Carbonaceous Nanoparticle Air Pollution: Toxicity and Detection in Biological Samples
Abstract
1. Introduction
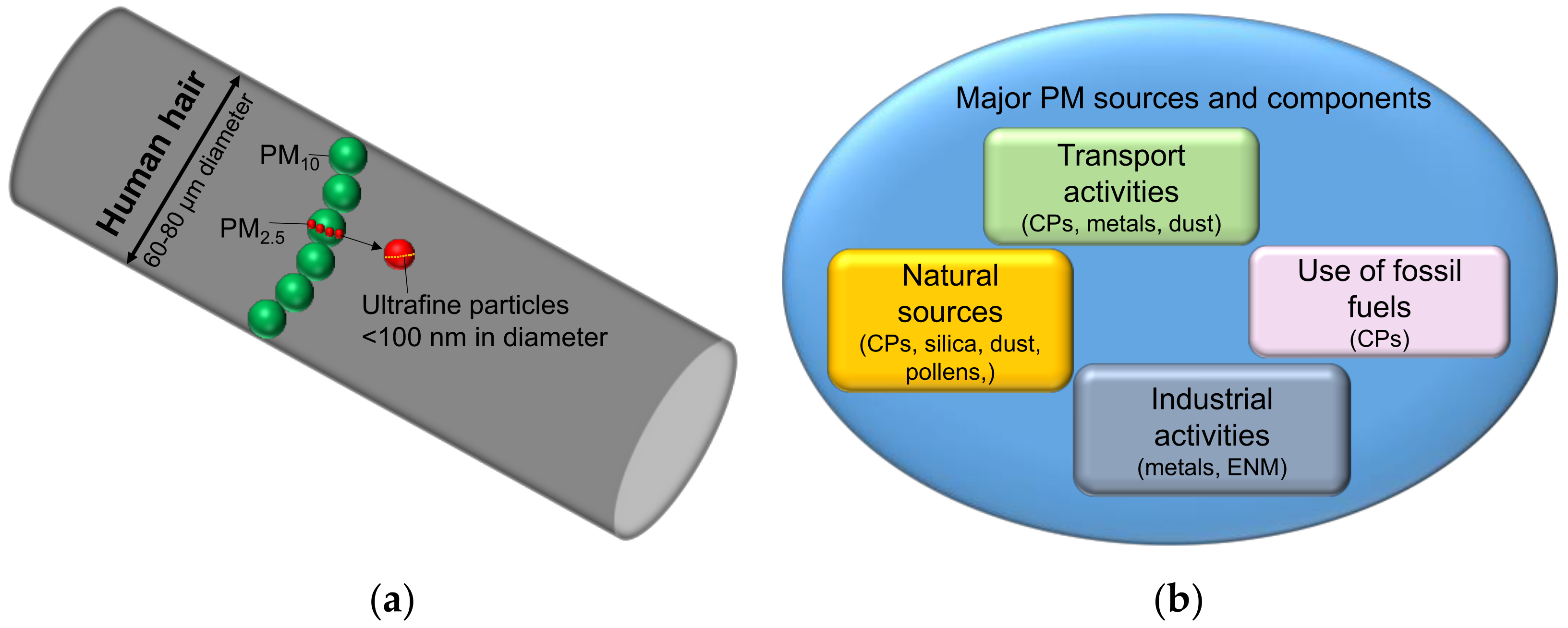
2. Carbonaceous Particle (CP) Air Pollution
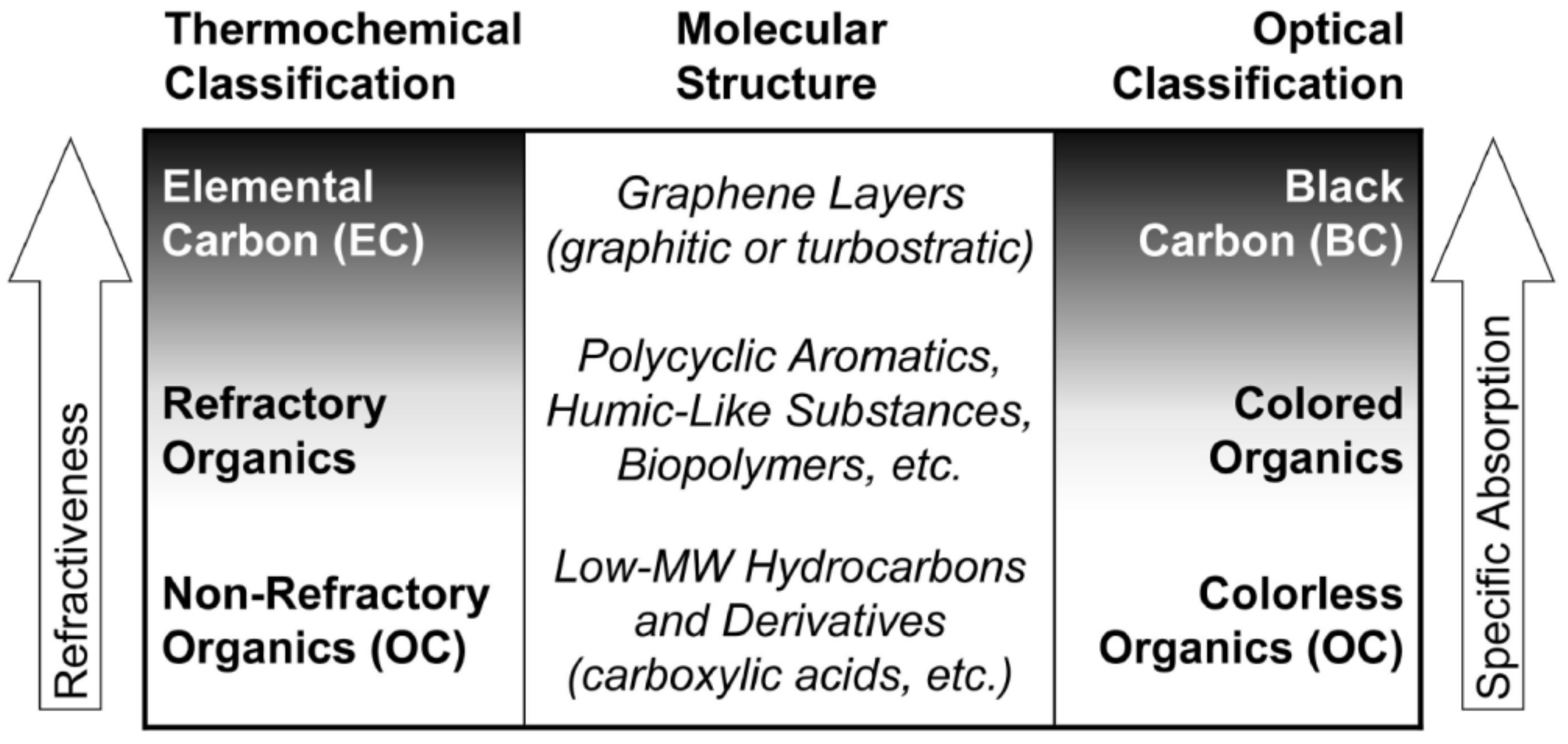
2.1. Classification of Different Components of CPs
2.1.1. Carbon Black (CB)
2.1.2. Black Carbon (BC)
2.1.3. Brown Carbon (BrC)
3. Environmental Impact of CPs
4. Health Effects of CPs
4.1. Pulmonary/Respiratory Effects
4.2. Cardiovascular Effects
4.3. Reproductive and Developmental Toxicity
4.4. Neurotoxic Effects
4.5. Genotoxic and Carcinogenic Effects
4.6. Dermal Toxicity
5. Characterization Techniques for Carbonaceous Particles (CPs)
5.1. Characterization Techniques for Atmospheric CPs
5.2. Characterization Techniques for CPs in Biological Samples
5.2.1. Isotope Tracing/Radiolabeling Detection of CPs
5.2.2. Mass Spectrometry Detection of CPs
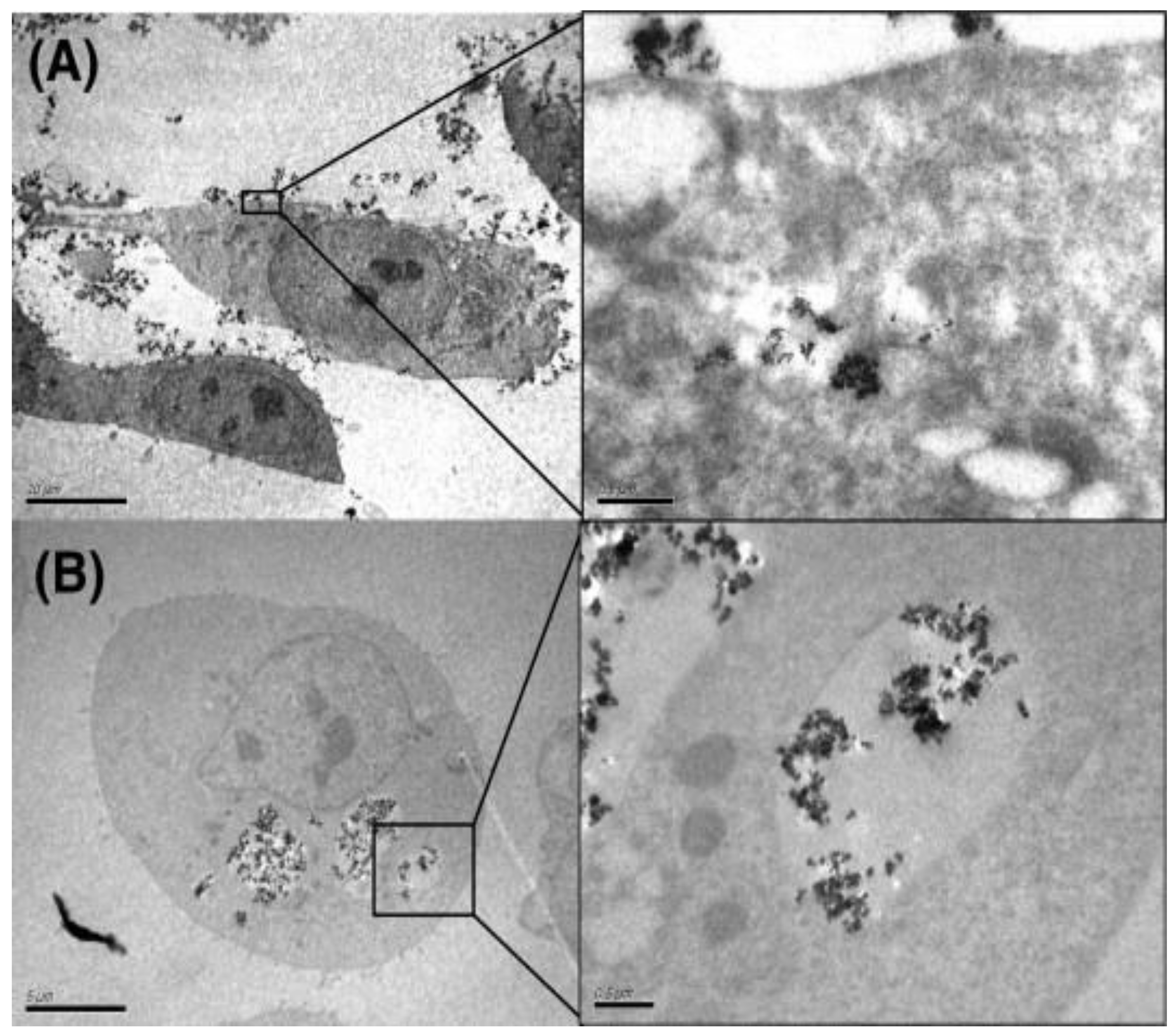
5.2.3. Electron Microscopy Detection of CPs
5.2.4. Optical Techniques for the Detection of CPs
- Absorption- and scattering-based microscopy
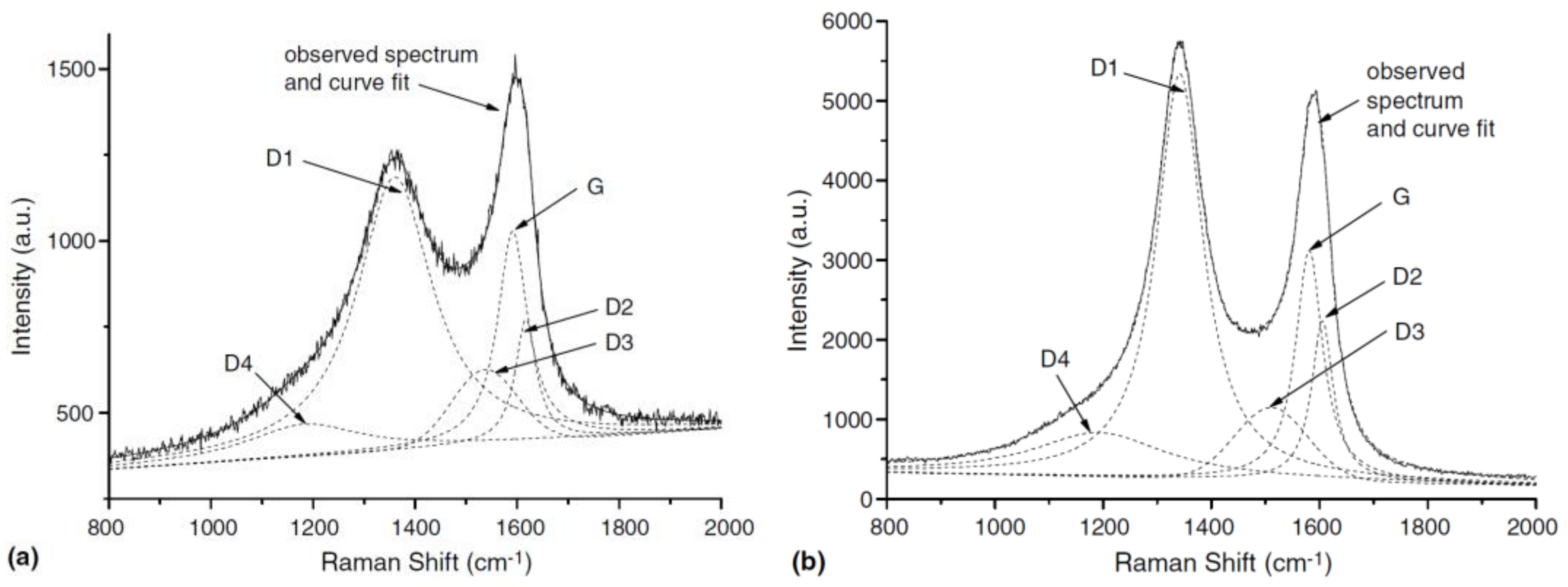
- Raman spectroscopy
- Photothermal pump–probe microscopy
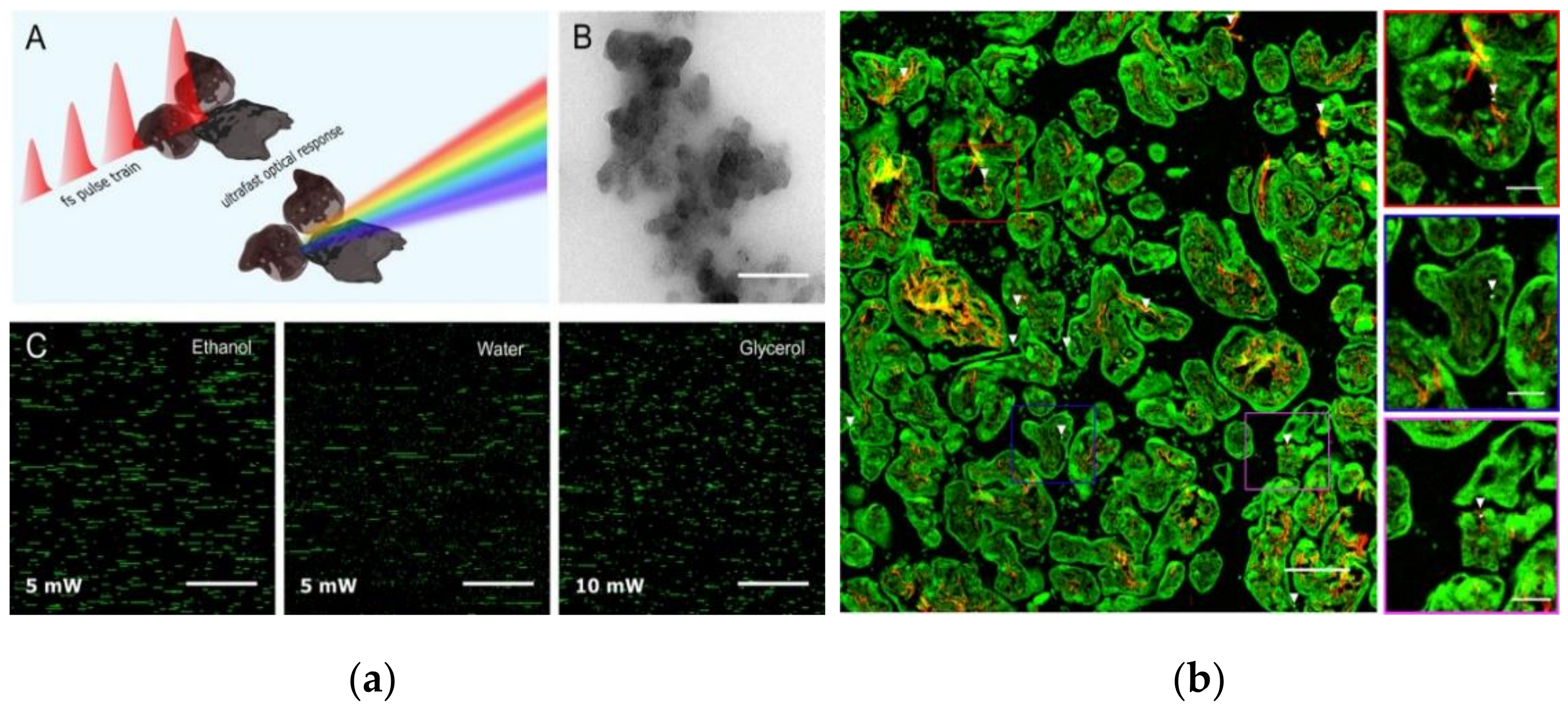
- Femtosecond-pulsed laser microscopy (non-incandescence-related white light generation) for CP detection
6. Conclusions and Outlook
6.1. Conclusions
6.2. Challenges and Outlook
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- OCDE. The Economic Consequences of Outdoor Air Pollution; Policy Highlights; OCDE: Paris, France, 2016; 116p. [Google Scholar] [CrossRef]
- Sicard, P.; Agathokleous, E.; De Marco, A.; Paoletti, E.; Calatayud, V. Urban population exposure to air pollution in Europe over the last decades. Environ. Sci. Eur. 2021, 33, 28. [Google Scholar] [CrossRef] [PubMed]
- Gakidou, E.; Afshin, A.; Abajobir, A.A.; Abate, K.H.; Abbafati, C.; Abbas, K.M.; Abd-Allah, F.; Abdulle, A.M.; Abera, S.F.; Aboyans, V.; et al. Global, regional, and national comparative risk assessment of 84 behavioural, environmental and occupational, and metabolic risks or clusters of risks, 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet 2017, 390, 1345–1422. [Google Scholar] [CrossRef]
- Neira, M.; Prüss-Ustün, A. Preventing disease through healthy environments: A global assessment of the environmental burden of disease. Toxicol. Lett. 2016, 259, S1. [Google Scholar] [CrossRef]
- Hamanaka, R.B.; Mutlu, G.M. Particulate Matter Air Pollution: Effects on the Cardiovascular System. Front. Endocrinol. 2018, 9, 680. [Google Scholar] [CrossRef] [PubMed]
- Newby, D.E.; Mannucci, P.M.; Tell, G.S.; Baccarelli, A.A.; Brook, R.D.; Donaldson, K.; Forastiere, F.; Franchini, M.; Franco, O.H.; Graham, I.; et al. Expert position paper on air pollution and cardiovascular disease. Eur. Heart J. 2015, 36, 83–93. [Google Scholar] [CrossRef] [PubMed]
- Brook, R.D.; Newby, D.E.; Rajagopalan, S. The Global Threat of Outdoor Ambient Air Pollution to Cardiovascular Health. JAMA Cardiol. 2017, 2, 353. [Google Scholar] [CrossRef]
- Schwarze, P.E.; Øvrevik, J.; Låg, M.; Refsnes, M.; Nafstad, P.; Hetland, R.B.; Dybing, E. Particulate matter properties and health effects: Consistency of epidemiological and toxicological studies. Hum. Exp. Toxicol. 2006, 25, 559–579. [Google Scholar] [CrossRef]
- Adams, K.; Greenbaum, D.S.; Shaikh, R.; van Erp, A.M.; Russell, A.G. Particulate matter components, sources, and health: Systematic approaches to testing effects. J. Air Waste Manag. Assoc. 2015, 65, 544–558. [Google Scholar] [CrossRef]
- Okubo, M.; Kuwahara, T. Emission regulations. In New Technologies for Emission Control in Marine Diesel Engines; Elsevier: Amsterdam, The Netherlands, 2020. [Google Scholar]
- Thorpe, A.; Harrison, R.M. Sources and properties of non-exhaust particulate matter from road traffic: A review. Sci. Total Environ. 2008, 400, 270–282. [Google Scholar] [CrossRef]
- Kelly, F.J.; Fussell, J.C. Size, source and chemical composition as determinants of toxicity attributable to ambient particulate matter. Atmos. Environ. 2012, 60, 504–526. [Google Scholar] [CrossRef]
- Louie, S.M.; Ma, R.; Lowry, G.V. Transformations of Nanomaterials in the Environment. In Frontiers of Nanoscience; Elsevier: Amsterdam, The Netherlands, 2014; ISBN 0013-936X. [Google Scholar]
- Keller, A.A.; McFerran, S.; Lazareva, A.; Suh, S. Global life cycle releases of engineered nanomaterials. J. Nanopart. Res. 2013, 15, 1692. [Google Scholar] [CrossRef]
- Kabir, E.; Ray, S.; Kim, K.-H.; Yoon, H.-O.; Jeon, E.-C.; Kim, Y.S.; Cho, Y.-S.; Yun, S.-T.; Brown, R.J.C. Current Status of Trace Metal Pollution in Soils Affected by Industrial Activities. Sci. World J. 2012, 2012, 1–18. [Google Scholar] [CrossRef]
- Hime, N.J.; Marks, G.B.; Cowie, C.T. A comparison of the health effects of ambient particulate matter air pollution from five emission sources. Int. J. Environ. Res. Public Health 2018, 15, 1206. [Google Scholar] [CrossRef]
- Reşitoʇlu, I.A.; Altinişik, K.; Keskin, A. The pollutant emissions from diesel-engine vehicles and exhaust aftertreatment systems. Clean Technol. Environ. Policy 2015, 17, 15–27. [Google Scholar] [CrossRef]
- Power, M.C.; Weisskopf, M.G.; Alexeeff, S.E.; Coull, B.A.; Spiro, A.; Schwartz, J. Traffic-Related Air Pollution and Cognitive Function in a Cohort of Older Men. Environ. Health Perspect. 2011, 119, 682–687. [Google Scholar] [CrossRef]
- Dumka, U.C.; Kaskaoutis, D.G.; Devara, P.C.S.; Kumar, R.; Kumar, S.; Tiwari, S.; Gerasopoulos, E.; Mihalopoulos, N. Year-long variability of the fossil fuel and wood burning black carbon components at a rural site in southern Delhi outskirts. Atmos. Res. 2019, 216, 11–25. [Google Scholar] [CrossRef]
- Fussell, J.C.; Kelly, F.J. Mechanisms underlying the health effects of desert sand dust. Environ. Int. 2021, 157, 106790. [Google Scholar] [CrossRef]
- Health Effects of Particulate Matter; World Health Organization: Copenhagen, Denmark, 2013; Available online: https://www.euro.who.int/__data/assets/pdf_file/0006/189051/Health-effects-of-particulate-matter-final-Eng.pdf (accessed on 20 October 2022).
- Manisalidis, I.; Stavropoulou, E.; Stavropoulos, A.; Bezirtzoglou, E. Environmental and Health Impacts of Air Pollution: A Review. Front. Public Health 2020, 8, 14. [Google Scholar] [CrossRef]
- Health Relevance of Particulate Matter from Various Sources; Report on a WHO Workshop. World Health Organization: Copenhagen, Denmark, 2007. Available online: https://www.euro.who.int/__data/assets/pdf_file/0007/78658/E90672.pdf (accessed on 20 October 2022).
- Lee, Y.M.; Lee, J.H.; Kim, H.C.; Ha, E. Effects of PM10 on mortality in pure COPD and asthma-COPD overlap: Difference in exposure duration, gender, and smoking status. Sci. Rep. 2020, 10, 2402. [Google Scholar] [CrossRef]
- Lewis, T.C.; Robins, T.G.; Dvonch, J.T.; Keeler, G.J.; Yip, F.Y.; Mentz, G.B.; Lin, X.; Parker, E.A.; Israel, B.A.; Gonzalez, L.; et al. Air pollution-associated changes in lung function among asthmatic children in Detroit. Environ. Health Perspect. 2005, 113, 1068–1075. [Google Scholar] [CrossRef]
- Du, Y.; Xu, X.; Chu, M.; Guo, Y.; Wang, J. Air particulate matter and cardiovascular disease: The epidemiological, biomedical and clinical evidence. J. Thorac. Dis. 2016, 8, E8–E19. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Zhou, R.; Zhang, J. Function of PM2.5 in the pathogenesis of lung cancer and chronic airway inflammatory diseases. Oncol. Lett. 2018, 15, 7506–7514. [Google Scholar] [CrossRef]
- Harrison, R.M. What is responsible for the carcinogenicity of PM2.5? Occup. Environ. Med. 2004, 61, 799–805. [Google Scholar] [CrossRef] [PubMed]
- Schraufnagel, D.E. The health effects of ultrafine particles. Exp. Mol. Med. 2020, 52, 311–317. [Google Scholar] [CrossRef] [PubMed]
- Oberdörster, G.; Celein, R.M.; Ferin, J.; Weiss, B. Association of particulate air pollution and acute mortality: Involvement of ultrafine particles? Inhal. Toxicol. 1995, 7, 111–124. [Google Scholar] [CrossRef]
- Oberdörster, G.; Sharp, Z.; Atudorei, V.; Elder, A.; Gelein, R.; Kreyling, W.; Cox, C. Translocation of inhaled ultrafine particles to the brain. Inhal. Toxicol. 2004, 16, 437–445. [Google Scholar] [CrossRef]
- Hong, G.; Jee, Y.K. Special issue on ultrafine particles: Where are they from and how do they affect us? Exp. Mol. Med. 2020, 52, 309–310. [Google Scholar] [CrossRef]
- Bai, L.; Chen, H.; Hatzopoulou, M.; Jerrett, M.; Kwong, J.C.; Burnett, R.T.; Van Donkelaar, A.; Copes, R.; Martin, R.V.; Van Ryswyk, K.; et al. Exposure to ambient ultrafine particles and nitrogen dioxide and incident hypertension and diabetes. Epidemiology 2018, 29, 323–332. [Google Scholar] [CrossRef]
- Schlesinger, R.B.; Kunzli, N.; Hidy, G.M.; Gotschi, T.; Jerrett, M. The health relevance of ambient particulate matter characteristics: Coherence of toxicological and epidemiological inferences. Inhal. Toxicol. 2006, 18, 95–125. [Google Scholar] [CrossRef]
- Stanek, L.W.; Sacks, J.D.; Dutton, S.J.; Dubois, J.J.B. Attributing health effects to apportioned components and sources of particulate matter: An evaluation of collective results. Atmos. Environ. 2011, 45, 5655–5663. [Google Scholar] [CrossRef]
- Krzyzanowski, M.; Dibbert, B.K.; Schneider, J. Health Effects of Transport Related Air Pollution; World Health Organization: Copenhagen, Denmark, 2005; Available online: https://www.euro.who.int/__data/assets/pdf_file/0006/74715/E86650.pdf (accessed on 20 October 2022).
- Donaldson, K.; Tran, L.; Jimenez, L.A.; Duffin, R.; Newby, D.E.; Mills, N.; MacNee, W.; Stone, V. Combustion-derived nanoparticles: A review of their toxicology following inhalation exposure. Part. Fibre Toxicol. 2005, 2, 10. [Google Scholar] [CrossRef]
- Janssen, N.A.H.; Hoek, G.; Simic-Lawson, M.; Fischer, P.; van Bree, L.; Ten Brink, H.; Keuken, M.; Atkinson, R.W.; Ross Anderson, H.; Brunekreef, B.; et al. Black carbon as an additional indicator of the adverse health effects of airborne particles compared with pm10 and pm2.5. Environ. Health Perspect. 2011, 119, 1691–1699. [Google Scholar] [CrossRef]
- Andreae, M.O.; Gelencsér, A. Black carbon or brown carbon? the nature of light-absorbing carbonaceous aerosols. Atmos. Chem. Phys. 2006, 6, 3131–3148. [Google Scholar] [CrossRef]
- Pöschl, U. Aerosol particle analysis: Challenges and progress. Anal. Bioanal. Chem. 2003, 375, 30–32. [Google Scholar] [CrossRef]
- Lyu, R.; Zhang, J.; Wu, J.; Feng, Y. Primary Carbonaceous Particle Emission from Four Power Plants with Ultralow Emission in China. ACS Omega 2021, 6, 1309–1315. [Google Scholar] [CrossRef]
- Long, C.M.; Nascarella, M.A.; Valberg, P.A. Carbon black vs. black carbon and other airborne materials containing elemental carbon: Physical and chemical distinctions. Environ. Pollut. 2013, 181, 271–286. [Google Scholar] [CrossRef]
- Lin, P.; Bluvshtein, N.; Rudich, Y.; Nizkorodov, S.A.; Laskin, J.; Laskin, A. Molecular Chemistry of Atmospheric Brown Carbon Inferred from a Nationwide Biomass Burning Event. Environ. Sci. Technol. 2017, 51, 11561–11570. [Google Scholar] [CrossRef]
- Medalia, A.I.; Rivin, D.; Sanders, D.R. A comparison of carbon black with soot. Sci. Total Environ. 1983, 31, 1–22. [Google Scholar] [CrossRef]
- Schnaiter, M.; Horvath, H.; Möhler, O.; Naumann, K.-H.; Saathoff, H.; Schöck, O.W. UV-VIS-NIR spectral optical properties of soot and soot-containing aerosols. J. Aerosol Sci. 2003, 34, 1421–1444. [Google Scholar] [CrossRef]
- Romshoo, B.; Müller, T.; Pfeifer, S.; Saturno, J.; Nowak, A.; Ciupek, K.; Quincey, P.; Wiedensohler, A. Optical properties of coated black carbon aggregates: Numerical simulations, radiative forcing estimates, and size-resolved parameterization scheme. Atmos. Chem. Phys. 2021, 21, 12989–13010. [Google Scholar] [CrossRef]
- Laskin, A.; Laskin, J.; Nizkorodov, S.A. Chemistry of Atmospheric Brown Carbon. Chem. Rev. 2015, 115, 4335–4382. [Google Scholar] [CrossRef] [PubMed]
- Russo, C.; Stanzione, F.; Alfè, M.; Ciajolo, A.; Tregrossi, A. Spectral Analysis in the UV-Visible Range for Revealing the Molecular Form of Combustion-Generated Carbonaceous Species. Combust. Sci. Technol. 2012, 184, 1219–1231. [Google Scholar] [CrossRef]
- Robertson, J. Hard amorphous (diamond-like) carbons. Prog. Solid State Chem. 1991, 21, 199–333. [Google Scholar] [CrossRef]
- Robertson, J.; O’Reilly, E.P. Electronic and atomic structure of amorphous carbon. Phys. Rev. B 1987, 35, 2946–2957. [Google Scholar] [CrossRef] [PubMed]
- Llamas-Jansa, I.; Jäger, C.; Mutschke, H.; Henning, T. Far-ultraviolet to near-infrared optical properties of carbon nanoparticles produced by pulsed-laser pyrolysis of hydrocarbons and their relation with structural variations. Carbon N. Y. 2007, 45, 1542–1557. [Google Scholar] [CrossRef]
- Duley, W.W.; Seahra, S. Graphite, Polycyclic Aromatic Hydrocarbons, and the 2175 Å Extinction Feature. Astrophys. J. 1998, 507, 874–888. [Google Scholar] [CrossRef]
- Henning, T.; Jäger, C.; Mutschke, H. Laboratory Studies of Carbonaceous Dust Analogs. Astrophys. Dust ASP Conf. Ser. 2004, 309, 603–628. [Google Scholar]
- Galvez, A.; Herlin-Boime, N.; Reynaud, C.; Clinard, C.; Rouzaud, J.-N. Carbon nanoparticles from laser pyrolysis. Carbon N. Y. 2002, 40, 2775–2789. [Google Scholar] [CrossRef]
- Wang, X.; Heald, C.L.; Sedlacek, A.J.; de Sá, S.S.; Martin, S.T.; Alexander, M.L.; Watson, T.B.; Aiken, A.C.; Springston, S.R.; Artaxo, P. Deriving brown carbon from multiwavelength absorption measurements: Method and application to AERONET and Aethalometer observations. Atmos. Chem. Phys. 2016, 16, 12733–12752. [Google Scholar] [CrossRef]
- Hegglin, M.I.; Krieger, U.K.; Koop, T.; Peter, T. Technical Note: Organics-Induced Fluorescence in Raman Studies of Sulfuric Acid Aerosols. Aerosol Sci. Technol. 2002, 36, 510–512. [Google Scholar] [CrossRef]
- Li, X.; Hu, M.; Wang, Y.; Xu, N.; Fan, H.; Zong, T.; Wu, Z.; Guo, S.; Zhu, W.; Chen, S.; et al. Links between the optical properties and chemical compositions of brown carbon chromophores in different environments: Contributions and formation of functionalized aromatic compounds. Sci. Total Environ. 2021, 786, 147418. [Google Scholar] [CrossRef]
- Lin, P.; Aiona, P.K.; Li, Y.; Shiraiwa, M.; Laskin, J.; Nizkorodov, S.A.; Laskin, A. Molecular Characterization of Brown Carbon in Biomass Burning Aerosol Particles. Environ. Sci. Technol. 2016, 50, 11815–11824. [Google Scholar] [CrossRef]
- Soleimanian, E.; Mousavi, A.; Taghvaee, S.; Shafer, M.M.; Sioutas, C. Impact of secondary and primary particulate matter (PM) sources on the enhanced light absorption by brown carbon (BrC) particles in central Los Angeles. Sci. Total Environ. 2020, 705, 135902. [Google Scholar] [CrossRef]
- Cachier, H.; Brémond, M.-P.; Buat-Ménard, P. Carbonaceous aerosols from different tropical biomass burning sources. Nature 1989, 340, 371–373. [Google Scholar] [CrossRef]
- Graber, E.R.; Rudich, Y. Atmospheric HULIS: How humic-like are they? A comprehensive and critical review. Atmos. Chem. Phys. 2006, 6, 729–753. [Google Scholar] [CrossRef]
- Bernauer, U.; Chaudhry, Q.; Coenraads, P.-J.; Degen, G.; Dusinska, M.; Lilienblum, W.; Nielsen, E.; Platzek, T.; Rousselle, C.; van Benthem, J. Opinion on Carbon Black (Nano-Form); Scientific Committee on Consumer Safety: Brussels, Belgium, 2013; ISBN 9789279301209. [Google Scholar]
- Singh, M.; Vander Wal, R. Nanostructure Quantification of Carbon Blacks. C 2018, 5, 2. [Google Scholar] [CrossRef]
- Association, I.C.B. Carbon Black User’s Guide, Safety, Health, & Environmental Information. Int. Carbon Black Assoc. 2016. Available online: https://cancarb.com/wp-content/uploads/2020/05/carbon-black-user-guide.pdf (accessed on 20 October 2022).
- WHO. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans; World Health Organization: Geneva, Switzerland, 1994. [Google Scholar] [CrossRef]
- Kirk-Othmer, Encyclopedia of Chemical Technology; John Wiley and Sons: Hoboken, NJ, USA, 1993.
- Scientific Opinion on the re-evaluation of vegetable carbon (E 153) as a food additive. EFSA J. 2012, 10, 2592. [CrossRef]
- Sids Initial Assessment Profile for Carbon Black, SIAM 22, 18–21 April. 2006. Available online: https://hpvchemicals.oecd.org/ui/handler.axd?id=b2394717-5545-46c3-9194-ae15f987df25 (accessed on 22 April 2022).
- Shrestha, G.; Traina, S.J.; Swanston, C.W. Black carbon’s properties and role in the environment: A comprehensive review. Sustainability 2010, 2, 294–320. [Google Scholar] [CrossRef]
- Dai, X.; Boutton, T.W.; Glaser, B.; Ansley, R.J.; Zech, W. Black carbon in a temperate mixed-grass savanna. Soil Biol. Biochem. 2005, 37, 1879–1881. [Google Scholar] [CrossRef]
- Epa, U. Report to Congress on Black Carbon: Executive Summary; United States Environmental Protection Agency: Washington, DC, USA, 2012. Available online: https://19january2017snapshot.epa.gov/www3/airquality/blackcarbon/2012report/fullreport.pdf (accessed on 20 October 2022).
- Dons, E.; Int Panis, L.; Van Poppel, M.; Theunis, J.; Wets, G. Personal exposure to Black Carbon in transport microenvironments. Atmos. Environ. 2012, 55, 392–398. [Google Scholar] [CrossRef]
- Byčenkiene, S.; Ulevicius, V.; Dudoitis, V.; Pauraite, J. Identification and characterization of black carbon aerosol sources in the East Baltic Region. Adv. Meteorol. 2013, 2013, 380614. [Google Scholar] [CrossRef]
- Ning, Z.; Chan, K.L.; Wong, K.C.; Westerdahl, D.; Močnik, G.; Zhou, J.H.; Cheung, C.S. Black carbon mass size distributions of diesel exhaust and urban aerosols measured using differential mobility analyzer in tandem with Aethalometer. Atmos. Environ. 2013, 80, 31–40. [Google Scholar] [CrossRef]
- Sonwani, S.; Yadav, A.; Saxena, P. Atmospheric Brown Carbon: A Global Emerging Concern for Climate and Environmental Health. Manag. Contam. Emerg. Concern Environ. 2021, 1, 225–247. [Google Scholar] [CrossRef]
- Yan, J.; Wang, X.; Gong, P.; Wang, C.; Cong, Z. Review of brown carbon aerosols: Recent progress and perspectives. Sci. Total Environ. 2018, 634, 1475–1485. [Google Scholar] [CrossRef] [PubMed]
- Bond, T.C. Spectral dependence of visible light absorption by carbonaceous particles emitted from coal combustion. Geophys. Res. Lett. 2001, 28, 4075–4078. [Google Scholar] [CrossRef]
- Lack, D.A.; Langridge, J.M.; Bahreini, R.; Cappa, C.D.; Middlebrook, A.M.; Schwarz, J.P. Brown carbon and internal mixing in biomass burning particles. Proc. Natl. Acad. Sci. USA 2012, 109, 14802–14807. [Google Scholar] [CrossRef]
- Alexander, D.T.L.; Crozier, P.A.; Anderson, J.R. Brown carbon spheres in East Asian outflow and their optical properties. Science 2008, 321, 833–836. [Google Scholar] [CrossRef]
- Lack, D.A.; Cappa, C.D. Impact of brown and clear carbon on light absorption enhancement, single scatter albedo and absorption wavelength dependence of black carbon. Atmos. Chem. Phys. 2010, 10, 4207–4220. [Google Scholar] [CrossRef]
- De Haan, D.O.; Corrigan, A.L.; Tolbert, M.A.; Jimenez, J.L.; Wood, S.E.; Turley, J.J. Secondary organic aerosol formation by self-reactions of methylglyoxal and glyoxal in evaporating droplets. Environ. Sci. Technol. 2009, 43, 8184–8190. [Google Scholar] [CrossRef]
- Powelson, M.H.; Espelien, B.M.; Hawkins, L.N.; Galloway, M.M.; De Haan, D.O. Brown carbon formation by aqueous-phase carbonyl compound reactions with amines and ammonium sulfate. Environ. Sci. Technol. 2014, 48, 985–993. [Google Scholar] [CrossRef]
- Bai, Z.; Zhang, L.; Cheng, Y.; Zhang, W.; Mao, J.; Chen, H.; Li, L.; Wang, L.; Chen, J. Water/Methanol-Insoluble Brown Carbon Can Dominate Aerosol-Enhanced Light Absorption in Port Cities. Environ. Sci. Technol. 2020, 54, 14889–14898. [Google Scholar] [CrossRef]
- Satish, R.; Rastogi, N.; Singh, A.; Singh, D. Change in characteristics of water-soluble and water-insoluble brown carbon aerosols during a large-scale biomass burning. Environ. Sci. Pollut. Res. 2020, 27, 33339–33350. [Google Scholar] [CrossRef]
- Jo, D.S.; Park, R.J.; Lee, S.; Kim, S.-W.; Zhang, X. A global simulation of brown carbon: Implications for photochemistry and direct radiative effect. Atmos. Chem. Phys. 2016, 16, 3413–3432. [Google Scholar] [CrossRef]
- Atwi, K.; Mondal, A.; Pant, J.; Cheng, Z.; El Hajj, O.; Ijeli, I.; Handa, H.; Saleh, R. Physicochemical properties and cytotoxicity of brown carbon produced under different combustion conditions. Atmos. Environ. 2021, 244, 117881. [Google Scholar] [CrossRef]
- Lima de Albuquerque, Y.; Berger, E.; Li, C.; Pardo, M.; George, C.; Rudich, Y.; Géloën, A. The Toxic Effect of Water-Soluble Particulate Pollutants from Biomass Burning on Alveolar Lung Cells. Atmosphere 2021, 12, 1023. [Google Scholar] [CrossRef]
- Marelle, L.; Thomas, J.L.; Ahmed, S.; Tuite, K.; Stutz, J.; Dommergue, A.; Simpson, W.R.; Frey, M.M.; Baladima, F. Implementation and Impacts of Surface and Blowing Snow Sources of Arctic Bromine Activation Within WRF-Chem 4.1.1. J. Adv. Model. Earth Syst. 2021, 13, e2020MS002391. [Google Scholar] [CrossRef]
- Jacobson, M.Z. Control of fossil-fuel particulate black carbon and organic matter, possibly the most effective method of slowing global warming. J. Geophys. Res. Atmos. 2002, 107, ACH 16-1–ACH 16-22. [Google Scholar] [CrossRef]
- Tollefson, J. Soot a major contributor to climate change. Nature 2013, 15, 15. [Google Scholar] [CrossRef]
- Zhang, Y.; Gao, T.; Kang, S.; Sprenger, M.; Tao, S.; Du, W.; Yang, J.; Wang, F.; Meng, W. Effects of black carbon and mineral dust on glacial melting on the Muz Taw glacier, Central Asia. Sci. Total Environ. 2020, 740, 140056. [Google Scholar] [CrossRef]
- Kang, S.; Zhang, Y.; Qian, Y.; Wang, H. A review of black carbon in snow and ice and its impact on the cryosphere. Earth-Sci. Rev. 2020, 210, 103346. [Google Scholar] [CrossRef]
- Ming, Y.; Ramaswamy, V.; Persad, G. Two opposing effects of absorbing aerosols on global-mean precipitation. Geophys. Res. Lett. 2010, 37, 1–4. [Google Scholar] [CrossRef]
- Bond, T.C.; Doherty, S.J.; Fahey, D.W.; Forster, P.M.; Berntsen, T.; Deangelo, B.J.; Flanner, M.G.; Ghan, S.; Kärcher, B.; Koch, D.; et al. Bounding the role of black carbon in the climate system: A scientific assessment. J. Geophys. Res. Atmos. 2013, 118, 5380–5552. [Google Scholar] [CrossRef]
- Brown, M.E.; Antle, J.M.; Backlund, P.; Carr, E.R.; Easterling, W.E.; Walsh, M.K.; Ammann, C.; Attavanich, W.; Barrett, C.B.; Bellemare, M.F.; et al. Climate Change, Global Food Security, and the U.S. Food System; International Food Policy Research Institute: Washington, DC, USA, 2015. [Google Scholar] [CrossRef]
- Gao, C.; He, J.; Cong, J.; Zhang, S.; Wang, G. Impact of forest fires generated black carbon deposition fluxes in Great Hinggan Mountains (China). L. Degrad. Dev. 2018, 29, 2073–2081. [Google Scholar] [CrossRef]
- Lu, Z.; Streets, D.G.; Winijkul, E.; Yan, F.; Chen, Y.; Bond, T.C.; Feng, Y.; Dubey, M.K.; Liu, S.; Pinto, J.P.; et al. Light absorption properties and radiative effects of primary organic aerosol emissions. Environ. Sci. Technol. 2015, 49, 4868–4877. [Google Scholar] [CrossRef] [PubMed]
- Saleh, R.; Hennigan, C.J.; McMeeking, G.R.; Chuang, W.K.; Robinson, E.S.; Coe, H.; Donahue, N.M.; Robinson, A.L. Absorptivity of brown carbon in fresh and photo-chemically aged biomass-burning emissions. Atmos. Chem. Phys. 2013, 13, 7683–7693. [Google Scholar] [CrossRef]
- Yue, S.; Zhu, J.; Chen, S.; Xie, Q.; Li, W.; Li, L.; Ren, H.; Su, S.; Li, P.; Ma, H.; et al. Brown carbon from biomass burning imposes strong circum-Arctic warming. One Earth 2022, 5, 293–304. [Google Scholar] [CrossRef]
- Myers-Smith, I.H.; Kerby, J.T.; Phoenix, G.K.; Bjerke, J.W.; Epstein, H.E.; Assmann, J.J.; John, C.; Andreu-Hayles, L.; Angers-Blondin, S.; Beck, P.S.A.; et al. Complexity revealed in the greening of the Arctic. Nat. Clim. Chang. 2020, 10, 106–117. [Google Scholar] [CrossRef]
- Riediker, M.; Zink, D.; Kreyling, W.; Oberdörster, G.; Elder, A.; Graham, U.; Lynch, I.; Duschl, A.; Ichihara, G.; Ichihara, S.; et al. Particle toxicology and health—where are we? Part. Fibre Toxicol. 2019, 16, 19. [Google Scholar] [CrossRef]
- Lelieveld, J.; Evans, J.S.; Fnais, M.; Giannadaki, D.; Pozzer, A. The contribution of outdoor air pollution sources to premature mortality on a global scale. Nature 2015, 525, 367–371. [Google Scholar] [CrossRef]
- Chen, C.; Xu, D.; He, M.Z.; Wang, Y.; Du, Z.; Du, Y.; Qian, Y.; Ji, D.; Li, T. Fine Particle Constituents and Mortality: A Time-Series Study in Beijing, China. Environ. Sci. Technol. 2018, 52, 11378–11386. [Google Scholar] [CrossRef]
- Olstrup, H.; Johansson, C.; Forsberg, B. The use of carbonaceous particle exposure metrics in health impact calculations. Int. J. Environ. Res. Public Health 2016, 13, 249. [Google Scholar] [CrossRef]
- Hvidtfeldt, U.A.; Geels, C.; Sørensen, M.; Ketzel, M.; Khan, J.; Tjønneland, A.; Christensen, J.H.; Brandt, J.; Raaschou-Nielsen, O. Long-term residential exposure to PM2.5 constituents and mortality in a Danish cohort. Environ. Int. 2019, 133, 105268. [Google Scholar] [CrossRef]
- Johnston, H.J.; Mueller, W.; Steinle, S.; Vardoulakis, S.; Tantrakarnapa, K.; Loh, M.; Cherrie, J.W. How Harmful Is Particulate Matter Emitted from Biomass Burning? A Thailand Perspective. Curr. Pollut. Rep. 2019, 5, 353–377. [Google Scholar] [CrossRef]
- Niranjan, R.; Thakur, A.K. The toxicological mechanisms of environmental soot (black carbon) and carbon black: Focus on Oxidative stress and inflammatory pathways. Front. Immunol. 2017, 8, 763. [Google Scholar] [CrossRef]
- Möller, W.; Felten, K.; Sommerer, K.; Scheuch, G.; Meyer, G.; Meyer, P.; Häussinger, K.; Kreyling, W.G. Deposition, Retention, and Translocation of Ultrafine Particles from the Central Airways and Lung Periphery. Am. J. Respir. Crit. Care Med. 2008, 177, 426–432. [Google Scholar] [CrossRef]
- Nitta, H.; Sato, T.; Nakai, S.; Maeda, K.; Aoki, S.; Ono, M. Respiratory Health Associated with Exposure to Automobile Exhaust. I. Results of Cross-sectional Studies in 1979, 1982, and 1983. Arch. Environ. Health Int. J. 1993, 48, 53–58. [Google Scholar] [CrossRef]
- English, P.; Neutra, R.; Scalf, R.; Sullivan, M.; Waller, L.; Zhu, L. Examining associations between childhood asthma and traffic flow using a geographic information system. Environ. Health Perspect. 1999, 107, 761–767. [Google Scholar] [CrossRef]
- Brauer, M.; Hoek, G.; Van Vliet, P.; Meliefste, K.; Fischer, P.H.; Wijga, A.; Koopman, L.P.; Neijens, H.J.; Gerritsen, J.; Kerkhof, M.; et al. Air Pollution from Traffic and the Development of Respiratory Infections and Asthmatic and Allergic Symptoms in Children. Am. J. Respir. Crit. Care Med. 2002, 166, 1092–1098. [Google Scholar] [CrossRef] [PubMed]
- Ozier, A.; Bara, I.; Girodet, P.-O.; Marthan, R.; Berger, P. Pathophysiology of asthma. Rev. Prat. 2011, 61, 339–345. [Google Scholar]
- Boland, S.; Baeza-Squiban, A.; Fournier, T.; Houcine, O.; Gendron, M.-C.; Chévrier, M.; Jouvenot, G.; Coste, A.; Aubier, M.; Marano, F. Diesel exhaust particles are taken up by human airway epithelial cells in vitro and alter cytokine production. Am. J. Physiol. Cell. Mol. Physiol. 1999, 276, L604–L613. [Google Scholar] [CrossRef]
- Dick, C.A.J.; Brown, D.M.; Donaldson, K.; Stone, V. The Role of Free Radicals in the Toxic and Inflammatory Effects of Four Different Ultrafine Particle Types. Inhal. Toxicol. 2003, 15, 39–52. [Google Scholar] [CrossRef]
- Wang, L.; Bao, S.; Liu, X.; Wang, F.; Zhang, J.; Dang, P.; Wang, F.; Li, B.; Lin, Y. Low-dose exposure to black carbon significantly increase lung injury of cadmium by promoting cellular apoptosis. Ecotoxicol. Environ. Saf. 2021, 224, 112703. [Google Scholar] [CrossRef]
- Elder, A.; Gelein, R.; Finkelstein, J.N.; Driscoll, K.E.; Harkema, J.; Oberdörster, G. Effects of Subchronically Inhaled Carbon Black in Three Species. I. Retention Kinetics, Lung Inflammation, and Histopathology. Toxicol. Sci. 2005, 88, 614–629. [Google Scholar] [CrossRef] [PubMed]
- Environment Canada Screening Assessment for the Challenge; Carbon Black Environment Canada, Health Canada: Ottawa, ON, Canada, 2013; pp. 1–48.
- Nemmar, A.; Hoet, P.H.M.; Vanquickenborne, B.; Dinsdale, D.; Thomeer, M.; Hoylaerts, M.F.; Vanbilloen, H.; Mortelmans, L.; Nemery, B. Passage of inhaled particles into the blood circulation in humans. Circulation 2002, 105, 411–414. [Google Scholar] [CrossRef] [PubMed]
- Shrey, K.; Suchit, A.; Deepika, D.; Shruti, K.; Vibha, R. Air pollutants: The key stages in the pathway towards the development of cardiovascular disorders. Environ. Toxicol. Pharmacol. 2011, 31, 1–9. [Google Scholar] [CrossRef]
- Grahame, T.J.; Schlesinger, R.B. Cardiovascular health and particulate vehicular emissions: A critical evaluation of the evidence. Air Qual. Atmos. Health 2010, 3, 3–27. [Google Scholar] [CrossRef]
- Simkhovich, B.Z.; Marjoram, P.; Kleinman, M.T.; Kloner, R.A. Direct and acute cardiotoxicity of ultrafine particles in young adult and old rat hearts. Basic Res. Cardiol. 2007, 102, 467–475. [Google Scholar] [CrossRef]
- Dockery, D.W.; Stone, P.H. Cardiovascular Risks from Fine Particulate Air Pollution. N. Engl. J. Med. 2007, 356, 511–513. [Google Scholar] [CrossRef]
- Meier, R.; Cascio, W.E.; Ghio, A.J.; Wild, P.; Danuser, B.; Riediker, M. Associations of Short-Term Particle and Noise Exposures with Markers of Cardiovascular and Respiratory Health among Highway Maintenance Workers. Environ. Health Perspect. 2014, 122, 726–732. [Google Scholar] [CrossRef]
- Kim, H.; Oh, S.-J.; Kwak, H.-C.; Kim, J.-K.; Lim, C.-H.; Yang, J.-S.; Park, K.; Kim, S.-K.; Lee, M.-Y. The Impact of Intratracheally Instilled Carbon Black on the Cardiovascular System of Rats: Elevation of Blood Homocysteine and Hyperactivity of Platelets. J. Toxicol. Environ. Health Part A 2012, 75, 1471–1483. [Google Scholar] [CrossRef]
- Gurgueira, S.A.; Lawrence, J.; Coull, B.; Krishna Murthy, G.G.; González-Flecha, B. Rapid increases in the steady-state concentration of reactive oxygen species in the lungs and heart after particulate air pollution inhalation. Environ. Health Perspect. 2002, 110, 749–755. [Google Scholar] [CrossRef]
- Ying, Z.; Kampfrath, T.; Thurston, G.; Farrar, B.; Lippmann, M.; Wang, A.; Sun, Q.; Chen, L.C.; Rajagopalan, S. Ambient Particulates Alter Vascular Function through Induction of Reactive Oxygen and Nitrogen Species. Toxicol. Sci. 2009, 111, 80–88. [Google Scholar] [CrossRef]
- Martinelli, N.; Olivieri, O.; Girelli, D. Air particulate matter and cardiovascular disease: A narrative review. Eur. J. Intern. Med. 2013, 24, 295–302. [Google Scholar] [CrossRef]
- Von Klot, S.; Cyrys, J.; Hoek, G.; Kühnel, B.; Pitz, M.; Kuhn, U.; Kuch, B.; Meisinger, C.; Hörmann, A.; Wichmann, H.-E.; et al. Estimated Personal Soot Exposure Is Associated with Acute Myocardial Infarction Onset in a Case-Crossover Study. Prog. Cardiovasc. Dis. 2011, 53, 361–368. [Google Scholar] [CrossRef]
- Bhaskaran, K.; Hajat, S.; Haines, A.; Herrett, E.; Wilkinson, P.; Smeeth, L. Effects of air pollution on the incidence of myocardial infarction. Heart 2009, 95, 1746–1759. [Google Scholar] [CrossRef]
- Büchner, N.; Ale-Agha, N.; Jakob, S.; Sydlik, U.; Kunze, K.; Unfried, K.; Altschmied, J.; Haendeler, J. Unhealthy diet and ultrafine carbon black particles induce senescence and disease associated phenotypic changes. Exp. Gerontol. 2013, 48, 8–16. [Google Scholar] [CrossRef]
- Larsen, P.B.; Mørck, T.A.; Nørgaard Andersen, D.; Hougaard, K.S. A Critical Review of Studies on the Reproductive and Developmental Toxicity of Nanomaterials; European Chemicals Agency: Helsinki, Finland, 2020; ISBN 9789294814234. [CrossRef]
- Umezawa, M.; Onoda, A.; Korshunova, I.; Jensen, A.C.Ø.; Koponen, I.K.; Jensen, K.A.; Khodosevich, K.; Vogel, U.; Hougaard, K.S. Maternal inhalation of carbon black nanoparticles induces neurodevelopmental changes in mouse offspring. Part. Fibre Toxicol. 2018, 15, 36. [Google Scholar] [CrossRef]
- Onoda, A.; Umezawa, M.; Takeda, K.; Ihara, T.; Sugamata, M. Effects of Maternal Exposure to Ultrafine Carbon Black on Brain Perivascular Macrophages and Surrounding Astrocytes in Offspring Mice. PLoS ONE 2014, 9, e94336. [Google Scholar] [CrossRef]
- Onoda, A.; Takeda, K.; Umezawa, M. Dose-dependent induction of astrocyte activation and reactive astrogliosis in mouse brain following maternal exposure to carbon black nanoparticle. Part. Fibre Toxicol. 2017, 14, 4. [Google Scholar] [CrossRef]
- Skovmand, A.; Jensen, A.C.Ø.; Maurice, C.; Marchetti, F.; Lauvås, A.J.; Koponen, I.K.; Jensen, K.A.; Goericke-Pesch, S.; Vogel, U.; Hougaard, K.S. Effects of maternal inhalation of carbon black nanoparticles on reproductive and fertility parameters in a four-generation study of male mice. Part. Fibre Toxicol. 2019, 16, 13. [Google Scholar] [CrossRef]
- Yokota, S.; Takeda, K.; Oshio, S. Spatiotemporal Small Non-coding RNAs Expressed in the Germline as an Early Biomarker of Testicular Toxicity and Transgenerational Effects Caused by Prenatal Exposure to Nanosized Particles. Front. Toxicol. 2021, 3, 32. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, N.; Ohsawa, M. Elevated serum immunoglobulin E to Cryptomeria japonica pollen in rats exposed to diesel exhaust during fetal and neonatal periods. BMC Pregnancy Childbirth 2002, 2, 2. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Dietert, R.R.; Holsapple, M.P. Methodologies for developmental immunotoxicity (DIT) testing. Methods 2007, 41, 123–131. [Google Scholar] [CrossRef] [PubMed]
- Manangama, G.; Audignon-Durand, S.; Migault, L.; Gramond, C.; Zaros, C.; Teysseire, R.; Sentilhes, L.; Brochard, P.; Lacourt, A.; Delva, F. Maternal occupational exposure to carbonaceous nanoscale particles and small for gestational age and the evolution of head circumference in the French Longitudinal Study of Children—Elfe study. Environ. Res. 2020, 185, 109394. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Kim, W.-H.; Kim, Y.-Y.; Park, H.-Y. Air Pollution and Central Nervous System Disease: A Review of the Impact of Fine Particulate Matter on Neurological Disorders. Front. Public Health 2020, 8, 575330. [Google Scholar] [CrossRef]
- Block, M.L.; Calderón-Garcidueñas, L. Air pollution: Mechanisms of neuroinflammation and CNS disease. Trends Neurosci. 2009, 32, 506–516. [Google Scholar] [CrossRef]
- Bandyopadhyay, A. Neurological Disorders from Ambient (Urban) Air Pollution Emphasizing UFPM and PM2.5. Curr. Pollut. Rep. 2016, 2, 203–211. [Google Scholar] [CrossRef]
- Costa, L.G.; Cole, T.B.; Coburn, J.; Chang, Y.-C.; Dao, K.; Roqué, P.J. Neurotoxicity of traffic-related air pollution. Neurotoxicology 2017, 59, 133–139. [Google Scholar] [CrossRef]
- Swardfager, W.; Lanctôt, K.; Rothenburg, L.; Wong, A.; Cappell, J.; Herrmann, N. A Meta-Analysis of Cytokines in Alzheimer’s Disease. Biol. Psychiatry 2010, 68, 930–941. [Google Scholar] [CrossRef]
- Hopkins, L.E.; Laing, E.A.; Peake, J.L.; Uyeminami, D.; Mack, S.M.; Li, X.; Smiley-Jewell, S.; Pinkerton, K.E. Repeated Iron–Soot Exposure and Nose-to-brain Transport of Inhaled Ultrafine Particles. Toxicol. Pathol. 2018, 46, 75–84. [Google Scholar] [CrossRef]
- Chen, J.-C.; Wang, X.; Wellenius, G.A.; Serre, M.L.; Driscoll, I.; Casanova, R.; McArdle, J.J.; Manson, J.E.; Chui, H.C.; Espeland, M.A. Ambient air pollution and neurotoxicity on brain structure: Evidence from women’s health initiative memory study. Ann. Neurol. 2015, 78, 466–476. [Google Scholar] [CrossRef]
- Jung, C.-R.; Lin, Y.-T.; Hwang, B.-F. Ozone, Particulate Matter, and Newly Diagnosed Alzheimer’s Disease: A Population-Based Cohort Study in Taiwan. J. Alzheimer’s Dis. 2015, 44, 573–584. [Google Scholar] [CrossRef]
- Ljungman, P.L.S.; Andersson, N.; Stockfelt, L.; Andersson, E.M.; Nilsson Sommar, J.; Eneroth, K.; Gidhagen, L.; Johansson, C.; Lager, A.; Leander, K.; et al. Long-Term Exposure to Particulate Air Pollution, Black Carbon, and Their Source Components in Relation to Ischemic Heart Disease and Stroke. Environ. Health Perspect. 2019, 127, 107012. [Google Scholar] [CrossRef]
- Tankersley, C.G.; Bierman, A.; Rabold, R. Variation in heart rate regulation and the effects of particle exposure in inbred mice. Inhal. Toxicol. 2007, 19, 621–629. [Google Scholar] [CrossRef]
- Bosetti, C.; Boffetta, P.; La Vecchia, C. Occupational exposures to polycyclic aromatic hydrocarbons, and respiratory and urinary tract cancers: A quantitative review to 2005. Ann. Oncol. 2007, 18, 431–446. [Google Scholar] [CrossRef]
- Seidler, A.; Heiskel, H.; Bickeböller, R.; Elsner, G. Association between diesel exposure at work and prostate cancer. Scand. J. Work. Environ. Health 1998, 24, 486–494. [Google Scholar] [CrossRef]
- Mauderly, J. Diesel exhaust is a pulmonary carcinogen in rats exposed chronically by inhalation. Fundam. Appl. Toxicol. 1987, 9, 208–221. [Google Scholar] [CrossRef]
- Mauderly, J.L.; Snipes, M.B.; Barr, E.B.; Belinsky, S.A.; Bond, J.A.; Brooks, A.L.; Chang, I.Y.; Cheng, Y.S.; Gillett, N.A.; Griffith, W.C. Pulmonary toxicity of inhaled diesel exhaust and carbon black in chronically exposed rats. Part I: Neoplastic and nonneoplastic lung lesions. Res. Rep. Health Eff. Inst. 1994, 1–75. Available online: https://www.healtheffects.org/publication/pulmonary-toxicity-inhaled-diesel-exhaust-and-carbon-black-chronically-exposed-rats-1 (accessed on 20 October 2022).
- Jacobsen, N.R.; Pojana, G.; White, P.; Møller, P.; Cohn, C.A.; Smith Korsholm, K.; Vogel, U.; Marcomini, A.; Loft, S.; Wallin, H. Genotoxicity, cytotoxicity, and reactive oxygen species induced by single-walled carbon nanotubes and C 60 fullerenes in the FE1-MutaTMMouse lung epithelial cells. Environ. Mol. Mutagen. 2008, 49, 476–487. [Google Scholar] [CrossRef]
- Gminski, R.; Decker, K.; Heinz, C.; Seidel, A.; Könczöl, M.; Goldenberg, E.; Grobéty, B.; Ebner, W.; Gieré, R.; Mersch-Sundermann, V. Genotoxic effects of three selected black toner powders and their dimethyl sulfoxide extracts in cultured human epithelial A549 lung cells in vitro. Environ. Mol. Mutagen. 2011, 52, 296–309. [Google Scholar] [CrossRef]
- Bond, J.A.; Johnson, N.F.; Snipes, M.B.; Mauderly, J.L.; Sega, G.A. DNA adduct formation in rat alveolar type II cells: Cells potentially at risk for inhaled diesel exhaust. Environ. Mol. Mutagen. 1990, 16, 64–69. [Google Scholar] [CrossRef] [PubMed]
- Gallagher, J.; Heinrich, U.; George, M.; Hendee, L.; Phillips, D.H.; Lewtas, J. Formation of DNA adducts in rat lung following chronic inhalation of diesel emissions, carbon black and titanium dioxide particles. Carcinogenesis 1994, 15, 1291–1299. [Google Scholar] [CrossRef] [PubMed]
- Danielsen, P.H.; Loft, S.; Jacobsen, N.R.; Jensen, K.A.; Autrup, H.; Ravanat, J.-L.; Wallin, H.; Møller, P. Oxidative Stress, Inflammation, and DNA Damage in Rats after Intratracheal Instillation or Oral Exposure to Ambient Air and Wood Smoke Particulate Matter. Toxicol. Sci. 2010, 118, 574–585. [Google Scholar] [CrossRef] [PubMed]
- Baccarelli, A.; Wright, R.O.; Bollati, V.; Tarantini, L.; Litonjua, A.A.; Suh, H.H.; Zanobetti, A.; Sparrow, D.; Vokonas, P.S.; Schwartz, J. Rapid DNA Methylation Changes after Exposure to Traffic Particles. Am. J. Respir. Crit. Care Med. 2009, 179, 572–578. [Google Scholar] [CrossRef]
- Mroz, R.M.; Schins, R.P.F.; Li, H.; Jimenez, L.A.; Drost, E.M.; Holownia, A.; MacNee, W.; Donaldson, K. Nanoparticle-driven DNA damage mimics irradiation-related carcinogenesis pathways. Eur. Respir. J. 2008, 31, 241–251. [Google Scholar] [CrossRef]
- Baccarelli, A.; Bollati, V. Epigenetics and environmental chemicals. Curr. Opin. Pediatr. 2009, 21, 243–251. [Google Scholar] [CrossRef]
- Wilson, M. Interactions between Ultrafine Particles and Transition Metals in Vivo and in Vitro. Toxicol. Appl. Pharmacol. 2002, 184, 172–179. [Google Scholar] [CrossRef]
- Corrêa, A.X.R.; Cotelle, S.; Millet, M.; Somensi, C.A.; Wagner, T.M.; Radetski, C.M. Genotoxicity assessment of particulate matter emitted from heavy-duty diesel-powered vehicles using the in vivo Vicia faba L. micronucleus test. Ecotoxicol. Environ. Saf. 2016, 127, 199–204. [Google Scholar] [CrossRef]
- Hardonnière, K.; Saunier, E.; Lemarié, A.; Fernier, M.; Gallais, I.; Héliès-Toussaint, C.; Mograbi, B.; Antonio, S.; Bénit, P.; Rustin, P.; et al. The environmental carcinogen benzo[a]pyrene induces a Warburg-like metabolic reprogramming dependent on NHE1 and associated with cell survival. Sci. Rep. 2016, 6, 30776. [Google Scholar] [CrossRef]
- Baudouin, C.; Charveron, M.; Tarroux, R.; Gall, Y. Environmental pollutants and skin cancer. Cell Biol. Toxicol. 2002, 18, 341–348. [Google Scholar] [CrossRef]
- Ushio, H.; Nohara, K.; Fujimaki, H. Effect of environmental pollutants on the production of pro-inflammatory cytokines by normal human dermal keratinocytes. Toxicol. Lett. 1999, 105, 17–24. [Google Scholar] [CrossRef]
- Fiorito, S.; Mastrofrancesco, A.; Cardinali, G.; Rosato, E.; Salsano, F.; Su, D.S.; Serafino, A.; Picardo, M. Effects of carbonaceous nanoparticles from low-emission and older diesel engines on human skin cells. Carbon N. Y. 2011, 49, 5038–5048. [Google Scholar] [CrossRef]
- Vierkötter, A.; Schikowski, T.; Ranft, U.; Sugiri, D.; Matsui, M.; Krämer, U.; Krutmann, J. Airborne Particle Exposure and Extrinsic Skin Aging. J. Investig. Dermatol. 2010, 130, 2719–2726. [Google Scholar] [CrossRef]
- Aam, B.B.; Fonnum, F. ROS scavenging effects of organic extract of diesel exhaust particles on human neutrophil granulocytes and rat alveolar macrophages. Toxicology 2007, 230, 207–218. [Google Scholar] [CrossRef]
- Ma, C.; Wang, J.; Luo, J. Activation of nuclear factor kappa B by diesel exhaust particles in mouse epidermal cells through phosphatidylinositol 3-kinase/Akt signaling pathway. Biochem. Pharmacol. 2004, 67, 1975–1983. [Google Scholar] [CrossRef]
- Chen, C.; Wang, H. Biomedical Applications and Toxicology of Carbon Nanomaterials; John Wiley & Sons: Hoboken, NJ, USA, 2016. [Google Scholar]
- Masiello, C.A. New directions in black carbon organic geochemistry. Mar. Chem. 2004, 92, 201–213. [Google Scholar] [CrossRef]
- Su, X.; Sutarlie, L.; Loh, X.J. Sensors and Analytical Technologies for Air Quality: Particulate Matters and Bioaerosols. Chem. – An Asian J. 2020, 15, 4241–4255. [Google Scholar] [CrossRef]
- Petzold, A.; Ogren, J.A.; Fiebig, M.; Laj, P.; Li, S.-M.; Baltensperger, U.; Holzer-Popp, T.; Kinne, S.; Pappalardo, G.; Sugimoto, N.; et al. Recommendations for reporting “black carbon” measurements. Atmos. Chem. Phys. 2013, 13, 8365–8379. [Google Scholar] [CrossRef]
- Migliorini, F.; De Iuliis, S.; Maffi, S.; Zizak, G. Environmental application of pulsed laser-induced incandescence. Appl. Phys. B 2013, 112, 433–440. [Google Scholar] [CrossRef]
- Weingartner, E.; Saathoff, H.; Schnaiter, M.; Streit, N.; Bitnar, B.; Baltensperger, U. Absorption of light by soot particles: Determination of the absorption coefficient by means of aethalometers. J. Aerosol Sci. 2003, 34, 1445–1463. [Google Scholar] [CrossRef]
- Adam, M.G.; Chiang, A.W.J.; Balasubramanian, R. Insights into characteristics of light absorbing carbonaceous aerosols over an urban location in Southeast Asia. Environ. Pollut. 2020, 257, 113425. [Google Scholar] [CrossRef] [PubMed]
- Sandradewi, J.; Prévôt, A.S.H.; Szidat, S.; Perron, N.; Alfarra, M.R.; Lanz, V.A.; Weingartner, E.; Baltensperger, U. Using Aerosol Light Absorption Measurements for the Quantitative Determination of Wood Burning and Traffic Emission Contributions to Particulate Matter. Environ. Sci. Technol. 2008, 42, 3316–3323. [Google Scholar] [CrossRef] [PubMed]
- Kirchstetter, T.W.; Novakov, T.; Hobbs, P.V. Evidence that the spectral dependence of light absorption by aerosols is affected by organic carbon. J. Geophys. Res. Atmos. 2004, 109, 1–12. [Google Scholar] [CrossRef]
- Good, N.; Mölter, A.; Peel, J.L.; Volckens, J. An accurate filter loading correction is essential for assessing personal exposure to black carbon using an Aethalometer. J. Expo. Sci. Environ. Epidemiol. 2017, 27, 409–416. [Google Scholar] [CrossRef] [PubMed]
- Baumgardner, D.; Popovicheva, O.; Allan, J.; Bernardoni, V.; Cao, J.; Cavalli, F.; Cozic, J.; Diapouli, E.; Eleftheriadis, K.; Genberg, P.J.; et al. Soot reference materials for instrument calibration and intercomparisons: A workshop summary with recommendations. Atmos. Meas. Tech. 2012, 5, 1869–1887. [Google Scholar] [CrossRef]
- Michelsen, H.A.; Schulz, C.; Smallwood, G.J.; Will, S. Laser-induced incandescence: Particulate diagnostics for combustion, atmospheric, and industrial applications. Prog. Energy Combust. Sci. 2015, 51, 2–48. [Google Scholar] [CrossRef]
- Stephens, M.; Turner, N.; Sandberg, J. Particle identification by laser-induced incandescence in a solid-state laser cavity. Appl. Opt. 2003, 42, 3726. [Google Scholar] [CrossRef]
- Axelsson, B.; Collin, R.; Bengtsson, P.-E. Laser-induced incandescence for soot particle size measurements in premixed flat flames. Appl. Opt. 2000, 39, 3683. [Google Scholar] [CrossRef]
- Moteki, N.; Kondo, Y. Method to measure time-dependent scattering cross sections of particles evaporating in a laser beam. J. Aerosol Sci. 2008, 39, 348–364. [Google Scholar] [CrossRef]
- Corbin, J.C.; Gysel-Beer, M. Detection of tar brown carbon with a single particle soot photometer (SP2). Atmos. Chem. Phys. 2019, 19, 15673–15690. [Google Scholar] [CrossRef]
- Miller, J.L.; Orr-Ewing, A.J. Cavity ring-down spectroscopy measurement of single aerosol particle extinction. II. Extinction of light by an aerosol particle in an optical cavity excited by a cw laser. J. Chem. Phys. 2007, 126, 174303. [Google Scholar] [CrossRef]
- Cignoli, F.; De Iuliis, S.; Manta, V.; Zizak, G. Two-dimensional two-wavelength emission technique for soot diagnostics. Appl. Opt. 2001, 40, 5370. [Google Scholar] [CrossRef]
- Huang, Q.; Wang, F.; Yan, J.; Chi, Y. Simultaneous estimation of the 3-D soot temperature and volume fraction distributions in asymmetric flames using high-speed stereoscopic images. Appl. Opt. 2012, 51, 2968. [Google Scholar] [CrossRef]
- Lin, P.; Liu, J.; Shilling, J.E.; Kathmann, S.M.; Laskin, J.; Laskin, A. Molecular characterization of brown carbon (BrC) chromophores in secondary organic aerosol generated from photo-oxidation of toluene. Phys. Chem. Chem. Phys. 2015, 17, 23312–23325. [Google Scholar] [CrossRef]
- Kong, H.; Zhang, Y.; Li, Y.; Cui, Z.; Xia, K.; Sun, Y.; Zhao, Q.; Zhu, Y. Size-Dependent Cytotoxicity of Nanocarbon Blacks. Int. J. Mol. Sci. 2013, 14, 22529–22543. [Google Scholar] [CrossRef]
- Oberdörster, G.; Sharp, Z.; Atudorei, V.; Elder, A.; Gelein, R.; Lunts, A.; Kreyling, W.; Cox, C. Extrapulmonary translocation of ultrafine carbon particles following whole-body inhalation exposure of rats. J. Toxicol. Environ. Health Part A 2002, 65, 1531–1543. [Google Scholar] [CrossRef]
- Wiebert, P.; Sanchez-Crespo, A.; Falk, R.; Philipson, K.; Lundin, A.; Larsson, S.; Möller, W.; Kreyling, W.; Svartengren, M. No significant translocation of inhaled 35-nm carbon particles to the circulation in humans. Inhal. Toxicol. 2006, 18, 741–747. [Google Scholar] [CrossRef]
- Brown, J.S.; Kim, C.S.; Reist, P.C.; Zeman, K.L.; Bennett, W.D. Generation of radiolabeled “soot-like” ultrafine aerosols suitable for use in human inhalation studies. Aerosol Sci. Technol. 2000, 32, 325–337. [Google Scholar] [CrossRef]
- Pourchez, J.; Albuquerque-Silva, I.M.D.; Cottier, M.; Clotagatide, A.; Vecellio, L.; Durand, M.; Dubois, F. Generation and characterization of radiolabelled nanosized carbonaceous aerosols for human inhalation studies. J. Aerosol Sci. 2013, 55, 1–11. [Google Scholar] [CrossRef]
- Mills, N.L.; Amin, N.; Robinson, S.D.; Anand, A.; Davies, J.; Patel, D.; De La Fuente, J.M.; Cassee, F.R.; Boon, N.A.; MacNee, W.; et al. Do inhaled carbon nanoparticles translocate directly into the circulation in humans? Am. J. Respir. Crit. Care Med. 2006, 173, 426–431. [Google Scholar] [CrossRef]
- Thrall, J. Nuclear Medicine and Molecular Imaging: The Requisites E-Book; Elsevier Health Sciences: Amsterdam, The Netherlands, 2020; ISBN 9780323550741. Available online: https://www.elsevier.com/books/nuclear-medicine-and-molecular-imaging-the-requisites/978-0-323-53037-8 (accessed on 20 October 2022).
- Sanchez-Crespo, A.; Klepczynska-Nyström, A.; Lundin, A.; Larsson, B.M.; Svartengren, M. 111 Indium-labeled ultrafine carbon particles; a novel aerosol for pulmonary deposition and retention studies. Inhal. Toxicol. 2011, 23, 121–128. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Klepczyńska-Nyström, A.; Sanchez-Crespo, A.; Andersson, M.; Falk, R.; Lundin, A.; Larsson, B.-M.; Svartengren, M. The pulmonary deposition and retention of indium-111 labeled ultrafine carbon particles in healthy individuals. Inhal. Toxicol. 2012, 24, 645–651. [Google Scholar] [CrossRef] [PubMed]
- Borges, J.B.; Velikyan, I.; Långström, B.; Sörensen, J.; Ulin, J.; Maripuu, E.; Sandström, M.; Widström, C.; Hedenstierna, G. Ventilation Distribution Studies Comparing Technegas and “Gallgas” Using 68 GaCl3 as the Label. J. Nucl. Med. 2011, 52, 206–209. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Huang, X.; Liu, Y.; Cao, D.; Lu, D.; Feng, Z.; Liu, Q.; Lin, Z.; Jiang, G. Identification, Quantification, and Imaging of the Biodistribution of Soot Particles by Mass Spectral Fingerprinting. Anal. Chem. 2021, 93, 6665–6672. [Google Scholar] [CrossRef] [PubMed]
- Houck, M.M.; Siegel, J.A. Fundamentals of Forensic Science; Elsevier: Amsterdam, The Netherlands, 2015; ISBN 9780128002315. Available online: https://www.elsevier.com/books/fundamentals-of-forensic-science/houck/978-0-12-800037-3 (accessed on 20 October 2022).
- Mandal, A.; Singha, M.; Addy, P.S.; Basak, A. Laser desorption ionization mass spectrometry: Recent progress in matrix-free and label-assisted techniques. Mass Spectrom. Rev. 2019, 38, 3–21. [Google Scholar] [CrossRef]
- Guilhaus, M. Special feature: Tutorial. Principles and instrumentation in time-of-flight mass spectrometry. Physical and instrumental concepts. J. Mass Spectrom. 1995, 30, 1519–1532. [Google Scholar] [CrossRef]
- Kehayias, J.J.; Heymsfield, S.B.; LoMonte, A.F.; Wang, J.; Pierson, R.N. In vivo determination of body fat by measuring total body carbon. Am. J. Clin. Nutr. 1991, 53, 1339–1344. [Google Scholar] [CrossRef]
- Alberts, B.; Johnson, A.; Lewis, J.; Raff, M.; Roberts, K.; Walter, P. Molecular Biology of the Cell, 4th ed.; Garland Science: New York, NY, USA, 2002; ISBN 0815340729. Available online: https://www.ncbi.nlm.nih.gov/books/NBK21054/ (accessed on 20 October 2022).
- Marquis, B.J.; Love, S.A.; Braun, K.L.; Haynes, C.L. Analytical methods to assess nanoparticle toxicity. Analyst 2009, 134, 425. [Google Scholar] [CrossRef]
- Brown, A.; Hondow, N. Electron Microscopy of Nanoparticles in Cells; Elsevier: Amsterdam, The Netherlands, 2013; Volume 5, pp. 95–120. [Google Scholar] [CrossRef]
- Jiang, S.; Shang, M.; Mu, K.; Jiang, N.; Wen, H.; Wang, R.; Wu, H.; Li, W. In vitro and in vivo toxic effects and inflammatory responses induced by carboxylated black carbon-lead complex exposure. Ecotoxicol. Environ. Saf. 2018, 165, 484–494. [Google Scholar] [CrossRef]
- Komatsu, T.; Tabata, M.; Kubo-Irie, M.; Shimizu, T.; Suzuki, K.; Nihei, Y.; Takeda, K. The effects of nanoparticles on mouse testis Leydig cells in vitro. Toxicol. Vitr. 2008, 22, 1825–1831. [Google Scholar] [CrossRef]
- Belade, E.; Armand, L.; Martinon, L.; Kheuang, L.; Fleury-Feith, J.; Baeza-Squiban, A.; Lanone, S.; Billon-Galland, M.-A.; Pairon, J.-C.; Boczkowski, J. A comparative transmission electron microscopy study of titanium dioxide and carbon black nanoparticles uptake in human lung epithelial and fibroblast cell lines. Toxicol. Vitr. 2012, 26, 57–66. [Google Scholar] [CrossRef]
- Li, L.; Mak, K.Y.; Shi, J.; Koon, H.K.; Leung, C.H.; Wong, C.M.; Leung, C.W.; Mak, C.S.K.; Chan, N.M.M.; Zhong, W.; et al. Comparative In Vitro Cytotoxicity Study on Uncoated Magnetic Nanoparticles: Effects on Cell Viability, Cell Morphology, and Cellular Uptake. J. Nanosci. Nanotechnol. 2012, 12, 9010–9017. [Google Scholar] [CrossRef][Green Version]
- Malatesta, M. Transmission Electron Microscopy as a Powerful Tool to Investigate the Interaction of Nanoparticles with Subcellular Structures. Int. J. Mol. Sci. 2021, 22, 12789. [Google Scholar] [CrossRef]
- Zhang, R.; Dai, Y.; Zhang, X.; Niu, Y.; Meng, T.; Li, Y.; Duan, H.; Bin, P.; Ye, M.; Jia, X.; et al. Reduced pulmonary function and increased pro-inflammatory cytokines in nanoscale carbon black-exposed workers. Part. Fibre Toxicol. 2014, 11, 73. [Google Scholar] [CrossRef]
- Bozzola, J.J.; Russell, L.D. Electron Microscopy: Principles and Techniques for Biologists; Jones & Bartlett Learning: Burlington, MA, USA, 1999; ISBN 0763701920. [Google Scholar]
- Hayat, M.A. Principles and Techniques of Electron Microscopy: Biological Applications; Macmillan Publishers: New York, NY, USA, 1974; pp. 229–231. [Google Scholar] [CrossRef]
- Mühlfeld, C.; Rothen-Rutishauser, B.; Vanhecke, D.; Blank, F.; Gehr, P.; Ochs, M. Visualization and quantitative analysis of nanoparticles in the respiratory tract by transmission electron microscopy. Part. Fibre Toxicol. 2007, 4, 11. [Google Scholar] [CrossRef]
- Parent, P.; Laffon, C.; Marhaba, I.; Ferry, D.; Regier, T.Z.; Ortega, I.K.; Chazallon, B.; Carpentier, Y.; Focsa, C. Nanoscale characterization of aircraft soot: A high-resolution transmission electron microscopy, Raman spectroscopy, X-ray photoelectron and near-edge X-ray absorption spectroscopy study. Carbon N. Y. 2016, 101, 86–100. [Google Scholar] [CrossRef]
- Wang, G.; Fang, N. Detecting and Tracking Nonfluorescent Nanoparticle Probes in Live Cells. Methods Enzymol. 2012, 504, 83–108. [Google Scholar] [CrossRef]
- Murphy, D.B. Fundamentals of Light Microscopy and Electronic Imaging; John Wiley & Sons: Hoboken, NJ, USA, 2002; ISBN 047123429X. [Google Scholar]
- Priest, L.; Peters, J.S.; Kukura, P. Scattering-based Light Microscopy: From Metal Nanoparticles to Single Proteins. Chem. Rev. 2021, 121, 11937–11970. [Google Scholar] [CrossRef]
- Wayne, R. Light and Video Microscopy; Elsevier: Amsterdam, The Netherlands, 2013; ISBN 9780124114845. [Google Scholar]
- You, R.; Lu, W.; Shan, M.; Berlin, J.M.; Samuel, E.L.; Marcano, D.C.; Sun, Z.; Sikkema, W.K.; Yuan, X.; Song, L.; et al. Nanoparticulate carbon black in cigarette smoke induces DNA cleavage and Th17-mediated emphysema. elife 2015, 4, e09623. [Google Scholar] [CrossRef]
- Modrzynska, J.; Berthing, T.; Ravn-Haren, G.; Jacobsen, N.R.; Weydahl, I.K.; Loeschner, K.; Mortensen, A.; Saber, A.T.; Vogel, U. Primary genotoxicity in the liver following pulmonary exposure to carbon black nanoparticles in mice. Part. Fibre Toxicol. 2018, 15, 2. [Google Scholar] [CrossRef]
- Pedata, P.; Stoeger, T.; Zimmermann, R.; Peters, A.; Oberdörster, G.; D’Anna, A. Are we forgetting the smallest sub 10 nm combustion generated nanoparticles. Part. Fibre Toxicol. 2015, 12, 34. [Google Scholar] [CrossRef] [PubMed]
- Sze, S.K.; Siddique, N.; Sloan, J.J.; Escribano, R. Raman spectroscopic characterization of carbonaceous aerosols. Atmos. Environ. 2001, 35, 561–568. [Google Scholar] [CrossRef]
- Beyssac, O.; Goffé, B.; Petitet, J.-P.; Froigneux, E.; Moreau, M.; Rouzaud, J.-N. On the characterization of disordered and heterogeneous carbonaceous materials by Raman spectroscopy. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2003, 59, 2267–2276. [Google Scholar] [CrossRef]
- Smith, E.; Dent, G. Modern Raman Spectroscopy—A Practical Approach; John Wiley & Sons, Ltd: Chichester, UK, 2004; ISBN 9780470011836. [Google Scholar]
- Bokobza, L.; Bruneel, J.-L.; Couzi, M. Raman spectroscopy as a tool for the analysis of carbon-based materials (highly oriented pyrolitic graphite, multilayer graphene and multiwall carbon nanotubes) and of some of their elastomeric composites. Vib. Spectrosc. 2014, 74, 57–63. [Google Scholar] [CrossRef]
- Dippel, B.; Heintzenberg, J. Soot characterization in atmospheric particles from different sources by NIR FT Raman spectroscopy. J. Aerosol Sci. 1999, 30, S907–S908. [Google Scholar] [CrossRef]
- Gruber, T.; Zerda, T.W.; Gerspacher, M. Raman studies of heat-treated carbon blacks. Carbon N. Y. 1994, 32, 1377–1382. [Google Scholar] [CrossRef]
- Sadezky, A.; Muckenhuber, H.; Grothe, H.; Niessner, R.; Pöschl, U. Raman microspectroscopy of soot and related carbonaceous materials: Spectral analysis and structural information. Carbon N. Y. 2005, 43, 1731–1742. [Google Scholar] [CrossRef]
- Wang, Y.; Alsmeyer, D.C.; McCreery, R.L. Raman spectroscopy of carbon materials: Structural basis of observed spectra. Chem. Mater. 1990, 2, 557–563. [Google Scholar] [CrossRef]
- Dippel, B.; Jander, H.; Heintzenberg, J. NIR FT Raman spectroscopic study of flame soot. Phys. Chem. Chem. Phys. 1999, 1, 4707–4712. [Google Scholar] [CrossRef]
- Cuesta, A.; Dhamelincourt, P.; Laureyns, J.; Martínez-Alonso, A.; Tascón, J.M.D. Raman microprobe studies on carbon materials. Carbon N. Y. 1994, 32, 1523–1532. [Google Scholar] [CrossRef]
- Knief, P.; Clarke, C.; Herzog, E.; Davoren, M.; Lyng, F.M.; Meade, A.D.; Byrne, H.J. Raman spectroscopy—A potential platform for the rapid measurement of carbon nanotube-induced cytotoxicity. Analyst 2009, 134, 1182. [Google Scholar] [CrossRef][Green Version]
- Čabanová, K.; Motyka, O.; Čábalová, L.; Hrabovská, K.; Bielniková, H.; Kuzníková, Ľ.; Dvořáčková, J.; Zeleník, K.; Komínek, P.; Kukutschová, J. Metal particles in mucus and hypertrophic tissue of the inferior nasal turbinates from the human upper respiratory tract. Environ. Sci. Pollut. Res. 2020, 27, 28146–28154. [Google Scholar] [CrossRef]
- Čabanová, K.; Motyka, O.; Bielniková, H.; Čábalová, L.; Handlos, P.; Zabiegaj, D.; Zeleník, K.; Dvořáčková, J.; Komínek, P.; Heviánková, S.; et al. Identification of the phase composition of solid microparticles in the nasal mucosa of patients with chronic hypertrophic rhinitis using Raman microspectroscopy. Sci. Rep. 2021, 11, 18989. [Google Scholar] [CrossRef]
- Fischer, M.C.; Wilson, J.W.; Robles, F.E.; Warren, W.S. Invited Review Article: Pump-probe microscopy. Rev. Sci. Instrum. 2016, 87, 031101. [Google Scholar] [CrossRef]
- Tong, L.; Liu, Y.; Dolash, B.D.; Jung, Y.; Slipchenko, M.N.; Bergstrom, D.E.; Cheng, J.-X. Label-free imaging of semiconducting and metallic carbon nanotubes in cells and mice using transient absorption microscopy. Nat. Nanotechnol. 2012, 7, 56–61. [Google Scholar] [CrossRef]
- Frank, H.A.; Cogdell, R.J. 8.6 Light Capture in Photosynthesis. In Comprehensive Biophysics; Elsevier: Amsterdam, The Netherlands, 2012; pp. 94–114. [Google Scholar]
- Hartland, G.V. Ultrafast studies of single semiconductor and metal nanostructures through transient absorption microscopy. Chem. Sci. 2010, 1, 303–309. [Google Scholar] [CrossRef]
- Chong, S.; Min, W.; Xie, X.S. Ground-State Depletion Microscopy: Detection Sensitivity of Single-Molecule Optical Absorption at Room Temperature. J. Phys. Chem. Lett. 2010, 1, 3316–3322. [Google Scholar] [CrossRef]
- Gaiduk, A.; Ruijgrok, P.V.; Yorulmaz, M.; Orrit, M. Detection limits in photothermal microscopy. Chem. Sci. 2010, 1, 343–350. [Google Scholar] [CrossRef]
- Steuwe, C.; Bové, H.; Clerinx, J.; VandeVen, M.; Fron, E.; Nawrot, T.; Ameloot, M.; Roeffaers, M. Rapid and label-free optical detection of individual carbon air pollutant nanoparticulates in biomedical samples. J. Biophotonics 2018, 11, e201700233. [Google Scholar] [CrossRef]
- Jiang, J.; Grass, D.; Zhou, Y.; Warren, W.S.; Fischer, M.C. Beyond intensity modulation: New approaches to pump-probe microscopy. Opt. Lett. 2021, 46, 1474. [Google Scholar] [CrossRef]
- Miyazaki, J.; Tsurui, H.; Hayashi-Takagi, A.; Kasai, H.; Kobayashi, T. Sub-diffraction resolution pump-probe microscopy with shot-noise limited sensitivity using laser diodes. Opt. Express 2014, 22, 9024. [Google Scholar] [CrossRef] [PubMed]
- Miyazaki, J.; Kawasumi, K.; Kobayashi, T. Resolution improvement in laser diode-based pump–probe microscopy with an annular pupil filter. Opt. Lett. 2014, 39, 4219. [Google Scholar] [CrossRef] [PubMed]
- Bové, H.; Steuwe, C.; Fron, E.; Slenders, E.; D’Haen, J.; Fujita, Y.; Uji-I, H.; Vandeven, M.; Roeffaers, M.; Ameloot, M. Biocompatible Label-Free Detection of Carbon Black Particles by Femtosecond Pulsed Laser Microscopy. Nano Lett. 2016, 16, 3173–3178. [Google Scholar] [CrossRef] [PubMed]
- Larson, A.M. Multiphoton microscopy. Nat. Photonics 2011, 5, 1. [Google Scholar] [CrossRef]
- Lévêque-Fort, S.; Georges, P. MICROSCOPY|Nonlinear Microscopy. In Encyclopedia of Modern Optics; Elsevier: Amsterdam, The Netherlands, 2005; pp. 92–103. [Google Scholar]
- Li, D.; Zheng, W.; Qu, J.Y. Two-photon autofluorescence microscopy of multicolor excitation. Opt. Lett. 2009, 34, 202–204. [Google Scholar] [CrossRef]
- Chen, X.; Nadiarynkh, O.; Plotnikov, S.; Campagnola, P.J. Second harmonic generation microscopy for quantitative analysis of collagen fibrillar structure. Nat. Protoc. 2012, 7, 654–669. [Google Scholar] [CrossRef]
- Borile, G.; Sandrin, D.; Filippi, A.; Anderson, K.I.; Romanato, F. Label-free multiphoton microscopy: Much more than fancy images. Int. J. Mol. Sci. 2021, 22, 2657. [Google Scholar] [CrossRef]
- Kobat, D.; Durst, M.E.; Nishimura, N.; Wong, A.W.; Schaffer, C.B.; Xu, C. Deep tissue multiphoton microscopy using longer wavelength excitation. Opt. Express 2009, 17, 13354–13364. [Google Scholar] [CrossRef]
- Aslam, I.; Roeffaers, M.B.J. Unique emissive behavior of combustion-derived particles under illumination with femtosecond pulsed near-infrared laser light. Nanoscale Adv. 2021, 3, 5355–5362. [Google Scholar] [CrossRef]
- Saenen, N.D.; Bové, H.; Steuwe, C.; Roeffaers, M.B.J.; Provost, E.B.; Lefebvre, W.; Vanpoucke, C.; Ameloot, M.; Nawrot, T.S. Children’s urinary environmental carbon load: A novel marker reflecting residential ambient air pollution exposure? Am. J. Respir. Crit. Care Med. 2017, 196, 873–881. [Google Scholar] [CrossRef]
- Bové, H.; Bongaerts, E.; Slenders, E.; Bijnens, E.M.; Saenen, N.D.; Gyselaers, W.; Van Eyken, P.; Plusquin, M.; Roeffaers, M.B.J.; Ameloot, M.; et al. Ambient black carbon particles reach the fetal side of human placenta. Nat. Commun. 2019, 10, 3866. [Google Scholar] [CrossRef]
- Witters, K.; Plusquin, M.; Slenders, E.; Aslam, I.; Ameloot, M.; Roeffaers, M.B.J.; Vangronsveld, J.; Nawrot, T.S.; Bové, H. Monitoring indoor exposure to combustion-derived particles using plants. Environ. Pollut. 2020, 266, 115261. [Google Scholar] [CrossRef]
- Aslam, I.; Fron, E.; Roeffaers, M.B.J. Label-free detection and size estimation of combustion-derived carbonaceous particles in a microfluidic approach. Nanoscale Adv. 2022, 4, 3272–3281. [Google Scholar] [CrossRef]
- Bongaerts, E.; Lecante, L.L.; Bové, H.; Roeffaers, M.B.J.; Ameloot, M.; Fowler, P.A.; Nawrot, T.S. Maternal exposure to ambient black carbon particles and their presence in maternal and fetal circulation and organs: An analysis of two independent population-based observational studies. Lancet Planet. Health 2022, 6, e804–e811. [Google Scholar] [CrossRef]
- Kim, K.-H.; Kabir, E.; Kabir, S. A review on the human health impact of airborne particulate matter. Environ. Int. 2015, 74, 136–143. [Google Scholar] [CrossRef]

| Characteristics | Carbon Black (CB) | Black Carbon (BC) | Brown Carbon (BrC) |
|---|---|---|---|
| Sources/Origin | Large-scale commercial production processes, abrasion of materials containing CB. | Biomass burning, diesel exhaust, forest fires, volcanic eruptions. | Biomass burning, coal burning, forest fires, biogenic sources (dust, humic matter, etc.). |
| Production/Emission estimates | ~9.8 million tons per year. | ~8.5 million tons per year. | ~6.9 million tons per year (carbon), variable. |
| Composition/Molecular structure | Elemental carbon > 97% | Source-dependent, often elemental carbon > 50% | Humic-like substances, PAHs, Biopolymers. |
| Primary particle sizes | ~15–300 nm | Smaller than primary CB particles: diesel exhaust around ~15–40 nm. | Various sizes, depending on the molecular structure. |
| General morphology (shape, form) | Aciniform aggregates and agglomerates. | Complex chains, aciniform aggregates, agglomerated spherical particles. | Individual particles, aggregates with PAHs, tarballs. |
| Exposure sources | Environmental and Occupational. | Environmental: anthropogenic and natural sources. | Environmental: anthropogenic and natural sources. |
| Environmental/Health effects | Cytotoxicity, inflammation and oxidative stress, cardiovascular and respiratory diseases. | Global warming, cytotoxicity, inflammation and oxidative stress, cardiovascular and respiratory diseases, carrier for harmful substances. | Global warming, cell apoptosis, carrier for carcinogenic NPs, persistent organic pollutants (POPs). |
| Optical properties | Absorbs light at all wavelengths from UV–Vis-nIR. | Absorbs light at all wavelengths from UV–Vis-nIR. | Absorbs light strongly in the UV region. |
| References | [42,62,63,64,65,67] | [39,42,69,70,71,72,73,74] | [39,43,61,80,81,82,83,84,85,86] |
| Detection Techniques | Detection Mechanism | Sample Types | Label-Free Approach | Model Studies | Detection in Real Samples | Limitations | References |
|---|---|---|---|---|---|---|---|
| Radiolabeling detection | Half-life probe | Cells, tissues, clinical studies | No | Possible | Not Possible | Individual gets exposed to ionizing, limited post-administration follow-up, limited to deliberate exposure. | [171,194,195,196,198] |
| Mass spectrometry | Laser desorption/ionization mass to charge ratio | Solid/liquid samples | Yes | Possible | No studies reported | Removal of impurities, complicated sample preparation, interference from the biological samples. | [86,201,203,204,205] |
| Electron microscopy | Using a focused electron beam | Dried samples | Yes | Possible | No studies reported | Complicated sample preparation, small field-of-view, investigation of dynamic cellular processes are not possible. | [207,208,210,211,212,213,215] |
| Absorption- and scattering-based microscopy | Light absorption and scattering | Solid/liquid samples | Yes | Possible | No studies reported | Low resolution, detection of very large aggregates, challenging to detect low concentrations. | [171,219,220,221,222,223,224] |
| Raman spectroscopy | Molecular vibrational fingerprinting | Solid/liquid samples | Yes | Possible | Possible, detected in human nasal mucus and hypertrophic tissues | Strong background signal from cells or tissues, long acquisition times. | [171,226,227,228,229,230,231,232,233,234,235] |
| Photothermal pump-probe microscopy | Contrast generation due to changes in the probe field intensity | Solid/liquid samples | Yes | Possible | No studies reported | Generation of spurious signal from non-CPs, cannot distinguish among different CPs, expensive. | [239,240,241,242,244,245,246,247,248] |
| Femtosecond pulsed laser microscopy (non-incandescence related WL generation) | White light emission, spectral information | Solid/liquid samples | Yes | Possible | Possible, detected in human urine and placenta samples | Possible interference from non-CP components of PM, size determination is challenging, difficult to distinguish among different CPs. | [249,250,251,252,253,255,256,257,258,259,261] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aslam, I.; Roeffaers, M.B.J. Carbonaceous Nanoparticle Air Pollution: Toxicity and Detection in Biological Samples. Nanomaterials 2022, 12, 3948. https://doi.org/10.3390/nano12223948
Aslam I, Roeffaers MBJ. Carbonaceous Nanoparticle Air Pollution: Toxicity and Detection in Biological Samples. Nanomaterials. 2022; 12(22):3948. https://doi.org/10.3390/nano12223948
Chicago/Turabian StyleAslam, Imran, and Maarten B. J. Roeffaers. 2022. "Carbonaceous Nanoparticle Air Pollution: Toxicity and Detection in Biological Samples" Nanomaterials 12, no. 22: 3948. https://doi.org/10.3390/nano12223948
APA StyleAslam, I., & Roeffaers, M. B. J. (2022). Carbonaceous Nanoparticle Air Pollution: Toxicity and Detection in Biological Samples. Nanomaterials, 12(22), 3948. https://doi.org/10.3390/nano12223948








