Electrochemical and X-ray Photoelectron Spectroscopy Surface Characterization of Interchain-Driven Self-Assembled Monolayer (SAM) Reorganization
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
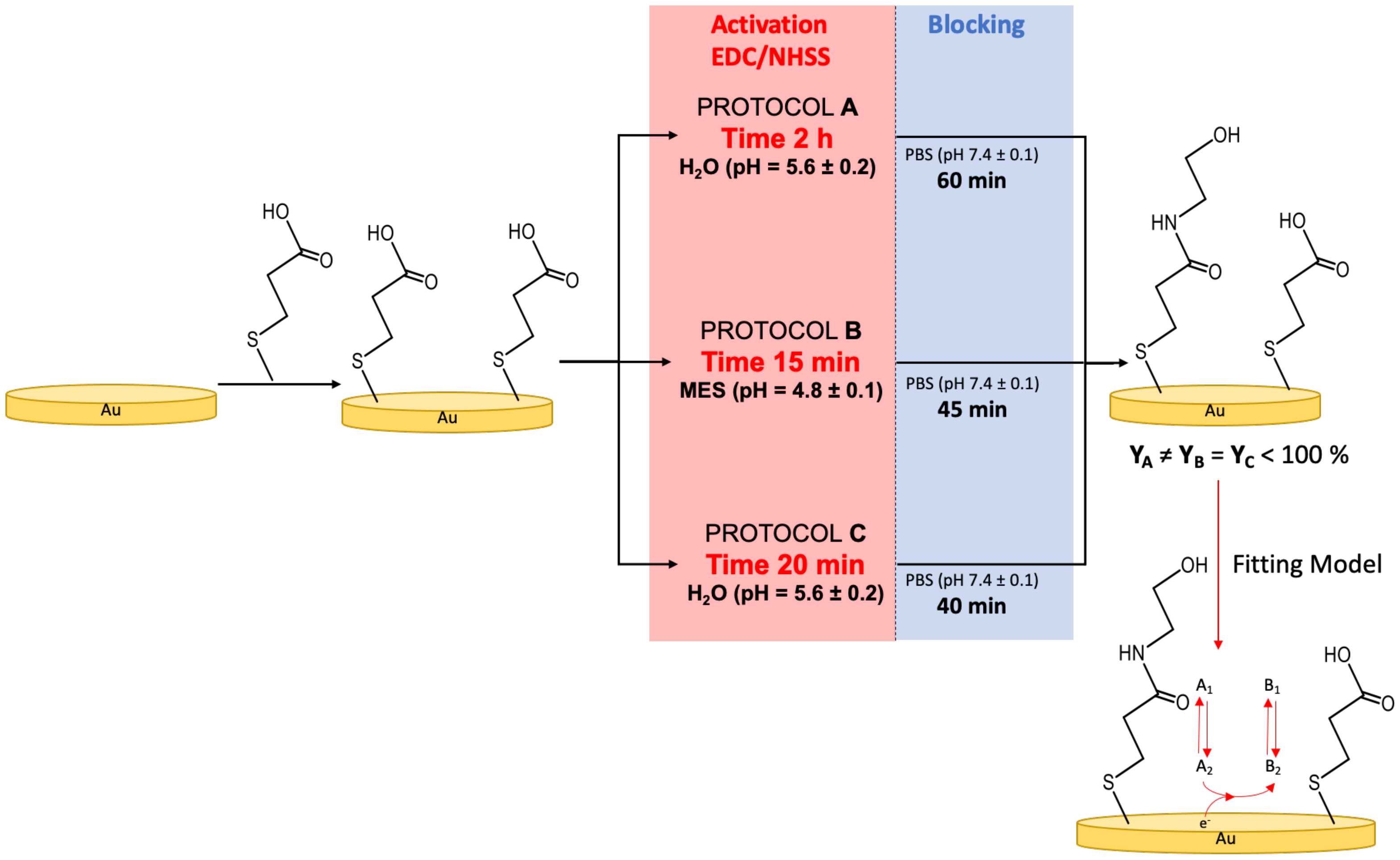
2.2. Electrode Preparation
- Deactivated SAMs, without specifying the protocols used.
- Protocol-A SAMs, Protocol-B SAMs, and Protocol-C SAMs, for describing an SAM functionalised using a specific protocol.
2.3. Electrochemical Measurements
2.4. Electrochemical Model
2.5. X-ray Photoelectron Spectroscopy (XPS) Analyses
3. Results and Discussion
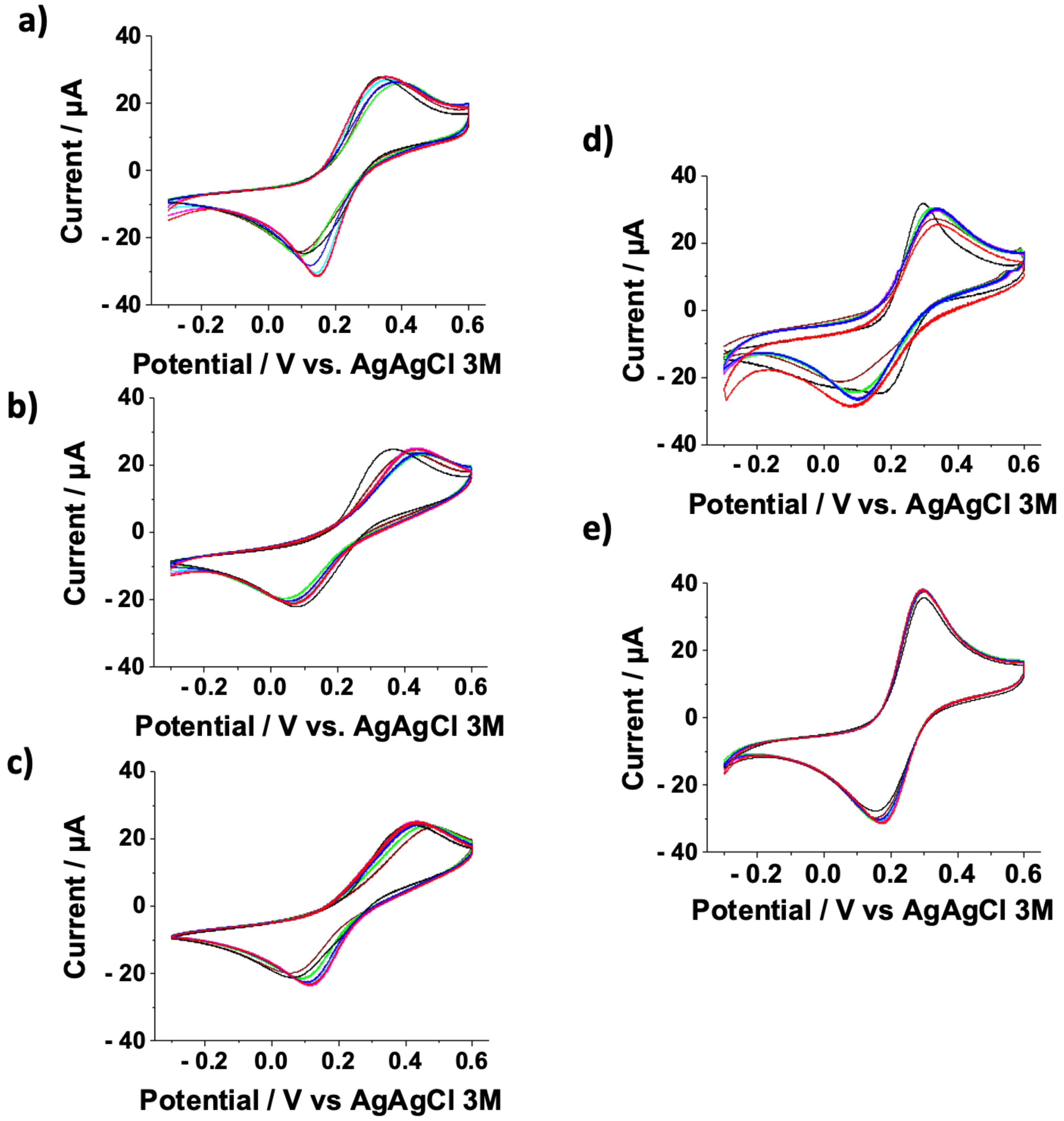
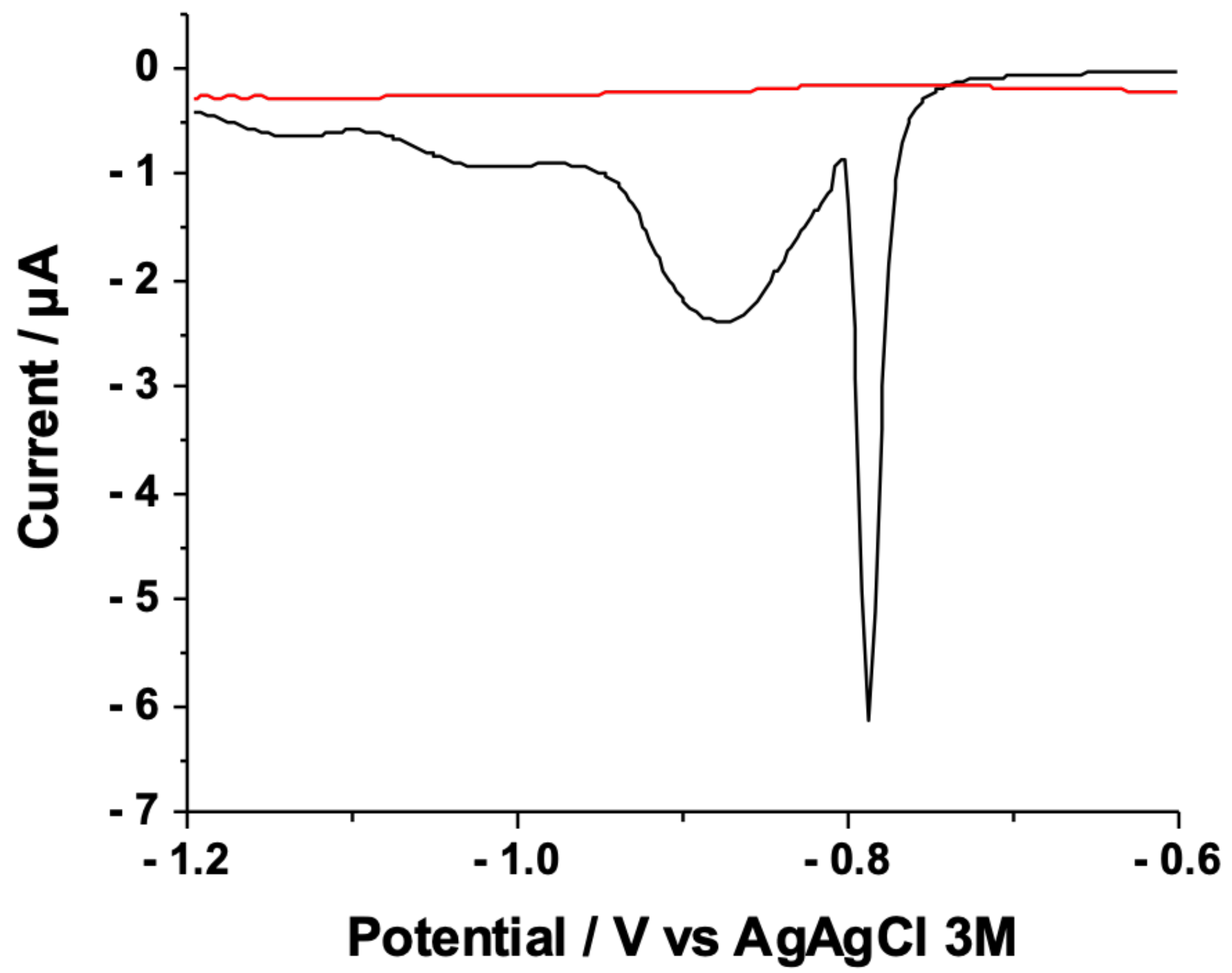
3.1. Cyclic Voltammetry Analyses on Au-SAMs
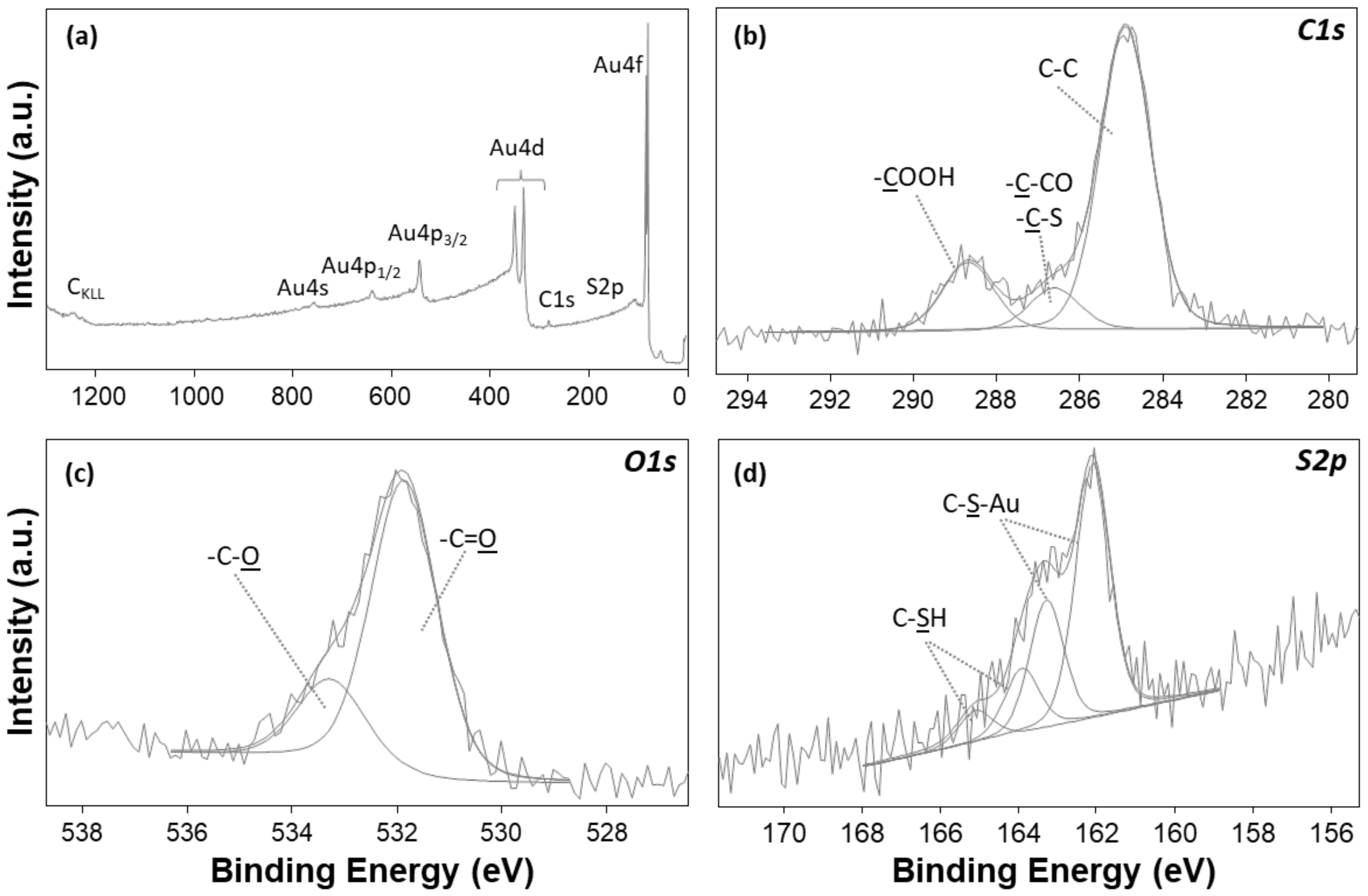
3.2. X-ray Photoelectron Spectroscopy (XPS) Characterization of Conformational Rearrangements of SAM Supramolecular Structure
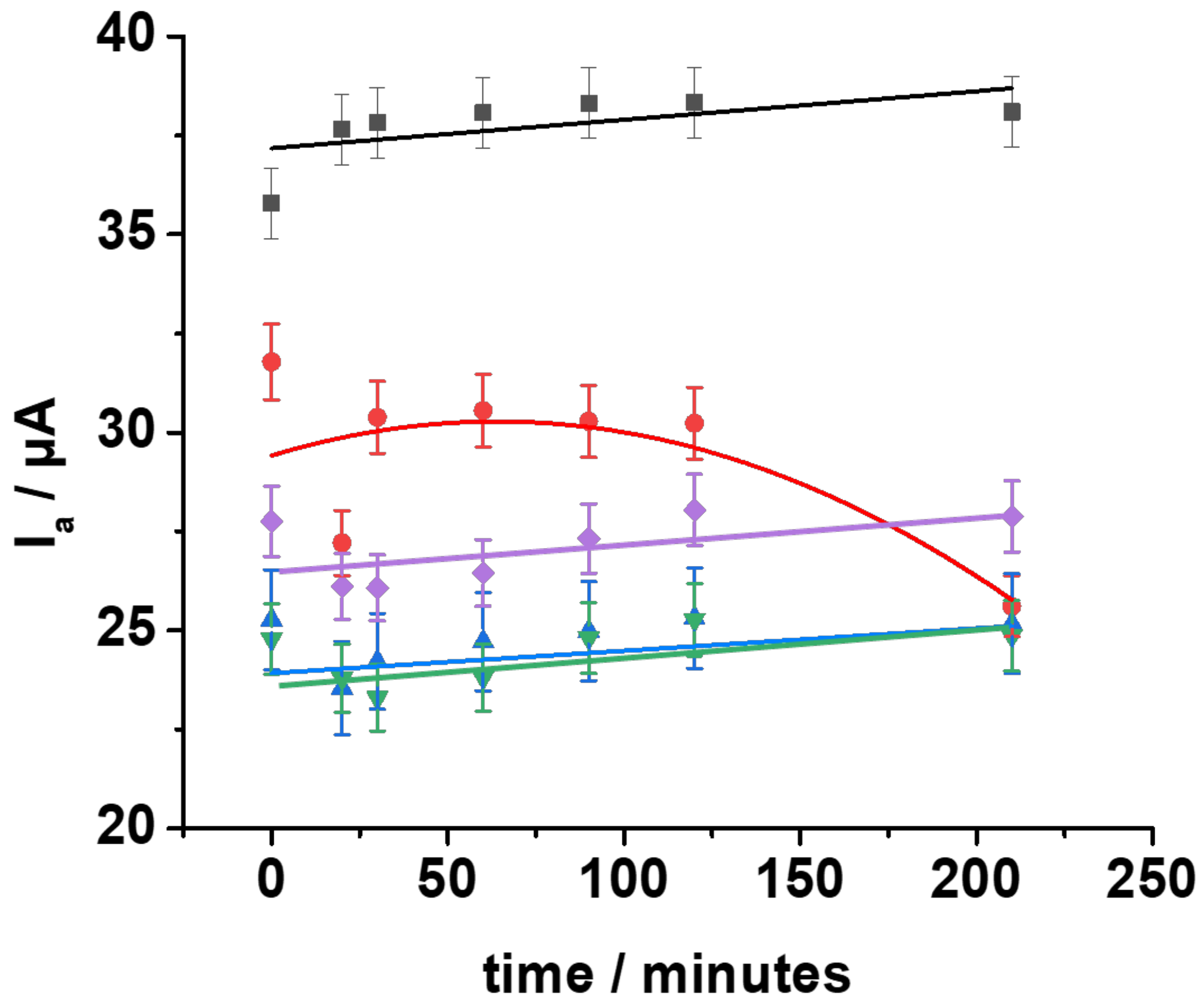
3.3. Surface Coverage Evaluation
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wink, T.; Van Zuilen, S.J.; Bult, A.; Van Bennekom, W.P. Self-assembled monolayers for biosensors. Analyst 1997, 122, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Bollella, P.; Hibino, Y.; Kano, K.; Gorton, L.; Antiochia, R. Highly Sensitive Membraneless Fructose Biosensor Based on Fructose Dehydrogenase Immobilized onto Aryl Thiol Modified Highly Porous Gold Electrode: Characterization and Application in Food Samples. Anal. Chem. 2018, 90, 12131–12136. [Google Scholar] [CrossRef] [PubMed]
- Sarcina, L.; Mangiatordi, G.F.; Torricelli, F.; Bollella, P.; Gounani, Z.; Österbacka, R.; Macchia, E.; Torsi, L. Surface plasmon resonance assay for label-free and selective detection of hiv-1 p24 protein. Biosensors 2021, 11, 180. [Google Scholar] [CrossRef] [PubMed]
- Poitras, C.; Tufenkji, N. A QCM-D-based biosensor for E. coli O157:H7 highlighting the relevance of the dissipation slope as a transduction signal. Biosens. Bioelectron. 2009, 24, 2137–2142. [Google Scholar] [CrossRef] [PubMed]
- Cotrone, S.; Ambrico, M.; Toss, H.; Angione, M.D.; Magliulo, M.; Mallardi, A.; Berggren, M.; Palazzo, G.; Horowitz, G.; Ligonzo, T.; et al. Phospholipid film in electrolyte-gated organic field-effect transistors. Org. Electron. 2012, 13, 638–644. [Google Scholar] [CrossRef] [Green Version]
- Cioffi, N.; Torsi, L.; Losito, I.; Sabbatini, L.; Zambonin, P.G.; Bleve-Zacheo, T. Nanostructured palladium–polypyrrole composites electrosynthesised from organic solvents. Electrochim. Acta 2001, 46, 4205–4211. [Google Scholar] [CrossRef]
- Torsi, L.; Dodabalapur, A.; Lovinger, A.J.; Katz, H.E.; Ruel, R.; Davis, D.D.; Baldwin, K.W. Rapid thermal processing of alpha.-hexathienylene thin-film transistors. Chem. Mater. 1995, 7, 2247–2251. [Google Scholar] [CrossRef]
- Macchia, E.; Romele, P.; Manoli, K.; Ghittorelli, M.; Magliulo, M.; Kovács-Vajna, Z.M.; Torricelli, F.; Torsi, L. Ultra-sensitive protein detection with organic electrochemical transistors printed on plastic substrates. Flex. Print. Electron. 2018, 3, 034002. [Google Scholar] [CrossRef]
- Nuzzo, R.G.; Allara, D.L. Adsorption of Bifunctional Organic Disulfides on Gold Surfaces. J. Am. Chem. Soc. 1983, 105, 4481–4483. [Google Scholar] [CrossRef]
- Lewis, P.A.; Smith, R.K.; Kelly, K.F.; Bumm, L.A.; Reed, S.M.; Clegg, R.S.; Gunderson, J.D.; Hutchison, J.E.; Weiss, P.S. The role of buried hydrogen bonds in self-assembled mixed composition thiols on Au{111}. J. Phys. Chem. B 2001, 105, 10630–10636. [Google Scholar] [CrossRef]
- Clegg, R.S.; Reed, S.M.; Smith, R.K.; Barron, B.L.; Rear, J.A.; Hutchison, J.E. Interplay of lateral and tiered interactions in stratified self-organized molecular assemblies. Langmuir 1999, 15, 8876–8883. [Google Scholar] [CrossRef]
- Love, J.C.; Estroff, L.A.; Kriebel, J.K.; Nuzzo, R.G.; Whitesides, G.M. Self-Assembled Monolayers of Thiolates on Metals as a Form of Nanotechnology. Chem. Rev. 2005, 105, 1103–1170. [Google Scholar] [CrossRef] [PubMed]
- Dai, Z.; Ju, H. Effect of chain length on the surface properties of ω-carboxy alkanethiol self-assembled monolayers. Phys. Chem. Chem. Phys. 2001, 3, 3769–3773. [Google Scholar] [CrossRef]
- Herrwerth, S.; Eck, W.; Reinhardt, S.; Grunze, M. Factors that Determine the Protein Resistance of Oligoether Self-Assembled Monolayers—Internal Hydrophilicity, Terminal Hydrophilicity, and Lateral Packing Density. J. Am. Chem. Soc. 2003, 125, 9359–9366. [Google Scholar] [CrossRef]
- Badia, A.; Lennox, R.B.; Reven, L. A dynamic view of self-assembled monolayers. Acc. Chem. Res. 2000, 33, 475–481. [Google Scholar] [CrossRef]
- Malinsky, M.D.; Kelly, K.L.; Schatz, G.C.; Van Duyne, R.P. Chain Length Dependence and Sensing Capabilities of the Localized Surface Plasmon Resonance of Silver Nanoparticles Chemically Modified with Alkanethiol Self-Assembled Monolayers. J. Am. Chem. Soc. 2001, 123, 1471–1482. [Google Scholar] [CrossRef]
- Martins, M.C.L.; Fonseca, C.; Barbosa, M.A.; Ratner, B.D. Albumin adsorption on alkanethiols self-assembled monolayers on gold electrodes studied by chronopotentiometry. Biomaterials 2003, 24, 3697–3706. [Google Scholar] [CrossRef]
- Cabarcos, O.M.; Shaporenko, A.; Weidner, T.; Uppili, S.; Dake, L.S.; Zharnikov, M.; Allara, D.L. Physical and electronic structure effects of embedded dipoles in self-assembled monolayers: Characterization of mid-chain ester functionalized alkanethiols on Au{111}. J. Phys. Chem. C 2008, 112, 10842–10854. [Google Scholar] [CrossRef]
- Clegg, R.S.; Hutchison, J.E. Hydrogen-bonding, self-assembled monolayers: Ordered molecular films for study of through-peptide electron transfer. Langmuir 1996, 12, 5239–5243. [Google Scholar] [CrossRef]
- Mendes, R.K.; Carvalhal, R.F.; Kubota, L.T. Effects of different self-assembled monolayers on enzyme immobilization procedures in peroxidase-based biosensor development. J. Electroanal. Chem. 2008, 612, 164–172. [Google Scholar] [CrossRef]
- Nuzzo, R.G.; Korenic, E.M.; Dubois, L.H. Studies of the temperature-dependent phase behavior of long chain n-alkyl thiol monolayers on gold. J. Chem. Phys. 1990, 93, 767–773. [Google Scholar] [CrossRef]
- Blasi, D.; Sarcina, L.; Tricase, A.; Stefanachi, A.; Leonetti, F.; Alberga, D.; Mangiatordi, G.F.; Manoli, K.; Scamarcio, G.; Picca, R.A.; et al. Enhancing the sensitivity of biotinylated surfaces by tailoring the design of the mixed self-assembled monolayers synthesis. ACS Omega 2020, 5, 16762–16771. [Google Scholar] [CrossRef] [PubMed]
- Welch, N.G.; Scoble, J.A.; Muir, B.W.; Pigram, P.J. Orientation and characterization of immobilized antibodies for improved immunoassays (Review). Biointerphases 2017, 12, 02D301. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arakawa, T.; Hobara, D.; Yamamoto, M.; Kakiuchi, T. Effect of the charged state of gold substrates on the surface composition of two-component alkanethiol self-assembled monolayers. Electrochem. Commun. 2005, 7, 848–852. [Google Scholar] [CrossRef]
- Bhadra, P.; Shajahan, M.S.; Bhattacharya, E.; Chadha, A. Studies on varying n-alkanethiol chain lengths on a gold coated surface and their effect on antibody-antigen binding efficiency. RSC Adv. 2015, 5, 80480–80487. [Google Scholar] [CrossRef]
- Briand, E.; Gu, C.; Boujday, S.; Salmain, M.; Herry, J.M.; Pradier, C.M. Functionalisation of gold surfaces with thiolate SAMs: Topography/bioactivity relationship—A combined FT-RAIRS, AFM and QCM investigation. Surf. Sci. 2007, 601, 3850–3855. [Google Scholar] [CrossRef]
- Patel, N.; Davies, M.C.; Heaton, R.J.; Roberts, C.J.; Tendler, S.J.B.; Williams, P.M. A scanning probemicroscopy study of the physisorption and chemisorption of protein molecules onto carboxylate terminated self-assembledmonolayers. Appl. Phys. A Mater. Sci. Process. 1998, 66, 569–574. [Google Scholar] [CrossRef]
- Campuzano, S.; Gálvez, R.; Pedrero, M.; De Villena, F.J.M.; Pingarrón, J.M. Preparation, characterization and application of alkanethiol self-assembled monolayers modified with tetrathiafulvalene and glucose oxidase at a gold disk electrode. J. Electroanal. Chem. 2002, 526, 92–100. [Google Scholar] [CrossRef]
- Lomant, A.J.; Fairbanks, G. Chemical probes of extended biological structures: Synthesis and properties of the cleavable protein cross-linking reagent [35S] dithiobis (succinimidyl propionate). J. Mol. Biol. 1976, 104, 243–261. [Google Scholar] [CrossRef]
- Staros, J.V.; Wright, R.W.; Swingle, D.M. Enhancement by N-hydroxysulfosuccinimide of water-soluble carbodiimide-mediated coupling reactions. Anal. Biochem. 1986, 156, 220–222. [Google Scholar] [CrossRef]
- Cuatrecasas, P.; Parikh, I. Adsorbents for affinity chromatography. Use of N-hydroxysuccinimide esters of agarose. Biochemistry 1972, 11, 2291–2299. [Google Scholar] [CrossRef] [PubMed]
- Tricase, A.; Blasi, D.; Favia, A.; Stefanachi, A.; Leonetti, F.; Colafemmina, G.; Torsi, L.; Scamarcio, G. Surface composition of mixed self-assembled monolayers on Au by infrared attenuated total reflection spectroscopy. Appl. Surf. Sci. 2021, 559, 149883. [Google Scholar] [CrossRef]
- Sam, S.; Touahir, L.; Salvador Andresa, J.; Allongue, P.; Chazalviel, J.N.; Gouget-Laemmel, A.C.; De Villeneuve, C.H.; Moraillon, A.; Ozanam, F.; Gabouze, N.; et al. Semiquantitative study of the EDC/NHS activation of acid terminal groups at modified porous silicon surfaces. Langmuir 2010, 26, 809–814. [Google Scholar] [CrossRef] [PubMed]
- Yan, Q.; Zheng, H.N.; Jiang, C.; Li, K.; Xiao, S.J. EDC/NHS activation mechanism of polymethacrylic acid: Anhydride versus NHS-ester. RSC Adv. 2015, 5, 69939–69947. [Google Scholar] [CrossRef]
- Eckermann, A.L.; Feld, D.J.; Shaw, J.A.; Meade, T.J. Electrochemistry of redox-active self-assembled monolayers. Coord. Chem. Rev. 2010, 254, 1769–1802. [Google Scholar] [CrossRef] [Green Version]
- Kim, S.J.; Gobi, K.V.; Iwasaka, H.; Tanaka, H.; Miura, N. Novel miniature SPR immunosensor equipped with all-in-one multi-microchannel sensor chip for detecting low-molecular-weight analytes. Biosens. Bioelectron. 2007, 23, 701–707. [Google Scholar] [CrossRef]
- Casalini, S.; Bortolotti, C.A.; Leonardi, F.; Biscarini, F. Self-assembled monolayers in organic electronics. Chem. Soc. Rev. 2017, 46, 40–71. [Google Scholar] [CrossRef]
- Tominaga, M.; Ohira, A.; Yamaguchi, Y.; Kunitake, M. Electrochemical, AFM and QCM studies on ferritin immobilized onto a self-assembled monolayer-modified gold electrode. J. Electroanal. Chem. 2004, 566, 323–329. [Google Scholar] [CrossRef]
- Dodabalapur, A.; Katz, H.E.; Torsi, L. Molecular orbital energy level engineering in organic transistors. Adv. Mater. 1996, 8, 853–855. [Google Scholar] [CrossRef]
- Longano, D.; Ditaranto, N.; Sabbatini, L.; Torsi, L.; Cioffi, N. Synthesis and antimicrobial activity of copper nanomaterials. In Nano-Antimicrobials; Cioffi, N., Rai, M., Eds.; Springer: Berlin/Heidelberg, Germany, 2012; pp. 85–117. ISBN 978-3-642-24428-5. [Google Scholar]
- Singh, M.; Manoli, K.; Tiwari, A.; Ligonzo, T.; Di Franco, C.; Cioffi, N.; Palazzo, G.; Scamarcio, G.; Torsi, L. The double layer capacitance of ionic liquids for electrolyte gating of ZnO thin film transistors and effect of gate electrodes. J. Mater. Chem. C 2017, 5, 3509–3518. [Google Scholar] [CrossRef]
- Magliulo, M.; De Tullio, D.; Vikholm-Lundin, I.; Albers, W.M.; Munter, T.; Manoli, K.; Palazzo, G.; Torsi, L. Label-free C-reactive protein electronic detection with an electrolyte-gated organic field-effect transistor-based immunosensor. Anal. Bioanal. Chem. 2016, 408, 3943–3952. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.J.; Huang, M.J.; Ang, J.J.; Liu, E.T.; Desai, K.V. Self-assembled monolayer-assisted silicon nanowire biosensor for detection of protein-DNA interactions in nuclear extracts from breast cancer cell. Biosens. Bioelectron. 2011, 26, 3233–3239. [Google Scholar] [CrossRef] [PubMed]
- Ladd, J.; Boozer, C.; Yu, Q.; Chen, S.; Homola, J.; Jiang, S. DNA-Directed Protein Immobilization on Mixed Self-Assembled Monolayers via a Streptavidin Bridge. Langmuir 2004, 20, 8090–8095. [Google Scholar] [CrossRef] [PubMed]
- Holzer, B.; Manoli, K.; Ditaranto, N.; Macchia, E.; Tiwari, A.; Di Franco, C.; Scamarcio, G.; Palazzo, G.; Torsi, L. Characterization of Covalently Bound Anti-Human Immunoglobulins on Self-Assembled Monolayer Modified Gold Electrodes. Adv. Biosyst. 2017, 1, 1700055. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Prime, K.L.; Whitesides, G.M. Self-Assembled Organic Monolayers: Model Systems for Studying Adsorption of Proteins at Aurfaces. Science 1991, 252, 1164–1167. [Google Scholar] [CrossRef] [Green Version]
- Arima, Y.; Iwata, H. Effect of wettability and surface functional groups on protein adsorption and cell adhesion using well-defined mixed self-assembled monolayers. Biomaterials 2007, 28, 3074–3082. [Google Scholar] [CrossRef]
- Macchia, E.; Manoli, K.; Holzer, B.; Di Franco, C.; Ghittorelli, M.; Torricelli, F.; Alberga, D.; Mangiatordi, G.F.; Palazzo, G.; Scamarcio, G.; et al. Single-molecule detection with a millimetre-sized transistor. Nat. Commun. 2018, 9, 3223. [Google Scholar] [CrossRef]
- Macchia, E.; Picca, R.A.; Manoli, K.; Di Franco, C.; Blasi, D.; Sarcina, L.; Ditaranto, N.; Cioffi, N.; Österbacka, R.; Scamarcio, G.; et al. About the amplification factors in organic bioelectronic sensors. Mater. Horiz. 2020, 7, 999–1013. [Google Scholar] [CrossRef] [Green Version]
- Macchia, E.; Sarcina, L.; Driescher, C.; Gounani, Z.; Tewari, A.; Osterbacka, R.; Palazzo, G.; Tricase, A.; Kovacs Vajna, Z.M.; Viola, F.; et al. Single-Molecule Bioelectronic Label-Free Assay of both Protein and Genomic Markers of Pancreatic Mucinous Cysts’ in Whole Blood Serum. Adv. Electron. Mater. 2021, 7, 2100304. [Google Scholar] [CrossRef]
- Besselink, G.A.J.; Beugeling, T.; Bantjes, A. N-Hydroxysuccinimide-activated glycine-sepharose. Appl. Biochem. Biotechnol. 1993, 43, 227. [Google Scholar] [CrossRef]
- Jennings, G.K.; Munro, J.C.; Yong, T.-H.; Laibinis, P.E. Effect of Chain Length on the Protection of Copper by n-Alkanethiols. Langmuir 1998, 14, 6130–6139. [Google Scholar] [CrossRef]
- Schoenfisch, M.H.; Pemberton, J.E. Air Stability of Alkanethiol Self-Assembled Monolayers on Silver and Gold Surfaces. J. Am. Chem. Soc. 1998, 120, 4502–4513. [Google Scholar] [CrossRef]
- Houseman, B.T.; Gawalt, E.S.; Mrksich, M. Maleimide-Functionalized Self-Assembled Monolayers for the Preparation of Peptide and Carbohydrate Biochips. Langmuir 2003, 19, 1522–1531. [Google Scholar] [CrossRef]
- Howarter, J.A.; Youngblood, J.P. Optimization of silica silanization by 3-aminopropyltriethoxysilane. Langmuir 2006, 22, 11142–11147. [Google Scholar] [CrossRef] [PubMed]
- Ito, Y.; Virkar, A.A.; Mannsfeld, S.; Oh, J.H.; Toney, M.; Locklin, J.; Bao, Z. Crystalline Ultrasmooth Self-Assembled Monolayers of Alkylsilanes for Organic Field-Effect Transistors. J. Am. Chem. Soc. 2009, 131, 9396–9404. [Google Scholar] [CrossRef] [PubMed]
- Okabayashi, N.; Paulsson, M.; Ueba, H.; Konda, Y.; Komeda, T. Inelastic tunneling spectroscopy of alkanethiol molecules: High-resolution spectroscopy and theoretical simulations. Phys. Rev. Lett. 2010, 104, 077801. [Google Scholar] [CrossRef]
- Liu, G.-Y.; Xu, S.; Qian, Y. Nanofabrication of Self-Assembled Monolayers Using Scanning Probe Lithography. Acc. Chem. Res. 2000, 33, 457–466. [Google Scholar] [CrossRef]
- Milosevic, M.; Milosevic, V.; Berets, S.L. Grazing Angle Attenuated Total Reflection Spectroscopy: Fields at the Interface and Source of the Enhancement. Appl. Spectrosc. 2007, 61, 530–536. [Google Scholar] [CrossRef]
- Mulcahy, M.; Berets, S.; Milosevic, M.; Michl, J. Enhanced Sensitivity in Single-Reflection Spectroscopy of Organic Monolayers on Metal Substrates (Pseudo-ATR). J. Phys. Chem. B 2004, 108, 1519–1521. [Google Scholar] [CrossRef]
- Tour, J.M.; Jones, L.; Pearson, D.L.; Lamba, J.J.S.; Burgin, T.P.; Whitesides, G.M.; Allara, D.L.; Parikh, A.N.; Atre, S. Self-Assembled Monolayers and Multilayers of Conjugated Thiols, alpha.,.omega.-Dithiols, and Thioacetyl-Containing Adsorbates. Understanding Attachments between Potential Molecular Wires and Gold Surfaces. J. Am. Chem. Soc. 1995, 117, 9529–9534. [Google Scholar] [CrossRef]
- Tricase, A.; Stefanachi, A.; Picca, R.A.; Macchia, E.; Favia, A.; Leonetti, F.; Scamarcio, G.; Blasi, D.; Bollella, P.; Torsi, L. Negatively charged ions to probe self-assembled monolayer reorganization driven by interchain interactions. J. Mater. Chem. C 2021, 9, 10935–10943. [Google Scholar] [CrossRef]
- Liu, B.T.; Hung, T.Y.; Gorji, N.E.; Mosavi, A.H. Fabrication and characterization of Cesium-doped Tungstate nanorods for Near-Infrared light absorption in dye sensitized solar cells. Results Phys. 2021, 29, 104804. [Google Scholar] [CrossRef]
- Carvalhal, R.F.; Freire, R.S.; Kubota, L.T. Polycrystalline gold electrodes: A comparative study of pretreatment procedures used for cleaning and thiol self-assembly monolayer formation. Electroanalysis 2005, 17, 1251–1259. [Google Scholar] [CrossRef]
- Sabbatini, L.; Malitesta, C.; De Giglio, E.; Losito, I.; Torsi, L.; Zambonin, P.G. Electrosynthesised thin polymer films: The role of XPS in the design of application oriented innovative materials. J. Electron. Spectros. Relat. Phenom. 1999, 100, 35–53. [Google Scholar] [CrossRef]
- Tabanlıgil Calam, T.; Hasdermir, E. Comparative characterizations of self-assembled monolayers of 1, 6-hexanedithiol and 1-hexanethiol formed on polycrystalline gold electrode. C. R. Acad. Bulg. Sci. 2019, 72, 316–326. [Google Scholar] [CrossRef]
- Tam-Chang, S.W.; Biebuyck, H.A.; Whitesides, G.M.; Jeon, N.; Nuzzo, R.G. Self-Assembled Monolayers on Gold Generated from Alkanethiols with the Structure RNHCOCH2SH. Langmuir 1995, 11, 4371–4382. [Google Scholar] [CrossRef]
- Hagenström, H.; Esplandi, M.J.; Kolb, D.M. Functionalized Self-Assembled Alkanethiol Monolayers on Au (111) Electrodes: 2. Silver Electrodeposition. Langmuir 2001, 17, 839–848. [Google Scholar] [CrossRef]
- Castner, D.G.; Hinds, K.; Grainger, D.W. X-ray Photoelectron Spectroscopy Sulfur 2p Study of Organic Thiol and Disulfide Binding Interactions with Gold Surfaces. Langmuir 1996, 12, 5083–5086. [Google Scholar] [CrossRef]
- Cavalleri, O.; Oliveri, L.; Daccà, A.; Parodi, R.; Rolandi, R. XPS measurements on l-cysteine and 1-octadecanethiol self-assembled films: A comparative study. Appl. Surf. Sci. 2001, 175–176, 357–362. [Google Scholar] [CrossRef]
- Trasatti, S.; Petrii, O.A. Real surface area measurements in electrochemistry. Pure Appl. Chem. 1991, 63, 711–734. [Google Scholar] [CrossRef]
- Sumi, T.; Uosaki, K. Electrochemical oxidative formation and reductive desorption of a self-assembled monolayer of decanethiol on a Au (111) surface in KOH ethanol solution. J. Phys. Chem. B 2004, 108, 6422–6428. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Suroviec, A.H. Determining surface coverage of self-assembled monolayers on gold electrodes. Chem. Educ. 2012, 17, 83–85. [Google Scholar] [CrossRef]
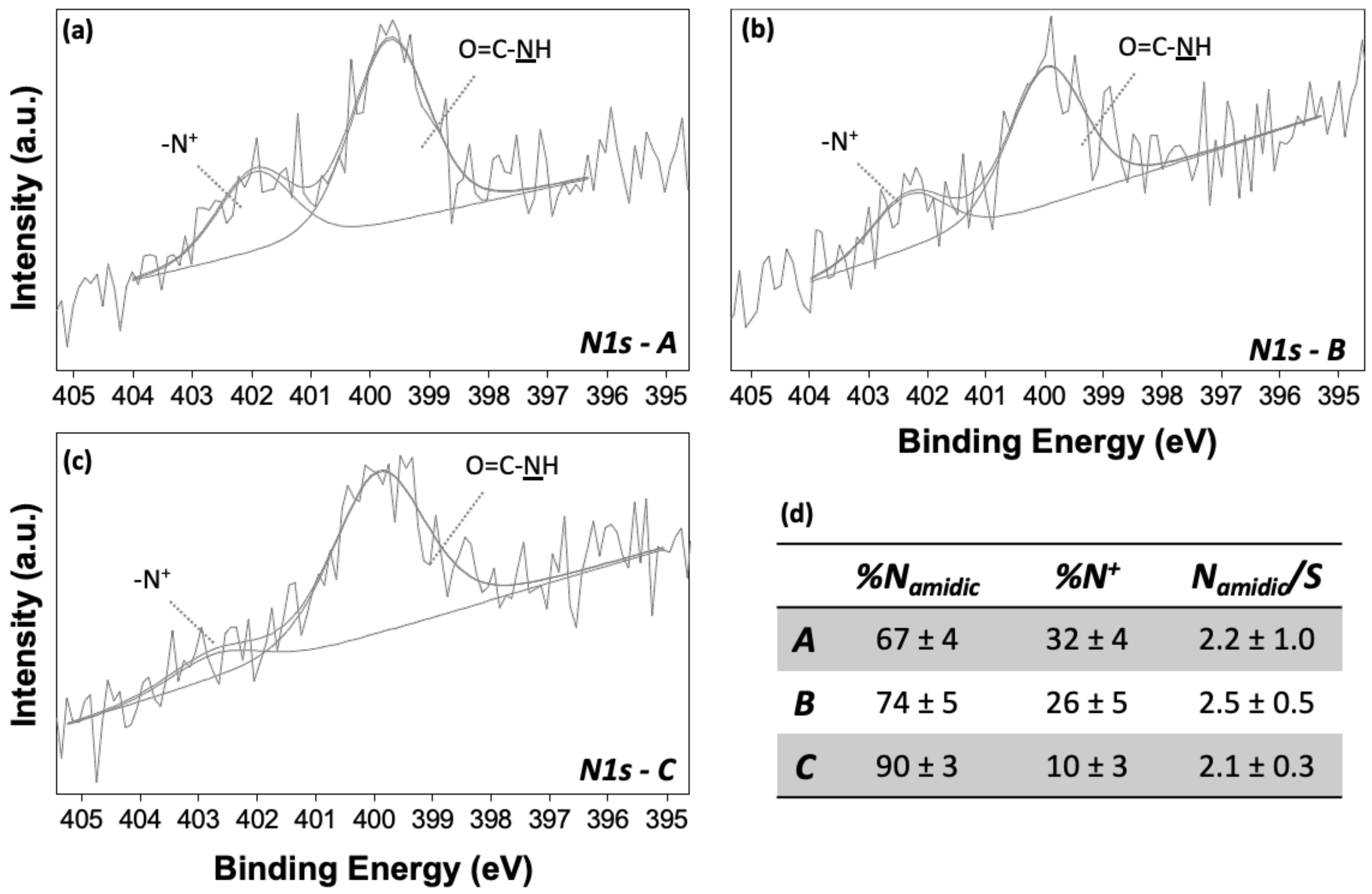
| %C | %N | %O | %S | %Au | |
|---|---|---|---|---|---|
| Bare Au | 18 ± 2 | n.d. | 8.4 ± 1.9 | n.d. | 73.8 ± 1.1 |
| 3MPA SAM | 36 ± 3 | n.d. | 12.7 ± 1.1 | 3.1 ± 0.5 | 48 ± 4 |
| Protocol A SAM | 30 ± 1 | 3.3 ± 1.7 | 17 ± 4 | 4.6 ± 0.5 | 45 ± 4 |
| Protocol B SAM | 32.1 ± 0.5 | 3.1 ± 0.5 | 18 ± 3 | 3.7 ± 0.5 | 43 ± 3 |
| Protocol C SAM | 29 ± 5 | 2.8 ± 0.7 | 14.8 ± 1.0 | 3.1 ± 0.5 | 50 ± 6 |
| %C | %N | %O | %S | %Au | |
|---|---|---|---|---|---|
| Protocol C SAM after reductive desorption | 23.3 ± 1.4 | 1.6 ± 1.4 | 3.9 ± 1.7 | n.d. | 71 ± 2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tricase, A.; Imbriano, A.; Ditaranto, N.; Macchia, E.; Picca, R.A.; Blasi, D.; Torsi, L.; Bollella, P. Electrochemical and X-ray Photoelectron Spectroscopy Surface Characterization of Interchain-Driven Self-Assembled Monolayer (SAM) Reorganization. Nanomaterials 2022, 12, 867. https://doi.org/10.3390/nano12050867
Tricase A, Imbriano A, Ditaranto N, Macchia E, Picca RA, Blasi D, Torsi L, Bollella P. Electrochemical and X-ray Photoelectron Spectroscopy Surface Characterization of Interchain-Driven Self-Assembled Monolayer (SAM) Reorganization. Nanomaterials. 2022; 12(5):867. https://doi.org/10.3390/nano12050867
Chicago/Turabian StyleTricase, Angelo, Anna Imbriano, Nicoletta Ditaranto, Eleonora Macchia, Rosaria Anna Picca, Davide Blasi, Luisa Torsi, and Paolo Bollella. 2022. "Electrochemical and X-ray Photoelectron Spectroscopy Surface Characterization of Interchain-Driven Self-Assembled Monolayer (SAM) Reorganization" Nanomaterials 12, no. 5: 867. https://doi.org/10.3390/nano12050867
APA StyleTricase, A., Imbriano, A., Ditaranto, N., Macchia, E., Picca, R. A., Blasi, D., Torsi, L., & Bollella, P. (2022). Electrochemical and X-ray Photoelectron Spectroscopy Surface Characterization of Interchain-Driven Self-Assembled Monolayer (SAM) Reorganization. Nanomaterials, 12(5), 867. https://doi.org/10.3390/nano12050867












