Size Effect of Ordered Mesoporous Carbon Nanospheres for Anodes in Li-Ion Battery
Abstract
:1. Introduction
2. Results and Discussion

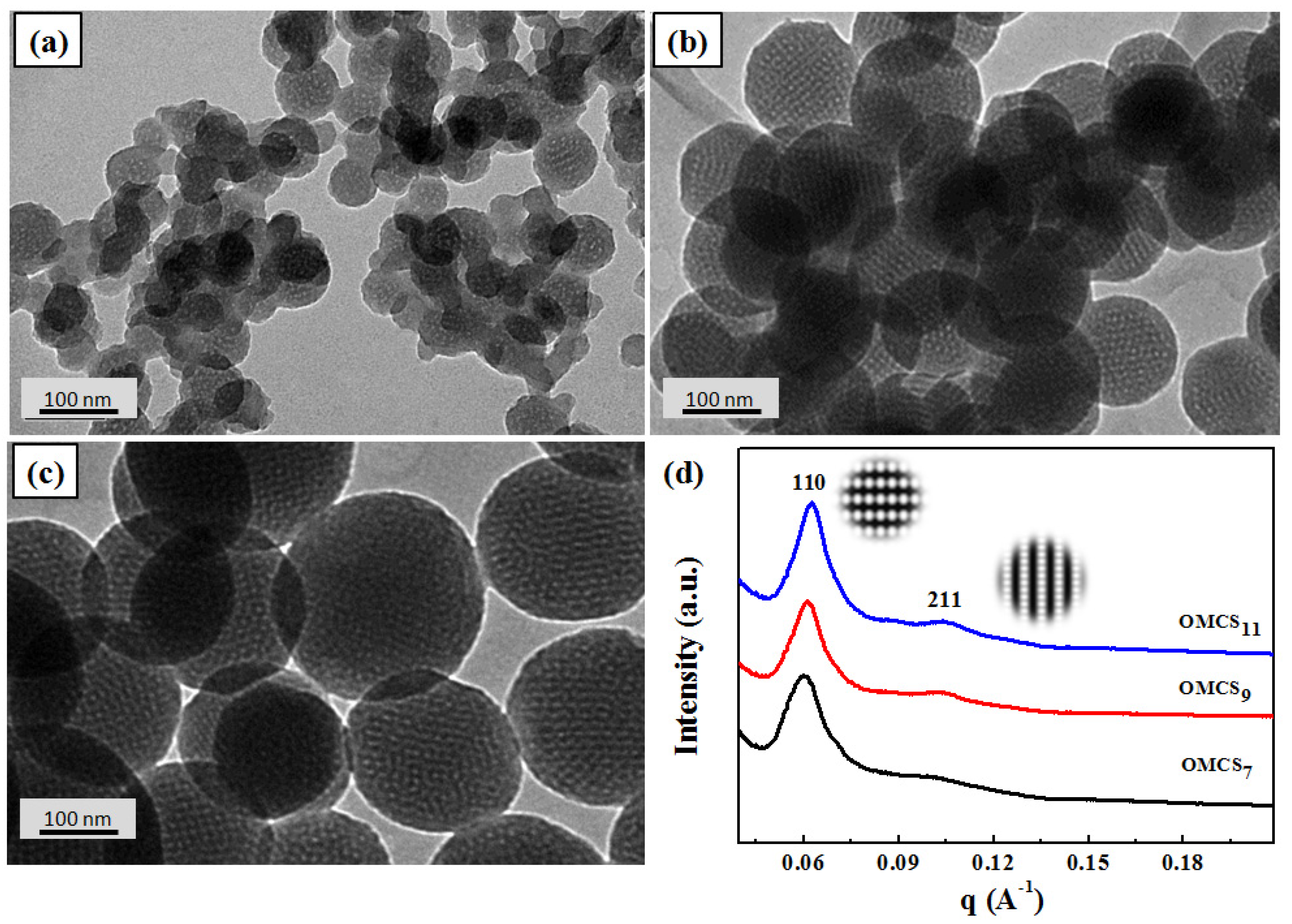
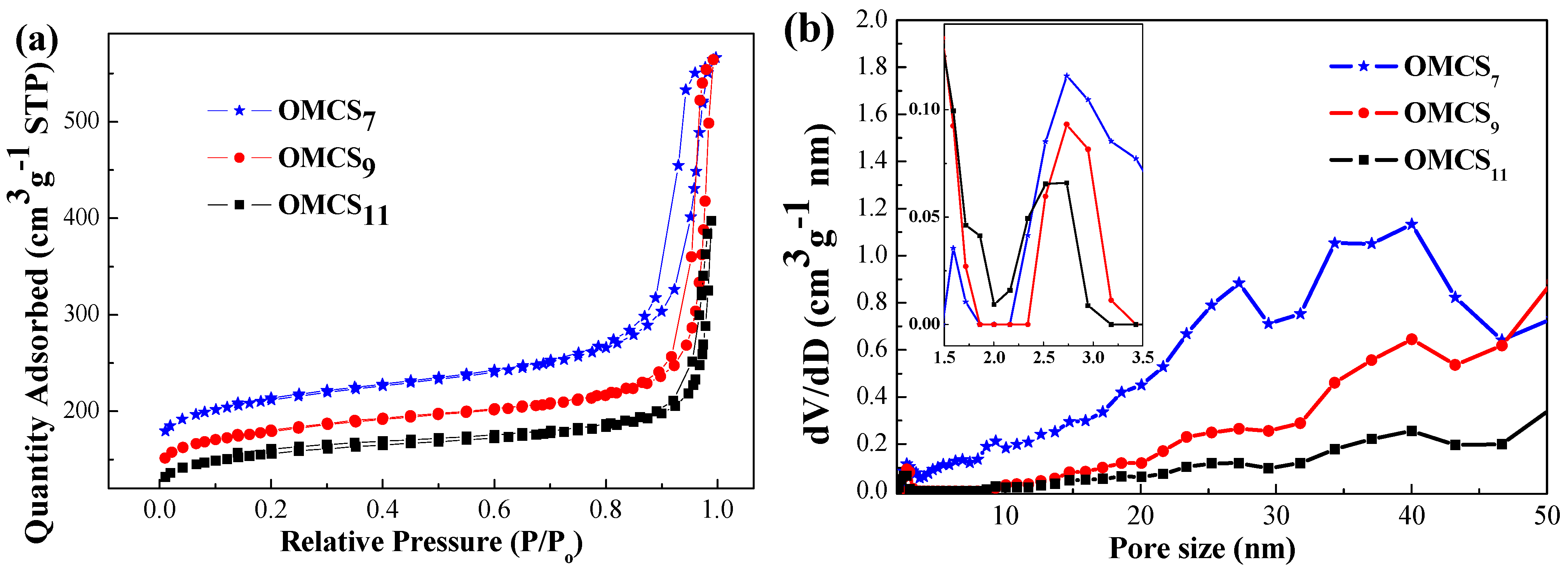
| Sample | SBET a (m2·g−1) | Pore Size b (nm) | Pore Volume b (cm3·g−1) | t-Plot Micropore Surface Area (m2·g−1) | Micropore/Total Surface Area Ratio |
|---|---|---|---|---|---|
| OMCS7 | 728 | 5.0 | 0.82 | 505 | 0.694 |
| OMCS9 | 615 | 3.4 | 0.67 | 425 | 0.691 |
| OMCS11 | 537 | 2.7 | 0.45 | 374 | 0.697 |
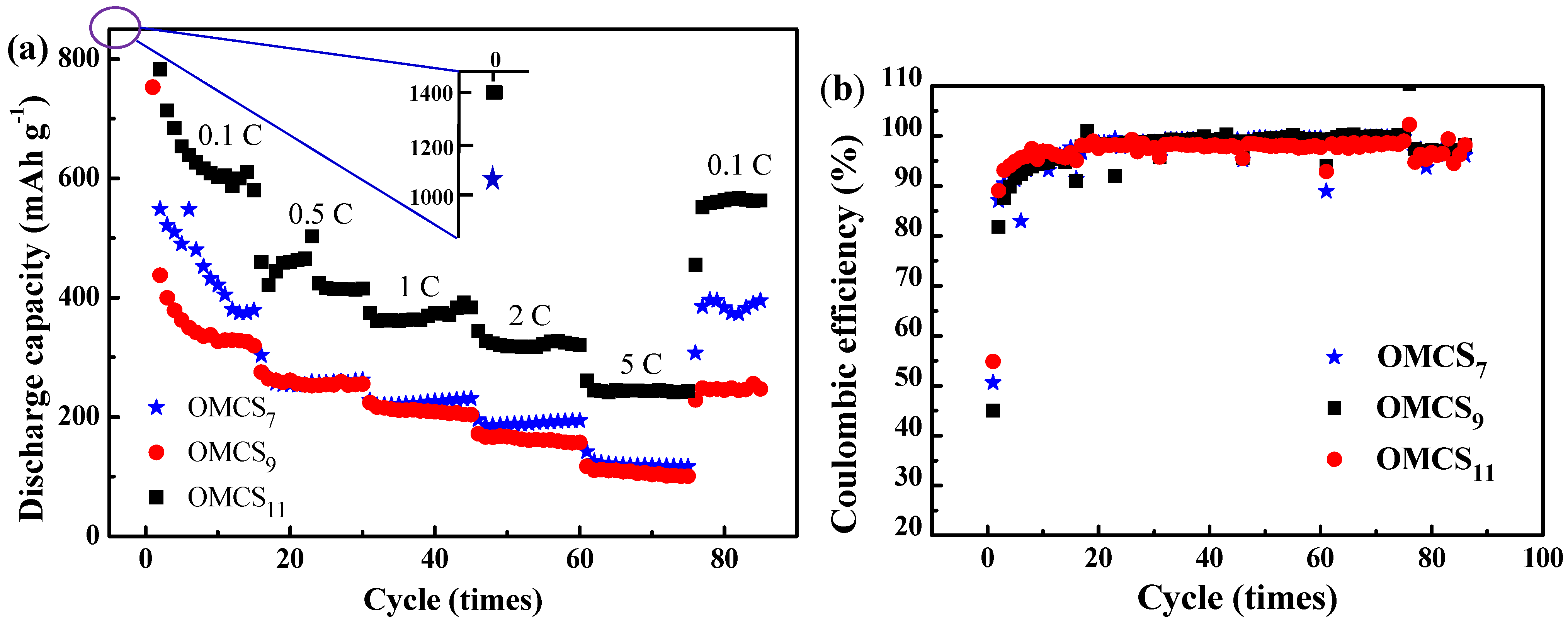
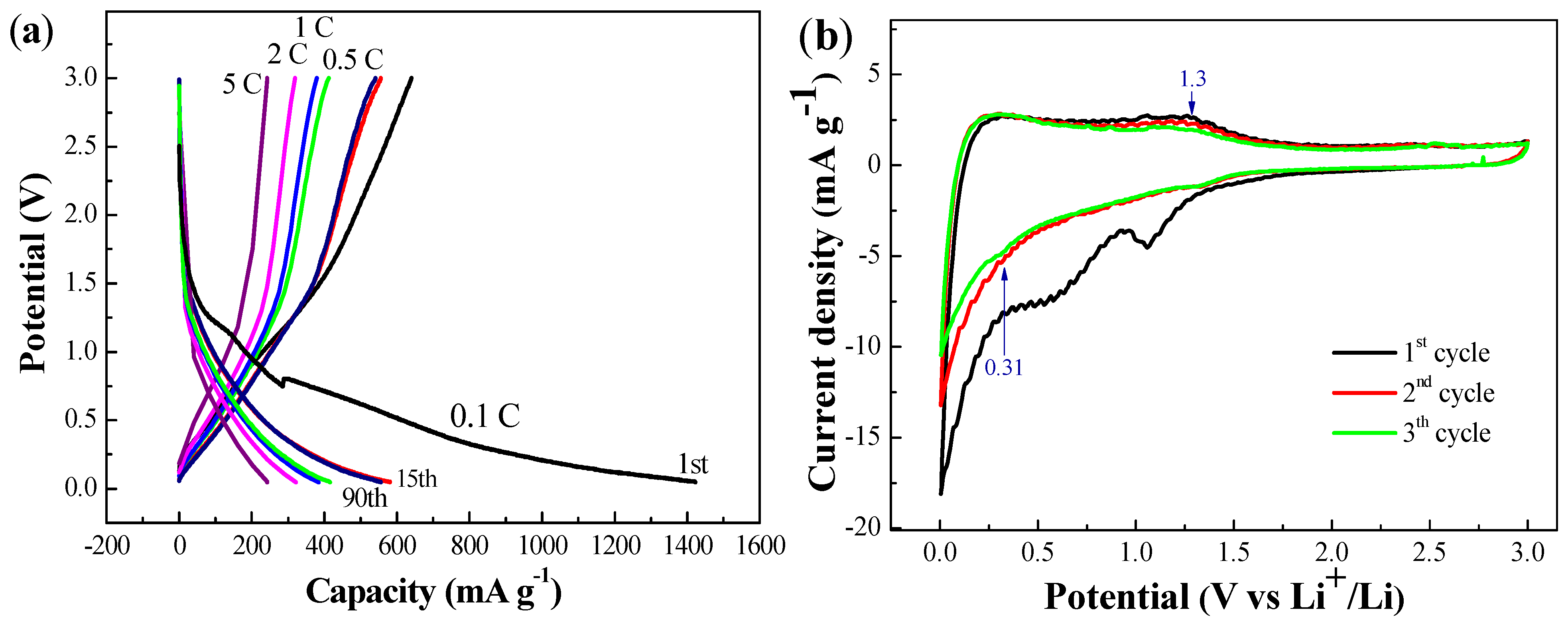

3. Experimental Section
3.1. Chemicals
3.2. Synthesis of Ordered Mesoporous Carbon Nanospheres (OMCS)
3.3. Characterization and Measurements
3.4. Electrochemical Measurements
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Su, D.S. Chemistry of energy conversion and storage. ChemSusChem 2014, 7, 1199–1200. [Google Scholar] [CrossRef] [PubMed]
- Shukla, A.K.; Kumar, T.P. Materials for next-generation lithium batteries. Curr. Sci. 2008, 94, 314–331. [Google Scholar]
- Landi, B.J.; Ganter, M.J.; Cress, C.D.; DiLeo, R.A.; Raffaelle, R.P. Carbon nanotubes for lithium ion batteries. Energy Environ. Sci. 2009, 2, 638–654. [Google Scholar] [CrossRef]
- Fujimoto, H. Development of efficient carbon anode material for a high-power and long-life lithium ion battery. J. Power Sources 2010, 195, 5019–5024. [Google Scholar] [CrossRef]
- Mabuchi, A.; Tokumitsu, K.; Fujimoto, H.; Kasuh, T. Charge-discharge characteristics of the mesocarbon miocrobeads heat-treated at different temperatures. J. Electrochem. Soc. 1995, 142, 1041–1046. [Google Scholar] [CrossRef]
- Yang, Z.; Zhang, J.; Kintner-Meyer, M.C.W.; Lu, X.; Choi, D.; Lemmon, J.P.; Liu, J. Electrochemical energy storage for green grid. Chem. Rev. 2011, 111, 3577–3613. [Google Scholar] [CrossRef] [PubMed]
- Armand, M.; Tarascon, J.M. Building better batteries. Nature 2008, 45, 652–657. [Google Scholar] [CrossRef] [PubMed]
- Endo, M.; Kim, C.; Nishimura, K.; Fujino, T.; Miyashita, K. Recent development of carbon materials for Li ion batteries. Carbon 2000, 38, 183–197. [Google Scholar] [CrossRef]
- Scrosati, B.; Garche, J. Lithium batteries: Status, prospects and future. J. Power Sources 2010, 195, 2419–2430. [Google Scholar] [CrossRef]
- Goodenough, J.B.; Park, K.S. The Li-ion rechargeable battery: A perspective. J. Am. Chem. Soc. 2013, 135, 1167–1176. [Google Scholar] [CrossRef] [PubMed]
- Winter, M.; Besenhard, J.O.; Spahr, M.E.; Novák, P. Insertion electrode materials for rechargeable lithium batteries. Adv. Mater. 1998, 10, 725–723. [Google Scholar] [CrossRef]
- Ji, L.; Lin, Z.; Alcoutlabi, M.; Zhang, X. Recent developments in nanostructured anode materials for rechargeable lithium-ion batteries. Energy Environ. Sci. 2011, 4, 2682–2699. [Google Scholar] [CrossRef]
- Fang, B.; Kim, M.S.; Kim, J.H.; Lim, S.; Yu, J.S. Ordered multimodal porous carbon with hierarchical nanostructure for high Li storage capacity and good cycling performance. J. Mater. Chem. 2010, 20, 10253–10259. [Google Scholar] [CrossRef]
- Roberts, A.D.; Li, X.; Zhang, H. Porous carbon spheres and monoliths: Morphology control, pore size tuning and their applications as Li-ion battery anode materials. Chem. Soc. Rev. 2014, 43, 4341–4356. [Google Scholar] [CrossRef] [PubMed]
- Tatsumi, K.; Iwashita, N.; Sakaebe, H.; Shioyama, H.; Higuchi, S.; Mabuchi, A.; Fujimoto, H. The influence of the graphitic structure on the electrochemical characteristics for the anode of secondary lithium batteries. J. Electrochem. Soc. 1995, 142, 716–720. [Google Scholar] [CrossRef]
- Nishihara, H.; Kyotani, T. Templated nanocarbons for energy storage. Adv. Mater. 2012, 24, 4473–4498. [Google Scholar] [CrossRef] [PubMed]
- Simon, P.F.W.; Ulrich, R.; Spiess, H.W.; Wiesner, U. Block copoplymer-ceramic hybrid materials from organically modified ceramic precursors. Chem. Mater. 2001, 13, 3464–3486. [Google Scholar] [CrossRef]
- Zhou, H.; Zhu, S.; Hibino, M.; Honma, I.; Ichihara, M. Lithium storage in ordered mesoporous carbon (CMK-3) with high reversible specific energy capacity and good cycling performance. Adv. Mater. 2003, 15, 2107–2111. [Google Scholar] [CrossRef]
- Tirado, J.L. Inorganic materials for the negative electrode of lithium-ion batteries: State-of-the-art and future prospects. Mater. Sci. Eng. 2003, 40, 103–136. [Google Scholar] [CrossRef]
- Kaskhedikar, N.A.; Maier, J. Lithium storage in carbon nanostructures. Adv. Mater. 2009, 21, 2664–2680. [Google Scholar] [CrossRef]
- Tokumitsu, K.; Fujimoto, H.; Mabuchi, A.; Kasuh, T. High capacity carbon anode for Li-ion battery a theoretical explanation. Carbon 1999, 37, 1599–1605. [Google Scholar] [CrossRef]
- Fu, L.J.; Liu, H.; Li, C.; Wu, Y.P.; Rahm, E.; Holze, R.; Wu, H.Q. Surface modifications of electrode materials for lithium ion batteries. Solid State Sci. 2006, 8, 113–128. [Google Scholar] [CrossRef]
- Niu, D.; Ma, Z.; Li, Y.; Shi, J. Synthesis of core-shell structured dual-mesoporous silica spheres with tunable pore size and controllable shell thickness. J. Am. Chem. Soc. 2010, 132, 15144–15147. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.; Gu, D.; Zou, Y.; Wu, Z.; Li, F.; Che, R.; Deng, Y.; Tu, B.; Zhao, D. A low-concentration hydrothermal synthesis of biocompatible ordered mesoporous carbon nanospheres with tunable and uniform size. Angew. Chem. Int. Ed. 2010, 49, 7987–7991. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.; Lv, Y.; Che, R.; Wu, H.; Zhang, X.; Gu, D.; Zheng, G.; Zhao, D. Two-dimensional mesoporous carbon nanosheets and their derived graphene nanosheets: Synthesis and efficient lithium ion storage. J. Am. Chem. Soc. 2013, 135, 1524–1530. [Google Scholar] [CrossRef] [PubMed]
- Meng, Y.; Gu, D.; Zhang, F.; Shi, Y.; Yang, H.; Li, Z.; Yu, C.; Tu, B.; Zhao, D. Ordered mesoporous polymers and homologous carbon frameworks: Amphiphilic surfactant templating and direct transformation. Angew. Chem. 2005, 117, 7215–7221. [Google Scholar] [CrossRef]
- Wan, Y.; Shia, Y.; Zhao, D. Designed synthesis of mesoporous solids via nonionic-surfactant-templating approach. Chem. Commun. 2007, 9, 897–926. [Google Scholar] [CrossRef] [PubMed]
- Qiao, Z.A.; Guo, B.; Binder, A.J.; Chen, J.; Veith, G.M.; Dai, S. Controlled synthesis of mesoporous carbon nanostructures via a “silica-assisted” strategy. Nano Lett. 2013, 13, 207–212. [Google Scholar] [CrossRef] [PubMed]
- Park, J.; Joo, J.; Kwon, S.G.; Jang, Y.; Hyeon, T. Synthesis of monodisperse spherical nanocrystals. Angew. Chem. Int. Ed. 2007, 46, 4630–4660. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.J.; Cui, W.J.; Jin, L.H.; Wang, C.X.; Xia, Y.Y. Preparation of three-dimensional ordered mesoporous carbon sphere arrays by a two-step templating route and their application for supercapacitors. J. Mater. Chem. 2009, 19, 3661–3667. [Google Scholar] [CrossRef]
- Yan, Y.; Zhang, F.; Meng, Y.; Tu, B.; Zhao, D. One-step synthesis of ordered mesoporous carbonaceous spheres by an aerosol-assisted self-assembly. Chem. Commun. 2007, 27, 2867–2869. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Cai, H.; Yu, H.; Sun, X.; Tu, B.; Zhao, D.Y. Highly ordered mesoporous carbonaceous frameworks from a template of a mixed amphiphilic triblock-copolymer system of PEO–PPO–PEO and reverse PPO–PEO–PPO. Chem. Asian J. 2007, 2, 1282–1289. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.S.; Bhattacharjya, D.; Fang, B.Z.; Yang, D.S.; Bae, T.S.; Yu, J.S. Morphology-dependent Li storage performance of ordered mesoporous carbon as anode material. Langmuir 2013, 29, 6754–6761. [Google Scholar] [CrossRef] [PubMed]
- Su, D.S.; Schlögl, R. Nanostructured carbon and carbon nanocomposites for electrochemical energy storage applications. ChemSusChem 2010, 3, 136–168. [Google Scholar] [CrossRef] [PubMed]
- Maiyalagan, T.; Nassr, A.B.A.; Alaje, T.O.; Bron, M.; Scott, K. Three-dimensional cubic ordered mesoporous carbon (CMK-8) as highly efficient stable Pd electro-catalyst support for formic acid oxidation. J. Power Sources 2012, 211, 147–153. [Google Scholar] [CrossRef]
- Maiyalagan, T.; Alaje, T.O.; Scott, K. Highly stable Pt–Ru nanoparticles supported on three-dimensional cubic ordered mesoporous carbon (Pt–Ru/CMK-8) as promising electrocatalysts for methanol oxidation. J. Phys. Chem. B 2012, 116, 2630–2638. [Google Scholar] [CrossRef]
- Su, F.; Zhao, X.S.; Wang, Y.; Zeng, J.; Zhou, Z.; Lee, J.Y. Synthesis of graphite ordered macroporous carbon with a three-dimensional interconnected pore structure for electrochemical applications. J. Phys. Chem. B 2005, 109, 20200–20206. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Fu, Z.Y.; Su, B.L. Hierarchically structured porous materials for energy conversion and storage. Adv. Funct. Mater. 2012, 22, 4634–4667. [Google Scholar] [CrossRef]
- Brownson, D.A.C.; Kampouris, D.K.; Banks, C.E. An overview of graphene in energy production and storage applications. J. Power Sources 2011, 196, 4873–4885. [Google Scholar] [CrossRef]
- Bae, S.H.; Karthikeyan, K.; Lee, Y.S.; Oh, I.K. Microwave self-assembly of 3D graphene-carbon nanotube-nickel nanostructure for high capacity anode material in lithium ion battery. Carbon 2013, 64, 527–536. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chang, P.-Y.; Bindumadhavan, K.; Doong, R.-A. Size Effect of Ordered Mesoporous Carbon Nanospheres for Anodes in Li-Ion Battery. Nanomaterials 2015, 5, 2348-2358. https://doi.org/10.3390/nano5042348
Chang P-Y, Bindumadhavan K, Doong R-A. Size Effect of Ordered Mesoporous Carbon Nanospheres for Anodes in Li-Ion Battery. Nanomaterials. 2015; 5(4):2348-2358. https://doi.org/10.3390/nano5042348
Chicago/Turabian StyleChang, Pei-Yi, Kartick Bindumadhavan, and Ruey-An Doong. 2015. "Size Effect of Ordered Mesoporous Carbon Nanospheres for Anodes in Li-Ion Battery" Nanomaterials 5, no. 4: 2348-2358. https://doi.org/10.3390/nano5042348
APA StyleChang, P.-Y., Bindumadhavan, K., & Doong, R.-A. (2015). Size Effect of Ordered Mesoporous Carbon Nanospheres for Anodes in Li-Ion Battery. Nanomaterials, 5(4), 2348-2358. https://doi.org/10.3390/nano5042348





