Metal Oxide Nanomaterial QNAR Models: Available Structural Descriptors and Understanding of Toxicity Mechanisms
Abstract
:1. Introduction
| Title | Summary | Ref |
|---|---|---|
| No time to lose—high throughput screening to assess nanomaterial safety | This review aims to provide a comprehensive introduction to the high throughput/content screening methodology employed for safety assessment of engineered nanomaterials, including data analysis and prediction of potentially hazardous material properties. | [3] |
| Exploring QNAR modeling as a tool for predicting biological effects of manufactured nanoparticles | The review discusses major approaches for model building and validation using both experimental and computed properties of nanomaterials by considering two different categories of nanomaterials datasets:(i) those comprising nanomaterials with diverse metal cores and organic decorations;(ii) those involving nanomaterials possessing the same core. | [20] |
| Predictive models for nanotoxicology: Current challenges and future opportunities | The review aims to provide researchers strategies for directing research towards predictive models and the ancillary benefits of such research. | [19] |
| Applying quantitative structure-activity relationship approaches to nanotoxicology: Current status and future potential | The purpose of this review is to provide a summary of recent key advances in the field of QNAR modelling, to identify the major gaps in research required to accelerate the use of QSAR methods, and to provide a road map for future research needed to achieve QSAR models useful for regulatory purposes. | [26] |
| Advancing risk assessment of engineered nanomaterials: Application of computational approaches | The purpose of this review is to present the current state of knowledge related to the risks of the engineered nanoparticles and to assess the potential of efficient expansion and development of new approaches, which are offered by application of theoretical and computational methods, applicable for evaluation of nanomaterials. | [27] |
| Nano(Q)SAR: Challenges, pitfalls and perspectives | This article aims to identify some of the pitfalls and challenges associated with (Q)NARs. Three major barriers were identified: the need to improve quality of experimental data in which the models are developed from, the need to have practical guidelines for the development of the (Q)NAR models and the need to standardise and harmonise activities for the purpose of regulation. | [28] |
2. Experimental Descriptors
2.1. Morphological Structural Properties
2.2. Physicochemical Properties
3. Theoretical Descriptors
3.1. Constitutional Properties
3.2. Electronic Properties
| Structural descriptors | Ref |
|---|---|
| Cation charges | Hu et al. [6] |
| The absolute electronegativity of the metal and of the metal oxide, the molar heat capacity and average of the alpha and beta LUMO | Pathakoti et al. [37] |
| Metal electronegativity, the charge of the metal cation, atomic number, valence electron number of the metal | Kar et al. [39] |
| Standard enthalpy of formation of metal oxide nanocluster, Mulliken’s electronegativity | Gajewicz et al. [40] |
| The enthalpy of formation of a gaseous cation with the same oxidation state as that in the metal-oxide structure | Puzyn et al. [41] |
4. Other Novel Descriptors
4.1. Liquid Drop Model
4.2. QSAR-Perturbation Approach Based Descriptors
4.3. Optimal SMILE-Based Descriptor
| Descriptors type | Advantages | Disadvantages |
|---|---|---|
| morphological structural properties | directly relate to the characteristics of metal oxide nanomaterials, easy to explain the toxicity mechanism | measuring error, some of the properties are difficult to quantitate |
| physicochemical properties | directly relate to characteristics of metal oxide nanomaterials, easy to explain the toxicity mechanism | measuring error |
| constitutional properties | easy to obtain | characteristics of metal oxide nanomaterial are not included |
| electronic properties or thermodynamic properties | easy to obtain , easy to explain the toxicity mechanism | the calculation system is relatively small |
| novel descriptors | directly relate to the characteristics of metal oxide nanomaterials, easy to explain the toxicity mechanism | the calculation method is complex |
5. Understanding Toxicity Mechanism(s) from Existing QNAR Models
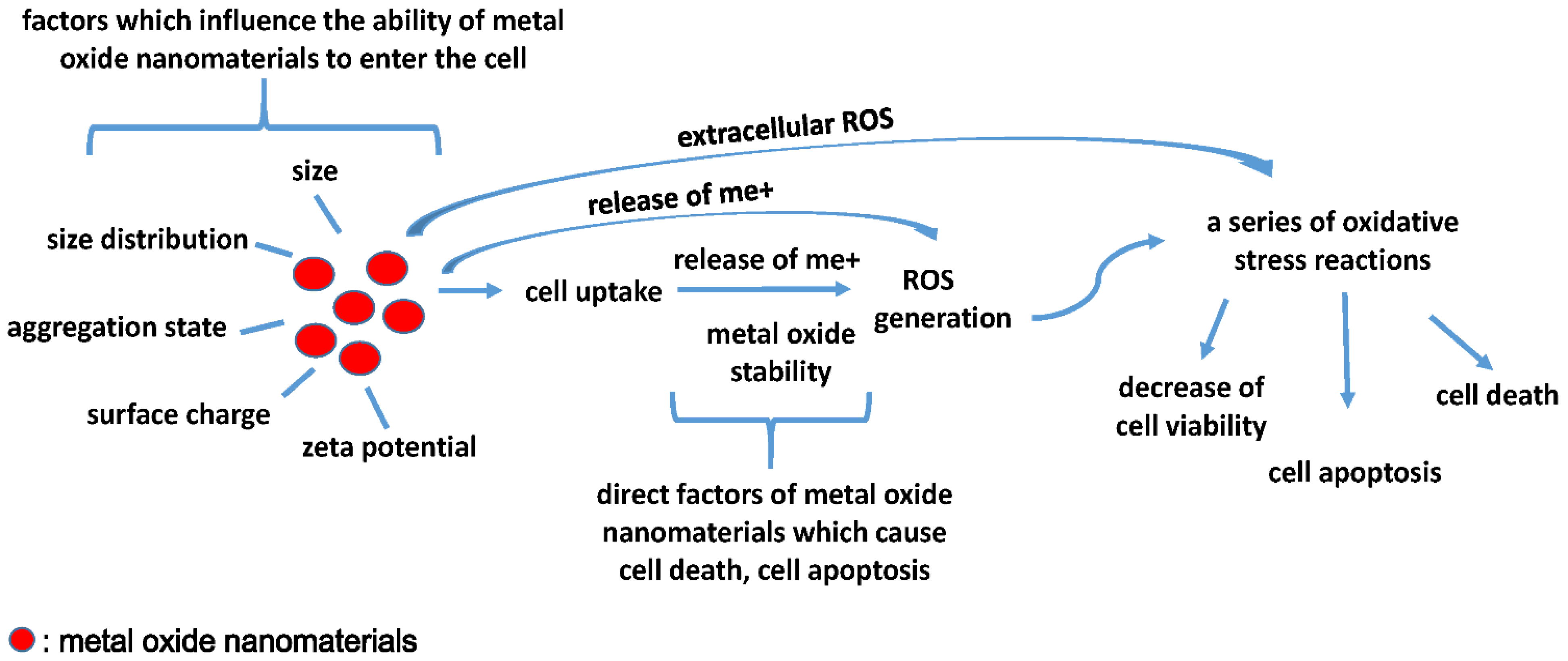
6. Conclusions and Outlook
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Fernández-Garcia, M.; Rodriguez, J.A. Encyclopedia of Inorganic and Bioinorganic Chemistry: Metal Oxide Nanoparticles; John Wiley & Sons, Ltd: New Yor, NY, USA, 2011; pp. 1–3. [Google Scholar]
- Teow, Y.; Asharani, P.V.; Hande, M.P.; Valiyaveettil, S. Health impact and safety of engineered nanomaterials. Chem. Commun. 2011, 47, 7025–7038. [Google Scholar] [CrossRef] [PubMed]
- Damoiseaux, R.; George, S.; Li, M.; Pokhrel, S.; Ji, Z.; France, B.; Xia, T.; Suarez, E.; Rallo, R.; Madler, L.; et al. No time to lose—High throughput screening to assess nanomaterial safety. Nanoscale 2011, 3, 1345–1360. [Google Scholar] [CrossRef]
- Gottschalk, F.; Nowack, B. The release of engineered nanomaterials to the environment. J. Exp. Med. 2011, 13, 1145–1155. [Google Scholar] [CrossRef] [PubMed]
- Heinlaan, M.; Ivask, A.; Blinova, I.; Dubourguier, H.C.; Kahru, A. Toxicity of nanosized and bulk ZnO, CuO and TiO2 to bacteria Vibrio fischeri and crustaceans Daphnia magna and Thamnocephalus platyurus. Chemosphere 2008, 71, 1308–1316. [Google Scholar] [CrossRef]
- Hu, X.; Cook, S.; Wang, P.; Hwang, H.M. In vitro evaluation of cytotoxicity of engineered metal oxide nanoparticles. The Sci. Total Environ. 2009, 407, 3070–3072. [Google Scholar] [CrossRef]
- Mahmoudi, M.; Hofmann, H.; Rothen-Rutishauser, B.; Petri-Fink, A. Assessing the in vitro and in vivo toxicity of superparamagnetic iron oxide nanoparticles. Chem. Rev. 2012, 112, 2323–2338. [Google Scholar] [CrossRef]
- Zuzana, M.; Alessandra, R.; Lise, F.; Maria, D. Safety assessment of nanoparticles cytotoxicity and genotoxicity of metal nanoparticles in vitro. J. Biomed. Nanotechnol. 2011, 7, 20–21. [Google Scholar] [CrossRef] [PubMed]
- Mahmoudi, M.; Simchi, A.; Milani, A.S.; Stroeve, P. Cell toxicity of superparamagnetic iron oxide nanoparticles. J. Colloid. Interf. Sci. 2009, 336, 510–518. [Google Scholar] [CrossRef] [PubMed]
- Karlsson, H.L.; Gustafsson, J.; Cronholm, P.; Moller, L. Size-dependent toxicity of metal oxide particles—A comparison between nano- and micrometer size. Toxicol. Lett. 2009, 188, 112–118. [Google Scholar] [CrossRef]
- Park, E.J.; Choi, J.; Park, Y.K.; Park, K. Oxidative stress induced by cerium oxide nanoparticles in cultured BEAS-2B cells. Toxicology 2008, 245, 90–100. [Google Scholar] [CrossRef] [PubMed]
- Lundqvist, M.; Stigler, J.; Elia, G.; Lynch, I.; Cedervall, T.; Dawson, K.A. Nanoparticle size and surface properties determine the protein corona with possible implications for biological impacts. Proc. Natl. Acad. Sci. USA 2008, 105, 14265–14270. [Google Scholar] [CrossRef] [PubMed]
- Hanna, L.; Karlsson, P.C.; Johanna, G.; Lennart, M. Copper oxide nanoparticles are highly toxic: A comparison between metal oxide nanoparticles and carbon nanotubes. Chem. Res. Toxicol. 2008, 21, 1726–1732. [Google Scholar]
- Wang, J.; Zhou, G.; Chen, C.; Yu, H.; Wang, T.; Ma, Y.; Jia, G.; Gao, Y.; Li, B.; Sun, J.; et al. Acute toxicity and biodistribution of different sized titanium dioxide particles in mice after oral administration. Toxicol. Lett. 2007, 168, 176–185. [Google Scholar] [CrossRef]
- Warheit, D.B.; Webb, T.R.; Sayes, C.M.; Colvin, V.L.; Reed, K.L. Pulmonary instillation studies with nanoscale TiO2 rods and dots in rats: Toxicity is not dependent upon particle size and surface area. Toxicol. Sci. 2006, 91, 227–236. [Google Scholar] [CrossRef] [PubMed]
- Jeng, H.A.; Swanson, J. Toxicity of metal oxide nanoparticles in mammalian cells. J. Environ. Sci. Health A 2006, 41, 2699–2711. [Google Scholar] [CrossRef]
- Hussain, S.M.; Hess, K.L.; Gearhart, J.M.; Geiss, K.T.; Schlager, J.J. In vitro toxicity of nanoparticles in BRL 3A rat liver cells. Toxicol. In Vitro 2005, 19, 975–983. [Google Scholar] [CrossRef] [PubMed]
- Burello, E.; Worth, A. Computational nanotoxicology: Predicting toxicity of nanoparticles. Nat. Nanotechnol. 2011, 6, 138–139. [Google Scholar] [CrossRef]
- Clark, K.A.; White, R.H.; Silbergeld, E.K. Predictive models for nanotoxicology: Current challenges and future opportunities. Regul. Toxicol. Pharm. 2011, 59, 361–363. [Google Scholar] [CrossRef]
- Fourches, D.; Pu, D.; Tropsha, A. Exploring quantitative nanostructure-activity relationships (QNAR) modeling as a tool for predicting biological effects of manufactured nanoparticles. Comb. Chem. High Throughput Screen. 2011, 14, 217–225. [Google Scholar] [CrossRef]
- Djurisic, A.B.; Leung, Y.H.; Ng, A.M.; Xu, X.Y.; Lee, P.K.; Degger, N.; Wu, R.S. Toxicity of metal oxide nanoparticles: Mechanisms, characterization, and avoiding experimental artefacts. Small 2015, 11, 26–44. [Google Scholar] [CrossRef]
- Landsiedel, R.; Ma-Hock, L.; Kroll, A.; Hahn, D.; Schnekenburger, J.; Wiench, K.; Wohlleben, W. Testing metal-oxide nanomaterials for human safety. Adv. Mater. 2010, 22, 2601–2627. [Google Scholar] [CrossRef]
- Ma, W.J.; Ruys, A.J.; Mason, R.S.; Martin, P.J.; Bendavid, A.; Liu, Z.; Ionescu, M.; Zreiqat, H. DLC coatings: Effects of physical and chemical properties on biological response. Biomaterials 2007, 28, 1620–1628. [Google Scholar] [CrossRef]
- Kroll, A.; Dierker, C.; Rommel, C.; Hahn, D.; Wohlleben, W.; Schulze-Isfort, C.; Gobbert, C.; Voetz, M.; Hardinghaus, F.; Schnekenburger, J. Cytotoxicity screening of 23 engineered nanomaterials using a test matrix of ten cell lines and three different assays. Part. Fibre Toxicol. 2011, 8, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Lauerent, S.; Saei, A.A.; Behzdi, S.; Panahifar, A.; Mahmoudi, M. Superpa ramagnetic iron oxide nanoparticles for delivery of therapeutic agents: Opportunities and challenges. Expert Opin. Drug Deliv. 2014, 11, 1449–1470. [Google Scholar] [CrossRef]
- Winkler, D.A.; Mombelli, E.; Pietroiusti, A.; Tran, L.; Worth, A.; Fadeel, B.; McCall, M.J. Applying quantitative structure-activity relationship approaches to nanotoxicology: Current status and future potential. Toxicology 2013, 313, 15–23. [Google Scholar] [CrossRef] [PubMed]
- Gajewicz, A.; Rasulev, B.; Dinadayalane, T.C.; Urbaszek, P.; Puzyn, T.; Leszczynska, D.; Leszczynski, J. Advancing risk assessment of engineered nanomaterials: Application of computational approaches. Adv. Drug Deliv. Rev. 2012, 64, 1663–1693. [Google Scholar] [CrossRef] [PubMed]
- Tantra, R.; Oksel, C.; Puzyn, T.; Wang, J.; Robinson, K.N.; Wang, X.Z.; Ma, C.Y.; Wilkins, T. Nano(Q)SAR: Challenges, pitfalls and perspectives. Nanotoxicology 2015, 9, 636–642. [Google Scholar] [CrossRef]
- Karelson, M. The QSPR/QSAR Approach for the Prediction of Properties of Fullerence Derivatives. Ph.D. Thesis, University of Tartu, Estonia, 2011. [Google Scholar]
- Ronald, H.; White, M.S.T. Assessing Nanomaterial Risks: Current Challenges and Future Opportunities for Risk Assessment. In Proceedings of the American Chemistry Council-Long Range Research Initiative Workshop: Navigating Obstacles towards Modernizing Risk Assessment, Washington, DC, USA, 24 February 2010.
- Yuan, J.; Pu, Y.; Yin, L. Prediction of binding affinities of PCDDs, PCDFs and PCBs using docking-based comparative molecular similarity indices analysis. Environ. Toxicol. Pharmacol. 2014, 38, 1–7. [Google Scholar] [CrossRef]
- Yuan, J.; Pu, Y.; Yin, L. Docking-based three-dimensional quantitative structure-activity relationship (3D-QSAR) predicts binding affinities to aryl hydrocarbon receptor for polychlorinated dibenzodioxins, dibenzofurans, and biphenyls. Environ. Toxicol. Pharmacol. 2013, 32, 1453–1458. [Google Scholar] [CrossRef] [PubMed]
- Todeschini, R.; Conso, V. Handbook of Molecular Descriptors; Wiley-VCH: Weinheim, Germany, 2000; pp. 927–933. [Google Scholar]
- Toropova, A.P.; Toropov, A.A.; Benfenati, E.; Korenstein, R.; Leszczynska, D.; Leszczynski, J. Optimal nano-descriptors as translators of eclectic data into prediction of the cell membrane damage by means of nano metal-oxides. Environ Sci. Pollut. Res. 2015, 22, 745–757. [Google Scholar] [CrossRef]
- Liu, R.; Rallo, R.; Bilal, M.; Cohen, Y. Quantitative structure-activity relationships for cellular uptake of surface-modified nanoparticles. Comb. Chem. High Throughput Screen. 2015, 18, 365–375. [Google Scholar] [CrossRef] [PubMed]
- Sizochenko, N.; Rasulev, B.; Gajewicz, A.; Kuz'min, V.; Puzyn, T.; Leszczynski, J. From basic physics to mechanisms of toxicity: The “liquid drop” approach applied to develop predictive classification models for toxicity of metal oxide nanoparticles. Nanoscale 2014, 6, 13986–13993. [Google Scholar] [CrossRef]
- Pathakoti, K.; Huang, M.J.; Watts, J.D.; He, X.; Hwang, H.M. Using experimental data of Escherichia coli to develop a QSAR model for predicting the photo-induced cytotoxicity of metal oxide nanoparticles. J. Photochem. Photobiol. B 2014, 130, 234–240. [Google Scholar] [CrossRef]
- Luan, F.; Kleandrova, V.V.; Gonzalez-Diaz, H.; Ruso, J.M.; Melo, A.; Speck-Planche, A.; Cordeiro, M.N. Computer-aided nanotoxicology: Assessing cytotoxicity of nanoparticles under diverse experimental conditions by using a novel QSTR-perturbation approach. Nanoscale 2014, 6, 10623–10630. [Google Scholar] [CrossRef] [PubMed]
- Kar, S.; Gajewicz, A.; Puzyn, T.; Roy, K.; Leszczynski, J. Periodic table-based descriptors to encode cytotoxicity profile of metal oxide nanoparticles: A mechanistic QSTR approach. Ecotoxicol. Environ. Saf. 2014, 107, 162–169. [Google Scholar] [CrossRef]
- Gajewicz, A.; Schaeublin, N.; Rasulev, B.; Hussain, S.; Leszczynska, D.; Puzyn, T.; Leszczynski, J. Towards understanding mechanisms governing cytotoxicity of metal oxides nanoparticles: Hints from nano-QSAR studies. Nanotoxicology 2015, 9, 313–325. [Google Scholar] [CrossRef] [PubMed]
- Puzyn, T.; Rasulev, B.; Gajewicz, A.; Hu, X.; Dasari, T.P.; Michalkova, A.; Hwang, H.M.; Toropov, A.; Leszczynska, D.; Leszczynski, J. Using nano-QSAR to predict the cytotoxicity of metal oxide nanoparticles. Nat. Nanotechnol. 2011, 6, 175–178. [Google Scholar] [CrossRef]
- Shaw, B.J.; Handy, R.D. Physiological effects of nanoparticles on fish: A comparison of nanometals versus metal ions. Environ. Int. 2011, 37, 1083–1097. [Google Scholar] [CrossRef]
- Han, D.W.; Hong, S.C.; Lee, J.H.; Lee, J.; Kim, H.Y.; Park, J.Y.; Cho, J.; Lee, J. Subtle cytotoxicity and genotoxicity differences in superparamagnetic iron oxide nanoparticles coated with various functional groups. Int. J. Nanomed. 2011, 6, 3219–3231. [Google Scholar] [CrossRef]
- Jensen, H.; Perdersen, H.; Jørgensen, J.E.; Pedersen, J.S.; Joensen, K.D.; Iversen, S.B.; Søgaard, E.G. Determination of size distributions in nanosized powders by TEM, XRD, and SAXS. J. Exp. Nanosci. 2006, 1, 355–373. [Google Scholar] [CrossRef]
- Bootz, A.; Vogel, V.; Schubert, D.; Kreuter, J. Comparison of scanning electron microscopy, dynamic light scattering and analytical ultracentrifugation for the sizing of poly (butyl cyanoacrylate) nanoparticles. Eur. J. Pharm. Biopharm. 2004, 57, 369–375. [Google Scholar] [CrossRef]
- Jillavenkatesa, A.; Kelly, J.F. Nanopowder characterization: Challenges and future directions. J. Nanopart. Res. 2002, 4, 463–468. [Google Scholar] [CrossRef]
- Warheit, D.B.; Frame, S.R. Characterization and reclassification of titanium dioxide-related pulmonary lesions. J. Occup. Environ. Med. 2006, 48, 1308–1313. [Google Scholar] [CrossRef] [PubMed]
- Seto, T.; Kawakami, Y.; Suzuki, N.; Hirasawa, M.; Kano, S.; Aya, N.; Sasaki, S.; Shimura, H. Evaluation of morphology and size distribution of silicon and titanium oxide nanoparticles generated by laser ablation. J. Nanopart. Res. 2001, 3, 185–191. [Google Scholar] [CrossRef]
- Fisker, R.; Carstensen, J.M.; Hansen, M.F.; Bødker, F.; Mørup, S. Estimation of nanoparticle size distributions by image analysis. J. Nanopart. Res. 2000, 2, 267–277. [Google Scholar] [CrossRef]
- Chu, B.; Liu, T. Characterization of nanoparticles by scattering techniques. J. Nanopart. Res. 2000, 2, 29–41. [Google Scholar] [CrossRef]
- Itoh, Y.; Lenggoro, W.; Okuyama, K.; Mädler, L.; Pratsinis, S.E. Size tunable synthesis of highly crystalline BaTiO3 nanoparticles using salt-assisted spray pyrolysis. J. Nanopart. Res. 2003, 5, 191–198. [Google Scholar] [CrossRef]
- Harano, A.; Shimada, K.; Okubo, T.; Sadakata, M. Crystal phases of TiO2 ultrafine particles prepared by laser ablation of solid rods. J. Nanopart. Res. 2002, 4, 215–219. [Google Scholar] [CrossRef]
- Liu, R.; Rallo, R.; George, S.; Ji, Z.; Nair, S.; Nel, A.E.; Cohen, Y. Classification nanoSAR development for cytotoxicity of metal oxide nanoparticles. Small 2011, 7, 1118–1126. [Google Scholar] [CrossRef]
- Epa, V.C.; Burden, F.R.; Tassa, C.; Weissleder, R.; Shaw, S.; Winkler, D.A. Modeling biological activities of nanoparticles. Nano Lett. 2012, 12, 5808–5812. [Google Scholar] [CrossRef]
- Burello, E.; Worth, A.P. QSAR modeling of nanomaterials. WIREs Nanomed. Nanobiotechnol. 2011, 3, 298–306. [Google Scholar] [CrossRef] [PubMed]
- Singh, K.P.; Gupta, S. Nano-QSAR modeling for predicting biological activity of diverse nanomaterials. RSC Adv. 2014, 4, 13215–13230. [Google Scholar] [CrossRef]
- Wang, X.Z.; Yang, Y.; Li, R.; McGuinnes, C.; Adamson, J.; Megson, I.L.; Donaldson, K. Principal component and causal analysis of structural and acute in vitro toxicity data for nanoparticles. Nanotoxicology 2014, 8, 465–476. [Google Scholar] [CrossRef] [PubMed]
- Fourches, D.; Pu, D.; Tassa, C.; Weissleder, R.; Shaw, S.Y.; Mumper, R.J.; Tropsha, A. Quantitative nanostructure-activity relationship modeling. ACS Nano 2010, 4, 5703–5712. [Google Scholar] [CrossRef]
- Horie, M.; Fujita, K.; Kato, H.; Endoh, S.; Nishio, K.; Komaba, L.K.; Nakamura, A.; Miyauchi, A.; Kinugasa, S.; Hagihara, Y.; et al. Association of the physical and chemical properties and the cytotoxicity of metal oxide nanoparticles: Metal ion release, adsorption ability and specific surface area. Metallomics 2012, 4, 350–360. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Ji, Z.X.; Xia, T.; Meng, H.; Low-Kam, C.; Liu, R.; Pokhrel, S.; Lin, S.; Wang, X.; Liao, Y.P.; et al. Use of metal oxide nanoparticle band gap to develop a predictive paradigm for oxidation stress and acute pulmonary inflammation. ACS Nano 2012, 6, 4349–4368. [Google Scholar] [CrossRef]
- Tantra, R.; Cackett, A.; Peck, R.; Gohil, D.; Snowden, J. Measurement of redox potential in nanoecotoxicological investigations. J. Toxicol. 2012, 2012, 270651. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Duffin, R.; Tran, L.; Brown, D.; Stone, V.; Donaldson, K. Proinflammogenic effects of low-toxicity and metal nanoparticles in vivo and in vitro: Highlighting the role of particle surface area and surface reactivity. Inhal. Toxicol. 2007, 19, 849–856. [Google Scholar] [CrossRef]
- Cho, W.S.; Duffin, R.; Thielbeer, F.; Bradley, M.; Megson, I.L.; Macnee, W.; Poland, C.A.; Tran, C.L.; Donaldson, K. Zeta potential and solubility to toxic ions as mechanisms of lung inflammation caused by metal/metal oxide nanoparticles. Toxicol. Sci. 2012, 126, 469–477. [Google Scholar] [CrossRef]
- Puzyn, T.; Cronin, M.T.D.; Leszczynski, J. Recent advances in QSAR studies: Methods and applications. Cronin. Eds. 2010, 8, 383–410. [Google Scholar]
- Kukreja, L.; Barik, S.; Misra, P. Variable band gap ZnO nanostructures grown by pulsed laser deposition. J. Cryst. Growth 2004, 105, 531–535. [Google Scholar] [CrossRef]
- Qu, Z.W.; Kroe, G.J. Theoretical study of the electronic structure and stability of titanium dioxide clusters (TiO2)n with n = 1–9. J. Phys. Chem. B 2006, 110, 8998–9007. [Google Scholar] [CrossRef]
- Burello, E.; Worth, A.P. A theoretical framework for predicting the oxidative stress potential of oxide nanoparticles. Nanotoxicology 2011, 5, 228–235. [Google Scholar] [CrossRef]
- Gajewicz, A.; Puzyn, T.; Rasulev, B.; Leszczynska, D.; Leszczynski, J. Metal oxide nanoparticles: Size-dependence of quantum-mechanical properties. Nanosci. Nanotechnol. Asia 2011, 1, 53–58. [Google Scholar]
- Katritzky, A.R.; Kulshyn, O.V.; Stoyanova-Slavova, I.; Dobchev, D.A.; Kuanar, M.; Fara, D.C.; Karelson, M. Antimalarial activity: A QSAR modeling using CODESSA PRO software. Bioorg. Med. Chem. 2006, 14, 2333–2357. [Google Scholar] [CrossRef] [PubMed]
- Tetko, I.V. Computing chemistry on the web. Drug Discov. Today 2005, 10, 1497–1500. [Google Scholar] [CrossRef]
- Puzyn, T.; Rostkowski, P.; Swieczkowski, A.; Jedrusiak, A.; Falandysz, J. Prediction of environmental partition coecients and the henrys law constants for 135 congeners of chlorodibenzothiophene. Chemosphere 2006, 62, 1817–1828. [Google Scholar] [CrossRef] [PubMed]
- Homepage of Molecular Operating Environment (MOE): Chemical Computing Group. Available online: http://www.chemcomp.com (accessed on 1 October 2010).
- Liu, R.; Zhang, H.Y.; Ji, Z.X.; Rallo, R.; Xia, T.; Chang, C.H.; Nel, A.; Cohen, Y. Development of structure-activity relationship for metal oxide nanoparticles. Nanoscale 2013, 5, 5644–5653. [Google Scholar] [CrossRef] [PubMed]
- Toropov, A.A.; Toropova, A.P. Quasi-QSAR for mutagenic potential of multi-walled carbon-nanotubes. Chemosphere 2015, 124, 40–46. [Google Scholar] [CrossRef]
- Wang, S.S.; Cheng, Y.B.; Lin, Y.C.; Liaw, C.C.; Chang, J.Y.; Kuo, Y.H.; Shen, Y.C. Nitrogen-containing diterpenoids, sesquiterpenoids, and nor-diterpenoids from Cespitularia taeniata. Mar. Drugs 2015, 13, 5796–5814. [Google Scholar] [CrossRef]
- Toropova, A.P.; Toropov, A.A.; Benfenati, E.; Puzyn, T.; Leszczynska, D.; Leszczynski, J. Optimal descriptor as a translator of eclectic information into the prediction of membrane damage: The case of a group of ZnO and TiO2 nanoparticles. Ecotoxicol. Environ. Saf. 2014, 108, 203–209. [Google Scholar] [CrossRef] [PubMed]
- Toropova, A.P.; Toropov, A.A.; Rallo, R.; Leszczynska, D.; Leszczynski, J. Optimal descriptor as a translator of eclectic data into prediction of cytotoxicity for metal oxide nanoparticles under different conditions. Ecotoxicol. Environ. Saf. 2015, 112, 39–45. [Google Scholar] [CrossRef]
- Hamilton, R.F.; Wu, N.; Porter, D.; Buford, M.; Wolfarth, M.; Holian, A. Particle length-dependent titanium dioxide nanomaterials toxicity and bioactivity. Part. Fibre Toxicol. 2009, 6, 1–11. [Google Scholar] [CrossRef]
- Frohlich, E. The role of surface charge in cellular uptake and cytotoxicity of medical nanoparticles. Int. J. Nanomed. 2012, 7, 5577–5591. [Google Scholar] [CrossRef] [PubMed]
- Rogers, S.N. Toxicological Effects of Cerium Oxide Nanoparticle Aggregates on Caenorhabditis elegans. Master Thesis, Marshall University, Huntington, WV, USA, 2013. [Google Scholar]
- Arvizo, R.R.; Miranda, O.R.; Thompson, M.A.; Pabelick, C.M.; Bhattacharya, R.; Robertson, J.D.; Rotello, V.M.; Prakash, Y.S.; Mukherjee, P. Effect of nanoparticle surface charge at the plasma membrane and beyond. Nano Lett. 2010, 10, 2543–2548. [Google Scholar] [CrossRef] [PubMed]
- Walkey, C.D.; Chan, W.C. Understanding and controlling the interaction of nanomaterials with proteins in a physiological environment. Chem. Soc. Rev. 2012, 41, 2780–2799. [Google Scholar] [CrossRef]
- Raemy, D.O.; Grass, R.N.; Stark, W.J.; Schumacher, C.M.; Clift, M.J.D.; Gehr, P.; Rothen-Rutishauser, B. Effects of flame made zinc oxide particles in human lung cells—A comparison of aerosol and suspension exposures. Part. Fibre Toxicol. 2012, 9, 1–18. [Google Scholar] [CrossRef]
- Calderon-Aparicio, A.; Strasberg-Rieber, M.; Rieber, M. Disulfiram anti-cancer efficacy without copper overload is enhanced by extracellular H2O2 generation: Antagonism by tetrathiomolybdate. Oncotarget 2015, 6, 29771–29781. [Google Scholar]
- Ge, Y.; Zhang, Y.; Xia, J.; Ma, M.; He, S.; Nie, F.; Gu, N. Effect of surface charge and agglomerate degree of magnetic iron oxide nanoparticles on KB cellular uptake in vitro. Colloids Surf. B 2009, 73, 294–301. [Google Scholar] [CrossRef] [PubMed]
- Osaka, T.; Nakanishi, T.; Shanmugam, S.; Takahama, S.; Zhang, H. Effect of surface charge of magnetite nanoparticles on their internalization into breast cancer and umbilical vein endothelial cells. Colloids Surf. B 2009, 71, 325–330. [Google Scholar] [CrossRef] [PubMed]
- Bellanger, X.; Billard, P.; Schneider, R.; Balan, L.; Merlin, C. Stability and toxicity of ZnO quantum dots: Interplay between nanoparticles and bacteria. J. Hazard. Mater. 2015, 283, 110–116. [Google Scholar] [CrossRef]
- Kumar, V.; Kumari, A.; Guleria, P.; Yadav, S.K. Evaluating the toxicity of selected types of nanochemicals. Rev. Environ. Contam. Toxicol. 2012, 215, 39–121. [Google Scholar]
- Linkov, I.; Steevens, J.; Adlakha-Hutcheon, G.; Bennett, E.; Chappell, M.; Colvin, V.; Davis, J.M.; Davis, T.; Elder, A.; Foss Hansen, S.; et al. Emerging methods and tools for environmental risk assessment, decision-making, and policy for nanomaterials: Summary of NATO advanced research workshop. J. Nanopart. Res. 2009, 11, 513–527. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ying, J.; Zhang, T.; Tang, M. Metal Oxide Nanomaterial QNAR Models: Available Structural Descriptors and Understanding of Toxicity Mechanisms. Nanomaterials 2015, 5, 1620-1637. https://doi.org/10.3390/nano5041620
Ying J, Zhang T, Tang M. Metal Oxide Nanomaterial QNAR Models: Available Structural Descriptors and Understanding of Toxicity Mechanisms. Nanomaterials. 2015; 5(4):1620-1637. https://doi.org/10.3390/nano5041620
Chicago/Turabian StyleYing, Jiali, Ting Zhang, and Meng Tang. 2015. "Metal Oxide Nanomaterial QNAR Models: Available Structural Descriptors and Understanding of Toxicity Mechanisms" Nanomaterials 5, no. 4: 1620-1637. https://doi.org/10.3390/nano5041620
APA StyleYing, J., Zhang, T., & Tang, M. (2015). Metal Oxide Nanomaterial QNAR Models: Available Structural Descriptors and Understanding of Toxicity Mechanisms. Nanomaterials, 5(4), 1620-1637. https://doi.org/10.3390/nano5041620






