ZIF-8@Rhodamine B as a Self-Reporting Material for Pollutant Extraction Applications
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Instrumentation
2.3. Synthesis of ZIF-8
2.4. Determination of Rhodamine B-Loading in ZIF-8 via UV-Vis Spectrophotometry
2.5. Buffers and Salts
- (1)
- Phosphate buffer solution (PBS) 1.0 M. Sodium chloride (8 g, 0.137 mol L−1), potassium chloride (0.2 g, 2.7 mmol L−1), sodium hydrogen phosphate (1.77 g, 10 mmol L−1), and potassium dihydrogen phosphate (0.24 g, 1.8 mmol L−1), were dissolved in 800 mL of Milli-Q water. The solution was adjusted to pH 7.4 using NaOH (1 mol L−1) and diluted to a final volume of 1 L with Milli-Q water.
- (2)
- Phosphate buffer solution 0.1× (PBS 0.1×). 10 mL of PBS was diluted using Milli-Q water to a volume of 100 mL to obtain a 0.1 M solution of PBS.
- (3)
- Ethylenediaminetetraacetic acid (EDTA). EDTA (2.92 g, 0.1 M) was dissolved in 100 mL of Milli-Q water.
- (4)
- Sodium chloride (NaCl). A 1.0 M solution of sodium chloride was prepared by dissolving 5.844 g NaCl into 100 mL of distilled water.
2.6. Mixed Phenol Extraction
3. Results and Discussion
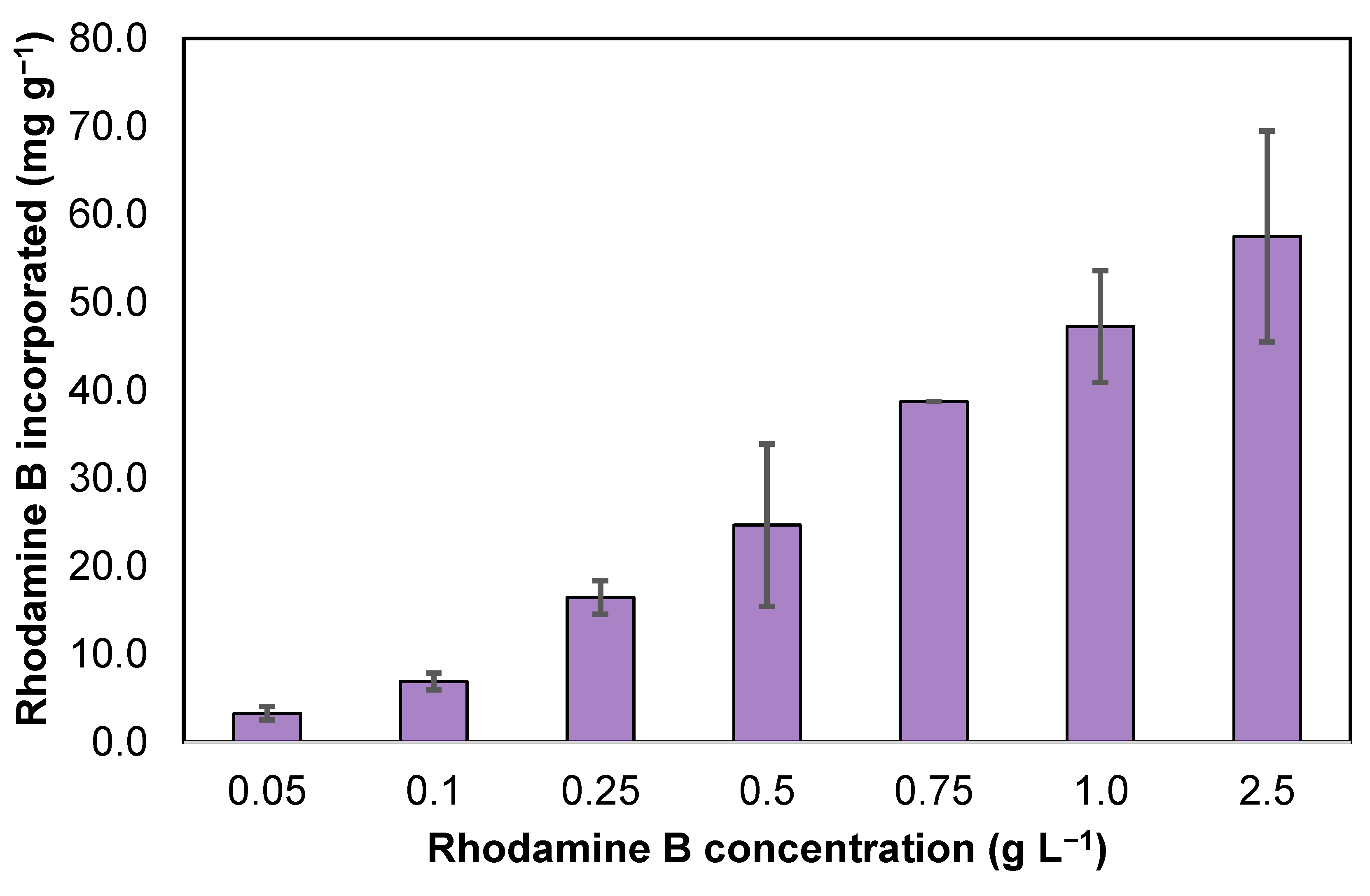
3.1. Screening of Rhodamine B Concentration in ZIF-8 Synthesis
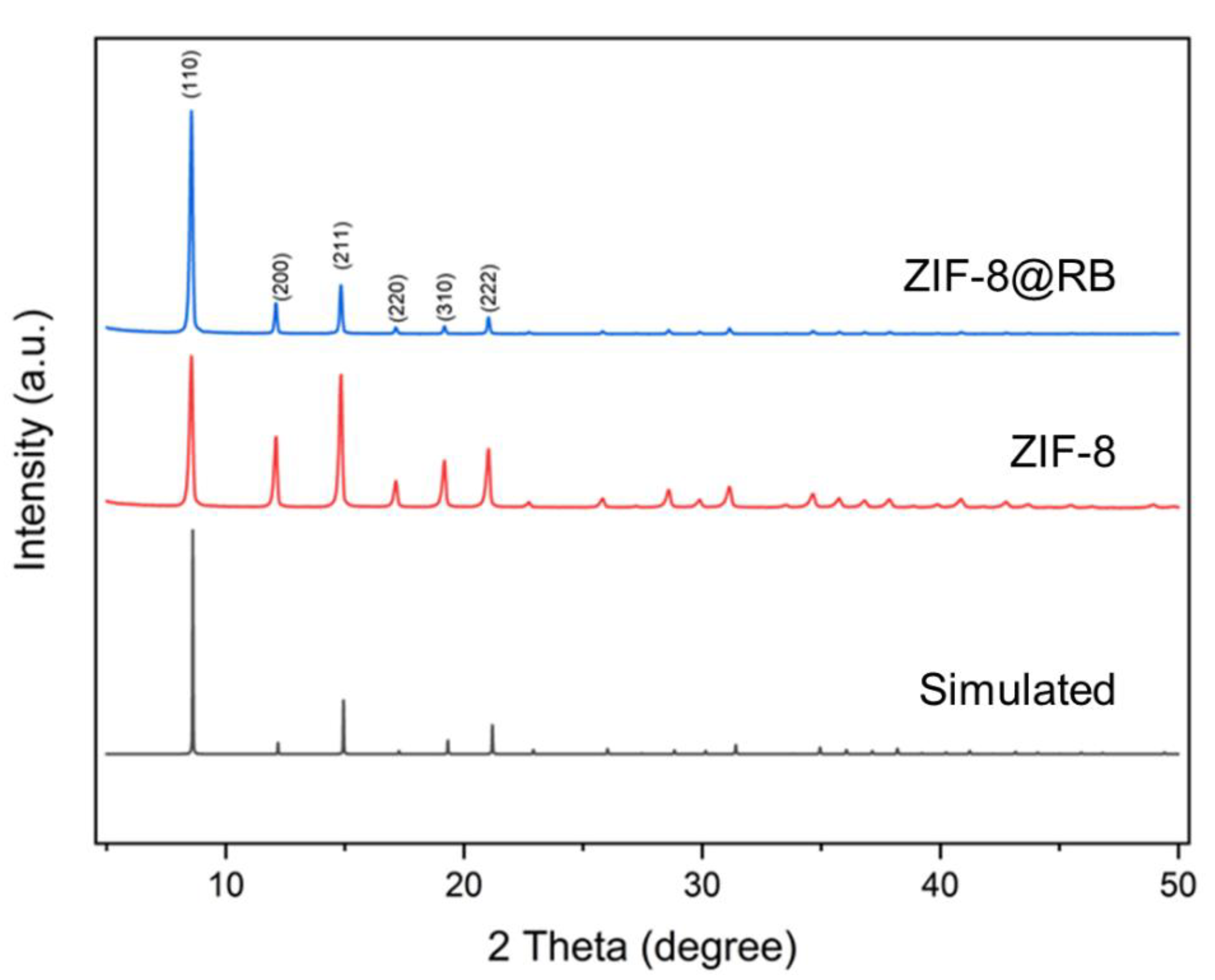
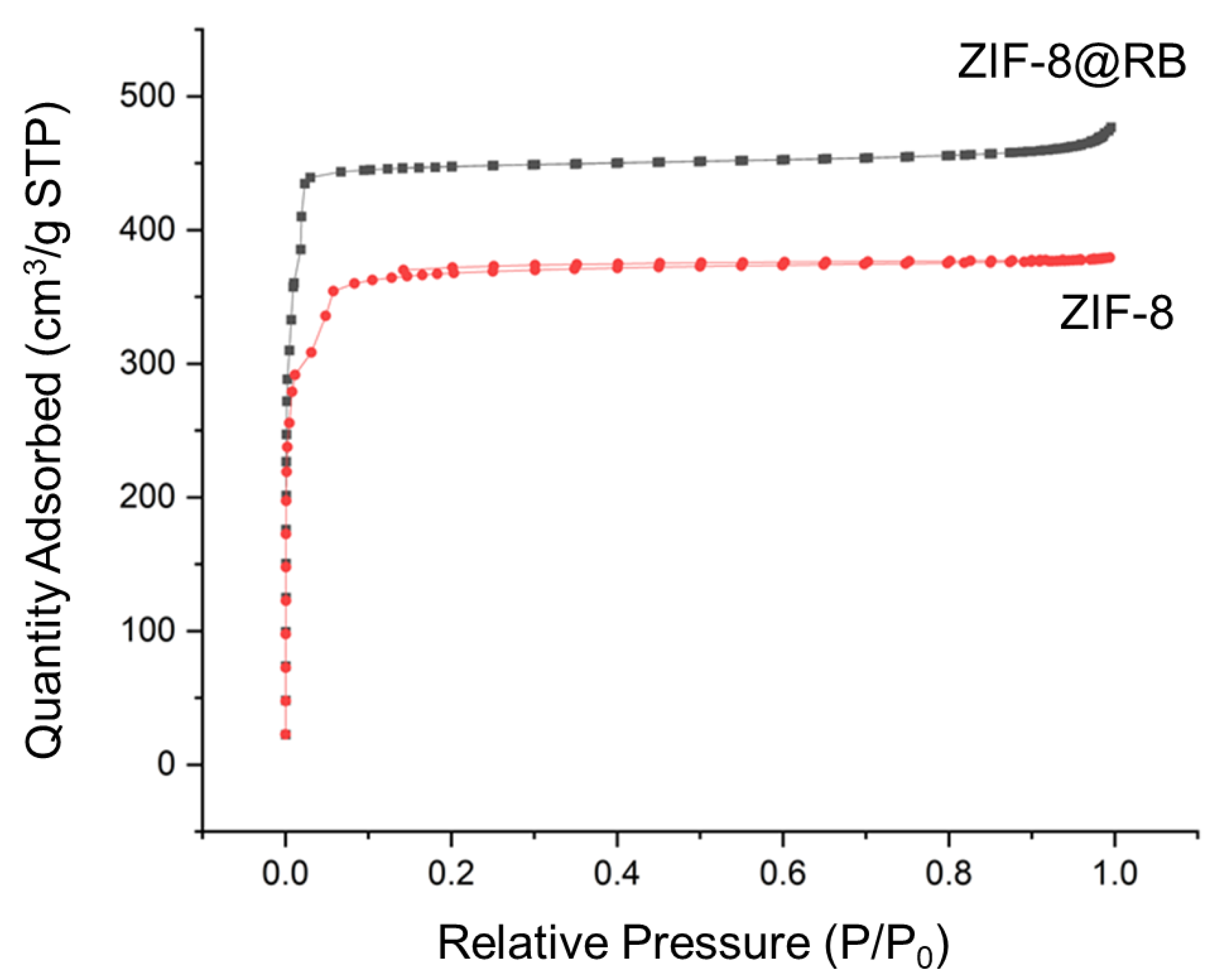
3.2. Characterization of ZIF-8@RB
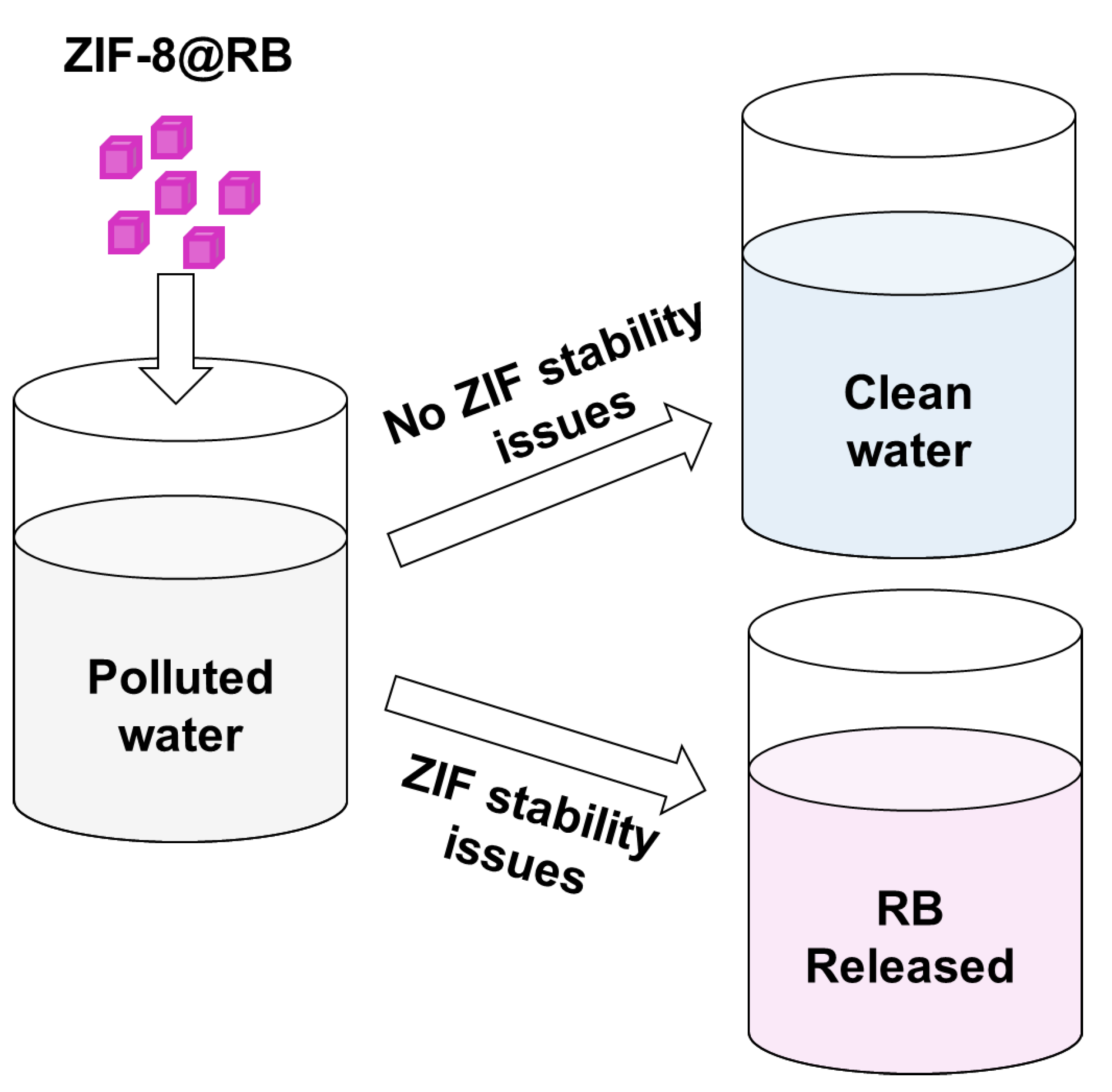
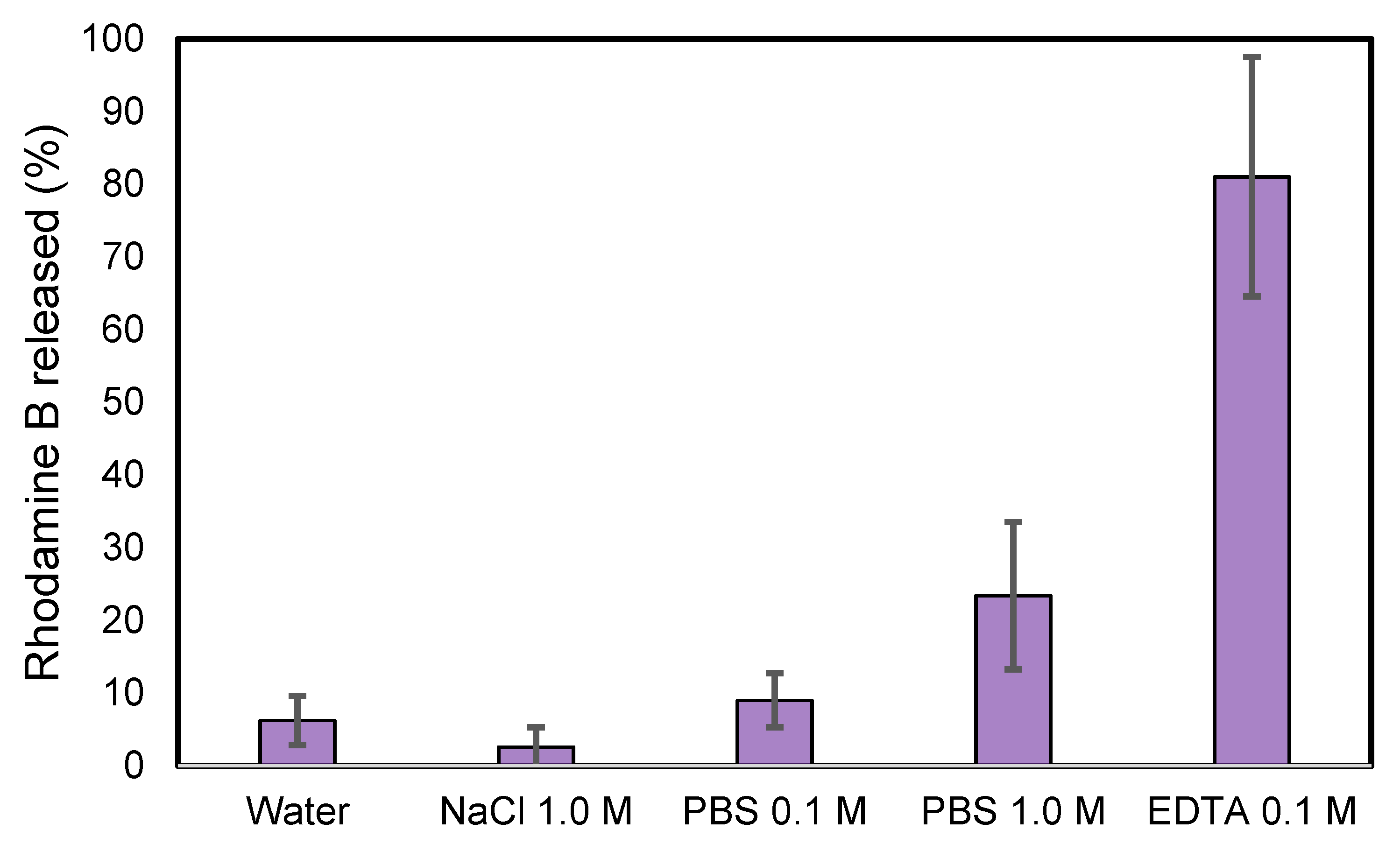
3.3. UV-Vis Spectrophotometry Monitoring of ZIF-8@RB Stability in Different Media
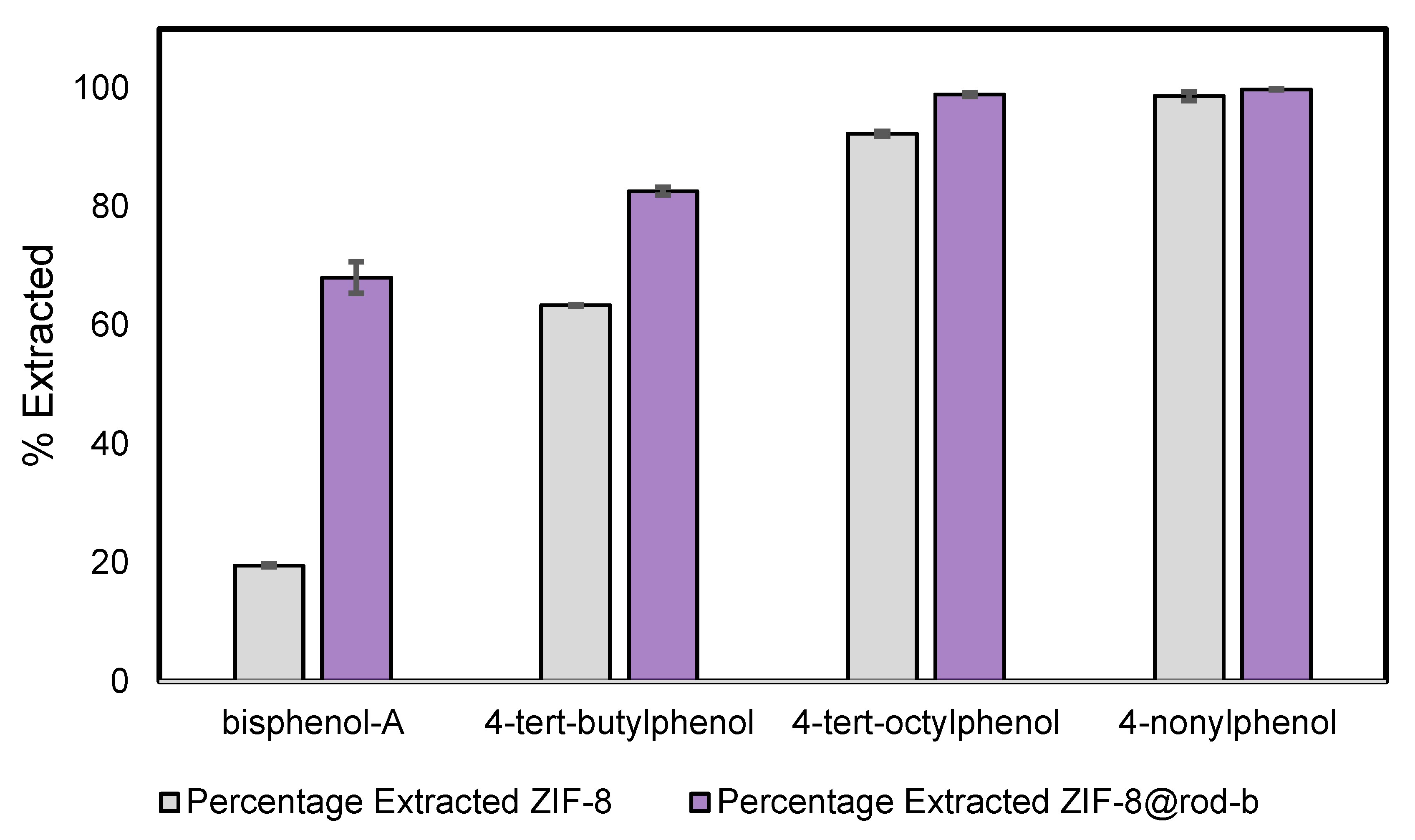
3.4. Simultaneous Extraction of Phenols from Water
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Li, H.; Eddaoudi, M.; O’Keeffe, M.; Yaghi, O.M. Design and synthesis of an exceptionally stable and highly porous metal-organic framework. Nature 1999, 402, 276–279. [Google Scholar] [CrossRef]
- Furukawa, H.; Cordova, K.E.; O’Keeffe, M.; Yaghi, O.M. The chemistry and applications of metal-organic frameworks. Science 2013, 341, 1230444. [Google Scholar] [CrossRef] [PubMed]
- Freund, R.; Zaremba, O.; Arnauts, G.; Ameloot, R.; Skorupskii, G.; Dincă, M.; Bavykina, A.; Gascon, J.; Ejsmont, A.; Goscianska, J.; et al. The current status of MOF and COF applications. Angew. Chem. Int. Ed. 2021, 60, 23975–24001. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Xiong, Y.; Liu, Y.; Han, J.; Duan, G.; Chen, Y.; He, S.; Mei, C.; Jiang, S.; Zhang, K. When MOFs meet wood: From opportunities toward applications. Chem 2022, 8, 2342–2361. [Google Scholar] [CrossRef]
- Zhu, L.; Liu, X.Q.; Jiang, H.L.; Sun, L.B. Metal–organic frameworks for heterogeneous basic catalysis. Chem. Rev. 2017, 117, 8129–8176. [Google Scholar] [CrossRef] [PubMed]
- Lázaro, I.A.; Forgan, R.S. Application of zirconium MOFs in drug delivery and biomedicine. Coord. Chem. Rev. 2019, 380, 230–259. [Google Scholar] [CrossRef]
- Zhang, X.; Chen, A.; Zhong, M.; Zhang, Z.; Zhang, X.; Zhou, Z.; Bu, X.H. Metal–organic frameworks (MOFs) and MOF-derived materials for energy storage and conversion. Electrochem. Energy Rev. 2019, 2, 29–104. [Google Scholar] [CrossRef]
- Ma, X.; Chai, Y.; Li, P.; Wang, B. Metal–organic framework films and their potential applications in environmental pollution control. ACC. Chem. Res. 2019, 52, 1461–1470. [Google Scholar] [CrossRef]
- Wen, Y.; Zhang, P.; Sharma, V.K.; Ma, X.; Zhou, H.-C. Metal-organic frameworks for environmental applications. Cell Rep. Phys. Sci. 2021, 2, 100348. [Google Scholar] [CrossRef]
- Rojas, S.; Horcajada, P. Metal–organic frameworks for the removal of emerging organic contaminants in water. Chem. Rev. 2020, 120, 8378–8415. [Google Scholar] [CrossRef]
- Huo, S.-H.; Yan, X.-P. Metal–organic framework MIL-100(Fe) for the adsorption of malachite green from aqueous solution. J. Mater. Chem. 2012, 22, 7449–7455. [Google Scholar] [CrossRef]
- Gao, C.; Chen, S.; Quan, X.; Yu, H.; Zhang, Y. Enhanced Fenton-like catalysis by iron-based metal organic frameworks for degradation of organic pollutants. J. Catal. 2017, 356, 125–132. [Google Scholar] [CrossRef]
- Figuerola, A.; Medina, D.A.; Santos-Neto, A.J.; Cabello, C.P.; Cerdà, V.; Palomino, G.T.; Maya, F. Metal–organic framework mixed-matrix coatings on 3D printed devices. Appl. Mater. Today 2019, 16, 21–27. [Google Scholar] [CrossRef]
- Park, K.S.; Ni, Z.; Côté, A.P.; Choi, J.Y.; Huang, R.; Uribe-Romo, F.J.; Chae, H.K.; O’Keeffe, M.; Yaghi, O.M. Exceptional chemical and thermal stability of zeolitic imidazolate frameworks. Proc. Natl. Acad. Sci. USA 2006, 103, 10186–10191. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.; Yang, Z.; Zhu, Y.; Xia, Y. Zeolitic imidazolate framework materials: Recent progress in synthesis and applications. J. Mater. Chem. A 2014, 2, 16811–16831. [Google Scholar] [CrossRef]
- Banerjee, R.; Phan, A.; Wang, B.; Knobler, C.; Furukawa, H.; O’Keeffe, M.; Yaghi, O.M. High-throughput synthesis of zeolitic imidazolate frameworks and application to CO2 capture. Science 2008, 319, 939–943. [Google Scholar] [CrossRef]
- Phan, A.; Doonan, C.J.; Uribe-Romo, F.J.; Knobler, C.B.; O’keeffe, M.; Yaghi, O.M. Synthesis, structure, and carbon dioxide capture properties of zeolitic imidazolate frameworks. ACC. Chem. Res. 2010, 43, 58–67. [Google Scholar] [CrossRef]
- Jian, S.; Ma, X.; Wang, Q.; Wu, J.; Wang, Y.; Jiang, S.; Xu, W.; Yang, W. Hierarchical porous Co3O4 nanocages with elaborate microstructures derived from ZIF-67 toward lithium storage. Vacuum 2021, 184, 109879. [Google Scholar] [CrossRef]
- Ma, X.; Guo, H.; Zhang, C.; Chen, D.; Tian, Z.; Wang, Y.; Chen, Y.; Wang, S.; Han, J.; Lou, Z.; et al. ZIF-67/wood derived self-supported carbon composites for electromagnetic interference shielding and sound and heat insulation. Inorg. Chem. Front. 2022, 9, 6305–6316. [Google Scholar] [CrossRef]
- Pan, Y.; Liu, Y.; Zeng, G.; Zhao, L.; Lai, Z. Rapid synthesis of zeolitic imidazolate framework-8 (ZIF-8) nanocrystals in an aqueous system. Chem. Commun. 2011, 47, 2071–2073. [Google Scholar] [CrossRef]
- Cliffe, M.J.; Mottillo, C.; Stein, R.S.; Bučar, D.-K.; Friščić, T. Accelerated aging: A low energy, solvent-free alternative to solvothermal and mechanochemical synthesis of metal–organic materials. Chem. Sci. 2012, 3, 2495–2500. [Google Scholar] [CrossRef]
- Liang, K.; Ricco, R.; Doherty, C.M.; Styles, M.J.; Bell, S.; Kirby, N.; Mudie, S.; Haylock, D.; Hill, A.J.; Doonan, C.J.; et al. Biomimetic mineralization of metal-organic frameworks as protective coatings for biomacromolecules. Nat. Commun. 2015, 6, 7240. [Google Scholar] [CrossRef] [PubMed]
- Astria, E.; Thonhofer, M.; Ricco, R.; Liang, W.; Chemelli, A.; Tarzia, A.; Alt, K.; Hagemeyer, C.E.; Rattenberger, J.; Schroettner, H.; et al. Carbohydrates@MOFs. Mater. Horiz. 2019, 6, 969–977. [Google Scholar] [CrossRef]
- Poddar, A.; Conesa, J.J.; Liang, K.; Dhakal, S.; Reineck, P.; Bryant, G.; Pereiro, E.; Ricco, R.; Amenitsch, H.; Doonan, C.; et al. Encapsulation, visualization and expression of genes with biomimetically mineralized zeolitic imidazolate framework-8 (ZIF-8). Small 2019, 15, 1902268. [Google Scholar] [CrossRef]
- Abdelhamid, H.N.; Huang, Z.; El-Zohry, A.M.; Zheng, H.; Zou, X. A fast and scalable approach for synthesis of hierarchical porous zeolitic imidazolate frameworks and one-pot encapsulation of target molecules. Inorg. Chem. 2017, 56, 9139–9146. [Google Scholar] [CrossRef]
- Liu, X.-Y.; Xing, K.; Li, Y.; Tsung, C.-K.; Li, J. Three models to encapsulate multicomponent dyes into nanocrystal pores: A new strategy for generating high-quality white light. J. Am. Chem. Soc. 2019, 141, 14807–14813. [Google Scholar] [CrossRef]
- Liu, Y.; Pang, H.; Wang, X.; Yu, S.; Chen, Z.; Zhang, P.; Chen, L.; Song, G.; Saleh Alharbi, N.; Omar Rabah, S.; et al. Zeolitic imidazolate framework-based nanomaterials for the capture of heavy metal ions and radionuclides: A review. Chem. Eng. J. 2021, 406, 127139. [Google Scholar] [CrossRef]
- Rodas, M.; Fikarová, K.; Pasanen, F.; Horstkotte, B.; Maya, F. Zeolitic imidazolate frameworks in analytical sample preparation. J. Sep. Sci. 2021, 44, 1203–1219. [Google Scholar] [CrossRef]
- Sun, Y.; Zhang, N.; Yue, Y.; Xiao, J.; Huang, X.; Ishag, A. Recent advances on the application of zeolitic imidazolate frameworks (ZIFs) in environmental remediation: A review. Environ. Sci. Nano 2022, 9, 4069–4092. [Google Scholar] [CrossRef]
- Jjagwe, J.; Olupot, P.W.; Menya, E.; Kalibbala, H.M. Synthesis and application of granular activated carbon from biomass waste materials for water treatment: A review. J. Bioresour. Bioprod. 2021, 6, 292–322. [Google Scholar] [CrossRef]
- Obey, G.; Adelaide, M.; Ramaraj, R. Biochar derived from non-customized matamba fruit shell as an adsorbent for wastewater treatment. J. Bioresour. Bioprod. 2022, 7, 109–115. [Google Scholar] [CrossRef]
- Velásquez-Hernández, M.d.J.; Ricco, R.; Carraro, F.; Limpoco, F.T.; Linares-Moreau, M.; Leitner, E.; Wiltsche, H.; Rattenberger, J.; Schröttner, H.; Frühwirt, P.; et al. Degradation of ZIF-8 in phosphate buffered saline media. CrystEngComm 2019, 21, 4538–4544. [Google Scholar] [CrossRef]
- Luzuriaga, M.A.; Benjamin, C.E.; Gaertner, M.W.; Lee, H.; Herbert, F.C.; Mallick, S.; Gassensmith, J.J. ZIF-8 degrades in cell media, serum, and some—But not all—Common laboratory buffers. Supramol. Chem. 2019, 31, 485–490. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Zhao, W.; Tsapatsis, M.; Stein, A. Three-dimensionally ordered macroporous mixed metal oxide as an indicator for monitoring the stability of ZIF-8. Chem. Mater. 2020, 32, 3850–3859. [Google Scholar] [CrossRef]
- Wu, M.; Pan, C.; Yang, M.; Xu, B.; Lei, X.; Ma, J.; Cai, L.; Chen, J. Chemical analysis of fish bile extracts for monitoring endocrine disrupting chemical exposure in water: Bisphenol A, alkylphenols, and norethindrone. Environ. Toxicol. Chem. 2016, 35, 182–190. [Google Scholar] [CrossRef] [PubMed]
- Pan, Y.; Heryadi, D.; Zhou, F.; Zhao, L.; Lestari, G.; Su, H.; Lai, Z. Tuning the crystal morphology and size of zeolitic imidazolate framework-8 in aqueous solution by surfactants. CrystEngComm 2011, 13, 6937–6940. [Google Scholar] [CrossRef]
- Careghini, A.; Mastorgio, A.F.; Saponaro, S.; Sezenna, E. Bisphenol A, nonylphenols, benzophenones, and benzotriazoles in soils, groundwater, surface water, sediments, and food: A review. Environ. Sci. Pollut. Res. 2015, 22, 5711–5741. [Google Scholar] [CrossRef]
- Zhu, S.; Khan, M.A.; Kameda, T.; Xu, H.; Wang, F.; Xia, M.; Yoshioka, T. New insights into the capture performance and mechanism of hazardous metals Cr3+ and Cd2+ onto an effective layered double hydroxide based material. J. Hazard. Mater. 2022, 426, 128062. [Google Scholar] [CrossRef]
- Zhu, S.; Chen, Y.; Khan, M.A.; Xu, H.; Wang, F.; Xia, M. In-depth study of heavy metal removal by an etidronic acid-functionalized layered double hydroxide. ACS Appl. Mater. Interfaces 2022, 14, 7450–7463. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moore, E.W.P.; Maya, F. ZIF-8@Rhodamine B as a Self-Reporting Material for Pollutant Extraction Applications. Nanomaterials 2023, 13, 842. https://doi.org/10.3390/nano13050842
Moore EWP, Maya F. ZIF-8@Rhodamine B as a Self-Reporting Material for Pollutant Extraction Applications. Nanomaterials. 2023; 13(5):842. https://doi.org/10.3390/nano13050842
Chicago/Turabian StyleMoore, Edward W. P., and Fernando Maya. 2023. "ZIF-8@Rhodamine B as a Self-Reporting Material for Pollutant Extraction Applications" Nanomaterials 13, no. 5: 842. https://doi.org/10.3390/nano13050842
APA StyleMoore, E. W. P., & Maya, F. (2023). ZIF-8@Rhodamine B as a Self-Reporting Material for Pollutant Extraction Applications. Nanomaterials, 13(5), 842. https://doi.org/10.3390/nano13050842





