Origin of Ferroelectricity in BiFeO3-Based Solid Solutions
Abstract
1. Introduction
2. DFT Calculations
3. Results and Discussion
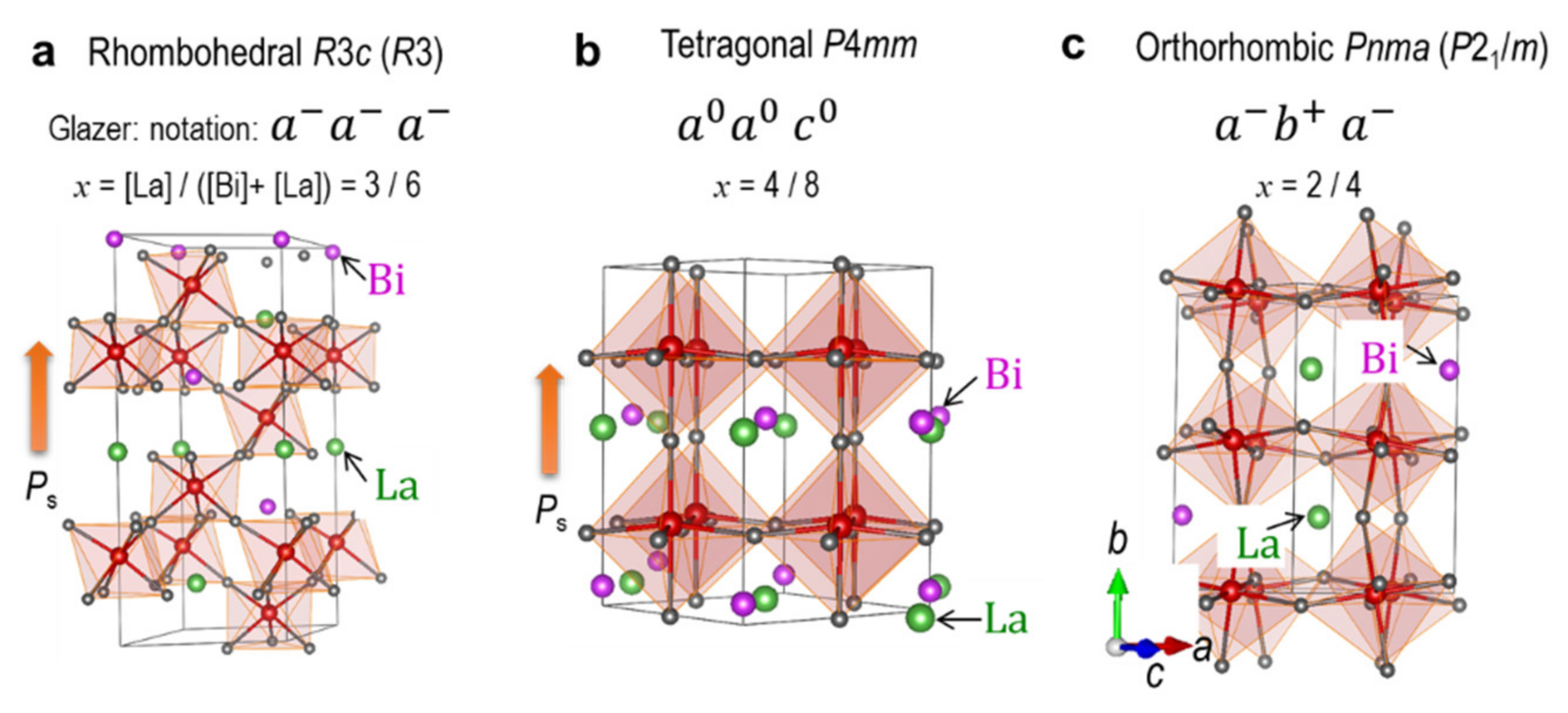
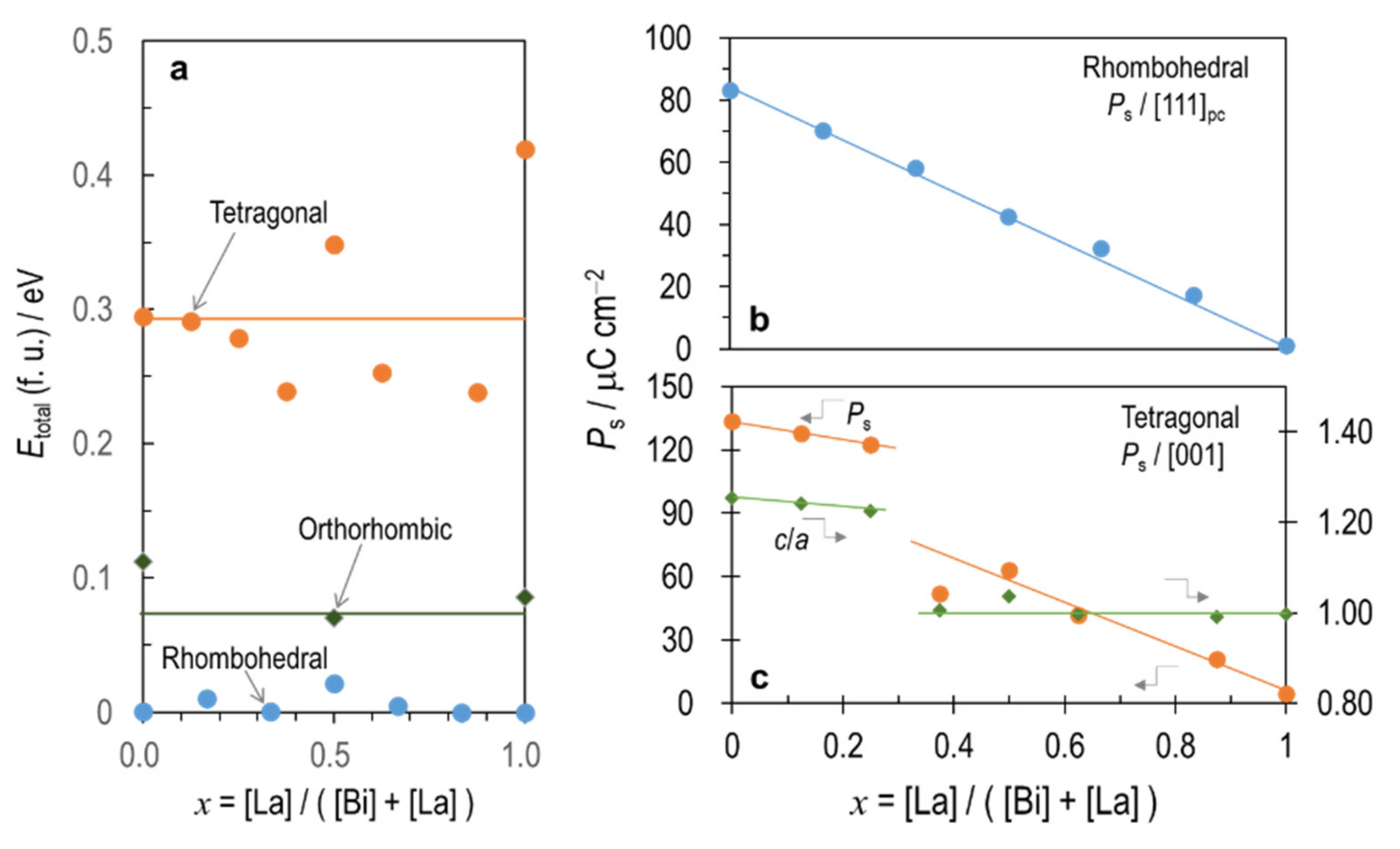
3.1. Ground-State Structures
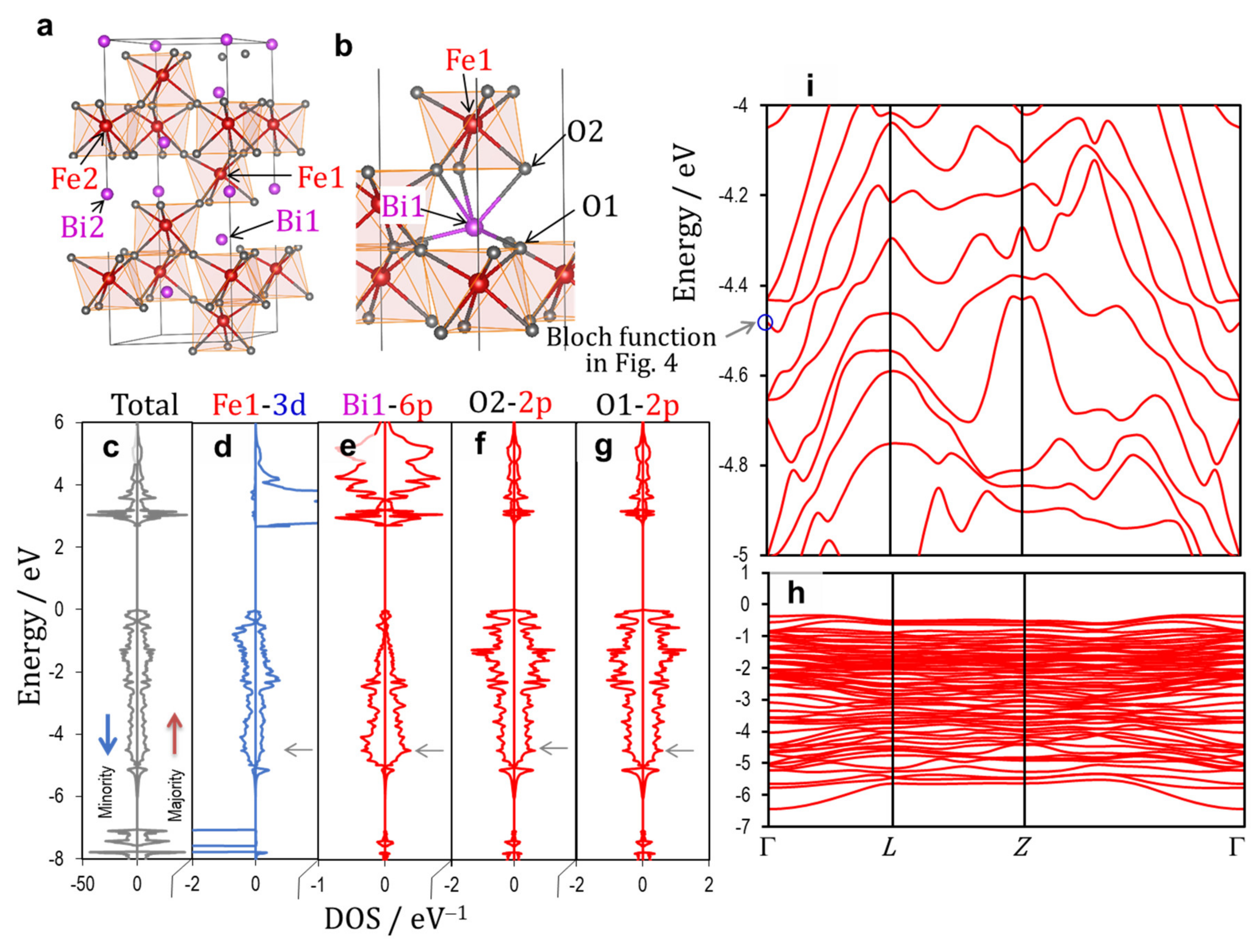
3.2. Electronic Structures
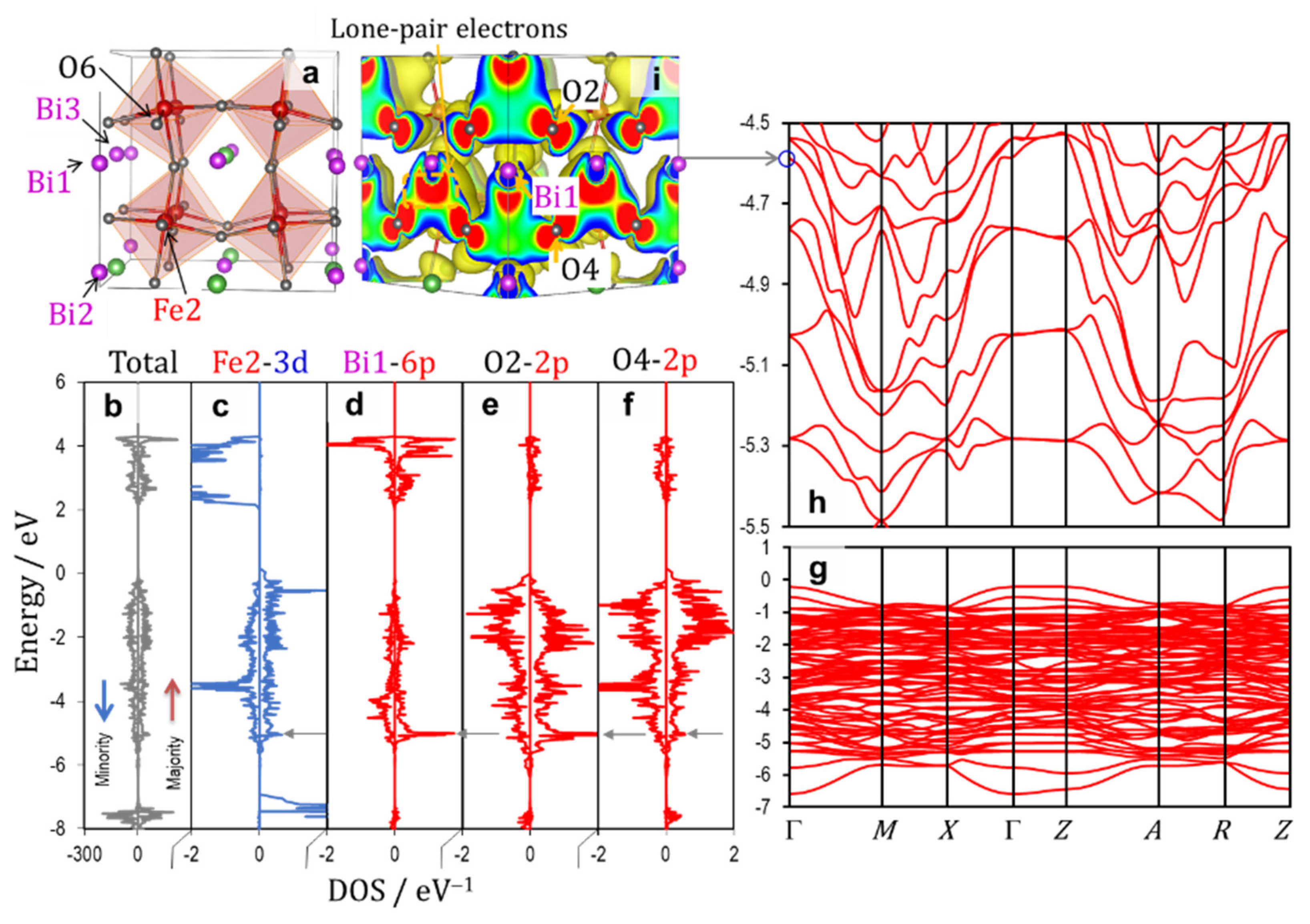
3.2.1. Ferroelectric Rhombohedral System
3.2.2. Ferroelectric Tetragonal System
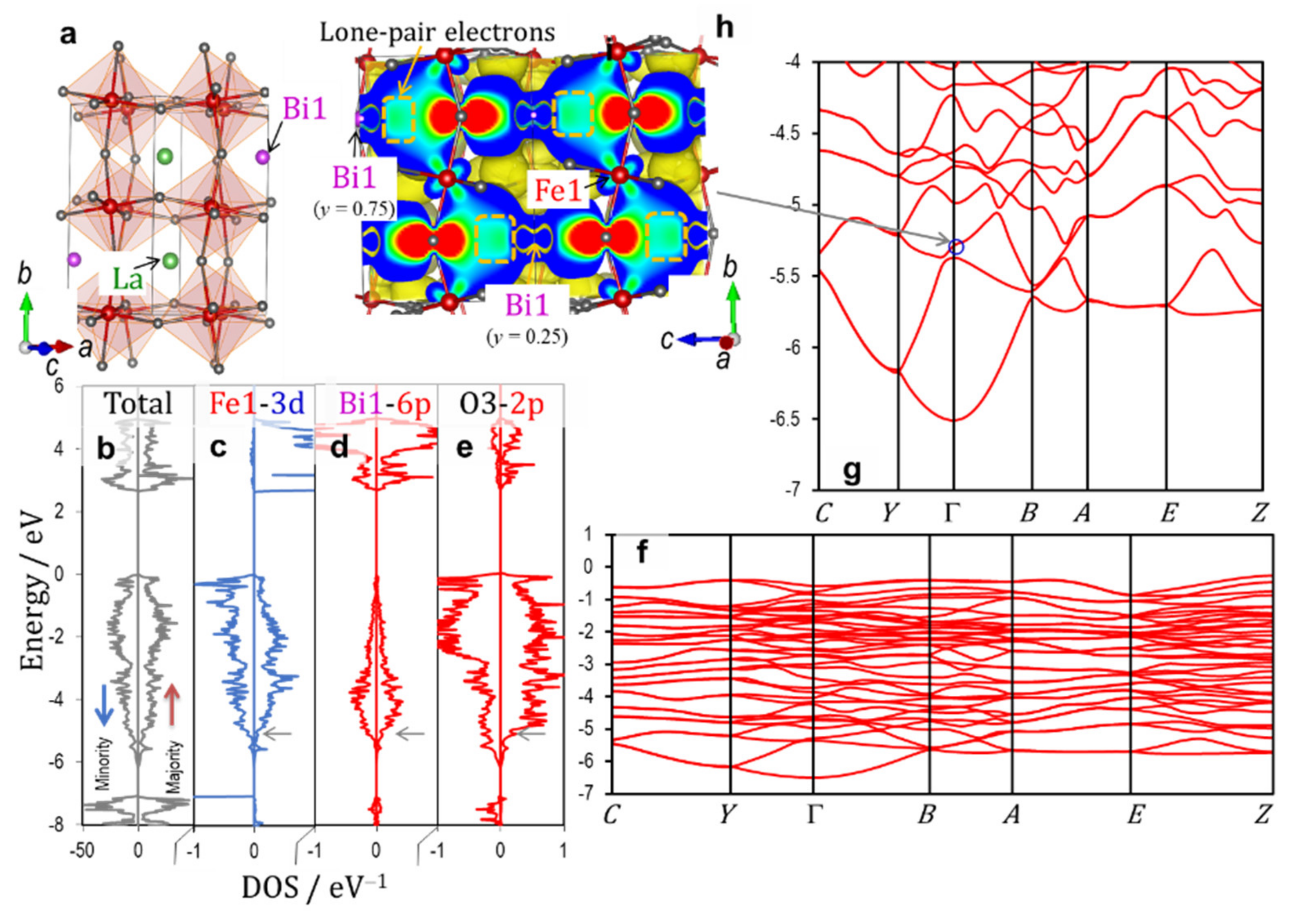
3.2.3. Paraelectric Orthorhombic System
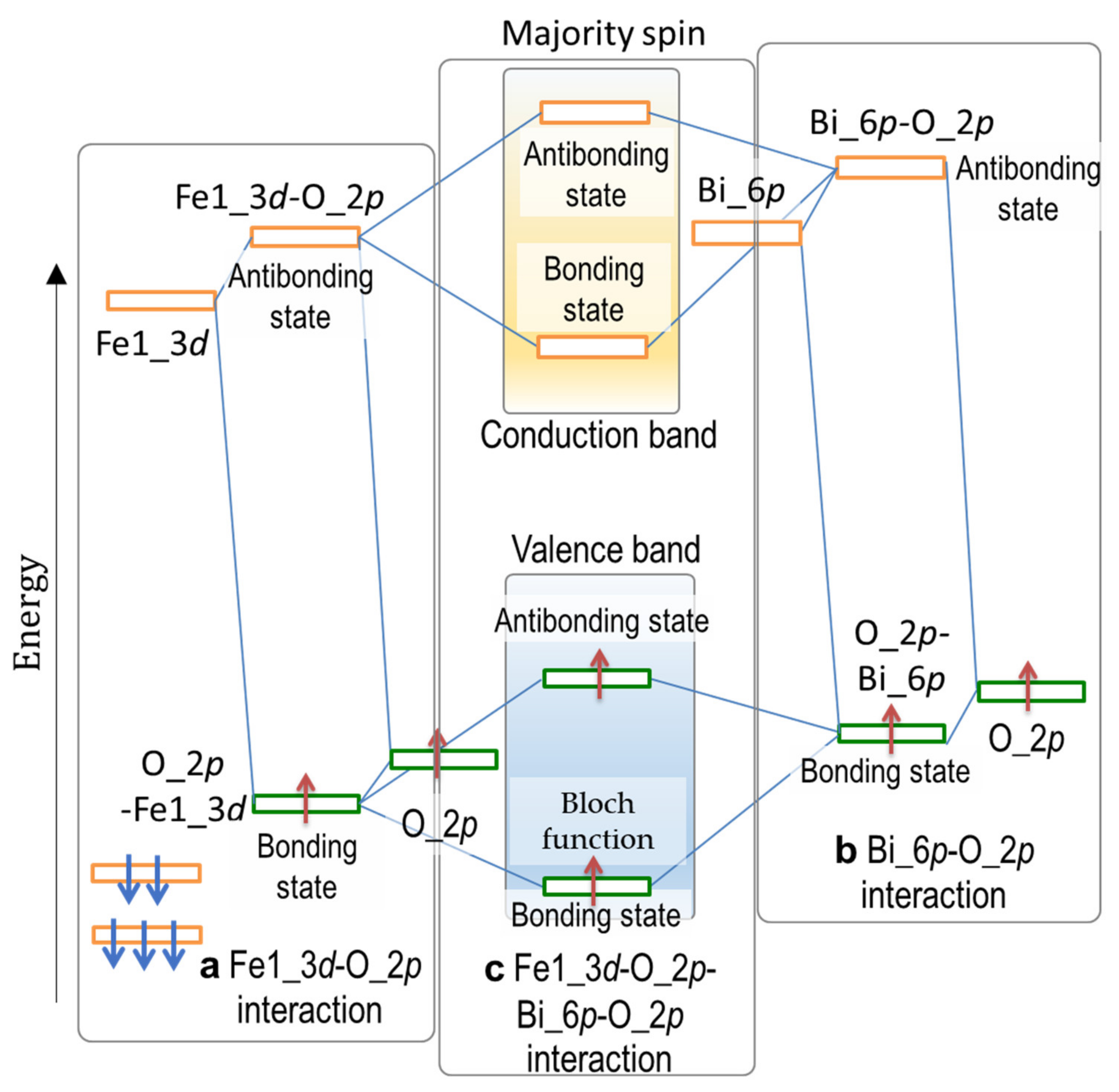
3.3. Factors Affecting Ferroelectricity
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bhide, V.G.; Deshmukh, K.G.; Hegde, M.S. Ferroelectric Properties of PbTiO3. Physica 1962, 28, 871–876. [Google Scholar] [CrossRef]
- Sawaguchi, E. Ferroelectricity versus Antiferroelectricity in the Solid Solutions of PbZrO3 and PbTiO3. J. Phys. Soc. Japan 1953, 8, 615–629. [Google Scholar] [CrossRef]
- Jaffe, B.; Roth, R.S.; Marzullo, S. Piezoelectric Properties of Lead Zirconate-Lead Titanate Solid-Solution Ceramics. J. Appl. Phys. 1954, 25, 809. [Google Scholar] [CrossRef]
- Noheda, B.; Gonzalo, J.; Cross, L.; Guo, R.; Park, S. Tetragonal-to-Monoclinic Phase Transition in a Ferroelectric Perovskite: The Structure of PbZr0.52Ti0.48O3. Phys. Rev. B 2000, 61, 8687–8695. [Google Scholar] [CrossRef]
- Kweon, S.-H.; Tani, K.; Kanda, K.; Nahm, S.; Kanno, I. Piezoelectric PZT Thin-Film Transformers with a Ring–Dot Structure. Jpn. J. Appl. Phys. 2020, 59, SPPD09. [Google Scholar] [CrossRef]
- Genenko, Y.A.; Glaum, J.; Hoffmann, M.J.; Albe, K. Mechanisms of Aging and Fatigue in Ferroelectrics. Mater. Sci. Eng. B Solid-State Mater. Adv. Technol. 2015, 192, 52–82. [Google Scholar] [CrossRef]
- Yamashita, T.; Takeshita, T.; Oouchi, A.; Kobayashi, T. Ultra-Thin PZT/Si Chip Integrated on Paper Substrates. Jpn. J. Appl. Phys. 2021, 60, SFFB12. [Google Scholar] [CrossRef]
- Fu, H.; Cohen, R.E. Polarization Rotation Mechanism for Ultrahigh Electromechanical Response in Single-Crystal Piezoelectrics. Nature 2000, 403, 281–283. [Google Scholar] [CrossRef]
- Damjanovic, D. A Morphotropic Phase Boundary System Based on Polarization Rotation and Polarization Extension. Appl. Phys. Lett. 2010, 97, 2010–2013. [Google Scholar] [CrossRef]
- Kutnjak, Z.; Petzelt, J.; Blinc, R. The Giant Electromechanical Response in Ferroelectric Relaxors as a Critical Phenomenon. Nature 2006, 441, 956–959. [Google Scholar] [CrossRef]
- Heitmann, A.A.; Rossetti, G.A. Thermodynamics of Ferroelectric Solid Solutions with Morphotropic Phase Boundaries. J. Am. Ceram. Soc. 2014, 97, 1661–1685. [Google Scholar] [CrossRef]
- Park, S.-E.; Shrout, T.R. Ultrahigh Strain and Piezoelectric Behavior in Relaxor Based Ferroelectric Single Crystals. J. Appl. Phys. 1997, 82, 1804. [Google Scholar] [CrossRef]
- Sun, E.; Cao, W. Relaxor-Based Ferroelectric Single Crystals: Growth, Domain Engineering, Characterization and Applications. Prog. Mater. Sci. 2014, 65, 124–210. [Google Scholar] [CrossRef]
- Luo, C.; Karaki, T.; Sun, Y.; Yamashita, Y.J.; Xu, J. Effect of Field Cooling AC Poling on Electrical and Physical Properties for Pb(Mg 1/3 Nb 2/3 )O3 -PbTiO3 -Based Single Crystals Manufactured by a Continuous-Feeding Bridgman Process. Jpn. J. Appl. Phys. 2020, 59, SPPD07. [Google Scholar] [CrossRef]
- Ederer, C.; Spaldin, N.A. Weak Ferromagnetism and Magnetoelectric Coupling in Bismuth Ferrite. Phys. Rev. B 2005, 71, 060401. [Google Scholar] [CrossRef]
- Yang, S.C.; Kumar, A.; Petkov, V.; Priya, S. Room-Temperature Magnetoelectric Coupling in Single-Phase BaTiO3-BiFeO3 System. J. Appl. Phys. 2013, 113, 144101. [Google Scholar] [CrossRef]
- Ramesh, R.; Spaldin, N.A. Multiferroics: Progress and Prospects in Thin Films. Nat. Mater. 2007, 6, 21–29. [Google Scholar] [CrossRef]
- Fiebig, M. Revival of the Magnetoelectric Effect. J. Phys. D. Appl. Phys. 2005, 38, R123. [Google Scholar] [CrossRef]
- Noguchi, Y.; Matsuo, H.; Kitanaka, Y.; Miyayama, M. Ferroelectrics with a Controlled Oxygen-Vacancy Distribution by Design. Sci. Rep. 2019, 9, 4225. [Google Scholar] [CrossRef]
- Kim, S.; Khanal, G.P.; Nam, H.W.; Fujii, I.; Ueno, S.; Moriyoshi, C.; Kuroiwa, Y.; Wada, S. Structural and Electrical Characteristics of Potential Candidate Lead-Free BiFeO3-BaTiO3 Piezoelectric Ceramics. J. Appl. Phys. 2017, 122, 164105. [Google Scholar] [CrossRef]
- Noguchi, Y.; Matsuo, H. Polarization and Dielectric Properties of BiFeO3-BaTiO3 Superlattice-Structured Ferroelectric Films. Nanomaterials 2021, 11, 1857. [Google Scholar] [CrossRef] [PubMed]
- Matsuo, H.; Noguchi, Y.; Miyayama, M.; Kiguchi, T.; Konno, T.J. Enhanced Photovoltaic Effects in Ferroelectric Solid Solution Thin Films with Nanodomains. Appl. Phys. Lett. 2020, 116, 132901. [Google Scholar] [CrossRef]
- Wang, J.; Neaton, J.B.; Zheng, H.; Nagarajan, V.; Ogale, S.B.; Liu, B.; Viehland, D.; Vaithyanathan, V.; Schlom, D.G.; Waghmare, U.V.; et al. Epitaxial BiFeO3 Multiferroic Thin Film Heterostructures. Science 2003, 299, 1719–1722. [Google Scholar] [CrossRef]
- Michel, C.; Moreau, J.M.; Achenbach, G.D.; Gerson, R.; James, W.J. The Atomic Structure of BiFeO3. Solid State Commun. 1969, 7, 701–704. [Google Scholar] [CrossRef]
- Kubel, F.; Schmid, H. Structure of a Ferroelectric and Ferroelastic Monodomain Crystal of the Perovskite BiFeO3. Acta Crystallogr. Sect. B Struct. Sci. 1990, 46, 698–702. [Google Scholar] [CrossRef]
- Zhang, J.X.; He, Q.; Trassin, M.; Luo, W.; Yi, D.; Rossell, M.D.; Yu, P.; You, L.; Wang, C.H.; Kuo, C.Y.; et al. Microscopic Origin of the Giant Ferroelectric Polarization in Tetragonal-like BiFeO3. Phys. Rev. Lett. 2011, 107, 147602. [Google Scholar] [CrossRef]
- Arnold, D. Composition-Driven Structural Phase Transitions in Rare-Earth-Doped Bifeo3 Ceramics: A Review. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 2015, 62, 62–82. [Google Scholar] [CrossRef]
- Karimi, S.; Reaney, I.M.; Han, Y.; Pokorny, J.; Sterianou, I. Crystal Chemistry and Domain Structure of Rare-Earth Doped BiFeO3 Ceramics. J. Mater. Sci. 2009, 44, 5102–5112. [Google Scholar] [CrossRef]
- Karpinsky, D.V.; Troyanchuk, I.O.; Tovar, M.; Sikolenko, V.; Efimov, V.; Kholkin, A.L. Evolution of Crystal Structure and Ferroic Properties of La-Doped BiFeO3 Ceramics near the Rhombohedral-Orthorhombic Phase Boundary. J. Alloys Compd. 2013, 555, 101–107. [Google Scholar] [CrossRef]
- Lee, J.-H.; Choi, H.J.; Lee, D.; Kim, M.G.; Bark, C.W.; Ryu, S.; Oak, M.-A.; Jang, H.M. Variations of ferroelectric off-centering distortion and 3 d− 4 p orbital mixing in La-doped BiFeO3 multiferroics. Phys. Rev. B 2010, 82, 045113. [Google Scholar] [CrossRef]
- González-Vázquez, O.E.; Wojdeł, J.C.; Diéguez, O.; Íñiguez, J. First-principles investigation of the structural phases and enhanced response properties of the BiFeO3-LaFeO3 multiferroic solid solution. Phys. Rev. B 2012, 85, 064119. [Google Scholar] [CrossRef]
- Mazumdar, D.; Knut, R.; Thöle, F.; Gorgoi, M.; Faleev, S.; Mryasov, O.N.; Shelke, V.; Ederer, C.; Spaldin, N.A.; Gupta, A.; et al. The Valence Band Electronic Structure of Rhombohedral-like and Tetragonal-like BiFeO3 Thin Films from Hard X-Ray Photoelectron Spectroscopy and First-Principles Theory. J. Electron Spectros. Relat. Phenom. 2016, 208, 63–66. [Google Scholar] [CrossRef]
- Chang, W.S.; Tu, C.-S.; Chen, P.-Y.; Chen, C.-S.; Lin, C.-Y.; Feng, K.-C.; Hsieh, Y.L.; Huang, Y.H. Effects of Fe3d–O2p and Bi 6sp–O2p Orbital Hybridizations in Nd Doped BiFeO3 Ceramics. J. Alloys Compd. 2017, 710, 670–679. [Google Scholar] [CrossRef]
- Tan, Q.; Wang, Q.; Liu, Y. Magnetic Properties and Spontaneous Polarization of La-, Mn- and N-Doped Tetragonal BiFeO3: A First-Principles Study. Materials 2018, 11, 985. [Google Scholar] [CrossRef]
- Mumtaz, F.; Nasir, S.; Jaffari, G.H.; Shah, S.I. Chemical Pressure Exerted by Rare Earth Substitution in BiFeO3: Effect on Crystal Symmetry, Band Structure and Magnetism. J. Alloys Compd. 2021, 876, 160178. [Google Scholar] [CrossRef]
- Kiyanagi, R.; Yamazaki, T.; Sakamoto, Y.; Kimura, H.; Noda, Y.; Ohyama, K.; Torii, S.; Yonemura, M.; Zhang, J.; Kamiyama, T. Structural and Magnetic Phase Determination of (1-x)BiFeO3–xBaTiO3 Solid Solution. J. Phys. Soc. Jpn. 2012, 81, 024603. [Google Scholar] [CrossRef]
- Leontsev, S.O.; Eitel, R.E. Dielectric and Piezoelectric Properties in Mn-Modified (1−x)BiFeO3-xBaTiO3 Ceramics. J. Am. Ceram. Soc. 2009, 92, 2957–2961. [Google Scholar] [CrossRef]
- Rusakov, D.A.; Abakumov, A.M.; Yamaura, K.; Belik, A.A.; Van Tendeloo, G.; Takayama-Muromachi, E. Structural Evolution of the BiFeO3−LaFeO3 System. Chem. Mater. 2011, 23, 285–292. [Google Scholar] [CrossRef]
- Troyanchuk, I.O.; Bushinsky, M.V.; Karpinsky, D.V.; Mantytskaya, O.S.; Fedotova, V.V.; Prochnenko, O.I. Structural Transformations and Magnetic Properties of Bi1-xLnxFeO3 (Ln = La, Nd, Eu) Multiferroics. Phys. Status Solidi 2009, 246, 1901–1907. [Google Scholar] [CrossRef]
- You, L.; Zheng, F.; Fang, L.; Zhou, Y.; Tan, L.Z.; Zhang, Z.; Ma, G.; Schmidt, D.; Rusydi, A.; Wang, L.; et al. Enhancing Ferroelectric Photovoltaic Effect by Polar Order Engineering. Sci. Adv. 2018, 4, eaat3438. [Google Scholar] [CrossRef]
- Langreth, D.C.; Perdew, J.P. Theory of Nonuniform Electronic Systems. I. Analysis of the Gradient Approximation and a Generalization That Works. Phys. Rev. B 1980, 21, 5469–5493. [Google Scholar] [CrossRef]
- Blöchl, P.E. Projector Augmented-Wave Method. Phys. Rev. B 1994, 50, 17953–17979. [Google Scholar] [CrossRef]
- Kresse, G.; Hafner, J. Ab Initio Molecular-Dynamics Simulation of the Liquid-Metal–Amorphous-Semiconductor Transition in Germanium. Phys. Rev. B 1994, 49, 14251–14269. [Google Scholar] [CrossRef] [PubMed]
- Perdew, J.P.; Ruzsinszky, A.; Csonka, G.I.; Vydrov, O.A.; Scuseria, G.E.; Constantin, L.A.; Zhou, X.; Burke, K. Restoring the Density-Gradient Expansion for Exchange in Solids and Surfaces. Phys. Rev. Lett. 2008, 100, 136406. [Google Scholar] [CrossRef] [PubMed]
- Anisimov, V.I.; Zaanen, J.; Andersen, O.K. Band Theory and Mott Insulators: Hubbard U Instead of Stoner I. Phys. Rev. B 1991, 44, 943–954. [Google Scholar] [CrossRef]
- Neaton, J.B.; Ederer, C.; Waghmare, U.V.; Spaldin, N.A.; Rabe, K.M. First-Principles Study of Spontaneous Polarization in Multiferroic BiFeO3. Phys. Rev. B 2005, 71, 014113. [Google Scholar] [CrossRef]
- Singh, S.K.; Ishiwara, H. Doping Effect of Rare-Earth Ions on Electrical Properties of BiFeO3 Thin Films Fabricated by Chemical Solution Deposition. Jpn. J. Appl. Phys. 2006, 45, 3194–3197. [Google Scholar] [CrossRef]
- Jangid, S.; Barbar, S.K.; Bala, I.; Roy, M. Structural, Thermal, Electrical and Magnetic Properties of Pure and 50% La Doped BiFeO3 Ceramics. Phys. B Condens. Matter 2012, 407, 3694–3699. [Google Scholar] [CrossRef]
- Gonze, X.; Lee, C. Dynamical Matrices, Born Effective Charges, Dielectric Permittivity Tensors, and Interatomic Force Constants from Density-Functional Perturbation Theory. Phys. Rev. B 1997, 55, 10355–10368. [Google Scholar] [CrossRef]
- Antonov, V.; Georgieva, I.; Trendafilova, N.; Kovacheva, D.; Krezhov, K. First Principles Study of Structure and Properties of La- and Mn-Modified BiFeO3. Solid State Sci. 2012, 14, 782–788. [Google Scholar] [CrossRef]
- Lee, J.-H.; Oak, M.-A.; Choi, H.J.; Son, J.Y.; Jang, H.M. Rhombohedral–Orthorhombic Morphotropic Phase Boundary in BiFeO3-Based Multiferroics: First-Principles Prediction. J. Mater. Chem. 2012, 22, 1667–1672. [Google Scholar] [CrossRef]
- Matsuo, H.; Kitanaka, Y.; Inoue, R.; Noguchi, Y.; Miyayama, M. Heavy Mn-Doping Effect on Spontaneous Polarization in Ferroelectric BiFeO3 Thin Films. Jpn. J. Appl. Phys. 2015, 54, 10NA03. [Google Scholar] [CrossRef]
- Yoneda, Y.; Kitanaka, Y.; Noguchi, Y.; Miyayama, M. Electronic and Local Structures of Mn-Doped BiFeO3 Crystals. Phys. Rev. B 2012, 86, 184112. [Google Scholar] [CrossRef]
- Matsuo, H.; Noguchi, Y.; Miyayama, M. Gap-State Engineering of Visible-Light-Active Ferroelectrics for Photovoltaic Applications. Nat. Commun. 2017, 8, 207. [Google Scholar] [CrossRef]
- Noguchi, Y.; Matsuo, H. Ferroelectric Polarization of Tetragonal BiFeO3—An Approach from DFT Calculations for BiFeO3–BaTiO3 Superlattices. Jpn. J. Appl. Phys. 2022, 61, SN1002. [Google Scholar] [CrossRef]
- Ravindran, P.; Vidya, R.; Kjekshus, A.; Fjellvåg, H.; Eriksson, O. Theoretical Investigation of Magnetoelectric Behavior in BiFe O3. Phys. Rev. B Condens. Matter Mater. Phys. 2006, 74, 224412. [Google Scholar] [CrossRef]
- Cohen, R.E. Origin of Ferroelectricity in Perovskite Oxides. Nature 1992, 358, 136–138. [Google Scholar] [CrossRef]
- Glazer, A.M. The Classification of Tilted Octahedra in Perovskites. Acta Crystallogr. Sect. B Struct. Crystallogr. Cryst. Chem. 1972, 28, 3384–3392. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Noguchi, Y.; Matsuo, H. Origin of Ferroelectricity in BiFeO3-Based Solid Solutions. Nanomaterials 2022, 12, 4163. https://doi.org/10.3390/nano12234163
Noguchi Y, Matsuo H. Origin of Ferroelectricity in BiFeO3-Based Solid Solutions. Nanomaterials. 2022; 12(23):4163. https://doi.org/10.3390/nano12234163
Chicago/Turabian StyleNoguchi, Yuji, and Hiroki Matsuo. 2022. "Origin of Ferroelectricity in BiFeO3-Based Solid Solutions" Nanomaterials 12, no. 23: 4163. https://doi.org/10.3390/nano12234163
APA StyleNoguchi, Y., & Matsuo, H. (2022). Origin of Ferroelectricity in BiFeO3-Based Solid Solutions. Nanomaterials, 12(23), 4163. https://doi.org/10.3390/nano12234163





