Integration of Different Graphene Nanostructures with PDMS to Form Wearable Sensors
Abstract
:1. Introduction
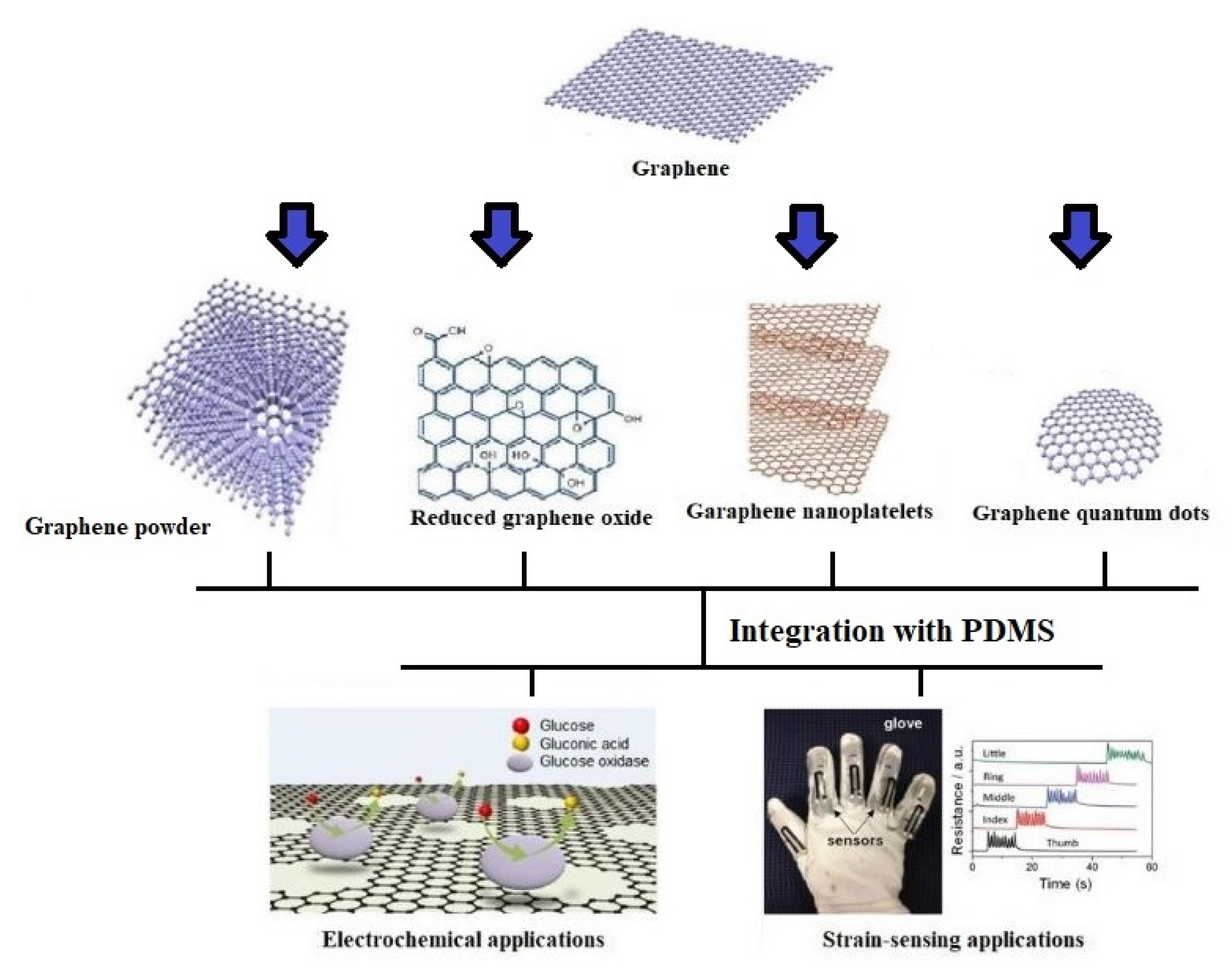
2. Graphene/PDMS-Based Sensors
2.1. Graphene Nanopowder-Based Devices
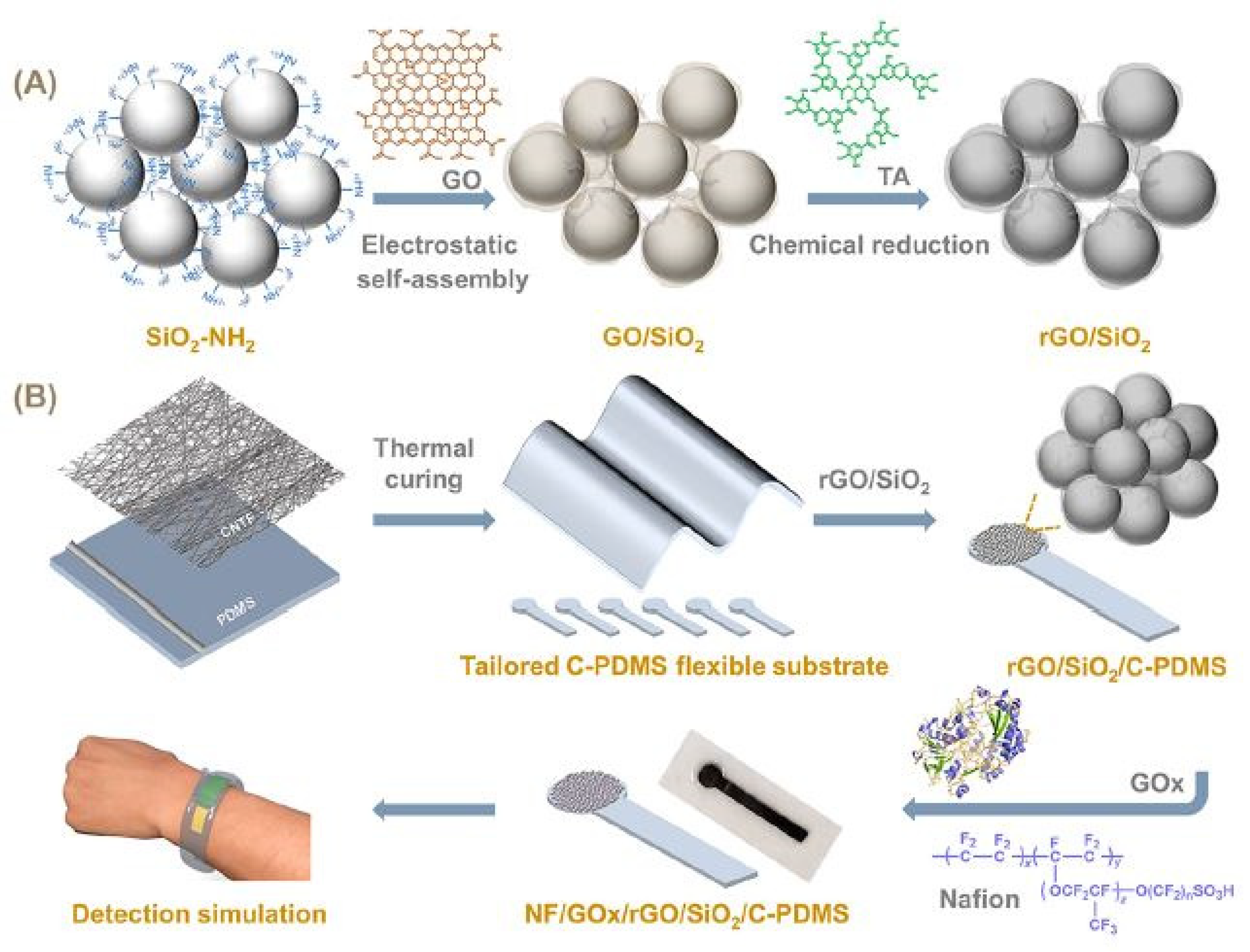
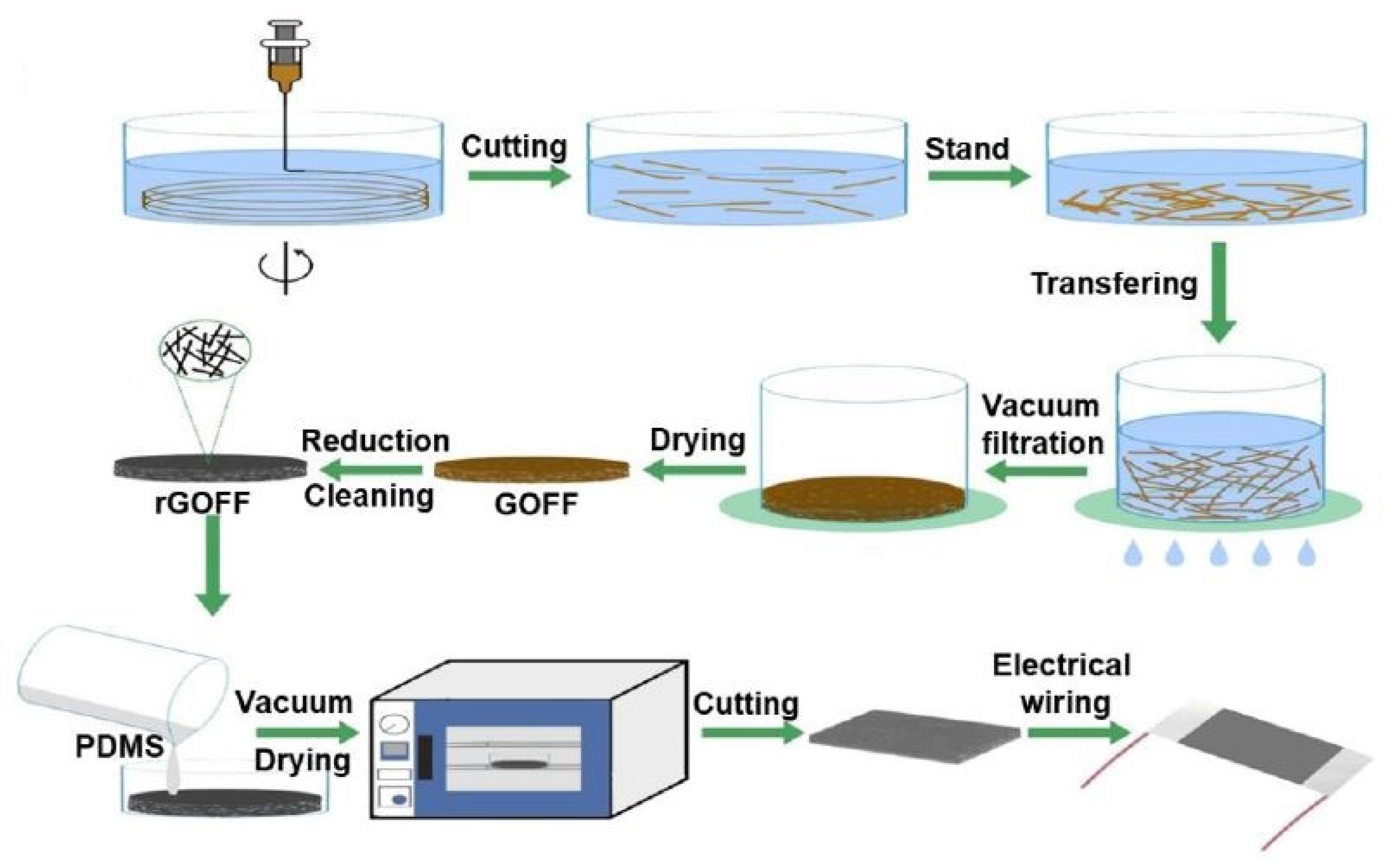
2.2. Reduced Graphene Oxide-Based Devices
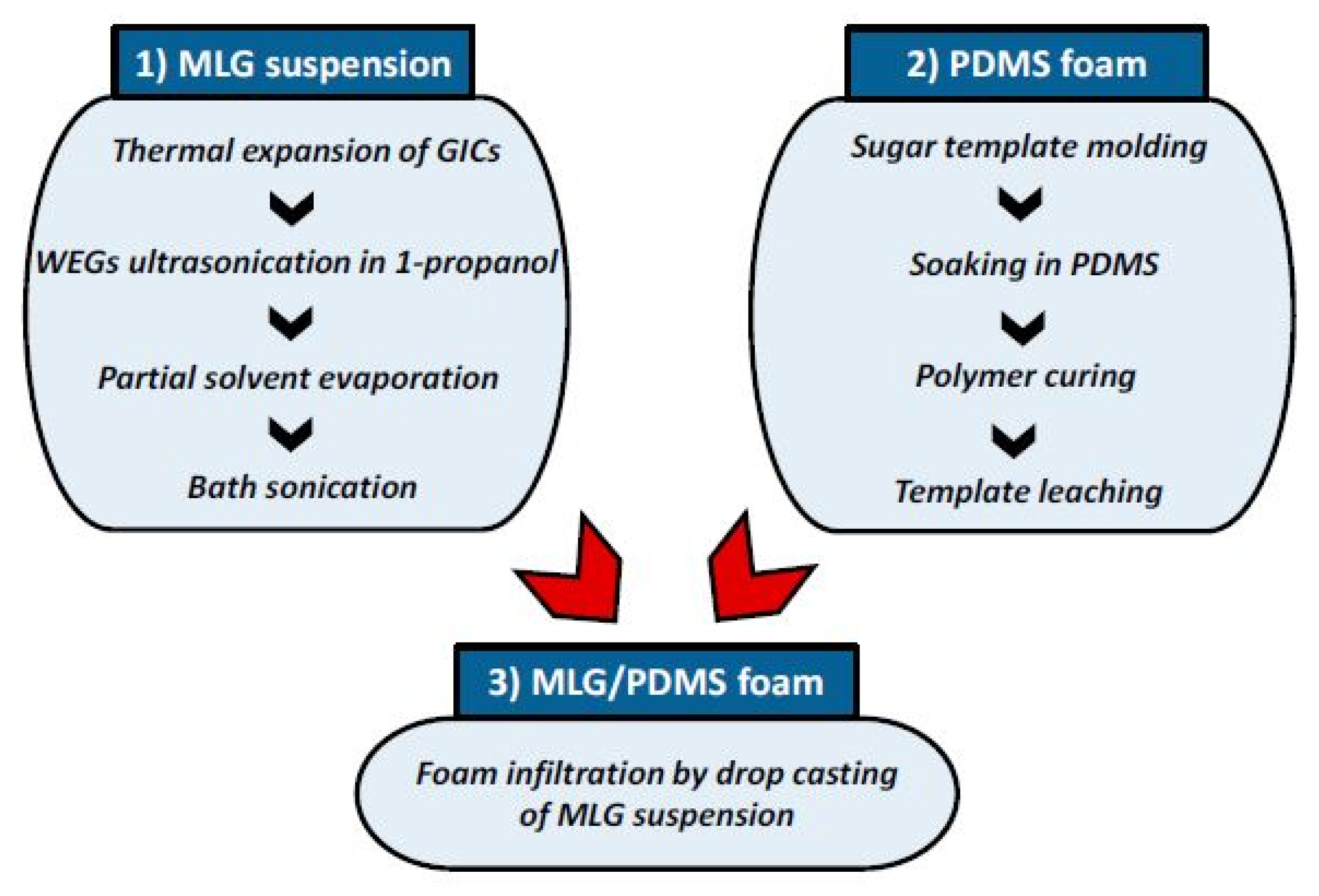
2.3. Graphene Nanoplatelets-Based Devices
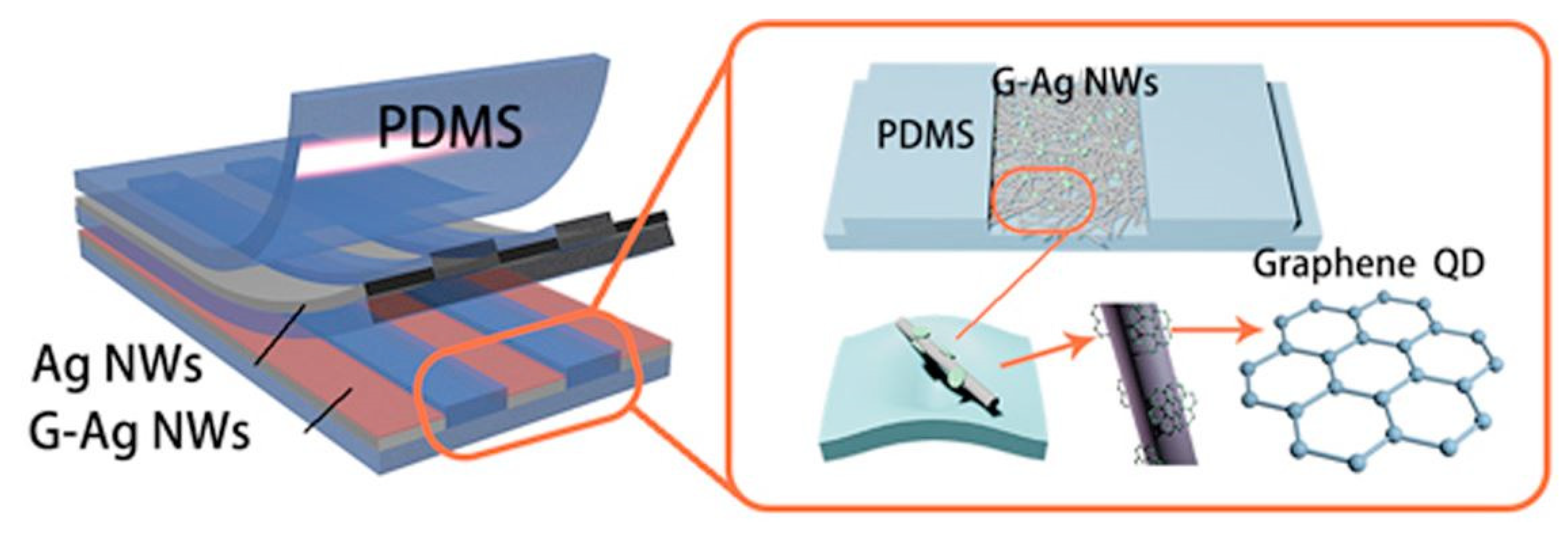
2.4. Graphene Quantum Dots-Based Devices
3. Challenges of the Existing Sensors
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Yan, T.; Wang, Z.; Pan, Z.-J. Flexible strain sensors fabricated using carbon-based nanomaterials: A review. Curr. Opin. Solid State Mater. Sci. 2018, 22, 213–228. [Google Scholar] [CrossRef]
- Zhang, S.; Sunami, Y.; Hashimoto, H. Mini review: Nanosheet technology towards biomedical application. Nanomaterials 2017, 7, 246. [Google Scholar] [CrossRef] [PubMed]
- Peng, B.; Zhao, F.; Ping, J.; Ying, Y. Recent Advances in Nanomaterial-Enabled Wearable Sensors: Material Synthesis, Sensor Design, and Personal Health Monitoring. Small 2020, 16, 2002681. [Google Scholar] [CrossRef] [PubMed]
- Nag, A.; Alahi, M.; Eshrat, E.; Mukhopadhyay, S.C.; Liu, Z. Multi-Walled Carbon Nanotubes-Based Sensors for Strain Sensing Applications. Sensors 2021, 21, 1261. [Google Scholar] [CrossRef]
- Moseley, P.T. Progress in the development of semiconducting metal oxide gas sensors: A review. Meas. Sci. Technol. 2017, 28, 082001. [Google Scholar] [CrossRef]
- Frasco, M.F.; Chaniotakis, N. Semiconductor quantum dots in chemical sensors and biosensors. Sensors 2009, 9, 7266–7286. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sze, S.M. Semiconductor Sensors; John Wiley & Sons: Hoboken, NJ, USA, 1994. [Google Scholar]
- Tilli, M.; Paulasto-Krockel, M.; Petzold, M.; Theuss, H.; Motooka, T.; Lindroos, V. Handbook of Silicon Based MEMS Materials and Technologies; Elsevier: Amsterdam, The Netherlands, 2020. [Google Scholar]
- Ejeian, F.; Azadi, S.; Razmjou, A.; Orooji, Y.; Kottapalli, A.; Warkiani, M.E.; Asadnia, M. Design and applications of MEMS flow sensors: A review. Sens. Actuators A Phys. 2019, 295, 483–502. [Google Scholar] [CrossRef]
- Nag, A.; Zia, A.I.; Li, X.; Mukhopadhyay, S.C.; Kosel, J. Novel sensing approach for LPG leakage detection: Part I—Operating mechanism and preliminary results. IEEE Sens. J. 2015, 16, 996–1003. [Google Scholar] [CrossRef] [Green Version]
- Nag, A.; Zia, A.I.; Li, X.; Mukhopadhyay, S.C.; Kosel, J. Novel sensing approach for LPG leakage detection—part II: Effects of particle size, composition, and coating layer thickness. IEEE Sens. J. 2015, 16, 1088–1094. [Google Scholar] [CrossRef] [Green Version]
- Alahi, M.E.E.; Xie, L.; Mukhopadhyay, S.; Burkitt, L. A temperature compensated smart nitrate-sensor for agricultural industry. IEEE Trans. Ind. Electron. 2017, 64, 7333–7341. [Google Scholar] [CrossRef]
- Yin, J.; Gao, W.; Zhang, Z.; Mai, Y.; Luan, A.; Jin, H.; Jian, J.; Jin, Q. Batch microfabrication of highly integrated silicon-based electrochemical sensor and performance evaluation via nitrite water contaminant determination. Electrochim. Acta 2020, 335, 135660. [Google Scholar] [CrossRef]
- Jha, R.K. Non-Dispersive Infrared Gas Sensing Technology: A Review. IEEE Sens. J. 2021, 22, 6–15. [Google Scholar] [CrossRef]
- Popa, D.; Hopper, R.; Ali, S.Z.; Cole, M.T.; Fan, Y.; Veigang-Radulescu, V.-P.; Chikkaraddy, R.; Nallala, J.; Xing, Y.; Alexander-Webber, J. A highly stable, nanotube-enhanced, CMOS-MEMS thermal emitter for mid-IR gas sensing. Sci. Rep. 2021, 11, 22915. [Google Scholar] [CrossRef] [PubMed]
- Drawbacks of MEMS. Available online: https://www.ecstuff4u.com/2020/03/advantages-disadvantages-mems.html (accessed on 8 March 2022).
- Disadvantages of MEMS. Available online: https://engineeringproductdesign.com/mems-micro-electro-mechanical-system/ (accessed on 8 March 2022).
- Khan, S.; Lorenzelli, L.; Dahiya, R.S. Technologies for printing sensors and electronics over large flexible substrates: A review. IEEE Sens. J. 2014, 15, 3164–3185. [Google Scholar] [CrossRef]
- Han, T.; Nag, A.; Afsarimanesh, N.; Mukhopadhyay, S.C.; Kundu, S.; Xu, Y. Laser-Assisted Printed Flexible Sensors: A Review. Sensors 2019, 19, 1462. [Google Scholar] [CrossRef] [Green Version]
- Xiao, Y.; Jiang, S.; Li, Y.; Zhang, W. Screen-printed flexible negative temperature coefficient temperature sensor based on polyvinyl chloride/carbon black composites. Smart Mater. Struct. 2021, 30, 025035. [Google Scholar] [CrossRef]
- Marra, F.; Minutillo, S.; Tamburrano, A.; Sarto, M.S. Production and characterization of Graphene Nanoplatelet-based ink for smart textile strain sensors via screen printing technique. Mater. Des. 2021, 198, 109306. [Google Scholar] [CrossRef]
- Kao, H.-L.; Cho, C.-L.; Chang, L.-C.; Chen, C.-B.; Chung, W.-H.; Tsai, Y.-C. A Fully Inkjet-Printed Strain Sensor Based on Carbon Nanotubes. Coatings 2020, 10, 792. [Google Scholar] [CrossRef]
- Barmpakos, D.; Tsamis, C.; Kaltsas, G. Multi-parameter paper sensor fabricated by inkjet-printed silver nanoparticle ink and PEDOT: PSS. Microelectron. Eng. 2020, 225, 111266. [Google Scholar] [CrossRef]
- He, S.; Feng, S.; Nag, A.; Afsarimanesh, N.; Han, T.; Mukhopadhyay, S.C. Recent Progress in 3D Printed Mold-Based Sensors. Sensors 2020, 20, 703. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nag, A.; Feng, S.; Mukhopadhyay, S.; Kosel, J.; Inglis, D. 3D printed mould-based graphite/PDMS sensor for low-force applications. Sens. Actuators A Phys. 2018, 280, 525–534. [Google Scholar] [CrossRef]
- Nomura, K.-I.; Kaji, R.; Iwata, S.; Otao, S.; Imawaka, N.; Yoshino, K.; Mitsui, R.; Sato, J.; Takahashi, S.; Nakajima, S.-I. A flexible proximity sensor formed by duplex screen/screen-offset printing and its application to non-contact detection of human breathing. Sci. Rep. 2016, 6, 19947. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Herrero, R.; Aresti, M.; Lasheras, M.; Landa, I.; Estella, J.; Arcos, S.L. New Developments in Printed Electronics using Offset Lithography on Paper Substrates. In Proceedings of the NIP & Digital Fabrication Conference; Society for Imaging Science and Technology: Springfield, VA, USA, 2018; pp. 82–86. [Google Scholar]
- Maddipatla, D.; Narakathu, B.B.; Avuthu, S.G.R.; Emamian, S.; Eshkeiti, A.; Chlaihawi, A.A.; Bazuin, B.J.; Joyce, M.K.; Barrett, C.W.; Atashbar, M.Z. A novel flexographic printed strain gauge on paper platform. In Proceedings of the 2015 IEEE Sensors, Busan, Korea, 1–4 November 2015; pp. 1–4. [Google Scholar]
- Wolfer, T.; Bollgruen, P.; Mager, D.; Overmeyer, L.; Korvink, J.G. Flexographic and inkjet printing of polymer optical waveguides for fully integrated sensor systems. Procedia Technol. 2014, 15, 521–529. [Google Scholar] [CrossRef] [Green Version]
- Peng, Y.; Du, B.; Xu, X.; Yang, J.; Lin, J.; Ma, C. Transparent triboelectric sensor arrays using gravure printed silver nanowire electrodes. Appl. Phys. Express 2019, 12, 066503. [Google Scholar] [CrossRef]
- Bariya, M.; Shahpar, Z.; Park, H.; Sun, J.; Jung, Y.; Gao, W.; Nyein, H.Y.Y.; Liaw, T.S.; Tai, L.-C.; Ngo, Q.P. Roll-to-roll gravure printed electrochemical sensors for wearable and medical devices. ACS Nano 2018, 12, 6978–6987. [Google Scholar] [CrossRef]
- Afsarimanesh, N.; Nag, A.; Sarkar, S.; Sabet, G.S.; Han, T.; Mukhopadhyay, S.C. A review on fabrication, characterization and implementation of wearable strain sensors. Sens. Actuators A Phys. 2020, 315, 112355. [Google Scholar] [CrossRef]
- Nag, A.; Mukhopadhyay, S.C.; Kosel, J. Wearable flexible sensors: A review. IEEE Sens. J. 2017, 17, 3949–3960. [Google Scholar] [CrossRef] [Green Version]
- Nag, A.; Alahi, M.E.E.; Feng, S.; Mukhopadhyay, S.C. IoT-based sensing system for phosphate detection using Graphite/PDMS sensors. Sens. Actuators A Phys. 2019, 286, 43–50. [Google Scholar] [CrossRef]
- Nag, A.; Afasrimanesh, N.; Feng, S.; Mukhopadhyay, S.C. Strain induced graphite/PDMS sensors for biomedical applications. Sens. Actuators A Phys. 2018, 271, 257–269. [Google Scholar] [CrossRef]
- Zhang, F.; Hu, H.; Hu, S.; Yue, J. Significant strain-rate dependence of sensing behavior in TiO2@ carbon fibre/PDMS composites for flexible strain sensors. J. Adv. Ceram. 2021, 10, 1350–1359. [Google Scholar] [CrossRef]
- Li, S.; Lin, P.; Zhao, L.; Wang, C.; Liu, D.; Liu, F.; Sun, P.; Liang, X.; Liu, F.; Yan, X. The room temperature gas sensor based on Polyaniline@ flower-like WO3 nanocomposites and flexible PET substrate for NH3 detection. Sens. Actuators B Chem. 2018, 259, 505–513. [Google Scholar] [CrossRef]
- Nag, A.; Mukhopadhyay, S.C.; Kosel, J. Tactile sensing from laser-ablated metallized PET films. IEEE Sens. J. 2016, 17, 7–13. [Google Scholar] [CrossRef]
- Altenberend, U.; Molina-Lopez, F.; Oprea, A.; Briand, D.; Bârsan, N.; De Rooij, N.F.; Weimar, U. Towards fully printed capacitive gas sensors on flexible PET substrates based on Ag interdigitated transducers with increased stability. Sens. Actuators B Chem. 2013, 187, 280–287. [Google Scholar] [CrossRef]
- Nag, A.; Mukhopadhyay, S.C.; Kosel, J. Sensing system for salinity testing using laser-induced graphene sensors. Sens. Actuators A Phys. 2017, 264, 107–116. [Google Scholar] [CrossRef] [Green Version]
- Nag, A.; Mukhopadhyay, S.C. Fabrication and implementation of printed sensors for taste sensing applications. Sens. Actuators A Phys. 2018, 269, 53–61. [Google Scholar] [CrossRef]
- Parmeggiani, M.; Zaccagnini, P.; Stassi, S.; Fontana, M.; Bianco, S.; Nicosia, C.; Pirri, C.F.; Lamberti, A. PDMS/Polyimide Composite as an Elastomeric Substrate for Multifunctional Laser-Induced Graphene Electrodes. ACS Appl. Mater. Interfaces 2019, 11, 33221–33230. [Google Scholar] [CrossRef]
- Serrano, I.G.; Panda, J.; Edvinsson, T.; Kamalakar, M.V. Flexible transparent graphene laminates via direct lamination of graphene onto polyethylene naphthalate substrates. Nanoscale Adv. 2020, 2, 3156–3163. [Google Scholar] [CrossRef]
- Lamanna, L.; Rizzi, F.; Guido, F.; Algieri, L.; Marras, S.; Mastronardi, V.M.; Qualtieri, A.; De Vittorio, M. Flexible and Transparent Aluminum-Nitride-Based Surface-Acoustic-Wave Device on Polymeric Polyethylene Naphthalate. Adv. Electron. Mater. 2019, 5, 1900095. [Google Scholar] [CrossRef]
- Jakob, M.H.; Gutsch, S.; Chatelle, C.; Krishnaraja, A.; Fahlteich, J.; Weber, W.; Zacharias, M. Flexible thin film pH sensor based on low-temperature atomic layer deposition. Phys. Status Solidi (RRL)–Rapid Res. Lett. 2017, 11, 1700123. [Google Scholar] [CrossRef]
- Deng, L.; Yuan, J.; Huang, H.; Xie, S.; Xu, J.; Yue, R. Fabrication of hierarchical Ru/PEDOT: PSS/Ti3C2Tx nanocomposites as electrochemical sensing platforms for highly sensitive Sudan I detection in food. Food Chem. 2022, 372, 131212. [Google Scholar] [CrossRef] [PubMed]
- Zeng, R.; Wang, W.; Chen, M.; Wan, Q.; Wang, C.; Knopp, D.; Tang, D. CRISPR-Cas12a-driven MXene-PEDOT: PSS piezoresistive wireless biosensor. Nano Energy 2021, 82, 105711. [Google Scholar] [CrossRef]
- Shen, G.; Chen, B.; Liang, T.; Liu, Z.; Zhao, S.; Liu, J.; Zhang, C.; Yang, W.; Wang, Y.; He, X. Transparent and Stretchable Strain Sensors with Improved Sensitivity and Reliability Based on Ag NWs and PEDOT: PSS Patterned Microstructures. Adv. Electron. Mater. 2020, 6, 1901360. [Google Scholar] [CrossRef]
- Kulkarni, S.; Navale, Y.; Navale, S.; Stadler, F.; Ramgir, N.; Patil, V. Hybrid polyaniline-WO3 flexible sensor: A room temperature competence towards NH3 gas. Sens. Actuators B Chem. 2019, 288, 279–288. [Google Scholar] [CrossRef]
- Gong, S.; Lai, D.T.; Wang, Y.; Yap, L.W.; Si, K.J.; Shi, Q.; Jason, N.N.; Sridhar, T.; Uddin, H.; Cheng, W. Tattoolike polyaniline microparticle-doped gold nanowire patches as highly durable wearable sensors. ACS Appl. Mater. Interfaces 2015, 7, 19700–19708. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Hu, D.; Guan, X.; Cai, S.; Shi, G.; Shuai, Z.; Zhang, J.; Peng, Q.; Wan, X. Brightening up Circularly Polarized Luminescence of Monosubstituted Polyacetylene by Conformation Control: Mechanism, Switching, and Sensing. Angew. Chem. Int. Ed. 2021, 60, 21918–21926. [Google Scholar] [CrossRef] [PubMed]
- Greco, E.; Shang, J.; Zhu, J.; Zhu, T. Synthesis of polyacetylene-like modified graphene oxide aerogel and its enhanced electrical properties. ACS Omega 2019, 4, 20948–20954. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tian, Y.; Kong, L.; Mao, H.; Shi, J.; Tong, B.; Cai, Z.; Dong, Y. A supramolecular approach for the synthesis of cross-linked ionic polyacetylene network gels. Mater. Chem. Front. 2020, 4, 645–650. [Google Scholar] [CrossRef]
- Qin, J.; Gao, J.; Shi, X.; Chang, J.; Dong, Y.; Zheng, S.; Wang, X.; Feng, L.; Wu, Z.S. Hierarchical ordered dual-mesoporous polypyrrole/graphene nanosheets as bi-functional active materials for high-performance planar integrated system of micro-supercapacitor and gas sensor. Adv. Funct. Mater. 2020, 30, 1909756. [Google Scholar] [CrossRef]
- Lima, R.M.; Alcaraz-Espinoza, J.J.; da Silva Jr, F.A.; de Oliveira, H.P. Multifunctional wearable electronic textiles using cotton fibers with polypyrrole and carbon nanotubes. ACS Appl. Mater. Interfaces 2018, 10, 13783–13795. [Google Scholar] [CrossRef]
- Jain, R.; Jadon, N.; Pawaiya, A. Polypyrrole based next generation electrochemical sensors and biosensors: A review. TrAC Trends Anal. Chem. 2017, 97, 363–373. [Google Scholar] [CrossRef]
- Niazvand, F.; Wagh, P.R.; Khazraei, E.; Dastjerdi, M.B.; Patil, C.; Najar, I.A. Application of carbon allotropes composites for targeted cancer therapy drugs: A review. J. Compos. Compd. 2021, 3, 140–151. [Google Scholar] [CrossRef]
- Nasir, S.; Hussein, M.Z.; Zainal, Z.; Yusof, N.A. Carbon-based nanomaterials/allotropes: A glimpse of their synthesis, properties and some applications. Materials 2018, 11, 295. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Villarreal, C.C.; Pham, T.; Ramnani, P.; Mulchandani, A. Carbon allotropes as sensors for environmental monitoring. Curr. Opin. Electrochem. 2017, 3, 106–113. [Google Scholar] [CrossRef]
- Ovid’Ko, I.; Valiev, R.; Zhu, Y. Review on superior strength and enhanced ductility of metallic nanomaterials. Prog. Mater. Sci. 2018, 94, 462–540. [Google Scholar] [CrossRef]
- Barabadi, H.; Najafi, M.; Samadian, H.; Azarnezhad, A.; Vahidi, H.; Mahjoub, M.A.; Koohiyan, M.; Ahmadi, A. A systematic review of the genotoxicity and antigenotoxicity of biologically synthesized metallic nanomaterials: Are green nanoparticles safe enough for clinical marketing? Medicina 2019, 55, 439. [Google Scholar] [CrossRef] [Green Version]
- Sáenz-Trevizo, A.; Hodge, A. Nanomaterials by design: A review of nanoscale metallic multilayers. Nanotechnology 2020, 31, 292002. [Google Scholar] [CrossRef] [PubMed]
- Nag, A.; Mukhopadhyay, S.C.; Kosel, J. Flexible carbon nanotube nanocomposite sensor for multiple physiological parameter monitoring. Sens. Actuators A Phys. 2016, 251, 148–155. [Google Scholar] [CrossRef] [Green Version]
- Han, T.; Nag, A.; Mukhopadhyay, S.C.; Xu, Y. Carbon nanotubes and its gas-sensing applications: A review. Sens. Actuators A Phys. 2019, 291, 107–143. [Google Scholar] [CrossRef]
- Gao, J.; He, S.; Nag, A.; Wong, J.W.C. A Review of the Use of Carbon Nanotubes and Graphene-Based Sensors for the Detection of Aflatoxin M1 Compounds in Milk. Sensors 2021, 21, 3602. [Google Scholar] [CrossRef] [PubMed]
- Nag, A.; Alahi, M.E.E.; Mukhopadhyay, S.C. Recent progress in the fabrication of graphene fibers and their composites for applications of monitoring human activities. Appl. Mater. Today 2021, 22, 100953. [Google Scholar] [CrossRef]
- Han, T.; Nag, A.; Simorangkir, R.B.; Afsarimanesh, N.; Liu, H.; Mukhopadhyay, S.C.; Xu, Y.; Zhadobov, M.; Sauleau, R. Multifunctional flexible sensor based on laser-induced graphene. Sensors 2019, 19, 3477. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nag, A.; Mitra, A.; Mukhopadhyay, S.C. Graphene and its sensor-based applications: A review. Sens. Actuators A Phys. 2018, 270, 177–194. [Google Scholar] [CrossRef]
- Tadakaluru, S.; Thongsuwan, W.; Singjai, P. Stretchable and flexible high-strain sensors made using carbon nanotubes and graphite films on natural rubber. Sensors 2014, 14, 868–876. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ren, T.-L.; Tian, H.; Xie, D.; Yang, Y. Flexible graphite-on-paper piezoresistive sensors. Sensors 2012, 12, 6685–6694. [Google Scholar] [CrossRef] [Green Version]
- Kano, S.; Fujii, M. All-painting process to produce respiration sensor using humidity-sensitive nanoparticle film and graphite trace. ACS Sustain. Chem. Eng. 2018, 6, 12217–12223. [Google Scholar] [CrossRef] [Green Version]
- Elahi, N.; Kamali, M.; Baghersad, M.H. Recent biomedical applications of gold nanoparticles: A review. Talanta 2018, 184, 537–556. [Google Scholar] [CrossRef] [PubMed]
- Fahmy, H.M.; Mohamed, F.M.; Marzouq, M.H.; Mustafa, A.B.E.-D.; Alsoudi, A.M.; Ali, O.A.; Mohamed, M.A.; Mahmoud, F.A. Review of green methods of iron nanoparticles synthesis and applications. BioNanoScience 2018, 8, 491–503. [Google Scholar] [CrossRef]
- Beyene, H.D.; Werkneh, A.A.; Bezabh, H.K.; Ambaye, T.G. Synthesis paradigm and applications of silver nanoparticles (AgNPs), a review. Sustain. Mater. Technol. 2017, 13, 18–23. [Google Scholar] [CrossRef]
- Nasr Esfahani, M.; Alaca, B.E. A Review on Size-Dependent Mechanical Properties of Nanowires. Adv. Eng. Mater. 2019, 21, 1900192. [Google Scholar] [CrossRef]
- Sannicolo, T.; Lagrange, M.; Cabos, A.; Celle, C.; Simonato, J.P.; Bellet, D. Metallic nanowire-based transparent electrodes for next generation flexible devices: A review. Small 2016, 12, 6052–6075. [Google Scholar] [CrossRef]
- Yogeswaran, U.; Chen, S.-M. A review on the electrochemical sensors and biosensors composed of nanowires as sensing material. Sensors 2008, 8, 290–313. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.; Zhao, X.; Li, P.; Huang, Y.; Wang, J.; Zhang, J. Highly sensitive Fe3O4 nanobeads/graphene-based molecularly imprinted electrochemical sensor for 17β-estradiol in water. Anal. Chim. Acta 2015, 884, 106–113. [Google Scholar] [CrossRef] [PubMed]
- Ma, Q.; Li, Y.; Lin, Z.-H.; Tang, G.; Su, X.-G. A novel ascorbic acid sensor based on the Fe3+/Fe2+ modulated photoluminescence of CdTe quantum dots@ SiO2 nanobeads. Nanoscale 2013, 5, 9726–9731. [Google Scholar] [CrossRef] [PubMed]
- Rajabi, H.R.; Roushani, M.; Shamsipur, M. Development of a highly selective voltammetric sensor for nanomolar detection of mercury ions using glassy carbon electrode modified with a novel ion imprinted polymeric nanobeads and multi-wall carbon nanotubes. J. Electroanal. Chem. 2013, 693, 16–22. [Google Scholar] [CrossRef]
- Gong, X.; Zhang, L.; Huang, Y.; Wang, S.; Pan, G.; Li, L. Directly writing flexible temperature sensor with graphene nanoribbons for disposable healthcare devices. RSC Adv. 2020, 10, 22222–22229. [Google Scholar] [CrossRef]
- Johnson, A.P.; Gangadharappa, H.; Pramod, K. Graphene nanoribbons: A promising nanomaterial for biomedical applications. J. Control. Release 2020, 325, 141–162. [Google Scholar] [CrossRef]
- Chen, Y.; Zhang, Y.; Yuan, F.; Ding, F.; Schmidt, O.G. A flexible PMN-PT ribbon-based piezoelectric-pyroelectric hybrid generator for human-activity energy harvesting and monitoring. Adv. Electron. Mater. 2017, 3, 1600540. [Google Scholar] [CrossRef] [Green Version]
- Sehrawat, P.; Julien, C.; Islam, S. E-textile based wearable thermometer from WS2-quantum dots. Nanotechnology 2021, 32, 335503. [Google Scholar]
- Molaei, M.J. Principles, mechanisms, and application of carbon quantum dots in sensors: A review. Anal. Methods 2020, 12, 1266–1287. [Google Scholar] [CrossRef]
- Molaei, M.J. A review on nanostructured carbon quantum dots and their applications in biotechnology, sensors, and chemiluminescence. Talanta 2019, 196, 456–478. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Su, J.; Song, Y.; Ye, R. Laser-Induced Graphene: En Route to Smart Sensing. Nano-Micro Lett. 2020, 12, 157. [Google Scholar] [CrossRef] [PubMed]
- Rajan, G.; Morgan, J.J.; Murphy, C.; Torres Alonso, E.; Wade, J.; Ott, A.K.; Russo, S.; Alves, H.; Craciun, M.F.; Neves, A.I. Low Operating Voltage Carbon–Graphene Hybrid E-textile for Temperature Sensing. ACS Appl. Mater. Interfaces 2020, 12, 29861–29867. [Google Scholar] [CrossRef] [PubMed]
- Yu, M.; Yu, G.; Dai, B. Graphene Fiber-Based Strain-Insensitive Wearable Temperature Sensor. IEEE Sens. Lett. 2020, 4, 2501304. [Google Scholar] [CrossRef]
- Vertuccio, L.; Guadagno, L.; Spinelli, G.; Lamberti, P.; Tucci, V.; Russo, S. Piezoresistive properties of resin reinforced with carbon nanotubes for health-monitoring of aircraft primary structures. Compos. Part B Eng. 2016, 107, 192–202. [Google Scholar] [CrossRef]
- Huang, H.; Su, S.; Wu, N.; Wan, H.; Wan, S.; Bi, H.; Sun, L. Graphene-based sensors for human health monitoring. Front. Chem. 2019, 7, 399. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kumar, S.; Bukkitgar, S.D.; Singh, S.; Singh, V.; Reddy, K.R.; Shetti, N.P.; Venkata Reddy, C.; Sadhu, V.; Naveen, S. Electrochemical sensors and biosensors based on graphene functionalized with metal oxide nanostructures for healthcare applications. ChemistrySelect 2019, 4, 5322–5337. [Google Scholar] [CrossRef]
- Nag, A.; Menzies, B.; Mukhopadhyay, S.C. Performance analysis of flexible printed sensors for robotic arm applications. Sens. Actuators A Phys. 2018, 276, 226–236. [Google Scholar] [CrossRef]
- Wang, T.; Huang, D.; Yang, Z.; Xu, S.; He, G.; Li, X.; Hu, N.; Yin, G.; He, D.; Zhang, L. A review on graphene-based gas/vapor sensors with unique properties and potential applications. Nano-Micro Lett. 2016, 8, 95–119. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, C.; Zhang, X.; Zhang, H.; Duan, X. Temperature Sensing at the Robot Fingertip Using Reduced Graphene Oxide-based Sensor on a Flexible Substrate. In Proceedings of the 2019 IEEE Sensors, Montreal, QC, Canada, 27–30 October 2019; pp. 1–4. [Google Scholar]
- Alahi, M.E.E.; Nag, A.; Mukhopadhyay, S.C.; Burkitt, L. A temperature-compensated graphene sensor for nitrate monitoring in real-time application. Sens. Actuators A Phys. 2018, 269, 79–90. [Google Scholar] [CrossRef]
- Guan, J.-F.; Huang, Z.-N.; Zou, J.; Jiang, X.-Y.; Peng, D.-M.; Yu, J.-G. A sensitive non-enzymatic electrochemical sensor based on acicular manganese dioxide modified graphene nanosheets composite for hydrogen peroxide detection. Ecotoxicol. Environ. Saf. 2020, 190, 110123. [Google Scholar] [CrossRef] [PubMed]
- Kaur, M.; Kaur, M.; Sharma, V.K. Nitrogen-doped graphene and graphene quantum dots: A review onsynthesis and applications in energy, sensors and environment. Adv. Colloid Interface Sci. 2018, 259, 44–64. [Google Scholar] [CrossRef] [PubMed]
- Kwak, Y.H.; Kim, W.; Park, K.B.; Kim, K.; Seo, S. Flexible heartbeat sensor for wearable device. Biosens. Bioelectron. 2017, 94, 250–255. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Jiang, Y.; Tai, H.; Liu, B.; Duan, Z.; Yuan, Z.; Pan, H.; Xie, G.; Du, X.; Su, Y. An integrated flexible self-powered wearable respiration sensor. Nano Energy 2019, 63, 103829. [Google Scholar] [CrossRef]
- Sun, S.; Liu, Y.; Chang, X.; Jiang, Y.; Wang, D.; Tang, C.; He, S.; Wang, M.; Guo, L.; Gao, Y. A wearable, waterproof, and highly sensitive strain sensor based on three-dimensional graphene/carbon black/Ni sponge for wirelessly monitoring human motions. J. Mater. Chem. C 2020, 8, 2074–2085. [Google Scholar] [CrossRef]
- Xia, S.; Song, S.; Jia, F.; Gao, G. A flexible, adhesive and self-healable hydrogel-based wearable strain sensor for human motion and physiological signal monitoring. J. Mater. Chem. B 2019, 7, 4638–4648. [Google Scholar] [CrossRef] [PubMed]
- Jiang, S.; Pang, Y.; Wang, D.; Yang, Y.; Yang, Z.; Yang, Y.; Ren, T.-L. Gait recognition based on graphene porous network structure pressure sensors for rehabilitation therapy. In Proceedings of the 2018 IEEE International Conference on Electron Devices and Solid State Circuits (EDSSC), Shenzhen, China, 6–8 June 2018; pp. 1–2. [Google Scholar]
- Kwon, S.S.; Shin, J.H.; Choi, J.; Nam, S.; Park, W.I. Defect-mediated molecular interaction and charge transfer in graphene mesh–glucose Sensors. ACS Appl. Mater. Interfaces 2017, 9, 14216–14221. [Google Scholar] [CrossRef] [PubMed]
- Irani, F.S.; Shafaghi, A.H.; Tasdelen, M.C.; Delipinar, T.; Kaya, C.E.; Yapici, G.G.; Yapici, M.K. Graphene as a Piezoresistive Material in Strain Sensing Applications. Micromachines 2022, 13, 119. [Google Scholar] [CrossRef] [PubMed]
- Yu, R.; Zhu, C.; Wan, J.; Li, Y.; Hong, X. Review of graphene-based textile strain sensors, with emphasis on structure activity relationship. Polymers 2021, 13, 151. [Google Scholar] [CrossRef] [PubMed]
- Cao, M.; Su, J.; Fan, S.; Qiu, H.; Su, D.; Li, L. Wearable piezoresistive pressure sensors based on 3D graphene. Chem. Eng. J. 2021, 406, 126777. [Google Scholar] [CrossRef]
- Miranda, I.; Souza, A.; Sousa, P.; Ribeiro, J.; Castanheira, E.; Lima, R.; Minas, G. Properties and applications of PDMS for biomedical engineering: A review. J. Funct. Biomater. 2022, 13, 2. [Google Scholar] [CrossRef] [PubMed]
- Herren, B.; Webster, V.; Davidson, E.; Saha, M.C.; Altan, M.C.; Liu, Y. PDMS Sponges with Embedded Carbon Nanotubes as Piezoresistive Sensors for Human Motion Detection. Nanomaterials 2021, 11, 1740. [Google Scholar] [CrossRef]
- Sainz-Urruela, C.; Vera-López, S.; San Andrés, M.P.; Díez-Pascual, A.M. Graphene-Based Sensors for the Detection of Bioactive Compounds: A Review. Int. J. Mol. Sci. 2021, 22, 3316. [Google Scholar] [CrossRef] [PubMed]
- Thangamani, G.; Deshmukh, K.; Kovářík, T.; Nambiraj, N.; Ponnamma, D.; Sadasivuni, K.K.; Khalil, H.A.; Pasha, S.K. Graphene oxide nanocomposites based room temperature gas sensors: A review. Chemosphere 2021, 280, 130641. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.; Guo, W.; Lu, X.; Xu, H.; Yang, Q.; Tan, J.; Zhang, W. Reduced graphene oxide nanocomposite based electrochemical biosensors for monitoring foodborne pathogenic bacteria: A review. Food Control 2021, 127, 108117. [Google Scholar] [CrossRef]
- Zhang, J.; Zhou, Q.; Cao, J.; Wu, W.; Zhang, H.; Shi, Y.; Mao, Q.; Ma, H. Flexible textile ion sensors based on reduced graphene oxide/fullerene and their potential applications of sweat characterization. Cellulose 2021, 28, 3123–3133. [Google Scholar] [CrossRef]
- Moriche, R.; Sánchez, M.; Jiménez-Suárez, A.; Prolongo, S.; Urena, A. Strain monitoring mechanisms of sensors based on the addition of graphene nanoplatelets into an epoxy matrix. Compos. Sci. Technol. 2016, 123, 65–70. [Google Scholar] [CrossRef]
- Cataldi, P.; Athanassiou, A.; Bayer, I.S. Graphene nanoplatelets-based advanced materials and recent progress in sustainable applications. Appl. Sci. 2018, 8, 1438. [Google Scholar] [CrossRef] [Green Version]
- Mousavi, S.M.; Hashemi, S.A.; Gholami, A.; Mazraedoost, S.; Chiang, W.-H.; Arjmand, O.; Omidifar, N.; Babapoor, A. Precise blood glucose sensing by nitrogen-doped graphene quantum dots for tight control of diabetes. J. Sens. 2021, 2021, 5580203. [Google Scholar] [CrossRef]
- Ahmed, S.R.; Kumar, S.; Ortega, G.A.; Srinivasan, S.; Rajabzadeh, A.R. Target specific aptamer-induced self-assembly of fluorescent graphene quantum dots on palladium nanoparticles for sensitive detection of tetracycline in raw milk. Food Chem. 2021, 346, 128893. [Google Scholar] [CrossRef]
- Shen, G. Recent advances of flexible sensors for biomedical applications. Prog. Nat. Sci. Mater. Int. 2021, 31, 872–882. [Google Scholar] [CrossRef]
- Nemčeková, K.; Labuda, J. Advanced materials-integrated electrochemical sensors as promising medical diagnostics tools: A review. Mater. Sci. Eng. C 2021, 120, 111751. [Google Scholar] [CrossRef]
- Chen, Z.; Yan, T.; Pan, Z. Review of flexible strain sensors based on cellulose composites for multi-faceted applications. Cellulose 2021, 28, 615–645. [Google Scholar] [CrossRef]
- Suzuki, K.; Nakagawa, R.; Zhang, Q.; Miura, H. Development of highly sensitive strain sensor using area-arrayed graphene nanoribbons. Nanomaterials 2021, 11, 1701. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Hao, Z.; Wang, X.; Huang, C.; Lin, Q.; Zhao, X.; Pan, Y. A Flexible and regenerative aptameric graphene–Nafion biosensor for cytokine storm biomarker monitoring in undiluted biofluids toward wearable applications. Adv. Funct. Mater. 2021, 31, 2005958. [Google Scholar] [CrossRef]
- Pu, Z.; Zou, C.; Wang, R.; Lai, X.; Yu, H.; Xu, K.; Li, D. A continuous glucose monitoring device by graphene modified electrochemical sensor in microfluidic system. Biomicrofluidics 2016, 10, 011910. [Google Scholar] [CrossRef] [Green Version]
- Xu, M.; Zhu, Y.; Gao, S.; Zhang, Z.; Gu, Y.; Liu, X. Reduced graphene oxide-coated silica nanospheres as flexible enzymatic biosensors for detection of glucose in sweat. ACS Appl. Nano Mater. 2021, 4, 12442–12452. [Google Scholar] [CrossRef]
- Kumar, V.; Matai, I.; Kumar, A.; Sachdev, A. GNP-CeO2-polyaniline hybrid hydrogel for electrochemical detection of peroxynitrite anion and its integration in a microfluidic platform. Microchim. Acta 2021, 188, 436. [Google Scholar] [CrossRef] [PubMed]
- Al-Ogaidi, I.; Gou, H.; Aguilar, Z.P.; Guo, S.; Melconian, A.K.; Al-Kazaz, A.K.A.; Meng, F.; Wu, N. Detection of the ovarian cancer biomarker CA-125 using chemiluminescence resonance energy transfer to graphene quantum dots. Chem. Commun. 2014, 50, 1344–1346. [Google Scholar] [CrossRef] [PubMed]
- Xie, F.; Cao, X.; Qu, F.; Asiri, A.M.; Sun, X. Cobalt nitride nanowire array as an efficient electrochemical sensor for glucose and H2O2 detection. Sens. Actuators B Chem. 2018, 255, 1254–1261. [Google Scholar] [CrossRef]
- Meng, W.; Wen, Y.; Dai, L.; He, Z.; Wang, L. A novel electrochemical sensor for glucose detection based on Ag@ ZIF-67 nanocomposite. Sens. Actuators B Chem. 2018, 260, 852–860. [Google Scholar] [CrossRef]
- Nikolaev, K.G.; Maybeck, V.; Neumann, E.; Ermakov, S.S.; Ermolenko, Y.E.; Offenhäusser, A.; Mourzina, Y.G. Bimetallic nanowire sensors for extracellular electrochemical hydrogen peroxide detection in HL-1 cell culture. J. Solid State Electrochem. 2018, 22, 1023–1035. [Google Scholar] [CrossRef]
- Wang, L.; Lu, W.; Zhu, W.; Wu, H.; Wang, F.; Xu, X. A photoelectrochemical sensor for highly sensitive detection of glucose based on Au–NiO1–x hybrid nanowires. Sens. Actuators B Chem. 2020, 304, 127330. [Google Scholar] [CrossRef]
- Yang, Y.; Cao, Z.; He, P.; Shi, L.; Ding, G.; Wang, R.; Sun, J. Ti3C2Tx MXene-graphene composite films for wearable strain sensors featured with high sensitivity and large range of linear response. Nano Energy 2019, 66, 104134. [Google Scholar] [CrossRef]
- Jiang, X.; Ren, Z.; Fu, Y.; Liu, Y.; Zou, R.; Ji, G.; Ning, H.; Li, Y.; Wen, J.; Qi, H.J. Highly compressible and sensitive pressure sensor under large strain based on 3D porous reduced graphene oxide fiber fabrics in wide compression strains. ACS Appl. Mater. Interfaces 2019, 11, 37051–37059. [Google Scholar] [CrossRef] [PubMed]
- Baloda, S.; Ansari, Z.A.; Singh, S.; Gupta, N. Development and Analysis of Graphene Nanoplatelets (GNPs)-Based Flexible Strain Sensor for Health Monitoring Applications. IEEE Sens. J. 2020, 20, 13302–13309. [Google Scholar] [CrossRef]
- Zhang, Q.; Yuan, Z.-Y.; Liu, L.-H.; Zhao, D.; Ji, J.-L.; Zhuo, K.; Zhang, H.-L.; Zhang, W.-D.; Sang, S.-B. Performance of CNTs/GQD-based flexible strain sensors. J. Nanosci. Nanotechnol. 2020, 20, 1013–1019. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.H.; Jang, N.S.; Ha, S.H.; Cho, J.H.; Kim, J.M. Highly sensitive and stretchable resistive strain sensors based on microstructured metal nanowire/elastomer composite films. Small 2018, 14, 1704232. [Google Scholar] [CrossRef] [PubMed]
- Duan, S.; Wang, Z.; Zhang, L.; Liu, J.; Li, C. A highly stretchable, sensitive, and transparent strain sensor based on binary hybrid network consisting of hierarchical multiscale metal nanowires. Adv. Mater. Technol. 2018, 3, 1800020. [Google Scholar] [CrossRef]
- Chen, J.; Zhang, J.; Luo, Z.; Zhang, J.; Li, L.; Su, Y.; Gao, X.; Li, Y.; Tang, W.; Cao, C. Superelastic, sensitive, and low hysteresis flexible strain sensor based on wave-patterned liquid metal for human activity monitoring. ACS Appl. Mater. Interfaces 2020, 12, 22200–22211. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.K.; Hong, S.; Cho, H.M.; Lee, J.; Suh, Y.D.; Ham, J.; Ko, S.H. Highly sensitive and stretchable multidimensional strain sensor with prestrained anisotropic metal nanowire percolation networks. Nano Lett. 2015, 15, 5240–5247. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Yang, R.; Shi, Z.; Zhang, L.; Shi, D.; Wang, E.; Zhang, G. Super-elastic graphene ripples for flexible strain sensors. ACS Nano 2011, 5, 3645–3650. [Google Scholar] [CrossRef]
- Moon, D.-B.; Bag, A.; Lee, H.-B.; Meeseepong, M.; Lee, D.-H.; Lee, N.-E. A stretchable, room-temperature operable, chemiresistive gas sensor using nanohybrids of reduced graphene oxide and zinc oxide nanorods. Sens. Actuators B Chem. 2021, 345, 130373. [Google Scholar] [CrossRef]
- Tang, Y.; Zhao, Z.; Hu, H.; Liu, Y.; Wang, X.; Zhou, S.; Qiu, J. Highly stretchable and ultrasensitive strain sensor based on reduced graphene oxide microtubes–elastomer composite. ACS Appl. Mater. Interfaces 2015, 7, 27432–27439. [Google Scholar] [CrossRef] [PubMed]
- Wiederoder, M.S.; Weiss, M.; Yoon, B.; Paffenroth, R.C.; McGraw, S.K.; Uzarski, J.R. Impact of graphene nanoplatelet concentration and film thickness on vapor detection for polymer based chemiresistive sensors. Curr. Appl. Phys. 2019, 19, 978–983. [Google Scholar] [CrossRef]
- Öztürk, A.; Özçelik, N.; Yurtcan, A.B. Platinum/graphene nanoplatelets/silicone rubber composites as polymer electrolyte membrane fuel cell catalysts. Mater. Chem. Phys. 2021, 260, 124110. [Google Scholar] [CrossRef]
- Rinaldi, A.; Tamburrano, A.; Fortunato, M.; Sarto, M.S. A flexible and highly sensitive pressure sensor based on a PDMS foam coated with graphene nanoplatelets. Sensors 2016, 16, 2148. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Younis, M.R.; He, G.; Lin, J.; Huang, P. Recent advances on graphene quantum dots for bioimaging applications. Front. Chem. 2020, 8, 424. [Google Scholar] [CrossRef]
- Ye, W.; Guo, J.; Bao, X.; Chen, T.; Weng, W.; Chen, S.; Yang, M. Rapid and sensitive detection of bacteria response to antibiotics using nanoporous membrane and graphene quantum dot (GQDs)-based electrochemical biosensors. Materials 2017, 10, 603. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, Z.; Wu, C.; Li, F.; Chen, W.; Guo, T.; Kim, T.W. Triboelectric electronic-skin based on graphene quantum dots for application in self-powered, smart, artificial fingers. Nano Energy 2018, 49, 274–282. [Google Scholar] [CrossRef]
- Johnson, D.W.; Dobson, B.P.; Coleman, K.S. A manufacturing perspective on graphene dispersions. Curr. Opin. Colloid Interface Sci. 2015, 20, 367–382. [Google Scholar] [CrossRef] [Green Version]
- Liu, W.; Webster, T. Toxicity and biocompatibility properties of nanocomposites for musculoskeletal tissue regeneration. In Nanocomposites for Musculoskeletal Tissue Regeneration; Elsevier: Amsterdam, The Netherlands, 2016; pp. 95–122. [Google Scholar]
- Sun, C.; Mikhaylov, R.; Fu, Y.; Wu, F.; Wang, H.; Yuan, X.; Xie, Z.; Liang, D.; Wu, Z.; Yang, X. Flexible printed circuit board as novel electrodes for acoustofluidic devices. IEEE Trans. Electron Devices 2020, 68, 393–398. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, H.; Liu, F.; Wu, X.; Xu, J.; Cui, H.; Wu, Y.; Xue, R.; Tian, C.; Zheng, B. Flexible printed circuit board based on graphene/polyimide composites with excellent thermal conductivity and sandwich structure. Compos. Part A Appl. Sci. Manuf. 2020, 138, 106075. [Google Scholar] [CrossRef]
- Baumbauer, C.L.; Anderson, M.G.; Ting, J.; Sreekumar, A.; Rabaey, J.M.; Arias, A.C.; Thielens, A. Printed, flexible, compact UHF-RFID sensor tags enabled by hybrid electronics. Sci. Rep. 2020, 10, 16543. [Google Scholar] [CrossRef] [PubMed]
- Bibi, F.; Guillaume, C.; Gontard, N.; Sorli, B. A review: RFID technology having sensing aptitudes for food industry and their contribution to tracking and monitoring of food products. Trends Food Sci. Technol. 2017, 62, 91–103. [Google Scholar] [CrossRef]
- Sodhro, A.H.; Sangaiah, A.K.; Pirphulal, S.; Sekhari, A.; Ouzrout, Y. Green media-aware medical IoT system. Multimed. Tools Appl. 2019, 78, 3045–3064. [Google Scholar] [CrossRef]
- Shen, M.; Deng, Y.; Zhu, L.; Du, X.; Guizani, N. Privacy-preserving image retrieval for medical IoT systems: A blockchain-based approach. IEEE Netw. 2019, 33, 27–33. [Google Scholar] [CrossRef]
- He, B.; Li, Y. Big data reduction and optimization in sensor monitoring network. J. Appl. Math. 2014, 2014, 294591. [Google Scholar] [CrossRef]
- Majumder, B.D.; Roy, J.K.; Padhee, S. Recent advances in multifunctional sensing technology on a perspective of multi-sensor system: A review. IEEE Sens. J. 2018, 19, 1204–1214. [Google Scholar] [CrossRef]

| Materials Used | Fabrication Method | Linear Range | Sensitivity | Limit of Detection | Ref. |
|---|---|---|---|---|---|
| GN, PDMS, Gold NPs | Micromolding | 0–162 mg/dL | 10 mA/cm2 | 1.44 mg/dL | [123] |
| rGO, PDMS, SiO2, Nafion | Electrostatic interaction, in situ reduction | 0.1–9 mM | 60.8 µA·mM−1·cm−2 | 3.7 µM | [124] |
| GNP, CeO2, PDMS, PANI | Magnetic stirring | 5–100 µM | 29.35 ± 1.4 μA·μM−1 | 0.14 µM | [125] |
| GQD, PDMS | Salination | 0.1–600 U/mL | - | 0.05 U/mL | [126] |
| Co3N NWs, Titanium | Autoclave treatment | 0.1 μM–25 mM | 3325.6 μA·mM−1·cm−2 | 50 nM | [127] |
| Co-based MOF, Ag@ZIF-67, Glassy carbon electrode | Sequential deposition-reduction | 2–1000 μM | 0.379 μA·μM−1·cm−2 | 0.66 μM | [128] |
| Gold, palladium naowires | Electrochemical nanowire assembly | 10−6–10−3 M | 18 μA·M−1 | 3 × 10−7 M | [129] |
| Gold-Nickel oxide nanowires | Plasmon method | 0.005–15 mM | 4.061 mA·cm−2·mM−1 | 0.005 mM | [130] |
| Materials Used | Fabrication Method | Linear Range | Gauge Factor | Limit of Detection | Ref. |
|---|---|---|---|---|---|
| GN, PDMS, Ti3C2Tx | Magnetic stirring | 0–74.1% | 190.8 (0–56%), 1148.2 (52.6–74.1%) | −0.025% (lower) | [131] |
| rGO fiber fabrics, PDMS | Vacuum filtration | 0.24–70% | 1668.48 | 1.17 Pa (higher) | [132] |
| GNP, PDMS | Coating | 0–65% | 62.5 | 65% (higher) | [133] |
| GQD, CNTs, PDMS, Copper wires | Drop casting | 7% | 841.2 | 19% (higher) | [134] |
| Ag NWs, Dragon skin | Embed-and-transfer | 0–150% | 81 (>130% strain) | 150% (higher) | [135] |
| Au NWs, PU nanofibers | Electrospinning | 0–70% | 12 (5% strain)–2379 (70% strain) | 70% (higher) | [136] |
| EGaIn, Ecoflex | Electroless plating | - | 21–25 | 320% (higher) | [137] |
| Ag NWs, PDMS | Vacuum filtration and trasfer | 0–10% | >20 | 35% (higher) | [138] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
He, S.; Zhang, Y.; Gao, J.; Nag, A.; Rahaman, A. Integration of Different Graphene Nanostructures with PDMS to Form Wearable Sensors. Nanomaterials 2022, 12, 950. https://doi.org/10.3390/nano12060950
He S, Zhang Y, Gao J, Nag A, Rahaman A. Integration of Different Graphene Nanostructures with PDMS to Form Wearable Sensors. Nanomaterials. 2022; 12(6):950. https://doi.org/10.3390/nano12060950
Chicago/Turabian StyleHe, Shan, Yang Zhang, Jingrong Gao, Anindya Nag, and Abdul Rahaman. 2022. "Integration of Different Graphene Nanostructures with PDMS to Form Wearable Sensors" Nanomaterials 12, no. 6: 950. https://doi.org/10.3390/nano12060950
APA StyleHe, S., Zhang, Y., Gao, J., Nag, A., & Rahaman, A. (2022). Integration of Different Graphene Nanostructures with PDMS to Form Wearable Sensors. Nanomaterials, 12(6), 950. https://doi.org/10.3390/nano12060950







