A Numerical Investigation of a Melting Rate Enhancement inside a Thermal Energy Storage System of Finned Heat Pipe with Nano-Enhanced Phase Change Material
Abstract
:1. Introduction
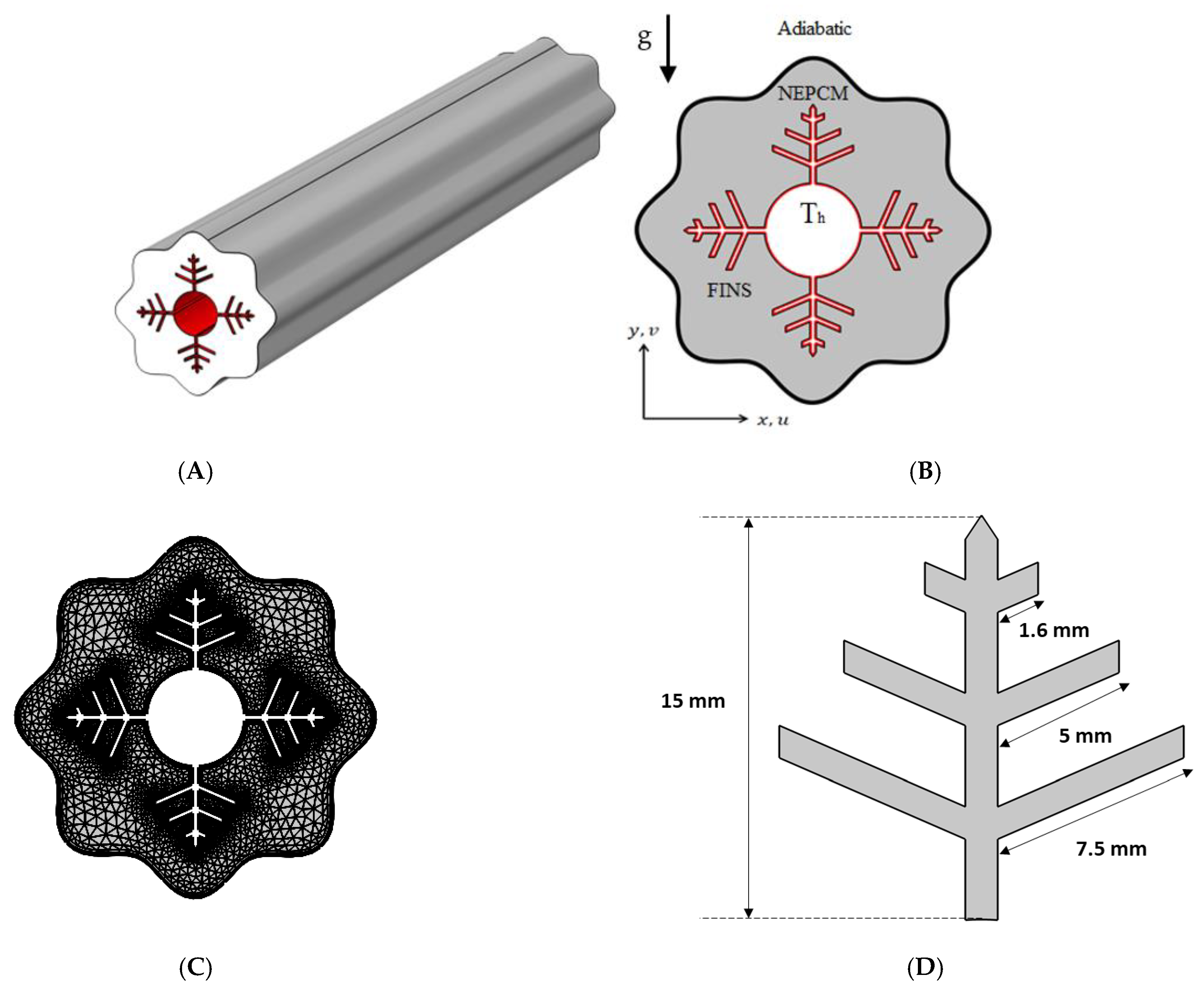
2. Problem Description
2.1. Mathematical Model
- The flow of liquid NePCM is regarded to be incompressible and laminar.
- Overlooking the volumetric impact of viscous dissipation and heat source.
- The shell wall is assumed to maintain a constant temperature, ignoring the heat transfer resistance of the container wall and the convective heat transfer process inside the tube.
- There is no heat transfer between the shell and its surroundings.
2.2. Validation and Mesh Independence Study
3. Results and Discussion
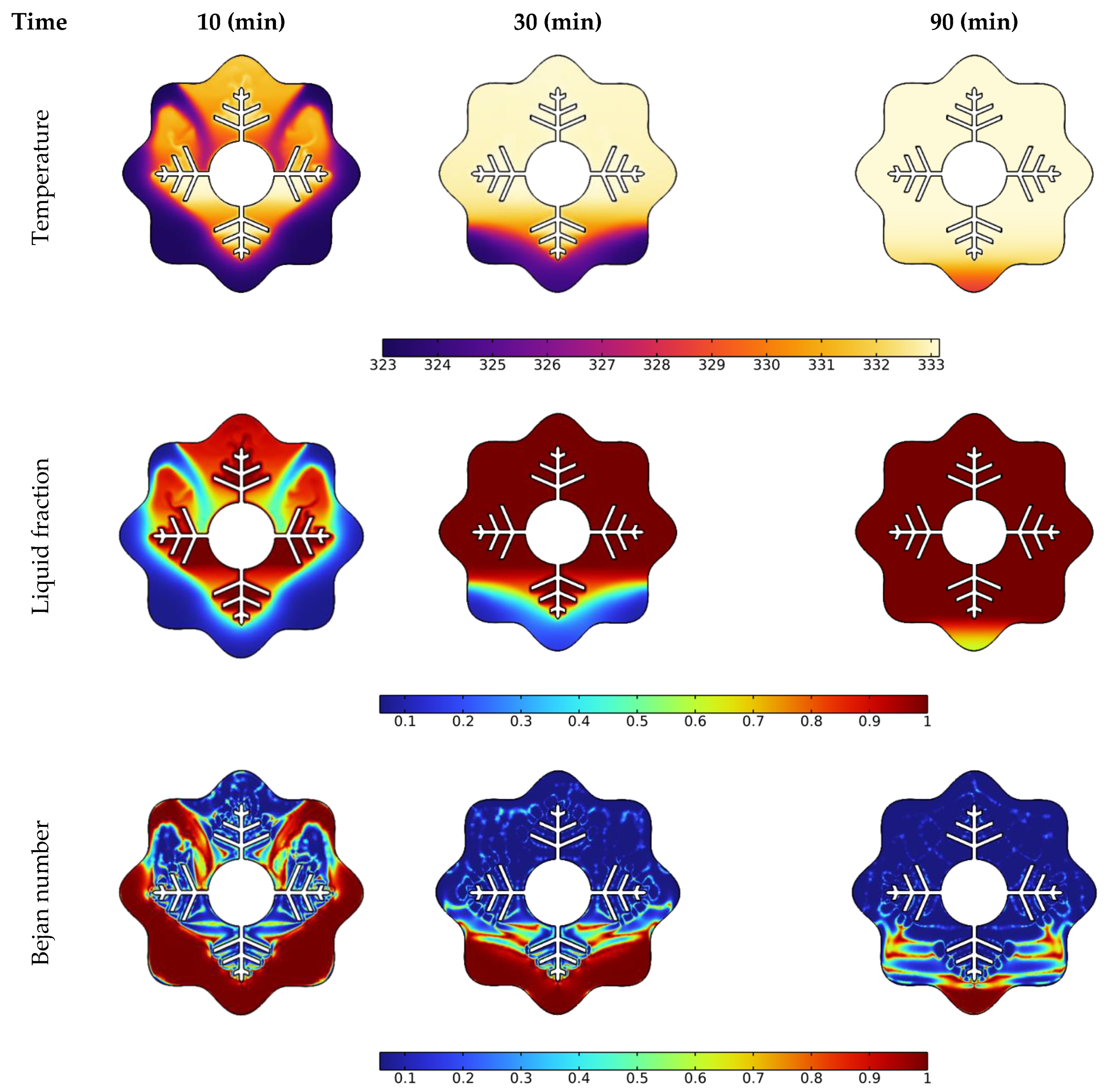
3.1. Time Needed for Melting NePCM
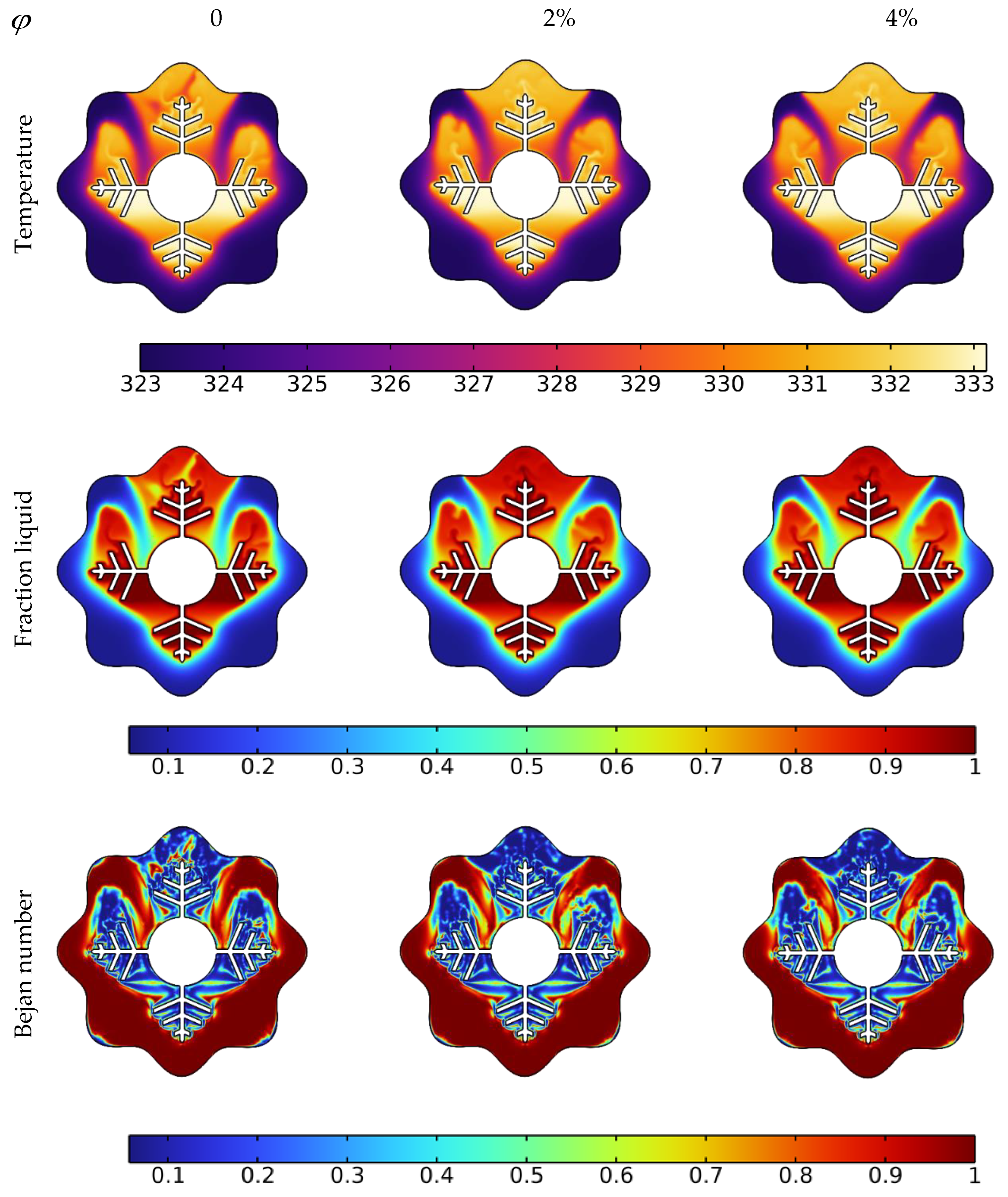
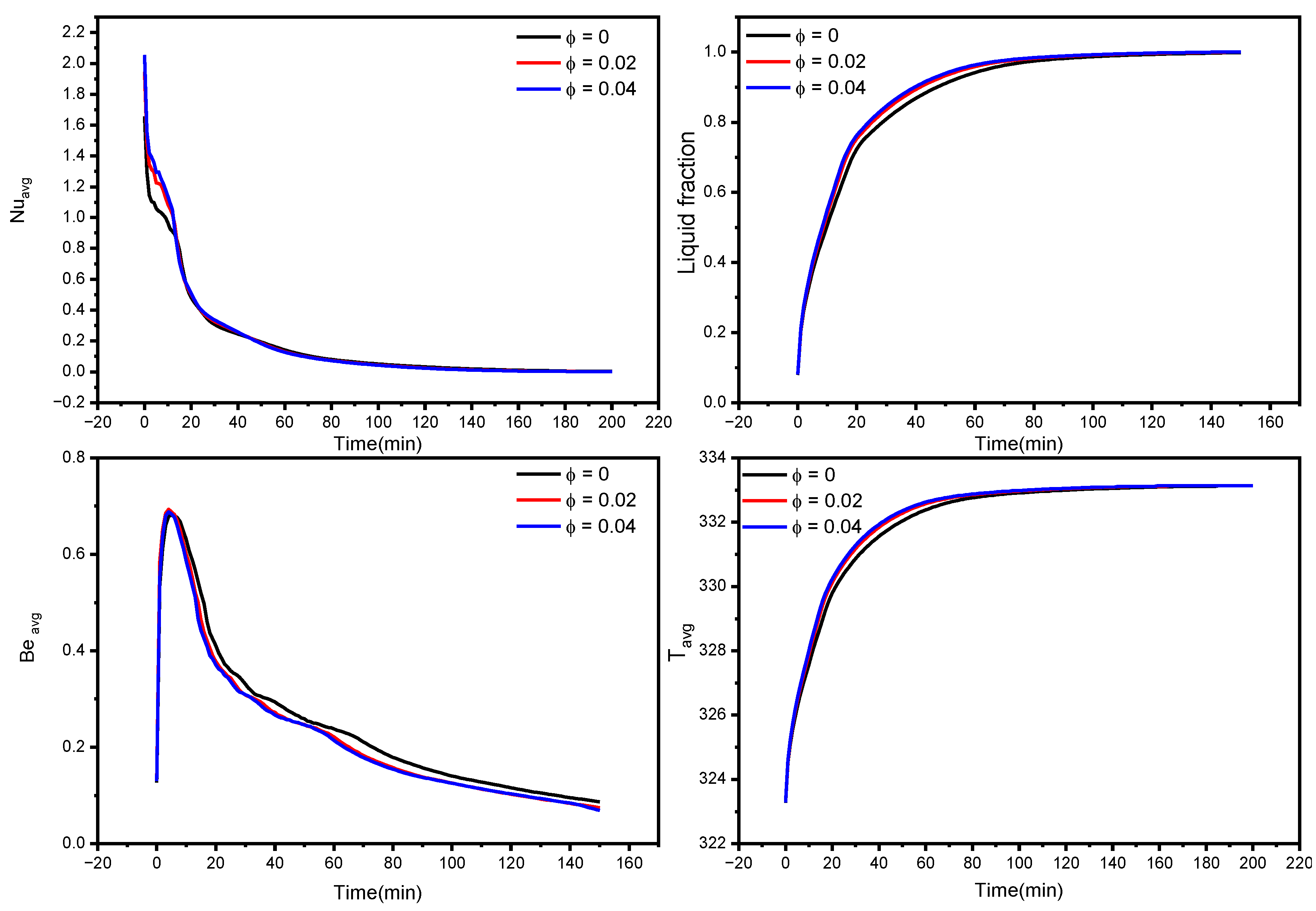
3.2. Nanoparticle’s Concentration Effect on the Acceleration of the Melting Process
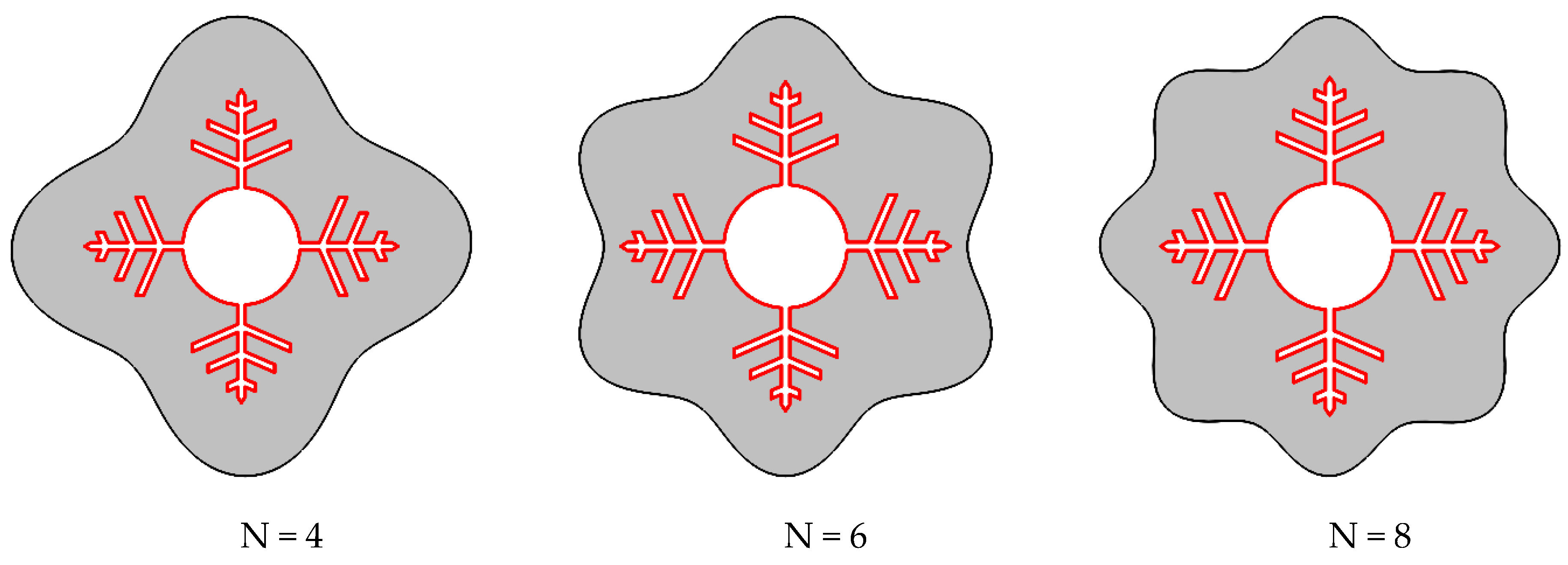
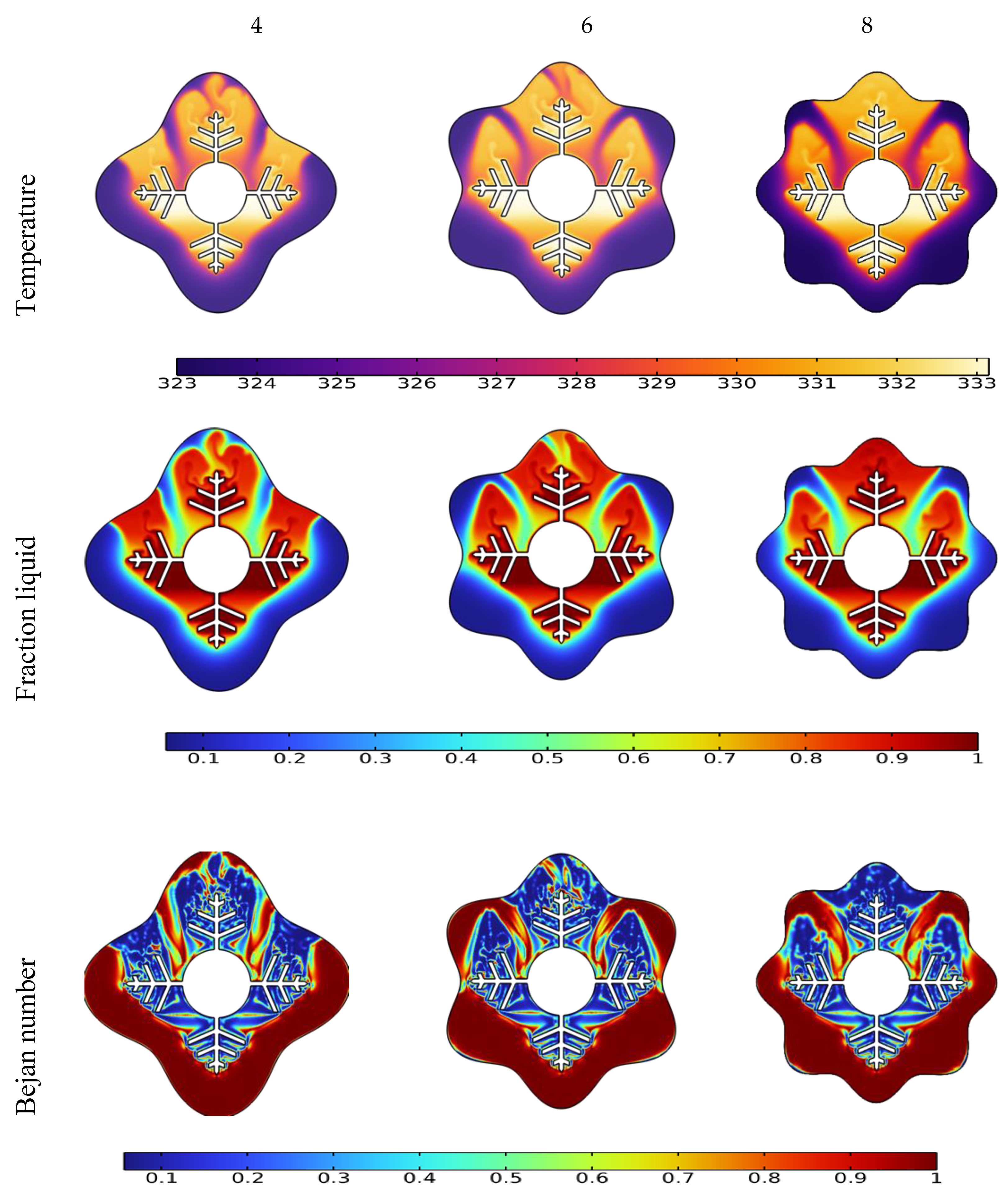
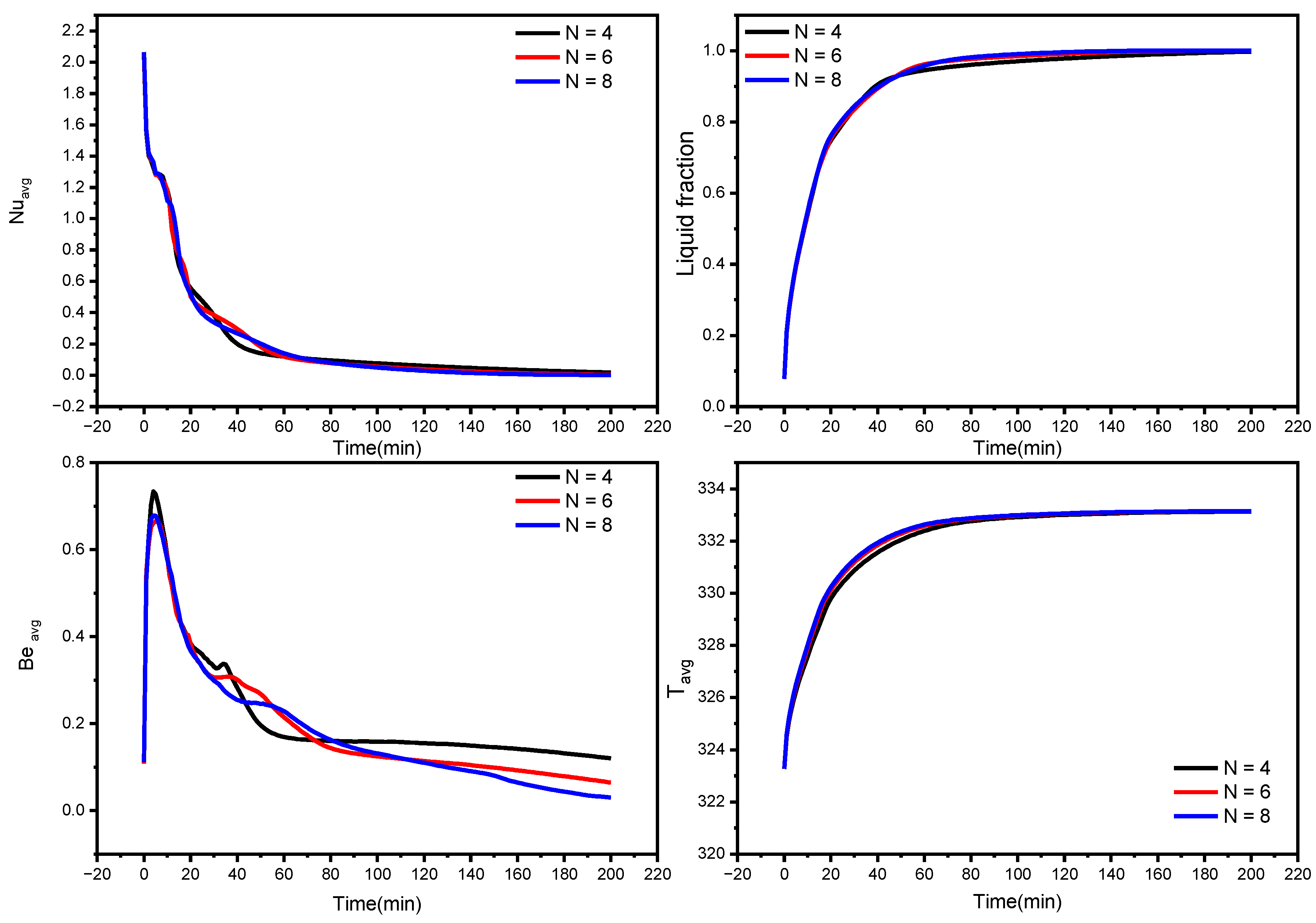
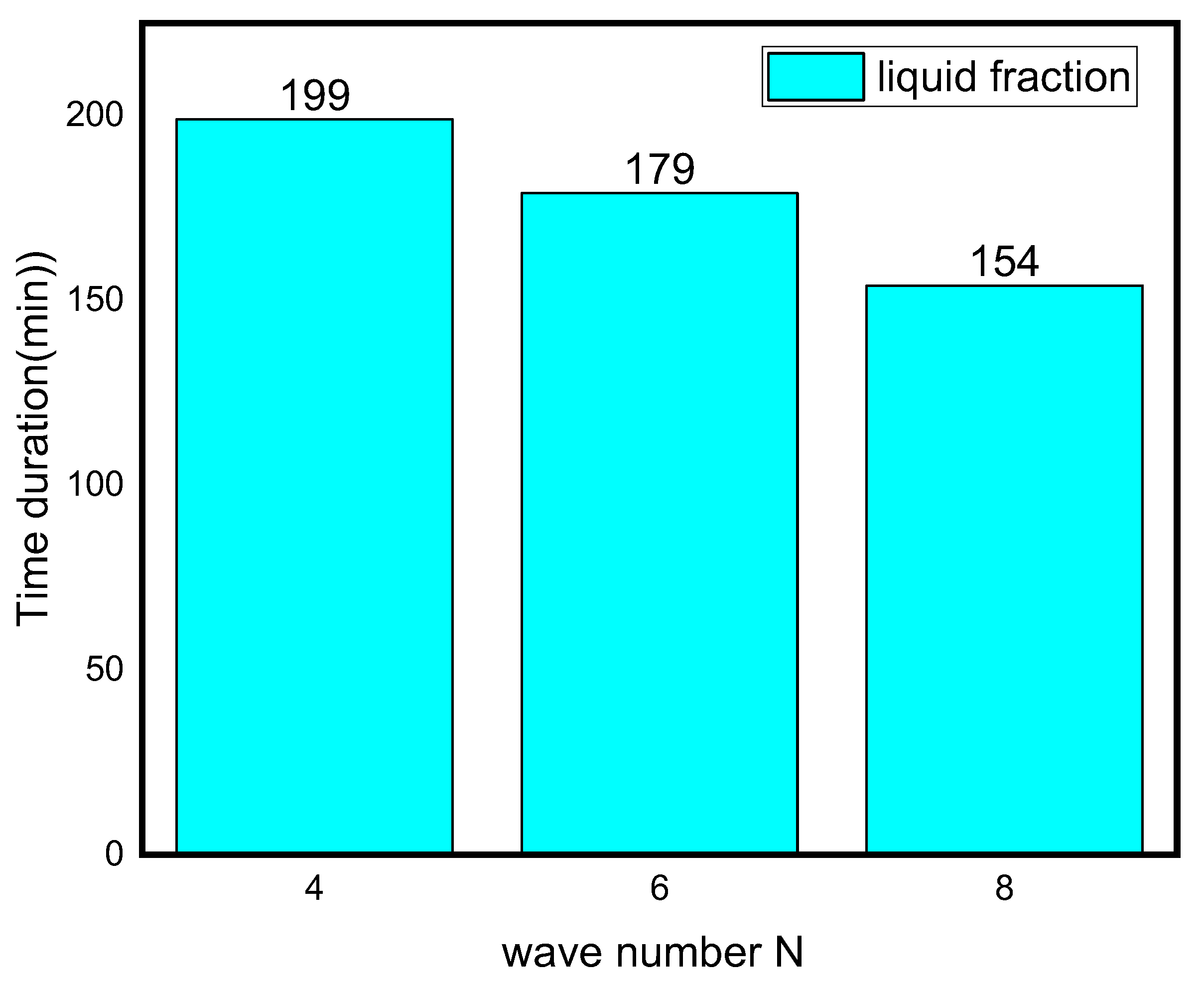
3.3. Influence of Waves Number on the Melting Process
4. Conclusions
- Owing to the presence of natural convection, the melting rate seen in the upper section of the analyzed annulus is significantly higher than that observed at its bottom portion;
- Depending on the volume fraction of nano-additive used, the melting time could be reduced from 3% to 14% with 2% and 4% nanoparticle concentrations, respectively;
- The phase transition process may be greatly accelerated by increasing the wave number N. When the wave number was increased from four to eight, the overall melting time decreased by 31%.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kober, T.; Schiffer, H.-W.; Densing, M.; Panos, E. Global energy perspectives to 2060—WEC’s World Energy Scenarios 2019. Energy Strateg. Rev. 2020, 31, 100523. [Google Scholar] [CrossRef]
- Aktar, M.A.; Alam, M.M.; Al-Amin, A.Q. Global economic crisis, energy use, CO2 emissions, and policy roadmap amid COVID-19. Sustain. Prod. Consum. 2021, 26, 770–781. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Cherian, J.; Shabbir, M.S.; Sial, M.S.; Li, J.; Mester, I.; Badulescu, A. Exploring the Relationship between Renewable Energy Sources and Economic Growth. The Case of SAARC Countries. Energies 2021, 14, 520. [Google Scholar] [CrossRef]
- Erdiwansyah; Mahidin; Husin, H.; Nasaruddin; Zaki, M.; Muhibbuddin. A critical review of the integration of renewable energy sources with various technologies. Prot. Control Mod. Power Syst. 2021, 6, 3. [Google Scholar] [CrossRef]
- Razmjoo, A.; Kaigutha, L.G.; Rad, M.A.V.; Marzband, M.; Davarpanah, A.; Denai, M. A Technical analysis investigating energy sustainability utilizing reliable renewable energy sources to reduce CO2 emissions in a high potential area. Renew. Energy 2021, 164, 46–57. [Google Scholar] [CrossRef]
- Shahzad, F.; Jamshed, W.; Sathyanarayanan, S.U.D.; Aissa, A.; Madheshwaran, P.; Mourad, A. Thermal analysis on Darcy-Forchheimer swirling Casson hybrid nanofluid flow inside parallel plates in parabolic trough solar collector: An application to solar aircraft. Int. J. 2021, 45, 20812–20834. [Google Scholar] [CrossRef]
- Kumar, A.; Tiwari, A.K.; Said, Z. A comprehensive review analysis on advances of evacuated tube solar collector using nanofluids and PCM. Sustain. Energy Technol. Assess. 2021, 47, 101417. [Google Scholar] [CrossRef]
- Jamshed, W.; Eid, M.R.; Aissa, A.; Mourad, A.; Nisar, K.S.; Shahzad, F.; Saleel, C.A.; Vijayakumar, V. Partial velocity slip effect on working magneto non-Newtonian nanofluids flow in solar collectors subject to change viscosity and thermal conductivity with temperature. PLoS ONE 2021, 16, e0259881. [Google Scholar] [CrossRef]
- Elashmawy, M.; Alhadri, M.; Ahmed, M.M.Z. Enhancing tubular solar still performance using novel PCM-tubes. Desalination 2021, 500, 114880. [Google Scholar] [CrossRef]
- Chaturvedi, R.; Islam, A.; Sharma, K. A review on the applications of PCM in thermal storage of solar energy. Mater. Today Proc. 2021, 43, 293–297. [Google Scholar] [CrossRef]
- Omara, A.A.M.; Mohammed, H.A.; al Rikabi, I.J.; Abuelnour, M.A.; Abuelnuor, A.A.A. Performance improvement of solar chimneys using phase change materials: A review. Sol. Energy 2021, 228, 68–88. [Google Scholar] [CrossRef]
- Mourad, A.; Aissa, A.; Said, Z.; Younis, O.; Iqbal, M.; Alazzam, A. Recent advances on the applications of phase change materials for solar collectors, practical limitations, and challenges: A critical review. J. Energy Storage 2022, 49, 104186. [Google Scholar] [CrossRef]
- Alothman, A.; Nandini, S.S.; Krishnaraj, L. A phase change material for building applications-A critical review. Mater. Today Proc. 2022, 56, 1858–1864. [Google Scholar] [CrossRef]
- Mousavi, S.; Rismanchi, B.; Brey, S.; Aye, L. PCM embedded radiant chilled ceiling: A state-of-the-art review. Renew. Sustain. Energy Rev. 2021, 151, 111601. [Google Scholar] [CrossRef]
- Preet, S. A review on the outlook of thermal management of photovoltaic panel using phase change material. Energy Clim. Chang. 2021, 2, 100033. [Google Scholar] [CrossRef]
- Agrawal, A.; Rakshit, D. Review on Thermal Performance Enhancement Techniques of Latent Heat Thermal Energy Storage (LHTES) System for Solar and Waste Heat Recovery Applications. In New Research Directions in Solar Energy Technologies; Springer: Berlin/Heidelberg, Germany, 2021; pp. 411–438. [Google Scholar]
- Ge, H.; Liu, J. Keeping smartphones cool with gallium phase change material. J. Heat Transf. 2013, 135, 054503. [Google Scholar] [CrossRef]
- Arshad, A.; Jabbal, M.; Sardari, P.T.; Bashir, M.A.; Faraji, H.; Yan, Y. Transient simulation of finned heat sinks embedded with PCM for electronics cooling. Therm. Sci. Eng. Prog. 2020, 18, 100520. [Google Scholar] [CrossRef]
- Fu, Z.; Liang, X.; Li, Y.; Li, L.; Zhu, Q. Performance improvement of a PVT system using a multilayer structural heat exchanger with PCMs. Renew. Energy 2021, 169, 308–317. [Google Scholar] [CrossRef]
- Peng, H.; Guo, W.; Li, M.; Feng, S. Melting behavior and heat transfer performance of gallium for spacecraft thermal energy storage application. Energy 2021, 228, 120575. [Google Scholar] [CrossRef]
- Mahani, R.B.; Mohammed, H.I.; Mahdi, J.M.; Alamshahi, F.; Ghalambaz, M.; Talebizadehsardari, P.; Yaïci, W. Phase change process in a zigzag plate latent heat storage system during melting and solidification. Molecules 2020, 25, 4643. [Google Scholar] [CrossRef]
- Memme, S.; Boccalatte, A.; Brignone, M.; Delfino, F.; Fossa, M. Simulation and design of a large thermal storage system: Real data analysis of a smart polygeneration micro grid system. Appl. Therm. Eng. 2022, 201, 117789. [Google Scholar] [CrossRef]
- Saha, S.; Ruslan, A.R.M.; Morshed, A.K.M.M.; Hasanuzzaman, M. Global prospects and challenges of latent heat thermal energy storage: A review. Clean Technol. Environ. Policy 2021, 23, 531–559. [Google Scholar] [CrossRef]
- Gupta, P.; Lohia, P.; Dwivedi, D.K. Phase change memory: Operation, current challenges and future prospects. Int. J. Eng. Sci. Technol. 2021, 13, 93–97. [Google Scholar] [CrossRef]
- Ahmed, S.E.; Abderrahmane, A.; Alotaibi, S.; Younis, O.; Almasri, R.A.; Hussam, W.K. Enhanced Heat Transfer for NePCM-Melting-Based Thermal Energy of Finned Heat Pipe. Nanomaterials 2022, 12, 129. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Opolot, M.; Liu, M.; Wang, J.; Bruno, F.; Mancin, S.; Hooman, K. Review of Analytical Studies of Melting Rate Enhancement with Fin and/or Foam Inserts. Appl. Therm. Eng. 2022, 207, 118154. [Google Scholar] [CrossRef]
- Zhang, J.; Cao, Z.; Huang, S.; Huang, X.; Liang, K.; Yang, Y.; Zhang, H.; Tian, M.; Akrami, M.; Wen, C. Improving the melting performance of phase change materials using novel fins and nanoparticles in tubular energy storage systems. Appl. Energy 2022, 322, 119416. [Google Scholar] [CrossRef]
- Kok, B. Examining effects of special heat transfer fins designed for the melting process of PCM and Nano-PCM. Appl. Therm. Eng. 2020, 170, 114989. [Google Scholar] [CrossRef]
- Sun, Z.; Fan, R.; Yan, F.; Zhou, T.; Zheng, N. Thermal management of the lithium-ion battery by the composite PCM-Fin structures. Int. J. Heat Mass Transf. 2019, 145, 118739. [Google Scholar] [CrossRef]
- Shehzad, S.A.; Alshuraiaan, B.; Kamel, M.S.; Izadi, M.; Ambreen, T. Influence of fin orientation on the natural convection of aqueous-based nano-encapsulated PCMs in a heat exchanger equipped with wing-like fins. Chem. Eng. Process.-Process Intensif. 2021, 160, 108287. [Google Scholar] [CrossRef]
- Sodhi, G.S.; Muthukumar, P. Compound charging and discharging enhancement in multi-PCM system using non-uniform fin distribution. Renew. Energy 2021, 171, 299–314. [Google Scholar] [CrossRef]
- Al-Omari, S.A.B.; Qureshi, Z.A.; Mahmoud, F.; Elnajjar, E. Thermal management characteristics of a counter-intuitive finned heat sink incorporating detached fins impregnated with a high thermal conductivity-low melting point PCM. Int. J. Therm. Sci. 2022, 175, 107396. [Google Scholar] [CrossRef]
- Sarani, I.; Payan, S.; Nada, S.A.; Payan, A. Numerical investigation of an innovative discontinuous distribution of fins for solidification rate enhancement in PCM with and without nanoparticles. Appl. Therm. Eng. 2020, 176, 115017. [Google Scholar] [CrossRef]
- Usman, H.; Ali, H.M.; Arshad, A.; Ashraf, M.J.; Khushnood, S.; Janjua, M.M.; Kazi, S.N. An experimental study of PCM based finned and un-finned heat sinks for passive cooling of electronics. Heat Mass Transf. 2018, 54, 3587–3598. [Google Scholar] [CrossRef]
- Aly, K.A.; El-Lathy, A.R.; Fouad, M.A. Enhancement of solidification rate of latent heat thermal energy storage using corrugated fins. J. Energy Storage 2019, 24, 100785. [Google Scholar] [CrossRef]
- Mosavi, A.; Mehdizadeh, H.; Abbasian-Naghneh, S.; Kalbasi, R.; Karimipour, A.; Cheraghian, G. Incorporation of horizontal fins into a PCM-based heat sink to enhance the safe operation time: Applicable in electronic device cooling. Appl. Sci. 2020, 10, 6308. [Google Scholar] [CrossRef]
- Williams, J.D.; Peterson, G.P. A Review of Thermal Property Enhancements of Low-Temperature Nano-Enhanced Phase Change Materials. Nanomaterials 2021, 11, 2578. [Google Scholar] [CrossRef] [PubMed]
- Tofani, K.; Tiari, S. Nano-Enhanced phase change materials in latent heat thermal energy storage systems: A review. Energies 2021, 14, 3821. [Google Scholar] [CrossRef]
- Bouzennada, T.; Mechighel, F.; Ghachem, K.; Kolsi, L. Numerical Simulation of the Impact of the Heat Source Position on Melting of a Nano-Enhanced Phase Change Material. Nanomaterials 2021, 11, 1425. [Google Scholar] [CrossRef]
- Kothari, R.; Sahu, S.K.; Kundalwal, S.I. Investigation on thermal characteristics of nano enhanced phase change material based finned and unfinned heat sinks for thermal management system. Chem. Eng. Process.-Process Intensif. 2021, 162, 108328. [Google Scholar] [CrossRef]
- Nižetić, S.; Jurčević, M.; Arıcı, M.; Arasu, A.V.; Xie, G. Nano-enhanced phase change materials and fluids in energy applications: A review. Renew. Sustain. Energy Rev. 2020, 129, 109931. [Google Scholar] [CrossRef]
- Babazadeh, H.; Sheremet, M.A.; Mohammed, H.A.; Hajizadeh, M.R.; Li, Z. Inclusion of nanoparticles in PCM for heat release unit. J. Mol. Liq. 2020, 313, 113544. [Google Scholar] [CrossRef]
- Arshad, A.; Jabbal, M.; Yan, Y. Preparation and characteristics evaluation of mono and hybrid nano-enhanced phase change materials (NePCMs) for thermal management of microelectronics. Energy Convers. Manag. 2020, 205, 112444. [Google Scholar] [CrossRef]
- Sivashankar, M.; Selvam, C.; Manikandan, S.; Harish, S. Performance improvement in concentrated photovoltaics using nano-enhanced phase change material with graphene nanoplatelets. Energy 2020, 208, 118408. [Google Scholar] [CrossRef]
- Faraji, H.; Faraji, M.; el Alami, M. Numerical survey of the melting driven natural convection using generation heat source: Application to the passive cooling of electronics using nano-enhanced phase change material. J. Therm. Sci. Eng. Appl. 2020, 12, 21005. [Google Scholar] [CrossRef]
- Mhiri, H.; Jemni, A.; Sammouda, H. Numerical and experimental investigations of melting process of composite material (nanoPCM/carbon foam) used for thermal energy storage. J. Energy Storage 2020, 29, 101167. [Google Scholar] [CrossRef]
- Shirazi, A.H.N.; Mohebbi, F.; Kakavand, M.R.A.; He, B.; Rabczuk, T. Paraffin nanocomposites for heat management of lithium-ion batteries: A computational investigation. J. Nanomater. 2016, 2016, 2131946. [Google Scholar] [CrossRef] [Green Version]
- Bondareva, N.S.; Buonomo, B.; Manca, O.; Sheremet, M.A. Heat transfer inside cooling system based on phase change material with alumina nanoparticles. Appl. Therm. Eng. 2018, 144, 972–981. [Google Scholar] [CrossRef]
- Ghalambaz, M.; Mehryan, S.A.M.; Veismoradi, A.; Mahdavi, M.; Zahmatkesh, I.; Kazemi, Z.; Younis, O.; Ghalambaz, M.; Chamkha, A.J. Melting process of the nano-enhanced phase change material (NePCM) in an optimized design of shell and tube thermal energy storage (TES): Taguchi optimization approach. Appl. Therm. Eng. 2021, 193, 116945. [Google Scholar] [CrossRef]
- Xuan, Y.; Li, Q. Heat transfer enhancement of nanofluids. Int. J. Heat Fluid Flow 2000, 21, 58–64. [Google Scholar] [CrossRef]
- Aman, S.; Khan, I.; Ismail, Z.; Salleh, M.Z. Impacts of gold nanoparticles on MHD mixed convection Poiseuille flow of nanofluid passing through a porous medium in the presence of thermal radiation, thermal diffusion and chemical reaction. Neural Comput. Appl. 2018, 30, 789–797. [Google Scholar] [CrossRef] [Green Version]
- Thirugnanam, C.; Marimuthu, P. Experimental analysis of latent heat thermal energy storage using paraffin wax as phase change material. Int. J. Eng. Innov. Technol. 2013, 3, 372–376. [Google Scholar]
- Saeed, A.M.; Abderrahmane, A.; Qasem, N.A.A.; Mourad, A.; Alhazmi, M.; Ahmed, S.E.; Guedri, K. A numerical investigation of a heat transfer augmentation finned pear-shaped thermal energy storage system with nano-enhanced phase change materials. J. Energy Storage 2022, 53, 105172. [Google Scholar] [CrossRef]
- Maneengam, A.; Ahmed, S.E.; Saeed, A.M.; Abderrahmane, A.; Younis, O.; Guedri, K.; Alhazmi, M.; Weera, W. Numerical Study of Heat Transfer Enhancement within Confined Shell and Tube Latent Heat Thermal Storage Microsystem Using Hexagonal PCMs. Micromachines 2022, 13, 1062. [Google Scholar] [CrossRef]
- Sharma, A.; Sharma, S.D.; Buddhi, D. Accelerated thermal cycle test of acetamide, stearic acid and paraffin wax for solar thermal latent heat storage applications. Energy Convers. Manag. 2002, 14, 1923–1930. [Google Scholar] [CrossRef]
- Al-Abidi, A.A.; Mat, S.; Sopian, K.; Sulaiman, M.Y.; Mohammad, A.T. Internal and external fin heat transfer enhancement technique for latent heat thermal energy storage in triplex tube heat exchangers. Appl. Therm. Eng. 2013, 53, 147–156. [Google Scholar] [CrossRef]
- Bendrer, B.A.; Abderrahmane, A.; Ahmed, S.E.; Raizah, Z.A.S. 3D magnetic buoyancy-driven flow of hybrid nanofluids confined wavy cubic enclosures including multi-layers and heated obstacle. Int. Commun. Heat Mass Transf. 2021, 126, 105431. [Google Scholar] [CrossRef]
- Al-Kouz, W.; Aissa, A.; Koulali, A.; Jamshed, W.; Moria, H.; Nisar, K.S.; Mourad, A.; Abdel-Aty, A.-H.; Khashan, M.M.; Yahia, I.S. MHD darcy-forchheimer nanofluid flow and entropy optimization in an odd-shaped enclosure filled with a (MWCNT-Fe3O4/water) using galerkin finite element analysis. Sci. Rep. 2021, 11, 22635. [Google Scholar] [CrossRef]
- Arasu, A.V.; Mujumdar, A.S. Numerical study on melting of paraffin wax with Al2O3 in a square enclosure. Int. Commun. Heat Mass Transf. 2012, 39, 8–16. [Google Scholar] [CrossRef]
- Ahmed, S.; Abderrahmane, A.; Saeed, A.M.; Guedri, K.; Mourad, A.; Younes, O.; Botmart, T.; Shah, N.A. Melting enhancement of PCM in a finned tube latent heat thermal energy storage. Sci. Rep. 2022, 12, 11521. [Google Scholar] [CrossRef]
| Properties of the Materials | PCM | Inner-Tube Wall | Nanoparticle | |
|---|---|---|---|---|
| (paraffin wax) | Copper | Copper | ||
| Solid | Liquid | |||
| Thermal conductivity, k, (W/mK) | 0.39 | 0.157 | 401 | 401 |
| Density, ρ, (kg/m3) | 775 | 833.6 | 8900 | 8954 |
| Kinematic viscosity (m2/s) | 8.31 × 10−5 | |||
| Thermal expansion coefficient, β, (1/K) | - | 7.14 × 10−3 | 1.67 × 10−5 | |
| Specific heat, cp, (kJ/kgK) | 2.44 | 2.384 | 0.385 | 0.385 |
| The melting point (°C) | 54.32 | |||
| Latent heat of fusion (kJ/kg) | 184.48 | |||
| The reference temperature (°C) | 50 | |||
| Mesh | G1 | G2 | G3 |
|---|---|---|---|
| Number of elements | 25,096 | 66,982 | 100,384 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jirawattanapanit, A.; Abderrahmane, A.; Mourad, A.; Guedri, K.; Younis, O.; Bouallegue, B.; Subkrajang, K.; Rajchakit, G.; Shah, N.A. A Numerical Investigation of a Melting Rate Enhancement inside a Thermal Energy Storage System of Finned Heat Pipe with Nano-Enhanced Phase Change Material. Nanomaterials 2022, 12, 2519. https://doi.org/10.3390/nano12152519
Jirawattanapanit A, Abderrahmane A, Mourad A, Guedri K, Younis O, Bouallegue B, Subkrajang K, Rajchakit G, Shah NA. A Numerical Investigation of a Melting Rate Enhancement inside a Thermal Energy Storage System of Finned Heat Pipe with Nano-Enhanced Phase Change Material. Nanomaterials. 2022; 12(15):2519. https://doi.org/10.3390/nano12152519
Chicago/Turabian StyleJirawattanapanit, Anuwat, Aissa Abderrahmane, Abe Mourad, Kamel Guedri, Obai Younis, Belgacem Bouallegue, Khanyaluck Subkrajang, Grienggrai Rajchakit, and Nehad Ali Shah. 2022. "A Numerical Investigation of a Melting Rate Enhancement inside a Thermal Energy Storage System of Finned Heat Pipe with Nano-Enhanced Phase Change Material" Nanomaterials 12, no. 15: 2519. https://doi.org/10.3390/nano12152519
APA StyleJirawattanapanit, A., Abderrahmane, A., Mourad, A., Guedri, K., Younis, O., Bouallegue, B., Subkrajang, K., Rajchakit, G., & Shah, N. A. (2022). A Numerical Investigation of a Melting Rate Enhancement inside a Thermal Energy Storage System of Finned Heat Pipe with Nano-Enhanced Phase Change Material. Nanomaterials, 12(15), 2519. https://doi.org/10.3390/nano12152519








