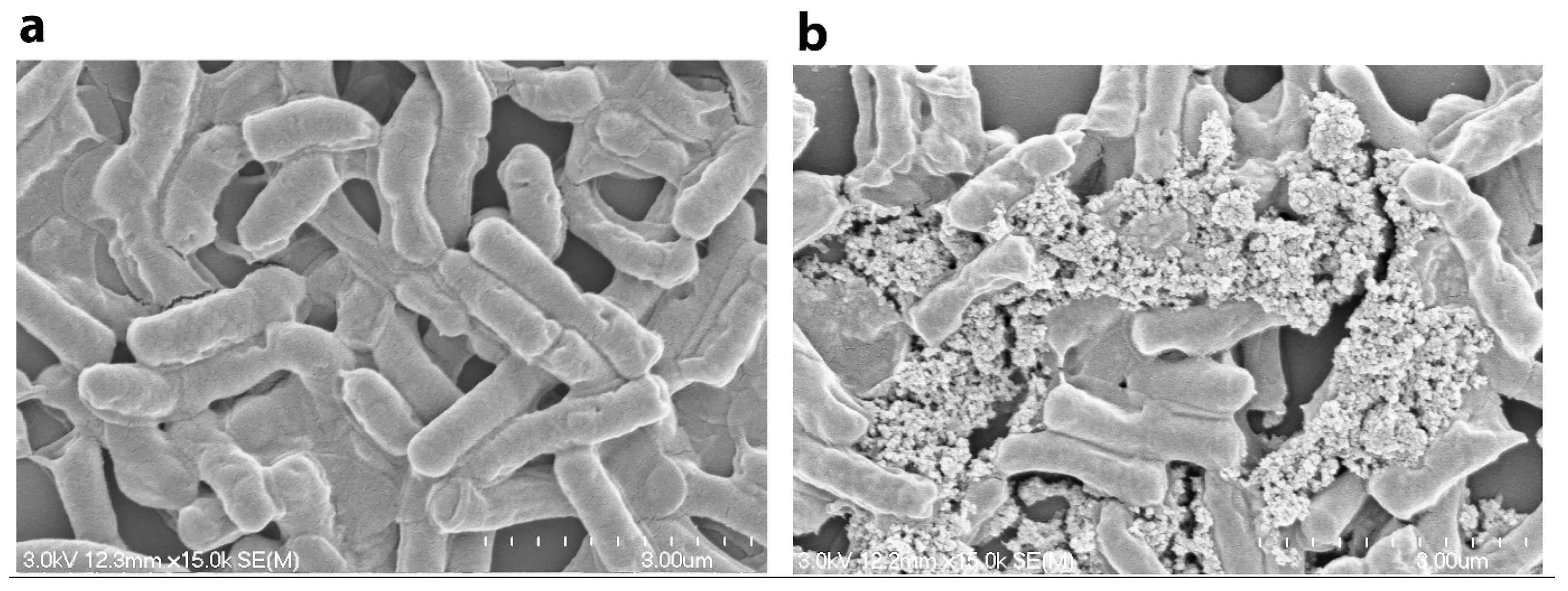
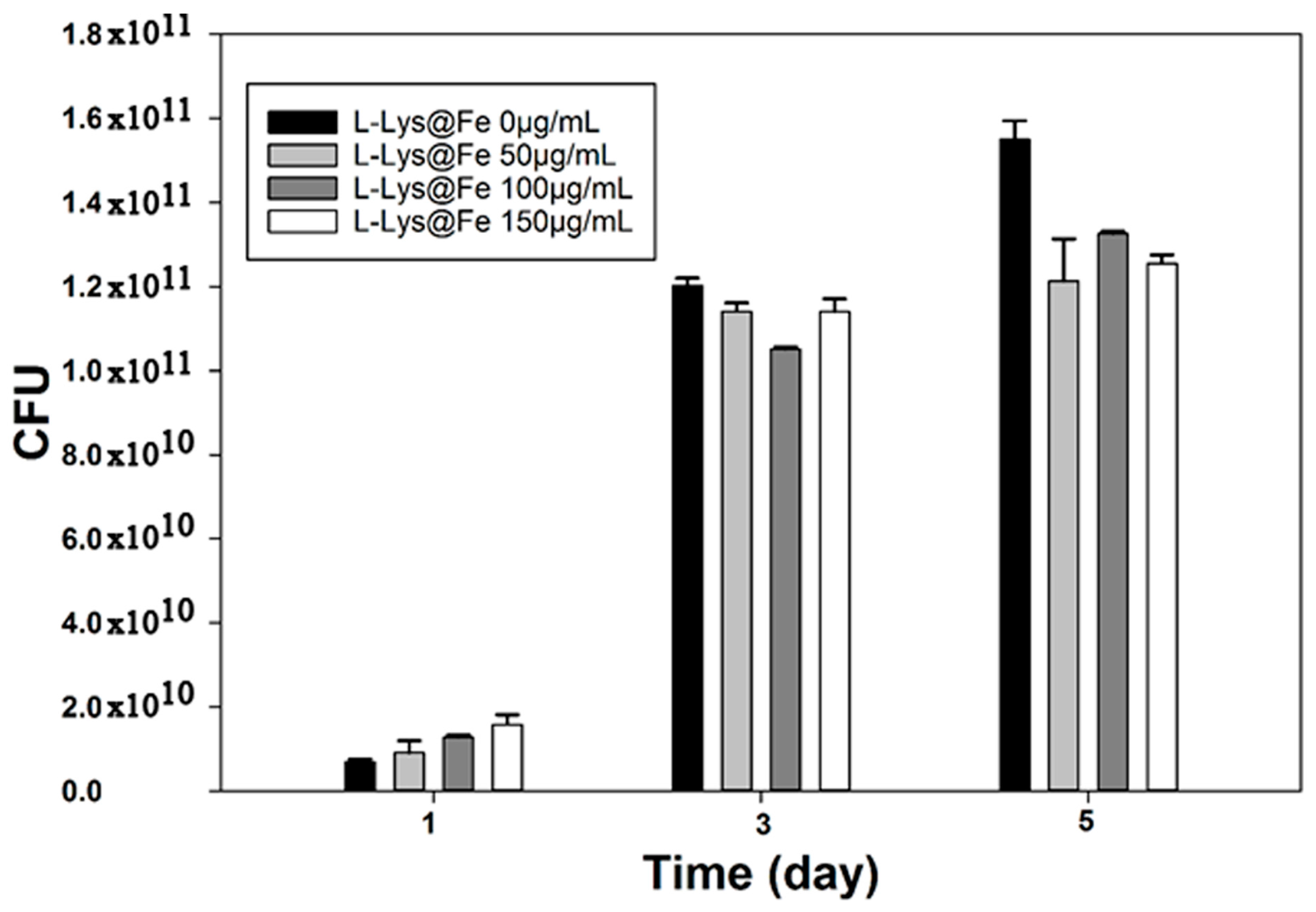
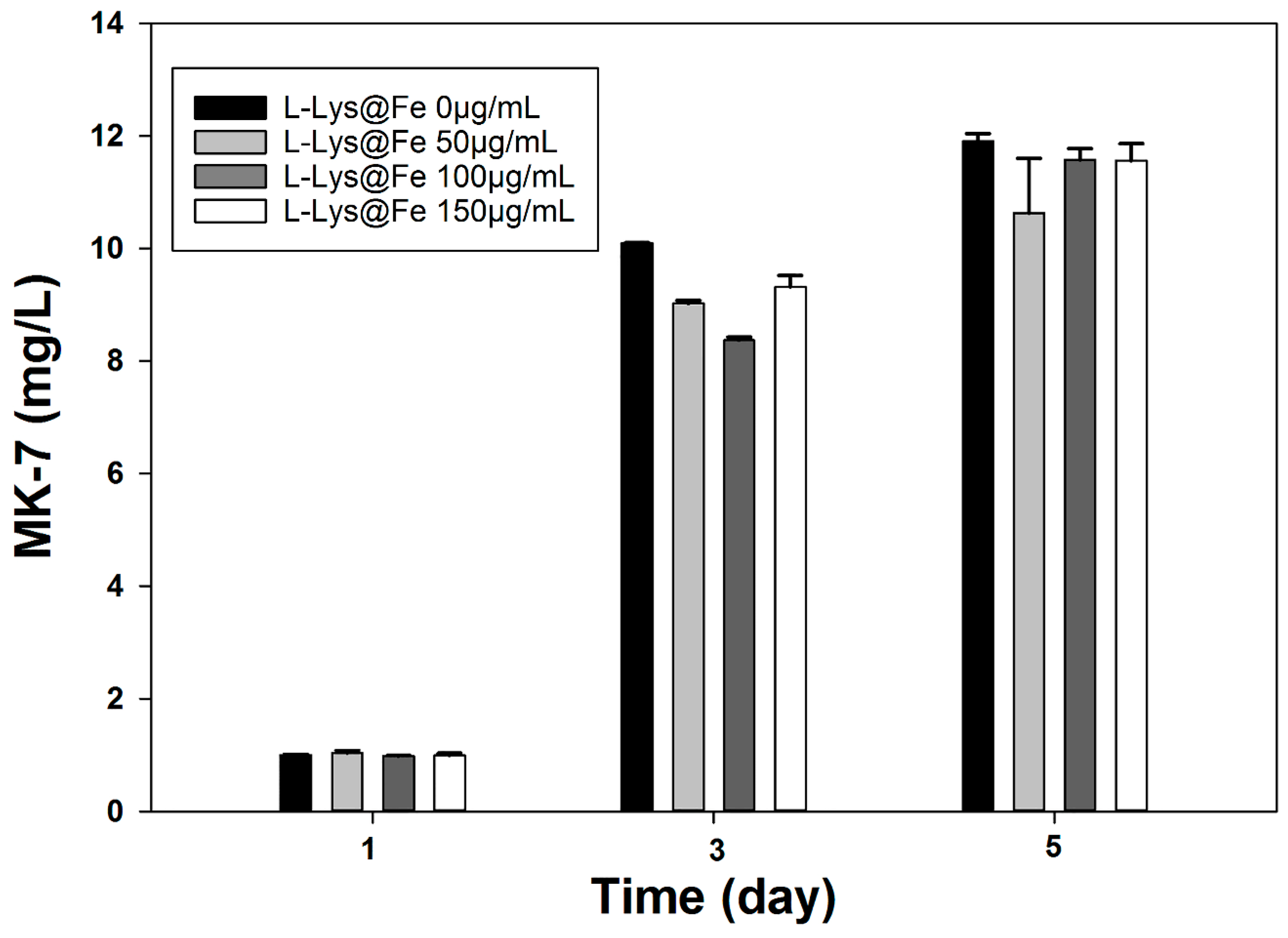
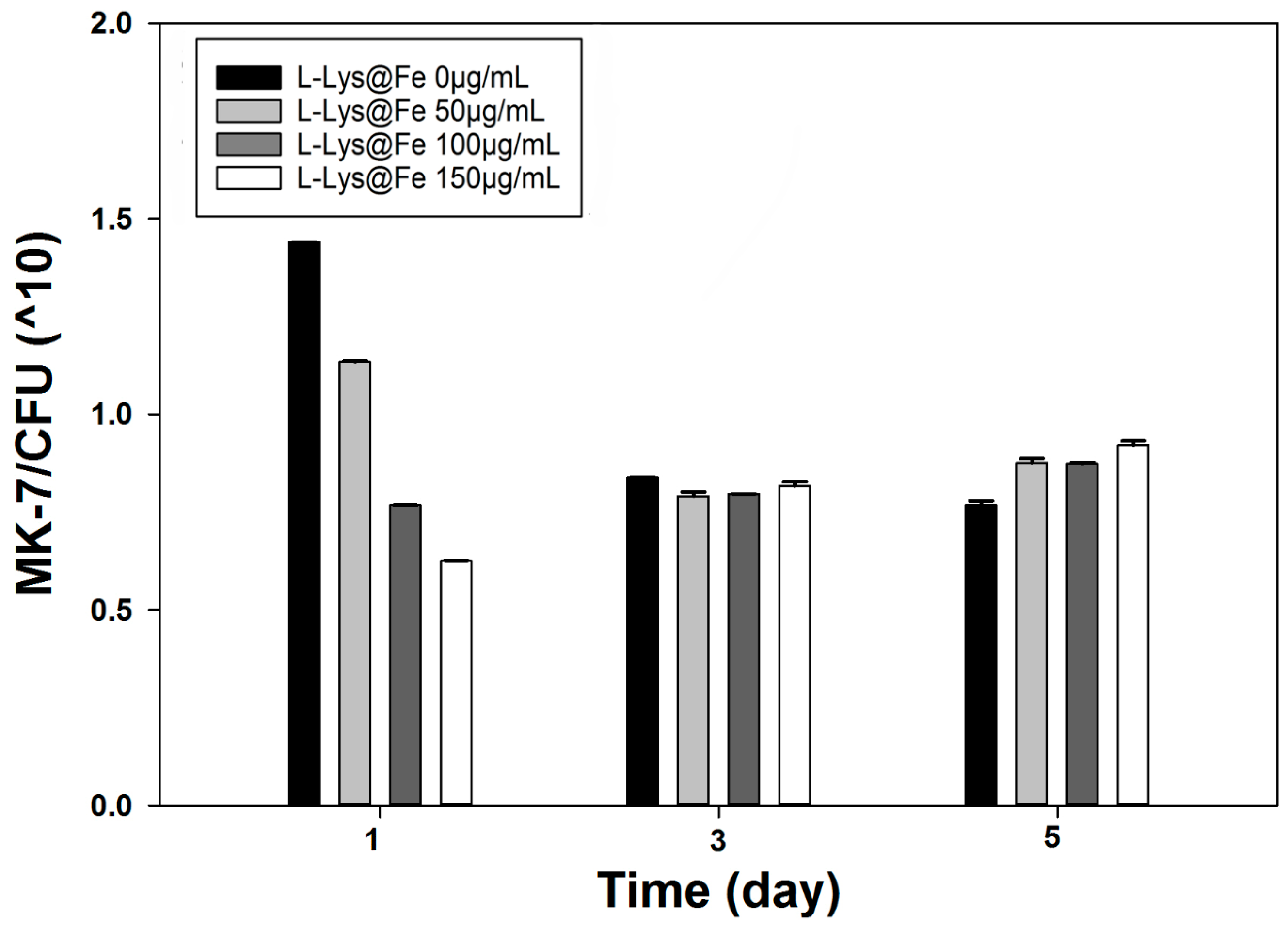
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Berenjian, A.; Mahanama, R.; Kavanagh, J.; Dehghani, F. Vitamin K series: Current status and future prospects. Crit. Rev. Biotechnol. 2015, 35, 174–184. [Google Scholar] [CrossRef] [PubMed]
- Berenjian, A.; Mahanama, R.; Talbot, A.; Regtop, H.; Kavanagh, J.; Dehghani, F. Designing of an intensification process for biosynthesis and recovery of menaquinone-7. Appl. Biochem. Biotechnol. 2014, 172, 1347–1357. [Google Scholar] [CrossRef] [PubMed]
- Wohlgemuth, R. The locks and keys to industrial biotechnology. New Biotechnol. 2009, 25, 204–213. [Google Scholar] [CrossRef] [PubMed]
- Vaghari, H.; Eskandari, M.; Sobhani, V.; Berenjian, A.; Song, Y.; Malmiri, H.J. Process intensification for production and recovery of biological products. Am. J. Biochem. Biotechnol. 2015, 11, 37–43. [Google Scholar] [CrossRef]
- Marques, M.P.C.; Fernandes, P. Microfluidic devices: Useful tools for bioprocess intensification. Molecules 2011, 16, 8368–8401. [Google Scholar] [CrossRef] [PubMed]
- Can, K.; Ozmen, M.; Ersoz, M. Immobilization of albumin on aminosilane modified superparamagnetic magnetite nanoparticles and its characterization. Colloid Surf. B 2009, 71, 154–159. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Guan, Y.; Yang, Y.; Ma, Z.; Wu, X.; Liu, H. Preparation of superparamagnetic immunomicrospheres and application for antibody purification. J. Appl. Polym. Sci. 2004, 94, 2205–2211. [Google Scholar] [CrossRef]
- Ansari, F.; Grigoriev, P.; Libor, S.; Tothill, I.E.; Ramsden, J.J. DBT degradation enhancement by decorating Rhodococcus erythropolis IGST8 with magnetic Fe3O4 nanoparticles. Biotechnol. Bioeng. 2009, 102, 1505–1512. [Google Scholar] [CrossRef] [PubMed]
- Ebrahiminezhad, A.; Davaran, S.; Amini, S.R.; Barar, J.; Moghadam, M.; Ghasemi, Y. Synthesis, characterization and anti-Listeria monocytogenes effect of amino acid coated magnetite nanoparticles. Curr. Nanosci. 2012, 8, 868–874. [Google Scholar] [CrossRef]
- Ebrahiminezhad, A.; Amini, S.R.; Davaran, S.; Barar, J.; Moghadam, M.; Ghasemi, Y. Impacts of iron oxide nanoparticles on the invasion power of Listeria monocytogenes. Curr. Nanosci. 2014, 10, 382–388. [Google Scholar] [CrossRef]
- Chatterjee, S.; Bandyopadhyay, A.; Sarkar, K. Effect of iron oxide and gold nanoparticles on bacterial growth leading towards biological application. J. Nanobiotechnol. 2011, 9, 34. [Google Scholar] [CrossRef] [PubMed]
- Grumezescu, A.M.; Mihaiescu, D.E.; Mogoşanu, D.E.; Chifiriuc, M.C.; Lažr, V.; Čluǧrescu, I.; Třistaru, V. In vitro assay of the antimicrobial activity of Fe3O4 and CoFe2O4/oleic acid–core/shell on clinical isolates of bacterial and fungal strains. Optoelectron. Adv. Mat. 2010, 4, 1798–1801. [Google Scholar]
- Ramteke, C.; Sarangi, B.K.; Chakrabarti, T.; Mudliar, S.; Dewanand, S.; Pandey, R.A. Synthesis and broad spectrum antibacterial activity of magnetite ferrofluid. Curr. Nanosci. 2010, 6, 587–591. [Google Scholar] [CrossRef]
- Ebrahiminezhad, A.; Amini, S.R.; Davaran, S.; Barar, J.; Ghasemi, Y. Impact of amino-acid coating on the synthesis and characteristics of iron-oxide nanoparticles (IONs). Bull. Kor. Chem. Soc. 2012, 33, 3957–3962. [Google Scholar] [CrossRef]
- Ebrahiminezhad, A.; Ghasemi, Y.; Rasoul-Amini, S.; Barar, J.; Davaran, S. Preparation of novel magnetic fluorescent nanoparticles using amino acids. Colloids Surf. B Biointerfaces 2013, 102, 534–539. [Google Scholar] [CrossRef] [PubMed]
- Ebrahiminezhad, A.; Amini, S.R.; Kouhpayeh, A.; Davaran, S.; Barar, J.; Ghasemi, Y. Impacts of amine functionalized iron oxide nanoparticles on HepG2 cell line. Curr. Nanosci. 2015, 11, 113–119. [Google Scholar] [CrossRef]
- Gholami, A.; Rasoul-amini, S.; Ebrahiminezhad, A.; Seradj, S.H.; Ghasemi, Y. Lipoamino acid coated superparamagnetic iron oxide nanoparticles concentration and time dependently enhanced growth of human hepatocarcinoma cell line (Hep-G2). J. Nanomater. 2015, 2015, 451405. [Google Scholar] [CrossRef]
- Mahanama, R.; Berenjian, A.; Valtchev, P.; Talbot, A.; Biffin, R.; Regtop, H.; Dehghani, F.; Kavanagh, J.M. Enhanced production of menaquinone 7 via solid substrate fermentation from Bacillus subtilis. Int. J. Food Eng. 2011, 7. [Google Scholar] [CrossRef]
- Mahanama, R.; Berenjian, A.; Dehghani, F.; Kavanagh, J. Modeling Menaquinone 7 production in tray type solid state fermenter. ANZIAM J. 2012, 53, 354–372. [Google Scholar]
- Larsen, M.U.; Seward, M.; Tripathi, A.; Shapley, N.C. Biocompatible nanoparticles trigger rapid bacteria clustering. Biotechnol. Prog. 2009, 25, 1094–1102. [Google Scholar] [CrossRef] [PubMed]
- Brunner, T.J.; Wick, P.; Manser, P.; Spohn, P.; Grass, R.N.; Limbach, L.K.; Bruinink, A.; Stark, W.J. In vitro cytotoxicity of oxide nanoparticles: Comparison to asbestos, silica, and the effect of particle solubility. Environ. Sci. Technol. 2006, 40, 4374–4381. [Google Scholar] [CrossRef] [PubMed]
- Epand, R.M.; Epand, R.F.; Savage, P.B. Ceragenins (cationic steroid compounds), a novel class of antimicrobial agents. Drug News Perspect. 2008, 21, 307–311. [Google Scholar] [CrossRef] [PubMed]
- Martin, S.T.; Morrison, C.L.; Hoffmann, M.R. Photochemical mechanism of size-quantized vanadium-doped TiO2 particles. J. Phys. Chem. 1994, 98, 13695–13704. [Google Scholar] [CrossRef]
- Berenjian, A.; Mahanama, R.; Talbot, A.; Biffin, R.; Regtop, H.; Valtchev, P.; Kavanagh, J.; Dehghani, F. Efficient media for high menaquinone-7 production: Response surface methodology approach. New Biotechnol. 2011, 28, 665–672. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ebrahiminezhad, A.; Varma, V.; Yang, S.; Ghasemi, Y.; Berenjian, A. Synthesis and Application of Amine Functionalized Iron Oxide Nanoparticles on Menaquinone-7 Fermentation: A Step towards Process Intensification. Nanomaterials 2016, 6, 1. https://doi.org/10.3390/nano6010001
Ebrahiminezhad A, Varma V, Yang S, Ghasemi Y, Berenjian A. Synthesis and Application of Amine Functionalized Iron Oxide Nanoparticles on Menaquinone-7 Fermentation: A Step towards Process Intensification. Nanomaterials. 2016; 6(1):1. https://doi.org/10.3390/nano6010001
Chicago/Turabian StyleEbrahiminezhad, Alireza, Vikas Varma, Shuyi Yang, Younes Ghasemi, and Aydin Berenjian. 2016. "Synthesis and Application of Amine Functionalized Iron Oxide Nanoparticles on Menaquinone-7 Fermentation: A Step towards Process Intensification" Nanomaterials 6, no. 1: 1. https://doi.org/10.3390/nano6010001
APA StyleEbrahiminezhad, A., Varma, V., Yang, S., Ghasemi, Y., & Berenjian, A. (2016). Synthesis and Application of Amine Functionalized Iron Oxide Nanoparticles on Menaquinone-7 Fermentation: A Step towards Process Intensification. Nanomaterials, 6(1), 1. https://doi.org/10.3390/nano6010001





