Surface, Structural, and Mechanical Properties Enhancement of Cr2O3 and SiO2 Co-Deposited Coatings with W or Be
Abstract
1. Introduction
2. Experimental Details
2.1. Sample Preparation
2.2. Analytical Techniques
3. Results
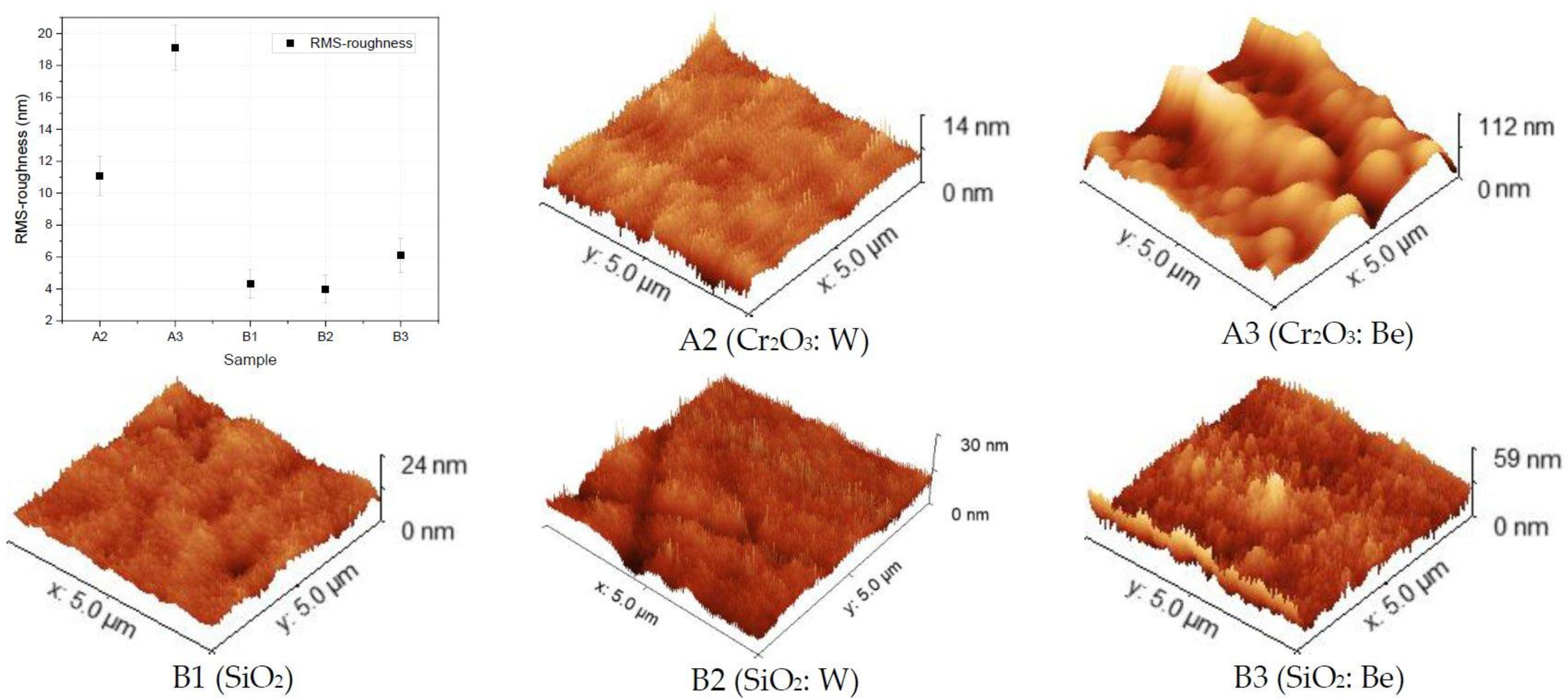
3.1. Surface Topology Characterization
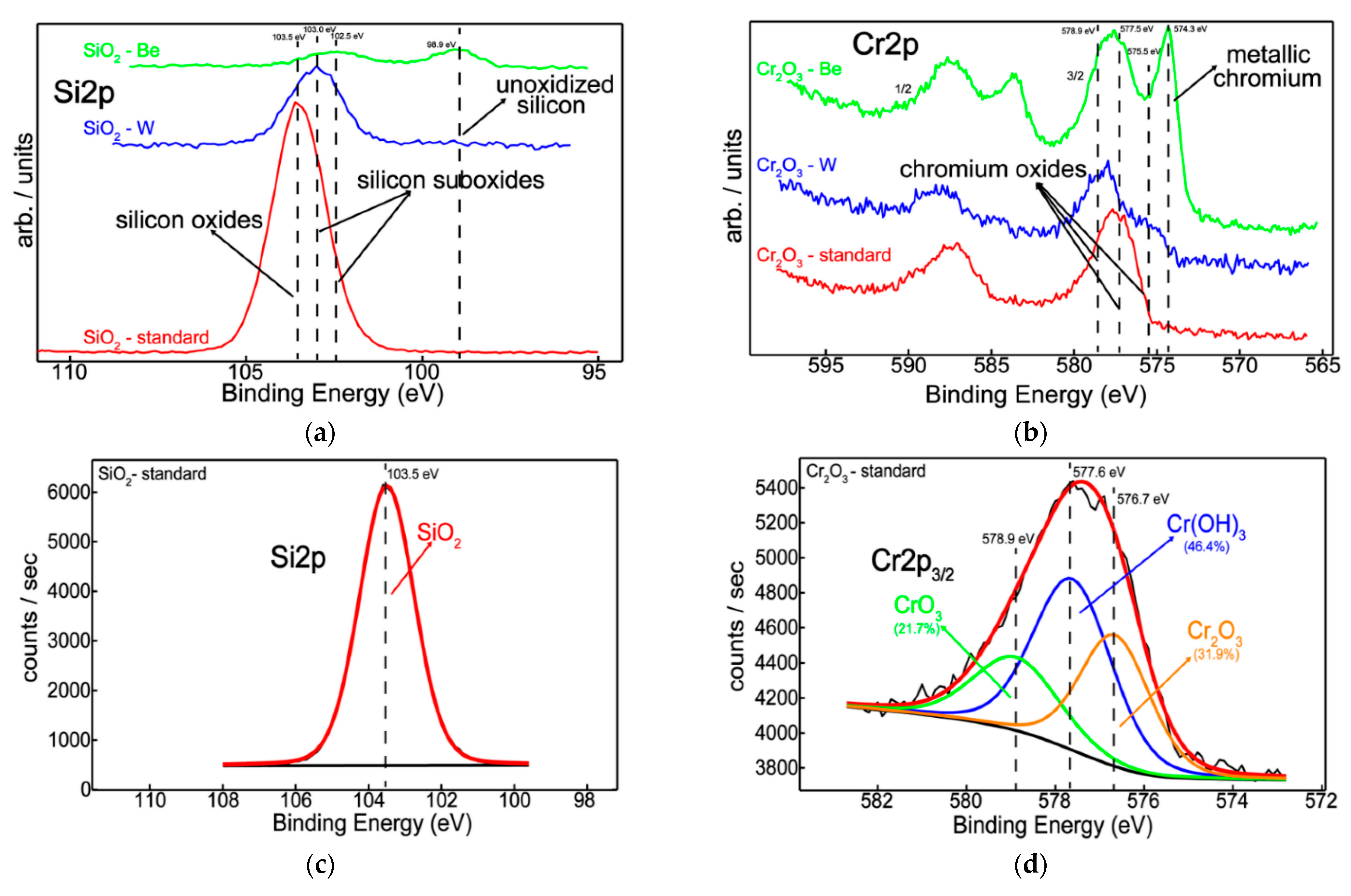
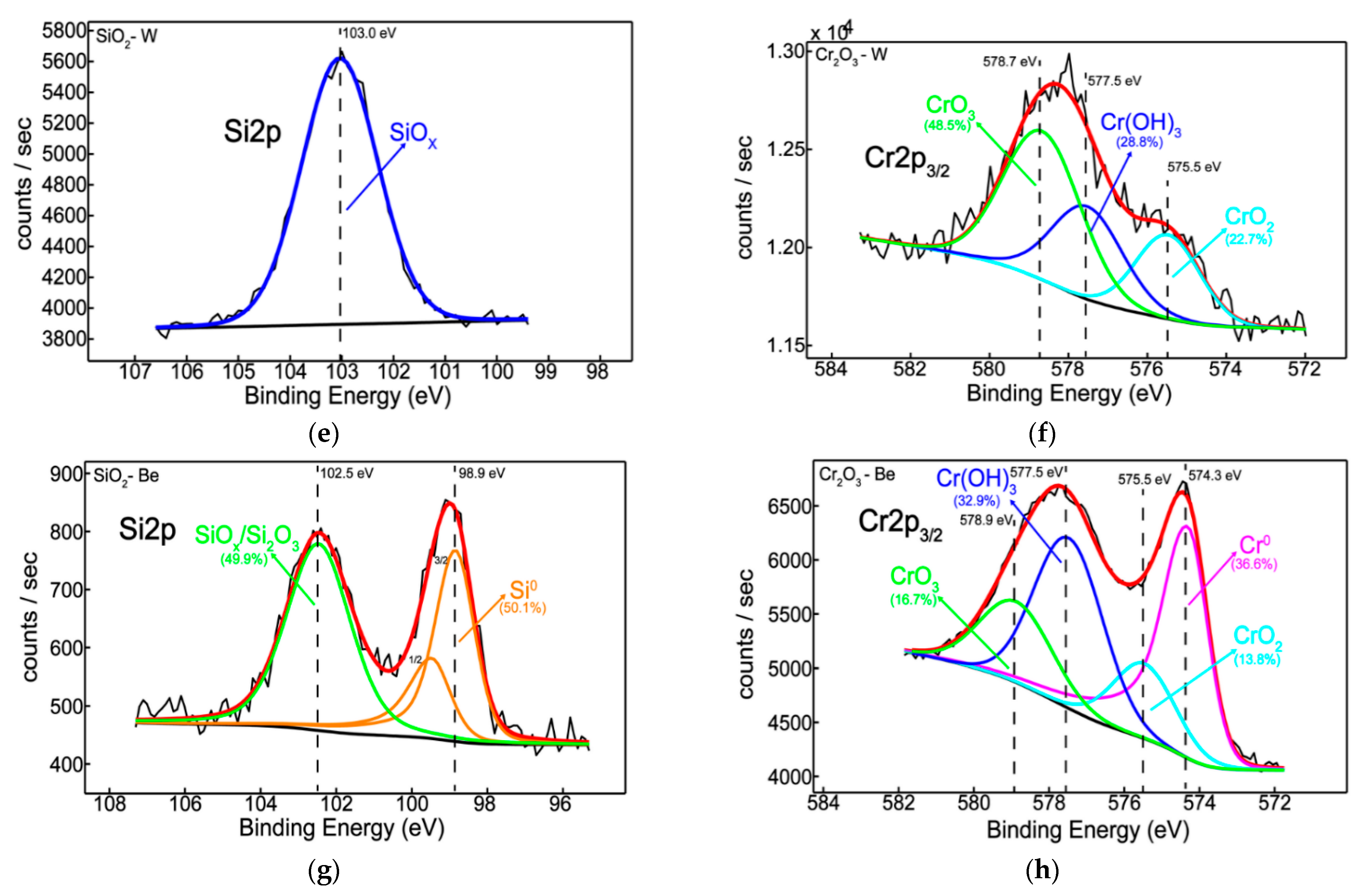
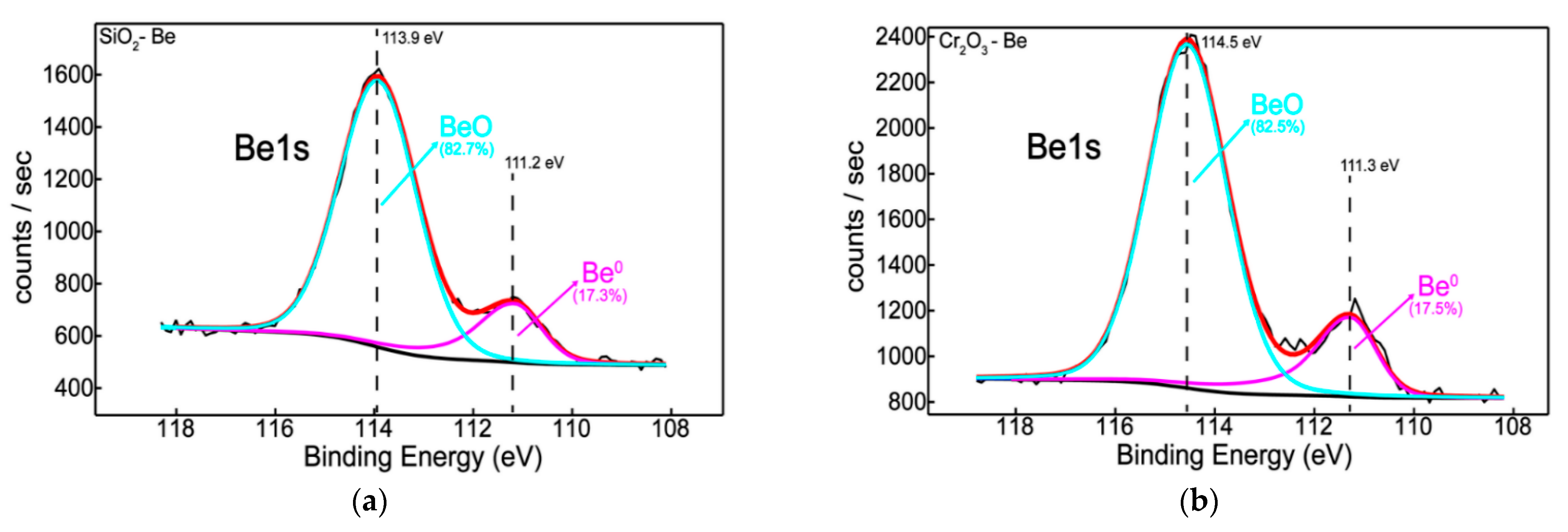
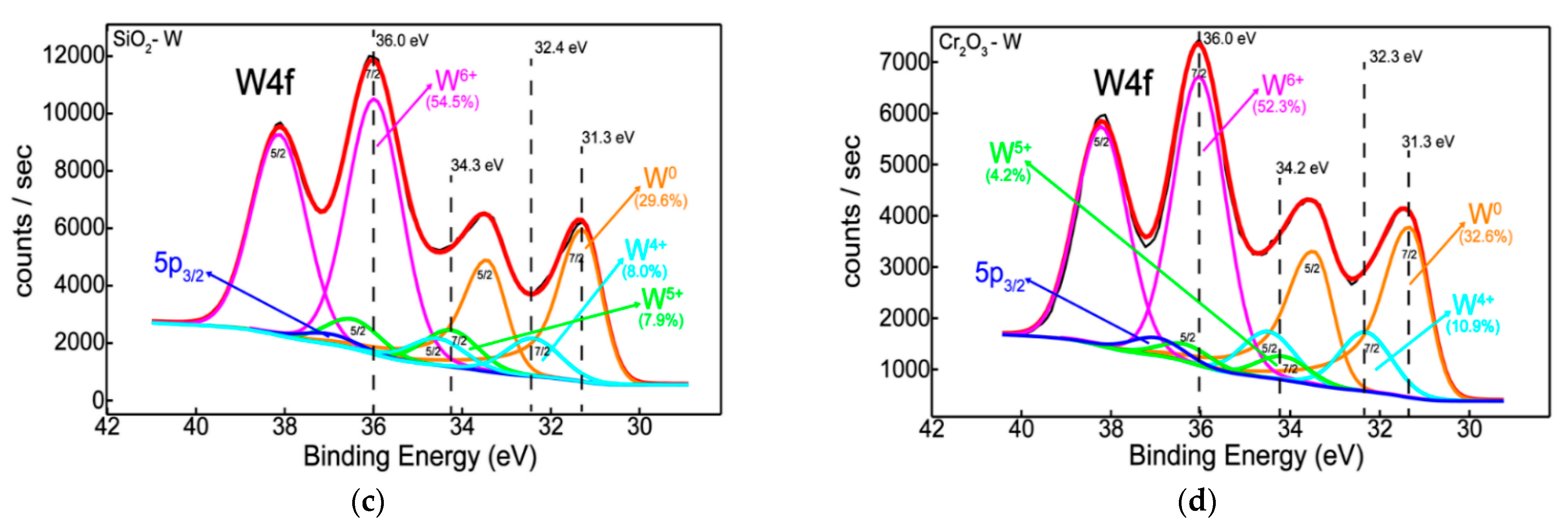
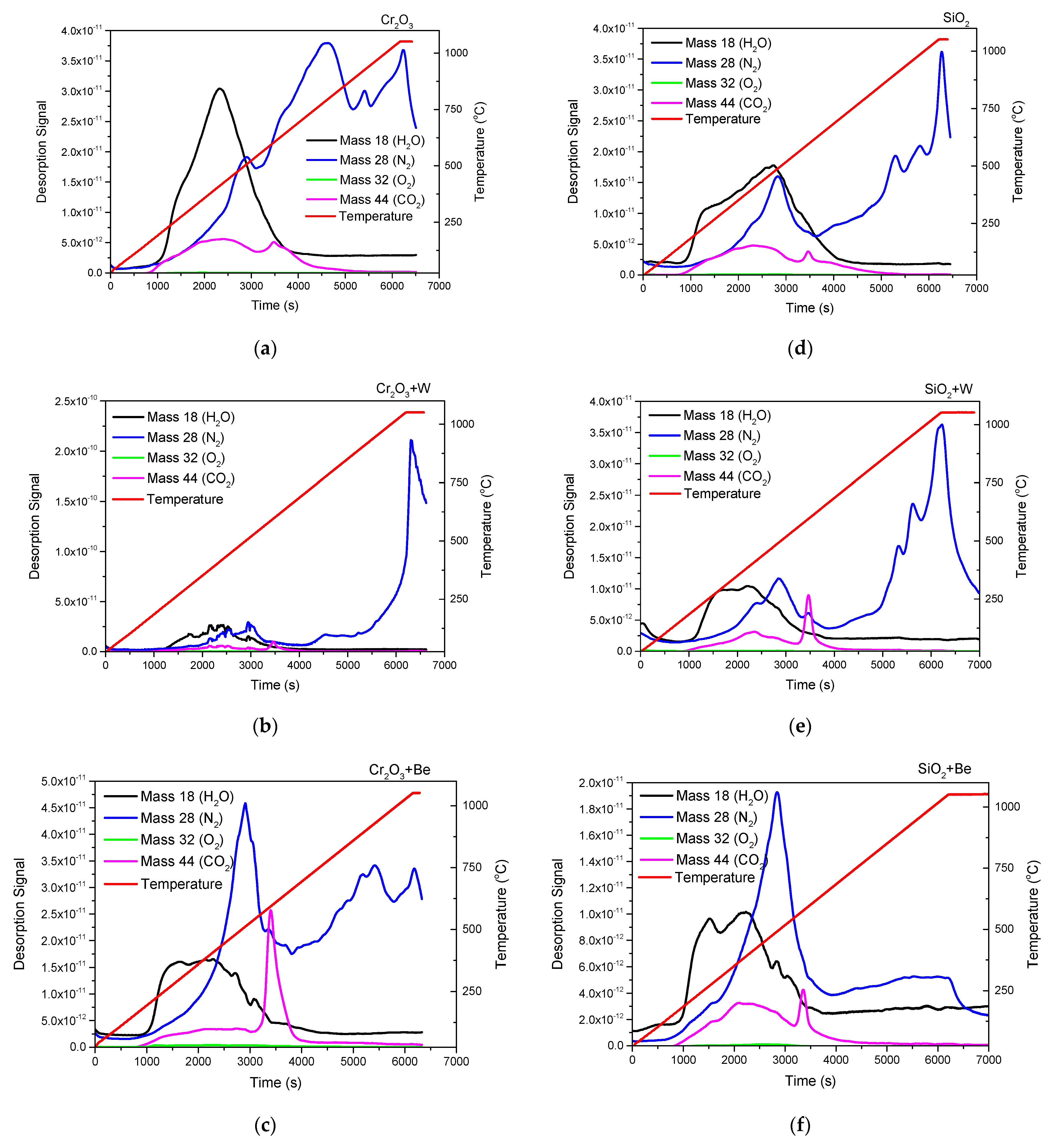
3.2. Chemical State, Structure, and Thermal Desorption Measurements
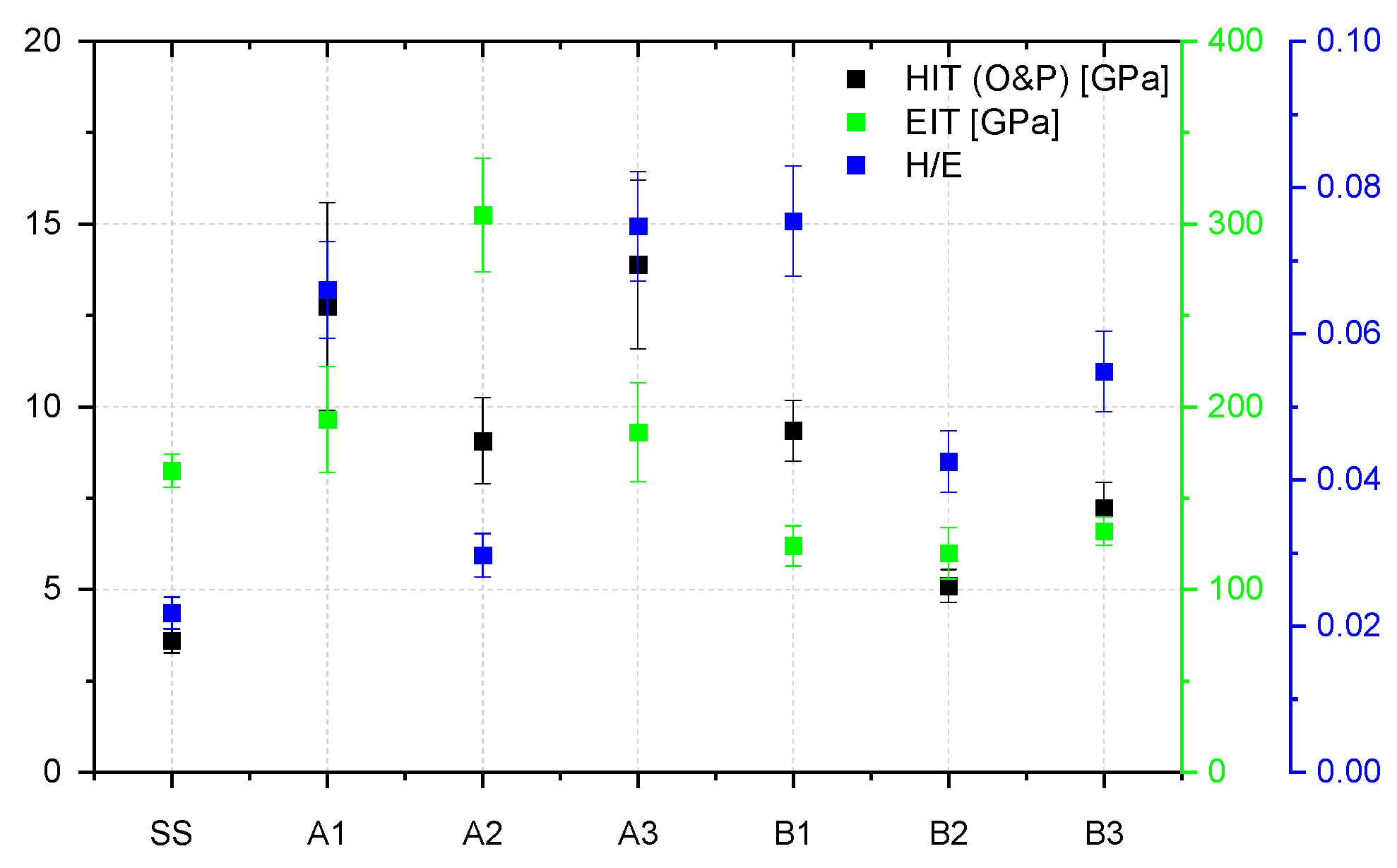
3.3. Mechanical Characterization
4. Conclusions
- Surface topology characterization: SEM images pointed out the surface morphology changes for the Cr2O3 and SiO2 oxides, while grain agglomerations promoting pore formations were observed for SiO2: Be configuration; Co-deposition with W could mitigate the RMS factor and homogenize the surface, as determined from the AFM measurements;
- Elemental distribution expressed as wt.% concentration was measured using EDX for oxide configurations (except SiO2: Be), thus evaluating the stoichiometry (O and W content); while TDS data suggest that the presence of an oxide bond at the surface has led to higher desorption peak temperatures observable for N2, excepting SiO2: Be. Moreover, defects present in the Cr2O3 matrix determined a significand desorption of H2O at 400 °C;
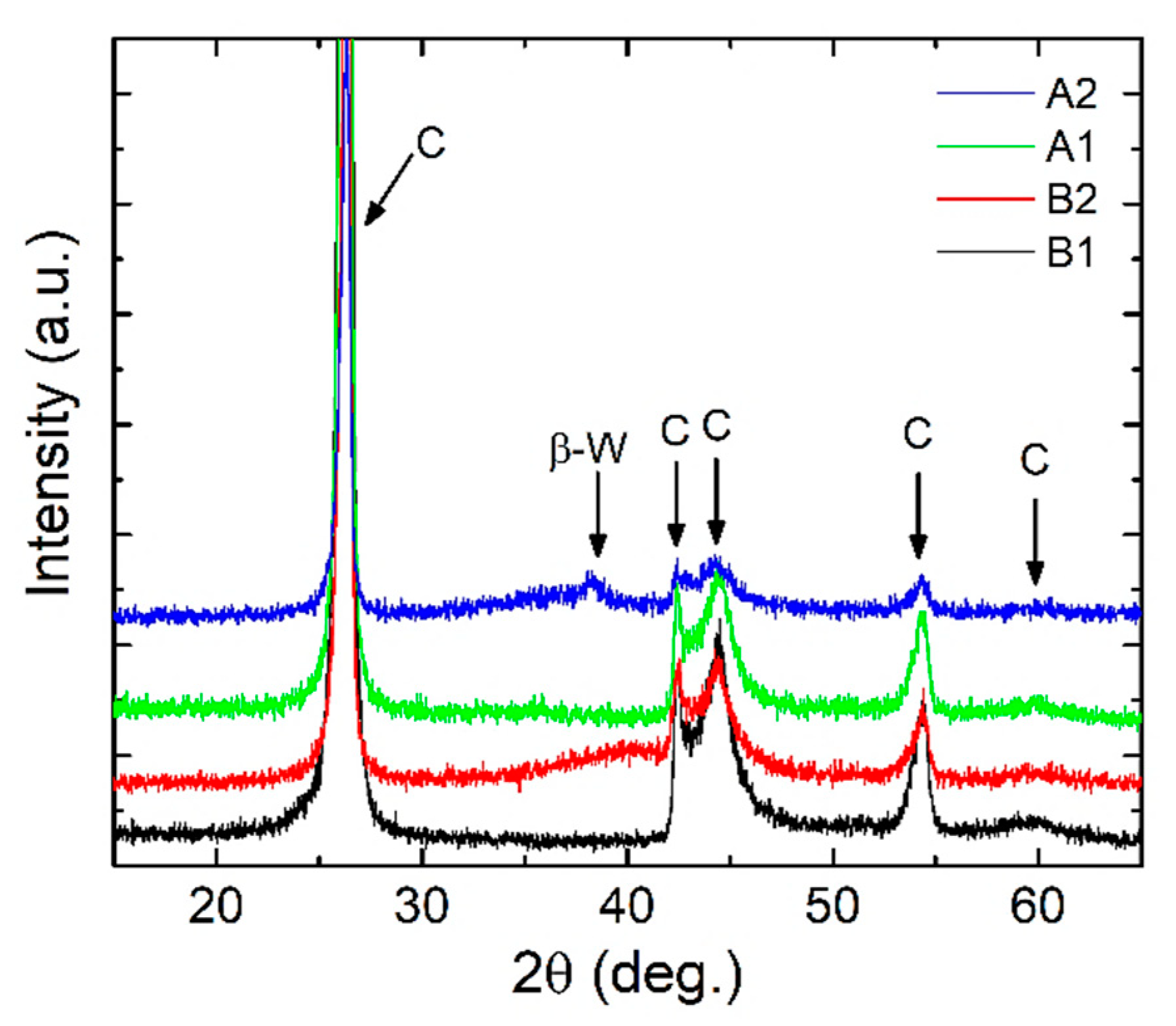
- XRD results indicated amorphous phase with no specific structural order for the measured configurations, commonly observable for oxides deposited by magnetron sputtering;
- Silicon and chromium-based systems showed improved protection against oxidation when tungsten and beryllium were embedded into the sample matrix during the deposition process of the films; also, the metallic feature among oxides of silicon and chromium can be seen only in the presence of beryllium.
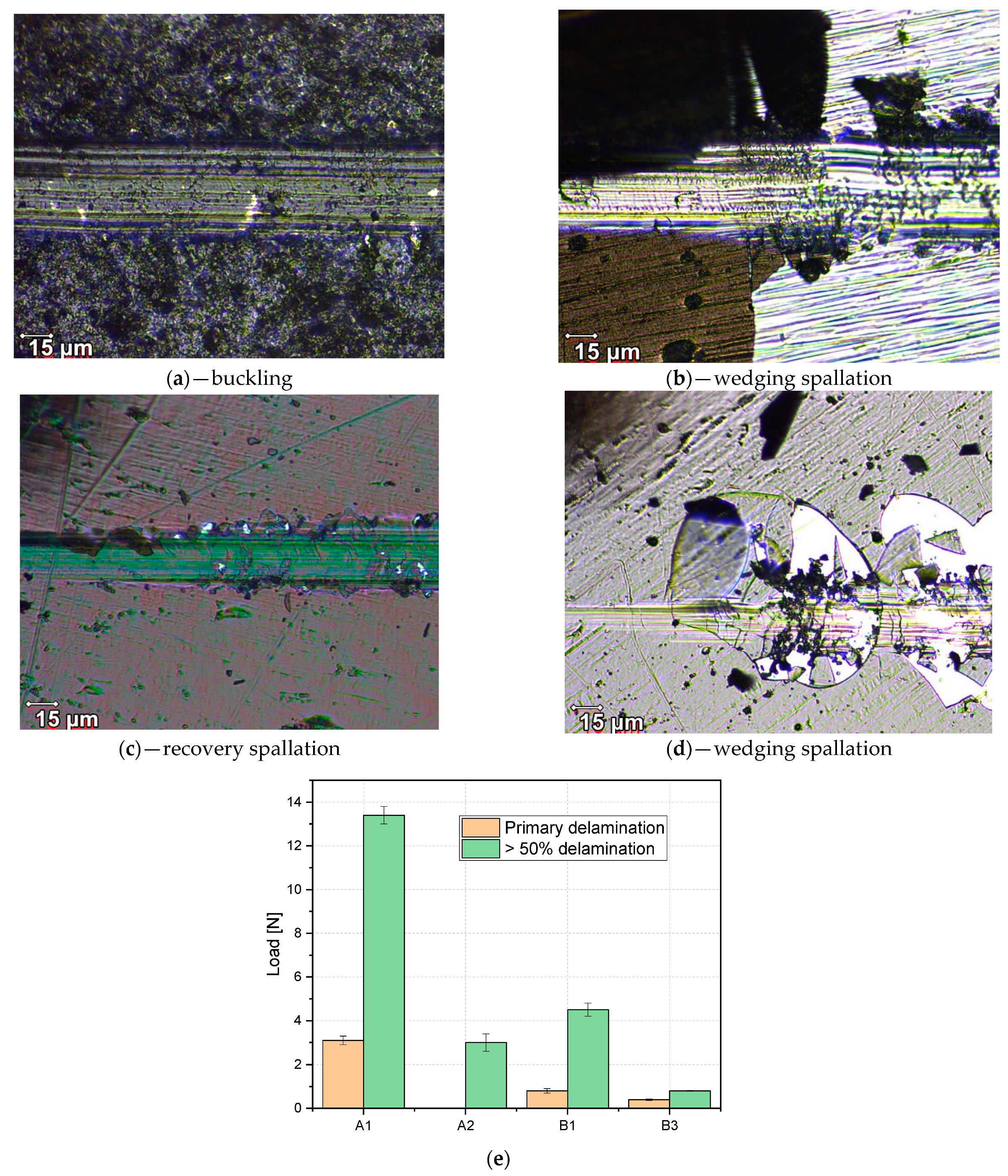
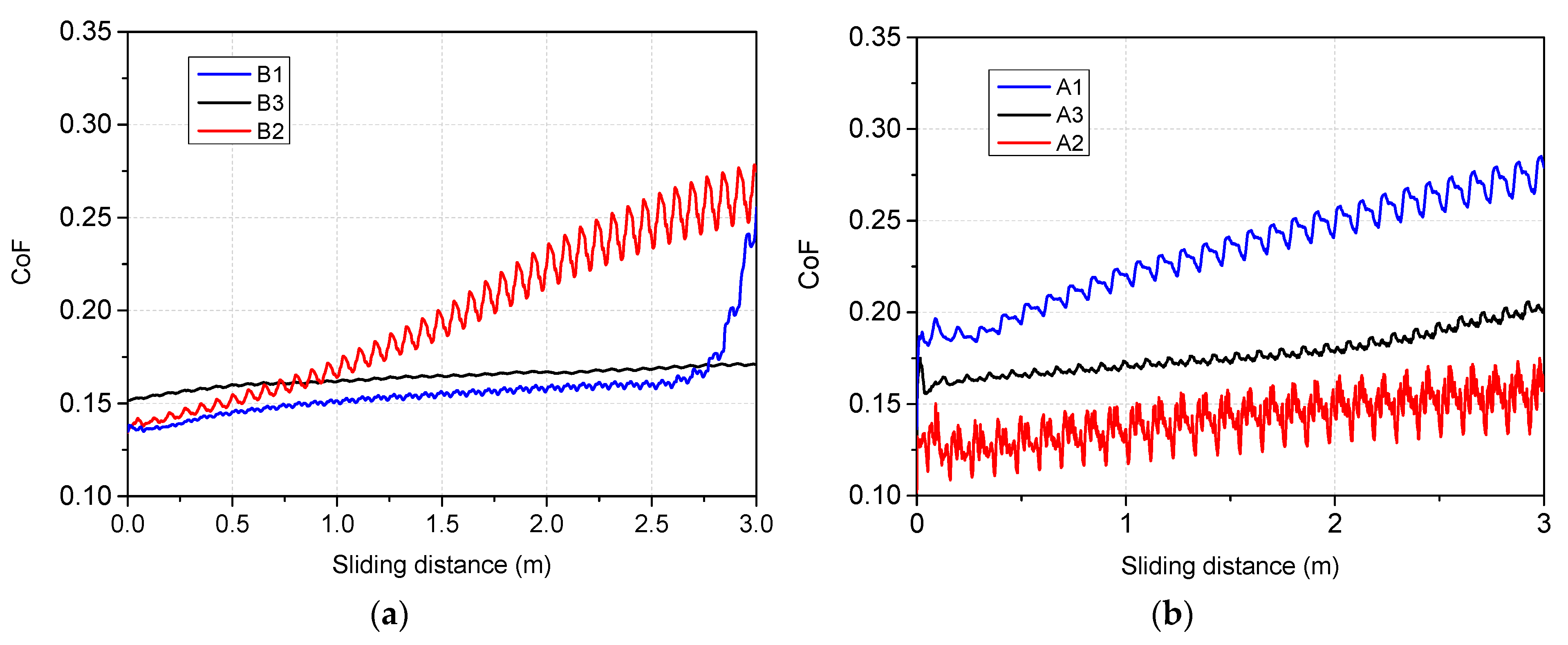
- Mechanical evaluation: at nano-scale, W in co-deposition with oxides tends to lower the hardness, while Be shows a higher probability of increasing the average hardness; Tribology measurements determined that for the SiO2 based films, the addition of W and Be leads to the increas in the coefficient of friction, while the enhancement of coating hardness is followed by the drawback of low adherence as observed for SiO2: W configuration; adherence evaluation provided several predictable results, since the buckling mode is associated with interface defects, wedge spallation is observable for rigid coatings (e.g., Cr2O3: W) and recovery spallation was observable for SiO2 in agreement with indentation results at micro-scale.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Litnovsky, A.; Wegener, T.; Klein, F.; Linsmeier, C.-H.; Rasinski, M.; Kreter, A.; Tan, X.; Schmitz, J.; Mao, Y.; Coenen, J.W.; et al. Advanced smart tungsten alloys for a future fusion power plant. Plasma Phys. Control. Fusion 2017, 59, 064003. [Google Scholar] [CrossRef]
- Nemanič, V. Hydrogen permeation barriers: Basic requirements, materials selection, deposition methods, and quality evaluation. Nucl. Mater. Energy 2019, 19, 451–457. [Google Scholar] [CrossRef]
- Lakdhar, I.; Alhussein, A.; Capelle, J.; Creus, J. Al-Ti-W alloys deposited by magnetron sputtering: Effective barrier to prevent steel hydrogen embrittlement. Appl. Surf. Sci. 2021, 567, 150786. [Google Scholar] [CrossRef]
- Hollenberg, G.W.; Simonen, E.P.; Kalinin, G.; Terlain, A. Tritium/hydrogen barrier development. Fusion Eng. Des. 1995, 28, 190–208. [Google Scholar] [CrossRef]
- Xin, Z.; Yin, X.; Ling, Y.; Zhang, Z.; Liu, X.; Liang, H.; Deng, X. Hydrogen permeation behavior and mechanism of Cr2O3/Al2O3 bipolar oxide film under plasma discharging environment. Int. J. Hydrog. Energy 2017, 42, 20869–20878. [Google Scholar] [CrossRef]
- Welter, T.; Müller, R.; Deubener, J.; Marzok, U.; Reinsch, S. Hydrogen Permeation Through Glass. Front. Mater. 2020, 6, 342. [Google Scholar] [CrossRef]
- Yu, Q.; Hao, L.; Li, S.; He, D.; Liu, X.; Jiang, L.; Wang, S. Microstructure and deuterium permeation resistance of the oxide scale prepared by initial oxidation method on vacuum solar receiver. Solid State Ion. 2013, 231, 5–10. [Google Scholar] [CrossRef]
- Luo, L.-M.; Liu, Y.-L.; Liu, D.-G.; Zheng, L.; Wu, Y.-C. Preparation technologies and performance studies of tritium permeation barriers for future nuclear fusion reactors. Surf. Coat. Technol. 2020, 403, 126301. [Google Scholar] [CrossRef]
- Chikada, T.; Suzuki, A.; Adelhelm, C.; Terai, T.; Muroga, T. Surface behavior in deuterium permeation through erbium oxide coatings. Nucl. Fusion 2011, 51, 063023. [Google Scholar] [CrossRef]
- Nakamichi, M.; Kawamura, H.; Sawai, T.; Ooka, Y.; Saito, M. Evaluation of the mechanical behaviour of Cr2O3 coating film under tensile load. Fusion Eng. Des. 1995, 29, 465–469. [Google Scholar] [CrossRef]
- Nakamichi, M.; Kawamura, H.; Teratani, T. Characterization of chemical densified coating as tritium permeation barrier. J. Nucl. Sci. Technol. 2001, 38, 1007–1013. [Google Scholar] [CrossRef][Green Version]
- Nakamichi, M.; Kawamura, H.; Teratani, T. Development of ceramic coating as tritium permeation barrier. Fusion Sci. Technol. 2002, 41, 939–942. [Google Scholar] [CrossRef]
- Checchetto, R.; Gratton, L.M.; Miotello, A.; Tosello, C. Deuterium permeation through SiO2 thin film deposited on stainless steel substrate. J. Non-Cryst. Solids 1997, 216, 65–70. [Google Scholar] [CrossRef]
- Philipps, V.; Mertens, P.; Matthews, G.F.; Maier, H.; JET-EFDA contributors. Overview of the JET ITER-like Wall Project. Fusion Eng. Des. 2010, 85, 1581–1586. [Google Scholar] [CrossRef]
- Kallenbach, A.; Neu, R.; Dux, R.; Fahrbach, H.-U.; Fuchs, J.C.; Giannone, L.; Gruber, O.; Herrmann, A.; Lang, P.T.; Lipschultz, B.; et al. Tokamak operation with high-Z plasma facing components. Plasma Phys. Control. Fusion 2005, 47, B207–B222. [Google Scholar] [CrossRef]
- Norajitra, P.; Giniyatulin, R.; Hirai, T.; Krauss, W.; Kuznetsov, V.; Mazul, I.; Ovchinnikov, I.; Reiser, J.; Ritz, G.; Ritzhaupt-Kleissl, H.-J.; et al. Current status of He-cooled divertor development for DEMO. Fusion Eng. Des. 2009, 84, 1429–1433. [Google Scholar] [CrossRef]
- Neu, R.; ASDEX Upgrade Team; EU PWI Taskforce; JET EFDA Contributors. Preparing the scientific basis for an all metal ITER. Plasma Phys. Control. Fusion 2011, 53, 124040. [Google Scholar] [CrossRef]
- Federici, G.; Skinner, C.H.; Brooks, J.N.; Coad, J.P.; Grisolia, C.; Haasz, A.A.; Hassanein, A.; Philipps, V.; Pitcher, C.S.; Roth, J.; et al. Plasma-material interactions in current tokamaks and their implications for next step fusion reactors. Nucl. Fusion 2001, 41, 1967. [Google Scholar] [CrossRef]
- Loarte, A.; Lipschultz, B.; Kukushkin, A.S.; Matthews, G.F.; Stangeby, P.C.; Asakura, N.; Counsell, G.F.; Federici, G.; Kallenbach, A.; Krieger, K.; et al. Chapter 4: Power and particle control. Nucl. Fusion 2007, 47, S203. [Google Scholar] [CrossRef]
- Roth, J.; Tsitrone, E.; Loarte, A.; Loarer, T.; Counsell, G.; Neu, R.; Philipps, V.; Brezinsek, S.; Lehnen, M.; Coad, P.; et al. Recent analysis of key plasma wall interactions issues for ITER. J. Nucl. Mater. 2009, 390–391, 1–9. [Google Scholar] [CrossRef]
- Philipps, V.; Wienhold, P.; Kirschner, A.; Rubel, M. Erosion and redeposition of wall material in controlled fusion devices. Vacuum 2002, 67, 399. [Google Scholar] [CrossRef]
- Wei, M.; Ruys, A.; Milthorpe, B.; Sorrell, C.C.; Evans, J.H. Electrophoretic Deposition of Hydroxyapatite Coatings on Metal Substrates: A Nanoparticulate Dual-Coating Approach. J. Sol-Gel Sci. Technol. 2001, 21, 39–48. [Google Scholar] [CrossRef]
- Nath, M.; Tripathi, H.S. Thermo-mechanical behavior of Al2O3–Cr2O3 refractories: Effect of TiO2. Ceram. Int. 2015, 41, 3109–3115. [Google Scholar] [CrossRef]
- Chen, S.; Ma, H.; Wang, S.; Shen, N.; Xiao, J.; Zhou, H.; Zhao, X.; Li, Y.; Yi, X. Vanadium oxide thin films deposited on silicon dioxide buffer layers by magnetron sputtering. Thin Solid Film. 2006, 497, 267–269. [Google Scholar] [CrossRef]
- Wu, W.-F.; Chiou, B.-S. Properties of radio frequency magnetron sputtered silicon dioxide films. Appl. Surf. Sci. 1996, 99, 237–243. [Google Scholar] [CrossRef]
- Wu, W.-F.; Chiou, B.-S. Optical and mechanical properties of reactively sputtered silicon dioxide films. Semicond. Sci. Technol. 1996, 11, 1317–1321. [Google Scholar] [CrossRef]
- Carneiro, J.O.; Machado, F.; Rebouta, L.; Vasilevskiy, M.I.; Lanceros-Méndez, S.; Teixeira, V.; Costa, M.F.; Samantilleke, A.P. Compositional, Optical and Electrical Characteristics of SiOx Thin Films Deposited by Reactive Pulsed DC Magnetron Sputtering. Coatings 2019, 9, 468. [Google Scholar] [CrossRef]
- Kawasaki, H.; Ohshima, T.; Yagyu, Y.; Ihara, T.; Shinohara, M.; Suda, Y. Preparation of Sn doped SiO2 thin films by magnetron sputtering deposition using metal and metal-oxide powder targets. Jpn. J. Appl. Phys. 2019, 58, SAAD04. [Google Scholar] [CrossRef]
- Thomson, S.; Pilatzke, K.; McCrimmon, K.; Castillo, I.; Suppiah, S. Tritium Permeation Characterization of Materials for Fusion and Generation IV Very High Temperature Reactors. Fusion Sci. Technol. 2015, 67. [Google Scholar] [CrossRef]
- Lungu, M.; Staicu, C.; Baiasu, F.; Marin, A.; Butoi, B.; Cristea, D.; Pompilian, O.G.; Locovei, C.; Porosnicu, C. Deposition, Morphological, and Mechanical Evaluation of W and Be-Al2O3 and Er2O3 Co-Sputtered Films in Comparison with Pure Oxides. Coatings 2021, 11, 1430. [Google Scholar] [CrossRef]
- Li, B.; Zhang, W.; Zhang, W.; Huan, Y. Preparation of Ni-W/SiC nanocomposite coatings by electrochemical deposition. J. Alloy. Compd. 2017, 702, 38–50. [Google Scholar] [CrossRef]
- Nečas, D.; Klapetek, P. Gwyddion: An open-source software for SPM data analysis. Cent. Eur. J. Phys. 2012, 10, 181–188. [Google Scholar] [CrossRef]
- Oliver, W.C.; Pharr, G.M. Measurement of hardness and elastic modulus by instrumented indentation: Advances in understanding and refinements to methodology. J. Mater. Res. 2004, 19, 3–20. [Google Scholar] [CrossRef]
- Henke, L.; Nagy, N.; Krull, U.J. An AFM determination of the effects on surface roughness caused by cleaning of fused silica and glass substrates in the process of optical biosensor preparation. Biosens. Bioelectron. 2002, 17, 547–555. [Google Scholar] [CrossRef]
- Beltowska-Lehman, E.; Bigos, A.; Indyka, P.; Chojnacka, A.; Drewienkiewicz, A.; Zimowski, S.; Kot, M.; Szczerba, M.J. Optimisation of the electrodeposition process of Ni-W/ZrO2 nanocomposites. J. Electroanal. Chem. 2018, 813, 39–51. [Google Scholar] [CrossRef]
- Jeong, S.-H.; Kim, J.-K.; Kim, B.-S.; Shim, S.-H.; Lee, B.-T. Characterization of SiO2 and TiO2 films prepared using RF magnetron sputtering and their application to anti-reflection coating. Vacuum 2004, 76, 507–515. [Google Scholar] [CrossRef]
- Liu, J.Y.; Hui, J.F.; Sun, M.Y.; Liu, X.S.; Lu, W.H.; Ma, C.H.; Zhang, Q.B. Sputtering pressure influence on growth morphology, surface roughness, and electrical resistivity for strong anisotropy beryllium film. Chin. Phys. B 2014, 23, 066804. [Google Scholar] [CrossRef]
- Petti, D.A.; Smolik, G.R.; Ander, R.A. On the mechanisms associated with the chemical reactivity of Be in steam. J. Nucl. Mater. 2000, 283–287 Pt II, 1390–1395. [Google Scholar] [CrossRef]
- Naumkin, A.V.; Kraut-Vass, A.; Gaarenstroom, S.W.; Powell, C.J. NIST X-ray Photoelectron Spectroscopy Database, NIST Standard Reference Database 20; version 4.1; National Institute of Standards and Technology: Gaithersburg, MD, USA, 2012.
- Chao, S.S.; Takagi, Y.; Lucovsky, G.; Pai, P.; Custer, R.C.; Tyler, J.E.; Keem, J.E. Chemical states study of Si in SiOx films grown by PECVD. Appl. Surf. Sci. 1986, 26, 575–583. [Google Scholar] [CrossRef]
- Olsson, C.-O.A.; Mathieu, H.-J.; Landolt, D. Angle-resolved XPS analysis of molybdenum and tungsten in passive films on stainless steel PVD alloys. Surf. Interface Anal. 2002, 34, 130–134. [Google Scholar] [CrossRef]
- Zhang, J.; Chang, X.; Li, C.; Li, A.; Liu, S.; Wang, T.; Gong, J. WO3 photoanodes with controllable bulk and surface oxygen vacancies for photoelectrochemical water oxidation. Mater. Chem. A 2018, 6, 3350–3354. [Google Scholar] [CrossRef]
- Mohammadtaheri, M.; Yang, Q.; Li, Y.; Corona-Gomez, J. The Effect of Deposition Parameters on the Structure and Mechanical Properties of Chromium Oxide Coatings Deposited by Reactive Magnetron Sputtering. Coatings 2018, 8, 111. [Google Scholar] [CrossRef]
- Zhao, C.; Zhao, L.; Liu, J.; Liu, Z.; Chen, Y. Effect of sputtering power on the properties of SiO2 films grown by radio frequency magnetron sputtering at room temperature. Opt. Quantum Electron. 2021, 53, 15. [Google Scholar] [CrossRef]
- Makepeace, C.; Pardanaud, C.; Roubin, P.; Borodkina, I.; Ayres, C.; Coad, P.; Baron-Wiechec, A.; Jepu, I.; Heinola, K.; Widdowson, A.; et al. The effect of beryllium oxide on retention in JET ITER-like wall tiles. Nucl. Mater. Energy 2019, 19, 346–351. [Google Scholar] [CrossRef]
- Zhou, B.; Prorok, B.C. A Discontinuous Elastic Interface Transfer Model of Thin Film Nanoindentation. Exp. Mech. 2010, 50, 793–801. [Google Scholar] [CrossRef]
- Bull, S.J.; Berasetegui, E.G. An overview of the potential of quantitative coating adhesion measurement by scratch testing. Tribol. Int. 2006, 39, 99–114. [Google Scholar] [CrossRef]
- Chang, Y.-Y.; Wang, D.-Y.; Chang, C.-H.; Wu, W. Tribological analysis of nano-composite diamond-like carbon films deposited by unbalanced magnetron sputtering. Surf. Coat. Technol. 2004, 184, 349–355. [Google Scholar] [CrossRef]
- Ye, T.; Ma, J.; Jia, Z.; Li, T.; Liu, W.; Yu, W. Microstructure, mechanical properties and low-temperature tribological behavior of Cr/Cr-W/W-DLC/DLC multilayer coatings on 5A06 Al alloy. J. Mater. Res. Technol. 2022, 18, 810–819. [Google Scholar] [CrossRef]


| Sample Id | Configuration | RF [W] | DC | Pressure [10−3 mbar] | Deposition Rate ×10−1 [nm/s] | Validated Layer Thickness [µm] | |
|---|---|---|---|---|---|---|---|
| U [kV] | I [A] | ||||||
| A1 | Cr2O3 | 100 | Not applicable | 4 | 0.2 | 5 | |
| A2 | Cr2O3: W | 0.28 | 0.04 | 4 | 0.2: 0.06 | 5 | |
| A3 | Cr2O3:Be | 0.55 | 0.15 | 4 | 0.2: 0.6 | 5 | |
| B1 | SiO2 | Not applicable | 1.25 | 0.4 | 1.6 | ||
| B2 | SiO2: W | 0.27 | 0.02 | 4 | 0.4: 0.3 | 5 | |
| B3 | SiO2: Be | 0.5 | 0.15 | 4 | 0.4: 0.6 | 5 | |
| Sample | Element | Weight % | Error % |
|---|---|---|---|
| A1 | O K | 31.2 | 5.7 |
| Fe L | 3.6 | 42.8 | |
| Cr K | 65.1 | 11.9 | |
| A2 | O K | 13.1 | 21.3 |
| W M | 48.5 | 10.1 | |
| Cr K | 38.4 | 22.7 | |
| A3 | O K | 32.4 | 5.3 |
| Fe L | 4.6 | 43.6 | |
| Cr K | 63.0 | 12.7 | |
| B1 | O K | 47.6 | 8.9 |
| Fe L | 3.6 | 32.3 | |
| Si K | 51.3 | 11.1 | |
| B2 | O K | 12.1 | 16.6 |
| Fe L | 6.4 | 45.5 | |
| Si K | 10.4 | 16.5 | |
| W M | 71.1 | 10.3 | |
| B3 | O K | 47.7 | 9.1 |
| Fe L | 5.8 | 26.3 | |
| Si K | 46.5 | 11.6 |
| Sample | Be1s | O1s | Si2p | W4f | Cr2p |
|---|---|---|---|---|---|
| A1 | - | 90.7 | - | - | 9.3 |
| A2 | - | 75.5 | - | 18.2 | 6.3 |
| A3 | 36.2 | 57.5 | - | - | 6.3 |
| B1 | - | 74.4 | 25.6 | - | - |
| B2 | - | 75.8 | 10.5 | 13.7 | - |
| B3 | 36.9 | 58.1 | 5.0 | - | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lungu, M.; Cristea, D.; Baiasu, F.; Staicu, C.; Marin, A.; Pompilian, O.G.; Butoi, B.; Locovei, C.; Porosnicu, C. Surface, Structural, and Mechanical Properties Enhancement of Cr2O3 and SiO2 Co-Deposited Coatings with W or Be. Nanomaterials 2022, 12, 2870. https://doi.org/10.3390/nano12162870
Lungu M, Cristea D, Baiasu F, Staicu C, Marin A, Pompilian OG, Butoi B, Locovei C, Porosnicu C. Surface, Structural, and Mechanical Properties Enhancement of Cr2O3 and SiO2 Co-Deposited Coatings with W or Be. Nanomaterials. 2022; 12(16):2870. https://doi.org/10.3390/nano12162870
Chicago/Turabian StyleLungu, Mihail, Daniel Cristea, Flaviu Baiasu, Cornel Staicu, Alexandru Marin, Oana Gloria Pompilian, Bogdan Butoi, Claudiu Locovei, and Corneliu Porosnicu. 2022. "Surface, Structural, and Mechanical Properties Enhancement of Cr2O3 and SiO2 Co-Deposited Coatings with W or Be" Nanomaterials 12, no. 16: 2870. https://doi.org/10.3390/nano12162870
APA StyleLungu, M., Cristea, D., Baiasu, F., Staicu, C., Marin, A., Pompilian, O. G., Butoi, B., Locovei, C., & Porosnicu, C. (2022). Surface, Structural, and Mechanical Properties Enhancement of Cr2O3 and SiO2 Co-Deposited Coatings with W or Be. Nanomaterials, 12(16), 2870. https://doi.org/10.3390/nano12162870







